Cerebrospinal Fluid Shunt Emergencies
Authors
Whitney M. Wroe, MD, Resident, Department of Pediatrics, University of Texas Health Sciences Center, San Antonio
Daniel J. Dire, MD, FACEP, FAAP, FAAEM, Clinical Professor, Departments of Emergency Medicine and Pediatrics, University of Texas Health Sciences Center, San Antonio
Peer Reviewer
Catherine A. Marco, MD, FACEP, Professor, Department of Emergency Medicine, Wright State University Boonshoft School of Medicine, Attending Physician, Miami Valley Hospital, Dayton, Ohio
Dr. Dire (author), Dr. Wroe (author), and Dr. Marco (peer reviewer) report no relationships with companies related to the field of study covered by this CME activity.
Executive Summary
- A high index of suspicion is needed to diagnose shunt malfunction; patients may present with non-specific signs and symptoms.
- Shunt failure is most common the first couple months after shunt placement; previous shunt revision and infection are known predictors of shunt failure.
- To assess for shunt function current image studies must be compared with prior exams.
- Quick-brain MRI is preferred over CT.
- Delayed recognition can lead to significant morbidity and mortality.
Cerebrospinal fluid (CSF) shunt failures account for more than 15,000 pediatric hospital admissions per year,1 and have an estimated mortality rate of 1-2%.2 The prompt recognition and treatment of shunt failure in the emergency department (ED) is of the utmost importance to limit morbidity and mortality.
— Ann M. Dietrich, MD, Editor
CSF shunts are mechanical conduits that drain CSF from the ventricular system to a distal site of absorption. They are used as a way to decrease elevated intracranial pressure (ICP) as a result of poor absorption and/or overproduction of CSF within the ventricular system.3,4
Hydrocephalus is one of the most common present-day reasons for pediatric shunt placement.3,5,6 Hydrocephalus refers to enlargement of the ventricles and can be a result of infection, hemorrhage, neoplasm, spinal dysraphism, trauma, or a congenital etiology.4,6 An estimated 125,000 U.S. children have shunt-dependent hydrocephalus,1 with an estimated prevalence of 1-1.5%.4,5
Shunt failure is common; 40% of shunts fail within the first year2,3,7-10 and 56-80% by 10 years.2,11,12 Between 8-10% of shunts eventually become infected.3,7,8,13 Shunt malfunction can lead to an acute increase in ICP, which may be a life-threatening emergency, making early detection and treatment critical to reducing morbidity and mortality.
CSF Shunts
Cerebrospinal fluid shunts drain CSF from the ventricular system to a site of absorption. Shunts are made up of four components: the proximal catheter, the reservoir, the valve, and the distal catheter. The proximal catheter originates in the lateral ventricle. The catheter exits the intra-cranium through a burr hole and connects to a reservoir in the overlying subcutaneous tissue. The reservoir allows access for CSF sampling and pressure monitoring. From the reservoir, a one-way valve controls flow into the distal catheter. There are five broad categories of valves: differential pressure, flow regulated, anti-siphon controlled, programmable, and gravitational. Each category represents the continued effort to improve the effects of over-drainage associated with hydrostatic pressure changes and reduce the need for surgical intervention when altering valve settings (see Table 1). A distal catheter is attached to this valve, which is then tunneled subcutaneously into another body cavity where CSF can easily be reabsorbed.11,14,15 The peritoneum is the most common site for distal catheter termination — when in such a location, the shunt is called a ventriculoperitoneal (VP) shunt. When the peritoneum is a non-viable option, ventriculoatrial, lumbarperitoneal, ventriculopleural, and ventriculogallbladder are alternative possibilities.2,8,16 Numerous types and brands of CSF shunts exist, each with their own advantages and disadvantages.14,17 Knowing the type of shunt a patient has is critical to patient management.
Table 1. Shunt Valve Classifications7,14,17,41-43 |
||
|
Type of Valve Can have a combination of the valves below |
Description |
Brand Examples |
|
Flow Regulated |
Maintains constant CSF flow through the valve. |
- Orbis-Sigma |
|
Anti-Siphon |
Reduces the siphoning, or over-drainage effect, that occurs in the upright position due to hydrostatic pressure differences. |
- Delta (Medtronic PS Medical) |
|
Programmable |
Allows valve pressure settings to be altered as an outpatient without the need for surgical intervention. |
- Strata (Medtronic PS Medical) - Codman-Hakim Programmable Shunt (Codman) - Certas Valve (Codman) |
|
Gravitational |
Posture dependent valves that change resistance based on a patient’s posture in efforts to overcome siphoning. |
- ProGAV (Aesculap) - Paedi-Gav (Aesculap) |
|
For more information on types of shunts, please visit the ISPN (international Society for Pediatric Neurosurgery) webpage at http://www.ispneurosurgery.org/ and view the shunt guide section. |
||
|
Differential Pressure |
Allows flow through the valve when the pressure gradient between the proximal and distal catheter reaches a set point. Valves are posture independent. |
- Medtronic Fixed Differential Pressure - Aesculap Fixed Differential Pressure - Codman Fixed Differential Pressure |
Signs and Symptoms of Shunt Failure
The signs and symptoms of shunt failure are vast and non-specific. Often, they mimic common childhood illnesses such as gastroenteritis, otitis media, appendicitis, migraines, and viral illnesses.3,4,18 Having a high index of suspicion for shunt failure is critical to diagnosis.
Clinical presentation varies according to age, duration of malfunction, and etiology of failure (infectious vs non-infectious).7,14 (See Table 2.) The most predictive factors of failure are bulging fontanel, fluid collection along the shunt, depressed level of consciousness, irritability, abdominal pain, nausea and vomiting, abnormal shunt pump test, accelerated head growth, and headache. Predictors more specific for an infectious etiology include: purulent drainage, skin erosion, meningismus, erythema, peritonitis, abdominal pain, CSF leakage, and irritability.3 Notably, although fever is strongly associated with shunt infection, it is not a requirement to make the diagnosis.3,14 Time since initial surgery and previous history of infection are other important factors to consider.19 To date, it is unclear whether seizures have any predictability in regards to shunt failure.2,18
Table 2. Signs and Symptoms of Shunt Failure |
|
Infectious and Non-infectious
|
When there is concern for repeat failure, knowing how a patient previously presented with shunt failure can be helpful.14 Younger age at time of insertion and short time interval since prior surgical revision are important predictors of repeated shunt failure.8
It is important to note that while the presence of certain signs and symptoms increases the likelihood of shunt failure, the same cannot be said for their absence. Thus, an astute physician must remain wary, as the lack of any signs and symptoms does little to rule out the possibility of shunt failure.3,18
Mechanisms of Shunt Failure — Non-Infectious Causes of Shunt Failure
Mechanical Failure
Fracture: Catheter fracture occurs due to the biomechanical forces of patient growth and catheter degradation from host reactions.8 Scar tissue surrounding the catheter provides a temporary conduit between catheter fragments, often causing a delay in symptomatology and detection.11 The neck is the most common region for fracture development.14,20
Disconnection: Catheter disconnection occurs due to surgical error or material defect. Disconnection is seen shortly after shunt surgery and typically located at the valve level.8,11,20
Migration: Migration occurs when the catheter (proximal or distal) moves from its original position to a location that inhibits proper drainage. This can occur due to improper length (both too short and too long), fracture, disconnection, tethering, or perforation.8,11,21 Evidence of catheter migration and subsequent erosion/perforation into viscera can be found in almost any body cavity, including the ventricular, intrathoracic, abdominal, and pelvic cavities.11
Misplacement: Misplacement occurs when the catheter tip (either proximal or distal) is inappropriately positioned during surgery, making for poor CSF flow. Misplaced shunts are typically noted shortly after surgery, but may also have a delayed presentation.8
Obstruction
Obstruction is the most common cause of shunt malfunction, making up 56-83% of shunt failures.2,9,15,21 The two most common places for obstruction to occur are at the proximal catheter tip and at the shunt valve. The proximal catheter tip can be obstructed due to ingrowth of the choroid plexus; the shunt valve can be blocked by blood and debris.8,11,14 Distal catheter obstruction is less common and occurs due to adhesions, scarring, migration, tube kinking (most commonly at the level of the valve connection or abdominal entry site), catheter twisting, or distal catheter obstruction (internal obstruction from inflammatory debris and tissue build-up or external obstruction from pseuduocyst formation).8,14,21
Over-drainage
Over-drainage occurs when a functioning shunt drains more CSF than appropriate. Acute over-drainage results in extra-axial fluid collections or subdural hematomas. Chronic over-drainage results in slit ventricle syndrome (SVS), the precise definition of which remains to be determined. Generally speaking, SVS refers to symptomatic small ventricles — small ventricles in association with intermittent symptoms suggestive of shunt malfunction (increased intracranial hypertension), typically associated with postural changes. Although small ventricles occur in 50% of shunted children, SVS only has a reported incidence of < 2%. Nonetheless, SVS accounts for a disproportionate number of shunt revisions in pediatric neurosurgery. The etiology of SVS is theorized to be due to chronic over-drainage during the period of rapid brain growth, leading to poor brain parenchymal compliance and obstruction of the catheter by the collapsed ventricular system.11,14,22
Ventricular Loculations
Ventricular loculations are non-communicating pockets of CSF within the ventricular system. They form gradually and are either congenitally acquired or develop after an episode of ventriculitis or hemorrhage. Shunt malfunction occurs due to a shunt’s inability to access and drain all the CSF so that a loculated compartment, separated from the shunt, continues to increase in size and creates symptoms of increased intracranial pressure.11,22 A special type of loculation, known as isolated or “trapped” fourth ventricle, occurs when the fourth ventricle is unable to communicate with both the third ventricle above and basal cisterns below. In the face of a functioning shunt, CSF is diverted away from the Sylvian aqueduct promoting its closure. If there is poor CSF absorption or fourth ventricular obstruction, CSF will accumulate in the fourth ventricle producing signs and symptoms of increased ICP. This phenomenon typically occurs in patients with a history of shunt revisions and post-infectious hydrocephalus.4,22
Abdominal Complications
Ascites: A rare complication that causes shunt malfunction in patients with comorbid heart, liver, or kidney disease. The ascites fluid increases pressure within the peritoneal cavity, which changes the CSF intracranial-peritoneal pressure differential and eventually leads to shunt malfunction.22
Constipation: Severe constipation can increase intra-abdominal pressure and thereby alter the CSF intracranial-peritoneal pressure differential, eventually leading to shunt malfunction.5
Inguinal Hernia: Increased intra-abdominal pressure following VP shunt placement increases the likelihood of subsequent inguinal hernia development, especially in neonates and males.21,23
Perforation: May occur acutely (intra-operatively) or chronically via erosion of the shunt through an abdominal or pelvic viscus.11,22 Chronic perforations typically present indolently with tubing emerging from the urethra or anus.22 Perforations into the bladder, vagina, diaphragm, intestine, gallbladder, and bronchial tree have all been documented.21
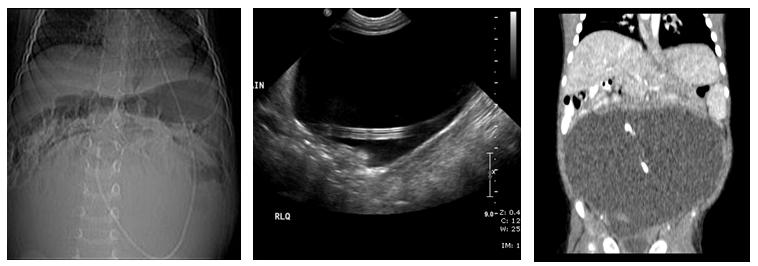
Pseudocyst: Pseudocyts are loculated intra-abdominal fluid collections centered around the terminal portion of the peritoneal catheter (see Figure 1).11,22 They are caused by adhesions due to infection, prior abdominal surgery, reactions to CSF proteins and shunt material, or migration of the omentum over the catheter tip.11,21 Pseudocysts present as abdominal masses (with or without pain), signs of abdominal obstruction, and/or neurological symptoms,22 and occurrence is 0.7-10%.21
Figure 1. Example of a CSFoma (Pseudocyst) with KUB (left), Abodominal Ultrasound (middle), and Coronal CT Image (right) of the Same Child |
 |
|
Images provided by Achint K. Singh, MD, University of Texas Health Sciences Center San Antonio |
Mechanisms of Shunt Failure — Infectious Causes of Shunt Failure
Approximately 8-10% of shunts become infected.8 Depending on the microbial source, shunt infection and subsequent failure can present both acutely or indolently. A high index of suspicion is needed in all patients presenting to the ED with nonspecific signs and symptoms, with or without fever, as the consequences of a missed/delayed diagnosis are severe.13,14,21,24 Most shunt infections occur within the first 4-6 months after placement and are caused by skin-colonizing organisms present at the time of surgery.13,20,25 Staphylococcus species account for up to 90% of infections,25 with coagulase negative staphylococci (CONS) alone accounting for 75%, due to its unique ability to create a biofilm (biofilms allow CONS to escape both antimicrobials and the host immune system).13 When present, Gram-negative organisms represent ICU colonization, neonatal pathogens, or abdominal pathology. Should cultures contain Enterobacteriaceae or anaerobes or be polymicrobial, bowel perforation by the peritoneal catheter should be a concern.13,21 Other infectious organisms that can be seen include Proprionibacterium species, Corynebacterium, Haemophilius influenza, and fungi.25
Known risk factors for infection include time since placement, age at insertion, history of prior infection, and history of revision (see Table 3).
Table 3. Known Risk Factors for Shunt Infection |
|
Mechanisms of Shunt Failure — Special Shunts
When placement of the distal shunt catheter into the peritoneum is contraindicated, alternative locations are used. Notably, these locations are less common and associated with a higher rate of failure.11
Ventriculoatrial Shunts
Due to their risk profile, ventriculatrial (VA) shunts are not as common as they once were. The atrium is accessed via the facial, subclavian, or internal jugular vein, making length critical to proper VA shunt functioning. Catheters that are too long allow for catheter migration into the right atrium, through a patent foramen ovale, into the pericardial space, or through the septum; if too short, migration with growth up the SVC may produce poor drainage.11 Other complications include bacterial endocartidits, pulmonary embolism, pulmonary hypertension, cor pulmonale, cardiac arrhythmias, cardiac tamponade, venous thrombosis, tricuspid valve pathology, infection, and shunt nephritis.2,8,11 Shunt nephritis is the result of activation of the complement cascade due to chronic infection and subsequent glomerular immune complex deposition. Associated symptoms include hypertension, nephrotic syndrome, hematuria, fever, anemia, hepatosplenomegaly, and non-thrombocytopenic purpura. Unlike VP shunts, due to the bloodstream location of VA catheters, when shunt infection is present, blood cultures are often positive. Likewise, if bacteremia from a non-catheter-related etiology occurs, colonization of the shunt subsequently ensues. Most VA complications are late occurrences.8,11
Ventriculopleural Shunts
Ventriculopleural shunts are used as a temporary measure, as the risks include hydrothorax, pneumothorax, fibrothorax, pleural emphyema, pleural effusion, chest pain, pneumonia, respiratory failure, and bronchial perforation.2,11,14
Lumboperitoneal Shunts
Lumboperitoneal shunts are associated with tonsillar herniation and arachnoiditis.26
Diagnostic Testing
Currently, there is no consensus guideline to aid physicians in the diagnostic workup of shunt failure. All imaging modalities have their unique advantages and disadvantages. Clinical judgment must be used in determining what type(s) of imaging modality to use.27
Shunt Pump Test
Pumping the shunt is a compression maneuver clinicians can perform to test for both proximal and distal shunt obstruction. How to perform the test depends on whether the shunt has a single or double reservoir (see Table 4). In general, resistance to reservoir compression is concerning for distal obstruction, whereas delayed refill with decompression is concerning for proximal obstruction.
Table 4. Pumping the Shunt |
|
Single Reservoir
Adapted from Greenberg4 and Magita10 |
Although previously thought to be helpful in the diagnosis of shunt malfunction, its utility is not as valuable as once believed.14 The reported sensitivity and specificity for detecting obstruction is 11-20% and 63-99%, respectively,10 with the specificity declining with increasing age.3 The positive predictive value ranges from 12% to 86% and the negative predictive value from 65% to 93%.8,10 Thus, a negative shunt pump test cannot rule out shunt obstruction,3,18 nor does a positive test in an asymptomatic individual necessitate a workup.28 Newer studies have even suggested that the test may actually cause obstruction by drawing the choroid plexus into the shunt.14 Thus, if the test is to be performed, it may only be used as an adjunct to clinical decision making, not to definitively rule in or out shunt obstruction.10
Shunt Series
Shunt series refers to the collection of radiographs that view the entire course of a shunt. It includes frontal and lateral views of the head and neck and frontal views of the chest and abdomen. It is used to evaluate shunt continuity (disconnections, fractures, catheter migrations) rather than shunt patency.11,14,21 Some valves and connectors are radiolucent while others are radiopaque. Normally, radiolucent material can be mistaken for shunt disconnection/fracture. Knowing what type of shunt the patient has can help distinguish this.11,14 Comparison to prior imaging is helpful.
Controversy surrounds the use of the shunt series in cases of possible dysfunction. Some studies suggest obtaining it in all cases of suspected shunt malfunction to identify the presence of shunt discontinuity; MRI or CT will only show ventricular size and morphology changes.29 Other studies suggest its use only when there is concern for a mechanical cause of shunt failure due to the low sensitivity of the test.20,27,30 Clinical judgment is advised as well as developing a protocol with your local neurosurgeon to determine in which instances a shunt series should be performed.
Quick-Brain MRI/CT
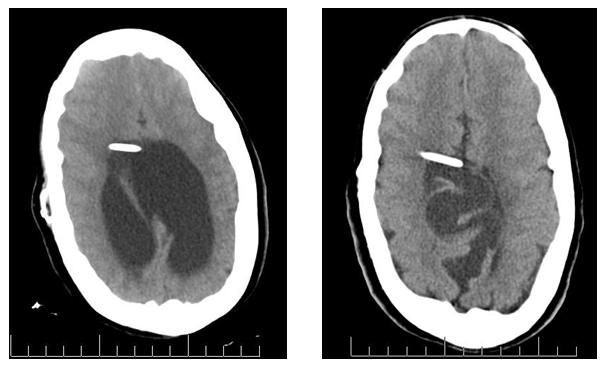
Quick-brain MRI and CT are imaging modalities used to provide information on ventricular morphology, shunt location, and shunt integrity to aid in detection of shunt failure. Shunt failure can present with enlarged, normal, or small-sized ventricles.14,21,31 Although ventricular enlargement is indicative of shunt failure,8,14,31 shunt malfunction occurs in 16-24% of patients with no ventricular changes on imaging.2 Comparison with prior imaging, if available, is imperative (see Figure 2).8,14,31 Comparison of ventricular morphology both from baseline imaging and/or prior imaging from past obstruction can guide a clinician’s judgment; changes are often time consistent and predictable, but not always.31 Secondary signs of obstruction include blurring of ventricular margins due to transependymal flow of CSF, peri-shunt edema, and subgaleal fluid collections.11,14 Infection can occasionally be detected by visualization of debris within the ventricles and/or leptomeningeal/ependymal enhancement. Care must be taken when interpreting pachymeningeal enhancement as this can be seen for months postoperatively.11,24 As an imaging modality, CT has good specificity but poor sensitivity (54-83%)1 and negative predictive value for predicting shunt obstruction;27 therefore, clinical findings in correlation with imaging are needed to make decisions regarding shunt management.
Figure 2. 16-year-old Female with VP Shunt Malfunction who Presented with Headache and Vomiting |
|
CT image on left was done in the ED showing enlargement of both lateral ventricles as compared to her previous CT performed when asymptomatic (right). |
|
Images provided by Daniel J. Dire, MD, University of Texas Health Sciences Center San Antonio |
Quick-brain MRI (also known as rapid-brain MRI, rapid-sequence MRI, fast-sequence MRI, single-shot fast-spin echo) is quickly replacing CT as the diagnostic imaging modality of choice in the workup of shunt malfunction.1,11,14,32-34 Quick-brain MRI works by taking 1-minute images of the brain in three separate planes, thus reducing the need for sedation and eliminating radiation exposure without altering the test characteristics for detecting shunt failure.1 Decreased radiation exposure is a significant benefit for patients with CSF shunts who receive an average of 2.6 head scans per year.12,32,34 Quick-brain is as sensitive as CT in detecting shunt malfunction with comparable sedation needs1,14,33 and minimal increase in time to complete the scan.1
Quick brain MRI is not without its limitations. Contraindications include pacemakers, defibrillators, and cochlear implants.1 Blood, air, and implanted devices can be difficult to visualize with quick-brain MRI and make CT a reasonable alternative when specifically trying to visualize a hemorrhage, pneumocephalus, or when contrast material is needed.32,33 Care must be taken with programmable shunt valves, as these may be adjusted by the MRI magnet. Prior to MRI imaging, the radiologist should confirm that the valve is resistant to reprograming at the magnetic field to be used. Post scan, it is good practice to check the valve to ensure that the settings have not changed. More recent valves have been made that are resistant to change at the 3T level.11
Shuntogram
Shuntogram is a radionucleotide shunt study used to evaluate shunt patency and velocity. Shunt obstruction is seen when the radionucleotide injected into the shunt reservoir fails to flow throughout the length of the catheter.11,14 This test is especially useful when there is concern for obstruction but no change in ventricular size.14 Notably, CT combined with shuntogram increases the sensitivity compared to CT alone.27
Ultrasound
Ultrasound is a new and emerging imaging modality being used as an adjunct in the evaluation of shunt malfunction. It is cheap, fast, easily accessible, and has the added benefit of no radiation exposure.35 Reports have been made about its use in detection of abdominal shunt-related pathology (pseudocyst, abscess),14 shunt discontinuity,36,37 and aiding the physician in accessing the shunt reservoir.36,37 Some studies have looked at shunt flow patterns and cerebral blood flow with Doppler ultrasound in attempts to extrapolate information regarding shunt function.36-39 Other studies have looked at using ultrasound to measure optic nerve sheath diameter as a predictor of increased ICP; however, these studies revealed that this test is neither sensitive nor specific enough to be used as a screening modality in the evaluation of shunt malfunction.35 Work continues to be done in the field of ultrasonography in hopes to further its use as an imaging modality in the workup of shunt malfunction.
Shunt Management
When there is concern for CSF shunt malfunction, always consult a neurosurgeon to assist in shunt evaluation. A neurosurgeon can tap the shunt for both therapeutic and diagnostic purposes, remove/repair the shunt, place a temporary extra-ventricular draining device, or perform a third ventriculostomy.4,40
Non-infectious
Any child that presents with a potential shunt dysfunction should be placed on a cardiac monitor and have frequent neuro checks performed. If the shunt is found to be dysfunctional, the child should be managed, preferably, at the site where the shunt was placed. An emergent transfer should be arranged in consultation with the accepting neurosurgeon. If a patient comes into the ED with concern for cerebral herniation and impending death from increased ICP (fixed and dilated pupils, presence of Cushing’s triad, unresponsiveness) and transfer to a higher level of care or a neurosurgeon is not immediately available, the ED physician may need to tap the shunt to emergently decrease the ICP.
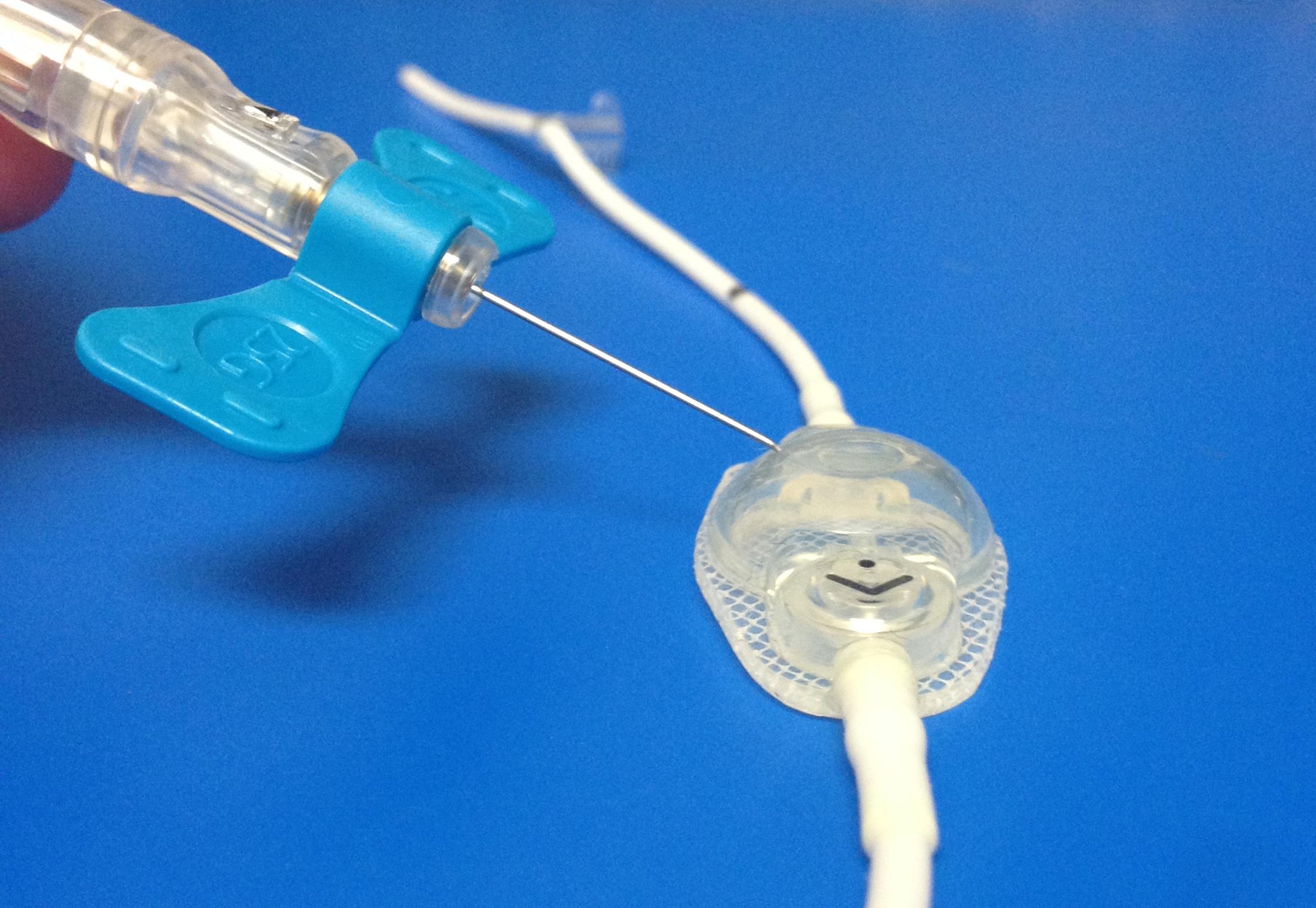
Prior to performing a procedure, the physician must know what kind of shunt the patient has and where to tap for each individual shunt type. The physician needs to be aware that there are significant risks associated with accessing the shunt, including infection, CSF leak, damage to the shunt, and local hematoma.40 The steps to perform a procedure are listed in Table 5 (see Figure 3).
Table 5. Steps to Performing a CSF Shunt Procedure4,10,15,40 |
|
Figure 3. Example Tapping a Shunt Reservoir with a 25 Gauge Butterfly Needle |
|
To obtain pressures, attach a manometer (not shown) to the distal end of the butterfly needle catheter. Additional photographs of a shunt tap can found at http://emedicine.medscape.com/article/81058-overview#a7. |
|
Image provided by Whitney M. Wroe, MD, University of Texas Health Sciences Center San Antonio |
Infectious
There are no universal guidelines for management of CSF shunt infections.13 Only a neurosurgeon should tap the shunt reservoir and obtain CSF cultures.11,13 This may be delayed until patient is transferred to a higher level of care or in conjunction with a neurosurgeon.
To increase detection of shunt pathogens, aerobic, anaerobic, and fungal cultures should be obtained and extended incubation times should be requested. Prolonging incubation time will lead to higher detection rates of fastidious organisms and microorganisms in patients pre-treated with antibiotics. New and emerging is the use of polymerase chain reaction as a diagnostic tool in detecting shunt pathogens. Current studies show that many negative cultures have tested positive with polymerase chain reaction.13
Empiric coverage should be directed toward common shunt pathogens and have the ability to penetrate the central nervous system.13 For Gram-positive coverage, vancomycin is typically used due to the emergence of methicillin-resistant Staphylococcus. Cefepime, ceftazidime, or meropenem are appropriate for Gram-negative coverage as they include coverage of Pseudomonas aeruginosa.13 When infection is present, a neurosurgeon will typically do one of the following:
- Remove the shunt, place a temporary external ventricular drain, and give IV antibiotics.13,24
- Replace the shunt and give IV antibiotics.13,24
- Serial ventricular taps and give IV antibiotics without surgical revision.13 Notably, there are lower success rates when hardware is kept in place.24
The duration of antibiotics varies, and the use of intraventricular antibiotics is controversial.13
Summary
Shunt failure is common. Symptoms are often non-specific and require a high index of suspicion for diagnosis. There is no guideline or consensus on diagnostic imaging, as each imaging modality has its advantages and disadvantages. Diagnosis of shunt malfunction ultimately is a clinical one, relying on the combination of symptoms, physical exam, and imaging results. Missed diagnosis can result in significant morbidity and mortality.
REFERENCES
- Yue EL, Meckler GD, Fleischman RJ, et al. Test characteristics of quick brain MRI for shunt evaluation in children: An alternative modality to avoid radiation. J Neurosurg Pediatr 2015;15:420-426.
- Kim TY, Stewart G, Voth M, et al. Signs and symptoms of cerebrospinal fluid shunt malfunction in the pediatric emergency department. Pediatr Emerg Care 2006;22:28-34.
- Piatt JH Jr, Garton HJ. Clinical diagnosis of ventriculoperitoneal shunt failure among children with hydrocephalus. Pediatr Emerg Care 2008;24:201-210.
- Greenberg MS. Hydrocephalus. In: Handbook of Neurosurgery. 7th ed. Tampa, FL: Greenberg Graphics; 2010:480-532.
- Powers CJ, George T, Fuchs HE. Constipation as a reversible cause of ventriculoperitoneal shunt failure. Report of two cases. J Neurosurg 2006;105(3 Suppl):227-230.
- Chazal J. Management of hydrocephalus in childhood. In: Sindou M, ed. Practical Handbook of Neurosurgery: From Leading Neurosurgeons. Springer Vienna; 2009:525-541.
- Drake JM, Kestle JR, Milner R, et al. Randomized trial of cerebrospinal fluid shunt valve design in pediatric hydrocephalus. Neurosurgery 1998;43:294-303; discussion 303-305.
- Browd SR, Ragel BT, Gottfried ON, Kestle JR. Failure of cerebrospinal fluid shunts: Part I: Obstruction and mechanical failure. Pediatr Neurol 2006;34:83-92.
- Kestle JR, Drake JM, Cochrane DD, et al. Lack of benefit of endoscopic ventriculoperitoneal shunt insertion: A multicenter randomized trial. J Neurosurg 2003;98:284-290.
- Migita R, Woodward T. Ventriculoperitoneal and other intracranial shunts. In: Baren JM, Rothrock SG, Brennan JA, Brown L, eds. Pediatric Emergency Medicine. Philadelphia, PA: Elsevier; 2008:1189-1193.
- Wallace AN, McConathy J, Menias CO, et al. Imaging evaluation of CSF shunts. AJR Am J Roentgenol 2014;202:38-53.
- Cohen JS, Jamal N, Dawes C, et al. Cranial computed tomography utilization for suspected ventriculoperitoneal shunt malfunction in a pediatric emergency department. J Emerg Med 2014;46:449-455.
- Adams DJ, Rajnik M. Microbiology and treatment of cerebrospinal fluid shunt infections in children. Curr Infect Dis Rep 2014;16:427. doi: 10.1007/s11908-014-0427-8.
- Sivaganesan A, Krishnamurthy R, Sahni D, Viswanathan C. Neuroimaging of ventriculoperitoneal shunt complications in children. Pediatr Radiol 2012;42:1029-1046.
- Madsen MA. Emergency department management of ventriculoperitoneal cerebrospinal fluid shunts. Ann Emerg Med 1986;15:1330-1343.
- Di Rocco F, Garnett M, Rouheau T, et al. Hydrocephalus. In: Lumenta CB, Di Rocco C, Haase J, Mooij JJ, eds. European Manual of Medicine: Neurosurgery. Springer; 2010:539-543.
- Symss NP, Oi S. Is there an ideal shunt? A panoramic view of 110 years in CSF diversions and shunt systems used for the treatment of hydrocephalus: From historical events to current trends. Childs Nerv Syst 2015;31:191-202.
- Garton HJ, Kestle JR, Drake JM. Predicting shunt failure on the basis of clinical symptoms and signs in children. J Neurosurg 2001;94:202-210.
- Simon TD, Whitlock KB, Riva-Cambrin J, et al. Revision surgeries are associated with significant increased risk of subsequent cerebrospinal fluid shunt infection. Pediatr Infect Dis J 2012;31:551-556.
- Desai KR, Babb JS, Amodio JB. The utility of the plain radiograph “shunt series” in the evaluation of suspected ventriculoperitoneal shunt failure in pediatric patients. Pediatr Radiol 2007;37:452-456.
- Coley BD, Kosnik EJ. Abdominal complications of ventriculoperitoneal shunts in children. Semin Ultrasound CT MR 2006;27:152-160.
- Browd SR, Gottfried ON, Ragel BT, Kestle JR. Failure of cerebrospinal fluid shunts: Part II: Overdrainage, loculation, and abdominal complications. Pediatr Neurol 2006;34:171-176.
- Wu JC, Chen YC, Liu L, et al. Younger boys have a higher risk of inguinal hernia after ventriculo-peritoneal shunt: A 13-year nationwide cohort study. J Am Coll Surg 2012;214:845-851.
- Greenberg MS. Infections. In: Handbook of Neurosurgery. 7th ed. Tampa, FL: Greenberg Graphics; 2010:533-612.
- Sciubba DM, Stuart RM, McGirt MJ, et al. Effect of antibiotic-impregnated shunt catheters in decreasing the incidence of shunt infection in the treatment of hydrocephalus. J Neurosurg 2005;103(2 Suppl):131-136.
- Chumas PD, Armstrong DC, Drake JM, et al. Tonsillar herniation: The rule rather than the exception after lumboperitoneal shunting in the pediatric population. J Neurosurg 1993;78:568-573.
- Ouellette D, Lynch T, Bruder E, et al. Additive value of nuclear medicine shuntograms to computed tomography for suspected cerebrospinal fluid shunt obstruction in the pediatric emergency department. Pediatr Emerg Care 2009;25:827-830.
- Piatt JH,Jr. Pumping the shunt revisited. A longitudinal study. Pediatr Neurosurg 1996;25:73-76; discussion 76-77.
- Pitetti R. Emergency department evaluation of ventricular shunt malfunction: Is the shunt series really necessary? Pediatr Emerg Care 2007;23:137-141.
- assilyadi M, Tataryn ZL, Alkherayf F, et al. The necessity of shunt series. J Neurosurg Pediatr 2010;6:468-473.
- Sellin JN, Cherian J, Barry JM, et al. Utility of computed tomography or magnetic resonance imaging evaluation of ventricular morphology in suspected cerebrospinal fluid shunt malfunction. J Neurosurg Pediatr 2014;14:160-166.
- Iskandar BJ, Sansone JM, Medow J, Rowley HA. The use of quick-brain magnetic resonance imaging in the evaluation of shunt-treated hydrocephalus. J Neurosurg 2004;101(2 Suppl):147-151.
- Patel DM, Tubbs RS, Pate G, et al. Fast-sequence MRI studies for surveillance imaging in pediatric hydrocephalus. J Neurosurg Pediatr 2014;13:440-447.
- Thompson EM, Baird LC, Selden NR. Results of a North American survey of rapid-sequence MRI utilization to evaluate cerebral ventricles in children. J Neurosurg Pediatr 2014;13:636-640.
- Le A, Hoehn ME, Smith ME, et al. Bedside sonographic measurement of optic nerve sheath diameter as a predictor of increased intracranial pressure in children. Ann Emerg Med 2009;53:785-791.
- Vega RA, Buscher MG, Gonzalez MS, Tye GW. Sonographic localization of a nonpalpable shunt: Ultrasound-assisted ventricular shunt tap. Surg Neurol Int 2013;4:101. doi: 10.4103/2152-7806.116151. eCollection 2013.
- Hamburg LM, Kessler DO. Rapid evaluation of ventriculoperitoneal shunt function in a pediatric patient using emergency ultrasound. Pediatr Emerg Care 2012;28:726-727.
- Mitra DK, Spicer RD, MacKinnon AE. Assessment of CSF shunts by doppler ultrasound. Z Kinderchir 1981;34:330-334.
- Kaplan M, Yakar H, Orhan H, Erol FS. Evaluation of doppler USG and CSF flow wave patterns in the diagnosis of ventriculoperitoneal shunt obstructions. Pediatr Neurosurg 2007;43:468-471.
- Horton C, Byrd L, Lucht H, Higby N. Emergency care of children with high-technology neurologic disorders. Clin Pediatr Emerg Med 2012;13:114-124.
- Lemcke J, Meier U, Muller C, et al. Safety and efficacy of gravitational shunt valves in patients with idiopathic normal pressure hydrocephalus: A pragmatic, randomised, open label, multicentre trial (SVASONA). J Neurol Neurosurg Psychiatry 2013;84:850-857.
- Pollack IF, Albright AL, Adelson PD. A randomized, controlled study of a programmable shunt valve versus a conventional valve for patients with hydrocephalus. hakim-medos investigator group. Neurosurgery 1999;45:1399-1408; discussion 1408-1411.
- Constantini S. The ISPN shunt guide. ISPN: International Society for Pediatric Neurosurgery Web site. Available at: http://ispn.guide/book/The%20ISPN%20Guide%20to%20Pediatric%20Neurosurgery/Hydrocephalus%20and%20Other%20Anomalies%20of%20CSF%20Circulation%20in%20Children/ispn-shunt-guide. Updated 2015. Accessed May 20, 2015.
- Tuan TJ, Thorell EA, Hamblett NM, et al. Treatment and microbiology of repeated cerebrospinal fluid shunt infections in children. Pediatr Infect Dis J 2011;30:731-735.
MONOGRAPH: The prompt recognition and treatment of shunt failure in the emergency department is of the utmost importance to limit morbidity and mortality.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.