Verbal consent and decision tree assist research community
Keeping up with changes is full-time job
As informed consent and IRB work evolve, new tools can help ease the transformation. For instance, IRBs and researchers might benefit from new informed consent tools and help with determining exemptions.
A dozen years ago, many IRBs worked with a simple, written informed consent (IC) form. There were fewer IC nuances, even with social-behavioral-educational research (SBER) studies.
In those days, if a researcher wanted to use verbal consent and the IRB agreed to waive written consent, then the IRB might offer the investigator a written consent form and tell him or her to remove the name, date, and signature line, says Carrie McDaniel, CIP, senior IRB regulatory analyst at the human subjects committee, social-behavioral IRB, Yale University in New Haven, CT.
“So the forms still looked very formal and didn’t work very well with research involving ethnographies or casual conversations where they needed a more casual consent,” McDaniel explains. “Over many years we revised the verbal informed consent to capture elements of informed consent, but more in a verbal script.”
The verbal consent template, revised this year, lends itself to a speaking, rather than written, voice.
The Yale University IRB also has learned over the years that the traditional IC form needs to be tweaked for other types of research, as well. These include surveys conducted online, McDaniel adds.
“You have to have something that doesn’t require a name, date, and signature,” she says. “For online, you need a click-through consent where they can click ‘yes.’”
IRB staff created a rough draft of the verbal IC template and consulted with the IRB chair and vice chair for their feedback on it, says Cathleen M. Montano, JD, CIP, IRB manager in the human research protection program at Yale University.
“Our typical process for making changes is we work on it in-house and then move it up to the chairs of the relevant IRBs for their review,” Montano says. “If there are further concerns raised, then we take it to the full board.”
The verbal IC is worded to make certain participants understand that their involvement in the study is entirely voluntary, McDaniel notes.
“You go through the entire paragraph so they know they do not have to participate and that the decision of whether or not to participate will not affect their relationship with any of those entities,” she explains. “So if I’m interviewing patients at a clinic setting and perhaps the clinic staff had referred them to research staff, we want them to know whether or not they participate will not affect services they are receiving at this clinic.”
When principal investigators adapt the template to their own studies, they submit their revised version to the IRB for approval, McDaniel says.
“The fact that it’s a verbal informed consent means it takes less time,” she adds. “There’s no need to have it be lengthy.”
The follwing is an example of the verbal consent script template’s language regarding risks:
• “You may experience description of risks (distress over the nature of the questions, potential for informational risks, etc.) OR (if no known risks, state the following) — There are no known or anticipated risks to you for participating. (If no benefit to participants, state) — Although this study will not benefit you personally, we hope that our results will add to the knowledge about (describe public good. If benefit to participants, state what they are and) — we hope that our results will add to the knowledge about (describe public good).”
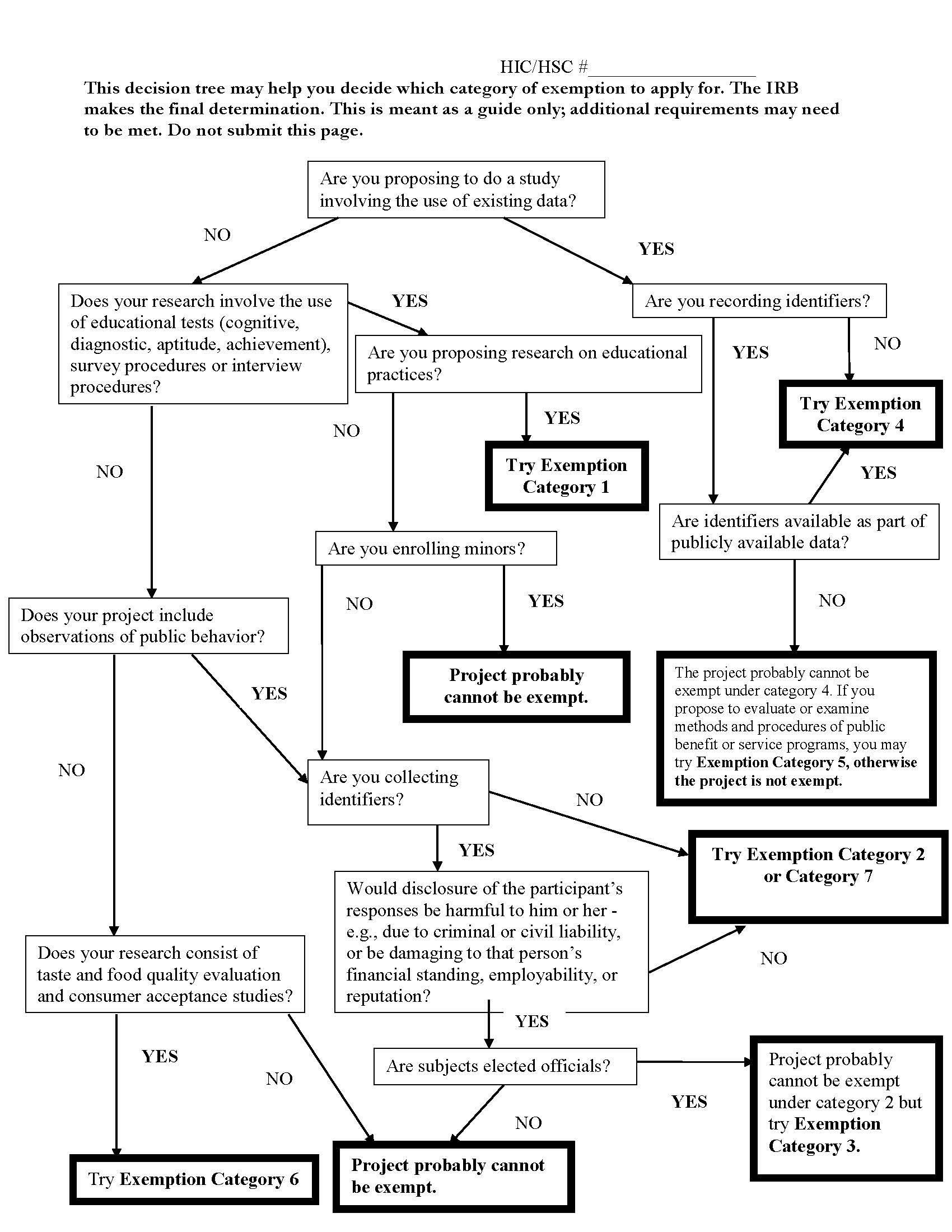
The Yale IRB also recently began to use a decision tree for determining standard or exempt research. A member of the IRB staff felt it would help researchers to have some type of guidance in determining whether their study fit into one of the exempt categories, Montano says.
“There hadn’t been a specialized application form to separate the types of research,” she adds. “So it occurred to our colleague that we ought to have a guidance document, such as a decision tree, to help researchers self-identify what they’re doing and whether it falls into an exempt category.”
The decision tree starts with the simple question, “Are you proposing to do a study involving the use of existing data?” (See Yale decision tree chart in this issue.)
So far, the decision tree has been well-received by investigators, McDaniel says.
“It looks complicated, but if you follow the arrows, it points you to the right form,” she notes.
“The feedback I have from investigators is that they love it; they can go through it and answer these questions without the IRB’s help,” she adds. “This has led to the IRB office receiving fewer inquiries about exempt or expedited reviews than before, and it empowers researchers to figure out what they’re doing and which form to use.”
Updating forms and tools is an important part of the IRB office’s work, so there is dedicated staff that are watchdogs for regulatory, guidance, and other changes necessitating a change, Montano says.
The IRB also has posted a table online that identifies the changes they’ve made to policies and procedures and forms, Montano notes.
“It’s a good resource for researchers to readily see what has been substantially changed,” she adds.
“We also receive feedback from our investigators, who let us know if there’s confusion about answering a question or if they have more questions about a topic,” she adds. “Our educational specialist is particularly in tune with changes and trends and what needs to be updated for our research community.”
As informed consent and IRB work evolve, new tools can help ease the transformation.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
