ID Grand Rounds — Stanford University
A 69-year-old male with a long-standing history of primary myelodysplastic syndrome (MDS) underwent non-myeloablative allogeneic, matched unrelated donor (MURD), hematopoietic stem cell transplantation (HSCT) in November 2013. Nine months following transplantation, the patient developed idiopathic thrombocytopenia purpura (ITP) that required IVIG administration coupled with high-dose prednisone (80 mg/day). Tacrolimus was discontinued upon ITP diagnosis. Eleven months after transplantation and 2 months after ITP was diagnosed, the patient presented to the outpatient hematology clinic due to high fever (39.5°C), right shoulder pain, pleuritic chest pain, dyspnea, and altered mental status. He was admitted to the hospital for further workup.
At admission, his immunosuppressive and antimicrobial prophylaxis regimen included prednisone (15 mg/daily), mycophenolate mofetil, atovaquone (750 mg three times per week), acyclovir (400 mg TID), and voriconazole (400 mg BID). He was a retired physician living in northern California. He reported active outdoor activities, including short-distance hikes, gardening, and construction at home that started 2 months preceding hospital admission.
PHYSICAL EXAMINATION
On physical examination, the patient had mild respiratory distress. His temperature was 39.7 °C, blood pressure was 127/68 mmHg, heart rate was 84 beats per minute, and respiratory rate was 20 breaths per minute. His oxygen saturation was 92% at 2 L/min of oxygen by nasal cannula. No oral mucosa lesions were appreciated. Hypoventilation and crackles were noted at the right lung base. Abdominal examination was benign, with no hepatosplenomegaly. He had 2+ lower extremity edema and multiple ecchymosis throughout skin. He appeared drowsy but answered questions appropriately. Cranial nerves examination was intact. No focal neurologic deficit was noted. The rest of the examination was unremarkable.
LAB TESTING AND IMAGING
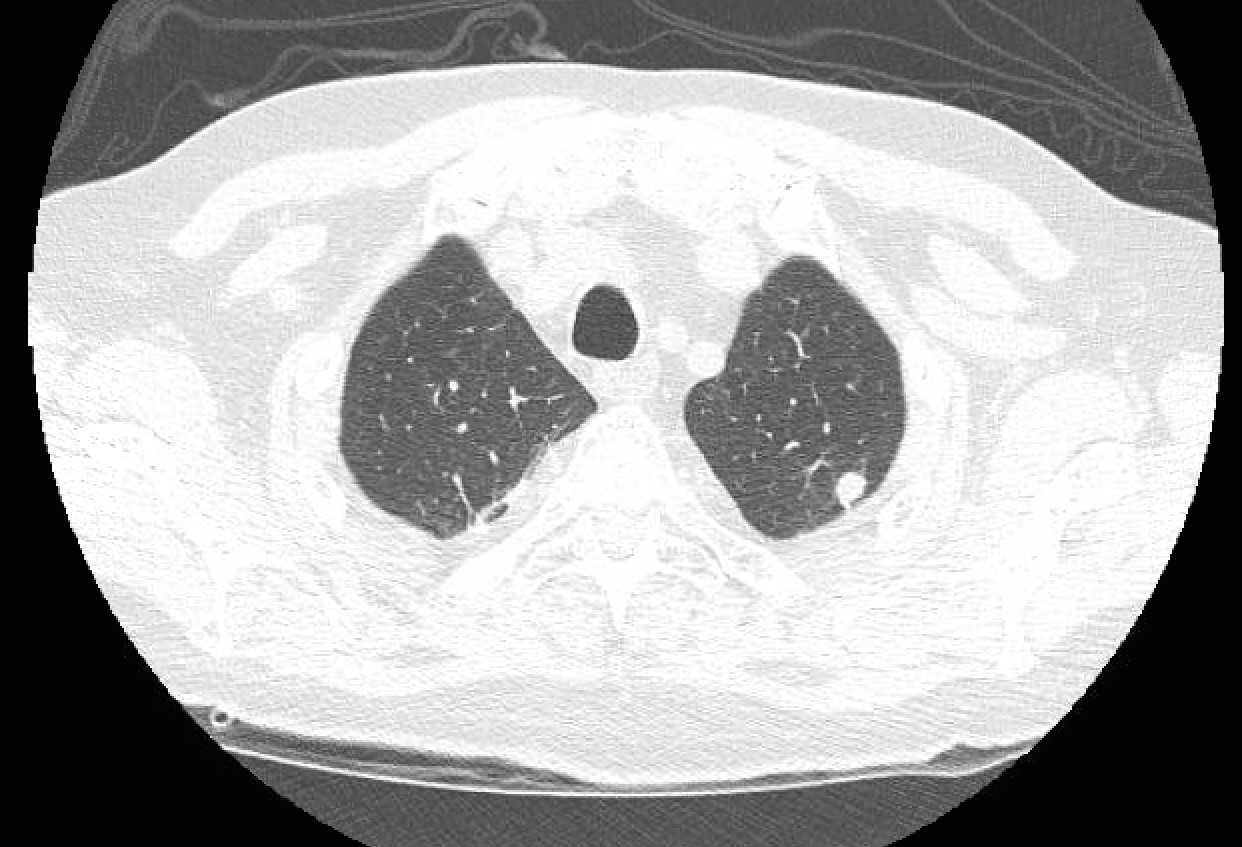
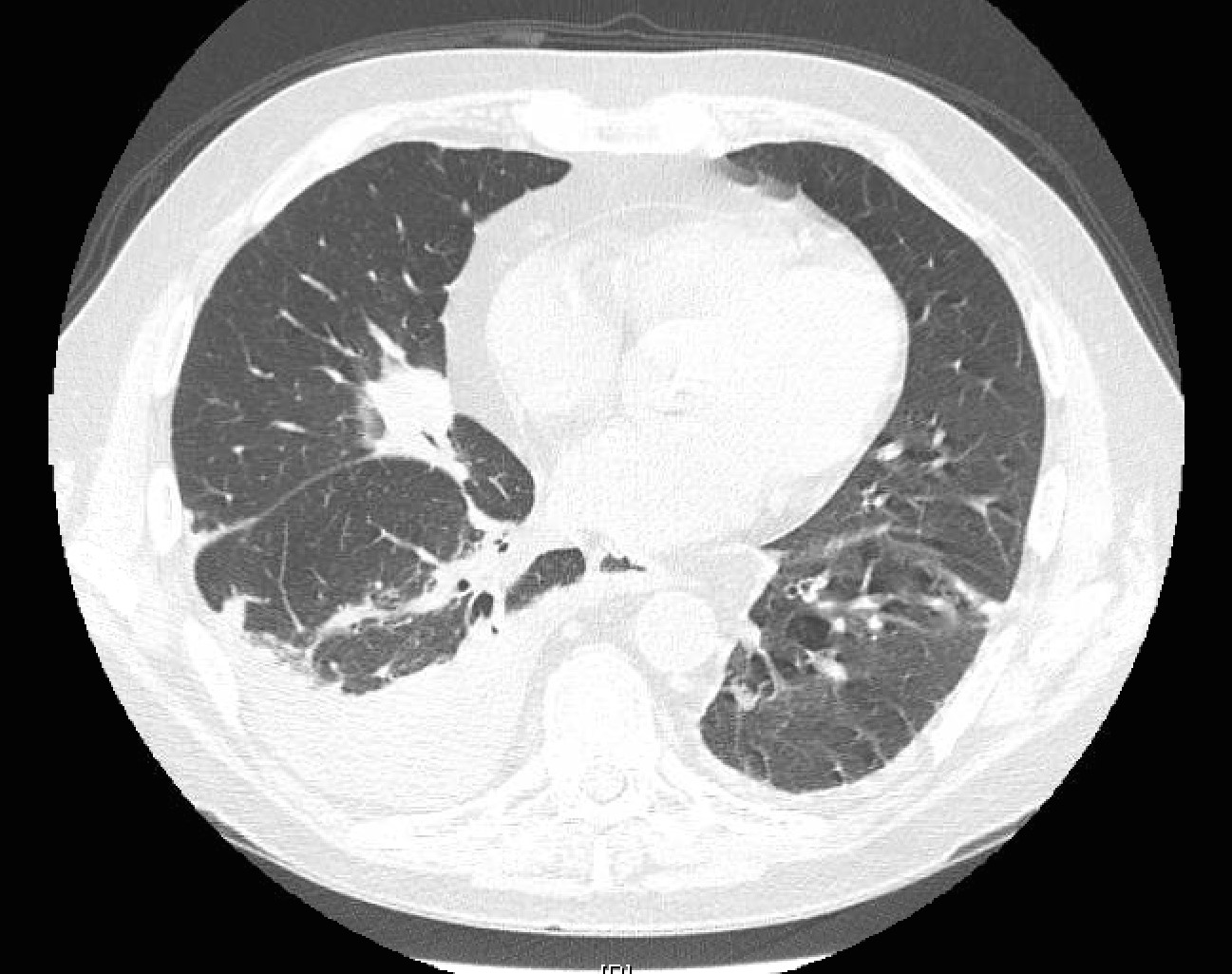
Laboratory investigation revealed leukopenia (2,100 WBC, 34% neutrophils, 44% lymphocytes); hemoglobin was 9.8 g/dL, and platelet count was 125.000 platelets per microliter of blood. His creatinine was 1.5 mg/dL. Liver function tests were normal. Blood cultures, pneumococcal urine antigen, and legionella urine antigen were negative. Blood cultures were drawn upon admission. Cryptococcal antigen by lateral flow testing, serum Aspergillus galactomannan antigen testing, Coccidioides immunodiffusion titers, and Histoplasma urine antigen were all negative. Initial chest X-ray showed multifocal areas of consolidation, scattered pulmonary nodules, and right-sided pleural effusion. A non-contrast CT of the chest showed multiple solid nodules and areas of alveolar consolidation, along with right pleural effusion. (See Figure 1.)
Figure 1: Non-contrast CT of the Thorax Showing Multiple Areas of Solid Nodules, Right Lower and Middle Lobe Consolidation, and Right Pleural Effusion.
CLINICAL COURSE
The patient was admitted to the medical ward. Empirical antibiotic therapy was started with IV vancomycin and cefepime. At day 2 following admission, the patient underwent fiberoptic bronchoscopy with bronchoalveolar lavage (BAL). Bacterial, fungal, and AFB cultures from BAL were non-diagnostic. Direct fluorescent antibody (DFA) testing for Pneumocystis was negative. At day 4 of hospital stay, blood cultures turned positive for Nocardia farcinica. This finding prompted brain imaging, and MRI of the brain showed multiple areas of rim-enhancing lesions localized across fronto-parietal-temporal and cerebellar areas bilaterally. No meningeal enhancement or hydrocephalus was noted.
The patient was started on IV trimethoprim- sulfamethoxazole (TMP-SMX), meropenem, and linezolid for empirical Nocardia therapy. Once antibiotic susceptibility testing performed by Etest confirmed TMP-SMX susceptibility, the patient was switched to oral TMP-SMX at doses of 2 DS tablets every 8 hours. Two months following hospital admission, the patient remains in good clinical condition. Follow-up brain MRI imaging showed interval improvement in the size of the lesions and overall decreased burden of disease. After 6 months of TMP-SMX therapy, the patient was switched to suppression doses (1 DS tab daily) for chronic secondary prophylaxis against Nocardia.
Diagnosis: Disseminated Nocardiosis
DISCUSSION
Here, we present a case of an allogeneic-HSCT recipient with disseminated nocardiosis manifested with bacteremia, pneumonia, and space-occupying lesions in the central nervous system (CNS). Brain abscess is a rare complication following HSCT. The differential diagnosis of space-occupying lesions in the CNS in HSCT patients includes invasive fungal disease (most commonly aspergillosis followed by other filamentous as well as endemic fungi), bacterial brain abscess, Toxoplasma gondii encephalitis, Nocardia infection, and non-infectious conditions such as infarction or post-transplant lymphoproliferative disorder (PTLD).
Nocardia are filamentous, aerobic, Gram-positive branching rods, appearing weakly acid-fast staining due to the mycolic acid content of their cell wall. The reduced intensity of staining relative to that of mycobacteria is the result of differences in their mycolic acids, with those of Nocardia containing only 22-60 carbon atoms, while those of mycobacteria contain 60-80. Nocardia is an environmental saprophyte, whose normal habitat includes soil and decaying organic material. The main route of Nocardia infection is via the respiratory tract, with potential dissemination to distant sites, especially the CNS. Nocardia infection is typically regarded as opportunistic in patients with T-cell mediated deficiency, patients with chronic steroid use, or those under immunosuppression for prevention of organ transplant rejection.
Localized disease manifests most commonly as primary skin infection consisting of sporotrochoid lesions at the site of inoculation, often leading to pyogenic abscess, sinus tract, or destructive disease. In the immunocompromised host, the lung and CNS are commonly affected. In cases of pulmonary disease, chronic productive or nonproductive cough, dyspnea, hemoptysis, fever, and radiographic imaging patterns of consolidation, nodules, or cavitary disease have been described. CNS infection may present as headache, altered mental status, local neurological deficit, or fever. CNS lesions can be insidious in onset or even asymptomatic in the immunocompromised host.
The diagnosis of Nocardia infection has been significantly enhanced in the last two decades due to better detection and identification procedures in clinical microbiology. The microbiology laboratory should be notified in advance regarding the suspicion of Nocardia infection. Samples collected from lower respiratory tract, aspirates, or biopsy specimens are typically processed for Gram and modified acid-fast stains. Growth of Nocardia species usually takes between 3 to 5 days, as demonstrated in our case. In the most recent years, MALDI-TOF (matrix-assisted laser desorption ionization-time-of-flight) has been introduced to expedite Nocardia species identification, thus avoiding the need for phenotypic, enzymatic, and 16srRNA gene sequencing testing. Antibiotic susceptibility testing should be performed in all isolates of Nocardia following CLSI recommendations (CLSI).
TMP-SMX is the mainstay of treatment; initial combination therapy with other anti-Nocardia active agents such as imipenem, meropenem, linezolid, ceftriaxone, or amikacin is recommended in cases of disseminated disease or severe infection involving an immunocompromised host. Nocardia speciation and antimicrobial susceptibility testing should guide definitive therapy. There are a paucity of studies available to guide duration of therapy. In cases with deep-seated infection, clinical and radiographic monitoring is recommended to assist determination of the appropriate length of therapy.
The epidemiology of Nocardia infection in transplantation has been described in the last two decades. In a large series of SOT patients including 5126 patients, the incidence of Nocardia infection was 3.6%, 2.5%, 1.3%, 0.2%, 0.1% in lung, heart, intestinal, kidney, and liver transplant recipients, respectively. In 20% of cases, CNS involvement was identified by brain imaging. CMV disease, high doses of prednisone, and high doses of calcineurin inhibitors (cyclosporine or tacrolimus) were associated with increased risk of Nocardia infection. In this large series, 69% of patients developed Nocardia infection despite antimicrobial prophylaxis with TMP-SMX; nonetheless, this antimicrobial agent was used as the cornerstone of Nocardia therapy, with a rate of cure of 89%.1
A case series from three large academic cancer centers reported an incidence of 0.3% of Nocardia infection among allogeneic HSCT patients.2 The median time onset of Nocardia infection following HSCT was 210 days. The most common symptoms at clinical presentation were fever, productive cough, pleuritic chest pain, and weight loss. In 40% of cases, Nocardia infection developed despite TMP-SMX prophylaxis. Although the survival rate was high (84%), clinical prognosis was highly determined by the presence of CNS disease.
In conclusion, Nocardia infection can present as an opportunistic pathogen in HSCT patients, leading to disseminated disease (most commonly pneumonia and brain abscess), especially in the context of enhanced immunosuppression and absence of TMP-SMX-based antimicrobial prophylaxis.
REFERENCES
- Peleg AY, et al. Risk factors, clinical characteristics, and outcome of Nocardia infection in organ transplant recipients: A matched case-control study. Clin Infect Dis 2007;44:1307-1314.
- Van Burik JA, et al. Nocardiosis after bone marrow transplantation: A retrospective study. Clin Infect Dis 1997;24:1154-1160.
- Clinical and Laboratory Standards Institute (CLSI). 2011. Susceptibility testing of Mycobacteria, Nocardiae, and Other Aerobic Acintomycetes. Approved Standard. Second Edition. CLSI Document M24-A2. Clinical and Laboratory Standards Institute, Wayne, PA.
A 69-year-old male with allogeneic hematopoietic stem cell transplant with a space-occupying lesion of the central nervous system.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.