Ultrasound for Trauma
Authors
Cindy H. Hsu, MD, PhD, Trauma/Surgical Critical Care Fellow,
R Adams Cowley Shock Trauma Center, University of Maryland School of Medicine, Baltimore.
Jay Menaker, MD, FACEP, Associate Professor Department of Surgery, Associate Professor Department of Emergency Medicine, University of Maryland School of Medicine, R Adams Cowley Shock Trauma Center, Baltimore.
Peer Reviewer
Eric Brader, MD, FAAEM, Director of Emergency Ultrasound, Allegheny General Hospital, Pittsburgh, PA.
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, Dr. Dietrich (editor in chief), Dr. Menaker (author), Dr. Hsu (author), Dr. Brader (peer reviewer), Ms. Behrens (nurse reviewer), Ms. Mark (executive editor), Ms. Leslie Coplin (executive editor), and Mr. Landenberger (continuing education and editorial director) report no relationships with companies related to this field of study.
Table 1. Commonly Used Ultrasound Transducers
Curvilinear probe (2-5 MHz)
- This probe is low frequency and thus enables deeper penetration at the expense of lower resolution. It can be used for FAST or E-FAST and obstetric studies.
Linear probe (5-10 MHz)
- This probe is high frequency and thus enables high resolution at the expense of less penetration. It can be used for E-FAST, vascular access, ocular ultrasound, soft tissue imaging, and fractures.
Phased-array probe (2-4 MHz)
- This probe possesses similar frequency and level of penetration compared to the curvilinear probe but has a smaller footprint. It can be used for FAST or E-FAST to enable better cardiac imaging in between the intercostal spaces.
It is important to be familiar with the common ultrasound physics vocabulary to enhance the interpretation and communication of image findings with other clinicians. Table 2 lists some key terms, while Table 3 illustrates the relationships between the image medium, image echogenicity, and level of ultrasound wave attenuation.
Table 2. Ultrasound Physics Terms
- Attenuation: Reduction in amplitude and intensity of the ultrasound wave as a function of distance through the imaging medium.
- Anechoic: Complete attenuation and thus complete absence of returning sound wave. The structure appears as a dark image.
- Hypoechoic: Large amount of attenuation and thus not much sound wave returned to the transducer. The structure will appear darker than surrounding tissue.
- Isoechoic: Produces ultrasound echoes equal to those of neighboring or of normal tissues.
- Hyperechoic: Opposite of hypoechoic due to poor propagation of sound wave through the medium. The structure will appear brighter than surrounding tissue.
Table 3. Medium Type, Image Appearance, and Ultrasound Wave Attenuation
Medium |
Image Appearance |
Level of Attenuation |
|
Air |
Poor propagation |
Low |
|
Bone |
Very echogenic (very bright) |
|
|
Muscle |
Echogenic (bright) |
|
|
Liver/Kidney |
Echogenic (less bright) |
Middle |
|
Fat |
Hypoechoic (dark) |
|
|
Blood |
Hypoechoic (darker) |
High |
|
Fluid |
Hypoechoic (very dark) |
The most important ultrasound mode is the “B-mode,” or brightness-mode. It provides structural information utilizing different shades of gray in a two-dimensional image. M-mode, or “motion mode,” captures returning echoes in only one line of the B-mode image and displays them over a time axis. Movement of structures positioned in that line can then be visualized. M-mode and B-mode both can be displayed simultaneously on the ultrasound monitor.
Focused Assessment with Sonography for Trauma
The term Focused Abdominal Sonography for Trauma was first used in 1996 to describe a standard set of ultrasound examinations for the evaluation of injured patients. The meaning of the acronym was later changed to Focused Assessment with Sonography for Trauma (FAST) to reflect the applications outside the abdomen.9 The goal of the FAST exam is to rapidly identify hemoperitoneum and hemopericardium.
The FAST exam is typically performed during the secondary survey according to the Advanced Trauma Life Support (ATLS) guidelines. However, in the appropriate clinical scenario, it may be performed during the primary survey to guide the decision for operative management of an unstable patient. Compared to the FAST exam, a CT scan is more sensitive for the detection of intraperitoneal hemorrhage and solid injury and, thus, is the study of choice for stable patients.10 However, CT scan warrants exposure to radiation and often requires patients to be transported out of the resuscitation area. Diagnostic peritoneal lavage (DPL), while highly sensitive for the detection of hemoperitoneum, carries a 1-2% complication rate and is now infrequently performed.11
The FAST exam consists of four views and 10 total spaces. (See Figure 1.) The curvilinear probe is often used for its larger footprint and excellent tissue penetration. However, the phased-array probe can also be used, as its smaller footprint enables easier cardiac imaging between the intercostal spaces. The probe indicator points toward the patient’s head for the right upper quadrant and left upper quadrant views, and points toward the patient’s right side for the subxiphoid and suprapubic views.
Figure 1. FAST Exam
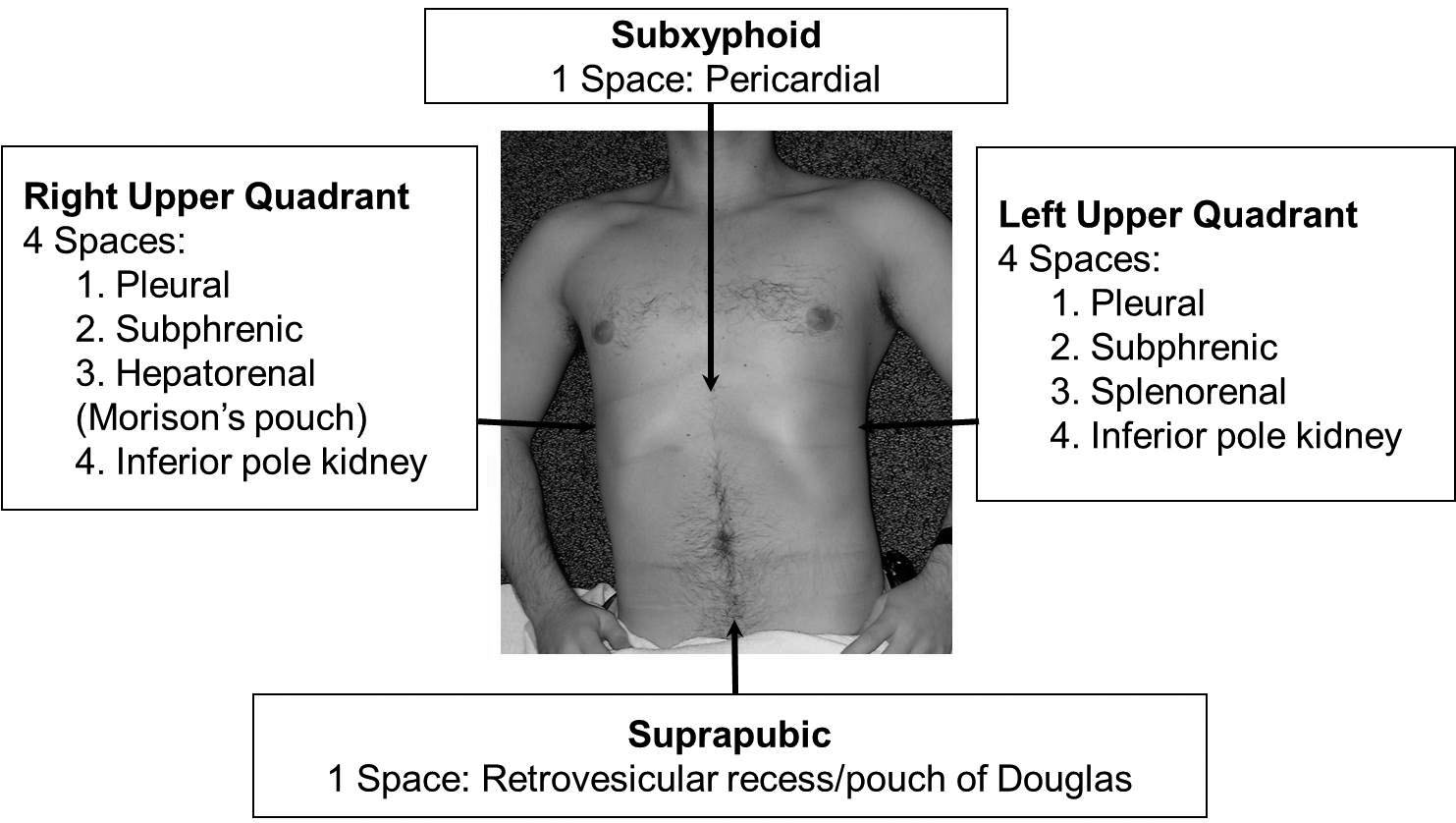
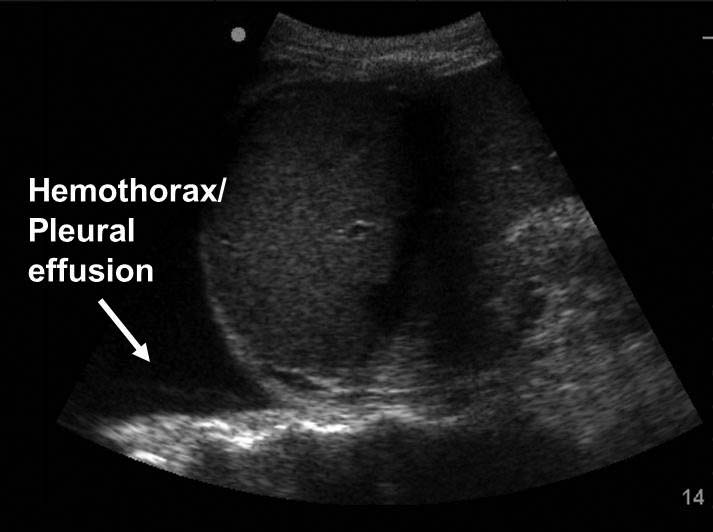
Right Upper Quadrant. This view consists of Morison’s pouch (hepatorenal space), right subphrenic, inferior pole of the right kidney, and right pleural space (see Figure 2A). Free fluid will first accumulate at the inferior pole of the right kidney as an anechoic stripe then progress to Morison’s pouch (see Figure 2B), the most dependent region in a supine patient. Thus, Morison’s pouch has the highest sensitivity for the detection of intraperitoneal fluid. The placement of the patient in the Trendelenburg and right lateral decubitus positions can further increase the sensitivity of FAST exam. Care also should be taken to carefully isolate the area between the dome of the liver and diaphragm to identify free fluid that may accumulate there. Slight cephalad movement of the transducer allows imaging of the right pleural space for free fluid (see Figure 2C). Caudal probe movement allows visualization of the inferior pole of the right kidney as well as the right paracolic gutter for free fluid assessment.11 One study showed that a good quality FAST exam with multiple views can reliably detect as little as 200 to 250 mL of free intraperitoneal fluid,3 while other studies showed up to 668 mL of fluid is required in supine and 443 mL in Trendelenburg patients.12
Figure 2A. Normal FAST Right Upper Quadrant View
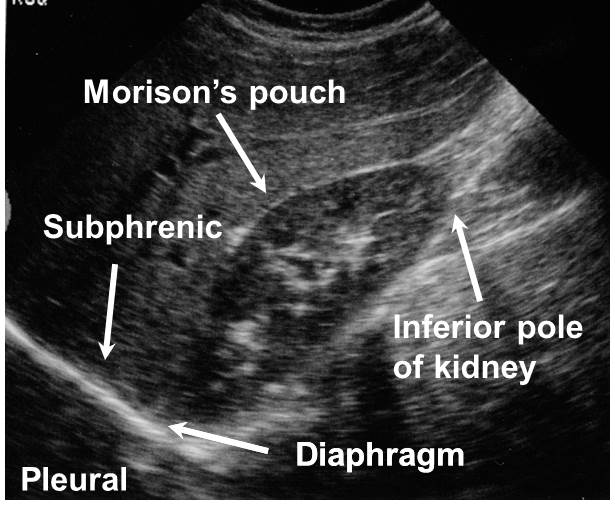
Figure 2B. FAST Free Fluid in Morison’s Pouch
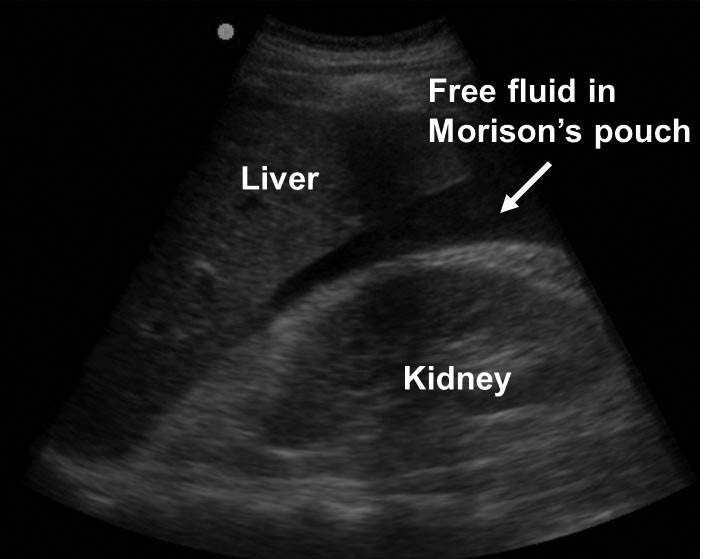
Figure 2C. Hemothorax/Pleural Effusion
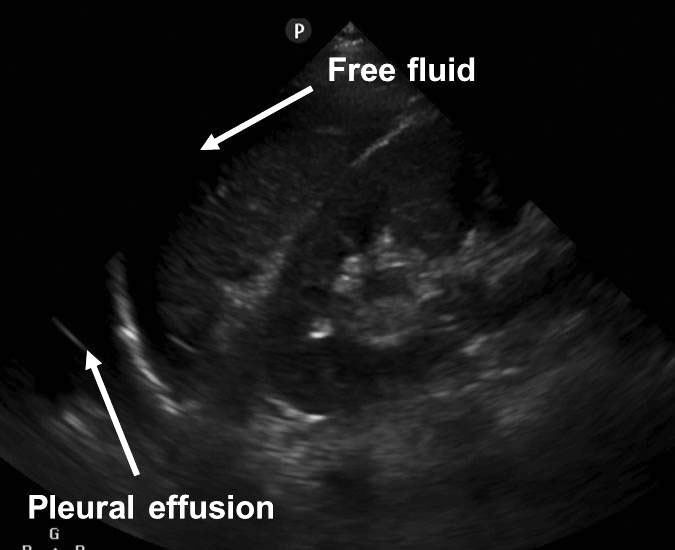
Left Upper Quadrant. This view consists of left subphrenic, inferior pole of the left kidney, splenorenal recess, and left pleural space (see Figures 3A and 3B). It is technically the most challenging view, as the splenorenal recess is more superior and posterior than the Morison’s pouch. The view can be improved by placing the probe in the posterior axillary line with the operator’s knuckles on the bed. The probe indicator can be rotated so that it is parallel to the rib spaces to minimize rib shadowing. The most dependent area of this view is the left subphrenic space and, thus, intraperitoneal fluid will first collect there (see Figure 3C).
Figures 3A and 3B. Normal FAST LUQ
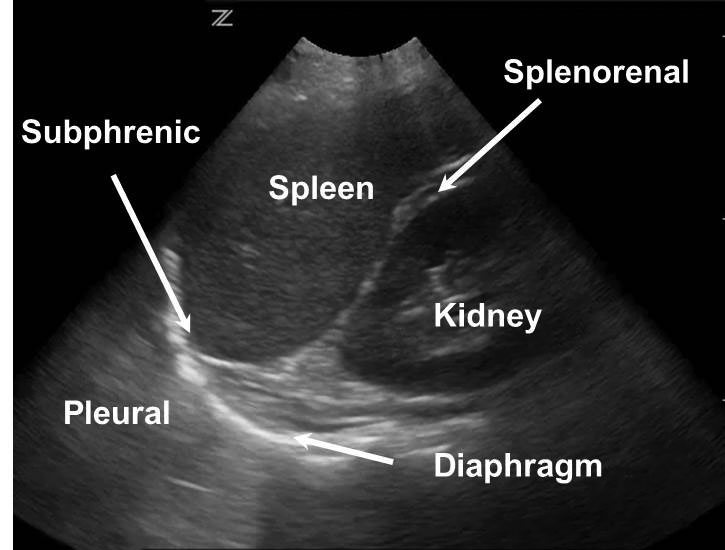
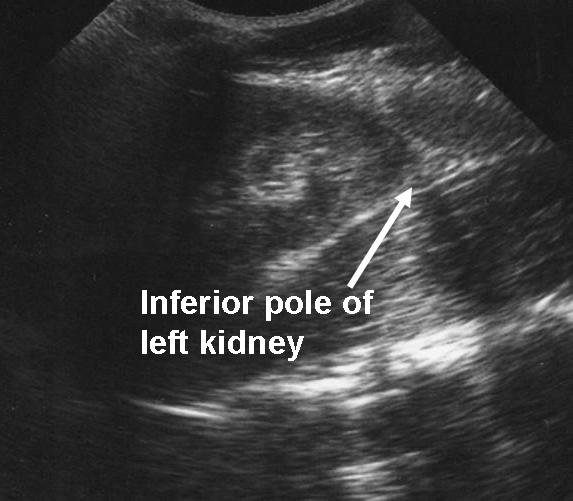
Figure 3C. Pleural Effusion and Free Fluid in Subphrenic Space
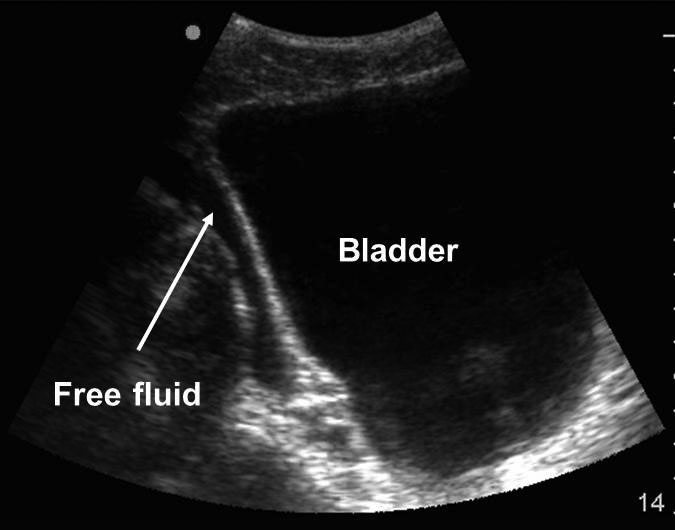
Suprapubic. This view allows assessment of the most dependent space in the peritoneum for free fluid, which is the retrovesicular recess for males and the pouch of Douglas for females. (See Figure 4A.) Analysis through a fluid-filled bladder may help with the assessment of pelvic fluid. The bladder should be scanned in its entirety in both the sagittal and transverse planes. When free fluid is present, it is noted most often posterior or superior to the bladder and uterus. (See Figure 4B.) Free fluid from an isolated pelvic injury would accumulate first in the pelvis, then spread up to Morison’s pouch, then the perisplenic area.11
Figure 4A. Normal Suprapubic
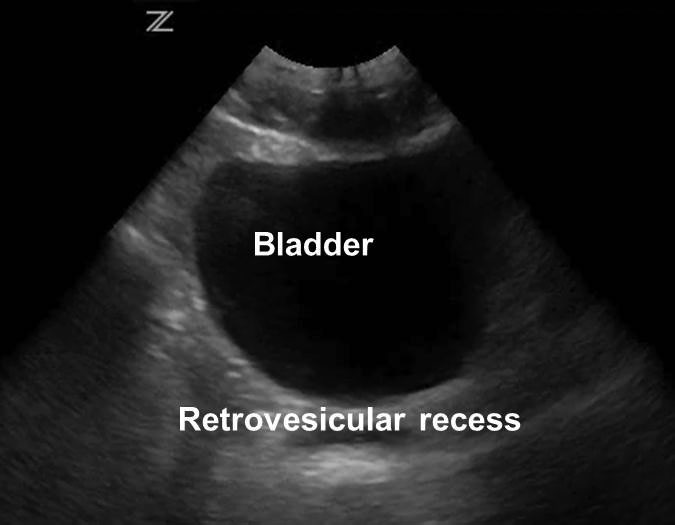
Figure 4B. Free Fluid Around Bladder
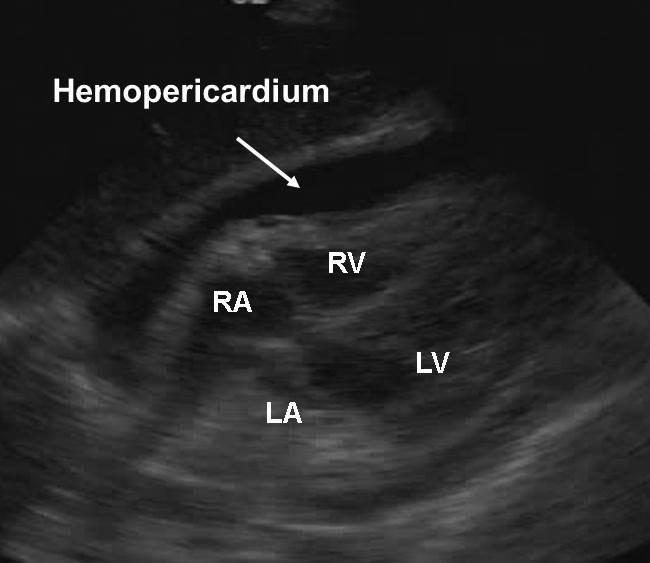
Subxiphoid. This view uses the left lobe of the liver as an acoustic window for analysis of the heart. (See Figure 5A.) Sometimes, alternative cardiac windows such as the parasternal or apical 4-chamber views may be necessary if an adequate subxiphoid view cannot be obtained.13 Fluid will appear as a dark anechoic stripe between the visceral and parietal pericardium. (See Figure 5B.) Epicardial fat pad may sometimes be mistaken for pericardial effusion. One can distinguish the two by noting that fat pads are attached to the heart and thus move with it during contractions; pericardial fluid or blood clots do not move with the heart.11 The presence of cardiac tamponade (right ventricular collapse during diastole) and cardiac function abnormalities can also be assessed. The detection of posterior hemopericardium is not as sensitive as that of anterior hemopericardium, especially in the presence of concomitant left hemothorax.11,14-17 The subxiphoid view should be repeated after the hemothorax has been evacuated by tube thoracostomy, although a false-negative remains possible if the hemopericardium continues to evacuate into the thoracic cavity.11
Figure 5A. Normal Subxiphoid View
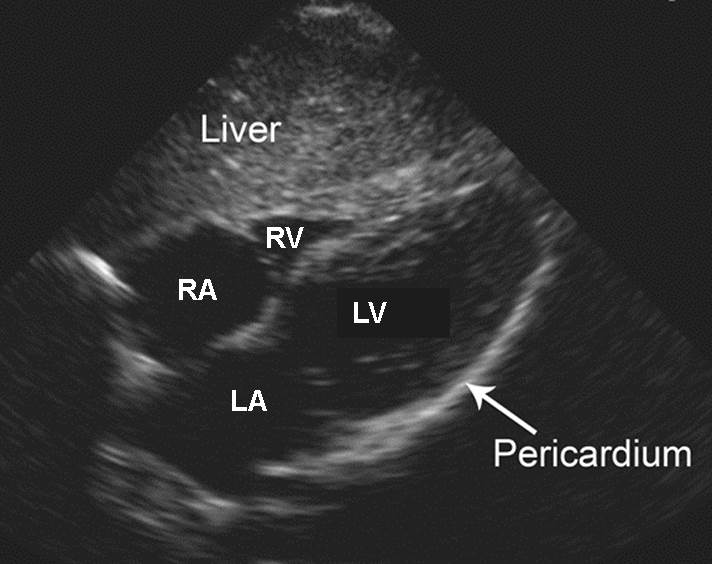
Figure 5B. Hemopericardium
Utility of FAST Exam. The reported sensitivity of FAST for detection of free intraperitoneal fluid varies, ranging from 62% to 99%, with negative predictive values of 89% to 99%.10,14,19 In most trauma management algorithms, if a patient is stable and needs further imaging, a CT scan is the highest sensitivity and specificity. It gives specific information about the hollow viscus and solid organs, as well as the retroperitoneum, which the FAST exam is unable to evaluate.11,20,21
The FAST exam has been shown to be most useful in unstable trauma patients, especially when the cause of hypotension is unclear.20,22,23 When transporting the patient to the CT scanner is not an option, FAST can be used for the initial evaluation and operative planning for an unstable patient. In addition, the detection of hemopericardium is also clinically useful for patients with penetrating trauma. The FAST exam can also be repeated during the patient’s stay for routine reassessment or as a consequence of clinical decompensation.11
For the hemodynamically stable patients with penetrating injuries to the thoracoabdominal region, the sensitivity of the FAST exam for the detection of intraperitoneal fluid is variable. In a meta-analysis of eight prospective observational studies,24 its sensitivity ranged from 24.2% to 100%, with a specificity of 94.1 to 100.0% and varied injury prevalence of 24.2% to 56.3%.25-31 No patients with an initial negative FAST exam in these studies died. Thus, a negative FAST exam does not preclude significant intraperitoneal injury after penetrating torso trauma in a stable patient and should prompt further imaging in those with a concerning mechanism or exam. Patients without evidence of peritonitis, hemodynamic instability, or hollow viscus injuries on CT scan may undergo trial nonoperative management that involves serial clinical examinations and laboratory tests, delayed laparoscopic evaluation of the diaphragm for left thoracoabdominal injuries, or endovascular interventions for solid organ injuries.32-33 Selective nonoperative management of both stab and gunshot injuries has been shown to be safe and can decrease the rate of unnecessary laparotomy, length of hospital stay, and management costs.32-34
For stable patients with blunt abdominal trauma, the FAST exam has been shown to lead to more accurate detection of intra-abdominal injury than physical examination,21 as well as reduced time to surgery, CT use, hospital stay, complications, and cost.35-36 Therefore, it is reasonable to use FAST as initial evaluation of blunt abdominal trauma, keeping in mind its limited sensitivity for intra-abdominal injuries.37
Limitations of FAST. The main limitation of the FAST examination is its limited sensitivity for the detection of injuries in the retroperitoneum, hollow viscus organs, diaphragm, or mesentery. Ultrasound may also be technically limited due to bowel gas, ascites, body habitus, subcutaneous emphysema, patient positioning, rate of bleeding, prior adhesions, and patient cooperation.13 Patients with concerning mechanisms or abnormal hemodynamics warrant additional investigations such as an exploratory laparotomy for unstable patients or additional CT imaging for stable patients. If it is clear that the patient will require emergent surgical intervention (e.g., bowel evisceration from a penetrating injury), then the FAST exam should be bypassed to avoid delays to definitive treatment. However, it may be necessary to exclude pericardial tamponade or pneumothorax before transferring a patient to the operating room for emergency laparotomy.13
Extended FAST
Pneumothoraces are common in trauma, but more than half are missed on a supine chest radiograph.38 POCUS has been shown to be equal or more sensitive than chest radiography, with high specificity for the diagnosis of pneumothorax in both blunt and penetrating trauma.39-49 The expansion of the FAST exam to evaluate the lungs for the presence of pneumothorax led to the term Extended-FAST (E-FAST).
The high-frequency probe is preferred by some operators to evaluate the thoracic cavity because it provides better resolution and detail of the pleura. However, the curvilinear or phased-array probe can also be used to expedite completion of the examination. To perform the lung exam, the operator places the probe on four locations of each chest.50 (See Table 4.)
Table 4. Probe Positions for Lung Ultrasound of E-FAST
- Anterior second intercostal space at the midclavicular line
- Fourth intercostal space at the anterior axillary line
- Sixth intercostal space at the midaxillary line
- Sixth intercostal space at the posterior axillary line
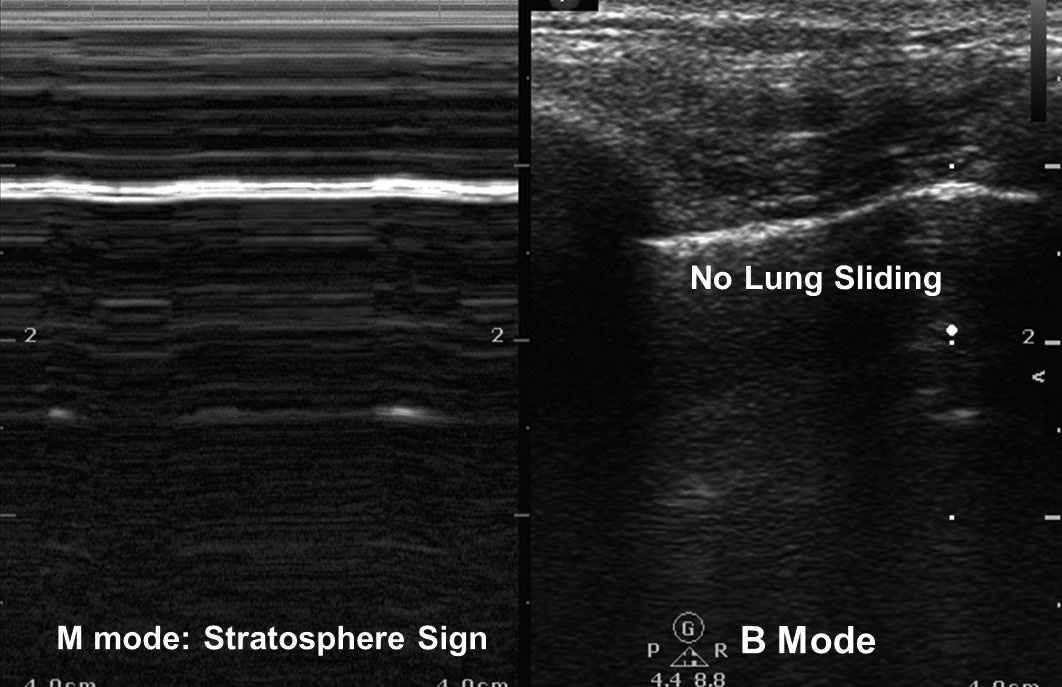
Lung sliding and the presence of comet tails are evidence of movement of the visceral on the parietal pleura. Comet tails are vertical reverberation artifacts arising from the pleural line. In M-mode, this lung sliding pattern is called the seashore sign.47 (See Figure 6A.) Pneumothorax leads to loss of contact between the visceral and parietal pleura and the loss of lung sliding. This leads to a distinct pattern on M-mode called the stratosphere or bar code sign. (See Figure 6B.) Lung sliding may be absent in patients who are not spontaneously breathing, even in the absence of pneumothorax. If no lung sliding is present, subtle cardiac pulsation of the parietal pleura at the lung periphery, known as the lung pulse, may be detected. This finding is equivalent to lung sliding.13,51 A lung point, or the transition between collapsed and normally expanded lung, is reportedly 100% specific for pneumothorax when present but may be difficult to detect.48
Figure 6A. Normal Lung
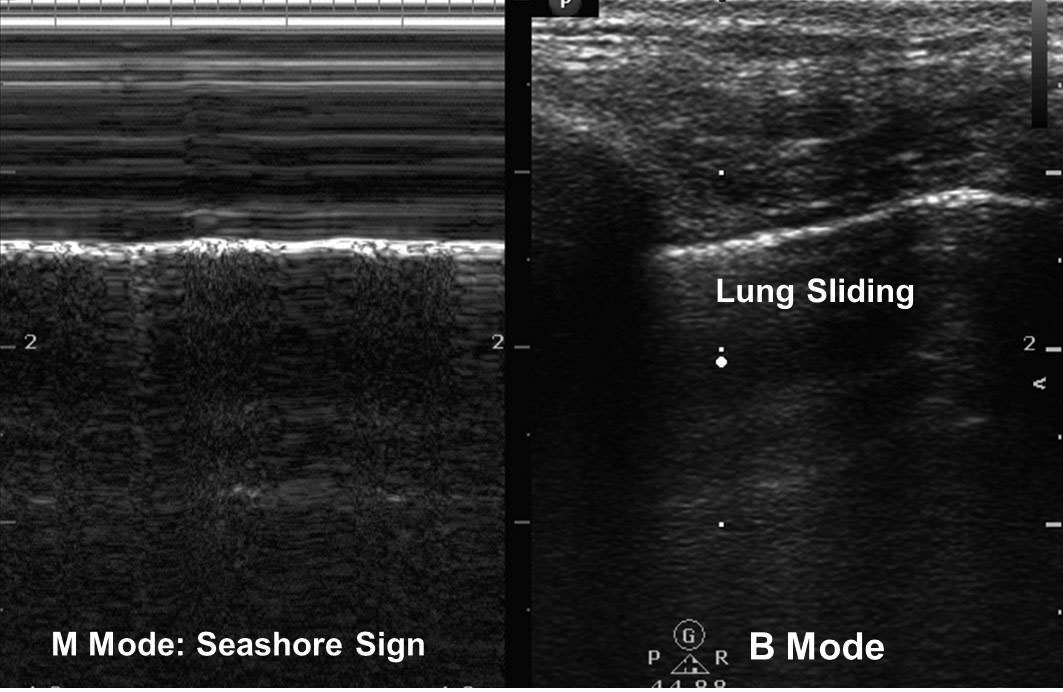
Figure 6B. Pneumothorax
Vascular Access
POCUS has been utilized as an adjunct for central venous catheter (CVC) placement, traditionally used for internal jugular and femoral vein access. In addition, ultrasound reduces the time and number of attempts for peripheral line placement,52 as well as the need for CVC placement due to difficult peripheral access.53-54 The use of ultrasound has not been shown to increase infection rates compared to traditionally placed lines.53-57 A linear probe is used with transverse and longitudinal views to optimize visualization for vascular access. Color doppler can also be used to differentiate an artery from a vein.
There has been growing interest in achieving subclavian vein access through the axillary vein, which can be visualized using ultrasound just distal to the subclavian vein.58 Ultrasound guidance resulted in faster access times and increased success at the first and second attempts for subclavian catheterization.59 In a recent meta-analysis of 10 randomized controlled trials (total of 2168 participants) of ultrasound compared to landmark technique for subclavian catheterization, the overall complication rates were reduced with ultrasound use compared to the landmark group. Subgroup analysis also demonstrated that dynamic 2D ultrasound also reduced inadvertent arterial puncture, pneumothorax, and hematoma formation.60
Volume Status
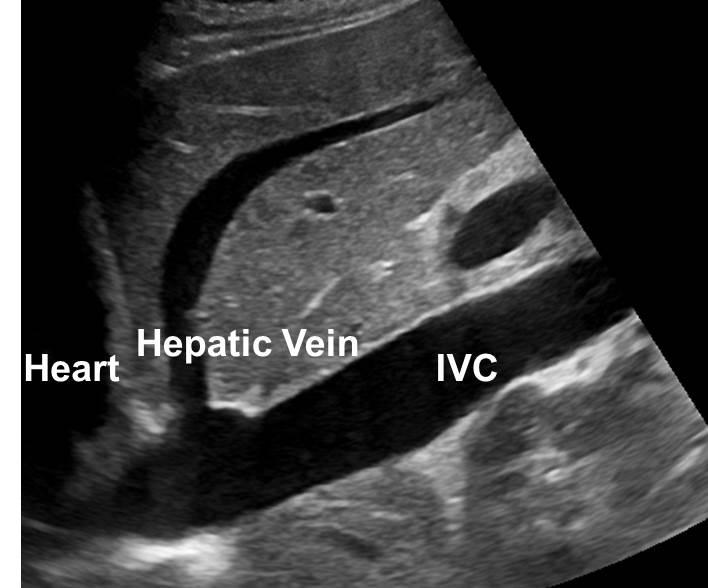
The volume status of an injured patient can be assessed by examining the diameter of the inferior vena cava (IVC) at end expiration.61-64 The curvilinear probe is positioned in the subxiphoid space with the indicator pointing toward the patient’s head. The IVC can be seen in its long axis as it enters the right atrium. (See Figure 7.) The IVC diameter should be assessed 2 cm distal to the hepatic vein’s entrance to the IVC.11 A diameter of less than 2 cm at end expiration of greater than 50% collapsibility index correlates with intravascular volume depletion.64-65
Figure 7. IVC Measurement for Volume Status
Ocular Ultrasound
Normal Eye. Ocular ultrasound can expedite the diagnosis and management of several ocular emergencies, including globe perforation, retrobulbar hematoma, retinal detachment, lens subluxation, vitreous hemorrhage, and intraocular foreign body.66-68 A linear transducer is most suitable for this examination. A large amount of water-soluble gel should be applied to the patient’s closed eyelid so that the transducer does not touch the eyelid. The globe should be scanned in both sagittal and transverse planes through closed eyelids.
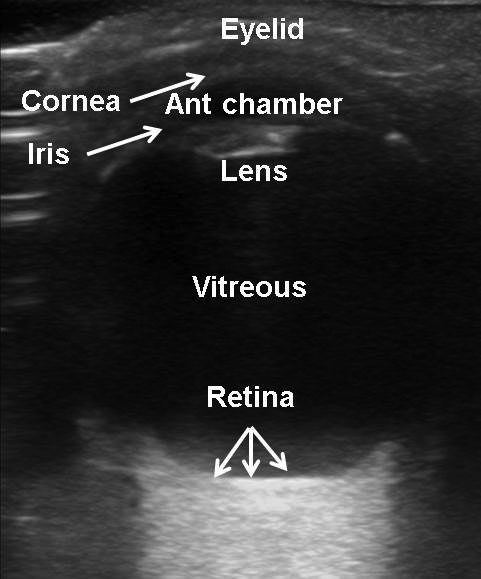
The normal eye appears as a circular hypoechoic structure. (See Figure 8A.) The cornea is seen as a thin hypoechoic layer parallel to the eyelid. The anterior chamber is filled with anechoic fluid and is bordered by the cornea, iris, and anterior reflection of the lens capsule. The iris and ciliary body are seen as echogenic linear structures extending from the peripheral globe toward the lens. The normal lens is anechoic. The normal vitreous chamber is filled with anechoic fluid. Vitreous is relatively echolucent in a young healthy eye. The optic nerve is visible posteriorly as a hypoechoic linear region radiating away from globe.50,69,70
Figure 8A. Normal Eye
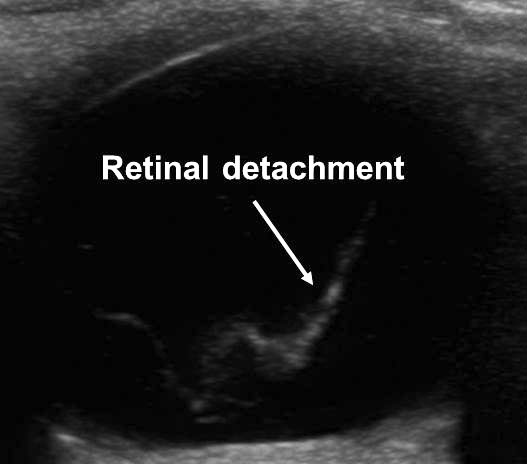
Ocular Injuries. A retinal detachment appears as a linear opacity within the vitreous chamber that moves in conjunction with eye movements. (See Figure 8B.) Vitreous hemorrhage consists of wavy linear or curved strands connecting with the retina that sway as the eye moves from side to side. A vitreous detachment occurs when the vitreous humor detaches from the posterior retina, which results in a mobile “swaying seaweed” appearance on ultrasound, where the vitreous appears separated from the retina.50,71
Figure 8B. Retinal Detachment
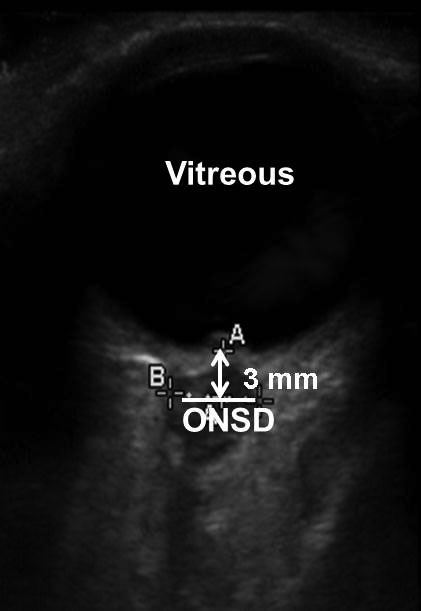
Intracranial Pressure Measurement. Blaivas et al described the use of ocular ultrasound to detect elevated intracranial pressure (ICP) among adult ED patients. A normal optic nerve sheath measures up to 5.0 mm in diameter on ultrasound. The optic nerve sheath diameter (ONSD) is measured 3 mm posterior to the globe for both eyes (see Figure 8C) because the results are more reproducible at this location due to greatest ultrasound contrast. The operator should average two ONSD measurements. An average ONSD greater than 5 mm is considered abnormal with suspected elevated ICP.70,72,73
Figure 8C. ONSD for ICP
Orthopedic and Soft Tissue Injuries
Shoulder Dislocation. Shoulder (glenohumeral) dislocation is a common orthopedic injury presenting to the ED, comprising about 50% of all major joint dislocations.74 Recent literature has demonstrated the superiority of POCUS in detecting both anterior and posterior shoulder dislocations.75-77 In addition, ultrasonography can increase the accuracy and efficacy of intra-articular anesthetic injections and, thus, eliminate the need for procedural sedation during shoulder reduction.78
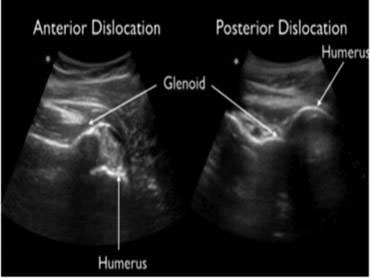
While standing behind the affected shoulder, the operator should place the curvilinear probe parallel to and just below the scapular spine, with the indicator pointing laterally, at the level of the glenoid. Adjust the depth so that both the glenoid and humeral head are seen clearly. The humeral head will appear as a circular object located just lateral to the glenoid fossa.79 (See Figure 9A.) With the more common anterior dislocation, the humeral head will be deep on the screen, while with a posterior dislocation, the humeral head will be closer to the probe and more superficial on the screen.79-81 (See Figure 9B.)
Figure 9A. Normal Shoulder
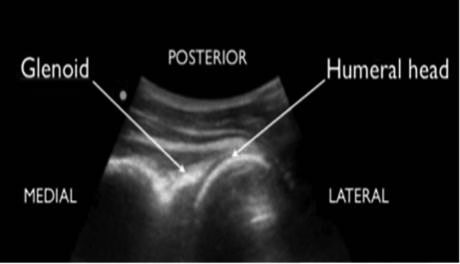
Figure 9B. Shoulder Dislocations
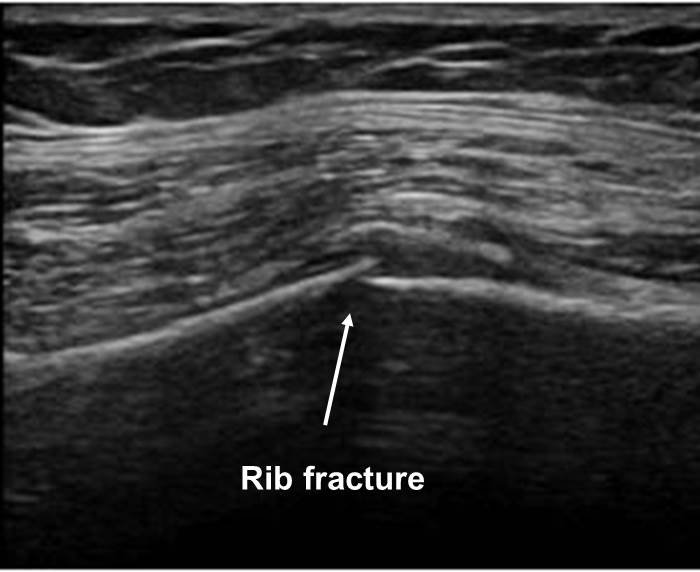
Fractures. Rib fractures can also be diagnosed using ultrasound with higher sensitivity than radiographs. Fractures of the rib, costochondral junction, and costal cartilage appear as a disruption of the anterior echogenic margin of the rib. The costal cartilage normally appears relatively hypoechoic compared with the osseous rib.50,82 Using a linear probe, a fracture is seen as a disruption of the bony contour as the rib is followed outward. (See Figure 10.) In addition, ultrasound has been used to diagnose other fractures, such as those of the clavicle, elbow, and distal forearm.83-85 Ultrasound is an especially useful tool in children and pregnant women in whom minimizing radiation exposure is paramount.
Figure 10. Fracture on Ultrasound
Foreign Body Detection. Retained foreign body from a penetrating injury is a common problem. Although radiograph is the most often used method for detection, POCUS enables the real-time localization and extraction of the foreign body. A high-frequency linear probe is most suitable for this task. All foreign bodies appear hyperechoic but will display variable degrees of artifact. Metal and glass tend to produce reverberation artifact that is caused by the sound wave bouncing back and forth between tissue and foreign body boundaries. Wood, gravel, and plastic are hyperechoic with a trailing shadow.86-87 Substances that have been present in the body longer than 24 hours typically have a small amount of surrounding inflammatory fluid, which appears as an anechoic halo surrounding the hyperechoic material.88
Nerve Blocks. Often, injured patients require anesthesia for painful procedures such as fracture reduction, incision and drainage, or complex laceration repairs. Ultrasound-guided nerve blocks can provide regional analgesia and minimize the need for large volumes of local anesthetic or procedural sedation. Nerve blocks targeting the femoral, posterior tibial, or popliteal nerves can facilitate lower extremity procedures,89-90 while those that target the interscalene, supraclavicular, median, ulnar, or radial nerves can provide anesthesia for the upper extremities.91-93 In addition, the elimination of procedural sedation by using nerve blocks can lead to decreased ED length-of-stay, shortened post-procedural observation periods, and high patient satisfaction.89,91-93
Pregnant Women
Trauma is the number one cause of pregnancy-associated maternal deaths in the United States.94 FAST is a safe, rapid method to identify intra-abdominal free fluid in pregnant trauma patients.95 In addition, ultrasound can assess gestational age, fetal viability, and fetal condition. Placental abruption after trauma occurs in 2% to 4% of minor accidents and in up to 50% of major injuries, and may occur without any external evidence of abdominal injury.96 Clinical findings that indicate abruption include vaginal bleeding, abdominal cramps, uterine tenderness, amniotic fluid leakage, maternal hypovolemia, a uterus larger than normal for the gestational age, or a change in the fetal heart rate. The transabdominal ultrasound is a first-line diagnostic method to confirm the presence of abruption, but is not as sensitive as cardiotocographic monitoring.96-98
The left lateral decubitus position of the third trimester pregnant hypotensive patient theoretically relieves the pressure of the uterus on the IVC, thereby improving venous return. However, Fields et al found that while 76% of patients showed an increased IVC size in left lateral decubitus position, the remaining patients showed the largest IVC measurement in the supine position. The utilization of POCUS to evaluate the IVC diameter during resuscitation may help to appropriately manage the positioning for injured pregnant patients.99
CONCLUSION
POCUS enables the rapid diagnosis of critical injuries and disposition/operative management of injured patients, as well as expedites and improves the outcome of bedside procedures. This review serves as a practical guide for any clinician who wants to become more proficient with this critical clinical skill.
REFERENCES
- Center for Disease Control and Prevention - National Center for Injury Prevention and Control. Leading causes of death. Available at: www.cdc.gov/injury/overview/leading_cod.html. Accessed Sept. 24, 2015.
- Woo J. A short history of the development of ultrasound in obstetrics and gynecology. Available at: www.ob-ultrasound.net/history1.html. 1998-2002. Accessed Sept. 27, 2015.
- Tiling T, Bouillon B, Schmid A. Ultrasound in blunt abdomino-thoracic trauma. In: Border J, Allgoewer M, Hansen S, eds. Blunt Multiple Trauma: Comprehensive Pathophysiology and Care. Marcel Dekker: New York,1990;415-433.
- Plummer D. Principles of emergency ultrasound and echocardiography. Ann Emerg Med 1989;18:1291-1297.
- Jehle D, Davis E, Evans T, et al. Emergency department sonography by emergency physicians. Am J Emerg Med 1989;7:605-611.
- Henwood PC, Mackenzie DC, Rempell JS, et al. A practical guide to self-sustaining point-of-care ultrasound education programs in resource-limited settings. Ann Emerg Med 2014;64:277-285.
- Hoffmann B, Rumsey H, Nixon MS. Physics and Technical Facts for the Beginner. Ultrasound Guide for Emergency Physicians. American College of Emergency Physicians, 2008. Available at: www.sonoguide.com/physics.html. Accessed Sept. 23, 2015.
- Turandot S, Del Rios Rivera M, Lewiss R. Ultrasound Image Quality. ACEP News. April 2011. Available at: http://www.acep.org/Content.aspx?id=79787. Accessed Sept. 26, 2015.
- Scalea TM, Rodriguez A, Chiu WC, et al. Focused Assessment with Sonography for Trauma (FAST): Results from an international consensus conference. J Trauma 1999;46:466-472.
- Shanmuganathan K, Mirvis SE, Sherbourne CD, et al. Hemoperitoneum as the sole indicator of abdominal visceral injuries: A potential limitation of screening abdominal US for trauma. Radiology 1999;212:423-430.
- Pariyadath M, Snead G. Emergency ultrasound in adults with abdominal and thoracic trauma. UpToDate 2015. Available at: www.uptodate.com/contents/emergency-ultrasound-in-adults-with-abdominal-and-thoracic-trauma. Accessed Sept. 22, 2015.
- Branney SW, Wolfe RE, Moore EE, et al. Quantitative sensitivity of ultrasound in detecting free intraperitoneal fluid. J Trauma 1995;39:375-380.
- American Institute of Ultrasound in Medicine, American College of Emergency Physicians. AIUM practice guideline for the performance of the focused assessment with sonography for trauma (FAST) examination. Available at: www.aium.org/resources/guidelines/fast.pdf. Accessed Sept. 23, 2015.
- Rozycki GS, Feliciano DV, Ochsner MG, et al. The role of ultrasound in patients with possible penetrating cardiac wounds: A prospective multicenter study. J Trauma 1999;46:543.
- Meyer DM, Jessen ME, Grayburn PA. Use of echocardiography to detect occult cardiac injury after penetrating thoracic trauma: A prospective study. J Trauma 1995;39:902.
- Ball CG, Williams BH, Wyrzykowski AD, et al. A caveat to the performance of pericardial ultrasound in patients with penetrating cardiac wounds. J Trauma 2009;67:1123.
- Nicol AJ, Navsaria PH, Beningfield S, et al. Screening for occult penetrating cardiac injuries. Ann Surg 2015;261:573.
- Rozycki GS, Ochsner MG, Schmidt JA, et al. A prospective study of surgeon-performed ultrasound as the primary adjuvant modality for injured patient assessment. J Trauma 1995;39:492-498.
- Gaarder C, Kroepelien F, Loekke R, et al. Ultrasound performed by radiologists — confirming the truth about FAST in trauma. J Trauma 2009;67:323–329.
- Natarajan B, Gupta P, Cemaj S, et al. FAST scan: Is it worth doing in hemodynamically stable blunt trauma patient? Surgery 2010;148:695-700.
- Nishijima DK, Simel DL, Wisner DH, et al. Does this adult patient have a blunt intra-abdominal injury? JAMA 2012;307:1517-1527.
- Kendall JL, Kestler AM, Whitaker KT, et al. Blunt abdominal trauma patients are at very low risk for intra-abdominal injury after emergency department observation. West J Emerg Med 2011;12:496-504.
- Miller MT, Pasquale MD, Bromberg WJ, et al. Not so FAST. J Trauma 2003;54:52-59.
- Quinn AC, Sinert R. What is the utility of the Focused Assessment with Sonography in Trauma (FAST) exam in penetrating torso trauma? Injury 2011;42:482.
- Udobi KF, Rodriguez A, Chiu WC, et al. Role of ultrasonography in penetrating abdominal trauma: A prospective clinical study. J Trauma 2001;50:475.
- Soffer D, McKenney MG, Cohn S, et al. A prospective evaluation of ultrasonography for the diagnosis of penetrating torso injury. J Trauma 2004;56:953.
- Biffl WL, Kaups KL, Cothren CC, et al. Management of patients with anterior abdominal stab wounds: A Western Trauma Association multicenter trial. J Trauma 2009;66:1294–1301.
- Boulanger BR, Kearney PA, Tsuei B, et al. The routine use of sonography in penetrating torso injury is beneficial. J Trauma 2001;51:320–325.
- Brooks A, Davies B, Smethhurst M, et al. Prospective evaluation of non-radiologist performed emergency abdominal ultrasound for haemoperitoneum. Emerg Med J 2004;21:e5.
- Kirkpatrick AW, Sirois M, Ball CG, et al. The hand-held ultrasound examination for penetrating abdominal trauma. Am J Surg 2004;187:660–665.
- Tayal VS, Beatty MA, Marx JA, et al. FAST (focused assessment with sonography in trauma) accurate for cardiac and intraperitoneal injury in penetrating anterior chest trauma. J Ultrasound Med 2004;23:467–472.
- Demetriades D, Hadjizacharia P, Constantinou C, et al. Selective nonoperative management of penetrating abdominal solid organ injuries. Ann Surg 2006;244:620-628.
- Berg RJ, Inaba K, Okoye O, et al. The contemporary management of penetrating splenic injury. Injury 2014;45:1394-400.
- Inaba K, Demetriades D. The nonoperative management of penetrating abdominal trauma. Adv Surg 2007;41:51-62.
- Melniker LA, Leibner E, McKenney MG, et al. Randomized controlled clinical trial of point-of-care, limited ultrasonography for trauma in the emergency department: The first sonography outcomes assessment program trial. Ann Emerg Med 2006;48:227.
- Ollerton JE, Sugrue M, Balogh Z, et al. Prospective study to evaluate the influence of FAST on trauma patient management. J Trauma 2006;60:785.
- Stengel D, Rademacher G, Ekkernkamp A, et al. Emergency ultrasound-based algorithms for diagnosing blunt abdominal trauma. Cochrane Database Syst Rev 2015;9:CD004446.
- Ball CG, Kirkpatrick AW, Laupland KB, et al. Factors related to the failure of radiographic recognition of occult posttraumatic pneumothoraces. Am J Surg 2005;189:550-556.
- Rowan KR, Kirkpatrick AW, Liu D, et al. Traumatic pneumothorax detection with thoracic US: Correlation with chest radiography and CT - initial experience. Radiology 2002;225:210.
- Kirkpatrick AW, Sirois M, Laupland KB, et al. Hand-held thoracic sonography for detecting post-traumatic pneumothoraces: The Extended Focused Assessment with Sonography for Trauma (EFAST). J Trauma 2004;57:288.
- Blaivas M, Lyon M, Duggal S. A prospective comparison of supine chest radiography and bedside ultrasound for the diagnosis of traumatic pneumothorax. Acad Emerg Med 2005;12:844.
- Soldati G, Testa A, Sher S, et al. Occult traumatic pneumothorax: Diagnostic accuracy of lung ultrasonography in the emergency department. Chest 2008;133:204.
- Wilkerson RG, Stone MB. Sensitivity of bedside ultrasound and supine anteroposterior chest radiographs for the identification of pneumothorax after blunt trauma. Acad Emerg Med 2010;17:11.
- Ku BS, Fields JM, Carr B, et al. Clinician-performed bedside ultrasound for the diagnosis of traumatic pneumothorax. West J Emerg Med 2013;14:103.
- Zhang M, Liu ZH, Yang JX, et al. Rapid detection of pneumothorax by ultrasonography in patients with multiple trauma. Crit Care 2006;10:R112.
- Dulchavsky SA, Schwarz KL, Kirkpatrick AW, et al. Prospective evaluation of thoracic ultrasound in the detection of pneumothorax. J Trauma 2001;50:201-205.
- Lichtenstein DA. Ultrasound in the management of thoracic disease. Crit Care Med 2007;35(5 Suppl):S250-S261.
- Lichtenstein D, Mezière G, Biderman P, et al. The “lung point”: An ultrasound sign specific to pneumothorax. Intensive Care Med 2000;26:1434-1440.
- Lichtenstein DA. General Ultrasound in the Critically Ill. 2005, Berlin; New York: Springer. ix, p199.
- Jain A, Menaker J. Ultrasound for trauma. Trauma Rep 2013;14:3.
- Lichtenstein D, Lascols N, Prin S, et al. The lung pulse: An early ultrasound sign of complete atelectasis. Intensive Care Med 2003;4:2187–2192.
- Randolph AG, Cook DJ, Gonzales CA, et al. Ultrasound guidance for placement of central venous catheters: A meta-analysis of the literature. Crit Care Med 1996;24:2053-2058.
- Gregg SC, Murthi SB, Sisley AC. Ultrasound-guided peripheral intravenous access in the intensive care unit. J Crit Care 2010;25:514-519.
- Shokoohi H, Boniface K, McCarthy M, et al. Ultrasound-guided peripheral intravenous access program is associated with a marked reduction in central venous catheter use in non-critically ill emergency department patients. Ann Emerg Med 2013;61:198-203.
- Constantino TG, Parikh AK, Satz WA, et al. Ultrasonography-guided peripheral intravenous access versus traditional approaches in patients with difficult intravenous access. Ann Emerg Med 2005;46:456-461.
- Dargin JM, Rebholz CM, Lowenstein RA, et al. Ultrasonography-guided peripheral intravenous catheter survival in ED patients with difficult access. Am J Emerg Med 2010;28:1-7.
- Adhikari S, Blaivas M, Morrison D. Comparison of infection rates among ultrasound-guided versus traditionally placed peripheral intravenous lines. J Ultrasound Med 2010;29:741-747.
- Glen H, Lang I, Christie L. Infraclavicular axillary vein cannulation using ultrasound in a mechanically ventilated general intensive care population. Intensive Care 2015;43:635-640.
- Maecken T, Heite L, Wolf B, et al. Ultrasound-guided catheterisation of the subclavian vein: Freehand vs needle-guided technique. Anaesthesia 2015 Aug 28.
- Lalu MM, Fayad A, Ahmed O, et al. Ultrasound-guided subclavian vein catheterization: A systematic review and meta-analysis. Crit Care Med 2015;43:1498-1507.
- Feissel M, Michard F, Faller JP, et al. The respiratory variation in inferior vena cava diameter as a guide to fluid therapy. Intensive Care Med 2004;30:1834–1837.
- Lyons M, Blaivas M, Brannam L. Sonographic measurement of the inferior vena cava as a marker of blood loss. Am J Emerg Med 2005;23:45–50.
- Moreno FL, Hagan AD, Holmen JR, et al. Evaluation of size and dynamics of the inferior vena cava as an index of right-sided cardiac function. Am J Cardiol 1984;53:579–585.
- Nagdev AD, Merchant RC, Tirado-Gonzalez A, et al. Emergency department bedside ultrasonographic measurement of the caval index for noninvasive determination of low central venous pressure. Ann Emerg Med 2010;55:290–295.
- Stawicki SP, Braslow BM, Panebianco NL. Intensivist use of hand-carried ultrasonography to measure IVC collapsibility in estimating intravascular volume status: Correlations with CVP. J Am Coll Surg 2009;209:55–61.
- Dewitz A. Soft tissue applications. In: Ma OJ, Mateer J, eds. Emergency Ultrasound. McGraw-Hill; New York; 2003:385.
- Whitcomb MB. How to diagnose ocular abnormalities with ultrasound. AAEP Proceedings 2002;48:272-275.
- Price D, Simon BC, Park RS. Evolution of emergency ultrasound. California J Emerg Med 2003;4:82-88.
- Cotton, J. Keeping an eye on intracranial pressure: Detecting elevated ICP using ocular ultrasound. SonoMojo, Sept. 14, 2014. Available at: http://sonomojo.org/2014/09/07/keeping-an-eye-on-intracranial-pressure-detecting-elevated-icp-using-ocular-ultrasound/. Accessed Sept. 27, 2015.
- Adhikari SR. Ocular Ultrasound. Ultrasound Guide for Emergency Physicians. American College of Emergency Physicians; 2008. Available at: www.sonoguide.com/smparts_ocular.html. Accessed Sept. 23, 2015.
- Shinar Z, Chan L, Orlinsky M. Use of ocular ultrasound for the evaluation of retinal detachment. J Emerg Med 2011;40:53–57.
- Howard Z, Gharahbaghian L. Emergency ultrasound tips and tricks — ocular ultrasound. Emergency Ultrasound Section Newsletter, April 2012. Available at: www.acep.org/Content.aspx?id=84879. Accessed Sept. 27, 2015.
- Blaivas M, Theodoro D, Sierzenski PR. Elevated intracranial pressure detected by bedside emergency ultrasonography of the optic nerve sheath. Acad Emerg Med 2003;10:376-381.
- Wolfson AB, Hendey GW, Ling LJ, et al. Harwood-Nuss’ Clinical Practice of Emergency Medicine. Lippincott Williams & Wilkins; 2012.
- Abbasi S, Molaie H, Hafezimoghadam P, et al. Diagnostic accuracy of ultrasonographic examination in the management of shoulder dislocation in the emergency department. Ann Emerg Med 2013:1-6.
- Blakeley CJ, Spencer O, Newman-Saunders T, et al. A novel use of portable ultrasound in the management of shoulder dislocation. Emerg Med J 2009;26:662-663.
- Beck S, Chilstrom M. Point-of-care ultrasound diagnosis and treatment of posterior shoulder dislocation. Am J Emerg Med 2013;31:449.e3-449.e5.
- Hunter, B, Wilbur, L MD. Can Intra-articular lidocaine supplant the need for procedural sedation for reduction of acute anterior shoulder dislocation? Ann Emerg Med 2012;59:513-514.
- Chilstrom M. Lecture presented at the Essentials of Emergency Medicine Conference, San Francisco, CA. Nov. 3, 2013.
- Dschaak T. Shoulder Ultrasound: Intra-articular injection and reduction. EMDocs, June 5, 2014. Available at: www.emdocs.net/shoulder-ultrasound-reduction-intra-articular-injection/. Accessed Sept. 27, 2015.
- Riguzzi C, Mantuani D, Nagdev A. How to use point-of-care ultrasound to identify shoulder dislocation. ACEP Now, Feb. 14, 2014. Aavailable at: http://www.acepnow.com/article/use-point-care-ultrasound-identify-shoulder-dislocation/. Accessed Sept. 25, 2015.
- Turk F, Kurt AB, Saglam S. Evaluation by ultrasound of traumatic rib fractures missed by radiography. Emerg Radiol 2010;17:473-477.
- Rabiner JE, Khine H, Avner JR, et al. Accuracy of point-of-care ultrasonography for diagnosis of elbow fractures in children. Ann Emerg Med 2012:196-0644.
- Chien M, Bulloch B, Garcia-Filion P, et al. Bedside ultrasound in the diagnosis of pediatric clavicle fractures. Pediatr Emerg Care 2011;27:1038-1041.
- Chaar-Alvarez FM, Warkentine F, Cross K, et al. Bedside ultrasound diagnosis of nonangulated distal forearm fractures in the pediatric emergency department. Pediatr Emerg Care 2011;27:1027-1032.
- Porter MD, Schriver JP. Ultrasound-guided Kopans’ needle location and removal of a retained foreign body. Surg Endosc 2000;14:500.
- Sidharthan S, Mbako AN. Pitfalls in diagnosis and problems in extraction of retained wooden foreign bodies in the foot. Foot Ankle Surg 2010;16:e18-20.
- American college of Emergency Physicians. Emergency ultrasound imaging criteria compendium. Available at: www.acep.org/workarea/DownloadAsset.aspx?id=80127. Accessed Sept. 23, 2015.
- Beaudoin FL, Nagdev A, Merchant RC, et al. Ultrasound-guided femoral nerve blocks in elderly patients with hip fractures. Am J Emerg Med 2010;28:76-81.
- Bunting LV. Ultrasound guided nerve blocks. Ultrasound Guide for Emergency Physics. American College of Emergency Physicians, 2008. Available at: www.sonoguide.com/nerve_block.html. Accessed on: Sept. 25, 2015.
- Liebmann O, Price D, Mills C, et al. Feasibility of forearm ultrasound-guided nerve blocks of the radial, ulnar and median nerves for hand procedures in the emergency department. Ann Emerg Med 2006;48:558-562.
- Stone MB, Wang R, Price DD. Ultrasound-guided supraclavicular brachial plexus nerve block vs procedural sedation for the treatment of upper extremity emergencies. Am J Emerg Med 2008;26:706-710.
- Blaivas M, Lyon M. Ultrasound-guided interscalene block for shoulder dislocation reduction in the ED. Am J Emerg Med 2006;24:293-296.
- Chang J, Berg C, Saltzman L, et al. A leading cause of injury deaths among pregnant and postpartum women in the United States, 1991-1999. Am J Public Health 2005;95:471.
- Goodwin H, Holmes JF, Wisner DH. Abdominal ultrasound examination in pregnant blunt trauma patients. J Trauma 2001;50:689-693.
- Kuhlman RS, Cruikshank DP. Maternal trauma during pregnancy. Clin Obstet Gynecol 1994;37: 274-293.
- Pearlman MD, Tintinalli JE, Lorenz RP. Blunt trauma during pregnancy. N Eng J Med 1990;323:1609-1613.
- Desjardins G. Management of the injured pregnant patient. Trauma.org. Available at: www.trauma.org/archive/resus/pregnancytrauma.html. Accessed Sept. 24, 2015.
- Fields JM, Catallo K, Au AK. Resuscitation of the pregnant patient: what is the effect of patient positioning on inferior vena cava diameter? Resuscitation 2013;84:304-308.
Point-of-care ultrasound is a critical clinical tool that facilitates the early diagnosis of many life-threatening injuries. As with any test, clinicians need to fully appreciate indications and limitations of the diagnostic tool and integrate where advantageous to their practice.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
