Severe Asthma Exacerbation in Pediatric Patients
Authors
Aaron N. Leetch, MD, Assistant Professor of Emergency Medicine & Pediatrics, Associate Residency Director, EM and EM/Pediatrics Programs, University of Arizona/Banner University Medical Center – Tucson Campus
Bryan Wilson, MD, Resident, Emergency Medicine/Pediatric Combined Residency, University of Arizona/Banner University Medical Center – Tucson Campus
Peer Reviewer
Taryn Taylor, MD, FAAP, FACEP, Assistant Professor of Pediatrics and Emergency Medicine, Emory University School of Medicine, Atlanta, GA
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Dietrich (editor), Dr. Skrainka (CME question reviewer), Ms. Wurster (nurse planner), Dr. Leetch (author), Dr. Wilson (author), Dr. Taylor (peer reviewer), Ms. Coplin (executive editor) and Ms. Mark (executive editor) report no relationships with companies related to the field of study covered by this CME activity.
Executive Summary
- The signs and symptoms of respiratory distress and impending respiratory failure can be subtle in the asthmatics, especially those with longstanding asthma who have become “poor perceivers” of the severity of their disease.
- Likewise, soft wheezing or a “silent chest” should be more concerning than loud wheezes, as it indicates a higher degree of obstruction.
- Children with concomitant food allergies and poor compliance with controller medications appear to have the highest rate of fatal asthma.
- Treatment for status asthmaticus includes first-line therapies, which include inhaled beta-agonists, inhaled ipratropium, and systemic corticosteroids. This is followed by second-line therapy of magnesium sulfate and third-line therapy with epinephrine, aminophylline, and ketamine. Third-line therapy can also include helium-oxygen mixtures and noninvasive ventilator support.
Acute asthma exacerbation is a common reason for emergency department (ED) visits during childhood. Most emergency physicians are comfortable managing a mild exacerbation or admitting/transferring a child who requires more than a few breathing treatments for further management. While inhaled beta-agonists and steroids will often easily and effectively manage most exacerbations, a small subset of children present in extremis requiring second- and third-line medications and airway management. These next steps in management are often less familiar to emergency physicians practicing outside of a pediatric ED. Failure to recognize or failure to reassess patients in status asthmaticus can lead to rapid deterioration and a more complicated, sicker child. Despite the best efforts, some patients will require aggressive airway support, including noninvasive ventilation and intubation. The decision to intubate an asthmatic is a difficult one and one that most emergency physicians hope they will not to have to make. The key to management of the critical asthmatic is early recognition, a strategy of multiple medical therapies, and noninvasive ventilation where appropriate. The goal of this article is to describe the initial assessment of children with severe asthma exacerbation and provide an outline and suggested hierarchy of medications and treatment strategies for stabilization, with intubation as a last resort.
— Ann M. Dietrich, MD, FAAP
Epidemiology
Asthma is a chronic disease of the airways characterized by recurrent episodes of inflammation and bronchospasm characterized by wheezing, coughing, shortness of breath, and chest tightness. Affecting approximately 1 in 10 children, asthma is estimated to cost $3300 per asthmatic per year in medical expenses, and costs the average asthmatic child 4 days of missed school per year.1 Asthma attacks are frequent, with more than half of children with asthma reporting an attack and 2.1 million ED visits for asthma exacerbations across all age groups in a 12-month period.1,2
Although only 3 per 100,000 asthmatics require hospitalization, there were 479,300 hospitalizations and 185 deaths across all age groups due to asthma reported in a 12-month period.1,2 The relative rarity of the critically ill asthmatic poses a challenge for provider comfort, research, and the establishment of standard practices. Two large multicenter retrospective studies of asthma exacerbations in pediatric intensive care units found a 1- to 2-day median length of stay and extremely variable practice patterns.3,4 Fortunately, death was rare even in this critically ill cohort. Of particular note, half of these critically ill patients initially presented to an outside ED, highlighting the importance of all emergency providers being capable of caring for the acutely ill asthmatic.4
Recognition
As with most pediatric emergencies, recognition is of the utmost importance. The signs and symptoms of respiratory distress and impending respiratory failure can be subtle in the asthmatic, especially those with longstanding asthma who have become “poor perceivers” of the severity of their disease.5 The Pediatric Assessment Triangle is a bedside tool that helps providers rapidly identify children with respiratory distress and failure.6 Many authors have attempted to define asthma severity scores that will reliably predict the need for admission for intensive therapy,7-9 but none have been externally validated with good inter-observer reliability.10-12 However, all of these scores focus on a combination of abnormal vital signs, pulse oximetry, and careful attention to the appearance, work of breathing, and breath sounds. (See Table 1.) Pediatric patients have an amazing ability to maintain increased respiratory effort for a prolonged period of time when compared to adults. However, fatigue, hypoxia, and hypercarbia often can be first noticed by the patient’s appearance. This is best identified by observing the patient's level of alertness, the appropriateness of the patient’s interaction with medical staff and a strange environment, and the patient’s response to stimuli such as needle sticks or the placement of a nebulizer on the face. The most critical patients will demonstrate a dulled or limited response. The work of breathing is seen as a function of the patient’s respiratory effort and the appearance of retractions. Accessory muscle use is limited in younger children, so signs such as head bobbing and grunting should be seen as significant effort. Retractions are seen with many obstructive pulmonary pathologies, but critical asthma often can cause intercostal, scalene, and even sternal retractions. Chest auscultation should focus on timing of wheeze and the amount of air movement. Wheezing usually occurs during the expiratory phase but often can become biphasic or inspiratory-only in the case of a severe exacerbation. Likewise, soft wheezing or a “silent chest” should be more concerning than loud wheezes, as it indicates a higher degree of obstruction. Asking an older patient to speak or recite the alphabet can help distinguish those with enough reserve to speak in sentences from those who are ill enough to speak only in single words. Peak flow measurements can be very helpful in patients who know their baseline values and can cooperate with the exam.
Table 1. Rapid Assessment of Pediatric Asthma Patients |
|
|
Appearance |
Level of alertness Interaction Response to stimuli |
|
Work of Breathing |
Respiratory rate Respiratory effort Retractions |
|
Breath Sounds |
Phase of wheezing Degree of air movement |
Concurrent with this brief initial assessment, a focused history should be obtained from the patient or family regarding high-risk factors for severe asthma or sudden death. (See Table 2.) Time is a precious commodity for critical asthma, so a history should be obtained in a hierarchal manner, with the most concerning risk factors (such as history of intubation, history of anaphylaxis, or previous ICU stay) being asked first. Children with concomitant food allergies and poor compliance with controller medications appear to have the highest rate of fatal asthma.13 Poor compliance with controller medications often leads to frequent albuterol use, which can down-regulate receptors and lead to a weaker response to rescue therapy in the ED.14 Non-white children and those who live in inner cities and have poor access to health care have a disproportionately high incidence of severe asthma and should be monitored carefully.15
Table 2. Historical Risk Factors for Severe Asthma and Sudden Death |
|
Previous intubation |
|
Previous ICU admission |
|
Frequent albuterol use |
|
Anaphylaxis |
|
Multiple controller medications |
|
Early age of diagnosis (< 5 years) |
|
Adapted from: Mitchell I, Tough SC, Semple LK, et al. Near-fatal asthma: A population-based study of risk factors. Chest 2008:121:1407-1413. |
Treatment Strategy
To effectively manage critical asthma, providers should have a treatment plan with multiple backup plans when successive medications fail. Table 3 shows a suggested plan for treating status asthmaticus. First-line therapies include inhaled beta-agonists, inhaled ipratropium, and systemic corticosteroids.16-20 This is followed by second-line therapy of magnesium sulfate and third-line therapy with epinephrine, aminophylline, and ketamine. Third-line therapy includes helium-oxygen mixtures or bi-level positive pressure assistance.
Table 3. Suggested Treatment Plan for Status Asthmaticus |
|
|
RECOGNIZE |
|
|
First-line Therapy |
Bronchodilators
Steroids
or
or
|
|
REASSESS |
|
|
Second-line Therapy |
|
|
REASSESS |
|
|
Third-line Therapy |
|
|
Intubation |
After failure of third-line therapy or at any time if patient clinically deteriorates |
Immediate Intubation
Initiation of medical therapy should include an assessment of whether the patient requires immediate intubation. Unfortunately, research-based recommendations are lacking, leaving current recommendations to be opinion-based. Clinical signs that strongly suggest the need for intubation include a decreasing mental status, cyanosis, respiratory fatigue, impending respiratory arrest, and cardiopulmonary arrest.18 Objective measures of elevated CO2 (pCO2 > 50 mmHg) or hypoxemia (pO2 < 60 mmHg) are also indicative of a need for advanced respiratory support.18 Essentially, the decision to intubate immediately relies on the presence of respiratory failure or hypoxia so severe that it requires aggressive and immediate reversal to avert cardiopulmonary arrest.
Tied to the decision of intubation is making a prediction of whether the patient is likely to respond to medical therapy. The patient’s ability to compensate can decline over time if significant respiratory effort continues. It is worth noting that although questions regarding frequency of symptoms, number of previous hospitalizations, number of ICU admissions, and number of previous intubations are mainstays of the asthma exacerbation history, 13% of patients admitted to the pediatric intensive care unit are newly diagnosed and 63% present with no hospitalization in the previous year.4 Additionally, frequent use of beta-agonists has been demonstrated to result in downregulation, so a history of such is concerning for a decreased response to medical therapy.14 A need for aggressive therapy in the ED and prior to arrival has been shown to be predictive of a continuing need for intensive care during the admission.18,21 Initial presentation and SpO2 have been found to be less predictive than the patient’s response at 30 minutes.22,23 Specifically, patients who at 30 minutes demonstrate a peak expiratory flow rate (PEFR) < 40% of predicted or personal best or < 60% improvement in PEFR are predicted to respond poorly to medical treatment and require intensive care unit admission.23 A reasonable strategy in the patient who does not require immediate intubation would be a 30-minute trial of aggressive medical therapy to assess the patient’s anticipated response, followed by a reassessment, particularly in a patient requiring transfer to another facility.
First-Line Therapy
Inhaled albuterol or salbutamol remains the initial treatment of choice for asthma exacerbations due to its short onset of action (approximately 5 minutes with a peak of action at 1 hour, peak duration of 2 hours, and total duration of 4-6 hours). Pre- and post-treatment assessments are helpful in guiding the frequency of dosing. In patients requiring maximum dosages of albuterol, continuous administration via nebulizer has been demonstrated effective and safe with only transient elevations in serum troponin and lactic acid.24 While nebulized solutions may be needed in critically ill patients unable to cooperate with a nebulizer, equal efficacy has been demonstrated with the proper use of a nebulizer or inhaler with a spacer.25-27 The R-enantiomer of albuterol, levalbuterol, is available on the market, but current studies have failed to demonstrate a notable benefit.28 Typical starting regimens for albuterol are either 2.5-5 mg nebulized or 4 to 8 puffs from a metered dose inhaler every 20 minutes for 1 hour.29 Patients who fail to respond or only minimally respond to the first hour of therapy may benefit from increasing doses of continuous albuterol or salbutamol.
Inhaled ipratropium acts as a useful adjunct to albuterol by contributing to further bronchodilation. Its longer onset of action (approximately 15 minutes with a duration of 4 hours), as well as clinical trials, demonstrate that ipratropium alone is inadequate in treating acute exacerbations.30 The use of ipratropium as an adjunct on initial presentation is well validated, but there remains a question of its efficacy beyond initial presentation.31,32,33 Of note, downregulation does not occur with frequent use of ipratropium as it does with beta-agonists. Typical starting regimens for ipratropium are 250-500 mcg nebulized 1-3 times given with the initial doses of albuterol.29
Systemic steroids complement the action of bronchodilators, not only by decreasing airway inflammation but also by acting synergistically with beta-agonists by upregulating receptors responsible for bronchodilation.22 They have a longer onset of action when compared to bronchodilators but also have a duration of action on the order of days, which is helpful in avoiding relapse. The duration of systemic steroid therapy varies by formulation and severity of the exacerbation. A 5-day course of prednisone for a patient discharged from the ED is typical, although research demonstrates 3 days of prednisone or even a single-dose dexamethasone to be equivalently efficacious, with few side effects and higher compliance rates.34-37 Steroids administered via oral (PO), intravenous (IV), or intramuscular (IM) routes are similarly efficacious and guided largely by what access is available.38-40 However, most patients with critical asthma are at risk for aspiration due to their work of breathing; thus, the IV or IM route is often preferred for these cases. There is evidence suggesting a possible benefit to the supplementation of systemic steroids with inhaled steroids.41 Evidence for the use of inhaled steroids alone is lacking.42,43 Typical doses of corticosteroids are:29
- Prednisone or prednisolone 1-2 mg/kg (max 60 mg) PO/IV;
- Methylprednisolone 1-2 mg/kg (max 80-100 mg) PO/IM/IV;
- Dexamethasone 0.6-1.0 mg/kg (max 16 mg) PO/IM/IV.
First-line therapy of moderate and severe asthma exacerbations is rounded out by the use of supplemental oxygen for hypoxia and the judicious use of IV fluids.16,17 Because of their marked tachypnea, these patients are at risk for significant fluid losses and dehydration, but this must be balanced against the risk of pulmonary edema, which would further exacerbate their respiratory distress.
Second-Line Therapy
Inhaled beta-agonists remain the mainstay of rescue medication in status asthmaticus, so patients who have not improved after the first round of therapy should have albuterol or salbutamol continuously nebulized at increasingly higher doses (max 20 mg/hour). Magnesium sulfate has become established as the second-line therapy for severe asthma exacerbations that have failed or are unlikely to adequately respond to bronchodilators and systemic steroids. It acts as a smooth-muscle relaxant and thus a bronchodilator, but in a different manner than first-line therapies. When given intravenously, it has been shown to reduce admission rates and increase pulmonary function.44-46 There has been some research into the use of nebulized magnesium but the current evidence is inconclusive.46,47 The greatest efficacy for magnesium has been demonstrated in severe exacerbations. Although the evidence supporting magnesium is not as robust as that of the bronchodilators and systemic steroids, magnesium's demonstrated benefit and safety make it a standard adjunct in the care of the moderate to severe asthma exacerbation. Typical dosing for magnesium sulfate is a single dose of 25-75 mg/kg (2 g maximum dosage) IV over 20 minutes.29
Third-line Therapy
A number of additional pharmacologic interventions have been investigated, though their role in the severe asthmatic has yet to be robustly defined, especially in the pediatric population. Consultation with a pediatric intensivist or someone familiar with these medications may be of value.
Epinephrine (an alpha- and beta-agonist) and terbutaline (a beta-agonist) are potential adjuncts or alternatives to albuterol. Terbutaline may be most useful in patients who have bronchospasm so severe that inhaled therapy cannot reach the target receptors; however, the added vasoconstriction effects of epinephrine can help decrease airway edema and may be a more favorable choice. Nebulized racemic epinephrine, in particular, has been demonstrated to be comparable to albuterol for acute asthma, though with a higher side effect profile and without a proven safety profile when multiple doses are administered.4,48,49 Although these medications may be nebulized as an adjunctive therapy, a more likely role for these medications is IV or IM administration in the patient whose exacerbation is so severe that pulmonary delivery of nebulized medication is compromised, though their efficacy is in question.50,51 Typical dosing for racemic epinephrine is 0.5 mL in 3-5 mL of normal saline nebulized.29 Typical dosing for epinephrine is 0.01 mg/kg (max 0.5 mg) IM every 5-15 minutes.29 Typical dosing for terbutaline is 2-10 mcg/kg for a loading dose followed by an infusion of 0.1-0.4 mcg/kg/min.29
Ketamine is a dissociative anesthetic frequently given in the ED setting for procedural sedation or induction for intubation. When given at dissociative doses, it has been demonstrated to improve respiratory distress through bronchodilation and prevent intubation.52-55 It is a well-accepted practice in adults, but pediatric literature is currently limited to case reports and small cohort studies.56 Ketamine is probably best used as a last attempt to avert intubation after all preparations for intubation have been made. While there is some debate, repeat boluses can be given, as the added benefit of a continuous infusion is questionable, and acquiring a ketamine infusion in a timely manner is often not possible in the ED.52,57 Typical dissociative dosing is 0.5-1 mg/kg IV (slowly over 60 seconds to minimize the risk of laryngospasm or respiratory depression) or 2-4 mg/kg IM.52 Given the faster onset and need for IV access in preparation of intubation, IV dosing is recommended.
The methylxanthines theophylline and aminophylline act as bronchodilators through competitive nonselective phosphodiesterase inhibition. Multiple studies and reviews have failed to demonstrate an improvement in outcomes.58-62 A single study reports increased length of stay and duration of symptoms.63 Given their poorly demonstrated efficacy and possibly worsened outcomes, there is likely an extremely limited role for these medications.
Montelukast, a leukotriene receptor antagonist, improves airway inflammation and is frequently used as an adjunct controller medication in asthmatics. A Cochrane review found no role for oral leukotriene in acute exacerbations and inadequate evidence to assess IV administration.64 Its role in acute asthma is likely extremely limited.
Nebulized furosemide has been demonstrated by limited evidence to improve pulmonary function in mild-to-moderate disease without significant adverse events.65 Studies specific to pediatrics and studies assessing outcomes such as admission, intubation, and mortality are lacking, but there is sufficient evidence to consider furosemide a reasonable adjunct. Typical study doses were 1 mg/kg of nebulized furosemide.
Respiratory Support
In the severe asthmatic who continues to decline or fails to improve, helium-oxygen (heliox) and noninvasive positive pressure ventilation (NIPPV) may help avoid intubation. Heliox is a mixture of helium and oxygen that is able to deliver higher FiO2 at lower gas densities. The benefit of this is a decreased respiratory effort when compared to standard air-O2 mixtures and subsequent improvement in respiratory fatigue. Current evidence supports the use of heliox but is limited to small case reports or case studies in the pediatric population.66-68
NIPPV similarly works to decrease respiratory effort. By providing positive airway pressure and pressure support for respiration, it works to decrease respiratory fatigue and increase the delivery of inhaled medications and oxygen. NIPPV is highly effective in improving oxygenation by maintaining airway pressures and improving ventilation by decreasing the work associated with breathing in a tired asthmatic. The most basic form of NIPPV is a high flow nasal cannula (HFNC) that supplies > 2 L per minute of humidified oxygen. Increasingly greater flow rates will provide increasing levels of positive airway pressure, though it is most effective on infants, and even then, variable from patient to patient. To provide a more consistent level of airway pressure, a nasal mask or a full face mask can be applied to create a tighter seal. With these devices, continuous positive airway pressure (CPAP) or bilevel positive airway pressure (BiPAP) can be delivered depending on the machine available. BiPAP allows respiratory support during both inspiration and expiration utilizing a high and low pressure vs CPAP, which delivers one pressure through the entire respiratory cycle. One study showed only 7% of pediatric patients on noninvasive positive pressure ventilation went on to require intubation.69 This therapy is generally well tolerated in patients who are alert and able to protect their airway, though children may need low dose sedation and continuous monitoring.
Intubation
The decision to intubate a pediatric patient should be made after all other options have been exhausted. Only a minority of patients with status asthmaticus require intubation, though nearly 8% of these patients will die on the ventilator.70,71 Those patients who cannot maintain their work of breathing, as evidenced by altered mentation, decreasing respiratory effort, and poor perfusion, must be intubated according to a provider’s best judgment. To date, no literature exists that demonstrates a laboratory value that predicts the need for intubation apart from clinical gestalt; however, blood gas values, such as hypercarbia and hypoxemia, can correlate to somnolence and air hunger, respectively.
The most experienced intubator should attempt intubation, as these patients are likely to rapidly desaturate. A cuffed endotracheal tube should be used if available to allow for adequate ventilator pressures. Preparation for intubation should include multiple backup plans in the event that rapid sequence induction fails. Patient should be pre-oxygenated with a non-rebreather or, if tolerated, positive pressure ventilation. Applying an HFNC at 10-15 L while attempting intubation is known as apneic oxygenation and may add valuable seconds to the intubation process.72 Ketamine has become the induction agent of choice when intubating patients in status asthmaticus due to its complementary bronchodilatory effects, minimal effect on hemodynamics, and ability to maintain spontaneous respirations.73,74 Induction should be with 1-2 mg/kg given over about 5 seconds.75
Once the patient has been successfully intubated, there will be a careful balance between providing enough pressure to oxygenate the patient but not so much as to cause barotrauma or prevent venous return and impair cardiac output. It is acceptable to optimize oxygenation and pulmonary pressures at the expense of hypercapnea.76-78 Mechanical ventilation modes that allow for pressure-regulated support and long expiratory times should be used to adequately balance these needs. Initial ventilator settings should allow for maximum pressures of 36-40 cm H2O, a respiratory rate of 12-14 breaths per minute, and an inspiratory-to-expiratory time ratio of 1:4-1:5.78 Peri- and post-intubation is a high-risk time for cardiac arrest. Deterioration of blood pressure or oxygenation should prompt an immediate assessment for sources utilizing the DOPE mnemonic (D – dislodgment, O – obstruction, P – pneumothorax, E – equipment malfunction).79 The patient should be taken off of the ventilator and manually bagged on 100% oxygen. The tube should be suctioned and its placement verified. Needle or tube thoracotomy should be performed for suspected pneumothorax if critical. Hyperinflation can cause severe obstructive shock and may be worsened acutely after intubation and positive pressure ventilation. Compression of the chest immediately after intubation may help deflate the lungs somewhat to reduce this pressure. In patients who continue to have poor oxygenation and/or hemodynamics despite aggressive ventilator strategies, aggressive airway clearance with bronchoscopy, inhaled volatile anesthetics, or even extracorporeal membrane oxygenation may be indicated.80-82
Pitfalls in Critical Asthma
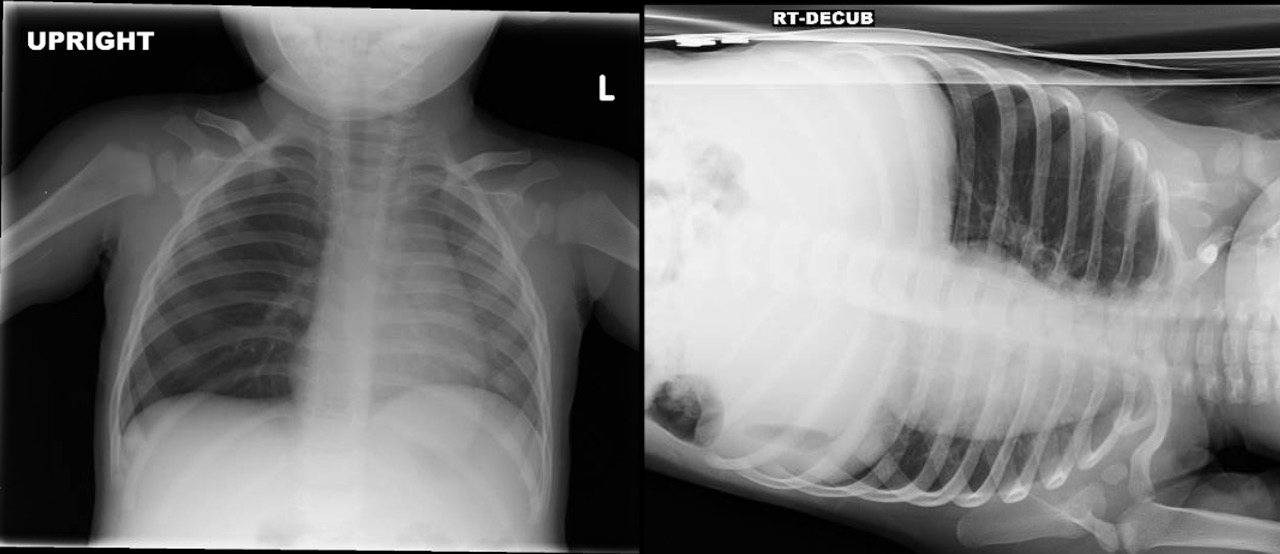
Patients with status asthmaticus often do not respond to initial therapy due to the severity of their disease. However, there are other conditions that may mimic asthma or complicate asthma that may prevent an appropriate response to typical therapy. (See Table 4.) Most laboratory studies are not helpful during the ED phase of a patient’s exacerbation, though a baseline potassium level may be helpful to the intensive care team, who will continue to monitor while the patient is on continuous albuterol. A chest X-ray should be obtained in most any severe asthma exacerbation. (See Figure 1.) Asthma patients with pneumonia or influenza may have a longer duration of symptoms and will be more difficult to treat.83 Such patients should be screened for pneumonia and influenza and may benefit from blood cultures if febrile. They also will require antibiotics and treatment according to sepsis guidelines. A chest X-ray may reveal evidence of a pneumothorax, which may require tube thoracostomy. Younger pediatric patients can also be prone to foreign body aspiration, sometimes without a witnessed event. Persistent cough, focal wheezing, or a recurrent focal pneumonia may be clues to the presence of a foreign body. The degree of wheezing is often dependent on the size of the foreign body, with larger foreign bodies creating a “ball-valve” effect and causing air trapping. (See Figure 2.) A chest X-ray with a left and right lateral decubitus view can sometimes reveal hyperinflation on the affected side due to air trapping, though this is not a sensitive test.84 The foreign body can sometimes be identified on chest X-ray, though the majority are food products and other radiolucent items.
Table 4. Asthma Comorbidities |
||
|
Infectious |
Traumatic |
Allergic |
|
|
|
Figure 1. Chest X-ray with Hyperinflation |
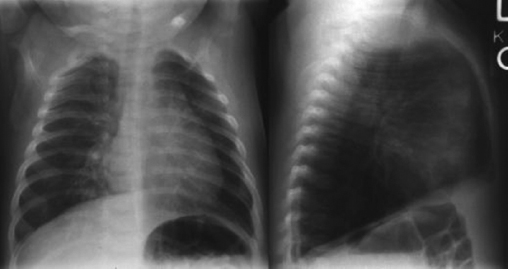 |
Figure 2. Chest X-ray with Bronchial Foreign Body |
 |
The “atopic triad” includes asthma, eczema, and allergies. Patients with asthma also may have significant allergies that manifest with anaphylaxis. A presentation of wheezing and respiratory distress may appear to be status asthmaticus at first glance, but will not respond to typical treatment due to a different pathophysiology. Anaphylaxis can be diagnosed by exposure to a likely allergen with involvement of two or more systems (gastrointestinal, skin, respiratory, or cardiovascular).85 Thus, patients with concomitant rash, hypotension, edema, vomiting, or diarrhea may benefit from a dose of epinephrine for both critical asthma and possible anaphylaxis. Also, if there is any concern the child has had skin exposure to an anaphylactic trigger the skin should be washed to terminate the exposure.
Disposition
Final disposition is a clinical decision. Patients requiring albuterol every 4 hours and breathing comfortably on room air can generally be discharged home. Patients requiring intubation or continuous bronchodilator nebulization require admission to the intensive care unit. Between these two extremes is a broad spectrum in which the disposition to the floor or intensive care unit relies on clinical judgement and local practice patterns without evidence-based decision rules. The following are some indicators that suggest admission to an intensive care unit:18,21
- Previous severe exacerbation;
- Two or more hospitalizations in the past year;
- Three or more ED visits in the past year;
- Hospitalization or ED visit in the past month;
- Use of more than two canisters of short-acting beta-agonist in the past month;
- Difficulty perceiving or articulating asthma symptoms;
- Persistence of PEFR < 40% predicted.
Patients requiring transfer to another facility warrant special consideration. The potential for decompensation en route and the limited resources available during transport, in comparison to what is available at the originating facility, must be carefully considered. Some specific considerations include:
- Avoid weaning albuterol to a lower frequency or dose than what the patient has proven to be able to tolerate.
- A dose of ipratropium prior to transport provides several hours of adjunctive bronchodilation.
- Early administration of steroids allows maximal effect during transport.
- Liberal administration of magnesium is preferable to decompensation en route.
- Carefully consider whether the patient needs pre-emptive intubation based on clinical status, improvement to current therapies, and the duration of transport.
Summary
Status asthmaticus can be frightening for patients and providers. Early recognition of severe asthma from the patients’ appearance, work of breathing, and breath sounds will lead to prompt response to their illness. A planned approach with multiple tiers of medications and clear endpoints can help direct the management of these patients. Prompt administration of first-line medications, including inhaled beta-agonists and anticholinergics with systemic steroids, may turn patients around quickly. Second-line therapy with magnesium sulfate can be added quickly to non-responders, in addition to continuous beta-agonist nebulization. Third-line agents can be started to treat refractory status asthmaticus, and consultation with a pediatric intensivist may be helpful at this time. Patients in severe respiratory distress who can tolerate a mask may benefit from noninvasive positive pressure ventilation. The decision to intubate an asthmatic must be based on clinical judgment, though the decision can be aided by hypercarbia or hypoxia on a blood gas. The most experienced intubator should make the first attempt, as the risk of cardiac arrest peri- and post-intubation is high for asthmatics. Ventilator managements should focus on preventing barotrauma, allowing for long exhalation times, and maximizing oxygenation. Chest radiography is recommended for all patients in status asthmaticus to look for alternate diagnosis or triggers for their exacerbation. Anaphylaxis should be considered for patients with comorbid allergies. Patients who require continuous nebulization or advanced respiratory support should be admitted or transferred to a pediatric intensive care unit for further management.
REFERENCES
- National Center for Environmental Health. US Centers for Disease Control and Prevention. Asthma in the US. CDC Vital Signs. Released May 3, 2011. Available at: http://www.cdc.gov/vitalsigns/pdf/2011-05-vitalsigns.pdf. Accessed April 14, 2015.
- Moorman JE, Akinbami LJ, Bailey CM, et al. National Surveillance of Asthma: United States, 2001–2010. National Center for Health Statistics. Vital Health Stat 3 2012;3:1-58.
- Bratton SL, Newth CJ, Zuppa AF, et al. Critical care for pediatric asthma: Wide care variability and challenges for study. Pediatr Crit Care Med 2012;13:407-414.
- Newth CJ, Meert KL, Clark AE, et al. Fatal and near-fatal asthma in children: The critical care perspective. J Pediatr 2012;161:214-221.
- Bijl-Hofland ID, Cloosterman SG, Folgering HT, et al. Relation of the perception of airway obstruction to the severity of asthma. Thorax 1999;54:15-19.
- Dieckmann RA, Brownstein D, Gausche-Hill M. The pediatric assessment triangle: A novel approach for the rapid evaluation of children. Pediatr Emerg Care 2010;26:312-315.
- Becker AB, Nelson NA, Simons FE. The pulmonary index: Assessment of a clinical score for asthma. Am J Dis Child 1984;138:574-576.
- Ducharme FM, Chalut D, Plotnick L, et al. The Pediatric Respiratory Assessment Measure: A valid clinical score for assessing acute asthma severity from toddlers to teenagers. J Pediatr 2008;152:476-480.
- Gorelick MH, Stevens MW, Schultz TR, et al. Performance of a novel clinical score, the Pediatric Asthma Severity Score (PASS), in the evaluation of acute asthma. Acad Emerg Med 2004;11:10-18.
- van der Windt DA, Nagelkerke AF, Bouter LM, et al. Clinical scores for acute asthma in pre-school children. J Clin Epidemiol 1994;47:635-646.
- Keogh KA, Macarthur C, Parkin PC, et al. Predictors of hospitalization in children with acute asthma. J Pediatr 2001;139:273-277.
- Gorelick M, Scribano PV, Stevens MW, et al. Predicting need for hospitalization in acute pediatric asthma. Pediatr Emerg Care 2008;24:735-744.
- Bergström SE, Boman G, Eriksson L, et al. Asthma mortality among Swedish children and young adults, a 10-year study. Respir Med 2008;102:1335-1341.
- Barnes PJ, Adcock IM. How do corticosteroids work in asthma? Ann Intern Med 2003;139(5 Pt 1):359-370.
- Cropp GJ. Regional differences in prevalence and risks of respiratory diseases in children. Pediatr Pulmonol Suppl 1999;18:37-40.
- National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): Guidelines for the Diagnosis and Management of Asthma-Summary Report 2007. J Allergy Clin Immunol 2007;120(5 Suppl):S94-138.
- British Thoracic Society Scottish Intercollegiate Guidelines Network. British Guideline on the Management of Asthma. Thorax 2008;63(Suppl 4):iv1-121.
- Jones BP, Paul A. Management of acute asthma in the pediatric patient: An evidence-based review. Pediatr Emerg Med Pract 2013;10:1-23.
- Nelson KA, Zorc JJ. Asthma update. Pediatr Clin North Am 2013;60:1035-1048.
- Rogers L, Reibman J. Pharmacologic approaches to life-threatening asthma. Ther Adv Respir Dis 2011;5:397-408.
- Pollack CV Jr, Pollack ES, Baren JM, et al. A prospective multicenter study of patient factors associated with hospital admission from the emergency department among children with acute asthma. Arch Pediatr Adolesc Med 2002;156:934-940.
- Keahey L, Bulloch B, Becker AB, et al. Initial oxygen saturation as a predictor of admission in children presenting to the emergency department with acute asthma. Ann Emerg Med 2002;40:300-307.
- Rodrigo C, Rodrigo G. Therapeutic response patterns to high and cumulative doses of salbutamol in acute severe asthma. Chest 1998;113:593-598.
- Camargo CA Jr, Spooner CH, Rowe BH. Continuous versus intermittent beta-agonists for acute asthma. Cochrane Database Syst Rev 2003;(4):CD001115.
- Cates CJ, Crilly JA, Rowe BH. Holding chambers (spacers) versus nebulisers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev 2006;(2):CD000052.
- Castro-Rodriguez JA, Rodrigo GJ. Beta-agonists through metered-dose inhaler with valved holding chamber versus nebulizer for acute exacerbation of wheezing or asthma in children under 5 years of age: A systematic review with meta-analysis. J Pediatr 2004;145:172-177.
- Wildhaber JH, Dore ND, Wilson JM, et al. Inhalation therapy in asthma: Nebulizer or pressurized metered-dose inhaler with holding chamber? In vivo comparison of lung deposition in children. J Pediatr 1999;135:28-33.
- Jat KR, Khairwa A. Levalbuterol versus albuterol for acute asthma: A systematic review and meta-analysis. Pulm Pharmacol Ther 2013;26:239-248.
- Tschudy MM, Arcara KM. Eds. The Harriet Lane Handbook :A Manual for Pediatric House Officers. Philadelphia, PA: Mosby Elsevier; 2012.
- Teoh L, Cates CJ, Hurwitz M, et al. Anticholinergic therapy for acute asthma in children. Cochrane Database Syst Rev 2012;(4):CD003797.
- Griffiths B, Ducharme FM. Combined inhaled anticholinergics and short-acting beta2-agonists for initial treatment of acute asthma in children. Cochrane Database Syst Rev 2013;(8):CD000060.
- Rodrigo GJ, Castro-Rodriguez JA. Anticholinergics in the treatment of children and adults with acute asthma: A systematic review with meta-analysis. Thorax 2005;60:740-746.
- Goggin N, Macarthur C, Parkin PC. Randomized trial of the addition of ipratropium bromide to albuterol and corticosteroid therapy in children hospitalized because of an acute asthma exacerbation. Arch Pediatr Adolesc Med 2001;155:1329-1334.
- Chang AB, Clark R, Sloots TP, et al. A 5- versus 3-day course of oral corticosteroids for children with asthma exacerbations who are not hospitalised: A randomised controlled trial. Med J Aust 2008;189:306-310.
- Qureshi F, Zaritsky A, Poirier MP. Comparative efficacy of oral dexamethasone versus oral prednisone in acute pediatric asthma. J Pediatr 2001;139:20-26.
- Greenberg RA, Kerby G, Roosevelt GE. A comparison of oral dexamethasone with oral prednisone in pediatric asthma exacerbations treated in the emergency department. Clin Pediatr 2008;47:817-823.
- Redman E, Powell C. Question 1: Prednisolone or dexamethasone for acute exacerbations of asthma: Do they have similar efficacy in the management of exacerbations of childhood asthma? Arch Dis Child 2013;98:916-919.
- Lahn M, Bijur P, Gallagher EJ. Randomized clinical trial of intramuscular vs oral methylyprednisolone in the treatment of acute asthma exacerbations following discharge from an emergency department. Chest 2004;126:362-368.
- Gries DM, Moffitt DR, Pulos E, Carter ER. A single dose of intramuscularly administered dexamethasone acetate is as effective as oral prednisone to treat asthma exacerbations in young children. J Pediatr 2000;136:298-303.
- Amir L, Hubermann H, Halevi A, et al. Oral betamethasone versus intramuscular dexamethasone for the treatment of mild to moderate viral croup: A prospective, randomized trial. Pediatr Emerg Care 2006;22:541-544.
- Edmonds ML, Milan SJ, Camargo CA Jr, et al. Early use of inhaled corticosteroids in the emergency department treatment of acute asthma. Cochrane Database Syst Rev 2012;(12):CD002308.
- Volovitz B. Inhaled budesonide in the management of acute worsenings and exacerbations of asthma: A review of the evidence. Respir Med 2007;101:685-695.
- Schramm CM, Carroll CL. Advances in treating acute asthma exacerbations in children. Curr Opin Pediatr 2009;21:326-332.
- Shan Z, Rong Y, Yang W, et al. Intravenous and nebulized magnesium sulfate for treating acute asthma in adults and children: A systematic review and meta-analysis. Respir Med 2013;107:321-330.
- Goodacre S, Cohen J, Bradburn M, et al. Intravenous or nebulised magnesium sulphate versus standard therapy for severe acute asthma (3Mg trial): A double-blind, randomised controlled trial. Lancet Respir Med 2013;1:293-300.
- Kew KM, Kirtchuk L, Michell CI. Intravenous magnesium sulfate for treating adults with acute asthma in the emergency department. Cochrane Database Syst Rev 2014;(5):CD010909.
- Powell CV, Kolamunnage-Dona R, Lowe J, et al. MAGNEsium Trial in Children (MAGNETIC): A randomised, placebo controlled trial and economic evaluation of nebulised magnesium sulphate in acute severe asthma in children. Health Technol Assess 2013;17:v-vi, 1-216.
- Wiebe K, Rowe BH. Nebulized racemic epinephrine used in the treatment of severe asthmatic exacerbation: A case report and literature review. CJEM 2007;9:304-308.
- Plint AC, Osmond MH, Klassen TP. The efficacy of nebulized racemic epinephrine in children with acute asthma: A randomized, double-blind trial. Acad Emerg Med 2000;7:1097-1103.
- Travers A, Jones AP, Kelly K, et al. Intravenous beta 2-agonists for acute asthma in the emergency department. Cochrane Database Syst Rev 2001;(1):CD002988.
- Stephanopoulos DE, Monge R, Schell KH, et al. Continuous intravenous terbutaline for pediatric status asthmaticus. Crit Care Med 1998;26:1744-1748.
- Allen JY, Macias CG. The efficacy of ketamine in pediatric emergency department patients who present with acute severe asthma. Ann Emerg Med 2005;46:43-50.
- Denmark TK, Crane HA, Brown L. Ketamine to avoid mechanical ventilation in severe pediatric asthma. J Emerg Med 2006;30:163-166.
- Petrillo TM, Fortenberry JD, Linger JF, et al. Emergency department use of ketamine in pediatric status asthmaticus. J Asthma 2001;38:657-664.
- Shlamovitz GZ, Hawthorne T. Intravenous ketamine in dissociating dose as a temporizing measure to avoid mechanical ventilation in adult patient with severe asthma exacerbation. J Emerg Med 2011;41:492-494.
- Green SM, Roback MG, Kennedy RM, Krauss B. Clinical practice guideline for emergency department ketamine dissociative sedation: 2011 update. Ann Emerg Med 2011;57:449-461.
- Howton JC, Rose J, Duffy S, et al. Randomized, double-blind, placebo-controlled trial of intravenous ketamine in acute asthma. Ann Emerg Med 1996;27:170-175.
- Yung M, South M. Randomised controlled trial of aminophylline for severe acute asthma. Arch Dis Child 1998;79:405-410.
- Ream RS, Loftis LL, Albers GM, et al. Efficacy of IV theophylline in children with severe status asthmaticus. Chest 2001;119:1480-1488.
- Mitra A, Bassler D, Goodman K, et al. Intravenous aminophylline for acute severe asthma in children over two years receiving inhaled bronchodilators. Cochrane Database Syst Rev 2005;(2):CD 001276.
- Goodman DC, Littenberg B, O’Connor GT, et al. Theophylline in acute childhood asthma: A meta-analysis of its efficacy. Pediatr Pulmonol 1996;21:211-218.
- Bien JP, Bloom MD, Evans RL, et al. Intravenous theophylline in pediatric status asthmaticus: A prospective, randomized, double-blind, placebo-controlled trial. Clin Pediatr 1995;34:475-481.
- Dalabih AR, Bondi SA, Harris ZL, et al. Aminophylline infusion for status asthmaticus in the pediatric critical care unit setting is independently associated with increased length of stay and time for symptom improvement. Pulm Pharmacol Ther 2014;27:57-61.
- Jat KR, Chawla D. Ketamine for management of acute exacerbations of asthma in children. Cochrane Database Syst Rev 2012;(11):CD009293.
- Inokuchi R, Aoki A, Aoki Y, Yahagi N. Effectiveness of inhaled furosemide for acute asthma exacerbation: A meta-analysis. Crit Care 2014;18:621.
- Gainnier M, Forel JM. Clinical review: Use of helium-oxygen in critically ill patients. Crit Care 2006;10:241.
- McGarvey JM, Pollack CV. Heliox in airway management. Emerg Med Clin North Am 2008;26:905-920.
- Myers TR. Use of heliox in children. Respir Care 2006;51:619-631.
- Mayordomo-Colunga J, Medina A, Rey C, et al. Non?invasive ventilation in pediatric status asthmaticus: A prospective observational study. Pediatr Pulmonol 2011;46:949-955.
- Krishnan V, Diette GB, Rand CS, et al. Mortality in patients hospitalized for asthma exacerbations in the United States. Am J Respir Crit Care Med 2006;174:633-638.
- McFadden ER Jr. Acute severe asthma. Am J Respir Crit Care Med 2003;168:740-759.
- Wimalasena Y, Burns B, Reid C, et al. Apneic oxygenation was associated with decreased desaturation rates during rapid sequence intubation by an Australian helicopter emergency medicine service. Ann Emerg Med 2015;65:371-376.
- L’Hommedieu CS, Arens JJ. The use of ketamine for the emergency intubation of patients with status asthmaticus. Ann Emerg Med 1987;16:568-571.
- Hemmingsen C, Nielsen PK, Odorico J. Ketamine in the treatment of bronchospasm during mechanical ventilation. Am J Emerg Med 1994;12:417-420.
- Papiris S, Kotanidou A, Malagari K, et al. Clinical review: Severe asthma. Crit Care 2002;6:30-44.
- Murata A, Ling PM. Asthma diagnosis and management. Emerg Med Clin North Am 2012;30:203-222.
- Sarnaik AP, Daphtary KM, Meert KL, et al. Pressure-controlled ventilation in children with severe status asthmaticus. Pediatr Crit Care Med 2004;5:133-138.
- Sabato K, Hanson JH. Mechanical ventilation for children with status asthmaticus. Respir Care Clin N Am 2000;6:171-188.
- Hoek TLV, Morrison LJ, Shuster M, et al. Part 12: Cardiac Arrest in Special Situations. 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2010;122(18 Suppl 3):S829-S861.
- Maggi JC, Nussbaum E, Babbitt C, et al. Pediatric fiberoptic bronchoscopy as adjunctive therapy in acute asthma with respiratory failure. Pediatr Pulmonol 2012;47:1180-1184.
- Masuda Y, Tatsumi H, Goto K, et al. Treatment of life-threatening hypercapnia with isoflurane in an infant with status asthmaticus. J Anesth 2014;28: 610-612.
- Hebbar KB, Petrillo-Albaran T, Coto-Puckett W, et al. Experience with use of extracorporeal life support for severe refractory status asthmaticus in children. Crit Care 2009;13:R29.
- Marrie TJ, Lau CY, Wheeler SL, et al. Predictors of symptom resolution in patients with community-acquired pneumonia. Clin Infect Dis 2000;31:1362-1367.
- Assefa D, Amin N, Stringel G, Dozor AJ. Use of decubitus radiographs in the diagnosis of foreign body aspiration in young children. Pediatr Emerg Care 2007;23:154-157.
- Sampson HA, Munoz-Furlong A, Campbell RL, et al. Second symposium on the definition and management of anaphylaxis: Summary report — second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network symposium. Ann Emerg Med 2006;47:373-380.
MONOGRAPH: A small subset of children present in extremis requiring second- and third-line medications and airway management. Here is how to treat them.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
