Acute Hepatitis in the Emergency Department
April 1, 2016
Reprints
Authors
Jean-Aine Pretanvil, MD, University of California San Diego Department of Emergency Medicine.
Michael Wilson, MD, PhD, FAAEM, Attending Physician, University of California San Diego Department of Emergency Medicine, Director, Department of Emergency Medicine Behavioral Emergencies Research (DEMBER) Lab, UC San Diego Health System.
Peer Reviewer
Frank LoVecchio, DO, FACEP, Vice-Chair for Research, Medical Director, Samaritan Regional Poison Control Center, Emergency Medicine Department, Maricopa Medical Center, Phoenix.
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Farel (CME question reviewer) owns stock in Johnson & Johnson. Dr. Stapczynski (editor) owns stock in Pfizer, Johnson & Johnson, Walgreens Boots Alliance Inc., GlaxoSmithKline, Bristol Myers Squibb, and AxoGen. Dr. Schneider (editor), Ms. Fessler (nurse planner), Dr. Pretanvil (author), Dr. Wilson (author), Dr. LoVecchio (peer reviewer), Ms. Mark (executive editor), Ms. Joy Dickinson (executive editor), and Mr. Landenberger (editorial and continuing education director) report no financial relationships with companies related to the field of study covered by this activity.
Introduction
The first descriptions of hepatitis (epidemic jaundice) generally are attributed to Hippocrates. Outbreaks of jaundice, probably hepatitis A, were reported in the 17th and 18th centuries, particularly in association with military campaigns. Hepatitis A (formerly called infectious hepatitis) was first differentiated epidemiologically from hepatitis B, which has a longer incubation period, in the 1940s.1
In the United States, the epidemiology of viral hepatitis, most commonly caused by hepatitis A, B, or C viruses, has changed dramatically over the past three decades. With the development of vaccines for hepatitis A and B viruses and the introduction of universal precautions and blood supply safety measures, hepatitis A virus (HAV) and hepatitis B virus (HBV) infection rates have declined by 88% and 64%, respectively, since 2000. By contrast, after years of stable or declining rates, the incidence of hepatitis C virus (HCV) infection has increased by 75% since 2010.2
In 2010, the U.S. Department of Health and Human Services (HHS) convened an interagency working group on viral hepatitis, comprised of experts throughout HHS, to develop a comprehensive strategy for addressing the prevention, screening, and treatment of viral hepatitis and for improving the coordination of care and treatment of individuals infected with viral hepatitis. The working group broadened the scope of expertise even further by soliciting information from other government agencies, professional organizations, community organizations, and members of the general public. As a result of many months of work by this diverse group of experts, HHS recently released “Combating the Silent Epidemic of Viral Hepatitis: U.S. Department of Health and Human Services Action Plan for the Prevention, Care and Treatment of Viral Hepatitis” (also called the “Viral Hepatitis Action Plan”).3
Viral hepatitis can be prevented and treated. Physicians and other health professionals should play an important role in promoting the prevention and treatment of viral hepatitis among their patients. Fulminant hepatic failure, also known as fulminant hepatitis or acute liver failure, is characterized by acute liver injury, hepatic encephalopathy, and an elevated prothrombin time/international normalized ratio (INR). Untreated, the prognosis is poor, so timely recognition and management of patients with acute liver failure is crucial. Fulminant hepatic failure can result from a wide variety of causes, which are summarized in Table 1.4
TABLE 1. CAUSES OF FULMINANY HEPATIC FAILURE
- Acetaminophen
- Idiosyncratic drug reactions
- Viral hepatitis
- Alcoholic hepatitis
- Autoimmune hepatitis
- Wilson disease
- Ischemic hepatopathy
- Budd-Chiari syndrome
- Veno-occlusive disease
- Acute fatty liver of pregnancy/HELLP (hemolysis, elevated liver enzymes, low platelets) syndrome
- Malignant infiltration (most often breast cancer, small cell lung cancer, lymphoma, melanoma, or myeloma)
- Partial hepatectomy
- Toxin exposure, including mushroom poisoning
- Sepsis
- Heat stroke
- Hemophagocytic lymphohistiocytosis (primarily a disorder of children)
FIGURE 1. INCIDENCE OF HEPATITIS A
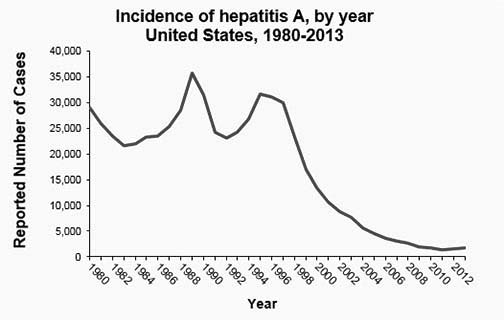
Source: Centers for Disease Control and Prevention
Viral and drug-induced hepatitis are the most common causes of acute liver failure in adults. This article will focus mainly on the different types of viral hepatitis.
COMMON FEATURES
The two important issues for patients with acute hepatitis are to identify the potential for hepatic failure and the cause of the liver damage. Prognosis in acute liver failure is dependent on etiology. Acute liver failure from certain causes demands immediate and specific treatment. It is also critical to identify early those patients who will be candidates for liver transplantation.
All patients with clinical or laboratory evidence of moderate or severe acute hepatitis should have immediate measurement of prothrombin time (PT) and careful evaluation of mental status. Liver enzymes such as AST and ALT are released by damaged liver cells. In fulminant damage, they rise, but may fall when damage is extensive. Prothrombin time is a measure of liver function. Prolongation of the PT or alteration in mental sensorium is grounds for hospital admission. A complete blood count (CBC) in patients with liver failure may reveal thrombocytopenia.
Laboratory test abnormalities typically seen in patients with acute liver failure include:
- Prolonged prothrombin time, resulting in an INR ≥ 1.5;
- Elevated aminotransferase levels (often markedly elevated);
- Elevated bilirubin level;
- Low platelet count (≤ 150,000/mm3).
Laboratory test findings often vary depending upon the specific cause of the acute liver failure. Patterns seen on laboratory testing may suggest a diagnosis, but additional laboratory and imaging testing is required prior to making a diagnosis. Some patterns that may be seen include:
- Hepatitis B: Aminotransferase levels of 1000 to 2000 international units/L are common, alanine aminotransferase (ALT) level that is higher than the aspartate aminotransferase (AST) level.
- Ischemic hepatic injury: Very high aminotransferase levels (25 to 250 times the upper limit of normal), elevated serum LDH levels.
- Wilson disease: Coombs-negative hemolytic anemia, aminotransferase levels < 2000 international units/L, AST to ALT ratio of > 2, normal or markedly subnormal alkaline phosphatase (< 40 international units/L), alkaline phosphatase (international units/L) to total bilirubin (mg/dL) ratio < 4, rapidly progressive renal failure, low uric acid levels.
- Acetaminophen: Very high aminotransferase levels (> 3500 international units/L), low bilirubin, high INR.
- Acute fatty liver of pregnancy/HELLP syndrome (hemolysis, elevated liver enzymes, and low platelets): Aminotransferase levels < 1000 international units/L, elevated bilirubin, low platelet count.
- Herpes simplex virus: Markedly elevated transaminases, leukopenia, low bilirubin.
- Reye’s syndrome, valproate toxicity, or tetracycline toxicity: Minor to moderate elevations in aminotransferase and bilirubin levels.
HEPATITIS A VIRUS INFECTION
Epidemiology. HAV infection occurs worldwide. In the United States, the incidence has declined substantially since vaccination was recommended for persons at increased risk (in 1996), children living in states with the highest incidence of HAV (in 1999), and for all United States–born infants (in 2006).5
HAV is spread via the fecal-oral route and is more prevalent in low socioeconomic areas where a lack of adequate sanitation and poor hygienic practices facilitate spread of the infection. The most common reported risk factor in the United States is international travel (up to 50% of cases), mainly to Mexico and Central and South America.6 Other risk factors include sexual and household contact with another person with hepatitis A (reported in about 10% of cases), homosexual activity in men (about 9%), foodborne or waterborne outbreaks (about 7%), child or employee in a daycare center (about 4%), living or working in small group home settings for the disabled, and injection drug use (about 3%). Community outbreaks due to contaminated water or food also have been described.7 Uncooked HAV-contaminated foods have been recognized as a source of outbreaks. Cooked foods can transmit HAV if the temperature during food preparation is inadequate to kill the virus or if food is contaminated after cooking, as occurs in outbreaks associated with infected food handlers. Waterborne outbreaks are infrequent in developed countries with well-maintained sanitation and water supplies. Maternal-fetal transmission has not been reported.
Clinical Evaluation. HAV infection usually results in an acute, self-limited illness and only rarely leads to fulminant hepatic failure.8 Fulminant hepatic failure occurs more commonly in patients with underlying liver disease, particularly chronic HCV infection. The manifestations also vary with age. In children younger than 6 years of age, 70% of infections are asymptomatic; if illness does occur, it is typically not accompanied by jaundice. Among older children and adults, infection is typically symptomatic, with jaundice occurring in more than 70% of patients. Table 2 shows individuals who are at increased risk for acquiring HAV infection.
TABLE 2. WHO IS AT INCREASED RISK FOR ACQUIRING HAV INFECTION?
- Travelers to countries with high or intermediate endemicity of HAV infection
- Men who have sex with men
- Users of injection and non-injection illegal drugs
- People with clotting factor disorders
- People working with nonhuman primates
The incubation period averages 30 days (range 15-49 days), after which the illness begins with the abrupt onset of prodromal symptoms including fatigue, malaise, nausea, vomiting, anorexia, fever, and right upper quadrant pain.9 Within a few days to one week, patients note dark urine, acholic stool (light-colored stools lacking bilirubin pigment), jaundice, and pruritus. The prodromal symptoms usually diminish when jaundice appears; jaundice typically peaks within 2 weeks. Symptoms usually last less than 2 months, although 10–15% of symptomatic patients have prolonged or relapsing disease for up to 6 months. The two most common physical findings are jaundice and hepatomegaly, which occur in 70% and 80% of symptomatic patients, respectively.10 Less common findings include splenomegaly, cervical lymphadenopathy, evanescent rash, arthritis, and, rarely, a leukocytoclastic vasculitis. Hepatitis A does not become chronic.
Diagnosis. The diagnosis of acute HAV infection is made by the detection of anti-HAV antibodies in a patient with the typical clinical presentation. Serum IgM anti-HAV is the gold standard for the detection of acute illness. The presence of serum IgM anti-HAV antibodies in adults without clinical features of viral hepatitis does not necessarily indicate acute infection. Such patients may have previous HAV infection with prolonged presence of IgM anti-HAV, a false-positive result, or asymptomatic infection (which is much more common in children younger than age 6 compared with older children and adults).11
Differential Diagnosis:
- Hepatitis B, C, D, and E: Hepatitis A and E are acute infections transmitted by the fecal-oral route, whereas hepatitis B, C, and D can present acutely or chronically and are transmitted by body fluids.
- Epstein-Barr and cytomegalovirus: Both Epstein-Barr and cytomegalovirus may present with liver function abnormalities, as well as fever, fatigue, and lymphadenopathy.
- Yellow fever virus: Yellow fever virus is transmitted by mosquitoes in regions where endemic; initial manifestations consist of malaise and other nonspecific symptoms, followed by acute illness with fever and gastrointestinal manifestations.
- Herpes simplex virus: Herpes simplex virus typically consists of oral, genital, and cutaneous manifestations; hepatitis is a rare complication that presents fulminantly.
- Adenovirus: Adenovirus infection typically involves the respiratory and gastrointestinal tracts; hepatitis is a complication of adenovirus infection in immunocompromised hosts.
- Malaria: Malaria is a mosquito-borne parasitic infection characterized by fever, anemia, and parasitemia; clinical manifestations include jaundice due to hemolysis. The diagnosis is established by peripheral smear.
- Leptospirosis: Leptospirosis is a bacterial infection characterized by fever, myalgia, headache, and conjunctival suffusion. Modest elevation of hepatic transaminases may be observed. The diagnosis is established by serology.
- Syphilis: Syphilis is a sexually transmitted infection; secondary syphilis consists of a number of clinical manifestations, including elevated serum alkaline phosphatase, often with normal or only slightly abnormal transaminases. The diagnosis is established by serology.
- Q fever: Q fever occurs as a result of infection with Coxiella burnetii; hepatic involvement includes transaminitis, hepatomegaly without jaundice, and granulomas on liver biopsy. The diagnosis is established by serology.
Treatment and Prognosis. Hepatitis A is usually self-limited; the treatment is supportive. Patients who develop fulminant infection require aggressive supportive therapy and should be transferred to a center capable of performing liver transplantation. Approximately 85% of HAV-infected individuals have full clinical and biochemical recovery within 3 months, and nearly all have complete recovery by 6 months.12 Fatalities due to hepatitis A are more common with advancing age and in patients with chronic hepatitis C.13
Prevention. Hepatitis A is transmitted predominantly by the fecal-oral route. The incubation period is 2-6 weeks; infected individuals are contagious during the incubation period and remain so for about a week after jaundice appears.14 Prevention can be aided by adherence to sanitary practices such as handwashing, heating foods appropriately, and avoiding water and foods from endemic areas. Handwashing is highly effective in preventing the transmission of the virus since HAV may survive for up to 4 hours on the fingertips.15 Chlorination and certain disinfecting solutions are sufficient to inactivate the virus.
Vaccination and Postexposure Prophylaxis. HAV infection is prevalent in many developing countries and is among the most common preventable infections acquired by travelers. Humans are the only known reservoir for hepatitis A virus; therefore, in theory, the virus could be eradicated if widespread immunization strategies were employed successfully. Since the availability of the hepatitis A vaccine in 1995, the rates of HAV infection in the United States have declined by 95%.16-18
Globally, the World Health Organization (WHO) recommends immunization for children 1 year old and older in places with intermediate endemicity on the basis of incidence of acute hepatitis A.19 In places of high endemicity, almost all people are asymptomatically infected with HAV in childhood, which provides immunity against clinical infection; therefore, universal vaccination in these countries is not recommended.
Adults: The Advisory Committee on Immunization Practices (ACIP) of the United States Centers for Disease Control and Prevention (CDC) recommends HAV vaccination for adults with the following risk factors:
- Individuals traveling to or working in countries with high or intermediate rates of hepatitis A;
- Individuals with chronic liver disease;
- Individuals with clotting factor disorders;
- Men who have sex with men;
- Illicit drug users (injection and noninjection);
- Individuals with close personal contact with an international adoptee from a country of high or intermediate endemicity during the first 60 days following arrival in the United States (e.g., household contact or regular babysitting);20-21
- Individuals working with HAV-infected primates or with HAV in a research laboratory;
- Individuals with recent exposure for postexposure prophylaxis.
Children: HAV vaccination was incorporated into the routine childhood vaccination schedule in the United States in 2006. Previously, HAV vaccination was recommended in regions of the United States where the incidence of HAV was greater than the national average (approximately 20 cases per 100,000 rather than 10 cases per 100,000).
Postexposure prophylaxis: Individuals with recent HAV exposure who have not previously received HAV vaccine should receive postexposure prophylaxis with either a single dose of single-antigen HAV vaccine or immune globulin (IG; 0.02 mL/kg) as soon as possible, within 2 weeks of exposure.22
HEPATITIS B VIRUS INFECTION
Epidemiology and Mode of Transmission. HBV infection is a global public health problem. It is estimated that there are 240 million HBV carriers in the world, of whom roughly 600,000 die annually from HBV-related liver disease.23 The predominant mode of transmission of HBV varies in different geographical areas. Perinatal infection is the predominant mode of transmission in high prevalence areas.24,25 Perinatal transmission is estimated to have occurred in 1058 of all U.S. births in 2009. In comparison, horizontal transmission, particularly in early childhood, accounts for most cases of chronic HBV infection in intermediate prevalence areas, while unprotected sexual intercourse and intravenous drug use in adults are the major routes of spread in low prevalence areas.26 In the United States, the most common modes of HBV transmission are sexual contact and injection drug use. Acute infection is symptomatic in up to 50% of adult cases; infants and children are usually asymptomatic. People with a compromised immune system are at elevated risk of developing chronic infection. HBV is considered hardy and remains infectious outside the body for about 7 days. To prevent HBV infection, people are advised to get vaccinated, avoid unprotected sexual contact, and avoid percutaneous or mucosal contact with infected blood, body fluids, and items that have been in contact with potentially infected blood or body fluids, such as needles and other equipment used to inject drugs.
Clinical Manifestations of Acute Hepatitis B Virus Infection. Fulminant hepatic failure is unusual, occurring in approximately 0.1-0.5% of patients. Fulminant hepatitis B is believed to be due to massive immune-mediated lysis of infected hepatocytes. This explains why many patients with fulminant hepatitis B have no evidence of HBV replication at presentation.27 The incubation period lasts 1-4 months. A serum sickness-like syndrome may develop during the prodromal period, followed by constitutional symptoms, anorexia, nausea, jaundice, and right upper quadrant discomfort. The symptoms and jaundice generally disappear after 1-3 months, but some patients have prolonged fatigue even after normalization of serum aminotransferase concentrations.
Treatment. Treatment for acute HBV is mainly supportive. In addition, appropriate measures should be taken to prevent infection in exposed contacts. The decision to hospitalize patients should be individualized. Patients who have a coagulopathy, are jaundiced, or are encephalopathic generally should be hospitalized. Hospitalization also might be considered in patients who are older, have significant comorbidities, cannot tolerate oral intake, or have poor social support systems. Not all the patients with acute HBV require antiviral treatment since the likelihood of fulminant hepatitis B is less than 1%, and in immunocompetent adults, the likelihood of progression to chronic HBV infection is less than 5%.28
The worldwide epidemics of hepatitis B and HIV have led to new understanding of the complicated interactions between these two viruses. Coinfection with HIV has a major impact on the natural history, diagnosis, progression, and morbidity and mortality related to HBV infection. The presence of chronic hepatitis B can also lead to an increased risk of hepatotoxicity related to the administration of potent antiretroviral therapy (ART). HBV and HIV often are diagnosed in the same patient because they share similar routes of transmission. HIV-infected patients should have routine testing for HBV infection.
ART is indicated in all HIV-infected patients with HBV coinfection; two of the three HIV agents (tenofovir, emtricitabine, and lamivudine) should have dual activity against HIV and HBV. However, treatment of HBV in the HIV-infected patient can be complicated by drug-induced hepatotoxicity, immune constitution syndromes, and toxicity related to medications. Furthermore, regardless of whether the patient is treated or not, surveillance for development of hepatocellular carcinoma is required.
Postexposure Prophylaxis. Healthcare personnel (HCP) are at risk for HBV and/or HCV infection if they are exposed to blood and/or certain other body fluids.29 An exposure can occur through:
- A percutaneous injury (e.g., a needlestick or cut with a sharp object);
- Contact with mucous membranes or nonintact skin (e.g., exposed skin that is chapped, abraded, or afflicted with dermatitis).
Blood is the most important source of HBV and HCV transmission in HCP. Other body fluids, such as cerebrospinal fluid, synovial fluid, pleural fluid, peritoneal fluid, and amniotic fluid, are considered potentially infectious.
Wound Care: Exposed mucous membranes should be flushed with water. Wounds and skin sites that have been in contact with blood or body fluids should be washed with soap and water.
Obtaining Information: Trained medical personnel (e.g., occupational health providers) should obtain information about the source patient, the exposed HCP, and the type of exposure to determine an appropriate postexposure treatment plan.
- Source Patient: The hepatitis B surface antigen (HBsAg) and hepatitis C antibody (anti-HCV) status of the source patient should be assessed.
-
HCP: The following information should be obtained from the injured HCP and verified from their medical/occupational health record:
- Dates of HepB immunizations;
- Postimmunization titer, if known;
- Previous testing (if available) for HBV and HCV;
- Tetanus immunization status;
- Current medications;
- Current or underlying medical conditions that might influence use of/response to vaccination.
-
Exposure: The following information regarding the exposure should be obtained:
- The date and time of the exposure;
- Nature of the exposure (i.e., nonintact skin, mucosal, percutaneous, human bite);
- Type of fluid (i.e., blood, blood contaminated fluid, or other contaminated fluid);
- Body location of the exposure and contact time with the contaminated fluid;
- For percutaneous injuries, a description of the injury (e.g., depth of wound, solid vs hollow needle, sharp use in source patient);
HBV exposure: Postexposure management of HCP with a possible exposure to HBV depends first upon the immune status of the HCP and, second, upon the HBsAg status of the source patient. If the HBsAg status of a source patient cannot be determined, the HCP should be managed as if the source patient is HBsAg-positive. The HBV status (i.e., HBsAg, anti-HBs) of all HCP should be known at the time of employment or shortly thereafter.
For HCP without evidence of prior HBV, the risk of infection depends upon their vaccine status, which is defined as follows:
- Vaccine responder: If the hepatitis B surface antibody (anti-HBs) level after receiving three to six doses of HepB vaccine is ≥ 10 mIU/mL, the individual is considered a vaccine responder and is thought to be protected against HBV infection.
- Vaccine nonresponder: If the anti-HBs remains < 10 mIU/mL after receiving the three-dose HepB vaccine series on two separate occasions (i.e., a total of six doses of HepB vaccine), the individual is considered a vaccine nonresponder.
- Vaccine response unknown: If the three-dose hepatitis B vaccine series was given, but post-vaccine serologic testing was not performed, the individual has an unknown vaccine response.
- Not vaccinated: Such individuals were not vaccinated or did not complete the three-dose series.
No postexposure management is required if the HCP has evidence of past HBV infection (anti-HBs-positive). Such individuals are thought to be protected against HBV infection and do not require postexposure treatment.
No postexposure management is required if the HCP received and adequately responded to the three-dose HepB vaccine series.
The source patient should be tested for HBsAg if the HCP is a vaccine nonresponder. If the HBsAg is positive, or if it cannot be obtained, the HCP should receive two doses of hepatitis B immunoglobulin (HBIG). The second dose of HBIG should be given 1 month after the first dose.
The source patient should be tested for HBsAg, and the HCP should be tested for anti-HBs if the vaccine response is unknown. These tests should be done as soon as possible after the exposure and should occur simultaneously.
The source patient should be tested for HBsAg status if the HCP has no documentation of being vaccinated and/or has not completed the hepatitis B vaccine series.
HBIG provides anti-HBs and generally protects against infection with HBV for 3-6 months. The standard adult dose is 0.06 mL/kg and should be given intramuscularly (preferably within 24 hours).
HCV exposure: There is no effective postexposure prophylaxis for people exposed to HCV. Thus, the management of HCP potentially exposed to HCV involves early diagnosis and treatment of HCV infection should it occur. After a potential exposure, the source patient should be tested for the presence of hepatitis C antibody (anti-HCV) and, if positive, additional testing for HCV RNA should be performed.
Don’t forget to consider other possible diseases like HIV.
COINFECTION WITH HCV OR HDV
Hepatitis B-infected patients may also be infected with HCV or hepatitis D virus (HDV).
HCV infection: Acute coinfection with HBV and HCV may shorten the duration of HBs antigenemia and lower the peak serum aminotransferase concentration compared with acute HBV infection alone.30 These findings suggest that HCV coinfection may interfere with the replication of HBV, and it may lead to attenuation of liver damage. However, acute coinfection of HCV and HBV, or acute HCV on pre-existing chronic HBV also have been reported to increase the risk of severe hepatitis and fulminant hepatic failure.31-32 Similarly, acute HBV in patients with chronic HCV can lead to severe hepatitis, but it also may lead to clearance of HCV.33 HCV superinfection in HBsAg carriers appears to reduce HBV DNA levels in serum and liver tissues and to increase the rate of HBsAg seroconversion.34-35 Most patients who have dual HCV and HBV infections have detectable serum HCV RNA but undetectable or low HBV DNA levels, indicating that HCV is the predominant cause of liver disease in these patients. On the other hand, levels of HBV DNA and HCV RNA fluctuate over time in about one-third of patients.36 Patients with dual HBV and HCV infection also may have a higher rate of hepatocellular carcinoma (HCC) compared with patients infected by either virus alone, particularly those who are anti-HCV and HBeAg positive.35
The treatment of HCV infection with peginterferon plus ribavirin in patients with concomitant HBV infection can lead to a sustained virologic response. The rate of sustained virologic response is similar to that observed in patients with HCV alone.37 However, HBV replication may increase after clearance of HCV.37 Thus, close monitoring of both viruses is required.
HDV infection: Although HDV can replicate autonomously, the simultaneous presence of HBV is required for complete virion assembly and secretion. As a result, individuals with hepatitis D are always dually infected with HDV and HBV. Acute HBV and HDV coinfection tends to be more severe than acute HBV infection alone and is more likely to result in fulminant hepatitis.38 HDV superinfection in patients with chronic HBV infection usually is accompanied by a suppression of HBV replication due to interference mechanisms that are not well understood.39
Vaccination. Three different classes of hepatitis B vaccine are available based upon how they are derived (from plasma, yeast, or mammalian cells).
- The first-generation hepatitis B vaccine was prepared by concentrating and purifying plasma from HBsAg carriers to produce 22 nm subviral particles, which contain HBsAg alone. Derivation from plasma has left lingering concerns regarding the potential to transmit bloodborne infections, although this vaccine has excellent efficacy and safety.40
- Yeast-derived recombinant hepatitis B vaccines were first introduced in the mid-1980s. They are produced by cloning of the HBV S gene in yeast cells. These vaccines contain non-glycosylated HBV small S protein as the envelope antigen, which must be released from the yeast during the manufacturing process.41
- The third class of hepatitis B vaccine is the mammalian cell-derived recombinant vaccine. Three vaccines of this class have been developed. In addition to the S antigen, one of these contains antigen from the pre-S2 region, while the other two contain antigens from both the pre-S1 and pre-S2 regions.
Hepatitis B vaccine is recommended for all neonates of HBsAg-positive mothers, and in many countries is also recommended for neonates of HBsAg-negative mothers.
Recommendations for screening the adult population have been issued by several major organizations, including the United States Preventive Services Task Force (USPSTF), the CDC, the American Association for the Study of Liver Disease (AASLD), and WHO.
All neonates: Neonates should be immunized against HBV. The timing of the first dose of hepatitis B vaccine and the need for hepatitis B immune globulin (HBIG) are determined by the mother’s hepatitis B surface antigen (HBsAg) status, the infant’s gestational age, and the infant’s birth weight. Universal vaccination of all newborns, regardless of maternal HBsAg status, is necessary for global eradication of HBV infection. Countries that have adopted universal vaccination of newborns have experienced a marked reduction in carrier rates as well as complications from HBV, including hepatocellular carcinoma.42,43
Neonates of HBsAg-positive mothers: Vaccination of neonates born to HBsAg-positive mothers is the most important step toward the eradication of chronic HBV infection. The standard regimen for neonates born to HBsAg-positive mothers consists of passive and active immunization. Hepatitis B vaccine and HBIG are given at the same time at two different sites within 12 hours of delivery. The neonates should then receive two additional doses of the hepatitis B vaccine at months 1-2 and months 6-12. This regimen has a protective efficacy of 95%.44
Catch-up vaccination: Catch-up vaccination refers to vaccination of children who were born before universal neonatal vaccination was implemented. All unvaccinated children and adolescents younger than 19 years should receive the HBV vaccine series.45
Other high-risk groups:
- Sexually active individuals with multiple sex partners and homosexual or bisexual males;
- Household contacts of patients with hepatitis B;
- Injection drug users;
- Healthcare workers;
- Patients on chronic hemodialysis and patients requiring repeated blood or blood product transfusion;
- Patients with chronic liver disease;
- All unvaccinated persons traveling to areas with intermediate to high levels of endemic HBV infection;
- Patients with diabetes.
HEPATITIS C VIRUS INFECTION
By convention, acute HCV infection refers to the first 6 months of HCV infection following presumed HCV exposure.46 While HCV infection is estimated to account for 15% of symptomatic cases of acute liver disease in the United States, the majority of patients with acute HCV go undetected.47 This lack of detection is due in large part to the fact that patients with acute HCV are typically asymptomatic.
Most patients infected with HCV in the United States and Europe acquired the disease through intravenous drug use or blood transfusion, the latter of which has become rare since routine testing of the blood supply for HCV began in 1990. Other types of parenteral exposure are important in specific regions in the world. Sexual transmission occurs rarely, although among both men who have sex with men and heterosexuals, the likelihood of sexual transmission rises with the number of sex partners and when sex partners are coinfected with HIV. In addition, there is evidence that men who have sex with men and take HIV pre-exposure prophylaxis may be at risk for sexual transmission of HCV if they have unprotected sex with HCV-infected partners.
Clinical Manifestations. Most patients who are acutely infected with HCV are asymptomatic. Symptomatic patients may experience jaundice, nausea, dark urine, and right upper quadrant pain. Patients with acute HCV infection typically have moderate to high serum aminotransferase elevations. These may go undetected in asymptomatic patients. Among patients who are symptomatic, symptoms typically develop 2-26 weeks after exposure to HCV, with a mean onset of 7-8 weeks.48 The acute illness usually lasts for 2-12 weeks. Fulminant hepatic failure due to acute HCV infection is very rare but may be more common in patients with underlying chronic HBV infection.49
Laboratory Findings. Aminotransferase levels are often greater than 10-20 times the upper limit of normal in patients with acute HCV, but can be highly variable.50 Among patients who develop symptoms, the aminotransferases start to increase shortly before the onset of clinical symptoms and usually before anti-HCV antibodies are detectable. However, the levels often fluctuate and may normalize.51 Thus, not all patients will have elevated aminotransferase levels at the time of presentation, and normalization of the serum aminotransferase concentrations after acute infection does not necessarily mean that the infection has cleared. Patients with acute HCV infection also may have elevated total bilirubin levels.52
Diagnosis. A detectable HCV RNA by polymerase chain reaction (PCR) in the setting of undetectable anti-HCV antibodies that subsequently become detectable within 12 weeks is generally considered definitive proof of acute HCV infection. Alternately, newly detectable HCV RNA and anti-HCV antibodies with documentation of negative tests within the prior 6 months are also suggestive of acute HCV infection.
Management of Side Effects of Treatment of Chronic HCV Infection. Side effects are observed in almost 80% of patients receiving peginterferon and ribavirin combination therapy for chronic HCV infection. Care of patients with chronic HCV infection depends upon recognition of those at increased risk for side effects, anticipation (and prevention) of side effects, and appropriate response when they occur. The side effects include:
- Hematologic effects: Hematologic side effects are common during treatment with peginterferon and ribavirin.53
- Anemia: Development of anemia is extremely common among patients taking peginterferon and ribavirin-containing therapy for chronic HCV infection. Primary management is with ribavirin dose reduction. In some cases, erythropoietic growth factors and red cell transfusions may be warranted.
- Neutropenia: Treatment with interferon is commonly associated with bone marrow suppression, and neutropenia (defined as an absolute neutrophil count [ANC] less than 1500/microL) is commonly observed during treatment for chronic HCV with an interferon-containing regimen. Selective peginterferon dose reduction is the primary management strategy for neutropenia.
- Thrombocytopenia: Thrombocytopenia is common in patients with advanced liver disease. Selective peginterferon dose reduction is the primary management strategy for treatment-associated thrombocytopenia.54
- Flu-like symptoms: The most common side effects associated with interferon-based therapy are flu-like symptoms (mainly muscle aches, headaches, and low-grade fevers), which are seen in more than 80% of patients. Flu-like symptoms are most severe in the first 48 hours after peginterferon administration and persist beyond the third month of therapy in only 10% of patients. It is suggested that patients take acetaminophen (1 gram 30 minutes before injection) and increase their fluid intake.
-
Other effects
- Nausea: Nausea has been reported in 29-43% of patients treated with peginterferon and ribavirin.55 Management includes the use of antiemetic agents such as prochlorperazine.
- Dermatologic complications: HCV infection itself has been associated with multiple dermatologic complications, including leukocytoclastic vasculitis, porphyria cutanea tarda, lichen planus, erythema nodosum, urticaria, erythema multiforme, and polyarteritis nodosa.56 Both interferon and ribavirin also have been associated with dermatologic complications. The most common is a nonspecific, pruritic rash associated with flaky dry skin. The rash resolves over time in many patients but may require a reduction in the dose (or discontinuation) of ribavirin if it is severe. All patients are advised to avoid powerful skin detergents and use regular non-perfumed skin moisturizers in addition to sunblock.
- Teratogenicity: Ribavirin produced significant embryonal or teratogenic effects in every animal species studied. Thus, it is contraindicated in pregnancy, and any woman who becomes pregnant while taking ribavirin should immediately notify her clinician and receive appropriate counseling about teratogenicity and the option for therapeutic abortion. In addition, women should not become pregnant during the 6 months after stopping ribavirin. It is recommended that all patients of childbearing potential practice at least two forms of contraception during and for 6 months following treatment with ribavirin. Interferon caused abortion in pregnant rhesus monkeys. Interferon is therefore not advisable in pregnancy.
- Infections: Acute infections are relatively common during therapy with an interferon-containing regimen. Of infections in which a pathogen was identified, influenza viruses, herpes viruses, Staphylococcus spp., and Candida spp. were the most commonly reported. In general, interferon therapy can be continued in patients with mild to moderate infections that can be managed as outpatients with oral antibiotic therapy (e.g., urinary tract, mild respiratory tract, or localized/uncomplicated skin and soft tissue infections). Patients taking interferon-based HCV therapy who have moderate to severe infections that require hospital admission for management typically should have their entire HCV regimen stopped unless otherwise decided in direct consultation with an expert in HCV therapy.
- Ophthalmologic disorders: Ophthalmologic disorders (retinal hemorrhages, cotton wool spots, loss of color vision, and, rarely, retinal artery or vein obstruction) can occur during interferon therapy. The most common ocular complication has been the development of a mild-to-moderate ischemic retinopathy. The risk may be greater in patients with a history of hypertension or diabetes mellitus.57 Patients should be cautioned about the possibility of visual disturbance when initiating a regimen that contains peginterferon. For patients with diabetes mellitus and hypertension, a baseline retinal examination performed by an ophthalmologist is suggested prior to treatment.
HEPATITIS D VIRUS INFECTION
HDV infection is closely associated with HBV infection. Although HDV can replicate autonomously,58 the simultaneous presence of HBV is required for complete virion assembly and secretion. As a result, individuals with hepatitis D always are dually infected with HDV and HBV. In most cases of HDV infection, HBV replication is suppressed to low levels by HDV. Liver damage in these patients essentially is due to HDV only. Occasionally, HBV and HDV replicate simultaneously, each virus contributing to the liver damage, thereby resulting in more severe liver disease.
Clinical Manifestations. Coinfection: Coinfection of HBV and HDV in an individual susceptible to HBV infection (i.e., anti-HBs-negative) results in acute hepatitis B + D. This entity is clinically indistinguishable from classic acute hepatitis B and is usually transient and self-limited. However, a high incidence of liver failure has been reported among injection drug users.59 The rate of progression to chronic infection is not different from that observed after classic acute hepatitis B, since persistence of HDV infection is dependent upon persistence of HBV infection.60
Superinfection: HDV superinfection of a chronic HBsAg carrier may present as a severe acute hepatitis in a previously unrecognized HBV carrier, or as an exacerbation of preexisting chronic hepatitis B. Progression to chronic HDV infection occurs in almost all patients. However, HBV replication is usually suppressed by HDV.61
Diagnosis. Due to the dependence of HDV on HBV, the presence of HBsAg is necessary for the diagnosis of HDV infection. The additional presence of IgM antibody to hepatitis B core antigen (IgM anti-HBc) is necessary for the diagnosis of acute HBV/HDV coinfection.
Detection of serum HDAg: Serum HDAg can be detected by microplate-based, enzyme-linked immunoassay (EIA) or radioimmunoassays (RIA).
Detection of serum HDV RNA: HDV RNA can be detected in serum by either molecular hybridization or reverse transcriptase-polymerase chain reaction (RT-PCR)-based assays.
Detection of anti-HDV antibody: Total (IgM and IgG) anti-HDV antibodies can be detected by EIAs or RIAs. These are the only commercially available assays for the diagnosis of HDV infection in the United States.
Tissue markers of HDV infection: Both HDAg and HDV RNA can be detected in liver tissues routinely processed for histopathologic evaluation.
Treatment. There is no specific treatment for acute hepatitis D. Foscarnet shows encouraging results, but they still need to be confirmed. Foscarnet is an inhibitor of some viral DNA polymerases. Its efficacy in fulminant hepatitis due to HBV/HDV coinfection may be secondary to its inhibition of HBV.62
HEPATITIS E VIRUS INFECTION
Epidemiology and Mode of Transmission. The epidemiology of hepatitis E virus (HEV), previously known as waterborne or enterically transmitted non-A, non-B hepatitis, is similar to that of HAV.63 However, HAV is more readily transmitted, causes more infections, and has a wider distribution worldwide, although HEV may be more widespread in industrialized countries than generally believed.64 The highest incidence of HEV infection is in Asia, Africa, the Middle East, and Central America. HEV is the second most common cause of sporadic hepatitis in North Africa and the Middle East.65 High attack rates are found in adults between 15 and 40 years of age. HEV is spread by fecally contaminated water in endemic areas. Person-to-person transmission is uncommon. HEV can be transmitted by blood transfusion, particularly in endemic areas. Sporadic cases that have been described in western countries have mostly been limited to visitors who have traveled to areas of the world that are endemic for HEV. However, sporadic cases that were unassociated with travel have been reported. HEV can infect pigs, which suggests that some cases of human transmission may be due to contact transmission across species.66
Clinical Manifestations. HEV generally causes a self-limited acute infection, although fulminant hepatitis can develop. Chronic hepatitis does not develop after acute HEV infection, except in the transplant setting and possibly in other settings of immunosuppression. The incubation period of HEV infection ranges from 15-60 days.67 The clinical signs and symptoms in patients with typical HEV infection are similar to those seen with other forms of acute viral hepatitis, although disease appears to be relatively severe compared with hepatitis A. Prolonged cholestasis has been described in up to 60% of patients. Jaundice is usually accompanied by malaise, anorexia, nausea, vomiting, abdominal pain, fever, and hepatomegaly. Other less common features include diarrhea, arthralgia, pruritus, and urticarial rash. Some patients have asymptomatic infection. Fulminant hepatitis can occur, which results in an overall case fatality rate of 0.5-3%. However, HEV does not appear to be an important cause of fulminant hepatitis in the United States.68
For reasons that are not understood, fulminant hepatic failure occurs more frequently during pregnancy, resulting in an inordinately high mortality rate of 15-25%, primarily in women in the third trimester. A contributing factor may be that pregnancy predisposes women to increased viral replication. Pregnant women with jaundice and acute viral hepatitis caused by HEV infection appear to have worse obstetric and fetal outcomes compared to pregnant women with jaundice and acute viral hepatitis due to other causes.69
Infection with HEV can lead to hepatic decompensation in patients with pre-existing liver disease and those who are malnourished.70 HEV infection also has been reported in recipients of solid organ transplants (kidney, liver, and kidney-pancreas).71
Diagnosis. The diagnosis of HEV is based upon the detection HEV in serum or stool by PCR or by the detection of IgM antibodies to HEV. Antibody tests against HEV alone are less than ideal since they have been associated with frequent false-positive and -negative results.72
Treatment. Treatment of HEV infection remains supportive, as the disease appears to be self-limiting in nonimmunocompromised patients. However, a number of approaches have been used in transplant recipients, as these patients are at increased risk for chronic HEV infection:
- Clearance of chronic infection has been reported after reducing or withdrawing immunosuppressive therapy.73
- Case reports and series have suggested a benefit from ribavirin monotherapy in solid-organ transplant recipients with chronic HEV, but prospective studies are needed to confirm these findings and to determine the dose, duration, and timing of ribavirin therapy.74
- Viral clearance was observed after 3 months of treatment with pegylated interferon in a series of three liver transplant recipients.75
CONCLUSION
The most important aspect of treatment for acute liver failure is to provide good intensive care support, including protection of the airway. Specific therapy is also dependent on the cause of the patient’s liver failure and the presence of any complications. Pay careful attention to the patient’s fluid management and hemodynamics. It is crucial to monitor their metabolic parameters, assess for infection, maintain nutrition, and promptly recognize gastrointestinal bleeding.76
REFERENCES
- Hamborsky J, Kroger A, eds. Epidemiology and prevention of vaccine-preventable diseases, E-Book: The Pink Book. Public Health Foundation, 2015.
- Centers for Disease Control and Prevention. Surveillance for viral hepatitis — United States, 2012. Atlanta; 2014. www.cdc.gov/hepatitis/Statistics/2012Surveillance/PDFs/2012HepSurveillanceRpt.pdf.
- Center for Substance Abuse Treatment. Addressing Viral Hepatitis in People With Substance Use Disorders. Rockville (MD): Substance Abuse and Mental Health Services Administration (US); 2011. (Treatment Improvement Protocol [TIP] Series, No. 53.) 1, Overview of Viral Hepatitis. Available from: www.ncbi.nlm.nih.gov/books/NBK92038/.
- Lee WM, Squires RH, Nyberg SL, et al. Acute liver failure: Summary of a workshop. Hepatology (Baltimore, Md). 2008;47:1401-1415.
- Wasley A, Samandari T, Bell BP. Incidence of hepatitis A in the United States in the era of vaccination. JAMA 2005;294:194-201.
- Klevens R, Miller JT, Iqbal K, et al. The evolving epidemiology of hepatitis A in the United States: Incidence and molecular epidemiology from population-based surveillance, 2005-2007. Arch Intern Med 2010;170:1811-1818.
- De Serres G, Cromeans TL, Levesque B, et al. Molecular confirmation of hepatitis A virus from well water: Epidemiology and public health implications. J Infect Dis 1999;179:37-43.
- Taylor RM, Davern T, Munoz S, et al. Fulminant hepatitis A virus infection in the United States. Incidence, prognosis, and outcomes. Hepatology (Baltimore, Md). 2006;44:1589-1597.
- Lednar WM, Lemon SM, Kirkpatrick JW, et al Frequency of illness associated with epidemic hepatitis A virus infections in adults. Am J Epidemiol 1985;122:226-233.
- Tong MJ, El-Farra NS, Grew ML. Clinical manifestations of hepatitis A: Recent experience in a community teaching hospital. J Infect Dis 1995;171:S15-S18.
- Centers for Disease Control and Prevention. Positive test results for acute hepatitis A virus infection among persons with no recent history of acute hepatitis — United States, 2002-2004. MMWR Morb Mortal Wkly Rep 20013;54:453-456.
- Koff RS. Clinical manifestations and diagnosis of hepatitis A virus infection. Vaccine 1992;10 (Suppl 1):S15–S17.
- Vento S, Garofano T, Renzini C, et al. Fulminant hepatitis associated with hepatitis A virus superinfection in patients with chronic hepatitis C. N Engl J Med 1998;338:286
- Richardson M, Elliman D, Maguire H, et al. Evidence base of incubation periods, periods of infectiousness and exclusion policies for the control of communicable diseases in schools and preschools. Pediatr Infect Dis J 2001;20:380–391.
- Mbithi JN, Springthorpe VS, Boulet JR, et al. Survival of hepatitis A virus on human hands and its transfer on contact with animate and inanimate surfaces. J Clin Microbiol 1992;30:757-763.
- Ly KN, Klevens RM. Trends in disease and complications of hepatitis A virus infection in the United States, 1999-2011: A new concern for adults. J Infect Dis 2015;212:176.
- Wasley A, Samandari T, Bell BP. Incidence of hepatitis A in the United States in the era of vaccination. JAMA 2005;294:194.
- Nelson KE. The changing epidemiology of hepatitis A virus infections in the United States. J Infect Dis 2015;212:171.
- WHO position paper on hepatitis A vaccines — June 2012. Wkly Epidemiol Rec 2012;87:261–276.
- Centers for Disease Control and Prevention, Advisory Committee on Immunization Practices. Updated recommendations from the Advisory Committee on Immunization Practices for use of hepatitis A vaccine in close contacts of newly arriving international adoptees. MMWR Morb Mortal Wkly Rep 2009;58:1006.
- American Academy of Pediatrics Committee on Infectious Diseases. Recommendations for administering hepatitis A vaccine to contacts of international adoptees. Pediatrics 2011;128:803.
- Advisory Committee on Immunization Practices (ACIP) Centers for Disease Control and Prevention (CDC). Update: Prevention of hepatitis A after exposure to hepatitis A virus and in international travelers. Updated recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep 2007;56:1080.
- Ott JJ, Stevens GA, Groeger J, et al. Global epidemiology of hepatitis B virus infection: New estimates of age-specific HBsAg seroprevalence and endemicity. Vaccine 2012;30:2212.
- Alter MJ, Hadler SC, Margolis HS, et al. The changing epidemiology of hepatitis B in the United States. Need for alternative vaccination strategies. JAMA 1990;263:1218.
- Beasley RP, Hwang LY, Lin CC, et al. Incidence of hepatitis B virus infections in preschool children in Taiwan. J Infect Dis 1982;146:198.
- Kim WR, Ishitani MB, Dickson ER, et al. Rising burden of hepatitis B in the United States: Should the other virus be forgotten? Hepatology 2002;36:222A.
- Wright TL, Mamish D, Combs C, et al. Hepatitis B virus and apparent fulminant non-A, non-B hepatitis. Lancet 1992;339:952.
- Kumar M, Satapathy S, Monga R, et al. A randomized controlled trial of lamivudine to treat acute hepatitis B. Hepatology 2007;45:97.
- Schillie S, Murphy TV, Sawyer M, et al. CDC guidance for evaluating health-care personnel for hepatitis B virus protection and for administering postexposure management. MMWR Recomm Rep 2013;62:1.
- Mimms LT, Mosley JW, Hollinger FB, et al. Effect of concurrent acute infection with hepatitis C virus on acute hepatitis B virus infection. BMJ 1993;307:1095.
- Sagnelli E, Coppola N, Messina V, et al. HBV superinfection in hepatitis C virus chronic carriers, viral interaction, and clinical course. Hepatology 2002;36:1285.
- Liaw YF, Chen YC, Sheen IS, et al. Impact of acute hepatitis C virus superinfection in patients with chronic hepatitis B virus infection. Gastroenterology 2004;126:1024.
- Sagnelli E, Coppola N, Pisaturo M, et al. HBV superinfection in HCV chronic carriers: A disease that is frequently severe but associated with the eradication of HCV. Hepatology 2009;49:1090.
- Jardi R, Rodriguez F, Buti M, et al. Role of hepatitis B, C, and D viruses in dual and triple infection: Influence of viral genotypes and hepatitis B precore and basal core promoter mutations on viral replicative interference. Hepatology 2001;34:404.
- Huang YT, Yang HI, Jen CL, et al. Suppression of hepatitis B virus replication by hepatitis C virus: Combined effects on risk of hepatocellular carcinoma (abstract). Hepatology 2005;42 (Suppl 1):230A.
- Raimondo G, Brunetto MR, Pontisso P, et al. Longitudinal evaluation reveals a complex spectrum of virological profiles in hepatitis B virus/hepatitis C virus-coinfected patients. Hepatology 2006;43:100.
- Bruix J, Sherman M, Practice Guidelines Committee, American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma. Hepatology 2005;42:1208.
- Smedile A, Farci P, Verme G, et al. Influence of delta infection on severity of hepatitis B. Lancet 1982;2:945.
- Pastore G, Monno L, Santantonio T, et al. Hepatitis B virus clearance from serum and liver after acute hepatitis delta virus superinfection in chronic HBsAg carriers. J Med Virol 1990;31:284.
- Szmuness W, Stevens CE, Harley EJ, et al. Hepatitis B vaccine: Demonstration of efficacy in a controlled clinical trial in a high-risk population in the United States. N Engl J Med 1980;303:833.
- Stephenne J. Development and production aspects of a recombinant yeast-derived hepatitis B vaccine. Vaccine 1990;8Suppl:S69.
- Poovorawan Y, Chongsrisawat V, Theamboonlers A, et al. Long-term benefit of hepatitis B vaccination among children in Thailand with transient hepatitis B virus infection who were born to hepatitis B surface antigen-positive mothers. J Infect Dis 2009;200:33.
- Liang X, Bi S, Yang W, et al. Evaluation of the impact of hepatitis B vaccination among children born during 1992-2005 in China. J Infect Dis 2009;200:39.
- Lee C, Gong Y, Brok J, et al. Hepatitis B immunisation for newborn infants of hepatitis B surface antigen-positive mothers. Cochrane Database Syst Rev 2006;CD004790.
- Mast EE, Margolis HS, Fiore AE, et al. A comprehensive immunization strategy to eliminate transmission of hepatitis B virus infection in the United States: Recommendations of the Advisory Committee on Immunization Practices (ACIP) part 1: Immunization of infants, children, and adolescents. MMWR Recomm Rep 2005;54:1.
- Blackard JT, Shata MT, Shire NJ, et al. Acute hepatitis C virus infection: A chronic problem. Hepatology 2008;47:321.
- Armstrong GL, Alter MJ, McQuillan GM, et al. The past incidence of hepatitis C virus infection: Implications for the future burden of chronic liver disease in the United States. Hepatology 2000;31:777.
- Marcellin P. Hepatitis C: The clinical spectrum of the disease. J Hepatol 1999;31Suppl 1:9.
- Chu CM, Yeh CT, Liaw YF. Fulminant hepatic failure in acute hepatitis C: Increased risk in chronic carriers of hepatitis B virus. Gut 1999;45:613.
- Maheshwari A, Thuluvath PJ. Management of acute hepatitis C. Clin Liver Dis 2010;14:169.
- Santantonio T, Sinisi E, Guastadisegni A, et al. Natural course of acute hepatitis C: A long-term prospective study. Dig Liver Dis 2003;35:104.
- Corey KE, Ross AS, Wurcel A, et al. Outcomes and treatment of acute hepatitis C virus infection in a United States population. Clin Gastroenterol Hepatol 2006;4:1278.
- Nachnani JS, Rao GA, Bulchandani D, et al. Predictors of hematological abnormalities in patients with chronic hepatitis C treated with interferon and ribavirin. Ann Hematol 2010;89:121.
- Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med 2002;347:975.
- Fried MW. Side effects of therapy of hepatitis C and their management. Hepatology 2002;36:S237.
- Berk DR, Mallory SB, Keeffe EB, et al. Dermatologic disorders associated with chronic hepatitis C: Effect of interferon therapy. Clin Gastroenterol Hepatol 2007;5:142.
- Vujosevic S, Tempesta D, Noventa F, et al. Pegylated interferon-associated retinopathy is frequent in hepatitis C virus patients with hypertension and justifies ophthalmologic screening. Hepatology 2012;56:455.
- Bichko V, Netter HJ, Wu TT, et al. Pathogenesis associated with replication of hepatitis delta virus. Infect Agents Dis 1994;3:94.
- Smedile A, Farci P, Verme G, et al. Influence of delta infection on severity of hepatitis B. Lancet 1982;2:945.
- Caredda F, d’Arminio Monforte A, Rossi E, et al. Prospective study of epidemic delta infection in drug addicts. Prog Clin Biol Res 1983;143:245.
- Smedile A, Dentico P, Zanetti A, et al. Infection with the delta agent in chronic HBsAg carriers. Gastroenterology 1981;81:992.
- Rasshofer R, Choi SS, Wolfl P, et al. Inhibition of HDV RNA replication in vitro by ribavirin and suramin. In: Viral Hepatitis and Liver Disease. Hollinger FB, Lemon SM, Margolis HS, eds. Williams & Wilkins: Baltimore; 1991:659.
- Balayan MS, Andjaparidze AG, Savinskaya SS, et al. Evidence for a virus in non-A, non-B hepatitis transmitted via the fecal-oral route. Intervirology 1983; 20:23.
- Quintana A, Sanchez L, Larralde O, et al. Prevalence of antibodies to hepatitis E virus in residents of a district in Havana, Cuba. J Med Virol 2005;76:69.
- Emerson SU, Purcell RH. Running like water — the omnipresence of hepatitis E. N Engl J Med 2004;351:2367.
- Ijaz S, Arnold E, Banks M, et al. Non-travel-associated hepatitis E in England and Wales: Demographic, clinical, and molecular epidemiological characteristics. J Infect Dis 2005;192:1166.
- Chauhan A, Jameel S, Dilawari JB, et al. Hepatitis E virus transmission to a volunteer. Lancet 1993; 341:149.
- Lee WM, Brown KE, Young NS, et al. Brief report: No evidence for parvovirus B19 or hepatitis E virus as a cause of acute liver failure. Dig Dis Sci 2006;51:1712.
- Patra S, Kumar A, Trivedi SS, et al. Maternal and fetal outcomes in pregnant women with acute hepatitis E virus infection. Ann Intern Med 2007;147:28.
- Kumar Acharya S, Kumar Sharma P, Singh R, et al. Hepatitis E virus (HEV) infection in patients with cirrhosis is associated with rapid decompensation and death. J Hepatol 2007;46:387.
- Kamar N, Selves J, Mansuy JM, et al. Hepatitis E virus and chronic hepatitis in organ-transplant recipients. N Engl J Med 2008; 358:811.
- Takahashi M, Kusakai S, Mizuo H, et al. Simultaneous detection of immunoglobulin A (IgA) and IgM antibodies against hepatitis E virus (HEV) is highly specific for diagnosis of acute HEV infection. J Clin Microbiol 2005;43:49.
- Aggarwal R, Jameel S. Hepatitis E. Hepatology 2011;54:2218.
- Kamar N, Izopet J, Tripon S, et al. Ribavirin for chronic hepatitis E virus infection in transplant recipients. N Engl J Med 2014;370:1111.
- Kamar N, Rostaing L, Abravanel F, et al. Pegylated interferon-alpha for treating chronic hepatitis E virus infection after liver transplantation. Clin Infect Dis 2010;50:e30.
- Stravitz RT, Kramer AH, Davern T, et al. Intensive care of patients with acute liver failure: Recommendations of the U.S. Acute Liver Failure Study Group. Crit Care Med 2007;35:2498-2508.
MONOGRAPH: Viral and drug-induced hepatitis are the most common causes of acute liver failure in adults.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
