Acute Knee Pain
Authors
Walter Valesky, MD, Attending Physician, Department of Emergency Medicine, Kings County Hospital Center, Brooklyn, NY.
Philipp Underwood, MD, FAAEM, FACEP, FAAFP, RMSK, Program Director, Sports Medicine Fellowship, Department of Emergency Medicine, North Shore University Hospital, Manhasset, NY; Assistant Professor, Hofstra Northwell School of Medicine.
Peer Reviewer
Brian L. Springer, MD, FACEP, Associate Professor, Department of Emergency Medicine, Wright State University Boonshoft School of Medicine, Dayton, OH.
After the knee has been observed, the examination should begin. The authors prefer that the patient be seated on the examination table with the feet dangling and not touching the ground. If the patient is unable to sit upright due to pain or other factors, examine the patient supine, preferably with the hips flexed and the soles of the feet on the stretcher, forcing the knees to flex to approximately 90 degrees. The anterior knee is examined first, assessing the quadriceps muscles that extend distally at the quadriceps tendon and attach on the superior pole of the patella. Tenderness on this tendon may indicate a quadriceps tendinopathy. If no tendon is felt at the superior patella, and a palpable gap is present, a quadriceps tendon rupture may be present. Pain over the patella can be an indication of patellar fracture. Swelling over the patella may be indicative of a prepatellar bursitis (housemaid’s knee). The examination continues distally to palpate over the patellar ligament (frequently referred to as the patellar tendon), originating at the distal patella and inserting at the tibial tubercle. Tenderness over this area may indicate a patellar tendinopathy or jumper’s knee. Finally, the insertion of the patellar tendon is palpated at the tibial tubercle, where enlargement, swelling, or tenderness may indicate Osgood-Schlatter disease (OSD).
After palpating the knee, and if tolerated by the patient, the examiner can place one hand over the patella and flex and extend the knee, feeling for crepitus in the knee. Movement may elicit pain, which may be indicative of arthropathy of the knee or a fractured patella.1 The clinician can examine the patellofemoral joint by placing posterior-directed pressure over the anterior patella with the knee extended. By applying a grinding-like movement to the patella, pain may be elicited, which is seen with patellofemoral arthritis or patellofemoral pain syndrome. Finally, the patella glide test can be performed by providing a lateral or medial directed force on the patella with the knee flexed to 20 degrees and assessing the degree of laxity of the patella. Medial glide of less than 25% of the width of the patella (one quadrant) is associated with lateral tightness (normal), whereas glide of greater than 75% of the width (three quadrants) in either direction is abnormal laxity and hypermobility that can predispose to patellar dislocation.2
The medial and lateral aspects of the knee are then examined. Pain over the medial joint space is indicative of possible meniscal injury or osteoarthritis. The bony prominences above and below the joint line should then be palpated for potential injury to the femoral condyle or tibial plateau, respectively. On the lateral aspect of the knee, the fibular head can be palpated by moving inferior and posterior from the lateral tibial plateau. The fibular head is situated at the level of the tibial tubercle.3 The leg then should be assessed for injury to the medial collateral ligament (MCL) or the lateral collateral ligament (LCL) by applying gentle valgus and varus stress to the knee, respectively, when flexed to 30 degrees.4 The degree of joint space opening, or gapping, may be used to assess the severity of injury to the collateral ligaments. Grade 1 tears (partial) have a joint opening of less than 5 cm, grade 2 have 5-10 cm, and grade 3 (complete tears) have a joint opening of more than 10 cm. Pain may prevent full assessment of the injury.
The posterior aspect of the knee is palpated for swelling and/or tenderness. Tenderness of the popliteal vein, which lies centrally or slightly lateral, is seen with deep venous thrombosis. More medially, between the semimembranosus tendon and the medial head of the gastrocnemius, is where popliteal cysts often arise, causing swelling and tenderness.
The range of motion of the knee should be assessed, with the typical knee showing motion between 0 and 135 degrees. Limitations in knee flexion or extension may indicate intra-articular pathology or a knee effusion. An effusion can be visualized by comparing the symptomatic knee side-by-side with the contralateral knee. Swelling may be seen superior and medial to the patella in the location of the suprapatellar bursa. Pressure on this space forces the fluid into the joint cavity, causing the patella to feel as if floating, distinguishing intra-articular effusion from prepatellar bursitis.1
As noted above, any complaints of knee pain must include a hip exam and vice versa, especially in the pediatric or geriatric patient, as pain from one joint may radiate to the other. Finally, no extremity exam is complete without assessment of the distal neurovascular status of the affected limb. Simple strength testing is often done in the sitting position. The patient raises the bended knee off the table, then straightens out the knee. Dorsi- and plantar-flexion of the foot tests the lower musculature.
When cruiciate ligament or meniscal injury is suspected, additional tests should be performed. The Lachman test is the most commonly used to assess for rupture of the anterior cruciate ligament (ACL), with one meta-analysis showing sensitivities and specificities of 0.81 and 0.81.5 This technique is performed with the patient supine and the knee flexed to 30 degrees. The examiner places one hand on the anterior distal thigh and the other hand on the anterior proximal tibia. The examiner pulls the tibia toward the examiner. Laxity (more than 2 mm of movement) or an endpoint that is “soft” compared to the contralateral knee is a positive test.
The anterior drawer test is another test to detect ACL rupture. The concerning knee is bent to 90 degrees, with the patient supine and the sole of the foot resting on the exam table. The examiner then sits or holds on the dorsum of the foot and applies an anterior force on the proximal tibia. This is then compared to the contralateral leg. A positive test is usually identified as laxity and inability to feel a firm endpoint. Although the specificity is similar to the Lachman test, it has a poor sensitivity of 0.38.5 The posterior drawer test is similar to the anterior drawer test, but the force on the tibia is directed in the posterior direction so that it may be used to detect injury to the posterior cruciate ligament. Like the anterior drawer test, laxity or a soft endpoint is considered positive.
Meniscal injuries should be considered in the patient with traumatic and nontraumatic knee pain. When these injuries are suspected, the most commonly used test is the McMurray test. However, the testing characteristics are poor, with one systematic review reporting a sensitivity of 0.55 and a specificity of 0.77.6 With the patient supine, the knee is flexed to greater than 90 degrees, and the clinician extends the knee with one hand on the foot and the other hand on the joint line of the knee. While the knee is extended, rotate the leg externally with the hand on the foot while the other hand applies a valgus force onto the knee. A positive test is noted with pain, clicking, or locking of the knee.1,7
Popliteal Cysts
Popliteal cysts, or Baker’s cysts as they are commonly called, are just one of many types of cystic lesions around the knee that can cause posterior knee and calf pain. The gastrocnemius-semimembranosus bursa is most commonly involved, lying between the medial head of the proximal gastrocnemius muscle and the semimembranosus tendon.8 The cysts form from degeneration or a “weak area” of the posterior joint capsule located between the two sections of the semimembranosus muscle medially (the oblique popliteal ligament and the popliteus muscle) and the posterior cruciate ligament (PCL) laterally.9 In other cases, a valve-like mechanism allows unidirectional flow from the joint into the communicating bursa due to the overlying gastrocnemius, semimembranosus, and semitendinosus tendons.10 Flexion of the knee opens this valve, allowing flow from the joint capsule to bursa, while extension of the knee causes the overlying tendons to become taut and compress the communication to the bursa.11,12
A popliteal cyst is usually associated with underlying joint pathology. In one series, popliteal cysts were associated with joint pathology in 94% of patients undergoing magnetic resonance imaging (MRI), with common findings being meniscal tears, chondral lesions, and ACL tears.13,14
However, popliteal cysts in pediatric patients do not have associated pathology, and most resolve spontaneously.15,16 Very rarely, these cysts are associated with other pathology such as osteochondritis dessicans (OCD) or juvenile rheumatoid arthritis. There is a single case report of intra-articular infection in a patient with a popliteal cyst. De Maeseneer et al reported that 6.3% of children referred for MRI for knee pain had a popliteal cyst, but no meniscal or ACL tears were noted.17
Clinically, patients with popliteal cysts will present with vague, posterior knee pain and a sensation of fullness in the calf or back of the knee. There may be a palpable mass present in the medial aspect of the posterior extended knee consistent with the location of the gastrocnemius-semimembranosus bursa. The examination should be repeated with the knee in flexion, as the cyst may be present with extension but may reduce in size with flexion to 45 degrees (Foucher’s sign). In extension,the overlying semimembranosus and gastrocnemius muscles relax and allow the cystic fluid to move into the joint.18 If the popliteal cyst ruptures (pseudothrombophlebitis), patients will complain of calf pain and swelling. This may be accompanied by ecchymosis of the calf or into the foot as the synovial fluid dissects down the fascial planes of the lower extremity. Because popliteal cysts and deep venous thrombosis are difficult to distinquish by symptoms or physical exam, imaging with duplex ultrasound should be strongly considered. On ultrasound, a popliteal cyst will appear as hypoechoic or anechoic fluid between the semimembranosus and medial gastrocnemius tendons (100% sensitivity, specificity, positive predictive value, and negative predictive value).19
Popliteal cysts in pediatric patients are rare. Ultrasound is generally diagnostic. They generally decrease in size and resolve spontaneously without injection or surgical intervention, but this may take 3-4 years.15 Nonurgent pediatric orthopedic surgery follow-up is advised.
In adult patients, cysts are believed to be secondary to intra-articular pathology. The treatment should be directed at the underlying pathology. An MRI is usually required to diagnose the underlying pathology, so the patient should be referred to an orthopedic surgeon or sports medicine physician. Corticosteroid injection intra-articularly or into the distended bursa itself often is used.20-22 There is limited evidence suggesting that direct injection into the cyst has a better outcome than intra-articular injection.23 Since many studies report follow-up of four weeks,20-23 this timeline should be used to obtain outpatient follow-up by either an orthopedic surgeon or sports medicine provider. Further treatments, such as sclerotherapy or surgical intervention, are outside the scope of practice of the emergency physician.
Jumper’s Knee
Jumper’s knee is a clinical diagnosis describing a symptomatic tendinopathy of the patellar ligament. Most commonly, pain is located at the proximal pole of the patellar ligament, but in some patients it is at the distal aspect of the patellar ligament or even at the distal quadriceps tendon (quadriceps tendinopathy). Although previously attributed to an acute inflammation of the tendon, current practices view this ailment as a degenerative condition due to repetitive mechanical overload of the tendon.
As its name implies, jumper’s knee is seen in athletes involved in jumping activities such as basketball and volleyball, with elite players in these sports showing a prevalence of 45% and 32%, respectively.24 Patients usually have no history of trauma and complain of insidious onset of anterior knee pain most often localized to the infrapatellar region. Initially, these symptoms may be worse either during or after sporting events, and may improve with rest. With time they become continuous and exacerbated by any movement that causes increased loading on the patellar ligament, even with minor activities of daily living.25,26 The physician should be cautious about a diagnosis of jumper’s knee if the patient describes catching or locking of the knee, swelling, or numbness/tingling in the knee. These symptoms typically are seen with other pathology, such as meniscal tears, and may warrant more investigative measures for diagnosis.
On physical exam, there is tenderness over the patellar ligament, most commonly over the inferior pole of the patella. Some patients experience tenderness over the distal patellar ligament or even the distal quadriceps tendon if the symptoms are related to a tendinopathy of the quadriceps tendon. An orthopedist will often diagnose this injury using a single-leg decline squat test. The patient stands on a 25-degree decline board, and the maximum angle of knee flexion that can be achieved until pain is compared to the contralateral knee.27 In time, patients may develop quadriceps wasting in the vastus medius obliquus, diminished thigh circumference, and calf muscle atrophy.28
Plain radiographs generally are not helpful unless the provider is concerned with other possible pathology. These patients rarely have acute trauma, so clinical decision rules, such as the Ottawa knee rule, are not applicable.29 However, if the patient’s symptoms have been chronic, then X-rays may be taken to exclude malignancies such as osteosarcoma.30,31 Ultrasound imaging or MRI may be useful to diagnose patellar tendinopathy.32 Increasingly, ultrasound is used; however, its accuracy may be operator-dependent. Neovascularization, abnormal tendon thickening at the point of maximal tenderness, and areas of hypoechogenicity are seen on ultrasound imaging.33
Initial treatment of jumper’s knee should focus on symptomatic treatments, such as pain control with nonsteroidal anti-inflammatory drugs (NSAIDs), and activity modification. Follow-up with an appropriate specialist within four to six weeks should be arranged if no improvement is noted by the patient. The prognosis can be poor; in one study, as many as one-third of patients were unable to return to sports within six months because of symptoms.34 If timely follow-up with an orthopedic surgeon or sports medicine provider is not feasible, specific exercises can be suggested. These include using a 25-degree decline squat as part of a physical therapy program.35 During the course of their physical therapy, these patients may follow-up with a specialist as scheduling permits.
Other treatment modalities used by specialists include extracorporeal shock wave therapy, low intensity pulsed ultrasound, injection therapy, and surgery.
Osgood-Schlatter Disease
Osgood-Schlatter disease (OSD) is a frequent cause of anterior knee pain in the adolescent population. It is seen most commonly 12- to 15-year-old boys and younger in girls.36 More commonly seen in the adolescent athlete, it is exacerbated by impact activities such as running, jumping, and cutting.37 It is most commonly seen unilaterally, but approximately 20-30% of patients will have a bilateral presentation.36
The characteristic age group correlates with the emergence of the tibial apophysis, a secondary ossification center.38 It is this apophysis that becomes avulsed and fragmented, which then causes pain in the anterior knee over the tibial tuberosity. Some authors suggest that a tight rectus femoris muscle or a short patella ligament is a possible cause of this disorder. Lateral knee X-rays may show patella alta or baja.39-41 Further etiologies, such as tibial rotation, also have been used to explain symptoms.42
Typically, patients are adolescents participating in sports. They describe pain over the tibial tuberosity or the distal patellar ligament. The pain often is exacerbated after activity. On examiation, pain is elicited by one- or two-legged squats or knee extension against resistance. There may be an enlarged tibial tubercle that will be tender to palpation. An area of “bony bumps” that are caused by the widening between the tubercle apophysis and the anterior tibia may be the hallmark of chronic OSD.43
Once the diagnosis is suspected, radiographs of the knee should be taken to confirm there is no tibial tubercle fracture and to confirm the symptoms are not from neoplastic disease. In OSD, the radiographs may reveal irregularities of the apophysis, widening of the physis, or separate bone fragments within the distal tendon at the insertion. Bony fragments may be seen in as many as 50% of cases; however, bony fragments must be accompanied by symptoms for the diagnosis of OSD.44 MRI may be done as an outpatient and is used to determine the stage of the disease. Ultrasound provides a quick way to confirm OSD and also can better elucidate the soft tissue structures that may not be adequately visualized on plain films. Typically, the provider will utilize a linear transducer longitudinally and midline to visualize the patellar ligament and its attachment at the tibial tuberosity. Such findings may include a delamination of the internal ossification center resulting in an “igloo-like” deformity of the physeal part of the tibial tuberosity, a fluid collection in the retrotendineal soft tissues representing infrapatellar bursitis, and diffuse thickening of the insertion portion of the patellar ligament.45,46
This disorder is typically self-limited and requires only supportive care, such as NSAIDs, ice, and activity modification and/or rest. When available, the emergency physician may refer for physical therapy with exercises focusing on quadricep, hamstring, and gastrocnemius strengthening. Cast immobilization has been used previously but is discouraged due to muscle atrophy. However, if a patient’s symptoms are severe and have been prolonged, a knee immobilizer for a few days may be considered.36 Multiple studies have shown these modalities achieve a cure rate of approximately 90% within 12-24 months.47-49 In one small case series, the use of an infrapatellar strap demonstrated improvement in pain in 79% of patients after six to eight weeks.50 Corticosteroid injections are not used in the ED, as most patients improve with conservative management. Additionally, they promote the added risk of subcutaneous atrophy and patellar ligament rupture. Orthopedic follow-up is encouraged.
Prepatellar Bursitis
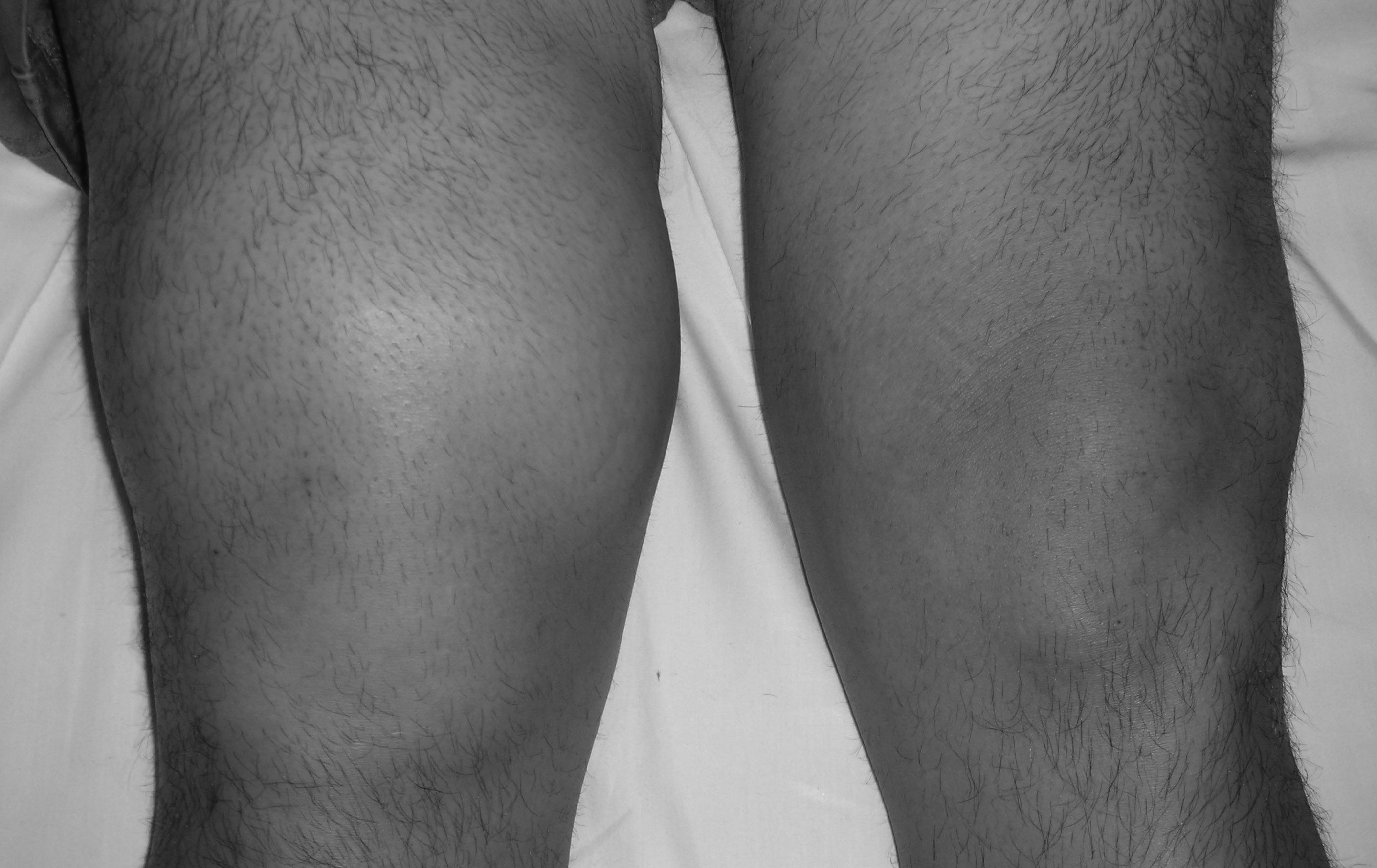
Prepatellar bursitis, or housemaid’s knee, is a common condition, particularly in individuals who spend a lot of time kneeling. It is a similar entity to olecranon bursitis and, as such, is frequently studied with olecranon bursitis in the literature. (See Figure 2.)
Figure 2. Prepatellar Bursitis
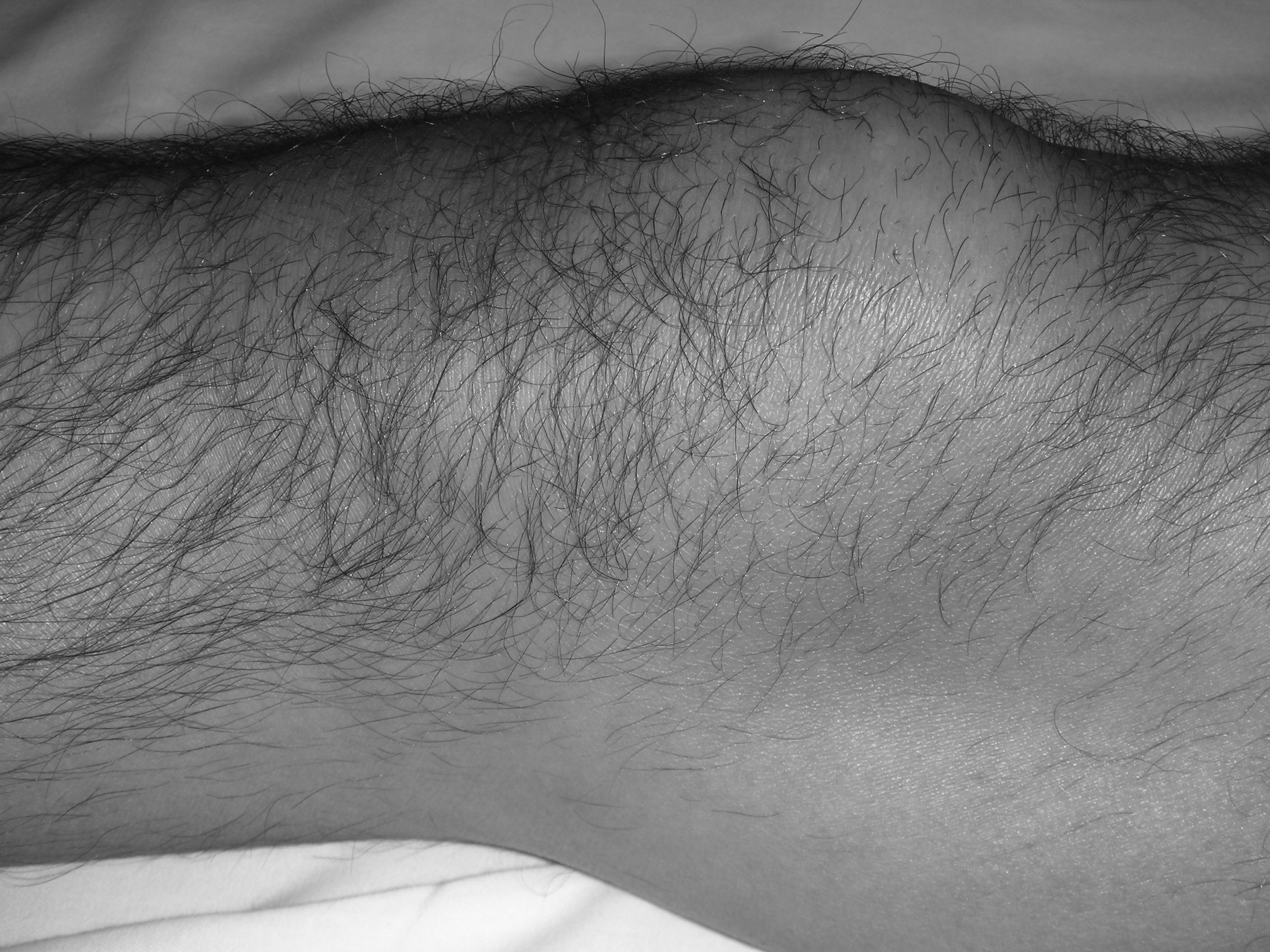
Image courtesy of J. Stephan Stapczynski, MD
Figure 3. Prepatellar Bursitis Aspiration
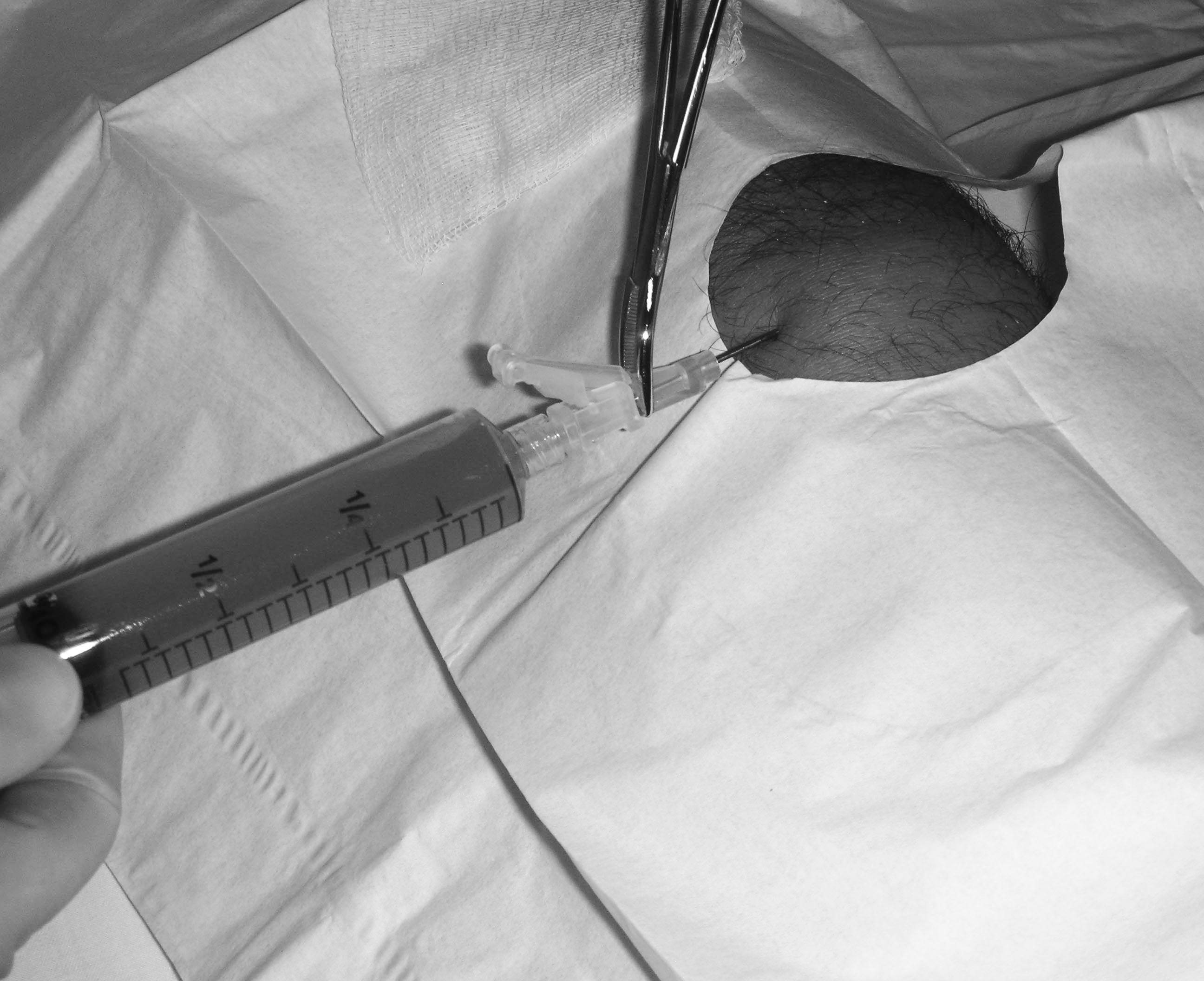
Image courtesy of J. Stephan Stapczynski, MD
The superficial infrapatellar bursa and the prepatellar bursa are the two main bursa surrounding the knee joint. Although they are two separate bursa, they are commonly referred to as the prepatellar bursa. Unlike the suprapatellar bursa, however, they do not communicate with the knee joint and are closed and sac-like.
Prepatellar bursitis is divided into septic bursitis (SB) or nonseptic bursitis (NSB), with NSB being further categorized as idiopathic, traumatic, or crystal-induced. Most patients are male between 40-60 years of age, with two-thirds of cases being NSB.51 NSB is an inflammation of the bursa classically caused by occupational trauma secondary to jobs that require kneeling, such as cleaners, gardeners, roofers, and carpenters. It also may be caused by overuse or direct trauma in athletes, such as wrestlers and volleyball players. It may be caused by systemic conditions such as gout, rheumatoid arthritis, systemic lupus erythematosus, or uremia.51
The emergency provider must distinguish NSB from SB, which is usually a result of direct inoculation into the bursa rather than hematogenous seeding. The most common organism is Staphylococcus aureus. It more commonly affects immunocompromised individuals with risk factors such as alcoholism, diabetes, as well as patients with repeated steroid exposure.52,53
Patients with prepatellar bursitis will present with either acute or chronic swelling over the anterior knee with tenderness to palpation. (See Figure 4.) Although movement of the knee may not cause pain, extreme flexion of the joint may cause pain knee (due to compression of the bursal compartment). Fever, local erythema, and warmth may be present, especially if it is SB. The patient may give a history of acute or repetitive trauma to the area consistent with NSB or prior local cellulitis suggesting SB. Unfortunately, no physical exam finding is specific or sensitive enough to adequately include or exclude an infectious etiology.
Figure 4. Prepatellar Septic Bursitis
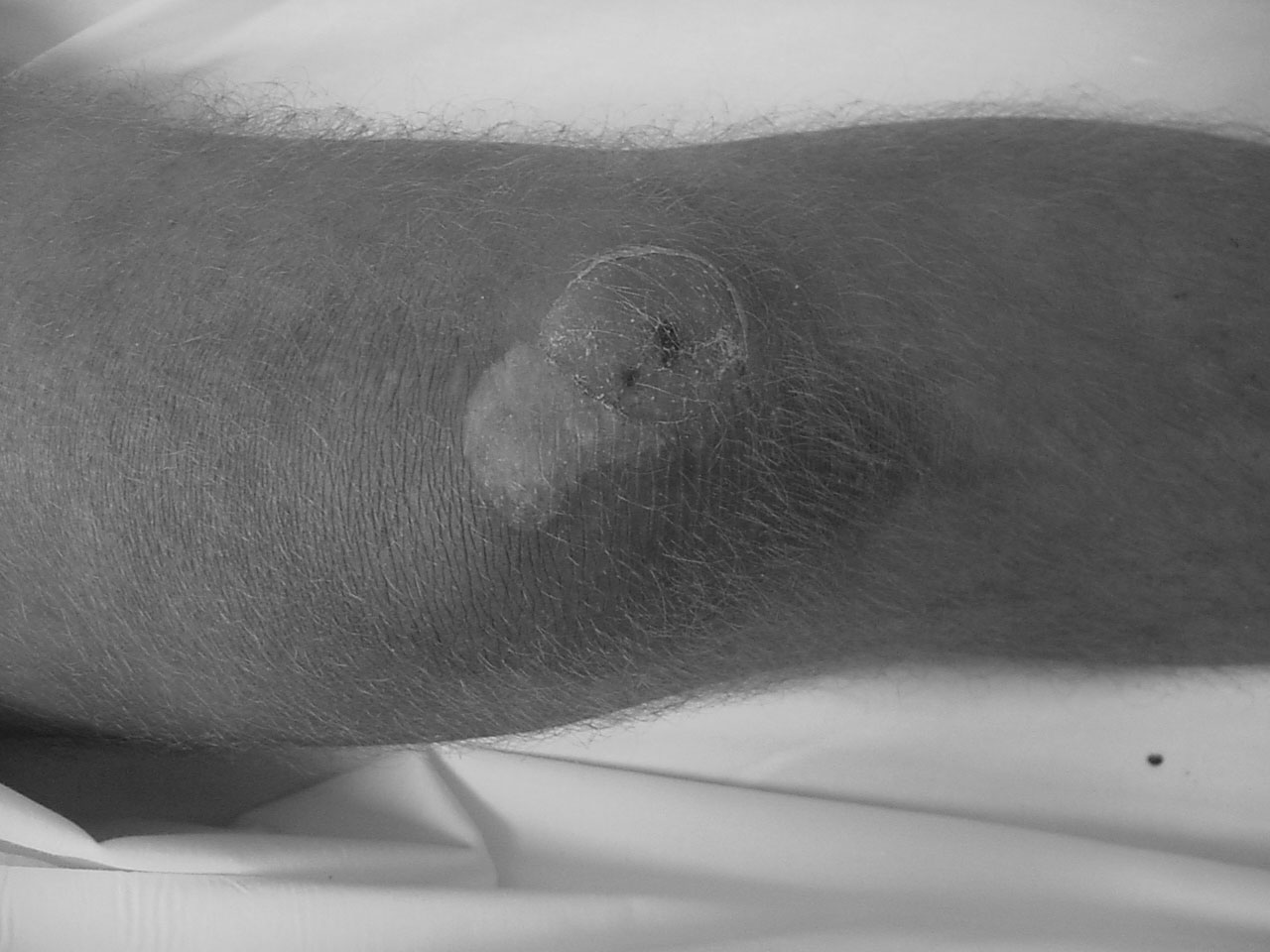
Image courtesy of J. Stephan Stapczynski, MD
If history and physical exam are not sufficient to adequately exclude SB, an aspiration can be attempted. Although some authors advocate for a lateral approach to the bursa for aspiration to prevent sinus tract formation, this has not been formally studied.54 The white cell counts of the fluids are lower than those seen with septic arthritis elsewhere. Cell counts of greater than 3,000 white cells/µL in the prepatellar fluid are sufficient for a diagnosis of septic bursitis. Gram stain and culture should be sent. Other markers of SB may include a fluid glucose level of less than 30 mg/dL or less than 50% of a simultaneous serum glucose.51,53 (See Figure 5.)
Figure 5. Prepatellar Septic Bursitis Aspiration
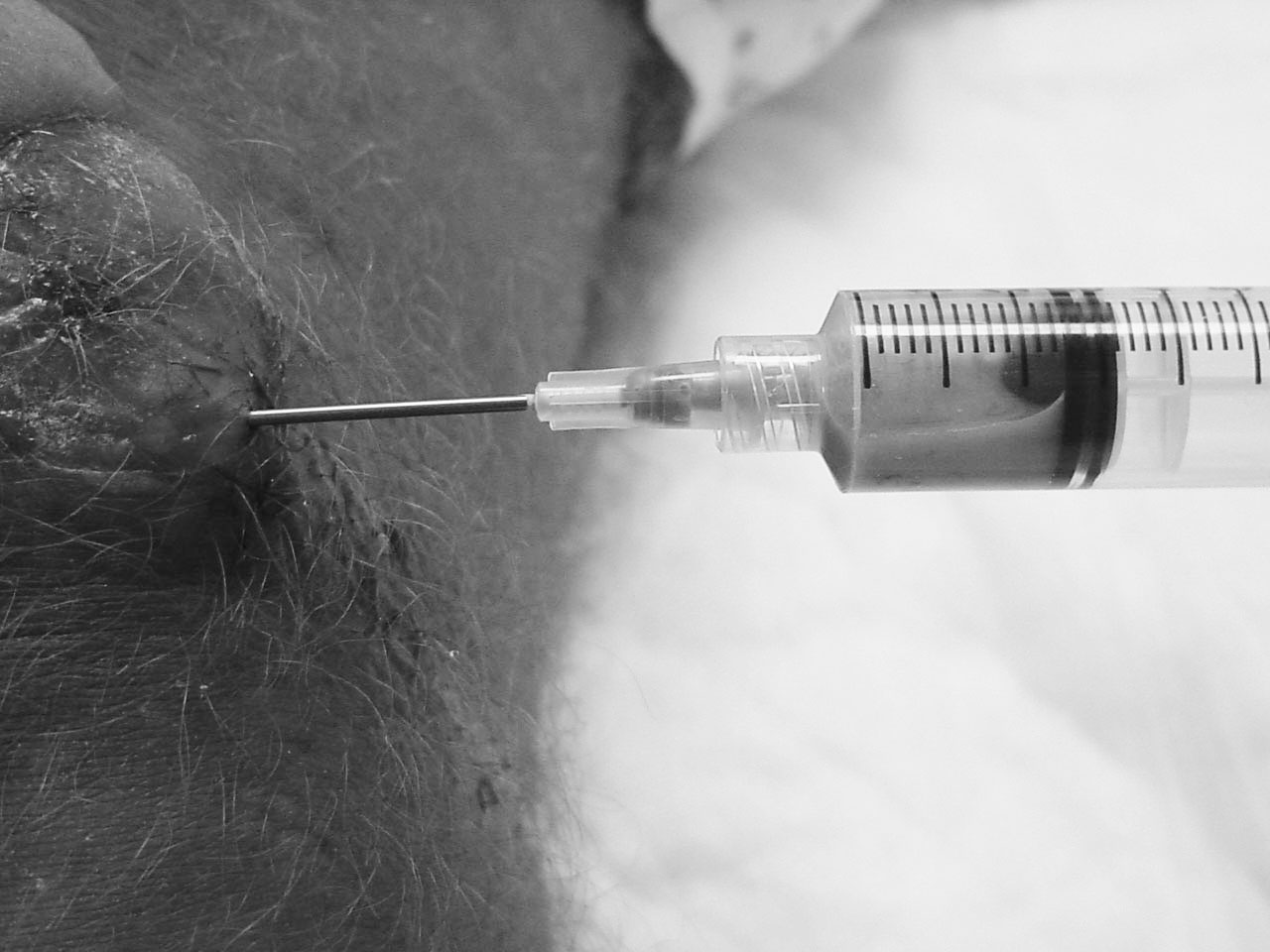
Image courtesy of J. Stephan Stapczynski, MD
Management of these cases is conservative, with most patients with NSB receiving compressive wrapping, elevation, NSAIDs, and therapeutic aspiration as needed. The evidence supporting intrabursal steroids is variable, although many physicians cite anecdotal evidence for its use. The best evidence must be extrapolated from studies on olecranon bursitis. Smith et al enrolled patients with olecranon bursitis to groups with 20 mg methylprednisolone intrabursal injection and oral naproxen 1 g/day for 10 days; 20 mg methylprednisolone intrabursal injection and oral placebo; oral naproxen 1 g/day for 10 days; and oral placebo for 10 days. Sustained improvement at six weeks and decreased aspirations at six months were seen in the groups utilizing an intrabursal injection of methylprednisolone. The author concluded that intrabursal steroids were the “most effective” treatment for nonseptic olecranon bursitis.55 In a retrospective study, Weinstein et al reported similar reductions in swelling when intrabursal steroid injections were used. However, he reported a “high incidence” of steroid-related side effects, such as skin atrophy, subsequent SB in previously NSB patients, and chronic pain.56 Unfortunately, it may difficult to extrapolate these data to prepatellar bursitis. Therefore, intrabursal steroid injections should be used only in those patients for whom conservative management regimens have failed or for those who desire a quicker resolution of symptoms.57 Orthopedic consultation is advisable.
Septic bursitis requires antibiotic therapy, which can be initiated as an outpatient with mild to moderate presentations. Patients who are immunocompromised or who are not able to follow up should be hospitalized. Typical treatment may include a penicillinase-resistant penicillin or a first-generation cephalosporin against S. aureus. However, in areas with increased prevalence of methicillin-resistant S. aureus, empiric therapy targeted toward this bacteria should be considered. Generally, a two-week course has been advised, but there is little evidence to support this strategy. If discharged from the ED, a two-day follow-up should be considered to assess for improvement in symptoms. However, in patients with comorbidities, more severe infection, or who appear ill, intravenous antibiotics and hospital admission are advised.53
Septic bursitis that does not improve with appropriate therapy requires orthopedic consultation for possible incision and debridement.
Osteoarthritis
Osteoarthritis of the knee (OAK) is a chronic condition that is estimated to affect the activity of up to 10% of all adults, and more than 20% of those 65 years of age. Typically, it is seen with greater prevalence in females, non-Hispanic whites, and African Americans.58 Risk factors include obesity, prior knee injury or surgery, and occupational bending and lifting.59 As the knee is made up of three compartments (the medial tibiofemoral, lateral tibiofemoral, and patellofemoral articulation), patients may present with a broad range of symptoms from mild osteoarthritis of one compartment to severe tricompartmental disease.
Clinically, patients present with pain with activity. They may describe pain when walking up or down steps, or with flexion of the knee to perform tasks such as bending or squatting. Patients may report seasonal or temporal variations of pain with OAK, usually described as worse in colder climates. Stiffness that is present upon awakening and that lasts less than 30 minutes also may be described. Frequently, patients may describe the leg “giving way” while walking, which may be a symptom of weakness of the muscles of the thigh and hamstring. Similar symptoms are seen with meniscal tears that may be either acute or degenerative.59
Often, the location of the pain is indicative of the affected compartments. Pain is anteromedial in medial compartment tibiofemoral OAK. It is felt behind the patella in patellofemoral OAK, and anterolateral in lateral tibiofemoral OAK.60 Often the pain is described as “deep,” or “in the knee.” Symptoms of OAK usually do not cause pain in the posterior knee; pain in that location is more often due to a Baker’s cyst or a deep venous thrombosis. In more progressive OAK, night pain may occur. It may be part of more generalized osteoarthritis, which is more common in the elderly, obese, or those with excess joint use. Radiation of symptoms may occur, as multiple joints may be affected, most notably the hip.
Physical findings include crepitus upon flexion and extension of the knee. The tibiofemoral joint lines should be palpated with the knee flexed to 90 degrees; this is most easily performed when the patient is sitting. Joint line pain also may be appreciated in those with OAK, with the medial aspect affected more commonly than the lateral joint line. Furthermore, as many as 36% of patients with radiographically diagnosed OAK may have a suprapatellar effusion, with half of them showing quadriceps atrophy.61
A diagnosis of OAK can be made on the basis of physical examination and history. Most cases do not require repetitive radiographs in the ED.
X-rays can be considered when there is a history of recent trauma or when pain suddenly becomes worse. Radiographs often do not correlate with the patient’s symptom severity.61,63 Patients with knee pain are found to have radiographic OAK in 15-76% of cases.63 Radiographic findings of OAK are less sensitive than those of MRI.61 (See Figure 6.)
Figure 6. Osteoarthritis Knee Radiograph
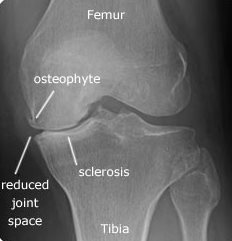
Image courtesy of J. Stephan Stapczynski, MD
Ultimately, when OAK progresses to a point where there is substantial cartilage erosion of the knee joint, or the patient experiences unremitting pain, total knee arthroplasty is a cost-effective treatment strategy.64 However, the majority of patients with OAK either are not ready for the procedure psychologically or their symptoms are manageable with conservative therapy. Initial treatment in the ED is symptomatic pain relief with NSAIDs or tramadol.65 Topical or oral non-narcotic medication are considered excellent first-line medications, although oral acetaminophen has been given an “inconclusive” or “uncertain“ recommendation because of concerns over toxicity.65,66 Physical therapy as an outpatient may be initiated in some EDs to expedite care. It has been shown that comprehensive programs combining strengthening, flexibility, and aerobic exercises provide the greatest probability of benefit.65,67 Additionally, valgus unloader braces and lateral wedge insoles have shown some benefit in patients with predominantly medial tibiofemoral arthritis, although the literature is mixed.65,66 Intra-articular steroid injections can be used for acute osteoarthritis flares. Two meta-analyses show the benefit of steroid injections with improvement in symptoms at short time intervals of two and four weeks.68,69 For this injection, the authors used 40 mg of either triamcinolone or methylprednisoloine mixed with 6-7 cc of 1% lidocaine without epinephrine. This was injected into the anteromedial or anterolateral joint space under sterile conditions.
Patients should follow up with their primary care provider, a sports medicine provider, or orthopedic surgeon. If physical therapy can be arranged from the ED, patients may not require follow-up until four to eight weeks later.
Pseudogout
Calcium pyrophosphate deposition disease (CPPD) or “pseudogout” is an understudied and underreported disorder most commonly affecting the knees, wrists, symphisis pubis, and hips.70 It occurs almost exclusively in the articular tissue affecting both the hyaline and fibrocartilage, and is the most common cause of chondrocalcinosis.71 In a recent study of the prevalence of musculoskeletal disorders, CPPD was the third most common inflammatory arthritis behind rheumatoid arthritis and gout (0.46% each), with a prevalence of 0.42%.72 This disease is rare before the age of 50 years, when the prevalence of CPPD is closer to 10% of the population.70 The true incidence is unknown, as the diagnosis requires arthrocentesis.73,74
Clinically, patients affected by CPPD present with knee pain, often identical to those with OAK.71 The presentation may resemble that of other crystal arthropathies with rapid development of pain, redness, stiffness, swelling, and effusion. The clinical course may be self-limiting, typically lasting three to four days and may commonly be regarded as a simple “arthritis flare.”71 In comparison with isolated osteoarthritis, CPPD may involve radiocarpal joints, shoulders, and elbows. It also tends to have more patellofemoral compartment involvement than osteoarthritis.71 CPPD is associated with many metabolic syndromes, such as hyperparathyroidism, hemochromatism, and hypomagnesemia, as well as previous joint injury.71 CPPD may follow arthroscopic lavage of the knee in patients with pre-existing chondrocalcinosis.75
The clinician may decide to obtain plain films of the knee depending on the clinical scenario. These films may show chondrocalcinosis, but the absence of this finding doesn’t exclude CPPD. (See Figure 7.) Additional findings may include patellofemoral compartment involvement, joint space narrowing, subchondral cysts, and osteophytes.76
Figure 7. Chondrocalcinosis Knee
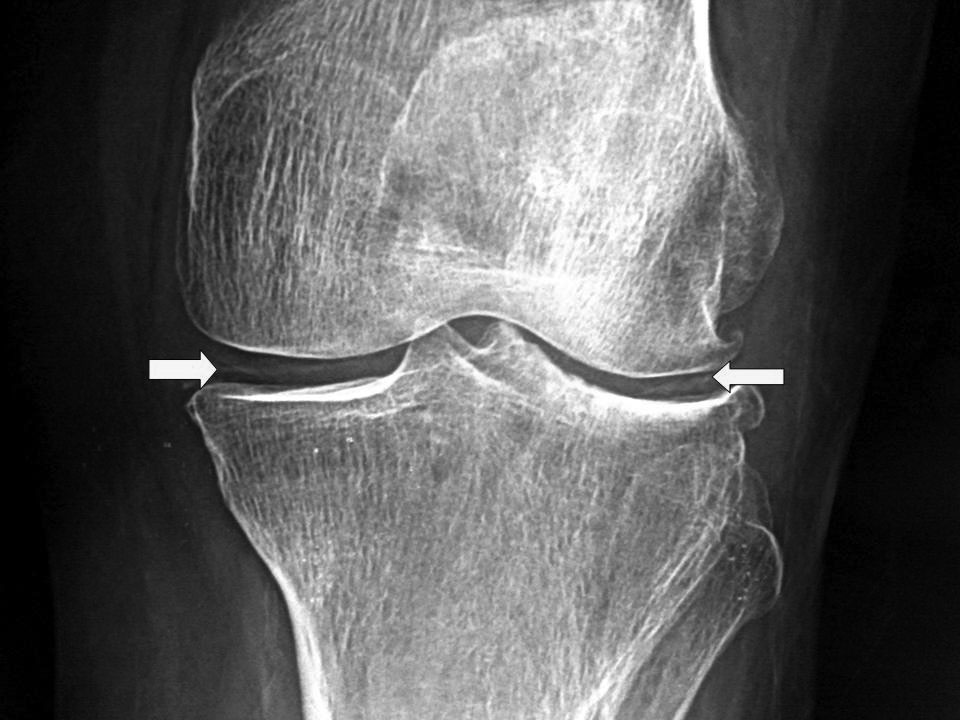
Image courtesy of J. Stephan Stapczynski, MD
The gold standard for diagnosis is joint aspiration and detection of rhomboid-shaped or parallelepedic crystals under polarizing light microscopy. The differential of CPPD includes septic arthritis (which may be seen in conjunction with CPPD), inflammatory arthritis (including gout and rheumatoid arthritis), and gonococcal arthritis. Joint aspiration is important to distinguish these disorders. Calcium pyrophosphate crystals are small and are much less bright than monosodium urate crystals, which are negatively birefringent. It also has been noted that some crystals lack birefringence.77,78 Fluid generally is sent for Gram stain and culture.
The first line of treatment of acute CPPD is NSAIDs and colchicine. The doses used are identical to those for acute gouty arthritis. As traditional methods of colchicine dosing almost certainly lead to side effects, newer expert opinion now recommends 0.5 mg up to three times daily with or without a 1 mg loading dose.79 Corticosteroids are an alternative in patients who may not tolerate NSAIDs. One small study showed equivalent efficacy between a single dosing of 7 mg intramuscular betamethasone or 125 mg intravenous methylprednisolone and diclofenac.80 Intra-articular steroids or oral steroids also may be used depending on patient preference. Although currently beyond the scope of practice of emergency medicine, interleukin-1 inhibitors have been used in acute and chronic CPP inflammatory arthritis.79
REFERENCES
- Orndorff D, Hart J, Miller M. Physical examination of the knee. Curr Sports Med Rep 2005;4:243-248.
- Lester J, Watson J, Hutchinson M. Physical examination of the patellofemoral joint. Clin Sports Med 2014;33:403-412.
- Hoppenfeld S. Physical Examination of the Spine and Extremities, 1st ed. Upper Saddle River, NJ: Prentice Hall; 1976.
- Miyamoto RG, Bosco JA Sherman OH. Treatment of medial collateral ligament injuries. J Am Acad Orthop Surg 2009;17:152-161.
- van Eck CF, van den Bekerom MP, Fu FH, et al. Methods to diagnose acute anterior cruciate ligament rupture: A meta-analysis of physical examination with and without anesthesia. Knee Surg Sports Traumatol Arthrosc 2013;21:1895-1903.
- Meserve B, Cleland JA, Boucher TR. A meta-analysis examining clinical test utilities for assessing meniscal injury. Clin Rehabil 2008;22:143-161.
- Shrier I, Boudier-Reveret M, Fahmy K. Understanding the different physical examination tests for suspected meniscal tears. Curr Sports Med Rep 2010;9:284-289.
- Stein D, Cantlon M, MacKay B, et al. Cysts about the knee: Evaluation and management. J Am Acad Orthop Surg 2013;21:469-479.
- Labropoulos N, Shifrin DA, Paxinos O. New insights into the development of popliteal cysts. Br J Surg 2004;91:1313-1318.
- Rauschning W. Anatomy and function of the communication between knee joint and popliteal bursae. Ann Rheum Dis 1980;39:354-358.
- Handy JR. Popliteal cysts in adults: A review. Semin Arthritis Rheum 2001;31:108-118.
- Herman AM, Marzo JM. Popliteal cysts: A current review. Orthopedics 2014;37:e678-e684.
- Sansone V, de Ponti A, Paluello GM, et al. Popliteal cysts and associated disorders of the knee. Critical review with MR imaging. Int Orthop 1995;19:275-279.
- Stone KR, Stoller D, De Carli A, et al. The frequency of Baker’s cysts associated with meniscal tears. Am J Sports Med 1996;24:670-671.
- Akagi R, Saisu T, Segawa Y, et al. Natural history of popliteal cysts in the pediatric popluation. J Pediatr Orthop 2013;33:262-268.
- Dinham J. Popliteal cysts in children. The case against surgery. J Bone Joint Surg Br 1975;57:69-71.
- De Maeseneer M, Debaere C, Desprechins B, et al. Popliteal cysts in children: Prevalence, appearance and associated findings at MR imaging. Pediatr Radiol 1999:29:605-609.
- Canoso J, Goldsmith M, Gerzof S, et al. Foucher’s sign of the Baker’s cyst. Ann Rheum Dis 1987;46:228-223.
- Ward E, Jacobson J, Fessell D, et al. Sonographic detection of Baker’s cysts: Comparison with MR imaging. AJR Am J Roentgenol 2001;176:373-380.
- Acebes J, Sanchez-Pernaute O, Diaz-Oca A, et al. Ultrasonographic assessment of Baker’s cysts after intra-articular corticosteroid injection in knee osteoarthritis. J Clin Ultrasound 2006;34:113-117.
- Di Sante L, Paoloni M, DiMaggio M, et al. Ultrasound-guided aspiration and corticosteroid injection compared to horizontal therapy for treatment of knee osteoarthritis complicated with Baker’s cyst: A randomized, controlled trial. Eur J Phys Rehabil Med 2012;48:561-567.
- Koroglu M, Callioglu M, Eris H, et al. Ultrasound guided percutaneous treatment and follow-up of Baker’s cyst in knee osteoarthritis. Eur J Radiol 2012;81:3466-3477.
- Bandinelli F, Fedi R, Generini S. Longitudinal ultrasound and clinical follow-up of Baker’s cysts injection with steroids in knee osteoarthritis. Clin Rheumatol 2012;31:727-731.
- Lian O, Engebretsen L, Bahr R. Prevalence of jumper’s knee among elite athletes from different sports: A cross-sectional study. Am J Sports Med 2005;33:561-567.
- Hyman G. Jumper’s knee in volleyball athletes: Advancements in diagnosis and treatment. Curr Sports Med Rep 2008;7:296-302.
- Christian R, Rossy W, Sherman O. Patellar tendinopathy — recent developments toward treatment. Bull Hosp Jt Dis 2014;72:217-224.
- Rudavsky A, Cook J. Physiotherapy management of patellar tendinopathy (jumper’s knee). J Physiother 2014;60:122-129.
- Cook J, Khan K, Maffulli N, et al. Overuse tendinosis, not tendonitis: Part 2: Applying the new approach to patellar tendinopathy. Phys Sports Med 2000;28:31-46.
- Bulloch B, Neto G, Lim R, et al. Validation of the Ottawa knee tule in children: A multicenter study. Ann Emerg Med 2003;42:48-55
- Krych A, Odland A, Rose P, et al. Oncologic conditions that simulate common sports injuries. J Am Acad Orthop Surg 2014;22:223-234.
- Muscolo D, Ayerza M, Makino A, et al. Tumors about the knee misdiagnosed as athletic injuries. J Bone Joint Surg Am 2003;85:1209-1214.
- Warden S, Kiss Z, Malara F. Comparative accuracy of mangetic resonance imaging and ultrasonography in confirming clinically diagnosed patellar tendinopathy. Am J Sports Med 2007;35:427-435.
- Hoksrud A, Ohberg L, Alfredson H, et al. Color Doppler ultrasound findings in patellar tendinopathy (jumper’s knee). Am J Sports Med 2008;36:1813-1820.
- Cook J, Khan K, Harcourt P, et al. A cross sectional study of 100 athletes with jumper’s knee managed conservatively and surgically. Br J Sports Med 1997;31:332-336.
- Purdam C, Johnsson P, Alfredson H, et al. A pilot study of the eccentric decline squat in the management of painful chronic patellar tendinopathy. Br J Sports Med 2004;38:395-397.
- Gholve P, Scher D, Khakharia S, et al. Osgood Schlatter syndrome. Curr Opin Pediatr 2007;19:44-50.
- Soprano J. Musculoskeletal injuries in the pediatric and adolescent athlete. Curr Sports Med Rep 2005;4:329-334.
- Ehrenborg G, Lagergren C. Roentgenologic changes in the Osgood–Schlatter lesion. Acta Chir Scand 1961;121:315–32.
- Aparicio G, Avril JC, Calvo E, et al. Radiologic study of patellar height in Osgood-Schlatter disease. J Pediatr Orthop 1997;17:63-66.
- Jakob R, Von Gumppenberg S, Engelhardt P. Does Osgood-Schlatter disease influence the position of the patella? J Bone Joint Surg Br 1981;63B:579-582.
- Lancourt JE, Cristini JA. Patella alta and patella infera. Their etiological role in patellar dislocation, chondromalacia and apophysitis of the tibial tubercle. J Bone Joint Surg Am 1975;57:1112-1115.
- Gigante A, Bevilacqua C, Bonetti M, et al. Increased external tibial torsion in Osgood-Schlatter disease. Acta Orthop Scand 2003;74:431-436.
- Gerbino G. Adolescent anterior knee pain. Oper Tech Sports Med 2006;14:203-211.
- Davis K. Imaging pediatric sports injuries: Lower extremity. Radiol Clin North Am 2010;48:1213–1235.
- Czyrny Z. Osgood-Schlatter disease in ultrasound diagnostics — a pictoral essay. Med Ultrason 2010;12:323-335.
- Blankstein A, Cohen I, Heim M, et al. Ultrasonography as a diagnostic modality in Osgood Shlatter disease: A clinical study and review of the literature. Arch Orthop Trauma Surg 2001;121:536-539.
- Beovich R, Fricker P. Osgood Schlatter’s disease. A review of the literature and an Australian series. Aust J Sci Med Sport 1988; 20:11-13.
- Hussain A, Hagroo G. Osgood Schlatter disease. Sports Exer Injury 1996;2:202-206.
- Mital MA, Matza RA, Cohen J. The so-called unresolved Osgood Schlatter lesion. J Bone Joint Surg Am 1980;62:732-739.
- Levine J, Kashyap S. A new conservative treatment of Osgood-Schlatter disease. Clin Orthop 1981;158:126-128.
- Baumbach SF, Lobo C, Badyine I, et al. Prepatellar and olecranon bursitis: Literature review and development of a treatment algorithm. Arch Orthop Trauma Surg 2014;134:359-370.
- Aaron DK, Patel A, Kayiaros S, et al. Four common types of bursitis: Diagnosis and mangagement. J Am Acad Orthop Surg 2011; 19:359-367.
- Hanrahan JA. Recent Developments in septic bursitis. Curr Infect Dis Rep 2013;15:421-425.
- McAfee J, Smith D. Olecranon and prepatellar bursitis fiagnosis and yreatment. West J Med 1988;149:607-610.
- Smith D, McAfee J, Lucas L, et al. Treatment of nonseptic olecranon bursitis. A controlled, blinded prospective trial. Arch Intern Med 1989;148:2527-2530.
- Weinstein P, Canoso J, Wohlgethan J. Long-term follow-up of corticosteroid injection for traumatic olecranon bursitis. Ann Rheum Dis 1984;43:44-46.
- McFarland E, Mamanee P, Queale W, et al. Olecranon and prepatellar bursitis: Treating acute, chronic, and inflamed. Treatment varies with bursitis, patient activities, and presence of infection. Phys Sports Med 2000;28:40-52.
- Helmick C, Felson D, Lawrence R, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States: Part I. Arthritis Rheum 2008;58:15-25.
- Felson DT. Clinical Practice. Osteoarthritis of the knee. N Engl J Med 2006;254:841-848.
- Abhishek A, Doherty M. Diagnosis and Clinical Presentation of Osteoarthritis. Rheum Dis Clin North Am 2013;39:45-66.
- Cibere J, Zhang H, Thorne A, et al. Association of Clinical Findings With Pre-Radiographic and Radiographic Knee Osteoarthritis in a Population-Based Study. Arthritis Care & Research 2010;62:1691-1698.
- Stiell I, Greenberg G, Wells G, et al. Prospective Validation of A Decision Rule for the Use of Radiography in Acute Knee Injuries. JAMA 1996;275:611-615.
- Bedson J, Croft P. The discordance between clinical and radiographic knee osteoarthritis: A systematic search and summary of the literature. BMC Musculoskeletal Disorder 2008;9:11.
- Losina E, Walensky R, Kessler C, et al. Cost-effectiveness of total knee arthroplasty in the United States: Patient risk and hospital volume. Arch Int Med 2009;169:1113-1121.
- Treatment of Osteoarthritis of the Knee. Evidenced-Based Guideline 2nd Ed. AAOS. Available at: www.aaos.org/research/guidelines/TreatmentofOsteoarthritisoftheKneeGuideline.pdf. Accessed July 6, 2016.
- McAlindon T, Bannuru R, Sullivan M, et al. OARSI Guidelines for the non-surgical management of osteoarthritis. Osteoarthritis Cartilage 2014;22:363-388.
- Uthman OA, van der Windt DA, Jordan JL et al. Exercise for lower limb osteoarthritis: Systematic review incorporating trial sequential analysis and network meta-analysis. BMJ 2013;347:f5555.
- Bannuru R, Natov N, Obadan I, et al. Therapeutic trajectory of hyaluronic acid versus corticosteroids in the treatment of knee osteoarthritis: A systematic review and meta-analysis. Arthritis Rheum 2009;61:1704–1711.
- Bellamy N, Campbell J, Robinson V, et al. Intraarticular corticosteroid for treatment of osteoarthritis of the knee. Cochrane Database Syst Rev 2006;19(2):CD005328.
- Abhishek A, Doherty M. Epidemiology of calcium pyrophosphate crystal arthritis and basic calcium phosphate crystal arthropathy. Rheum Dis Clin North Am 2014;40:177-191.
- Zhang W, Doherty M, Bardin T, et al. European League against Rheumatism recommendations for calcium pyrophosphate deposition. Part I: Terminology and diagnosis. Ann Rheum Dis 2011;70:563-570.
- Salaffi F, De Angelis R, Grassi W, on behalf of the MArche Pain Prevalence Investigation Group (MAPPING) Study. Prevalence of musculoskeletal conditions in an Italian population sample: Results of a community-based study. 1. The MAPPING study. Clin Exp Rheumatol 2005;23:819-828.
- Ledingham J, Regan M, Jones A, et al. Radiographic patterns and associations of osteoarthritis of the knee in patients referred to hospital. Ann Rheum Dis 1993;52:520-526.
- Pattrick M, Hamilton E, Wilson R, et al. Association of radiographic changes of osteoarthritis, symptoms, and synovial fluid particles in 300 knees. Ann Rheum Dis 1993;52:97-103.
- Pasquetti P, Selvi E, Righeschi K, et al. Joint lavage and pseudogout. Ann Rheum Dis 2004;63:1529-1530.
- Sagarin M. Pseudogout. J Emerg Med 2000;18:373-374.
- Rosenthal AK. Update in calcium deposition diseases. Curr Opin Rheumatol 2007;19:158-162.
- Durcan L, Bolster F, Kavanagh, McCarthy G. The structural consequences of calcium crstal depositions. Rheum Dis Clin North Am 2014;40:311-328.
- Zhang W, Doherty M, Pasual E, et al. European League against Rheumatism recommendations for calcium pyrophosphate deposition. Part II: Management. Ann Rheum Dis 2011;70:571-574.
- Werlen D, Gabay C, Vischer T. Corticosteroid therapy for the treatment of acute attacks of crystal-induced arthritis: An effective alternative to nosteroidal anti-inflammatory drugs. Rev Rhum Engl Ed 1996;63:248-254.
MONOGRAPH: The physical exam of the knee in the ED and selected nontraumatic conditions causing knee pain.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.