Noninvasive Ventilation and Acute Respiratory Failure
September 1, 2016
Reprints
AUTHORS
Erin Tromble, MD, University of Arizona, Department of Emergency Medicine and Pediatrics, Tucson
Aaron N. Leetch, MD, Assistant Professor of Emergency Medicine & Pediatrics, Assistant Residency Director, EM and EM/Peds Programs, Banner University Medical Center, Tucson, AZ
PEER REVIEWER
Larry B. Mellick, MD, MS, FAAP, FACEP, Professor of Emergency Medicine, Professor of Pediatrics, Augusta University, Augusta, GA
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Dietrich (editor), Dr. Skrainka (CME question reviewer), Ms. Wurster (nurse planner), Dr. Tromble (author), Dr. Leetch (author), Dr. Mellick (peer reviewer), Ms. Coplin (executive editor) and Ms. Mark (executive editor) report no relationships with companies related to the field of study covered by this CME activity.
EXECUTIVE SUMMARY
- In addition to avoiding upper airway trauma associated with endotracheal intubation, noninvasive ventilation (NIV) allows patients to maintain their own airway clearance and often decreases the need for patient sedation. NIV devices are easier to apply or remove and carry fewer complications when accidentally displaced.
- The positive intrathoracic pressure produced by NIV can lead to hypotension and hemodynamic instability due to decreased cardiac preload with hyper-expanded lungs and positive intrathoracic pressures, although these risks have been shown to be less severe with NIV when compared to invasive mechanical ventilation.
- Conditions in which improved oxygenation is the primary goal and in which the patient’s degree of respiratory distress is less severe may respond well to continuous positive airway pressure (CPAP) alone.
- By convention, both CPAP and the expiratory positive airway pressure setting of bi-level positive airway pressure (which both provide positive end-expiratory pressure [PEEP]) generally are started at 5 cm H2O. This is estimated to be the baseline PEEP in healthy lungs.
- Identifying patients who likely will deteriorate on noninvasive therapy is crucial to avoid delays to intubation. The available pediatric literature suggests several predictors of NIV failure: high severity of illness at presentation as indicated by the Pediatric Risk of Mortality score, individual physiologic markers including high FiO2 requirements and high PaCO2 on admission, inability to wean FiO2 and absence of improvement after 1-2 hours of therapy, and underlying etiology of respiratory compromise (acute respiratory distress syndrome, sepsis, oncologic processes, and immune deficiencies).
For a long time, endotracheal intubation with invasive mechanical ventilation was the only tool available for children with acute respiratory failure. The decision used to be to intubate or not to intubate. The advent of new tools, such as noninvasive ventilation, has provided an incredible resource for avoiding intubation and its complications. Noninvasive ventilation allows patients to maintain their own airway clearance (decreasing ventilator-associated pneumonia rates) and often decreases the need for patient sedation. The authors review noninvasive ventilation indications, contraindications, and the growing body of literature that supports its use in a variety of clinical scenarios.
— Ann M. Dietrich, MD, FAAP, FACEP
Acute respiratory failure, one of the most common medical emergencies encountered by pediatric providers, is defined as failure of adequate gas exchange demonstrated either by PaO2 < 60 mmHg, PaCO2 > 50 mmHg, or both.
Until recently, endotracheal intubation with invasive mechanical ventilation (IMV) has been nearly the only management tool available for acute respiratory failure. However, in recent years, noninvasive ventilation (NIV) has gained significant attention as an alternative to IMV.1-4 Its utility in treating adult respiratory failure, particularly in the management of chronic obstructive pulmonary disease and cardiogenic pulmonary edema, is well established.2,5-10 The evidence supporting NIV use in pediatric populations is less robust, but a growing body of literature supports its use in certain clinical settings.11
Considering risks and benefits, there are several theoretic advantages of NIV over IMV. In addition to avoiding the upper airway trauma associated with endotracheal intubation, NIV allows patients to maintain their own airway clearance (decreasing ventilator-associated pneumonia rates) and often decreases the need for patient sedation.11,12 Additionally, NIV devices are easier to apply or remove and carry far fewer complications when accidentally displaced.
NIV is not without any risks, however. (See Table 1.) Like IMV, NIV can lead to barotrauma and possible pneumothorax.13 It also leaves an unsecured airway and has an increased risk of aspiration compared to IMV if the patient’s mental status does not allow for adequate airway clearance.14 Difficulty fitting NIV devices to achieve an adequate seal is not uncommon, leading to tissue injuries when the fit is too tight and air leaks when the fit is too loose.15,16 Further, like IMV, the positive intrathoracic pressure produced by NIV can lead to hypotension and hemodynamic instability due to decreased cardiac preload with hyper-expanded lungs and positive intrathoracic pressures, although these risks have been shown to be less severe with NIV when compared to IMV.17
Table 1. Potential Complications of Noninvasive Ventilation |
|
|
SOURCE: Author created. |
Mechanism
NIV provides support through several mechanisms. (See Table 2.) Positive pressure helps maintain airway patency and allows improved exhalation in patients with lower airway obstructive processes (e.g., asthma, bronchiolitis) and decreased work of breathing in patients with dynamic upper airway obstruction (e.g., laryngomalacia).11,18-25 Additionally, NIV aids in alveolar recruitment through the addition of positive end-expiratory pressure (PEEP), thus assisting with oxygenation and decreasing V-Q mismatch.11,25,26 Inspiratory positive pressure also decreases the force that respiratory muscles must generate during inhalation, decreasing total energy expenditure.11,25,26 In short, in select pediatric patients, NIV has the potential to improve gas exchange and decrease work of breathing in respiratory failure, thus forestalling or even entirely preventing the deterioration into respiratory arrest.
Table 2. Mechanisms of Respiratory Support Provided by Noninvasive Ventilation |
|
|
SOURCE: Author created. |
Mode
NIV can be employed through a variety of methods, which range from high flow nasal cannula (HFNC) to noninvasive positive pressure ventilation (NIPPV) controlled by a ventilator. All will provide the same overall mechanics but with varying degrees of control and efficacy.
High Flow Nasal Cannula
HFNC has been investigated as an alternative to continuous positive airway pressure (CPAP). The exact definition of HFNC remains somewhat nebulous, but generally it is used to refer to flow rates at or above 1-2 L/minute of oxygen that is warmed, blended, and humidified. Heating and humidifying oxygen replaces some of the physiologic role of the upper airway and is believed to reduce energy expenditure. Studies in preterm infants and neonates have found that HFNC can deliver positive pressures in the range typically administered via CPAP and can have similar physiologic benefits to CPAP.27,28
Further advantages of HFNC include its relative ease of initiation, without requiring use of a ventilator. However, the exact pressure support delivered to the patient cannot be measured directly, offering less control over the level of respiratory support. Higher flow rates also convey a risk of mucosal damage, airway desiccation and bleeding, and excessive nasopharyngeal airway pressures. These risks can be somewhat minimized by ensuring an adequate air leak around the nasal cannula. Lastly, studies demonstrating its efficacy in older children are lacking.27,28
Continuous Positive Airway Pressure
CPAP delivers a constant level of pressure support throughout the respiratory cycle via a ventilator. Consequently, CPAP offers the functional equivalent of PEEP, aiding in alveolar recruitment, oxygenation, and V-Q mismatch.11,16,19
Bi-level Positive Airway Pressure
Alternatively, bi-level positive airway pressure (BiPAP) provides two different levels of pressure, which vary during different phases of respiration. The higher pressure is administered during inspiration and is called inspiratory positive airway pressure (IPAP). The lower pressure, delivered during exhalation, is called expiratory positive airway pressure (EPAP). By setting two pressure modes, BiPAP allows for respiratory support both by creating PEEP (through EPAP) and by assisting respiratory effort by creating a driving pressure (the difference between EPAP and IPAP) with each breath. BiPAP may be synchronized, with each driving pressure administered with the patient’s own spontaneous effort, or unsynchronized, with a fixed back-up respiratory rate if the patient does not initiate a minimum number of spontaneous breaths per minute.11,16,19
Selection of CPAP vs. BiPAP should take into consideration the patient’s full clinical picture, including the underlying condition, current respiratory status, and ability to tolerate the selected mode of ventilation. Conditions in which improved oxygenation is the primary goal and in which the patient’s degree of respiratory distress is less severe may respond well to CPAP alone. Further, the lower continuous pressure provided by CPAP may be easier for some patients to tolerate. In contrast, some patients find the dual pressure of BiPAP easier to tolerate, as it more closely mimics a physiologic respiratory cycle despite its higher peak pressures. BiPAP also confers a greater degree of respiratory support than CPAP and may be more appropriate for those with more severe distress.
Interface Choice
If the decision is made to initiate CPAP or BiPAP, the next step is selection of the interface. These devices can include nasal prong devices, nasal masks, full-face masks, and helmets. (See Table 3.)
Table 3. Interface Advantages and Disadvantages |
||
|
Interface Type |
Advantages |
Disadvantages |
|
Nasal Prongs |
|
|
|
Nasal Mask |
|
|
|
Full-face Mask |
|
|
|
Helmet |
|
|
|
SOURCE: Author created. |
||
Nasal Prongs
Nasal prongs, which are most often used in infants, are small and relatively easy to place. The shorter pronged cannulas are similar to a typical nasal cannula but with a larger gauge to allow for more flow. They fit snugly into the anterior nares but often can leak if not held in place. Other HFNC have longer prongs that must be placed in the posterior nares similar to a nasal trumpet. Both types must be monitored closely to ensure they do not cause pressure sores and erosion of the nares or septum. Additionally, if the patient opens his or her mouth, an air leak is created via the mouth, and the positive pressure delivery to the airways and lungs is lost. Pacifiers and chinstraps are sometimes used in infants to help with this problem.16,29,30
Nasal Masks
Nasal masks, like nasal prongs, are less cumbersome and obtrusive than full-face masks and often are tolerated better. They avoid the risk of erosion to the internal nose and nares caused by nasal prongs but still can result in pressure sores to the face if not fitted and adjusted properly. Like nasal prongs, if patients open their mouths during speech or sleep, positive pressure ventilation is lost.16,29,30
Full-face Masks
Full-face masks, which cover both the nose and mouth, provide more consistent control over the pressure delivered. They administer positive pressure ventilation regardless of the position of the patient’s mouth and provide a complete seal to prevent leaks. However, many patients, especially younger children and infants, develop agitation with full-face masks, and some patients may experience a sensation of claustrophobia. Anxiolysis or mild sedation or dissociation may help the patient cooperate better.16,29,30
Helmets
Helmets also deliver pressure support reliably, avoid the risk of pressure injuries, and tend to be well tolerated.31 These devices are large and often cumbersome to use and may predispose to CO2 rebreathing.31 Additionally, they tend to have limited availability.
Ultimately, the device chosen requires consideration of the individual patient’s needs as well as the availability at each institution. Most facilities carry a limited range of equipment types and sizes. Using an inappropriately fitted device should be avoided, as it can lead to increased complications such as pressure sores from over-tightening an ill-fitting mask and air leaks from poor fit and seal.
Initial Settings
By convention, both CPAP and the EPAP setting of BiPAP (which both provide PEEP) generally are started at 5 cm H2O.16 This is estimated to be the baseline PEEP in healthy lungs. However, the underlying respiratory disorder and oxygenation status of the patient must be considered when choosing these settings.16 Initial IPAP levels often range from 8-10 cm H2O.33
Patient tolerance, hemodynamic status, oxygen status, tidal volumes, and mean airway pressures must be monitored immediately after initiation of NIPPV. Adjustments in pressure often are made in increments of 2 cm H2O at a time. When using BiPAP, IPAP should be titrated to a target tidal volume of 6 to 8 mL/kg. If the patient’s FiO2 level cannot be weaned to appropriate levels, EPAP or CPAP likely will need to be increased, and transition to IMV should be considered.16,33 (See Figure 1.)
Figure 1. Steps to Initiation of Noninvasive Ventilation |
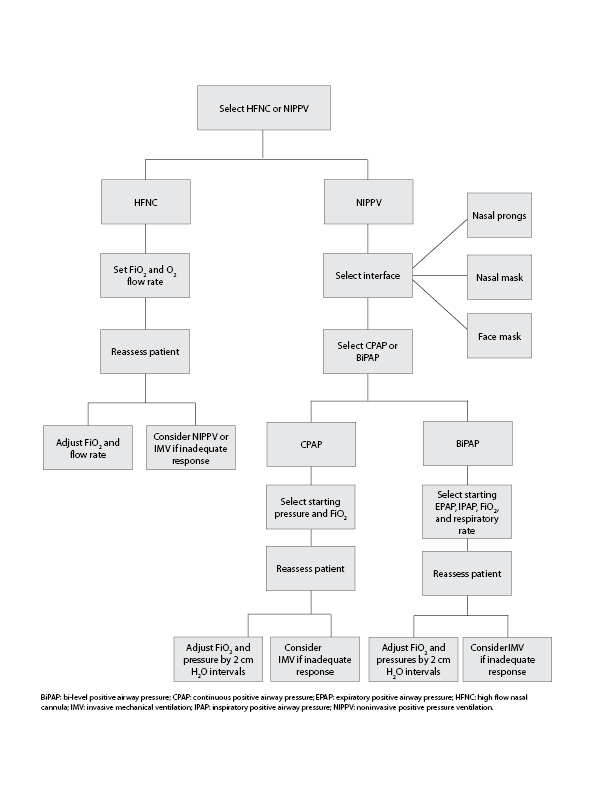 |
|
SOURCE: Author created. |
Initial settings up to EPAP/CPAP of 10 cm H2O are believed to be safe in pediatric populations, and IPAP settings of up to 20 cm H2O are generally accepted.16,34 Inspiratory levels above this should be employed with caution, as nasal and facial masks may lose their seal, air leaks may form, and barotrauma is more likely. High pressures also can overcome lower esophageal sphincter tone, insufflating the stomach, and increasing the risk of aspiration.
Acclimatization of Pediatric Patients to NIV
A unique challenge faced when initiating NIV in pediatric patients is acclimatizing the patient to the therapy. Many adults poorly tolerate positive pressure ventilation and the associated interfaces, which may be augmented further in younger patients. In general, adolescent and older pediatric patients tend to tolerate NIV better than younger patients, although many preterm and younger infants also do well. In patients old enough and alert enough to understand their circumstances, it is important to provide as much orientation and hands-on modeling to NIV as is feasible. It may be helpful to begin with low pressures and gradual increases so that patients can adjust to the sensation of breathing with positive pressure. When possible, providing a trial of different interfaces can increase tolerance based on patient preferences. Intermittent changes of the delivery interface also may help patients tolerate ongoing therapy.16
Indications for Initiation of NIV
As previously discussed, although there is abundant evidence demonstrating the benefits of NIV use in adult patients, the literature is limited for pediatric populations. The majority of pediatric research involves case series with relatively few randomized, controlled trials. One notable prospective randomized, controlled trial, published by Yanez et al in 2008, found NIPPV used for treatment of acute respiratory failure reduced intubation rates and improved physiologic markers of respiratory distress.23
It is best to start NIV in patients who have underlying disease processes that are likely to reverse with its use. Existing evidence suggests that NIV can reduce intubation rates and improve respiratory status in cases of both hypoxemic and hypercapneic respiratory failure.26 Table 4 provides a list of conditions and circumstances that current evidence suggests are responsive to NIV use.19,21-24,35-55
Table 4. Conditions with Possible Benefit from Noninvasive Ventilation |
|
|
SOURCE: Author created. |
In the setting of lower airway obstruction, such as bronchiolitis and asthma, EPAP helps maintain lower airway patency, thus helping decrease lung hyperinflation and auto-PEEP. Additionally, IPAP provides respiratory muscle support and decreases work of breathing. Conversely, patients with dynamic upper airway obstruction, such as croup and laryngo/tracheomalacia, may benefit from upper airway stenting provided via CPAP or IPAP. In parenchymal lung disease, EPAP aids alveolar recruitment and, therefore, assists with oxygenation, while IPAP again appears to improve the work of breathing.19
Chronic neuromuscular disorders represent a common group of diseases among pediatric patients in which NIV is applied routinely. In this population, muscular weakness of both the upper and lower respiratory muscles is the primary mechanism of respiratory failure. As such, positive pressure administered via IPAP serves to support the lower respiratory muscles, while EPAP or CPAP can help maintain patency of the upper airway in the setting of obstructive sleep apnea.35
In apnea of prematurity, NIV offers similar support to those conditions previously discussed via offloading the work of lower respiratory muscles, stenting upper and lower airways, and alveolar recruitment. In this condition, however, NIV is theorized also to stimulate an augmented inspiratory reflex (inspiratory effort triggered by inflation of the lung), which helps to establish a functional residual capacity and maintain alveolar patency. Such an effect is not seen in term infants or older patients. However, patients with apnea must be monitored closely after initiation of noninvasive respiratory support to ensure the apnea appropriately resolves. If these patients continue to have persistent apneic episodes, transition to IMV should be considered.36
Conversely, there is minimal research into the use of NIV in the setting of trauma. To date, the existing evidence in adult literature suggests that there may be some utility to NIV in the setting of blunt chest trauma (e.g., flail chest, pulmonary contusion). In these conditions, positive pressure is believed to minimize alveolar collapse and ease work of breathing. However, clinical judgment remains paramount in the context of trauma, and NIV should not be used in patients with altered mental status, aspiration risk, or facial trauma.53-55
Research also has found that the use of NIV following extubation may reduce rates of re-intubation in pediatric populations, and it also may be used to optimize respiratory and oxygenation status prior to intubation.19,46,58
Contraindications
NIV requires patients to be able to cooperate with machine-delivered breaths and is contraindicated in patients with altered mental status. Additionally, NIV is not appropriate in patients for whom a secure airway is indicated or those who have a risk of airway loss (e.g., expanding neck hematoma; epiglottitis). Further contraindications to NIV use include hemodynamic instability, facial trauma (which may limit ability to fit masks), poor secretion clearance, vomiting, and upper GI bleeds.56 (See Table 5.)
Table 5. Absolute Contraindications to Noninvasive Ventilation |
|
|
SOURCE: Author created. |
Patient selection also should take into account the availability of the necessary supplies and in the appropriate sizes, the ability of the patient to tolerate treatment, and the availability of appropriate support staff (nurses and respiratory therapists) with the capability to manage pediatric NIV.
Predictors of Failure
Identifying patients who likely will deteriorate on noninvasive therapy is crucial to avoid delays to intubation. The available pediatric literature suggests several predictors of NIV failure. (See Table 6.) One such predictor is high severity of illness at presentation as indicated by the Pediatric Risk of Mortality score.19,43,57,58 Individual physiologic markers that have been found to be predictors of failure include high FiO2 requirements and high PaCO2 on admission.19,43,50,58 Inability to wean FiO2 and absence of improvement after 1-2 hours of therapy also may predict failure.47,57,60,61 Lastly, the underlying etiology of respiratory compromise is an important factor. Patients with underlying acute respiratory distress syndrome, sepsis, oncologic processes, and immune deficiencies have been found to have high failure rates on NIV.47,59
Table 6. Predictors of Noninvasive Ventilation Failure |
|
|
Severe disease |
|
|
Poor initial response to therapy |
|
|
Underlying process |
|
|
SOURCE: Author created. |
|
Signs of NIV Failure
After NIV has been initiated, the patient must be reassessed continually for evidence of improved respiratory status vs. signs of therapy failure. Each patient requires individualized evaluation of his or her own clinical circumstances and underlying pathology. As such, multiple variables should be considered when assessing the success or failure of NIV use. Signs of NIV failure may include persistent tachypnea or respiratory distress, inability to wean FiO2 (with target < 0.6), or persistently elevated FiO2 of > 60 mmHg. (See Table 7.) Other physiologic markers of NIV failure may include persistent tachycardia or hypertension. Additionally, clinical tolerance of NIV should be monitored. Patient agitation also suggests a need for escalation of therapy.62,63
Table 7. Markers of Noninvasive Ventilation Failure |
|
|
Source: Author created. |
Conclusion
NIV increasingly is used by both adult and pediatric providers. Although the advantages of NIV in certain populations are clear, appropriate patient selection and understanding of the risks and benefits of noninvasive techniques are paramount to their successful implementation. Further investigation is needed to adequately delineate the appropriate use of NIV in pediatric populations. Nevertheless, NIV continues to develop as a valuable tool in the management of acute and pending respiratory failure in pediatric patients.
REFERENCES
- Fanning JJ, Lee KJ, Bragg DS, Gedeit RG. U.S. attitudes and perceived practice for noninvasive ventilation in pediatric acute respiratory failure. Pediatr Crit Care Med 2011;12:e187-e194.
- Liesching T, Kwok H, Hill NS. Acute applications of noninvasive positive pressure ventilation. Chest 2003;124:699-713.
- Sinuff T, Cook DJ, Randall J, Allen CJ. Evaluation of a practice guideline for noninvasive positive-pressure ventilation for acute respiratory failure. Chest 2003;123:2062-2073.
- Maheshwari V, Paioli D, Rothaar R, Hill NS. Utilization of noninvasive ventilation in acute care hospitals: A regional survey. Chest 2006;129:1226-1233.
- Brochard L, Mancebo J, Wysocki M, et al. Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. N Engl J Med 1995;333:817-822.
- Keenan SP, Sinuff T, Cook DJ, Hill NS. Which patients with acute exacerbation of chronic obstructive pulmonary disease benefit from noninvasive positive-pressure ventilation? A systematic review of the literature. Ann Intern Med 2003;138:861-870.
- Plant PK, Owen JL, Elliott MW. Early use of non-invasive ventilation for acute exacerbations of chronic obstructive pulmonary disease on general respiratory wards: A multicentre randomised controlled trial. Lancet 2000;355:1931-1935.
- Gray A, Goodacre S, Newby DE, et al. Noninvasive ventilation in acute cardiogenic pulmonary edema. N Engl J Med 2008;359:142-151.
- Weng CL, Zhao YT, Liu QH, et al. Meta-analysis: Noninvasive ventilation in acute cardiogenic pulmonary edema. Ann Intern Med 2010;152:590-600.
- Vital FM, Ladeira MT, Atallah AN. Non-invasive positive pressure ventilation (CPAP or bilevel NPPV) for cardiogenic pulmonary edema. Cochrane Database Syst Rev 2008;CD005351.
- Deis JN, Abramo TJ, Crawley L. Noninvasive respiratory support. Pediatr Emerg Care 2008;24:331-338.
- Antonelli M, Conti G, Rocco M, et al. A comparison of noninvasive positive-pressure ventilation and conventional mechanical ventilation in patients with acute respiratory failure. N Engl J Med 1998;339:429-435.
- Hess DR. Noninvasive positive-pressure ventilation and ventilator-associated pneumonia. Respir Care 2005;50:924-931.
- Carroll CL, Zucker AR. Barotrauma not related to type of positive pressure ventilation during severe asthma exacerbations in children. J Asthma 2008;45:421-424.
- Mehta S, Hill NS. Noninvasive ventilation. Am J Respir Crit Care Med 2001;163:540-577.
- Akingbola OA, Hopkins RL. Pediatric noninvasive positive pressure ventilation. Pediatr Crit Care Med 2001;2:164-169.
- Girault C, Briel A, Benichou J, et al. Interface strategy during noninvasive positive pressure ventilation for hypercapnic acute respiratory failure. Crit Care Med 2009;37:124-131.
- Confalonieri M, Gazzaniga P, Gandola L, et al. Haemodynamic response during initiation of non-invasive positive pressure ventilation in COPD patients with acute ventilatory failure. Respir Med 1998;92:331-337.
- Najaf-Zadeh A, Leclerc F. Noninvasive positive pressure ventilation for acute respiratory failure in children: A concise review. Ann Intensive Care 2011;1:15.
- Thia LP, McKenzie SA, Blyth TP, et al. Randomised controlled trial of nasal continuous positive airways pressure (CPAP) in bronchiolitis. Arch Dis Child 2008;93:45-47.
- Carroll CL, Schramm CM. Noninvasive positive pressure ventilation for the treatment of status asthmaticus in children. Ann Allergy Asthma Immunol 2006;96:454-459.
- Beers SL, Abramo TJ, Bracken A, Wiebe RA. Bilevel positive airway pressure in the treatment of status asthmaticus in pediatrics. Am J Emerg Med 2007;25:6-9.
- Yanez LJ, Yunge M, Emilfork M, et al. A prospective, randomized, controlled trial of noninvasive ventilation in pediatric acute respiratory failure. Pediatr Crit Care Med 2008;9:484-489.
- Essouri S, Nicot F, Clement A, et al. Noninvasive positive pressure ventilation in infants with upper airway obstruction: Comparison of continuous and bilevel positive pressure. Intensive Care Med 2005;31:574-580.
- Teague WG. Noninvasive ventilation in the pediatric intensive care unit for children with acute respiratory failure. Pediatr Pulmonol 2003;35:418-426.
- Calderini E, Chidini G, Pelosi P. What are the current indications for noninvasive ventilation in children? Curr Opin Anesthesiol 2010;23:368-374.
- Cummings JJ, Polin RA; Committee on Fetus and Newborn, American Academy of Pediatrics. Noninvasive respiratory support. Pediatrics 2016;137: doi: 10.1542/peds.2015-3758.
- Marohn K, Panisello JM. Noninvasive ventilation in pediatric intensive care. Curr Opin Pediatr 2013;25:290-296.
- Ramirez A, Delord V, Khirani S, et al. Interfaces for long-term noninvasive positive pressure ventilation in children. Intensive Care Med 2012;38:655-662.
- de Carvalho WB, Johnston C. The fundamental role of interfaces in noninvasive positive pressure ventilation. Pediatr Crit Care Med 2006;7:495-496.
- Chidini G, Calderini E, Cesana BM, et al. Noninvasive continuous positive airway pressure in acute respiratory failure: Helmet versus facial mask. Pediatrics 2010;126:e330-e336.
- Taccone P, Hess D, Caironi P, Bigatello LM. Continuous positive airway pressure delivered with a “helmet”: Effects on carbon dioxide rebreathing. Crit Care Med 2004;32:2090-2096.
- Abadesso C, Nunes P, Silvestre C, et al. Non-invasive ventilation in acute respiratory failure in children. Pediatr Rep 2012;4:e16.
- Cavari Y, Sofer S, Rozovski U, Lazar I. Non invasive positive pressure ventilation in infants with respiratory failure. Pediatr Pulmonol 2012;47:1019-1025.
- Benditt JO. Initiating noninvasive management of respiratory insufficiency in neuromuscular disease. Pediatrics 2009;123(Suppl 4):S236-S238.
- Cummings JJ, Polin RA; Committee on Fetus and Newborn, American Academy of Pediatrics. Noninvasive respiratory support. Pediatrics 2016;137: doi: 10.1542/peds.2015-3758.
- Needleman JP, Sykes J, Schroeder S, Singer L. Noninvasive positive pressure ventilation in the treatment of pediatric status asthmaticus. Pediat Asthma Allergy Immunol 2004;17:272-277.
- Akingbola OA, Simakajornboon N, Hadley Jr EF, Hopkins RL. Noninvasive positive-pressure ventilation in pediatric status asthmaticus. Pediatr Crit Care Med 2002;3:181-184.
- Thill PJ, McGuire JK, Baden HP, et al. Noninvasive positive-pressure ventilation in children with lower airway obstruction. Pediatr Crit Care Med 2004;5:337-342.
- Cambonie G, Milesi C, Jaber S, et al. Nasal continuous positive airway pressure decreases respiratory muscles overload in young infants with severe acute viral bronchiolitis. Intensive Care Med 2008;34:1865-1872.
- Javouhey E, Barats A, Richard N, et al. Non-invasive ventilation as primary ventilatory support for infants with severe bronchiolitis. Intensive Care Med 2008;34:1608-1614.
- Larrar S, Essouri S, Durand P, et al. [Effects of nasal continuous positive airway pressure ventilation in infants with severe acute bronchiolitis]. [Article in French]. Arch Pediatr 2006;13:1397-1403.
- Campion A, Huvenne H, Leteurtre S, et al. [Non-invasive ventilation in infants with severe infection presumably due to respiratory syncytial virus: Feasibility and failure criteria]. [Article in French]. Arch Pediatr 2006;13:1404-1409.
- Padman R, Henry M. The use of bilevel positive airway pressure for the treatment of acute chest syndrome of sickle cell disease. Del Med J 2004;76:199-203.
- Muñoz-Bonet JI, Flor-Macian EM, Rosello PM, et al. Noninvasive ventilation in pediatric acute respiratory failure by means of a conventional volumetric ventilator. World J Pediatr 2010;6:323-330.
- Mayordomo-Colunga J, Medina A, Rey C, et al. Non invasive ventilation after extubation in paediatric patients: A preliminary study. BMC Pediatr 2010;10:29. Doi: 10.1186/1471-2431-10-29.
- Essouri S, Chevret L, Durand P, et al. Noninvasive positive pressure ventilation: Five years of experience in a pediatric intensive care unit. Pediatr Crit Care Med 2006;7:329-334.
- Kovacikova L, Dobos D, Zahorec M. Non-invasive positive pressure ventilation for bilateral diaphragm paralysis after pediatric cardiac surgery. Interact Cardiovasc Thorac Surg 2009;8:171-172.
- Salvo V, Lista G, Lupo E, et al. Noninvasive ventilation strategies for early treatment of RDS in preterm infants: An RCT. Pediatrics 2015;135:444-451.
- Joshi G, Tobias JD. A five-year experience with the use of BiPAP in a pediatric intensive care unit population. J Intensive Care Med 2007;22:38-43.
- Young AC, Wilson JW, Kotsimbos TC, Naughton MT. Randomised placebo controlled trial of non-invasive ventilation for hypercapnia in cystic fibrosis. Thorax 2008;63:72-77.
- Birnkrant DJ, Pope JF, Eiben RM. Topical review: Pediatric noninvasive nasal ventilation. J Child Neurol 1997;12:231-236.
- Hernandez G, Fernandez R, Lopez-Reina P, et al. Noninvasive ventilation reduces intubation in chest trauma-related hypoxemia: A randomized clinical trial. Chest 2010;137:74-80.
- Duggal A, Perez P, Golan E, et al. Safety and efficacy of noninvasive ventilation in patients with blunt chest trauma: A systematic review. Crit Care 2013;17:R142. Doi: 10/1186/cc12821.
- Richter T, Ragaller M. Ventilation in chest trauma. J Emerg Trauma Shock 2011;4:251-259.
- Evans TW. International Consensus Conferences in Intensive Care Medicine: Non-invasive positive pressure ventilation in acute respiratory failure. Intensive Care Med 2001;27:166-178.
- Mayordomo-Colunga J, Medina A, Rey C, et al. Predictive factors of non invasive ventilation failure in critically ill children: A prospective epidemiological study. Intensive Care Med 2009;35:527-536.
- Lum LC, Abdel-Latif ME, de Bruyne JA, et al. Noninvasive ventilation in a tertiary pediatric intensive care unit in a middle-income country. Pediatr Crit Care Med 2011;12:e7-e13.
- Marohn K, Panisello JM. Noninvasive ventilation in pediatric intensive care. Curr Opin Pediatr 2013;25:290-296.
- Munoz-Bonet JI, Flor-Macian EM, Brines J, et al. Predictive factors for the outcome of noninvasive ventilation in pediatric acute respiratory failure. Pediatr Crit Care Med 2010;11:675-680.
- Bernet V, Hug MI, Frey B. Predictive factors for the success of noninvasive mask ventilation in infants and children with acute respiratory failure. Pediatr Crit Care Med 2005;6:660-664.
- Padman R, Lawless ST, Kettrick RG. Noninvasive ventilation via bilevel positive airway pressure support in pediatric practice. Crit Care Med 1998;26:169-173.
- James CS, Hallewell CP, James DP, et al. Predicting the success of non-invasive ventilation in preventing intubation and re-intubation in the paediatric intensive care unit. Intensive Care Med 2011;37:1994-2001.
MONOGRAPH: Noninvasive ventilation indications, contraindications, and the growing body of literature supporting its use in a variety of clinical scenarios.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
