New Prescription Drug Labeling for Pregnant and Nursing Women
By Larry Lim, PharmD, and Kayleen Thompson, PharmD
Dr. Lim is Commander, U.S. Public Health Service, and Team Lead, FDA Division of Drug Information.
Dr. Thompson is a graduate of the University of Arizona College of Pharmacy, Tucson.
Dr. Lim and Dr. Thompson report no financial relationships relevant to this field of study.
On average in the United States, there are more than 6 million pregnancies annually, and an expecting mother or woman will take three to five prescription medications while pregnant.1 To better assist obstetricians and other healthcare providers with assessing drug benefits and risks for pregnant patients and nursing mothers taking medications, the FDA has updated the requirements for the pregnancy and lactation sections of drug labeling. The rule addresses shortcomings with the current prescription drug labeling information, and requires content and format changes to the requirements under the Physician Labeling Rule (PLR). Overall, the changes allow pregnant women and their healthcare providers to be better informed about the risks and benefits of medications while pregnant or nursing. The new Pregnancy and Lactation Labeling Rule (PLLR) went into effect in June 2015 and is being phased in over the next three to five years.
Labeling Through the Years
Prescription drug labeling is a communication tool. Its principal objective is to make available to healthcare providers the detailed prescribing information necessary for the safe and effective use of a drug, and to do so in a manner that is clear and useful when counseling patients about prescriptions.
Regulations on labeling for human prescription drug use during pregnancy, labor, and delivery and by nursing mothers were issued originally in 1979. In May 2008, the FDA published rules on the content and format of labeling for human prescription drug and biologic products. Nearly 30 years after the codification of the first regulation that provided standardized guidance to healthcare professionals about the use of prescription drugs in pregnant women and nursing mothers, the FDA proposed to eliminate the pregnancy categories because they are often viewed as “… confusing and overly simplistic and, therefore, not adequate to effectively communicate risk of reproductive and developmental toxicity.”2 In December 2014, the FDA published an update known as the PLLR. The new requirements went into effect in June 2015.
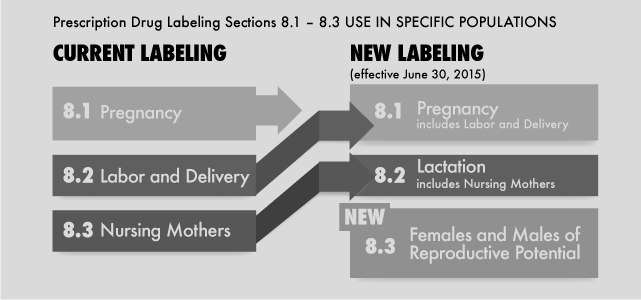
The following questions and answers provide information that may be helpful to obstetricians and other prescribers about medication use while pregnant or nursing.
Q. What are key changes to the prescribing information that went into effect in June 2015?
The former subsections of the labeling rule (Pregnancy, Labor and Delivery, and Nursing Mothers) will be replaced by three new subsections entitled Pregnancy, Lactation, and Females and Males of Reproductive Potential. Table 1 outlines the content of these new subsections. The PLLR also requires the removal of the pregnancy categories – A, B, C, D, and X – from all prescription drug product labeling. Additionally, the PLLR requires the labeling to be updated as new information becomes available.
Q. Why did the FDA make these changes?
Prescribing decisions during pregnancy and lactation are highly individualized and involve complex maternal, fetal, and infant risk–benefit considerations. The pregnancy categories in drug labeling often were viewed as confusing and overly simplistic and didn’t effectively communicate the risk a drug may have during pregnancy. The FDA believes that a narrative structure is best able to capture and convey the potential risks of drug exposure.
Q. Which medications are affected?
All prescription drugs and biologic products that follow the PLR (those approved since June 30, 2001) will be affected. Prescription drugs and biologic products submitted for approval on or after June 30, 2015, will use the new format, while labeling for prescription drugs approved June 30, 2001 through June 29, 2015, will be phased-in. For products approved prior to June 30, 2001, manufacturers are required to remove the pregnancy category within three years of the effective date of the final rule. Labeling for over-the-counter (OTC) medicines will not change; OTC drug products are not affected by the final rule.
Q. What is a pregnancy exposure registry and why does the FDA’s Office of Women’s Health keep a list?
A pregnancy exposure registry is an observational study that collects health information from women who take prescription drugs or vaccines when they are pregnant. Pregnancy exposure registries maintain data on the effects of approved drugs that are prescribed to and used by pregnant women. The FDA does not conduct any studies collected in the pregnancy exposure registries, and does not endorse any registry, but may recommend or require that a drug company implement a pregnancy exposure registry based on certain criteria. The FDA’s Office of Women’s Health maintains a list of registries, which is posted on the FDA’s website at: http://www.fda.gov/ScienceResearch/SpecialTopics/WomensHealthResearch/ucm134848.htm. Enrolling in a pregnancy exposure registry can help improve safety information for medicines used during pregnancy and can be used to update drug labeling.
Q. If the labeling must be continuously updated as more information becomes available, what is the best way for me to obtain up-to-date information?
The drug labeling information for medications marketed in the United States can be found on FDA’s Drugs@FDA at http://www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm and the National Library of Medicine’s DailyMed at http://dailymed.nlm.nih.gov.
The changes to the regulations for the content and format of the Pregnancy subsection in prescription drug labeling attempt to address the lack of information available to healthcare professionals, pregnant women, and nursing mothers. The pregnancy categories A, B, C, D, and X that were once a necessary reference and a fairly accurate guide now appear to be an archaic tool given the advances in medicine and data collection. Not only does new pregnancy labeling assist healthcare professionals with assessing risk vs. benefit and subsequent counseling, it also allows women to make informed and educated decisions for themselves and their children.
Table 1: Label Changes by Subsection |
||
Subsection |
Subheadings |
Description |
|
8.1 Pregnancy |
Pregnancy Exposure Registry |
|
|
Risk Summary |
|
|
|
Clinical Considerations |
|
|
|
Data (if applicable) |
|
|
|
8.2 Lactation |
Risk Summary |
|
|
Clinical Considerations (if applicable) |
|
|
|
Data (if applicable) |
|
|
|
8.3 Females |
Pregnancy Testing (if applicable) |
|
|
Contraception (if applicable) |
|
|
|
Infertility (if applicable) |
|
|
REFERENCES
- Barbor M. Medscape Medical News from the American Society of Health-System Pharmacists (ASHP) 2015 Midyear Clinical Meeting. FDA Issues New Drug Safety Information for Pharmacists. Published December 10, 2015.
- Content and Format of Labeling for Human Prescription Drug and Biological Products; Requirements for Pregnancy and Lactation Labeling, 2008.
The FDA has updated the requirements for the pregnancy and lactation sections of drug labeling to allow pregnant women and their healthcare providers to be better informed about the risks and benefits of medications while pregnant or nursing. This article discusses the new Pregnancy and Lactation Labeling Rule that went into effect in June 2015 and is being phased in over the next three to five years.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
