Evaluation and Management of Cervical Artery Dissection in the Emergency Department
November 15, 2016
Reprints
Authors
J. Michael Ballester, MD, Assistant Professor, Emergency Medicine; Program Director, Emergency Medicine, Wright State University Boonshoft School of Medicine, Dayton, OH.
Thomas Dailey, MD, Emergency Medicine, Wright State University Boonshoft School of Medicine, Dayton, OH.
Peer Reviewer
Matthew E. Fink, MD, FAAN, FAHA, FANA, Louis and Gertrude Feil Professor, Chairman, Neurology, Assistant Dean of Clinical Affairs, Weill Cornell Medicine, New York-Presbyterian Hospital, New York, NY.
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Farel (CME question reviewer) owns stock in Johnson & Johnson. Dr. Stapczynski (editor) owns stock in Pfizer, Johnson & Johnson, Walgreens Boots Alliance Inc., GlaxoSmithKline, Bristol Myers Squibb, and AxoGen. Dr. Schneider (editor), Ms. Fessler (nurse planner), Dr. Ballester (author), Dr. Dailey (author), Dr. Fink (peer reviewer), Ms. Mark (executive editor), Ms. Coplin (executive editor), and Mr. Landenberger (editorial and continuing education director) report no financial relationships with companies related to the field of study covered by this CME activity.
EXECUTIVE SUMMARY
- Cervical artery dissection is classified as extracranial/intracranial, vertebral/carotid artery. Carotid dissections are more common than vertebral ones.
- Intracranial extension can result in a stroke or subarachnoid hemorrhage. Arterial embolism is the main cause of ischemic strokes.
- Pain over the face or neck, often refractory to common analgesics, is often the first sign. If untreated, this may progress to neurologic deficits.
- A CT angiogram is the preferred diagnostic test. MRI and magnetic resonance angiography also may demonstrate the lesion but are more time-consuming, and less available to the ED. A regular head CT may show some suggestion of a dissection, but these are often missed.
- Once diagnosed, treatment should begin as soon as possible. Treatment with anticoagulation or antiplatelet agents is considered first-line care. Endovascular repair or even open surgical repair is done in special circumstances or as part of a clinical trial.
A few years ago, a healthy young colleague suddenly developed signs of a stroke. Testing in the emergency department (ED) determined he had a cervical artery dissection. With treatment, he made a full recovery and returned to work.
This episode got me thinking. Before CT angiography (CTA) of the head and neck were part of the routine workup of a stroke, how many dissections had I missed? Should every patient with new neurologic findings get a CTA? What about hospitals that barely can get a CT in the off hours? This paper will begin to answer these questions. But here, still, are a lot of unanswered questions. When is a headache, or neck pain, really a dissection? Should all trauma patients with a head/neck injury and an unreliable neurologic exam receive specialized imaging?
Cervical artery dissection can have devastating results if not treated correctly and promptly. The problem is thinking of the diagnosis.
— Sandra M. Schneider, MD, Editor
Definition of the Problem
Patients with cervical artery dissection (CeAD), including vertebral and carotid dissections, present to the ED in a myriad of ways. Symptoms can vary from clear indication of dissection at onset, to vague symptoms, such as headache, neck pain, facial pain, stroke-like symptoms, and neurological deficits, that can make a diagnosis very difficult. In all cases, prompt treatment of CeAD is essential to mitigate morbidity and mortality. The goal of this paper is to help emergency physicians in their diagnosis, management, and treatment of this rare but potentially devastating condition.
Epidemiology
The majority of morbidity and mortality with CeAD results when cerebral ischemia or hemorrhage is present. Combined occurrence of vertebral and carotid artery dissection is reported to have an incidence of 2.6-3.01 per 100,000.1 It is believed that the recorded incidence of CeAD has increased since the mid 1990s because of previous under-detection. Magnetic resonance imaging (MRI) became available around that time.1
The overall incidence of ischemic stroke caused by CeAD has been reported to range from 2-13%.2 Fortunately, recurrence of ischemic stroke due to cervical artery dissection is very low (1-2%).3,4
CeAD is the leading cause of ischemic stroke in adults younger than 42 years of age. The mean age for cervical artery dissection is reported to be 45.8 years, with the average age of CeAD causing ischemic stroke being 49 years.5
Cervical artery dissection is classified as extracranial or intracranial, and vertebral or carotid dissection. The incidence of extracranial vertebral dissection is more common in a younger population, and internal dissection is more common in older age groups.6 Traumatic or increased mechanical stress is associated with extracranial dissection.5 Interestingly, there are more physicians among CeAD stroke patients than non-CeAD stroke patients, and CeAD with local symptoms has a higher reported incidence for physicians than non-physicians.4 This may be because physicians may have an increased awareness of the significance of their symptoms, or they may seek centers with more advanced testing facilities.
Etiology
CeAD etiology can result from traumatic or spontaneous dissection. Spontaneous dissection refers to cases in which a cause is not immediately identifiable, or the presence of major trauma is lacking. Spontaneous dissection has been the most studied, as it often affects young people, resulting in devastating morbidity and mortality. Aside from specifics on susceptibility of dissection at certain locations along the vertebral and internal carotid arteries, there have been few studies looking at traumatic etiology.
Traumatic Dissection
Traumatic etiology is considered when blunt or penetrating trauma has occurred to the head and/or neck. In 1974, Crissey and Bernstein identified four mechanisms of injury to the carotid artery. Type I injuries result from a direct blow to the neck. Type II injuries, which are the most common, result from hyperextension and contralateral rotation of the head and neck. Type III injuries result from intraoral trauma. Type IV injuries are associated with skull base fractures that involve the sphenoid or petrous bones.7 Minor or unidentifiable (but suspected) trauma leads to the CeAD being classified as spontaneous. Minor trauma may include sudden head movements, neck manipulation, coughing, sneezing, and performing the Valsalva maneuver.
Spontaneous Dissection
There have been conflicting studies on whether traditional cardiac risk factors have an association with CeAD in cases of spontaneous dissection. Cardiac risk factors, which include hypertension, hypercholesterolemia, diabetes mellitus, and smoking, previously have been thought to be associated with vertebral artery and internal carotid artery dissection.5,8 This is contrary to the only prospective study of risk factors associated with CeAD, where preliminary results support the belief that there is no causative relationship between general cardiovascular risk factors, with the exception of migraine, and CeAD.9 Recent infection was reported in only 21% of CeAD.10
Spontaneous dissections are thought to have some hereditary etiology that may independently increase the risk of CeAD, or increase the risk of CeAD in minor trauma. These hereditary factors may include connective tissue disorders such as Ehlers-Danlos syndrome, Marfan syndrome, polycystic kidney disease, osteogenesis imperfecta, fibromuscular dysplasia, alpha 1 antitrypsin, and homocysteinuria.11 All of these disorders lead to connective tissue abnormalities within the vessel wall structure. CeAD was found to be the initial manifestation for 12.1% of fibromuscular dysplasia patients.12 Studies have demonstrated a link in first-degree relatives who have connective tissue disorders for patients with spontaneous cervical artery dissection.
However, even with all of these genetic and familial relations, there is no definite heritable risk, as relatives of CeAD patients had fewer strokes at a young age than relatives of non-CeAD ischemic stroke patients.13 The largest retrospective review on familial association of connective tissue disorders found a very low association with CeAD ischemic stroke. Specifically, less than 1.0% of ischemic stroke CeADs have any association with familial connective tissue disorders.14 It is has been reported that the majority of studies that found a positive relationship between CeAD and connective tissue disorders had significant sampling bias. This is important knowledge to impart to patients with connective tissue disorders who may be concerned about their risk of CeAD.
There are genetic factors that have an association with cerebrovasculopathy, which subsequently have been found to correlate with CeAD. These include genes that have a role in homocystinuria, venous thrombosis, subarachnoid hemorrhage, Ehlers-Danlos syndrome, familial dermal connective tissue abdnormalities, and Loeys-Dietz syndrome. Elevated homocysteine levels are an independent risk factor for atherosclerosis and stroke. High levels of homocysteine contribute to endothelial damage and susceptibility to arterial thrombosis through increased oxidative stress. Individuals with elevated homocysteine levels and mutations in methylene tetrahydrofolate reductase (MTHFR), an enzyme that helps with the metabolism of a co-substrate for homocysteine metabolism, have a positive association with CeAD.15 Furthermore, lowering homocysteine levels with vitamin B and folic acid supplements may have a role in prevention and therapy for CeAD.
Although it is difficult to make an association between rare genetic disorders and CeAD because of its low prevalence, there have been a few other genes associated with CeAD. ICAM is involved in cell migration across endothelium. COL3A1 is involved in type III collagen production (part of extracellular membrane). COL5A2 is involved in type V collagen (part of fibrillar formation). TGFBR2 regulates smooth muscle cell migration.16 PHACTR1, a gene that encodes for a protein involved in tubule formation in endothelial cells, has an increased association with myocardial infarction, migraines, and CeAD.17 All of these genes have demonstrated varying associations with CeAD through clinical studies.
There have even been several case reports of CeAD in postpartum women. It is difficult to determine whether the intense Valsalva during pregnancy, numerous hemodynamic factors (increased blood volume, increased CO, increased wall forces), hormonal changes, or advanced maternal age (increased arterial stiffness) during pregnancy play a role in the pathophysiology of CeAD in peripartum women.18,19 There is no increased risk of recurrent CeAD during pregnancy for women who had a previous CeAD.20
Other seemingly benign mechanisms, such as riding on roller coasters and neck manipulation, have been associated with cervical artery dissection, likely due to shearing forces.10 Historically, education on CeAD almost always will mention neck manipulation as a risk factor, yet there is little statistical evidence to demonstrate this relationship.21 Studies typically fail to demonstrate causation. Futhermore, patients with neck pain often seek chiropractic manipulation, when in fact the neck pain is the initial symptom of the CeAD. It becomes nearly impossible to determine which came first. These patients actually may have presented to the chiropractor with an existing dissection. Many studies rely on self-reporting and suffer from recall bias, and improper coding for CeAD associated with spinal manipulation. A large majority of studies are case reports.22 Because of this, the positive predictive value in spinal manipulation with CeAD has been low. Cai et al re-analyzed their own data with the appropriate diagnosis, which revealed a much higher positive predictive value between spinal manipulation therapy and cervical artery dissection.23 Conclusions that appeared at first glance to be more speculative and anecdotal actually may be statistically significant.
Multivessel Dissection
Single vessel CeAD is relatively rare, and, subsequently, multiple vessel CeAD is even more rare. Underlying vasculopathy, such as fibromuscular dysplasia, and environmental triggers, such as recent infection, cervical manipulation, and remote history of head and neck surgery, have an increased risk of multiple vessel CeAD.24 Multivessel vs. single-vessel involvement does not change the risk of stroke.21
Pathophysiology
Cervical dissection pathophysiology is no different from that of any other vascular dissection. It often begins with an intimal tear that results in stenosis, occlusion, or dissecting aneurysm (also known as pseudoaneurysm). The intimal tear may explain the pathophysiology in spontaneous dissection, but aneurysmal formation is the major cause in developing a CeAD due to trauma.
The majority of dissections result in stenosis 48% of the time, followed by occlusion (35%) and dissecting aneurysm (17%).1 (See Figures 1 and 2.)
Figure 1. Internal Carotid Artery with Dissection and Occlusion
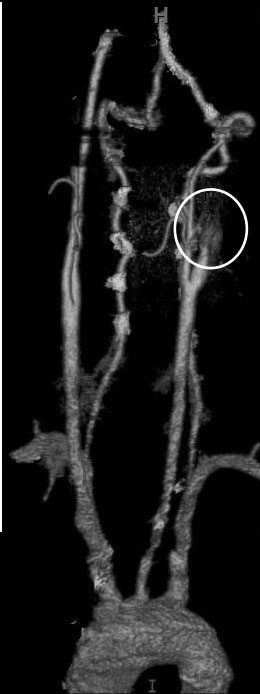
Figure 2. Complete Right Internal Carotid Artery Occlusion Secondary to Dissection
>
Images courtesy of Dr. Linda Heier, Chief of Neuroradiology at Weill Cornell Medical Center, New York, NY.
Spontaneous dissection pathophysiology becomes less evidence-based and more speculative when the question arises of whether an intimal tear always occurs and why it occurs. Most of the theories in spontaneous dissection result from research performed on connective tissue disorders. For example, it has been demonstrated that medial degeneration involves inflammatory infiltration in patients with Marfan syndrome, and it is possible that these defective components of the extracellular matrix are potent attractants for macrophage chemotaxis and subsequent dissection along the media.25 In addition to defective arterial wall structure, it is thought that increased wall forces may be a significant contributor in the development of CeAD. The tear may not be obvious or even present in some cases of spontaneous dissection.
Subintimal dissections form between the intima and media, and usually result in intramural hematomas, which lead to stenosis of the vessel. Subadventitial tears are those that form between the media and adventitia, and usually result in aneurysmal dilation. This defect will extend or dissect along the plane of injury. Extracranial vs. intracranial dissections have different rates of intramural vs. aneurysm formation.
Depending on the origin of dissection and vessel involved (carotid vs. vertebral), the rates of extension intracranially vary. Intracranial extension of vertebral dissections is particularly disabling, with a high incidence of ischemic stroke or subarachnoid hemorrhage.26 There is a higher risk of stroke with vertebral artery dissection than with internal carotid dissection.1 Most often, an intracerebral hemorrhage due to CeAD is subarachnoid. Intracranial to extracranial extension also has been described in patients after aneurysmal subarachnoid hemorrhage.27
Similar to aortic dissection, treatment is focused on controlling shear wall forces. However, intracranial pressures are not exposed to the same systemic wall forces as aortic dissections.
Reversible cerebral vasoconstriction syndrome (RCVS), diagnosed by MRI and clinical features characterized by severe headaches and, infrequently, seizures and focal neurologic symptoms, have a positive correlation with CeAD.28 The mechanism for RCVS is thought to involve sympathetic dysregulation of arterial tone with subsequent increased wall forces and susceptibility to dissection.
Although dissection and resultant intramural hematoma/aneurysm certainly can cause symptomatic findings and damage, in and of itself, the resultant thrombus with thromboembolic sequelae leads to the majority of morbidity and mortality related to stroke and intracerebral hemorrhage. Arterial embolization is thought to be the main mechanism of ischemic stroke. There is an immediate threat of thromboembolic phenomena from cervical artery dissection due to fresh clot formation, which is subject to rapid flow of the arterial system. This clot is fragile during the first several hours of this hyperacute phase. When actual brain infarction occurs, it is usually during this hyperacute phase and caused by thromboemboli.13 Spontaneous dissection is thought to result more often in thromboembolic phenomena than traumatic dissection.
Traumatic CeAD occurs more frequently at the level of the first and second cervical vertebrae. At the level of C2, there is some natural arterial vulnerability due to the rotational forces at the atlantoaxial junction, and even seemingly minor neck motion can cause stretching and compression of the vertebral artery at this level. This susceptibility should be considered in cases of children, who have increased mobility and larger craniums.29 Spontaneous CeAD occurs more commonly at the level of the fifth cervical vertebra, and is characterized by increased incidence in people older than 50 years of age, and is associated with coronary artery disease, hypertension, and hyperlipidemia.30 Fractures and direct trauma to C5 and C6 predispose the artery to dissection, since it enters the cervical foramen at these locations.
Clinical Features
The primary presenting symptoms of CeAD include headache, neck pain, speech disturbance, limb weakness, balance disturbance, and ptosis. (See Table 1.) Pain and blurred vision are the most common antecedent manifestations.6,10 Pain is usually unilateral over the face, head, and neck.6 Unilateral facial and neck pain should always alert the provider that a more serious underlying condition may be present, especially if this pain is refractory to treatment, as is often the case with CeAD. Radiculopathy from compression or irritation of the cervical roots also has been described in cases of CeAD.31 Patients may experience cerebellar symptoms, including dizziness, vertigo, ataxia, or brain stem and cranial nerve deficits. Horner’s syndrome may develop in some cases, as the sympathetic fibers are being stretched over the external carotid sheath. Consider a dissection in a patient who presents with pain and Horner’s syndrome. There is a higher association of non-ischemic dissections than ischemic dissections with Horner’s syndrome.21
Table 1. Risk Factors, Symptoms, and Signs of Cervical Artery Dissection
|
Risk factors |
Major blunt or traumatic injury Minor trauma: • Sudden movement, neck manipulation, coughing, Valsalva, riding roller coasters Hypertension, hyperlipidemia, diabetes mellitus, smoking Connective tissue disorders Vasculopathy |
|
Symptoms |
Headache, ptosis, unilateral facial and/or neck pain Cerebral vascular accident symptoms Cerebellar symptoms |
|
Signs |
No specific exam findings unless causing a cerebral vascular accident or Horner’s syndrome. Primarily present with subjective symptoms listed above. |
Overall, extracranial lesions usually present with less severe and more clinically vague symptoms, including dizziness, vertigo, tinnitus, nausea, and vomiting. Intracranial lesions are more likely to rupture because of extension of the dissection into the subadventitium, since the media is thinner, so subarachnoid hemorrhage is the most common presenting symptom of intracranial CeAD.32 Most patients with two-vessel CeAD present with symptoms from a single vessel.21
Diagnostic Studies
Although imaging is the only incontestable way to diagnose a CeAD, there are also laboratory tests that may be considered on the path to diagnosis. (See Table 2.) CeAD is associated with high white blood cell (WBC) counts in many cases of spontaneous dissection. In these patients, imaging reveals signs of local inflammation around the affected vessels.18 D-dimer has been proposed as a screening tool in aortic arterial dissections, although the false-positive rate is fairly high.33 Unfortunately, application of this tool has not been extended to CeAD, as D-dimers were found to be lower in cases of CeAD than in cases of aortic dissections.34 It is thought that with dissections in general, the clotted blood is not exposed to the systemic blood flow as it develops an intramural hematoma.
Table 2. Tests for Cervical Artery Dissection
- White blood count (usually high)
- D-dimer — not helpful
- 3D magnetic resonance imaging
- CT angiography — preferred
Angiography is classically considered the gold standard in diagnosis of CeAD. A serious limitation in using angiography is the failure to delineate luminal abnormalities, as well as the invasive and less expeditious nature of the test. These shortcomings make angiography a less than ideal diagnostic study in the ED.
While MRI may not be entirely applicable to the ED setting, it is better at showing luminal abnormalities and vessel wall hematomas, but is limited for vertebral artery dissection. There has been extensive research with regard to MRI quality. Two-dimensional MRI may be useful for diagnosis in cases of intramural hematomas, but three-dimensional MRI has been shown to easily identify these smaller, localized intramural hematomas with high sensitivity and specificity.35 Three-dimensional MRI also is much better for visualizing segments of the vertebral arteries, including subacute intramural hematomas.36,37
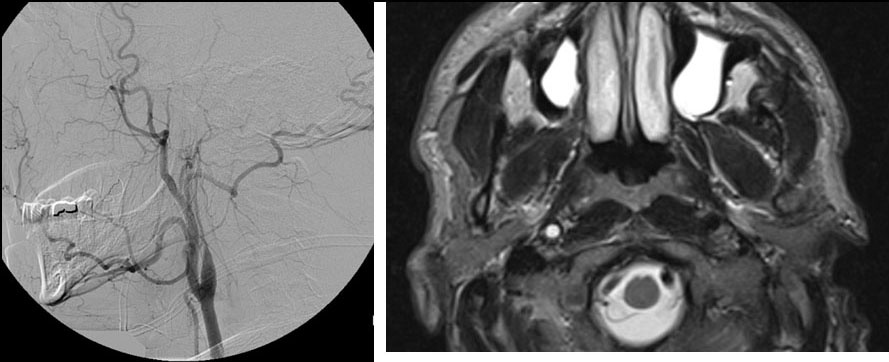
CT angiography (CTA) is faster and less invasive than angiography, and it has better imaging for the vertebral artery than magnetic resonance angiography (MRA). (See Figures 2-4.)
Figure 3. Internal Carotid with Intimal Flap
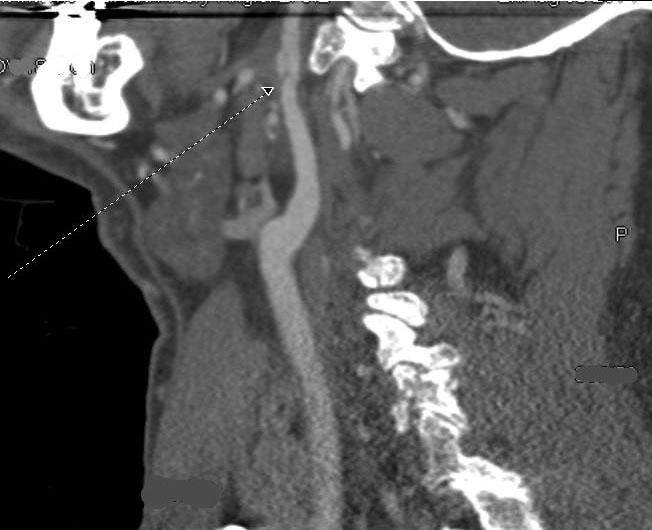
Figure 4. Internal Carotid with Intimal Flap
Non-contrast head CT, which is frequently ordered as part of the typical stroke workup, typically does not reveal any specific signs of CeAD, and it is incapable of confirming the presence of an intramural hematoma.38 Non-contrast CT does demonstrate nonspecific findings, including asymmetry, thickening, and reduced diameter of lumen, which can lead to suspicions of CeAD that may be confirmed through further diagnostic testing.
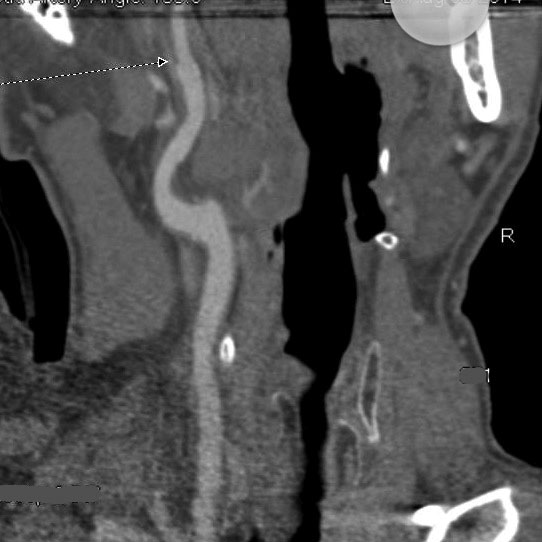
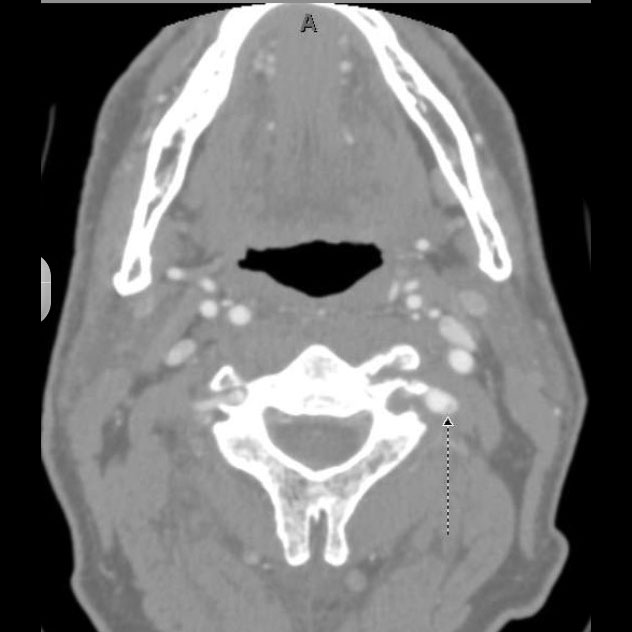
On its own, a non-contrast head CT does not diagnose CeAD. A CTA of the head and neck is necessary to lead to a positive diagnosis of CeAD. Further assessment of the effects on the lumen, including stenosis, occlusion, and pseudoaneurysm, can be done with an MRA. (See Figure 5.) Diagnosis of cerebral ischemia can be accomplished with diffusion-weighted MRI. T1-weighted, 3D MRI is the definitive test for diagnosis of CeAD because it allows for identification of the mural hematoma. Duplex ultrasound can be considered as an option to monitor progression of dissection following diagnosis.
Figure 5. Vertebral Dissection with Pseudoaneurysm
Differential Diagnosis
Differential diagnosis of CeAD varies significantly depending on presenting symptoms. As the majority of symptoms of CeAD can be either vague or very specific stroke symptoms, the differential can be broad or narrow. Signs and symptoms of CeAD vary and are similar to other more benign etiologies that are encountered more frequently, so differential diagnosis is dependent on factors that lead to a high index of suspicion.
Management
Regardless of the number of vessels involved, the most important aspect of management of patients with CeAD is early treatment, which significantly reduces the chances of death and disability from major stroke. This is especially true in patients presenting with mild, non-disabling symptoms of CeAD.39 Patients with CeAD have an increased risk of stroke within 24 hours, and have an increased risk of recurrent symptoms or initial stroke within seven days. Treatment is targeted at preventing an immediate acute event within 24 hours and preventing the recurrence or delayed primary stroke within seven days.40 (See Table 3.) Treatment focuses on minimizing neurologic insult with restoration of blood flow and prevention of bleeding. To date, there have been no studies that have compared outcomes resulting from antithrombotic, endovascular repair, thrombolysis, or open surgical repairs or interventions.
Table 3. Treatment for Cervical Artery Dissection
Type of Dissection |
Treatment |
|
Extracranial dissection |
Anticoagulation |
|
Intracranial dissection + pseudoaneurysm, CVA |
Antiplatelet Endovascular stenting |
|
Traumatic dissection |
Thrombolysis |
The choice of antithrombotic therapy in the management CeAD is the subject of an ongoing debate. Confounding factors related to additional injury, including type of therapy, timing of antithrombotic delivery, location of dissection (extracranial vs. intracranial), and mechanism of dissection (traumatic vs. spontaneous), influence the choice of therapy.
A meta-analysis by Sarikaya et al in 2013 comparing antiplatelet therapy to anticoagulant therapy demonstrated that there was a large treatment effect in favor of antiplatelet medications in preventing ischemic stroke, intracranial hemorrhage, and death within three months of treatment initiation for all CeAD.41 Before this, the Cochrane review in 2010 published a large meta-analysis that compared treatments of carotid artery dissection. The authors found no significant difference in death or occurrence of ischemic stroke with either the use of antiplatelet or anticoagulant treatments. With the use of anticoagulation, the rate of death and disability was more favorable, but clinically relevant intracranial hemorrhage and major extracranial hemorrhage still occurred without a notable difference in frequency.42
The CADISS trial is a landmark study, as it is the first randomized, controlled study to compare antiplatelet therapy to anticoagulation treatment of extracranial cervical artery dissection. The study enrolled patients who had either local symptoms or stroke within a seven-day onset. The antiplatelet group was treated with either aspirin, clopidogrel, dipyridamole, or both aspirin and clopidogrel. The anticoagulant group received heparin and warfarin, or just warfarin. There was no significant difference in outcome of stroke or death between the treatment groups.43 One major limitation of this study was that the mean time of onset in treatment after symptom onset was 10.8 days. It is thought that most strokes develop within the first seven days of brain ischemia.44
One confounding problem when studying the treatment of CeAD is that the onset of symptoms may not correlate with the onset of the dissection. There are a large majority of patients without symptoms who have CeAD.21
Of note, novel oral anticoagulants (NOACs), which include dabigatran, rivaroxaban, and apixaban, have similar rates of recurrent stroke and fewer hemorrhagic complications than warfarin or heparin, but have increased rates of worsening radiographic stenosis.45 The majority of studies that evaluate antithrombotic therapies are using warfarin or heparin, not the NOACs.
Due to the pathophysiology of intimal tearing with either severe stenosis and occlusion or thromboembolic sequelae from rapid clot formation, there is an urgency to treat patients with CeAD within 24 hours of presentation. This has led the majority of physicians to prefer anticoagulation rather than antiplatelet therapy as initial treatment for extracranial dissections.46 Others have argued that while anticoagulants may help with the theoretical prevention of clot embolization, they also may be detrimental to a favorable outcome because of potential extension of the intramural hemorrhage, which may further worsen the dissection. Most experts agree that antiplatelets should be used when there is a contraindication to anticoagulants, such as spinal cord hematoma, large intracerebral infarct, or intracranial extension of the dissection, which may cause subarachnoid hemorrhage.47 However, no definitive difference in outcome has been noted in patients treated with antiplatelets vs. anticoagulants, even in studies evaluating intracranial dissection.7 For these patients, there is a theoretical increased risk, which has not been statistically proven, of hemorrhagic transformation. Within these studies, the only patients who experienced hemorrhagic stroke after the onset of a CeAD were those who were treated with anticoagulants.48 New ischemic stroke occurs in up to a quarter of patients with CeAD, regardless of antiplatelet or anticoagulant therapy.49
Medical therapy is the primary treatment for extracranial CeAD. Through 2014, there have been no studies to support the use of endovascular repair (stent or coils) over medical therapy for either extracranial or intracranial CeAD. Endovascular repair generally is reserved for patients with CeAD who have intracranial complications. Extracranial lesions preferably are treated with antithrombotic medication, since the occurrence of subsequent stroke and intracranial hemorrhage is relatively low.
Endovascular stenting or proximal occlusion (coils) may be the best options for intracranial dissections with complications, such as pseudoaneurysm rupture, impending infarct in the basilar system, posterior system ischemia, refractory medical therapy, or other general contraindications to antiplatelets.16 Although endovascular repair is reserved primarily for intracranial complications, the use of stenting has been described in cases of completely occluded bilateral extracranial internal carotid artery (ICA).50 Interventional radiology has become more involved in the routine management of aneurysm and pseudoaneurysm with permanent vessel sacrifice. While there is already significant potential morbidity with vessel sacrifice, there is also the threat of a thromboembolic process with the coils, especially with newer methods that combine coils with liquid embolic agents such as Onyx.51
Thrombolysis is indicated in certain scenarios, but it comes with significant risks, which include distal embolic sequela when the clot breaks apart and risk of converting intramural hematoma to dissecting aneurysm. The use of thrombolysis is generally limited to cases of traumatic dissection. Surgical repair options for the treatment of CeADs include cervical artery ligation, thromboendarterectomy, extracranial to intracranial bypass, and interposition grafts.
Significant morbity and mortality are associated with these procedures.
Regardless of the methods employed to manage cases of CeAD, stroke severity is the best predictor of outcome. Outcomes from thrombolysis in ischemic stroke are similar whether the stroke is due to CeAD or other causes.
Additional Factors
Not only is it important that the emergency physician has a good understanding of the laboratory and specific imaging studies important to diagnosis and management of CeADs, but it is also of paramount importance for diagnosis, management, and disposition that the facility have the capability to perform these procedures. Comprehensive stroke centers are specialized and equipped to provide necessary multimodal neuroimaging for diagnosis of complex cases that lead to better patient outcomes through more adequate treatment. This is especially true for CeAD-related ischemic strokes, where it was found that these centers have an increased frequency of diagnosis of CeAD. Early diagnosis of CeAD correlates with an increased number of revascularization therapies conducted in the acute phase, which contributes to better outcomes in patients with more severe CeAD-related strokes.52
The incidence of recurrent CeAD is higher than initially thought because of a lack of specific symptoms. In one study, 79% of patients with recurrent CeAD were asymptomatic.53 Age and basilar artery involvement are independent predictors of unfavorable outcomes in cases of CeAD with ischemic stroke.9 Neurologic deterioration occurs more frequently in women, bilateral vertebral artery involvement, and intracranial vessel involvement.54 Neurologic deterioration, specifically National Institutes of Health Stroke Score > 8 during the first 24 hours in ischemic stroke patients, often is related to CeAD and hemorrhagic transformation.55 This warrants immediate repeat imaging. Compared to the general population, there is an increased frequency of post-traumatic stress disorder in patients who recover from CeAD, and they also have decreased mental quality of life and coping abilities.56
SUMMARY
Untreated, the risk of subsequent stroke after CeAD remains high. Up to 56% of patients will suffer a stroke within 24 hours, and as many as 82% will suffer a stroke within seven days of symptom onset. It is imperative that patients are identified early and that appropriate imaging be performed immediately. Traumatic CeAD needs to be evaluated on mechanism and symptoms alone, and one should not delay an evaluation if neurologic symptoms are not present. Patients will need hospitalization and evaluation by appropriate trauma and/or comprehensive neuro-interventional services.
REFERENCES
- Lee VH, Brown RD Jr., Mandrekar JN, Mokri B. Incidence and outcome of cervical artery dissection: A population-based study. Neurology 2006;67:1809-1812.
- Debette S, Leys D. Cervical artery dissections: Predisposing factors, diagnosis, and outcome. Lancet Neurol 2009;8:668-678.
- Debette S, Grond-Ginsbach C, Bodenant M, et al. Differential features of carotid and vertebral artery dissections: The CADISP study. Neurology 2011;77:1174-1181.
- Grond-Ginsbach C, Lichy C, Debette S, et al. Cervical Artery Dissection (CeAD) in physicians. Cerebrovasc Dis 2015;39:72-74.
- Béjot Y, Daubail B, Debette S, et al. Incidence and outcome of cerebrovascular events related to cervical artery dissection: The Dijon Stroke Registry. Int J Stroke 2014;9:879-882.
- Kobayashi H, Morishita T, Ogata T, et al. Extracranial and intracranial vertebral artery dissections: A comparison of clinical findings. J Neurol Sci 2016;362:244-250.
- Crissey MM, Bernstein EF. Delayed presentation of carotid intimal tear following blunt craniocervical trauma. Surgery 1974;75:543-549.
- Kim BM, Kim SH, Kim DI, et al. Outcomes and prognostic factors of intracranial unruptured vertebrobasilar artery dissection. Neurology 2011;76:1735-1741.
- Thomas LC, Rivett DA, Attia JR, Levi C. Risk factors and clinical presentation of cervical arterial dissection: Preliminary results of a prospective case-control study. J Orthop Sports Phys Ther 2015;45:503-511.
- Badve MS, Bhuta S. Roller coasters and cervical artery dissection. Med J Aust 2015;203:339.
- Giossi A, Ritelli M, Costa P, et al. Connective tissue anomalies in patients with spontaneous cervical artery dissection. Neurology 2014;83:2032-2037.
- O’Connor SC, Poria N, Gornik HL. Fibromuscular dysplasia: An update for the headache clinician. Headache 2015;55:748-755.
- Kloss M, Grond-Ginsbach C, Pezzini A, et al; Cervical Artery Dissection and Ischemic Stroke Patients (CADISP) Study Group. Stroke in first-degree relatives of patients with cervical artery dissection. Eur J Neurol 2014;21:1102-1107.
- Debette S, Goeggel Simonetti B, Schilling S, et al. Familial occurrence and heritable connective tissue disorders in cervical artery dissection. Neurology 2014;83:2023-2031.
- Luo H, Liu B, Hu J, et al. Hyperhomo-cysteinemia and methylenetetrahydrofolate reductase polymorphism in cervical artery dissection: A meta-analysis. Cerebrovasc Dis 2014;37:313-322.
- Southerland AM, Meschia JF, Worrall BB. Shared associations of nonatherosclerotic, large-vessel, cerebrovascular arteriopathies: Considering intracranial aneurysms, cervical artery dissection, moyamoya disease and fibromuscular dysplasia. Curr Opin Neurol 2013;26:13-28.
- Debette S, Kamantani Y, Metso TM, et al. Common variation in PHACTR1 is associated with susceptibility to cervical artery dissection. Nat Genet 2015;47:78-83.
- Simon EL, Griffin G, Bosman E. Bilateral carotid and vertebral artery dissection: A life-threatening cause of postpartum headache. Am J Emerg Med 2015;33:600.e1-600.e3.
- 19.Kelly JC, Safain MG, Roguski M, et al. Postpartum internal carotid and vertebral arterial dissections. Obstet Gynecol 2014;123:848-856.
- Reinhard M, Munz M, von Kannen A, et al. Risk of recurrent cervical artery dissection during pregnancy, childbirth and puerperium. Eur J Neurol 2015;22:736-739.
- Biller J, Sacco RL, Albuquerque FC, et al; American Heart Association Stroke Council. Cervical arterial dissections and association with cervical manipulative therapy A statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2014;45:3155-3174.
- Wynd S, Westaway M, Vohra S, Kawchuk G. The quality of reports on cervical arterial dissection following cervical spinal manipulation. PloS One 2013;8:1-8.
- Cai X, Razmara A, Paulus JK, et al. Case misclassification in studies of spinal manipulation and arterial dissection. J Stroke Cerebrovasc Dis 2014;23:2031-2035.
- Béjot Y, Aboa-Eboulé C, Debette S, et al; CADISP Group. Characteristics and outcomes of patients with multiple cervical artery dissection. Stroke 2014;45:37-41.
- Grond-Ginsbach C, Giossi A, Aksay SS, et al; CADISP group. Elevated peripheral leukocyte counts in acute cervical artery dissection. Eur J Neurol 2013;20:1405-1410.
- Arauz A, Ruiz A, Pacheco G, et al. Aspirin versus anticoagulation in intra- and extracranial vertebral artery dissection. Eur J Neurol 2013;20:167-172.
- Chatani M, Nakagawa I, Wada T, et al. Gradual growth of spontaneous dissecting aneurysm of the extracranial internal carotid artery after aneurysmal subarachnoid hemorrhage. Turk Neurosurg 2015;25:649-652.
- Mawet J, Boukobza M, Franc J, et al. Reversible cerebral vasoconstriction syndrome and cervical artery dissection in 20 patients. Neurology 2013;81:821-824.
- Agostini M, Lupica MM, Mostert M, et al. Cervical arterial dissection and ischaemic stroke in children: Two cases. Acta Pædiatr 2013;102:e142-e146.
- Lleva P, Ahluwalia BS, Marks S, et al. Traumatic and spontaneous carotid and vertebral artery dissection in a level 1 trauma center.
J Clin Neurosci 2012;19:1112-1114. - Eberhardt O, Topka H. Compressive cervical radiculopathy due to vertebral artery dissection. J Stroke Cerebrovasc Dis 2015;24:e115-e116.
- Hernández-Durán S, Ogilvy CS. Clinical outcomes of patients with vertebral artery dissection treated endovascularly: A meta-analysis. Neurosurg Rev 2014;37:569-577.
- Asha SE, Miers JW. A systematic review and meta-analysis of D-dimer as a rule-out test for suspected acute aortic dissection. Ann Emerg Med 2015;66:368-378.
- Nagamine Y, Fukuoka T, Hayashi T, et al. Research article: Clinical characteristics of isolated anterior cerebral artery territory infarction due to arterial dissection. J Stroke Cerebrovasc Dis 2014;23:2907-2913.
- Edjlali M, Roca P, Rabrait C, et al. 3D fast spin-echo T1 black-blood imaging for the diagnosis of cervical artery dissection. AJNR Am J Neuroradiol 2014;34:E103-E106.
- Cuvinciuc V, Viallon M, Momjian-Mayor I, et al. 3D fat-saturated T1 SPACE sequence for the diagnosis of cervical artery dissection. Neuroradiology 2013;55:595-602.
- Cuvinciuc V, Viallon M, Momjian-Mayor I, et al. 3D fat-saturated T1 SPACE sequence for the diagnosis of cervical artery dissection. Neuroradiology 2013;55:595-602.
- Ben Hassen W, Machet A, Edjlali-Goujon M, et al. Imaging of cervical artery dissection. Diagn Interv Imaging 2014;95:1151-1161.
- Gallerini S, Bartalucci M, Marconi R. Letter by Gallerini et al regarding article, Characteristics and outcomes of patients with multiple cervical artery dissection. Stroke 2014;45:e90.
- Ellanti P, Hurson C. Traumatic intracerebral infarction due to vertebral and carotid artery dissection. BMJ Case Rep 2014; Dec 22.
- Sarikaya H, da Costa BR, Baumgartner RW, et al. Antiplatelets versus anticoagulants for the treatment of cervical artery dissection: Bayesian meta-analysis. PLoS One 2013;8:e72697.
- Lyrer P, Engelter S. Antithrombotic drugs for carotid artery dissection. Cochrane Database Syst Rev 2010;(10):CD000255.
- Kennedy F, Lanfranconi S, Hicks C, et al; CADISS Investigators. Antiplatelets vs anticoagulation for dissection: CADISS nonrandomized arm and meta-analysis. Neurology 2012;79:686-689.
- Markus HS. Author Response: Antiplatelets vs anticoagulation for dissection: CADISS nonrandomized arm and meta-analysis. Neurology 2013;80:970-971.
- Caprio FZ, Bernstein RA, Alberts MJ, et al. Efficacy and safety of novel oral anticoagulants in patients with cervical artery dissections. Cerebrovasc Dis 2014;38:247-253.
- Norris JW. Anticoagulants versus antiplatelet drugs for cervical artery dissection: Case for anticoagulants. J Neural Transm (Vienna) 2013;120:333-334.
- Gross BA, Albuquerque FC. Antiplatelets versus anticoagulation for cervical arterial dissection. Word Neurosurg 2015;84:18-25.
- Chowdhury MM, Sabbagh CN, Jackson D, et al. Antithrombotic treatment for acute extracranial carotid artery dissections: A meta-analysis. Eur J Vasc Endovasc Surg 2015;50:148-156.
- Gensicke H, Ahlhelm F, Jung S, et al. New ischaemic brain lesions in cervical artery dissection stratified to antiplatelets or anticoagulants. Eur J Neurol 2015;22:859-865,e61.
- Goyal N, Male S, Doss VT, et al. Spontaneous dissection of the bilateral internal carotid and vertebral arteries: A rationale for endovascular management. J Neurol Sci 2015;350:112-114.
- Chalouhi N, Starke RM, Tjourmakeris SI, et al. Carotid and vertebral artery sacrifice with a combination of onyx and coils: Technical note and case series. Neuroradiology 2013;55:993-998.
- Almendrote M, Millán M, Prats LA, et al. Impact of a comprehensive stroke centre on the care of patients with acute ischaemic stroke due to cervical artery dissection. Neurología (Barcelona, Spain) 2015;30:331-338.
- Grond-Ginsbach C, Metso TM, Metso AJ, et al. Cervical artery dissection goes frequently undiagnosed. Med Hypotheses 2013;80:787-790.
- Hassan AE, Jadhav V, Zacharatos H, et al. Determinants of neurologic deterioration and stroke-free survival after spontaneous cervicocranial dissections: A multicenter study. J Stroke Cerebrovasc Dis 2013;22:389-396.
- Ntaios G, Lambrou D, Cuendet D, Michel P. Early major worsening in ischemic stroke: Predictors and outcome. Neurocrit Care 2013;19:287-292.
- Speck V, Noble A, Kollmar R, Schenk T. Diagnosis of spontaneous cervical artery dissection may be associated with increased prevalence of posttraumatic stress disorder. J Stroke Cerebrovasc Dis 2014;23:335-342.
The goal of this paper is to help emergency physicians in their diagnosis, management, and treatment of this rare but potentially devastating condition.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
