Assessment of Tetanus Risk in the Pediatric Emergency Department
December 1, 2016
Reprints
Related Articles
-
Is Artificial Intelligence Coming for Your Job?
-
Can Pulmonary Artery Pressure Help in the Decision to Operate in Chronic Aortic Regurgitation?
-
Combined TAVR and LAAO Studied in a Randomized Controlled Trial
-
Another Hemodynamic Variant of Aortic Stenosis
-
Clinical Features of Tachyarrhythmia-Induced Cardiomyopathy
AUTHORS
Anthony R. Arredondo, DO, Resident, Department of Pediatrics, University of Texas Health Science Center San Antonio
Daniel J. Dire, MD, FACEP, FAAP, FAAEM, Clinical Professor, Departments of Emergency Medicine and Pediatrics, University of Texas Health Science Center San Antonio
PEER REVIEWER
James A. Wilde, MD, FAAP, Professor of Emergency Medicine, Associate Professor of Pediatrics, Augusta University, Augusta, GA
EXECUTIVE SUMMARY
- The following factors have contributed to the reduction in tetanus since 1900: 1) the use of tetanus antitoxin for wound management; and 2) the introduction of tetanus toxoid vaccines in the 1930s and 1940s, which led to universal childhood immunization and the addition of decennial tetanus toxoid boosters for adults.
- Inadequate immunization (non-immunized, partially immunized, or fully immunized individuals who fail to maintain adequate immunity with periodic booster doses) continues to be a vital cause of tetanus.
- Clostridium tetani is a rod-shaped, obligate anaerobic, gram-positive species that is motile and heat sensitive. The main site of infection is usually a wound, which can range anywhere from minor to severe, especially puncture wounds or contaminated wounds.
- Early on, tetanus may present as local muscular rigidity in the clostridium-containing wound or with dysphagia; however, the most common presentation is generalized muscle spasms. It usually follows a descending pattern, first causing trismus or “lockjaw” and then manifesting with muscle rigidity of the neck, throat, abdomen, and extremities. Reflex spasms also develop in most patients, triggered by minimal sensory stimuli.
- In cases of suspected tetanus, wounds should be cultured; however, only about 30% of cases actually recover C. tetani.
- The spatula test is a simple diagnostic bedside test that involves touching the oropharynx with a tongue blade (spatula). In normal circumstances, this elicits a gag reflex, and patients try to expel the blade (i.e., a negative test result). If tetanus is present, patients develop a reflex spasm of the masseters and bite the blade (i.e., a positive test result).
- The treatment of tetanus includes wound care, management of the symptoms and complications including treatment of muscle spasm, prevention of respiratory and metabolic complications, neutralization of circulating toxin to prevent the continued spread, and elimination of the source.
- In a sample of 200 patients, a comparison of the Tetanos Quick Stick (TQS) test to an enzyme-linked immunosorbent assay found the former had an 88.1% sensitivity and 97.6% specificity compared with the latter, and the positive and negative predictive values of the TQS test were 99.3% and 66.1%, respectively.
Tetanus is a life-threatening, preventable disease. It is most commonly acquired from a wound site; however, it can be obtained via injuries of other tissues. Both primary and secondary measures of prevention contribute to the eradication of tetanus. Therefore, appropriate tetanus prophylaxis should be administered in a timely fashion when patients present with wounds or infected tissue. Because tetanus prevention can be so effective, it is imperative to review, document, and address any tetanus deficiencies.
— Ann M. Dietrich, MD, FAAP, FACEP, Editor
Tetanus is a life-threatening but preventable disease that is characterized by an acute onset of painful muscular contractions (usually in a descending pattern, starting with trismus or lockjaw) and generalized rigidity.1 It is caused by the toxin of Clostridium tetani, a spore-forming bacterium. The spores can be found in soil, dust, and human and animal feces, entering the body through breaks in the skin and germinating under low-oxygen conditions.1,2 The most successful intervention against tetanus in history is prevention through effective vaccination, which has led to a dramatic decline in the incidence of tetanus.3
Tetanus occurs in all parts of the world, most frequently in hot and wet climates where the soil contains large amounts of organic matter. Although rare in the United States, with an average of 29 reported cases per year from 1996 through 2009, tetanus has not been eradicated by vaccination programs.2 While early diagnosis and intervention are lifesaving, prevention is the ultimate management strategy.4 According to the U.S. Centers for Disease Control and Prevention (CDC), nearly all cases of tetanus occur in children and adults who have never received a tetanus vaccine or adults who do not stay up-to-date on their 10-year booster shots.2
Although records from the fifth century B.C. contain clinical descriptions of tetanus, Carle and Rattone in 1884 first produced tetanus in animals by injecting them with pus from fatal human cases. During the same year, Nicolaier produced tetanus in animals by injecting them with soil. In 1889, Kitasato isolated the organism from a human victim, showed that it produced tetanus when injected into animals, and reported that the toxin could be neutralized by specific antibodies. In 1897, Nocard demonstrated the protective effect of passively transferred antitoxin; passive immunization in humans was used for treatment and prophylaxis during World War I. In the early 1920s, Ramon developed a method for inactivating tetanus toxin with formaldehyde, which led to the development of a toxoid by Descombey in 1924. This toxoid was first widely used during World War II.5
Background and Surveillance
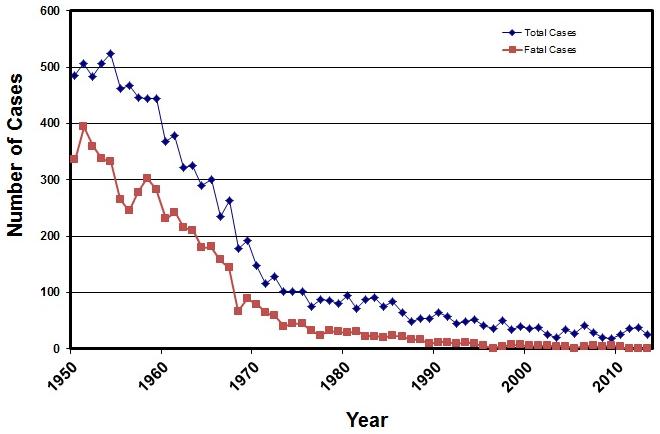
Since 1947, there has been a dramatic reduction in the number of tetanus cases in the United States; however, cases do continue to occur. (See Figure 1.) The number of cases per year has dropped from a range of 445-524 in the 1950s to a range of 26-37 from 2010-2013.6 Even the U.S. mortality rate has declined significantly, from a high of 394 deaths in 1951 to only three deaths in 2010.6 The following factors have contributed to this reduction in tetanus since 1900: 1) the use of tetanus antitoxin for wound management; and 2) the introduction of tetanus toxoid vaccines in the 1930s and 1940s, which led to universal childhood immunization and the addition of decennial tetanus toxoid boosters for adults. In addition to scheduled immunizations, which have helped increase maternal levels of immunity, improvement in childbirth practices (sanitary delivery) also has led to the near elimination of neonatal tetanus in the United States.4 Other factors contributing to decreased exposure include improved wound care management and increased rural-to-urban migration during the first half of the 20th century.1
Figure 1. Number of Total and Fatal Cases of Tetanus Reported to the CDC from 1955 to 20146 |
 |
Immunization almost exclusively has eliminated tetanus in the United States. However, inadequate immunization (non-immunized, partially immunized, or fully immunized individuals who fail to maintain adequate immunity with periodic booster doses) continues to be a vital cause of tetanus.1 From 1972 to 2008, only 142 out of 2,037 tetanus cases (excluding neonatal tetanus) had received three or more prior doses of tetanus toxoid-containing vaccines; however, just slightly more than half of those cases (1,185) had a known vaccination status, with 1,042 being inadequately vaccinated altogether.7,8
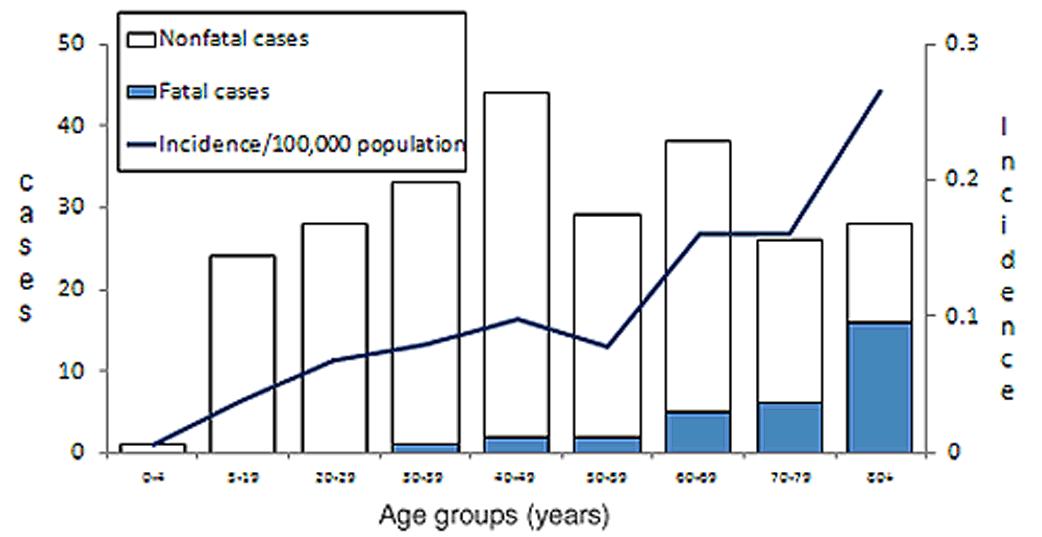
Currently, adults primarily are affected by tetanus because they have a higher rate of being unvaccinated or inadequately vaccinated.4 Between 2001 and 2008, about 90% (210 out of 233) of the tetanus cases were among persons aged 20 years or older, with the highest percentage of occurrence among those aged 20-64 years.8 However, the highest risk of mortality was among patients older than 65 years of age. (See Figure 2.) Despite the greater prevalence in adults, tetanus still occurs in children in the United States; 22 out of the same 233 cases were in patients younger than 20 years of age, including one case of neonatal tetanus.8 Therefore, it is imperative to review the immunization status of all patients who present to the emergency department, regardless of age and especially when there is any concern for an open wound.
Figure 2. Reported Cases of Tetanus, Survival Status of Patients, and Average Annual Incidence Rates by Age Group—United States, 2001-2009 |
 |
|
SOURCE: http://www.cdc.gov/vaccines/pubs/surv-manual/chpt16-tetanus.html |
The National Diseases Surveillance System (NNDSS) monitors national tetanus surveillance; however, it relies on physicians to report tetanus cases to state and local health departments, who then report these cases to the NNDSS.2,8,9 The reports are then transmitted to the CDC.
Worldwide, tetanus is predominantly a disease of underdeveloped countries located in warm, damp climates. Tetanus affects all age groups, with the highest prevalence found in newborns and young people. In 1992, an estimated 578,000 infant deaths were due to neonatal tetanus.4 In 1998, there were 215,000 tetanus deaths, with more than 50% occurence in people on the African continent.4 Overall, the annual incidence of tetanus is 0.5-1 million cases.10 The World Health Assembly first called for elimination of neonatal tetanus in 1989. In 1999, the goal was expanded to include elimination of maternal tetanus.11 At that time, 57 countries still had not eliminated maternal and neonatal tetanus. Since 2009, tetanus has been one of the target diseases of the World Health Organization Expanded Program on Immunization. As of 2016, there has been a 90% reduction in neonatal tetanus mortality since 1988.11
Etiology
Tetanus can be acquired outdoors as well as indoors. The incubation period usually ranges from three to 21 days, and averages about 7-10 days.12
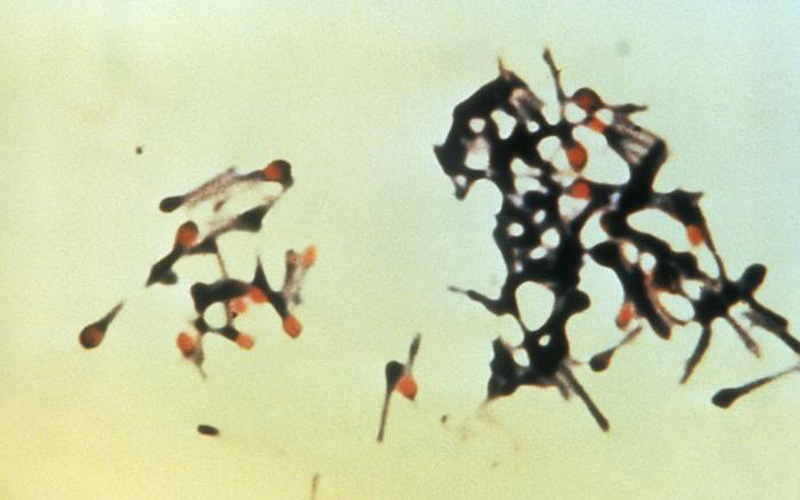
C. tetani is a rod-shaped, obligate anaerobic, gram-positive species that is motile and heat sensitive. It forms terminal spores that are resistant to heat and antiseptics, giving it the classic drumstick appearance on Gram stain.2 (See Figure 3.) The species is found mainly in soil and animal intestines; however, it also can be found in human feces, on skin surfaces, and in contaminated heroin.1,2,4
Figure 3. Micrograph Depicting Group of Clostridium tetani Spores |
 |
The main site of infection is usually a wound, which can range anywhere from minor to severe, especially puncture wounds or contaminated wounds.2,4 Often, treatment is not sought with the initial incident. The site of antecedent acute injury is the lower extremity (52%), upper extremity (34%), and head or trunk (5%).4 Other common ways tetanus enters the body are through burns and injuries with devitalized tissue.2 Less common routes of exposure include dental infections or procedures, surgical procedures, compound fractures, chronic sores and acute infections (skin ulcers, abscesses, gangrene), penetrating eye injuries, otitis media, tattoos, intramuscular/subcutaneous injections, and intravenous (IV) or subcutaneous illicit drug use.
See Table 1 for tetanus surveillance data in the United States (130 cases with one unknown injury history) between 1998 and 2000.9
Table 1. Tetanus Surveillance Data in the United States (1998-2000) |
|
Pathophysiology
Under favorable anaerobic conditions, such as in dirty, necrotic wounds, tetanus spores germinate and may produce tetanospasmin, an extremely potent neurotoxin.12 Tetanospasmin affects both the peripheral and central nervous systems and causes the clinical manifestations of tetanus.1,2,4,13 The estimated minimum lethal dose is 2.5 nanograms per kilogram of body weight.14 Once the toxin is produced, it disseminates via the blood and lymphatic system to eventually reach and enter all motor neurons.1,2,4 The toxin then travels in a retrograde manner from multiple nerve terminals to the spinal cord and ultimately into central inhibitory neurons.1,2,14,15
Once in the central inhibitory neurons, tetanospasmin interferes with the release of neurotransmitters,1,2,4 particularly the major inhibitory ones. Consequently, both gamma-aminobutyric acid (GABA)-containing and glycine-containing vesicles are not released into the synapses, leading to the loss of central inhibitory impulses on both motor and autonomic neurons.15 This results in uncontrolled muscle spasms in response to normal sensory stimuli (touch, loud noises, and bright lights) and autonomic dysfunction (hyper/hypotension, tachy/bradycardia, etc.).2,4,13,16
The shortest peripheral nerves are the first to deliver the toxin to the central nervous system, which leads to early symptoms of facial distortion and back and neck stiffness. Once the toxin becomes fixed to neurons, it cannot be neutralized with antitoxin. Recovery of nerve function from tetanus toxins requires sprouting new nerve terminals and forming new synapses to then allow appropriate release of inhibitory neurotransmitters.4 Complete recovery could take months.2
Clinical Presentation
Early on, tetanus may present as local muscular rigidity in the clostridium-containing wound or with dysphagia; however, the most common presentation is generalized muscle spasms.2,4 It usually follows a descending pattern, first causing trismus or “lockjaw” and then manifesting with muscle rigidity of the neck, throat, abdomen, and extremities.1,2,4,17 Reflex spasms also develop in most patients, triggered by minimal sensory stimuli.4 The spasms can last for seconds to minutes, continue for three to four weeks, increase in intensity or frequency, and cause complications such as apnea (intercostal and diaphragm involvement), asphyxia (laryngeal involvement), fractures, dislocations, or rhabdomyolysis.2,4 Other presenting complaints or symptoms include headache, rapid heart rate, sweating, restlessness, and elevated blood pressure.2,3,13,16 Autonomic dysfunction becomes progressively evident as the level of toxin in the central nervous system increases.3,4,16
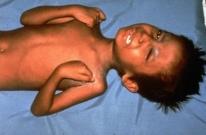
Risus sardonicus is a condition in which there is sustained contraction of facial muscles, creating a sneering grin expression.4,13 (See Figure 4.) Severe tetanus can result in opisthotonos, which is backward arching of the head, neck, and spine due to severe muscular spasms. Tetanic seizures also may develop, usually indicating a poorer prognosis if present since the frequency and severity of seizures are related to severity of the disease.2,4 These seizures seem to resemble epileptic seizures with the sudden onset of tonic contractions; however, there is no loss of consciousness, and the patient is usually in severe pain. 2,4
Figure 4. Risus sardonicus |
|
Risus sadonicus is a fixed “sarcastic” grimace and anxious expression, with drawing up of the eyebrows and corners of the mouth due to spasms of the masseter and other facial muscles, usually accompanied by rigidity of neck and trunk muscles and arching of back.
|
|
SOURCE: http://www.immunizenevada.org/vaccines/vaccine-preventable-diseases |
Clinical Types of Tetanus
Tetanus can be categorized into the following four clinical types: generalized, localized, cephalic, and neonatal.
Generalized Tetanus
The initial presenting symptom of generalized tetanus, which is the most common form seen in 75-81% of cases, typically is trismus or “lockjaw.”1,2,4,17 Other than a primary care physician, a dentist or oral surgeon may be the first person to see the patient. The muscle rigidity then starts to progress downward from the face.3 Depending on the distance from the site of the injury to the central nervous system, symptoms may manifest earlier or later, correlating with a broad incubation period of 3-21 days.5 Generalized tetanus also can affect the autonomic nervous system and includes symptoms such as diaphoresis, hyperthermia, arrhythmias, and fluctuating blood pressure.1,3,5,16,17 Patients remain lucid in most cases. Other possible features include reflex spasms, sustained facial contraction (risus sardonicus), opisthotonos, and generalized seizure-like activity.3,4,5,12
Localized Tetanus
The next most common presentation of tetanus is localized tetanus, which occurs in up to 15% of cases.4,5 Localized tetanus generally involves the extremity with the contaminated wound and has a highly variable severity.2 In milder cases, patients may have some persistent muscular contraction; in more severe cases, they may have intense, painful spasms.2,4,12 Local tetanus may persist for several weeks, but it usually is self-limited, with only about 1% of cases resulting in death.1,4,5 More severe cases even may precede the onset of generalized tetanus.2,5
Cephalic Tetanus
Cephalic tetanus is a rare form of the disease that usually is associated with chronic otitis media or head trauma, and it has a poor prognosis.2,4,5 Involvement of the cranial nerves typically results, especially in the facial region (most frequently CN VII).2,5 Ophthalmoplegic tetanus is a variant that develops after penetrating eye injuries and results in CN III palsies and ptosis.18 Cephalic tetanus usually has a shorter incubation period of 1-2 days with a higher risk of mortality and can progress quite rapidly.2 Like localized tetanus, cephalic tetanus can remain localized or develop into generalized tetanus if untreated.2,4
Neonatal Tetanus
Although it is extremely rare in the United States, neonatal tetanus is still common in developing countries and has a very poor prognosis.2,19 Worldwide, this disease accounts for 5-7% of neonatal mortality, but the mortality is as high as 50% in some developing nations.20 Neonatal tetanus is a form of generalized tetanus that occurs in newborns, resulting from infection.2 The usual causative factor is the use of contaminated (unsterile) delivery instruments when dealing with the umbilical cord of newborns of unimmunized mothers (resulting in no neonatal protective passive immunity).2,19 This disease is sometimes referred to as “the disease of the seventh day” because the typical incubation period is 3-14 days after birth, with an average of about seven days.2,12 Presenting symptoms include the inability to suck, poor feeding, irritability, rigidity, facial grimacing, severe spasms, and opisthotonos.4,12 The World Health Organization defines neonatal tetanus as an illness occurring in a neonate who has the normal ability to suck and cry in the first two days of life, but who loses this ability between days three and 28 and becomes rigid and has spasms.12 In neonates, sepsis can accompany tetanus, exacerbating the severity of illness.20 The mortality rate is very high, exceeding 70%, but it is only 10-20% when modern ICU care is available.12,20 Some survivors of neonatal tetanus have been reported to have neurological damage, ranging from cerebral palsy and severe psychomotor retardation to subtle intellectual and behavioral abnormalities.20
Diagnosis
The diagnosis of tetanus is made strictly on clinical grounds.20 In cases of suspected tetanus, wounds should be cultured; however, only about 30% of cases actually recover C. tetani.1 It is important to note that C. tetani can be isolated from the wounds of patients who do not have tetanus.1 The differential diagnosis includes other causes of trismus. (See Table 2.) Seizures, meningitis, and metabolic disorders, such as hypoglycemia and hypocalcemia, must be ruled out in neonates.20
Table 2. Differential Diagnosis of Tetanus 3,20 |
|
No laboratory tests specific for the diagnosis of tetanus exist.4,12 A moderate peripheral leukocytosis may be present on a complete blood count along with an increased opening pressure on cerebrospinal fluid (CSF) studies, especially during muscular spasms; otherwise, these laboratory studies are unremarkable. In addition, elevated serum levels of muscle enzymes (creatine kinase, aldolase) may occur.
Serologic testing is another consideration. The minimum level of protective antibody in the normal population is between 0.01 and 0.15 IU/mL. The majority of vaccinated individuals should demonstrate protective levels of antibody > 0.15 IU/mL. After a complete primary series of tetanus vaccination (three properly spaced doses in persons 7 years of age and older, or four doses in children younger than 7 years of age), nearly 100% of patients achieve protective levels in their blood.2 Antitoxin levels, however, decrease over time, thus leading to the recommendation of routine tetanus boosters every 10 years.2 Although quite rare, fatal tetanus cases still occur despite having protective levels; therefore, serology alone cannot exclude the diagnosis of tetanus.1,20 Unfortunately, assays for antitoxin levels are not readily available to emergency physicians.
The spatula test is a simple diagnostic bedside test that involves touching the oropharynx with a tongue blade (spatula).21 In normal circumstances, this elicits a gag reflex, and patients try to expel the blade (i.e., a negative test result). If tetanus is present, patients develop a reflex spasm of the masseters and bite the blade (i.e., a positive test result). In 400 patients, this test had a sensitivity of 94% and a specificity of 100%. No adverse sequelae (e.g., laryngeal spasm) were reported.21
Treatment
The treatment of tetanus includes wound care, where required, as well as management of the symptoms and complications directed toward the treatment of muscle spasm, prevention of respiratory and metabolic complications, neutralization of circulating toxin to prevent the continued spread, and elimination of the source.4,12 Tetanus is a medical emergency, usually requiring admission to an ICU. Because of the risk of reflex spasms, the patient should stay in a dark and quiet environment. Clinicians should avoid unnecessary procedures and manipulations.
Hospitalization, along with the administration of both tetanus toxoid vaccine and tetanus immunoglobulin (TIG), is required in addition to other supportive measures.2,12 TIG helps neutralize any unbound toxin and may prevent further progression, but it may not ameliorate the existing symptoms.12 A dose of 3,000-10,000 units intramuscularly should be given as soon as the diagnosis of tetanus is considered, with part of the dose infiltrated around the wound.4,22 Management also includes antispasmodic medications, aggressive wound care, antibiotics, and the ability to maintain an adequate airway.2,22 Intubation and ventilation is required in 67% of patients.4 Adequate hydration and nutrition are also important, especially since tetanus can result in such high metabolic demands.
Antibiotics are recommended universally for patients with tetanus, although their efficacy in altering the clinical course is unclear.4,22 Metronidazole is the preferred drug of choice, but penicillin G also is acceptable.
Generalized muscle spasms are life-threatening since they can cause respiratory failure, lead to aspiration, and induce generalized exhaustion in patients.22 Several drugs may be used to control these spasms; benzodiazepines (such as diazepam) are the drugs of choice. The dosing is adjusted according to the clinical response, but large doses or prolonged continuous IV infusion may be needed in severe cases. There is a risk of precipitating metabolic acidosis due to the preservative propylene glycol when using large doses of diazepam.3 Benzodiazepines should be tapered gradually as the patient recovers to avoid benzodiazepine withdrawal syndrome.
Baclofen is a central acting GABA-B receptor agonist, but it has poor penetration across the blood–brain barrier and hence is ineffective in tetanus.3 Intrathecal baclofen has been used anecdotally and in case series to wean patients off the ventilator and stop the diazepam infusion.4 Intrathecal baclofen is 600 times more potent than orally administered baclofen. Repeated intrathecal injections have been efficacious in limiting the duration of artificial ventilation or preventing intubation. The daily baclofen dose infused has ranged from 500-2,000 µg in many case reports. At these doses, respiratory depression as well as cardiovascular instability were observed.3
Magnesium sulfate (dosage below) or morphine sulfate (0.5 to 1.0 mg/kg per hour IV continuous infusion) can help with autonomic dysfunction control and painful spasms, while antihypertensive medications, such as beta-blockers, also may assist with autonomic control.13,16,23,24 In a randomized, double-blind trial in 256 patients with severe tetanus in Vietnam, magnesium sulfate infusion was compared with placebo.25 The patients were randomly assigned to magnesium sulfate (loading dose 40 mg/kg over 30 minutes, followed by continuous infusion of either 2 g/hr for patients > 45 kg or 1.5 g/hr for patients ≤ 45 kg) vs. placebo. Magnesium infusion significantly reduced the requirement for other drugs to control muscle spasms, and patients treated with magnesium were 4.7 times less likely to require verapamil to treat cardiovascular instability (95% confidence interval, 1.4-15.9). Magnesium sulfate infusion did not reduce the need for mechanical ventilation.
Labetalol (0.25-1.0 mg/min) frequently has been administered to tetanus patients in the ICU because of its dual alpha- and beta-blocking properties. Beta-blockade alone (e.g., propranolol) should be avoided because of reports of sudden death.22
Tetanus is one of the few bacterial diseases that does not confer immunity following recovery; therefore, patients still require active immunization with a tetanus toxoid vaccine to help prevent recurrence.2,5
Complications
Prior to 1954, asphyxia from tetanic spasms was the usual cause of death; however, with the advent of neuromuscular blockers, mechanical ventilation, and pharmacological control of spasms, sudden cardiac death has become the leading cause of death. This has been attributed to excessive catecholamine productions, direct action of tetanospasmin or tetanolysin on the myocardium. Other complications include: long bone fractures, glenohumeral joint and temporomandibular joint dislocations, hypoxic injury, aspiration pneumonia, pulmonary emboli, paralytic ileus, pressure sores, urinary retention, malnutrition, stress ulcers, coma, nerve palsies, neuropathies, psychological after-effects, and flexion contractures.2,4,5
Prognosis
Tetanus tends to be less severe among patients who have received a primary series of tetanus vaccines compared to those who are partially vaccinated or unvaccinated. The prognosis depends on the incubation period, the time from spore inoculation to first symptom, the time from first symptom to first tetanic spasm, or any associated secondary complication.4 In general, shorter intervals indicate more severe tetanus and a poorer prognosis. Interestingly, shorter incubation periods (< 7 days) are associated with more contaminated wounds, more severe disease, the development of complications, and a higher risk of mortality.2,26 Although recovery can be quite slow (around 2-4 months), patients usually return to their baseline state of health.
Historically, the overall mortality rate for generalized tetanus in the United States was 30% and higher (52%) for patients older than 60 years of age, with both cephalic and neonatal tetanus being more severe and having higher mortality rates. 4 However, in the last 20 years, the reported U.S. mortality rates have been less than 5%. (See Figure 1.) From 2001-2008, 77% of U.S. deaths were in patients older than 60 years of age.8 Other associated risk factors for mortality include patients who require mechanical ventilation, contaminated lesions of the face and head, and early onset of convulsions.4 Autonomic dysfunction tends to correlate with a higher rate of mortality as well.27
Vaccination and Prevention
Protection against tetanus is antibody-dependent and can be achieved only through active (tetanus vaccine) or passive (tetanus-specific immunoglobulin) immunization.12 Neonates born to a properly immunized mother develop a protective passive immunity, in which maternal antibodies wane over a period of 6-12 months. Therefore, beyond early infancy, active immunity via a primary vaccine series is required for further protection, in addition to subsequent timely booster doses — hence primary prevention.1,2 Passive protection with anti-tetanus antibody (TIG) may be required, depending on a patient’s vaccination status and type of wound, a component of secondary prevention. (See Figure 5.)
Figure 5. Summary Guide to Tetanus Prophylaxis in Routine Wound Management |
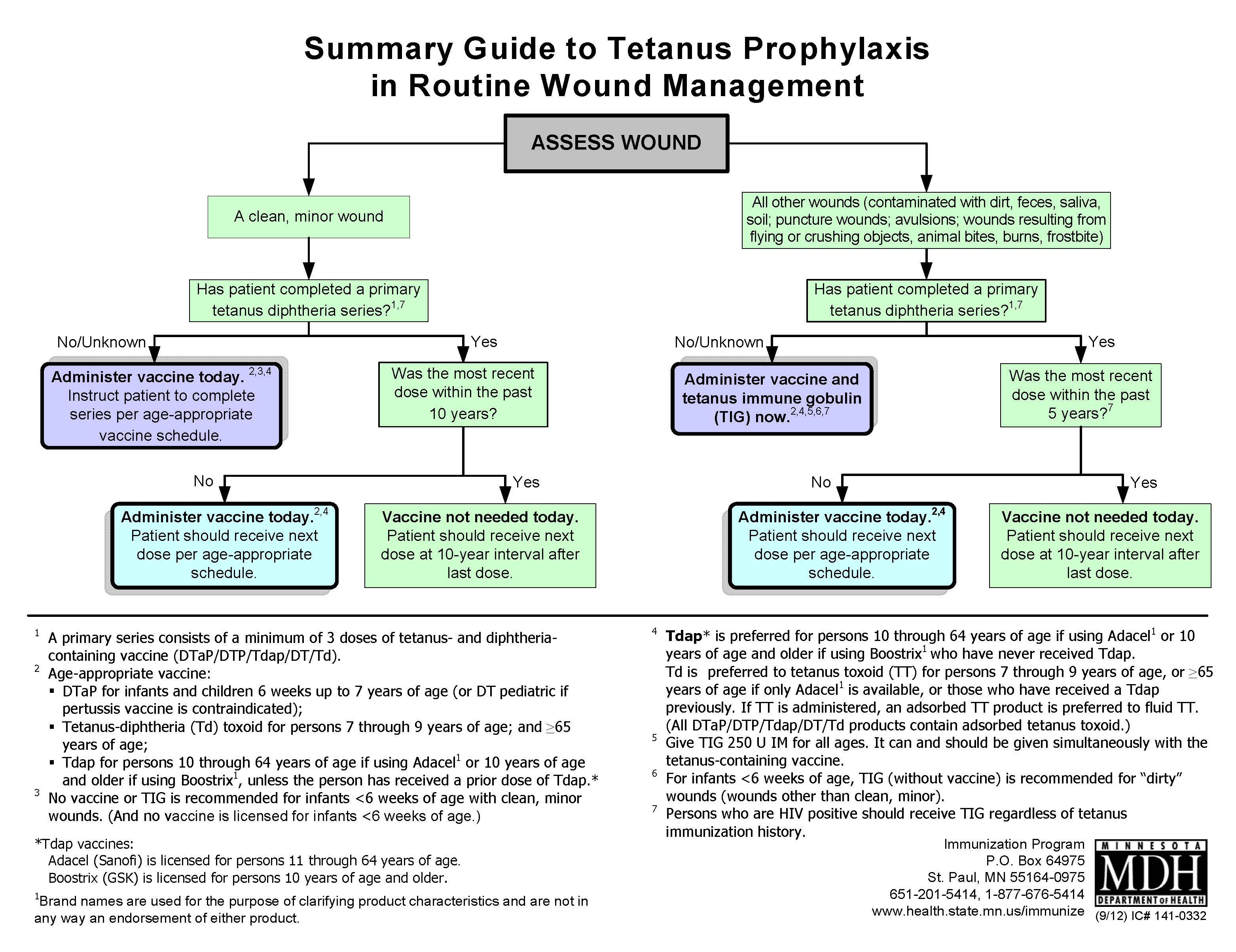 |
Various formulations of tetanus toxoid-containing vaccines are available in the United States. (See Figure 6.) Tetanus toxoid is available in combination with both diphtheria and acellular pertussis in both pediatric and adult formulations.2 DTaP (tetanus with diphtheria toxoids and acellular pertussis) and DT (diphtheria and tetanus toxoids) are licensed for infants and children younger than 7 years of age, while Td (tetanus and diphtheria toxoids), Tdap (tetanus with diphtheria toxoids and acellular pertussis), and TT (tetanus toxoid) are licensed for both children 7 years of age and older and adults.1,2 In 2005, two brands of Tdap, Boostrix (now approved for patients 10 years of age and older) and Adacel (approved for patients 11-64 years of age) were licensed for adolescents and adults; however, according to the CDC, either Tdap product may be used in patients 65 years of age and older.1,2,28,29 Pediarix (DTaP-IPV-HepB) and Pentacel (DTaP-IPV-Hib) are other pediatric combination vaccines that include tetanus and other antigens.
Figure 6. Available Tdap- and DTaP-containing Vaccinations |
 |
Pediatric formulations (DT and DTaP) have a similar amount of tetanus toxoid as adult Td, but contain three to four times as much diphtheria toxoid.2 Tetanus toxoid should always be administered in combination with diphtheria toxoid given that periodic boosting is needed for both diseases.2
Primary Prevention
Primary tetanus vaccination is recommended for all infants and children aged 6 weeks through 6 years, with DTaP being the preferred choice.1,2 This primary prevention consists of a three-dose series, administered at ages 2 months, 4 months, and 6 months, followed by two booster doses at 12-18 months (fourth dose) and 4-6 years (fifth dose). There is a minimum interval of four weeks between each of the first three doses. The fourth dose must be given at least six months after the third dose. The fifth dose is not necessary if the fourth dose was administered at age 4 years or older. A single dose of Tdap then is recommended for adolescents at age 11-18 years if they have not previously received Tdap, followed by routine Td booster vaccination every 10 years for adults. For adults aged 19 years and older who previously have not received a dose of Tdap, a single dose of Tdap should replace a single decennial Td booster dose.1,22,28,29
For patients with a history of never being vaccinated or having incomplete or unknown tetanus vaccination, a primary three- or four-dose series is recommended for ages 7 years and older and for those younger than 7 years of age, respectively.1,22,28,29 For children younger than 7 years of age, DTaP should be given every four weeks between each of the first three doses along with a fourth (booster) dose given at least six months after the third dose, and a fifth (booster) dose at 4-6 years of age with at least a six-month interval after the fourth dose (again, the fifth dose is not necessary if the fourth dose was given at 4 years of age or older).1,2,30 Children 7 years of age and older should receive Tdap as the initial dose, followed by Td (the adult formulation) as needed in the catch-up series. The preferred schedule for children 7 years and older, after the initial dose of Tdap, is a dose of Td given at least four weeks later, followed by another dose of Td in 6-12 months.1,2,30 The routine Td boosters then are recommended every 10 years.
In patients with an uncertain vaccination history, consider serologic testing for antibody titers to tetanus (protective level of > 0.1 IU/mL) or using a Tetanos Quick Stick (TQS; Nephrotek Laboratory, Rungis, France), a bedside immunochromatographic dipstick test, with the goal of avoiding unnecessary vaccination. In a sample of 200 patients, a comparison of the TQS test to an enzyme-linked immunosorbent assay found the former had an 88.1% sensitivity and 97.6% specificity compared with the latter, and the positive and negative predictive values of TQS test were 99.3% and 66.1%, respectively.31 The authors noted the TQS test to be appropriate and cost-effective for emergency department use in evaluating patients who do not remember or cannot give their tetanus immunization history.31
In 2010, the Advisory Committee on Immunization Practices approved additional recommendations for the use of Tdap: 1) use of Tdap regardless of interval since the last tetanus- or diphtheria-toxoid containing vaccine; 2) use of Tdap in certain adults aged 65 years and older; and 3) use of Tdap in undervaccinated children ages 7 through 10 years.29,32 Part of the recommendation stems from the recent increase of pertussis in the United States, therefore facilitating the use of Tdap to reduce the burden of disease and risk for transmission to infants.29,32 In 2011, the FDA approved an expanded age indication for Tdap to include persons aged 65 years and older.28
One dose of Tdap vaccine is recommended for pregnant adolescents during each pregnancy, preferably during the third trimester between 27-36 weeks’ gestation; however, it can be given at any time, regardless of the time interval since the last Td or Tdap vaccination.30,32 This recommendation aims to optimize strategies for preventing pertussis morbidity and mortality in infants. For pregnant women as part of standard wound management, a tetanus booster may be administered if five or more years have elapsed since the last tetanus shot. If a tetanus booster is indicated, Tdap should be administered. Pregnant women with an unknown or incomplete tetanus vaccination history should be treated the same as patients 7 years of age and older, as discussed above.
The safety profile of tetanus vaccination is good. Most reactions are mild and local (fever, erythema, swelling, and soreness), usually self-limited, and require no therapy.33 Other moderate conditions, such as an exaggerated local (Arthus-like) reaction, and seizures are not common, while more severe reactions, such as a severe allergic reactions, are quite rare. Anaphylaxis following a prior tetanus vaccine is a contraindication to subsequent doses, unless the risk of withholding the tetanus vaccine for certain wounds and significant illness outweighs the risk of vaccine administration.2
Secondary Prevention
Secondary prevention of tetanus, which varies with previous vaccination history, is accomplished post-exposure through wound prophylaxis and administration of TIG and toxoid. Tetanus-prone wounds include grossly contaminated wounds, wounds that have been present for more than six hours, deep wounds (> 1 cm), wounds exposed to saliva or feces, stellate-shaped wounds, and ischemic or infected wounds (including abscesses, avulsions, punctures, or crush injuries).4 If indicated, debridement should be undertaken only after the patient has been stabilized, and any wound manipulation should be delayed until several hours after antitoxin administration because of the risk of releasing tetanus toxin into the bloodstream. Excision of at least 2 cm of healthy-appearing tissue around the wound margins is recommended. Abscesses should be treated with incision and drainage when possible.
In a wound that is not tetanus-prone, patients should be given a tetanus toxoid-containing vaccine (Td or Tdap) if they have received fewer than three doses of tetanus toxoid previously or if more than 10 years have passed since their last dose. (See Figure 5.) Tdap is preferred to Td for those who have not previously received Tdap, and is indicated only once with subsequent Td doses.28,29
Patients with tetanus-prone wounds should receive a tetanus toxoid (Td or Tdap) if they have received fewer than three prior doses of tetanus toxoid or if it has been more than five years since their last dose. Additionally, TIG (250-500 units IM) also is indicated in patients who have previously received fewer than three doses of tetanus toxoid, always in the opposite extremity to the tetanus vaccine. Although not shown in Figure 5, patients 60 years of age and older also should receive TIG, especially if they are not current on their tetanus immunization.4
Summary
Although tetanus can be life-threatening, it is a preventable disease. Tetanus is most commonly acquired from a wound site; however, it can be obtained via injuries of other tissues. Tetanospasmin, the neurotoxin responsible for the clinical manifestations, can affect the nervous system by suppressing the inhibitory regulating neurotransmitters (GABA and glycine), causing anywhere from mild-to-severe muscle spasms in addition to other complications. Both primary and secondary measures of prevention contribute to the eradication of tetanus. Therefore, appropriate tetanus prophylaxis should be administered in a timely fashion when patients present with wounds or infected tissue.
Because tetanus prevention can be so effective, it is imperative to review and appropriately document the immunization status of all patients who present to the emergency department for any care (regardless of chief complaint) and to update tetanus vaccination status accordingly. If more than 10 years have elapsed since their last tetanus shot, a booster dose should be administered in the ED.
REFERENCES
- Tejpratap SP, Tiwari MD. Tetanus. In: Manual for the Surveillance of Vaccine-Preventable Diseases. 6th ed. Centers for Disease Control and Prevention, Atlanta, GA; 2013.
- Centers for Disease Control and Prevention. Tetanus. Available at: http://www.cdc.gov/tetanus/index.html. Updated: Jan. 9, 2013. Accessed Oct. 3, 2016.
- Rodrigo C, Ferando D, Rajapakes S. Pharmacological management of tetanus: An evidence-based review. Crit Care 2014;18:217-226.
- Dire DJ. Tetanus. eMedicine Journal. May 18, 2001. Available at: http://www.vaccinationnews.org/DailyNews/May2001/TetanusEMedicine.htm. Accessed Oct. 11, 2016.
- Hamborsky J, Kroger A, Wolfe C. Tetanus. In: Epidemiology and Prevention of Vaccine-Preventable Diseases. 13th ed. Centers for Disease Control and Prevention, Atlanta, GA; 2015.
- Hamborsky J, Kroger A, Wolfe C. Appendix E: Data and Statistics. In: Epidemiology and Prevention of Vaccine-Preventable Diseases. 13th ed. Centers for Disease Control and Prevention, Atlanta, GA; 2015.
- Srivastava P, Brown K, Chen J, et al. Trends in Tetanus Epidemiology in the United States, 1972-2001 (online presentation). Available at: https://cdc.confex.com/cdc/nic2005/techprogram/paper_7813.htm. March 22, 2005.
- Centers for Disease Control and Prevention. Tetanus surveillance — United States, 2001-2008. MMWR Morb Mortal Wkly Rep 2011:60:365-369.
- Centers for Disease Control and Prevention. Pascual FB, McGinley EL, Zanardi LR, et al. Tetanus surveillance — United States, 1998-2000. MMWR Surveil Summ 2003;52:1-8
- World Health Organization. Tetanus Fact Sheet. Available at: http://www.wpro.who.int/mediacentre/factsheets/fs_20120307_tetanus/en/. March 7, 2012. Updated Feb. 23, 2016. Accessed Oct. 14, 2016.
- UNICEF. Elimination of Maternal and Neonatal Tetanus. Available at: http://www.unicef.org/health/index_43509.html. Updated July 1, 2016. Accessed Oct. 22, 2016.
- World Health Organization. Tetanus Vaccine: WHO Position Paper. Weekly Epidemiol Rec 2006;81:197-208.
- World Health Organization. Current recommendations for treatment of tetanus during humanitarian emergencies. Available at: http://www.who.int/diseasecontrol_emergencies/publications/who_hse_gar_dce_2010.2/en/. January 2010. Accessed Oct. 3, 2016.
- Gill DM. Bacterial toxins: A table of lethal amounts. Microbiol Rev 1982;46:86-94.
- Yeh FL, Dong M, Yao J, et al. SV2 mediates entry of tetanus neurotoxin into central neurons. PLoS Pathog 2010;6:e1001207.
- Lin T, Chen L, Lin T, et al. Autonomic dysfunction because of severe tetanus in an unvaccinated child. Pediatr Neonatol 2011;52:169-171.
- Pearce JM. Notes on tetanus (lockjaw). J Neurol Neurosurg Psychiatry 1996;60:332-332.
- Saltissi S, Hakin RN, Pearce J. Ophthalmoplegic tetanus. Br Med J 1976;1:437.
- Glezen WP. Prevention of neonatal tetanus. Am J Public Health 1998;88:871-872.
- Roper MH, Vandelaer JH, Gasse FL. Maternal and neonatal tetanus. Lancet 2007;370:1947-1959.
- Apte NM, Karnad DR: Short report: The spatula test: A simple bedside test to diagnose tetanus. Am J Trop Med Hyg 1995;53:386-387.
- Sexton DJ. Tetanus. In: UpToDate. Bartlett JG, Thorner AR (eds). Available at: http://www.uptodate.com/contents/tetanus. Updated April 28, 2016. Accessed Oct. 5, 2016.
- Ceneviva GD, Thoma NJ, Kees-Folts, D. Magnesium sulfate for control of muscle rigidity and spasms and avoidance of mechanical ventilation in pediatric tetanus. Pediatr Crit Care Med 2003;4:480-484.
- Rodrigo C, Samarakoon L, Fernando SD, et al. A meta-analysis of magnesium for tetanus. Anaesthesia 2012;67:1370-1374.
- Thwaites CL, Yen LM, Loan HT, et al. Magnesium sulphate for treatment of severe tetanus: A randomised controlled trial. Lancet 2006;368:1436.
- Patel JC, Mehta BC. Tetanus: A study of 8697 cases. Indian J Med Sci 1999;53:393-401.
- Cook TM, Proteroe PT, Heindel JM. Tetanus: A review of the literature. Br J Anaesth 2001;87:477-487.
- Centers for Disease Control and Prevention. FDA Approval of expanded age indication for a tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccine. MMWR 2011;60:1279-1280.
- Centers for Disease Control and Prevention. Updated Recommendations for Use of Tetanus Toxoid, Reduced Diphtheria Toxoid and Acellular Pertussis (Tdap) Vaccine from the Advisory Committee on Immunization Practices, 2010. MMWR 2011;60:13-15.
- Centers for Disease Control and Prevention. Catch-up Schedule. Available at: http://www.cdc.gov/vaccines/schedules/hcp/imz/catchup.html. Updated Feb. 1, 2016. Accessed Oct. 4, 2016.
- Hatamabadi HR, Abdalvand A, Safari S, et al. Tetanus quick stick as an applicable and cost-effective test in assessment of immunity status. Am J Emerg Med 2001;29:717-712.
- Centers for Disease Control and Prevention. Updated Recommendations for Use of Tetanus Toxoid, Reduced Diphtheria Toxoid, and Acellular Pertussis Vaccine (Tdap) in Pregnant Women — Advisory Committee on Immunization Practices (ACIP), 2012. MMWR 2013;62:131-135.
- Centers for Disease Control and Prevention. Possible Side-effects from Vaccines. (n.d.). Available at: http://www.cdc.gov/vaccines/vac-gen/side-effects.htm#dtap. Updated July 20, 2016. Accessed Oct. 3, 2016.
Tetanus is a life-threatening, preventable disease. It is most commonly acquired from a wound site; however, it can be obtained via injuries of other tissues. Both primary and secondary measures of prevention contribute to the eradication of tetanus. Therefore, appropriate tetanus prophylaxis should be administered in a timely fashion when patients present with wounds or infected tissue. Because tetanus prevention can be so effective, it is imperative to review, document, and address any tetanus deficiencies.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.