Understanding and Managing Angioedema in the Emergency Department
December 1, 2016
Reprints
AUTHORS
Daniel Fisher, DO, FACEP, Clinical Assistant Professor, Division of Emergency Medicine, Medical University of South Carolina, Charleston.
Hesham F. Abukhdeir, MD, MPH, Division of Emergency Medicine, Medical University of South Carolina, Charleston.
PEER REVIEWER
Joseph J. Moellman, MD, Professor of Emergency Medicine, University of Cincinnati College of Medicine, Medical Director, Deer Park/Silverton EMS.
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Farel (CME question reviewer) owns stock in Johnson & Johnson. Dr. Stapczynski (editor) owns stock in Pfizer, Johnson & Johnson, Walgreens Boots Alliance Inc., GlaxoSmithKline, Bristol Myers Squibb, and AxoGen. Dr. Moellman (peer reviewer) reports that he has received research/grant support from Shire, Janssen, J&J, Medtronic, Gilead, and Becton/Dickinson. Dr. Schneider (editor), Ms. Fessler (nurse planner), Dr. Fisher (author), Dr. Abukhdeir (author), Ms. Mark (executive editor), Ms. Coplin (executive editor), and Mr. Landenberger (editorial and continuing education director) report no financial relationships with companies related to the field of study covered by this CME activity.
EXECUTIVE SUMMARY
- Allergic angioedema, particularly if associated with anaphylaxis or airway compromise, should be treated with IM epinephrine 0.01 mg/kg (up to 0.3 mg in children and 0.5 mg in adults). Of note, the labeling of epinephrine recently has changed. The drug previously labeled 1:1000 is now labeled
1 mg/mL. - Patients with hereditary angioedema and severe disease generally are treated with icatibant or ecallantide.
- Most patients with hereditary angioedema have decreased C1-INH levels. Drawing a C4 and C1-INH level during an acute attack can be helpful.
- Unlike allergic reactions that are mediated by histamine, hereditary angioedema is mediated by bradykinin. Epinephrine, diphenhydramine, and corticosteroids do not reverse the disease. They do not worsen it either. So if there is doubt, patients with airway symptoms and no prior diagnosis should receive epinephrine.
After completing this educational activity, participants should be able to recognize and demonstrate an understanding of angioedema, discuss both the differential diagnosis and range of complications associated with the condition, and apply state-of-the-art diagnostic and therapeutic techniques in the evaluation and management of angioedema in the emergency department (ED).
Introduction
Angioedema is the swelling of the deep dermal, subcutaneous, and submucosal tissues due to increased vascular permeability. The first detailed account of angioedema was documented in 1876 by Dr. John Laws Milton in London, who originally called the condition “giant urticaria.” In 1882, Heinrich Quincke provided another description of angioedema, which subsequently was given its original name of “Quincke’s disease.”1 In 1888, William Osler identified hereditary angioedema (HAE) as a separate disease after studying a family with bouts of angioedema spanning five generations, and in 1963, Donaldson and Evans reported the cause of HAE as decreased levels of C1 esterase inhibitor (type I).1-4 A second form of HAE was found with normal C1 esterase inhibitor levels but abnormal function, leading to the discovery that both forms were related to a mutation in the C1 inhibitor gene (SERPING1) (type II). However, a new form of HAE has been found with normal SERPING1.1
In addition to the hereditary deficiencies of C1 inhibitor, there are also acquired types. These are thought to be secondary to autoantibodies resulting from existent disease processes, with lymphoproliferative disorders being the most common etiology.1 Additional forms include angiotensin-converting enzyme (ACE) inhibitor-induced angioedema and non-histaminergic idiopathic angioedema. The unifying theme for these variants revolves around the regulation of bradykinin. Another form of angioedema is histamine-mediated, as is seen in acute allergic reactions and anaphylaxis.
The peptide bradykinin and amine histamine are both powerful vasodilators, and excess levels can cause vascular permeability. This vascular permeability can result in transient swelling in the deep dermis and subcutaneous layers of the skin, as well as the submucosal layer of the respiratory and gastrointestinal tracts. Angioedema, also known as angioneurotic and Quincke’s edema, describes the swelling that occurs in these particular tissue layers secondary to conditions of excess histamine or bradykinin.5
Normally, angioedema is non-pitting, can be skin-colored or erythematous, and is often in areas where the skin is loose (i.e., face, genitalia). (See Figures 1 and 2.) Heat and pain may be present with the swelling, but it generally is not pruritic.2 A similar type of reaction is seen in urticaria; however, urticaria is swelling due to vascular permeability that occurs in more superficial layers of the skin. It is typically pruritic. For emergency medicine physicians, it is important to know the similarities and differences between urticaria and angioedema. (See Table 1.)
Figure 1. Extremity Urticaria
This patient photograph shows the localized erythematous wheals typical in urticaria. Reproduced with permission from The Urtikaria Network
© www.urtikaria.net.
Figure 2. Angioedema of the Lips
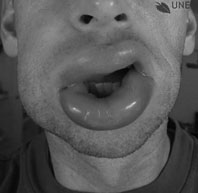
This patient photograph demonstrates the localized submucosal swelling seen in acute exacerbations of angioedema.
Reproduced with permission from The Urtikaria Network © www.urtikaria.net.
Table 1. Comparison of Features of Angioedema and Urticaria2 |
||
Feature |
Urticaria |
Angioedema |
|
Location |
Skin only |
Skin and mucosa, particularly the eyelids and lips |
|
Duration |
Transitory (usually lasts |
Transitory (usually lasts between 24-48 hours) |
|
Physical signs |
Red patches and wheals |
Pink or skin-colored swelling |
|
Symptoms |
Usually associated with an itch Pain and tenderness uncommon |
May or may not be itchy Can be accompanied by pain and tenderness |
|
Pathology |
Extensive vasodilation, little edema, and sparse perivascular infiltrate of mainly neutrophils, eosinophils, monocytes, and T-lymphocytes in the papillary dermis layer |
Extensive edema, little vasodilation, and little or no cellular infiltrate, except in allergic angioedema where eosinophils may be seen in the reticular dermal, subcutaneous/submucosal layers |
Epidemiology and Pathophysiology
Hereditary Angioedema. HAE with C1-INH deficiency is an autosomal dominant disease with almost complete penetrance and has no propensity for sex or race. Type I makes up approximately 85% of cases, with the rest being attributed to type II. The prevalence of HAE is 1:30,000 to 1:80,000 of the general population.1 About 20-25% of type I cases are due to a spontaneous mutation.5 In contrast, HAE with normal C1-INH (formerly HAE type III) is also autosomal dominant but with a predominance for women. (See Table 2.) Population data at this time do not exist because of lack of clear diagnostic criteria. Men with this disease have a less severe version.1 The cause of HAE with normal C1-INH is unknown, but it may be due to factor XII gene miscues.5,6
Table 2. Types of Angioedema1,2
Syndrome |
Pathophysiology |
Patients Affected |
Prevalence |
C4 Level |
C1-INH Antigen |
C1-INH Function |
C1q Level |
|
Type I HAE |
Mutation in SERPING1 gene causing C1 inhibitor deficiency |
All |
~1 : 50,000 |
Low |
Low |
Low |
Normal |
|
Type II HAE |
Mutation in SERPING1 gene causing functional C1 inhibitor deficiency |
All |
~1 : 250,000 |
Low |
Normal |
Low |
Normal |
|
HAE with normal C1-INH (formerly called type III HAE) |
Unknown |
All, but many more women |
Unknown |
Normal |
Normal |
Normal |
Normal |
|
Acquired C1-INH deficiency |
Excessive consumption of C1 inhibitor leading to deficiency |
Older patients |
~1 : 250,000 |
Low |
Low |
Low |
Low |
|
ACE inhibitor–induced |
Inhibition of bradykinin catabolism |
All, but increased in African-Americans |
~1 : 250 |
Normal |
Normal |
Normal |
Normal |
|
Allergic |
Sensitization of mast cells by IgE |
All |
Unknown |
Unknown |
Unknown |
Unknown |
Unknown |
|
Idiopathic |
Sensitization of mast cells by IgG, linked with autoimmune thyroid disease |
All |
Unknown |
Unknown |
Unknown |
Unknown |
Unknown |
|
Nonhistaminergic idiopathic |
Unknown |
Unknown |
Unknown |
Normal |
Normal |
Normal |
Normal |
Types I and II HAE are due to a deficiency of C1-INH1 caused by mutations in the SERPING1 gene. C1-INH regulates many proteases. It is involved in inhibition of the classical pathway of complement, kinin, plasmin, and clotting systems.7 C1-INH does this by combining with the target protease to form a composite protease that is cleared as a whole. Type I and II HAE are autosomal dominant diseases, with more than 280 different mutations of the SERPING1 gene currently reported. Type II almost exclusively is caused by missense mutations, causing the production of C1-INH incapable of forming a composite protease.1,7
Type I can be any type of mutation resulting in a lack C1-INH, in turn leading to an excess of bradykinin as a cause of edema, while another implicated cause of edema is excess C2 kinin.1,7 The levels of these two peptides are increased when there is a lack of inhibition. These peptides act on the postcapillary venule, causing endothelial cells to retract, forming a space through which plasma can leak and ultimately accumulate as edema.7 Both of these types of angioedema appear to activate the complement cascade without the use of C1q (the first subcomponent of the C1 complex), as evidenced by laboratory testing demonstrating normal levels of C1q and reduced levels of C4.
The pathophysiology of HAE with normal C1-INH (previously called HAE type III) is not well understood. However, approximately 25% of these patients have been found to have mutations in factor XII. An increase in bradykinin has not been proven, but case reports have shown that treatments directed at inhibiting bradykinin production have been the most successful in reducing swelling.1
Acquired Angioedema. Acquired angioedema (AA) also involves a deficiency in C1-INH; however, this deficiency is not due to any genetic mutation. AA is seen in middle-aged and older patients with prevalence between 1:100,000-1:500,000, although the population statistics may not be accurate as a result of cases not being diagnosed correctly.1 Two types of acquired C1-INH deficiency have been described, both caused by a disproportionate compounding and clearing of C1-INH compared to the amount produced. This is often secondary to an underlying disease, causing a continuous activation of the complement pathway (type I).1 The most common diseases involved are lymphoproliferative conditions. However, AA patients also may produce autoantibodies against C1-INH that cause it to be cleaved into an inactive form without inactivating the proteases with which they bind (type II). Because there is no prognostic difference between acquired C1-INH with or without autoantibodies, there is no longer a diagnostic distinction between the two types. This decrease in C1-INH invariably leads to increased levels of bradykinin. Of note, AA differs from the hereditary types in that it consumes both C1q and C4 when activating the complement cascade.
ACE Inhibitor and Drug-induced Angioedema. ACE inhibitors are a commonly prescribed class of antihypertensives. ACE inhibitor-induced angioedema has an incidence of 0.2–0.7% among patients taking these medications, with a much higher risk for African-Americans. In fact, the condition has been shown to be up to five times more common in African-Americans than in whites.2 There is also an increased risk for patients taking immunosuppressive therapy, who have a history of smoking, are older, or are female.1,8-10 One multicenter study found that ACE inhibitor-induced angioedema makes up about one-third of angioedema patients who present to the ED, but these only accounted for 0.7 cases in 10,000 ED visits.11
Angiotensin-converting enzyme cleaves bradykinin and substance P into inactive proteins. Logically, ACE inhibitor medications thus cause increased levels of bradykinin. The level to which patients may be susceptible to angioedema may depend on the presence of other enzymes that break down bradykinin. Dipeptidyl-peptidase 4 (DPP-4) is one such enzyme. Patients taking both ACE inhibitors and DPP-4 inhibitors, a class of oral hypoglycemic medications including sitagliptin used in the treatment of diabetes, have a higher risk of developing angioedema. Immunosuppressive drugs also can cause a decrease in DPP-4, which may explain the increased risk of angioedema in these patients.1,8-10 However, the mechanism of angioedema with ACE inhibitors is not believed to be immunologic because attacks can happen within minutes to years after first taking these medications.12 Another drug that has been implicated in affecting this same pathway is the thrombolytic alteplase, which activates kinin and has been shown to cause orolingual angioedema after use in treating acute stroke patients.13 Thus, care should be taken when using these drugs together, as the possibility of angioedema is greatly increased.
Allergic Angioedema. Allergic angioedema is due to the type I immediate hypersensitivity response. It can present alone or with other signs and symptoms of anaphylaxis, and is the result of IgE-mediated release of histamine and other mediators from mast cells and basophils.5 The role of histamine in the body is primarily immunologic, and of the four known histamine receptors, types 1 and 2 are active in causing vasodilation, vascular permeability, and fluid accumulation in the interstitium, ultimately leading to the development of angioedema.4 Allergic angioedema is considered to be one of the most commonly encountered forms; in fact, a 2007 study from Mahidol University in Thailand found that an estimated 45.7% of all angioedema patients enrolled had experienced allergic angioedema, with 41.7% of these cases caused by food and 39.6% by drugs.14
Idiopathic and Other Types of Angioedema. Idiopathic angioedema can occur at any age, but it is most common in the 40- to 50-year-old age group. Women are affected more frequently than men, although it may be more common in men when there is no urticaria present.2 The specific cause of chronic idiopathic angioedema is unknown, but episodes can recur for five or more years, and the disease exhibits an extremely low risk of airway complications.5
Nonhistaminergic idiopathic angioedema is used to describe recurrent angioedema in patients who do not respond to high-dose antihistamines, have no family history of angioedema, and have elevated levels of bradykinin. The cause of these elevated levels of bradykinin currently is not well understood, and this diagnosis is made only after all other investigations have failed to explain recurrent symptoms in patients experiencing three or more attacks in a six- to 12-month period.1,15 Other forms of angioedema related to vibration, temperature extremes, or solar radiation (often accompanied by urticaria) are thought to be histamine-mediated via mast cell degranulation secondary to these physical stimuli. Antihistamines can provide effective treatment in these cases.16-18
Complications
There are many manifestations of angioedema, largely due to the sheer complexity and range of causes of the disease process. Location and severity of angioedema depend on its type and the extent of involvement in each patient. Thus, complications of this disease range from simply uncomfortable, such as extremity edema, to life-threatening; angioedema has the potential to cause significant morbidity and mortality.
Abdominal pain is relatively common in HAE. One study showed that 153 patients with HAE reported a total of 33,671 abdominal pain attacks in their lifetime.19 Another retrospective trial of HAE patients showed that 195 of 209 patients had recurrent abdominal pain, with a total of 62,503 abdominal attacks.20 Abdominal pain secondary to angioedema may be associated with nausea, vomiting, diarrhea, symptoms of hypotension, and ascites. Some associated symptoms that are less common include dysuria, hemorrhagic diarrhea, tetany, and intussusception, and patients can also experience circulatory shock due to hypovolemia.19 Abdominal symptoms primarily are due to visceral discomfort secondary to edema of the bowel wall. Thus, it is important to consider angioedema as a potential cause of acute abdominal pain in the ED. Knowledge that a patient has a personal or family history of angioedema or risks for the disease (i.e., ACE inhibitor use) might help to prevent unnecessary surgical intervention.21
The most dangerous and immediately concerning complication of angioedema is airway compromise. Patients most at risk for airway compromise include those with evidence of swelling involving the face, lips, larynx, uvula, and tongue. Edema in these areas can evolve into subjective or objective dyspnea, difficulty speaking (i.e., hot potato voice), stridor, and eventual loss of airway. These initial signs and symptoms of airway compromise must be recognized in a timely fashion to facilitate rapid and potentially lifesaving action.
Differential Diagnosis
There are many disease processes that can be mistaken for angioedema. Table 3 lists several of these, including both rare and common conditions.
Table 3. Differential Diagnosis of Angioedema: Pseudoangioedema2,27-30
Disease |
Clinical Clues to Differentiate from AE |
|
Facial cellulitis |
Indurated, painful, often associated with fever, can be present for days |
|
Acute contact: allergic or photodermatitis |
Associated with exposure history, itching, vesiculation, lasts longer than 24-48 h, type IV hypersensitivity |
|
Crohn’s disease of mouth and lips |
Causes granulomatous swelling of lips complicated by lymphedema Patients often show cobblestone thickening of buccal mucous membranes |
|
Hypocomplementemic urticarial vasculitis syndrome (HUVS) |
Leukocytoclastic vasculitis, severe angioedema, laryngeal edema, associated with chronic obstructive pulmonary disease, arthritis, arthralgia, glomerulonephritis, and uveitis Presence of anti-C1q antibodies |
|
Dermatomyositis, systemic lupus erythematosus, polymyositis, Sjögren’s syndrome |
Presents with heliotrope (violaceous) edema of the eyelids (dermatomyositis only), malar rash, photosensitivity, oral ulcers and arthralgias |
|
Hydrostatic edema |
Accumulation of soft tissue edema from increased capillary hydrostatic pressure secondary to various causes, including heart failure |
|
Tumid discoid lupus |
Red edematous plaques on light-exposed areas of the face; persistent; may cause scarring |
|
Ascher syndrome |
Recurrent episodes of eyelid edema leading to blepharochalasis |
|
Melkersson-Rosenthal syndrome |
Granulomatous swelling of lips that is persistent; can be associated with lingua plicata and/or Bell’s palsy |
|
Superior vena cava syndrome |
Edema of the eyelids and lips, associated with venous engorgement |
|
Gleich syndrome: episodic angioedema with eosinophilia |
Present with fever, weight gain, eosinophilia, increased serum major basic protein |
|
Schnitzler’s syndrome: macroglobulinemia secondary to monoclonal IgM |
Nonpruritic urticarial eruption and edema, fevers, bone pain, increased erythrocyte sedimentation rate |
|
Muckle-Wells syndrome |
Periodic attacks of urticaria, fever, and limb pains, progressive perceptive nerve deafness, renal amyloidosis, autosomal dominant trait |
|
Cheilitis granulomatosa/Miecher’s cheilitis |
Idiopathic episodes of recurrent edema that lead to permanent enlargement of lips |
|
Hypothyroidism |
Puffiness of face and lips; cold, dry skin; dry, coarse, brittle hair; pretibial myxedema |
|
Trichinosis |
Periorbital edema, history of eating raw/undercooked meat, myalgias, abdominal pain, diarrhea, vomiting |
|
Filarial parasites |
Lymphangitis that looks like chronic edematous swelling in lower extremities, localized areas of edema where adult worms live in subcutaneous tissues, history of visiting Central/West Africa |
|
Facial lymphedema |
Classically associated with rosacea or, rarely, Melkersson-Rosenthal syndrome |
|
Oncotic edema |
Accumulation of soft tissue edema from decreased plasma colloid osmotic pressure secondary to hypoproteinemia, often related to liver or renal disease |
Prehospital Management
Prehospital management of angioedema should focus on the ABCs (airway, breathing, circulation). Depending on the etiology and involved location, patients can develop hemodynamic collapse and airway compromise. Patients presenting with hemodynamic or respiratory changes will require immediate cardiac monitoring, continuous pulse oximetry, and capnography when available, and should receive two large-bore IVs, supplemental oxygen, and nebulized beta2-agonists as indicated (i.e., evidence of bronchospasm). Patients with known or possible allergic angioedema associated with anaphylactic shock should receive immediate intramuscular (IM) epinephrine, as well as intravenous (IV) fluids and supplementation with IV or IM antihistamines and steroids. Prehospital airway management (including intubation or other airway procedures as indicated and based on provider training and local protocols) should be undertaken if the patient is losing his or her airway from angioedema.
ED Evaluation
Patients presenting with angioedema should be triaged rapidly, with assessment of vital signs as well as airway patency and hemodynamic stability. Immediately concerning or unstable patients, including those with abnormal vital signs or edema of the head and/or neck, should be roomed immediately in the ED, specifically in resuscitation rooms as indicated. Emergent cardiac monitoring, continuous pulse oximetry, airway maneuvers, and IV access with appropriate medical therapy should be initiated. Patients who are stable with normal vitals and no evidence or risk of airway or circulatory compromise do not require immediate ED bedding but should be re-assessed regularly until they can be evaluated fully.
History
The ED history is vital in determining the cause and appropriate management of every angioedema presentation. Information including time of onset, prodrome, progression, exposures, and associated symptoms will be important to elicit. Emergency physicians should inquire about involved areas of swelling, such as the lips or tongue, as well as any voice changes or sensation of dyspnea or throat closure. Other history includes current medications, prior episodes or family history of angioedema, recent procedures or trauma, and the patient’s general medical history.3
Physical Examination
More commonly affected body regions include the face and extremities, as well as the gastrointestinal (GI) tract as previously discussed, especially if the patient has a history of hereditary angioedema. Urticaria and pruritus can accompany allergic angioedema, but such signs and symptoms only rarely present with other forms. Airway compromise is the most concerning complication of angioedema, and evaluation for this should be of primary importance during the physical exam and ED evaluation. In addition to visual inspection for rashes and swelling, emergency providers can assess for evidence of airway edema by listening and auscultating for telltale changes such as hoarseness, stridor, wheezing, and abnormal provoked humming and vocal sounds.3
Diagnostic Evaluation
The primary focus of ED evaluation and management of angioedema centers on determining the presence and extent of airway involvement, as well as evaluating for causes that could benefit from emergency medical treatment, such as the allergic and hereditary etiologies. Left untreated, any obstructive angioedema of the upper airway carries a mortality rate ranging from 25-40%.3 Similarly, estimates of lifetime mortality from hereditary angioedema have been as high as 30%, with airway obstruction implicated as the primary cause of death.22
The true etiology of any given angioedema presentation often is difficult to ascertain in the ED, and there are limited emergency diagnostic tests helpful in evaluating these patients. Thus, an emergency physician’s clinical evaluation of the airway and hemodynamic stability of these patients should always take priority.
Evaluation for the presence or absence of airway edema preferably involves direct visualization via awake video laryngoscopy or fiberoptic nasopharyngoscopy, where available. In 2010, Saxena et al demonstrated that the presence and level of airway edema is difficult to judge based only on a patient’s history and clinical appearance, and that supraglottic swelling often does not correlate with tongue swelling.23 With the appropriate equipment and training, emergency physicians can use video and fiberoptic techniques to rapidly assess for laryngeal and supraglottic edema in awake and upright patients, requiring no adjuncts beyond some local anesthetic spray or gel (can also administer lidocaine via nebulizer) to improve patient tolerance. However, if such equipment is not available and the provider believes the patient is stable without any risk of acute decompensation, computed tomography (CT) can be used to assess airway edema. Less sensitive or reliable methods to evaluate for airway edema include plain radiographs, laryngeal ultrasonography, and indirect laryngoscopy using a mirror (can be helpful but not commonly available in the ED). If the patient is very stable, and consultation is immediately available, the expertise of consultants, such as otolaryngology and anesthesia, can be considered to further assist in the evaluation of airway edema and to guide treatment and disposition.
CT and ultrasonography can be helpful in the evaluation of bowel wall edema and ascites, which can be seen in up to 93% of patients with hereditary angioedema. Contrast-enhanced CT is the modality of choice to evaluate for intestinal angioedema.24,25 Plain radiography, CT, and ultrasonography also can be helpful in evaluating acutely hypoxic patients for the presence of pulmonary edema as a complication of HAE.26
Laboratory evaluation of angioedema rarely provides timely information in the ED setting; however, it is important to order C1-INH levels during an acute attack in patients with suspected but not diagnosed HAE. Other testing can be done during the admission. Complement testing for HAE often begins with serum C4 testing. If C4 levels are less than 30% of normal, C1-INH level and function are subsequently tested. Low C1-INH level and function indicate HAE type I, normal C1-INH level but low function indicates HAE type II, and normal C1-INH level and function in the setting of known family history of angioedema defines HAE with normal C1-INH. However, C4 screening tests have not proven to demonstrate consistently low levels in HAE patients, which has brought into question the reliability of this screening method.31
Additional complement testing includes evaluation of C1q levels to differentiate between hereditary (normal C1q) and type I acquired (decreased C1q) angioedema. The presence of anti-C1-INH autoantibodies is required in the diagnosis of acquired angioedema type II. Diagnosis of hereditary and acquired cases of angioedema requires a high index of suspicion from the provider, and these diagnoses historically have been delayed. In 1977, the average delay in diagnosing HAE was 22 years from onset of symptoms to diagnosis, and more recent data from 2013 found a diagnostic delay of about 8.5 years.32,33
There is currently no established test to diagnose ACE inhibitor-induced angioedema, which is currently a clinical diagnosis despite evidence of predisposition. Allergic angioedema can be evaluated via measurement of serum tryptase level, a sensitive marker of mast cell degranulation that increases during exacerbations, as well as more specific IgE testing as indicated.34
ED Management and General Treatment
The initial ED evaluation of patients presenting with angioedema will provide vital information regarding that patient’s cardiopulmonary stability. (See Figures 3 and 4.) Patients exhibiting any airway or hemodynamic risk or compromise should receive IV access immediately, supplemental O2, continuous pulse oximetry, capnography (when available), and a cardiac monitor. As providers proceed through the primary survey, the airway must be examined properly to ensure patency, and, if compromised, a decision should be made quickly regarding when and how the airway should be secured. After this decision point and subsequent management, determining the type of angioedema involved will dictate treatment. (See Table 4.) If the type of angioedema is unknown and the emergency physician believes the patient could be having an anaphylactic reaction, IM epinephrine 0.01 mg/kg of concentration 1 mg/mL (up to 0.3 mg in children and 0.5 mg in adults) should be given. (Note that as of May 2016, epinephrine 1:1000 is labeled 1 mg/mL). Oral (PO), IV, or IM H1 antagonists (i.e., diphenhydramine 50 mg), and PO, IV, or IM corticosteroids (i.e., IV solumedrol 1-2 mg/kg up to 125 mg/dose) will treat allergic angioedema effectively but also can be used to treat individuals with angioedema of unknown etiology. An oral or IV H2 antagonist, such as famotidine, may be added if potential histaminergic symptoms do not seem to be alleviated with H1 antagonists alone.36-38
Figure 3. Angioedema Airway Algorithm
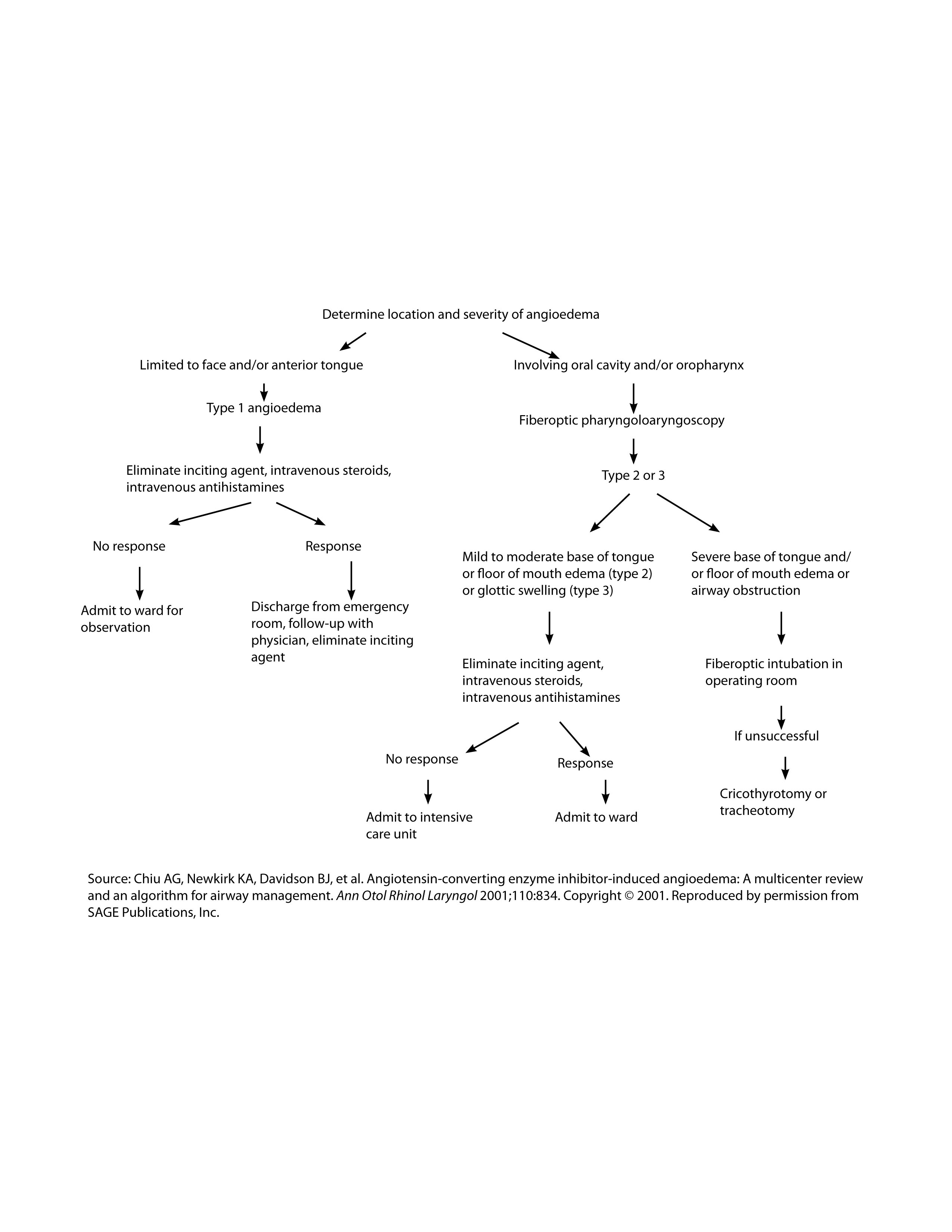 <>
<>
Figure 4. Treatment Algorithm35,37,49-51
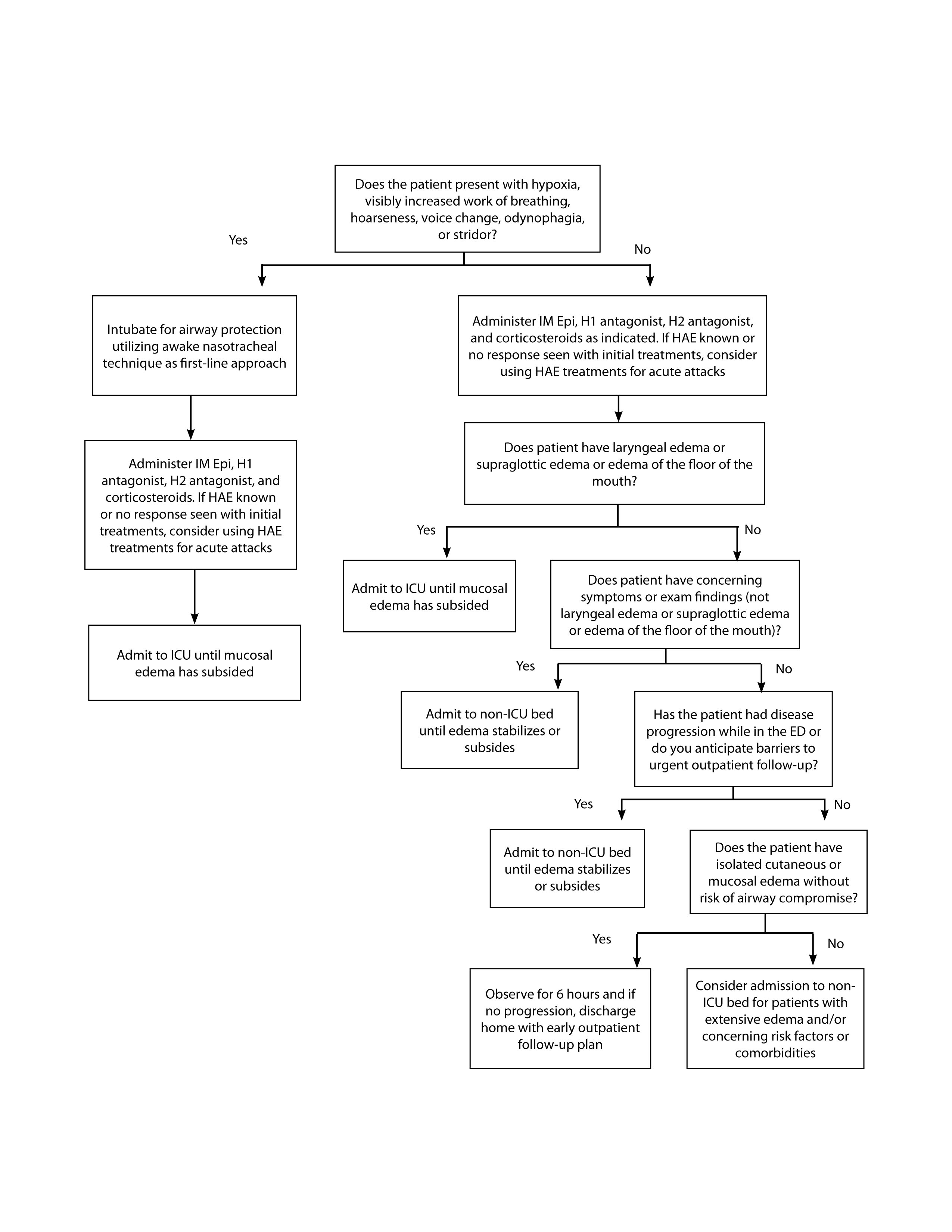
Table 4. Treatment of Acute Attacks of HAE and Acquired Angioedema2,36,38,44-47
Drug |
Brand Name |
Mechanism of Action |
Dose |
Route of Administration |
Approved for self-administration? |
Time to Symptom Relief |
Side Effects |
|
Plasma derived C1-INH |
Berinert |
C1-INH replacement |
20 U/kg 500-1,000 U in less severe attacks |
IV |
Yes |
30 min (high dose) |
Anaphylaxis, blood-borne infection risk |
|
Cinryze |
C1-INH replacement |
1,000 U, 2nd dose 1 hour later |
IV |
Yes |
4 hours |
Anaphylaxis, blood-borne infection risk. |
|
|
rhC1-INH |
Ruconest, Rhucin |
C1-INH replacement |
50 U/kg or 4,200 U |
IV |
No |
90 min |
Anaphylaxis in rabbit-sensitized patients |
|
Icatibant |
Firazyr |
Bradykinin B2-receptor antagonist |
30 mg q 6 hours 90 mg max/24 hours |
SC |
Yes |
2 hours |
Transient erythema, wheal/pain at injection site |
|
Ecallantide |
Kalbitor |
Kallikrein inhibitor |
30 mg q 1 hour 60 mg max/24 hours |
SC |
No |
90 min |
Anaphylaxis, antidrug antibodies |
|
FFP |
C1-INH replacement |
1-3 U |
IV |
No |
45 min |
CHF exacerbation, possibly worsened symptoms |
Patients on beta-blocker therapy may not respond effectively to IM epinephrine, and may require doses of glucagon to mitigate the activity of their beta-blocker and allow the full effect of any administered epinephrine (1-5 mg IV glucagon over five minutes for adults, can also be given as an infusion).39
Nebulized racemic epinephrine (0.5-0.75 mL of 2.25% solution mixed with 2-3 mL of normal saline) has been suggested to reduce oral and laryngeal edema temporarily while preparing for airway intervention.40 Patients who are hypotensive or hemodynamically unstable should receive aggressive intravenous fluids and pressors.
For patients presenting with significant intraoral and respiratory tract swelling, endotracheal intubation may be necessary. (See Figure 5.) Supraglottic rescue devices, such as laryngeal mask airways (LMA) and King Laryngeal Tubes, have limited use and increased risk of failure in acute angioedema presentations because of the potential for vocal cord and epiglottis involvement. Awake fiberoptic nasotracheal intubation is consistently the preferred technique to intubate patients with acute oral angioedema.41 Awake intubation techniques utilizing sedatives and local anesthetics are favored, as they allow patients to protect their airway for as long as possible while an endotracheal tube is passed into the trachea. In comparison, standard orotracheal rapid sequence intubation (RSI) using induction and paralytic medication causes relaxation of the edematous epiglottis and other intraoral tissues, which can sag and obstruct the larygnoscopist’s view. This decreased visibility greatly reduces the likelihood of successfully passing an endotracheal tube and could lead to a disastrous “cannot intubate, cannot ventilate” scenario. RSI should be used with caution in these patients.
Figure 5. Intubated Patient with Oral Angioedema
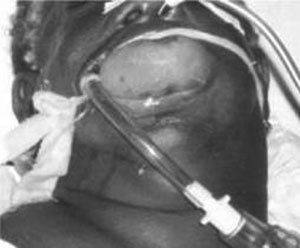
Photo reproduced with permission from the West Indian Medical Journal.
Attempts at awake fiberoptic intubation can be performed in either the ED or operating room, and, when emergently available, involve anesthesia and surgical staff. The physician should be prepared to perform an emergent cricothyrotomy.35,41,42 A difficult airway cart, if available, should be at the bedside to provide a variety of airway options. Video laryngoscopes and fiberoptic intubation equipment, if available, may be useful. A recent study by Chomansky et al showed decreased time to intubation with airway angioedema via video laryngoscope compared to fiberoptic nasotracheal intubation, and no statistical difference in first pass attempts, although this has not been validated in a larger study.43
In 1999, to aid providers in airway angioedema decision-making, Ishoo et al published a staging system guiding angioedema management and disposition that was based on the location of a patient’s airway edema. Patients exhibiting only facial and lip changes were considered stage I, and staging increased as edema approached the larynx, with stage II for edema of the soft palate, stage III for lingual edema, and stage IV for laryngeal edema. Stage III usually (67%) and stage IV always (100%) necessitated ICU admission, and acute airway intervention was required in 7% of stage III and 24% of stage IV patients.48 More recently, Chiu et al created an algorithm for intubation based on a grading system of the degree of angioedema at the time of patient presentation.35 They used three classifications as follows: type 1, limited to the face and oral cavity, excluding floor of mouth; type 2, edema of the floor of mouth and/or oropharynx; and type 3, oropharyngeal edema that extended to supraglottic/glottis structures. (See Figure 3.)
In the management of hereditary angioedema, antihistamines and corticosteroids will have no effect on the pathology of the disease,2 but they also are unlikely to negatively affect the patient. Thus, the empiric approach described above, if initiated in the ED, is neither harmful nor unreasonable. In the past, fresh frozen plasma (FFP) and C1-inhibitor concentrate were the mainstay of treatment of HAE; however, newer treatments are now available. FFP is successful in treating HAE when it includes C1-INH. It is dosed between 1-3 units (U) and is successful in improving symptoms within 45 minutes.45-47 However, some patients actually have become more edematous after receiving FFP, and, thus, it is not recommended in cases of life-threatening laryngeal edema.3 Beinert is a C1-inhibitor concentrate that is administered intravenously at various doses, depending on the severity of the reaction, and is effective in relieving symptoms within 30 minutes at the highest dose. Life-threatening attacks, such as laryngeal edema or severe oral edema, require 20 U/kg IV. Non-life-threatening attacks can be treated with 500 U or 1,000 U IV.37 Cinryze is another C1-INH concentrate effective for relieving symptoms within four hours. It is dosed at 1,000 U, with a second dose given one hour later, if necessary. Cinryze has been associated with thrombotic events, but no causal link or frequency has been demonstrated.52 Recombinant human C1-INH (rhC1INH) is given in doses of 50 U/kg or 4,200 U IV and is effective for relieving symptoms within 90 minutes. Ecallantide is a kallikrein inhibitor that is given subcutaneously at a dose of 30 mg, relieves symptoms within 90 minutes, and may be repeated once.36,38,44 Icatibant is a bradykinin B2 receptor antagonist that is given subcutaneously at a dose of 30 mg, relieves symptoms within two hours, and can be repeated up to two times every six hours in a 24-hour period.36,38,44 The most exciting aspect of these newer treatments is their ability to be taken at home, excluding ecallantide and rhC1INH.
C1-INH concentrates, icatibant, and ecallantide are expensive ($5,000-$10,000/dose).53
In stark contrast to the above, investigations into effective medical treatment of ACE inhibitor-induced angioedema have been largely fruitless to date. Numerous studies evaluating the efficacy of therapies used for other angioedema types have shown no benefit, and currently the best treatment for ACE inhibitor-induced angioedema continues to be airway management and supportive care. Emergency providers often will administer medications, including antihistamines and corticosteroids, empirically to these patients. In 2015, Bas et al evaluated the use of icatibant compared to standard antihistamine and steroid therapy for ACE inhibitor-induced angioedema, and found that patients receiving icatibant had resolution of symptoms 70% faster than the control group (8.0 hours vs. 27.1 hours, P = 0.002).54 However, Shire’s CAMEO study, also in 2015, found no statistically significant difference in time that patients with ACE inhibitor-induced angioedema improved to meet discharge criteria after receiving icatibant when compared to placebo (P = 0.63), as well as no statistical difference in time to symptom relief.55 Similarly, a 2014 study by Lewis et al found that ecallantide failed to improve ACE inhibitor-induced angioedema when compared to placebo.56
The most important message emergency providers should take from the above results is that ACE inhibitor-induced angioedema, which is understood to be bradykinin-mediated, should not be expected to respond to antihistamines and glucocorticoids. In addition, current evidence also shows no consistent efficacy in the use of bradykinin receptor antagonists or kallikrein inhibitors. Thus, in all cases of ACE inhibitor-induced angioedema in the ED, airway management and stabilization should be of paramount importance, and medical therapies can not yet be relied upon.
Disposition
As recommended by Chiu in the algorithm in Figure 3, disposition of patients with non-allergic angioedema will depend on whether there was airway involvement and what level of acute management was required. Intubated patients should be admitted to the ICU for further evaluation and treatment as indicated. Patients with airway edema not requiring emergent airway procedures can be admitted to the floor or ICU, or even discharged home, depending on the location and extent of involvement.
Patients with allergic angioedema are at risk for biphasic anaphylactic reactions, which typically occur within 10 hours after initial treatment. Disposition of patients with anaphylaxis should be guided by the severity of their symptoms and response to treatment in the ED. According to the 2015 practice parameter update from the Joint Council of Allergy, Asthma, and Immunology, patients with non–life-threatening symptoms can be observed for four to eight hours if initial treatment is effective, and discharged if there is no recurrence. Patients with recurrence requiring a second dose of epinephrine, patients with history of an ingested allergen, or those patients with particularly severe or concerning presentations, such as evidence of significant cardiovascular and respiratory involvement, warrant either longer observation in the ED or admission for observation.57
In all cases of angioedema, after relief or resolution of angioedema attacks, potential causes should be investigated. Patients should discontinue suspected inciting medications (such as ACE inhibitors, new antibiotic prescriptions, etc.) immediately, should avoid suspected inciting allergens, and should receive appropriate warnings and precautions regarding these suspected causes. It is recommended that all patients with new allergic reactions be referred to an allergist. ACE inhibitors should be replaced with antihypertensive medications not in the ACE inhibitor, angiotensin II receptor blocker, or direct renin inhibitor classes; patients should be instructed to follow up with their primary physician to review any medication changes.58-60
Patients being discharged after complete resolution of an allergic angioedema and/or anaphylactic reaction should be given an auto-injectable epinephrine prescription and should be instructed personally in its use; however, these patients do not need to be prescribed antihistamines or corticosteroids upon discharge.57 Chronic cases of allergic angioedema can be treated with H1-receptor antagonists such as cetirizine, or even long-term, low-dose steroids in refractory cases.61
Several established therapies, such as attenuated androgens, C1-INH concentrates, and recombinant C1-INH, currently are used to help prevent attacks of HAE. (See Table 5.) Low-dose androgens include danazol and stanozolol and are dosed at 50-300 mg/day and 2-4 mg/day, respectively.2,38 Cynryze is the only C1-INH concentrate currently approved for preventive use with a dose of 1,000 U every 3-4 days. rhC1-INH also has been shown to be effective in prevention when dosed at 50 U/kg weekly. Tranexamic acid is another preventive medication for HAE and is dosed at 1 g twice daily in adults or 20 mg/kg in children. The above treatments are similarly efficacious in preventing attacks of acquired angioedema when coupled with active treatment of the underlying disease.
Table 5. Prophylaxis of HAE and Acquired Angioedema
Drug |
Brand Name |
Mechanism of Action |
Dose |
Route of Administration |
Side Effects |
|
Plasma derived C1-INH |
Cinryze |
C1-INH replacement |
1,000 U every 3-4 days |
IV |
Anaphylaxis, blood-borne infection risk |
|
rhC1-INH |
Ruconest, Rhucin |
C1-INH replacement |
50 U/kg/week |
IV |
Anaphylaxis, blood-borne infection risk |
|
Androgen |
Danazol |
Unknown |
50-300 mg/day |
PO |
Hepatotoxicity, liver tumors, male secondary sex characteristics in women |
|
Stanozolol |
Unknown |
2-4 mg/day |
PO |
Hepatotoxicity, liver tumors, male secondary sex characteristics in women |
|
|
Tranexamic acid |
Cyklokapron, Lysteda |
Binds plasminogen binding sites |
Adults: 1 g twice daily Peds: 20 mg/kg/day |
PO |
Anaphylaxis, seizures, thromboembolism, cerebral edema |
In the treatment of idiopathic angioedema, low doses of H2 antagonists during the day along with H1 antagonists at night have been shown to be very effective. For patients who fail to respond to antihistamine therapy, cyclosporine in dosages of 2.5-4 mg/kg/day for a maximum of three months can be considered, with one trial demonstrating 80% of patients experiencing near or complete resolution.2,62 Those who did not respond were given another round of cyclosporine or started instead on methotrexate. IVIG, plasmapheresis, and low doses of corticosteroids also may be given.
Summary
Angioedema is a complex and potentially deadly condition that has multiple causes, not all of which are well understood. While some etiologies do have medical therapies available, all variants can present with life-threatening airway edema, which must be addressed rapidly if found. Angioedema is a diagnosis that cannot be missed when it enters the ED. Approach each suspected case with a thorough history, physical exam, and the appropriate clinical suspicion. Determine if a patient is presenting with urticaria, angioedema, anaphylaxis, or combinations of these. Remember that angioedema, in contrast to urticaria, takes place in the subcutaneous and submucosal layers, lasts between 24-48 hours, exhibits pink/skin-colored edema, is more likely accompanied by pain than itching, and can be complicated by abdominal pain and airway compromise. Be familiar with the differential diagnosis of angioedema, and utilize a thorough history and physical exam in assessing for these pseudoangioedemas.
The role of prehospital providers should focus on assessment and stabilization of the ABCs while rapidly transporting these patients to the nearest ED. If patients show any evidence of hemodynamic or respiratory changes, both prehospital and ED staff should immediately obtain IV access (two large-bore IVs preferred), provide supplemental oxygen, and connect the patient to continuous pulse oximetry and a heart monitor. During the primary survey, providers should determine their comfort level with the patient’s airway and whether or not intervention is necessary using the algorithm depicted in Figure 3. Once the airway is considered stable or has been protected, providers then can proceed through the primary and secondary surveys, intervening as indicated. Knowing the type of angioedema will help expedite the correct treatment for these patients. Providers should be familiar with the availability and accessibility of associated pharmacological agents and should ensure there are personnel trained in the appropriate preparation and administration of these medications. Finally, providers can utilize the algorithm depicted in Figure 4 to develop comfort and understanding of the appropriate disposition of patients with angioedema, whether they require admission or can be safely discharged home.
REFERENCES
- Hereditary Angioedema and Bradykinin-Mediated Angioedema. In: Adkinson NF Jr., Bochner BS, Burks AW, et al. Middleton’s Allergy: Principles and Practice. Saunders; 2014:588-601.
- Kaplan AP, Greaves MW. Angioedema. J Am Acad Dermatol 2005;53:373–88. quiz 389-392.
- Toxicology/Infections and Disorders of the Neck and Upper Airway. In: Tintinalli JE, Stapczynski JS, Jo O. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 7th ed. New York: McGraw Hill; 2011:183-190, 1276,1591.
- Diseases of Immunity. In: Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Elsevier; 2015:64, 245.
- Urticaria, Angioedema and Pruritis. In: Habif TP. Clinical Dermatology: A Color Guide to Diagnosis and Therapy, 6th ed. Saunders; 2016:178-217.
- Bork K, Wulff K, Hardt J, et al. Hereditary angioedema caused by missense mutations in the factor XII gene: Clinical features, trigger factors, and therapy. J Allergy Clin Immunol 2009;124:129.
- Complement. In: Male D, Brostoff J, Roth D, Roitt I. Immunology, 8th ed. Saunders; 2013:71-84.
- Mackenzie M, Wood LA. Lingual angioedema associated with everolimus. Acta Oncol 2010;49:107.
- Mahé E, Morelon E, Lechaton S, et al. Angioedema in renal transplant recipients on sirolimus. Dermatology 2007;214:205.
- Sendur OF, Turan Y, Berkit IK, Tastaban E. Angio-oedema in a patient treated with etanercept for rheumatoid arthritis. Basic Clin Pharmacol Toxicol 2009;104:488.
- Banerji A, Clark S, Blanda M, et al. Multicenter study of patients with angiotensin-converting enzyme inhibitor-induced angioedema who present to the emergency department. Ann Allergy Asthma Immunol 2008;100:327.
- Agostoni A, Cicardi M, Cugno M, et al. Angioedema due to angiotensin-converting enzyme inhibitors. Immunopharmacology 1999;44:21-25.
- Ottomeyer C, Hennerici MG, Szabo K. Raising awareness of orolingual angioedema as a complication of thrombolysis in acute stroke patients. Cerebrovasc Dis 2009;27:307.
- Kulthanan K, Jiamton S, Boochangkool K, Jongjarearnprasert K. Angioedema: Clinical and etiological aspects. Clin Dev Immunol 2007;2007:26438.
- Frigas E, Nzeako UC. Angioedema. Pathogenesis, differential diagnosis, and treatment. Clin Rev Allergy Immunol 2002;23:217-231.
- Bentley B 2nd. Cold-induced urticaria and angioedema: Diagnosis and management. Am J Emerg Med 1993;11:43-46.
- Lawlor F, Black AK, Breathnach AS, Greaves MW. Vibratory angioedema: Lesion induction, clinical features, laboratory and ultrastructural findings and response to therapy. Br J Dermatol 1989;120:93-99.
- Rose RF, Bhushan M, King CM, Rhodes LE. Solar angioedema: An uncommonly recognized condition? Photodermatol Photoimmunol Photomed 2005;21:226-228.
- Bork K, Staubach P, Eckardt AJ, Hardt J. Symptoms, course, and complications of abdominal attacks in hereditary angioedema due to C1 inhibitor deficiency. Am J Gastroenterol 2006;101:619.
- Bork K, Meng G, Staubach P, Hardt J. Hereditary angioedema: New findings concerning symptoms, affected organs, and course. Am J Med 2006;119:267.
- Bloom AS, Schranz C. Angiotensin-converting enzyme inhibitor-induced angioedema of the small bowel: A surgical abdomen mimic. J Emerg Med 2015;48:e127.
- Agostoni A, Aygoren-Pursun E, Binkley KE, et al. Hereditary and acquired angioedema: Problems and progress: Proceedings of the third C1 esterase inhibitor deficiency workshop and beyond. J Allergy Clin Immunol 2004;114(3 Suppl):S51-S131.
- Saxena S, Gierl B, Eibling DE. Supraglottic swelling may not correlate with tongue swelling in angiotensin converting enzyme inhibitor-induced angioedema. Laryngoscope 2010;120:62-64.
- Bork K, Meng G, Staubach P, Hardt J. Hereditary angioedema: New findings concerning symptoms, affected organs, and course. Am J Med 2006;119:267-274.
- Scheirey CD, Scholz FJ, Shortsleeve MJ, Katz DS. Angiotensin-converting enzyme inhibitor-induced small-bowel angioedema: Clinical and imaging findings in 20 patients. AJR Am J Roentgenol 2011;197:393-398.
- da Costa JT, da Silva JM, Cunha L, et al. Hereditary angioedema presenting with adult respiratory distress syndrome. Chest 1994;105:1898-1899.
- Van Dellen RG, Maddox DE, Dutta EJ. Masqueraders of angioedema and urticaria. Ann Allergy Asthma Immunol 2002;88:10.
- Gleich GJ, Schroeter AL, Marcoux JP, et al. Episodic angioedema associated with eosinophilia. N Engl J Med 1984;310:1621.
- Jara LJ, Navarro C, Medina G, et al. Hypocomplementemic urticarial vasculitis syndrome. Curr Rheumatol Rep 2009;11:410.
- Charlesworth EN. Differential diagnosis of angioedema. Allergy Asthma Proc 2002;23:337.
- Tarzi MD, Hickey A, Forster T, et al. An evaluation of tests used for the diagnosis and monitoring of C1 inhibitor deficiency: Normal serum C4 does not exclude hereditary angio-oedema. Clin Exp Immunol 2007;149:513-516.
- Zuraw BL. Clinical practice. Hereditary angioedema. N Engl J Med 2008;359:1027-1036.
- Zanichelli A, Magerl M, Longhurst H, et al. Hereditary angioedema with C1 inhibitor deficiency: Delay in diagnosis in Europe. Allergy, Asthma Clin Immunol 2013;9:29.
- Temino VM, Peebles RS Jr. The spectrum and treatment of angioedema. Am J Med 2008;121:282-286.
- Chiu AG, Newkirk KA, Davidson BJ, et al. Angiotensin-converting enzyme inhibitor-induced angioedema: A multicenter review and an algorithm for airway management. Ann Otol Rhinol Laryngol 2001;110:834.
- Cicardi M, Bork K, Caballero T, et al. Evidence-based recommendations for the therapeutic management of angioedema owing to hereditary C1 inhibitor deficiency: Consensus report of an International Working Group. Allergy 2012;67:147.
- American Academy of Emergency Medicine. Clinical practice guideline: Initial evaluation and management of patients presenting with acute urticaria or angioedema. Available at: http://www.aaem.org/em-resources/position-statements/2006/clinical-practice-guidelines. Accessed Nov. 27, 2016.
- Bork K. An evidence based therapeutic approach to hereditary and acquired angioedema. Curr Opin Allergy Clin Immunol 2014;14:354.
- Sampson HA, Munoz-Furlong A, Campbell RL, et al. Second symposium on the definition and management of anaphylaxis: Summary report — second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network Symposium. Ann Emerg Med 2006;47:373-380.
- Abdelmalak B, Doyle J. Anesthesia for Otolaryngologic Surgery, 1st ed. Cambridge University Press; 2013:102.
- Nadel ES, Brown DF. Angioedema. J Emerg Med 1998;16:477-479.
- Bentsianov BL, Parhiscar A, Azer M, Har-El G. The role of fiberoptic nasopharyngoscopy in the management of the acute airway in angioneurotic edema. Laryngoscope 2000;110:2016-2019.
- Wood A, Choromanski D, Orlewicz M. Intubation of patients with angioedema: A retrospective study of different methods over three year period. Int J Crit Illn Inj Sci 2013;3:108-112.
- Bork K, Bernstein JA, Machnig T, Craig TJ. Efficacy of different medical therapies for the treatment of acute laryngeal attacks of hereditary angioedema due to C1-esterase inhibitor deficiency. J Emerg Med 2016;50:567.
- Hassen GW, Kalantari H, Parraga M, et al. Fresh frozen plasma for progressive and refractory angiotensin-converting enzyme inhibitor-induced angioedema. J Emerg Med 2013;44:764.
- Pekdemir M, Ersel M, Aksay E, et al. Effective treatment of hereditary angioedema with fresh frozen plasma in an emergency department. J Emerg Med 2007;33:137.
- Hill BJ, Thomas SH, McCabe C. Fresh frozen plasma for acute exacerbations of hereditary angioedema. Am J Emerg Med 2004;22:633.
- Ishoo E, Shah UK, Grillone GA, et al. Predicting airway risk in angioedema: Staging system based on presentation. Otolaryngol Head Neck Surg 1999;121:263-268.
- Bowen T, Cicardi M, Farkas H, et al. 2010 International consensus algorithm for the diagnosis, therapy and management of hereditary angioedema. Allergy Asthma Clin Immunol 2010;6:24.
- Betschel S, Badiou J, Binkley K, et al. Canadian hereditary angioedema guideline. Allergy Asthma Clin Immunol 2014;10:50.
- Dykewicz MS. Cough and angioedema from angiotensin-converting enzyme inhibitors: New insights into mechanisms and management. Curr Opin Allergy Clin Immunol 2004;4:267.
- Gandhi PK, Gentry WM, Bottorff MB. Thrombotic events associated with C1 esterase inhibitor products in patients with hereditary angioedema: Investigation from the United States Food and Drug Administration adverse event reporting system database. Pharmacotherapy 2012;32:902.
- Cicardi M, Zuraw B. Hereditary angioedema: Treatment of acute attacks. In: UpToDate, Post TW (Ed), UpToDate, Waltham, MA. Accessed Nov. 22, 2016.
- Baş M, Gree J, Stelter K, et al. A randomized trial of icatibant in ACE-inhibitor-induced angioedema. N Engl J Med 2015;372:418-425.
- Sinert, et al. Blinded safety and efficacy placebo controlled study of icatibant for angiotensin converting enzyme inhibitor angioedema. CAMEO study by Shire Pharmaceuticals Limited, not published, discontinued September 2015.
- Lewis LM, Graffeo C, Crosely P, et al. Ecallantide for the acute treatment of angiotensin-converting enzyme inhibitor-induced angioedema: A multicenter, randomized, controlled trial. Ann Emerg Med 2015;65:204-213.
- Lieberman P, Nicklas RA, Randolph C, et al. Anaphylaxis — a practice parameter update 2015. Ann Allergy Asthma Immunol 2015;115:341-384.
- Toh S , Reichman ME, Houstoun M, et al. Comparative risk for angioedema associated with the use of drugs that target the renin-angiotensin-aldosterone system. Arch Intern Med 2012;172:1582-1589.
- Vasekar M, Craig TJ. ACE inhibitor-induced angioedema. Curr Allergy Asthma Rep 2012;12:72.
- Byrd JB, Adam A, Brown NJ. Angiotensin-converting enzyme inhibitor-associated angioedema. Immunol Allergy Clin North Am 2006;26:725.
- Kaplan AP. Clinical practice. Chronic urticaria and angioedema. N Engl J Med 2002;346:175.
- Grattan CE, O’Donnell BF, Francis DM, et al. Randomized double-blind study of cyclosporin in chronic ‘idiopathic’ urticaria. Br J Dermatol 2000;143: 365-372.
Angioedema is a complex and potentially deadly condition that has multiple causes, not all of which are well understood.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
