Non-Traumatic Ocular Emergencies
June 1, 2017
Reprints
AUTHORS
Ademola Adewale, MD, FAAEM, Director of Research/Medical Simulation, Assistant Program Director, Florida Hospital Emergency Medicine Residency, Orlando.
Chasi Skinner, DO, Emergency Medicine, PGY3, Florida Hospital Orlando.
PEER REVIEWER
Steven M. Winograd, MD, FACEP, Assistant Professor of Emergency Medicine, New York Institute of Technology College of Osteopathic Medicine, Old Westbury, NY.
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Farel (CME question reviewer) owns stock in Johnson & Johnson. Ms. Light (nurse planner) is a consultant for Bard Medical. Dr. Schneider (editor), Dr. Stapczynski (editor), Dr. Adewale (author), Dr. Skinner (author), Dr. Winograd (peer reviewer), Ms. Mark (executive editor), Ms. Coplin (executive editor), and Ms. Hatcher (AHC Media Editorial Group Manager) report no financial relationships with companies related to the field of study covered by this CME activity.
EXECUTIVE SUMMARY
- Chlamydial neonatal conjunctivitis is treated with oral therapy.
- Herpes simplex and herpes zoster keratitis both are treated with antivirals and topical steroids.
- An important differentiation between preseptal and orbital cellulitis is impaired globe motility with orbital infection.
- Chemical burns of the eye should be irrigated until the pH of tear fluid is normal: 7.0 to 7.3.
- Central retinal artery occlusion causes profound visual impairment and should be treated aggressively.
- Pupillary dysfunction along with a third cranial nerve palsy supports the likelihood of a posterior communicating artery aneurysm.
Introduction
Ocular-related problems represent roughly 2% of all patients who present to the emergency department (ED).1 From 2006-2011, approximately 12 million visits to the ED were for eye-related problems, which equates to about 2 million visits per year.2 Of these visits, 44.3% were nonemergent, while approximately 41.2% were deemed emergent.3
The majority of infectious etiologies affect the outer fibrous layer of the cornea and sclera. Chemical exposures also affect the outer fibrous layers, but can penetrate deeply into the vascular layer. Finally, vascular and neurodegenerative processes occur within the posterior segments, particularly near the optic disc and the retinal artery and vein.
Evaluating eye-related complaints in the ED requires a good understanding of the anatomy and potential implications of failure to treat. Although most ED presentations are nonemergent, it is imperative that the emergency physician identify the emergent presentations and manage them promptly to prevent potential vision loss. This paper will focus on non-traumatic ocular emergencies. The discussion will emphasize the following four categories: infectious, vascular, chemical, and neurologic causes of non-traumatic eye emergencies.
Presentation and Management
Infectious
Neonatal Conjunctivitis. Conjunctivitis is an inflammation of the bulbar and palpebral conjunctiva caused by viral, bacterial, mechanical, allergic, and toxic or caustic agents.4 Special consideration includes the newborn with conjunctival infection. Gonorrhoeae is present in roughly 60 of every 100,000 births. Chlamydia is present in roughly 400 per 100,000 births, and 30-40% of neonates born to mothers with Chlamydia trachomatis develop conjunctivitis, which makes this infection the leading cause of blindness worldwide.5
Neisseria gonorrhoeae should be suspected if symptoms manifest in the first two to four days after birth. These patients need emergent ophthalmology consultation, as rapid and severe corneal involvement ensues and subsequent corneal perforation can occur within two days. Infected neonates suspected of having gonorrhoeae can be treated with a single dose of ceftriaxone 125 mg intramuscular and topical polymyxin B-bacitracin ointment one drop every three hours for 10 days.
Similarly, C. trachomatis should be suspected if neonates present with conjunctival abnormalities on days 5-14 after birth. Since topical treatment alone is ineffective for neonatal Chlamydial conjunctivitis, this infection is treated with oral erythromycin 50 mg/kg per day divided QID for 14 days, often with adjunctive topical erythromycin ointment.5 For all forms of conjunctivitis, steroids generally should be avoided, as they can exacerbate infection, increase ocular pressure, and have been associated with cataract formation.6
Herpes Simplex Keratitis. Infections with ocular herpes simplex may be primary or a reactivation of preexisting disease. The physical exam often reveals only a red eye, but consider this diagnosis carefully, as the patient may or may not exhibit classic herpetic vesicles on the lids or conjunctiva. On slit lamp examination, lesions may appear as superficial punctate keratitis, ulcer, or may have the classic dendritic lesions. Management includes topical antiviral agents such as trifluridine 1% every two hours for 14 days.7
Prior to the Herpetic Eye Disease Study (HEDS) report, the standard therapy for all forms of herpes simplex virus keratitis was topical antivirals. The HEDS report was undertaken to assess the effect of adding corticosteroids to conventional therapy with trifluridine (TFT). It was a prospective, randomized, double-masked, placebo-controlled, multicenter study divided into several trials. Three were therapeutic trials. The first therapeutic group evaluated “Herpes Stromal Keratitis, Not on Corticosteroids.” Compared with the placebo group, patients receiving prednisolone phosphate drops had faster resolution and fewer treatment failures.8 The second therapeutic group evaluated “Herpes Stromal Keratitis, on Corticosteroid Treatment.” There was no apparent benefit in adding oral acyclovir to topical corticosteroids and TFT. However, visual acuity had improved over six months in more patients on acyclovir.9 The third group looked at “Herpes Simplex Virus Iridocyclitis, Receiving Topical Corticosteroids.” More treatment failures occurred in the placebo group vs. the acyclovir group, indicating a benefit to adding oral acyclovir (antiviral) to topical corticosteroids. Therefore, the emergency physician should include topical corticosteroids for faster resolution and to decrease treatment failures, and should also include antivirals to help improve long-term visual acuity.
Herpes Zoster Ophthalmicus. This infection occurs as activation of the herpes virus along the V2 ophthalmic division of the trigeminal nerve. Up to 250,000 cases occur annually in the United States, second in frequency only to thoracic dermatomal distribution. The disease process usually begins with a prodrome of influenza-like illness with fatigue, malaise, and low-grade fever, followed by skin hyperesthesia.1 The rash then follows dermatomal patterns and involves the forehead and upper eyelid, producing significant pain.
Careful attention should be paid to potential involvement of the nasociliary branch of the trigeminal nerve, which is manifested by zoster lesions on the tip of the nose. This physical exam finding is associated with a 76% risk of ocular involvement. Ophthalmic herpes zoster accounts for approximately 10-20% of all zoster cases and mandates emergent ophthalmologic consultation.7
Treatment is complex, with use of topical or intravenous (IV) antivirals, such as acyclovir 800 mg five times a day for 7-10 days, and consideration of topical steroids, such as dexamethasone. Antivirals can shorten the duration of symptoms and also shorten the duration of pain if taken within the first three days of symptom onset.10 Previous randomized, controlled studies support that oral administration of antivirals results in substantial vitreous penetration and is as effective as IV therapies.1
Uveitis. Uveitis is a broad term used to describe the inflammation of one or more layers of the eye. Its peak is in the middle-age or older population. Various etiologies, such as infectious or idiopathic, are responsible for uveitis, and milder presentations usually are seen if the injury is traumatic. Uveitis more commonly affects those with autoimmune disorders and is seen in 60% of patients with Wegener’s granulomatosis, 25% of patients with sarcoidosis, 20% of patients with ankylosing spondylitis, and 5% of patients with inflammatory bowel disease.11
Anterior inflammation is known as iritis, middle or vitreous inflammation is known as vitritis, and posterior inflammation is known as chorioretinitis. Acute anterior uveitis presents with a ciliary flush, and a perilimbal deep red injection is seen on physical examination. Patients will experience deep eye pain and photophobia, commonly consensual photophobia. Decreased visual acuity, irregularity of the pupil, and even nonreactive pupils can be seen. A slit lamp examination may reveal leukocyte cells and hypopyons or flare, both of which are characteristic in anterior uveitis.
Goals of treatment include reducing flare and inflammation and preserving vision, which is achieved by mydriatics such atropine 1% one drop twice a day or scopolamine 0.25% one drop two to four times a day. Topical corticosteroids, such as 1% prednisolone one drop every four to six hours, help treat active inflammation and prevent complications. Anterior uveitis usually resolves in days to weeks with treatment.12
Middle and posterior uveitis also presents with a red eye, but in contrast to anterior uveitis, it is painless. Patients may have impaired visual acuity that is associated with floaters, if caused from the middle eye, or symptoms of macular degeneration, if caused from the posterior eye. Slit lamp examination may show inflammatory cells or edema of the optic disc, which often is seen more in posterior uveitis.
Again, goals of therapy include reducing inflammation and preserving vision using systemic steroids and periocular therapy. Refractory uveitis occurs, and patients may require immunosuppressants such as mycophenolate or azathioprine. Middle and posterior uveitis usually has a more prolonged course compared to anterior uveitis. Uveitis should be evaluated thoroughly by ophthalmology. If a cause is not established, the patient likely needs to be evaluated for systemic illnesses, as uveitis is a complicating factor in many disease processes.13,14
Dacryocystitis. Dacryocystitis is an acute suppurative inflammation of the lacrimal sac due to obstruction of the nasolacrimal duct. It can occur acutely or as an exacerbation of chronic dacryocystitis. It is seen more commonly in children than in adults because of the lack of maturity of the nasolacrimal ducts. Nasolacrimal duct obstruction in adults can be primary, idiopathic, or secondary to a wide variety of infectious, inflammatory, traumatic, mechanical, toxic, or neoplastic causes.
The clinical manifestation of this condition starts as a dull aching to severe throbbing pain around the area of the sac. In addition, the area appears red, hot, and tender along the medial canthus. (See Figure 1.) The patient might present with constitutional symptoms, such as fever, headache, malaise, and watering eyes. The most important concerns with this condition are the complications that could occur if not properly managed. These complications may include lacrimal fistula with lacrimal abscess opening through the skin, pyocele, orbital cellulitis, and cavernous sinus thrombosis (the most dangerous complication). This infection also is a potential source of endophthalmitis.15 The most common organism in the pathology is Staphylococcus aureus, but streptococcus, pneumococcus, and anaerobes sometimes may be involved.
The treatment depends on its manifestation. In the case of cellulitis, systemic and topical antibiotics often are required, in addition to nonsteroidal anti-inflammatory drugs and pain control. For patients with lacrimal abscess, treatment may require incision and drainage, with or without dacryocystorhinectomy. In the case of fistula formation, antibiotics and fistulectomy plus dacryocystorhinectomy is the treatment of choice.
Figure 1. Dacryocystitis
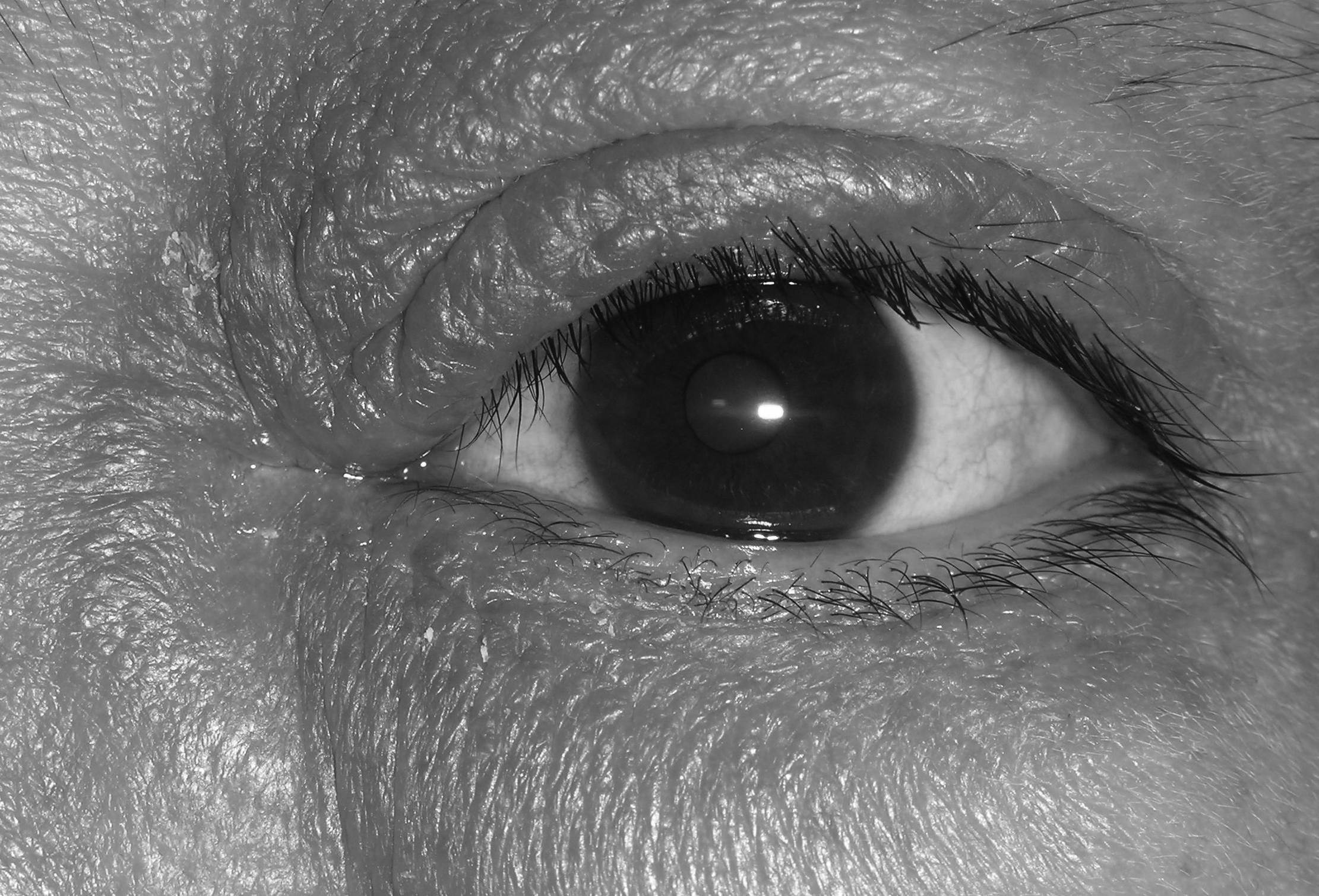
Image courtesy of J. Stephan Stapczynski, MD
Endophthalmitis. Endophthalmitis is an emergent, sight-threatening infection that usually occurs after ocular surgery, but may result from trauma, especially with globe penetration, or via hematogenous spread from a distal source. Other predisposing factors include diabetes and intravenous drug abuse, especially in fungal endophthalmitis. Both the anterior and posterior segments of the eye are involved in endophthalmitis. Onset is acute and painful, accompanied by redness and very rapidly progressing loss of vision. Irreparable visual loss can occur within 24-48 hours. Examination is very difficult because of pain and extreme photophobia. Chemosis and vitreous haze are common on examination. Causative organisms include S. aureus, Streptococcus pneumoniae, Bacillus cereus, and Candida albicans.16
In 70% of cases, endophthalmitis occurs as a complication of surgery. Postoperative endophthalamitis is divided into three categories. Acute endophthalmitis occurs within six weeks of surgery (usually within two weeks), with the peak incidence three to five days afterward. Delayed-onset endophthalmitis occurs more than six weeks after surgery. There also is a bleb-associated presentation of endophthalmitis, which can occur anytime after filtering surgery for glaucoma.
The initial complaint in patients with endophthalmitis is blurred vision.17 Other signs of endophthalmitis include edema and erythema of the lids, conjunctival chemosis, purulent ocular discharge, and corneal edema and opacification. Fever also can be present. A hypopyon often is seen in the inferior anterior chamber on physical examination. Proptosis is a late sign in the infection. Sudden onset of pain and vision loss is worrisome for endophthalmitis in a patient who has never undergone eye surgery and should prompt evaluation for hematogenous spread from a distant site. There are even reported cases of endophthalmitis causing bacteremia resulting in hematogenous spread.18
Although most cases are postoperative, with an incidence of 0.16% in ocular surgical patients, prophylactic antibiotics still are controversial. A systematic literature review failed to find any high-quality, randomized, controlled trials of prophylactic measures to prevent endophthalmitis following cataract surgery but concluded that current literature most strongly supports prevention using preoperative povidone-iodine antisepsis.19
Treatment requires immediate consultation with an ophthalmologist. For postoperative etiologies, intravitreal medications, such as vancomycin or ceftazidime, are the standard of care and should be given and managed by an ophthalmologist. This regimen also may be supplemented with topical antibiotics. Topical antibiotics such as vancomycin and amikacin plus atropine and prednisolone are good for empiric initial therapy until evaluation by ophthalmology is completed. Systemic antibiotics have not been shown to improve visual outcomes in acute postoperative endophthalmitis.
Most postoperative patients can be managed as outpatients with close continued care by ophthalmology. Cases of endophthalmitis resulting from hematogenous spread likely will need close monitoring with inpatient care.20 The most important prognostic factor for determining visual outcome is the patient’s visual acuity at presentation. Up to half of patients with endophthalmitis suffer major and irreparable visual loss.21
Corneal Ulcers. With potential complications that can rapidly and permanently affect sight, corneal ulcers are another ocular emergency. This infection typically starts as a keratitis, or inflammation of the cornea, and then proceeds to an ulcer. Patients present with decreased vision, pain, lid edema, tearing, and photophobia and may have visible redness. (See Figure 2.) A decrease in visual acuity is typical, and hypopyons commonly are seen. Although the corneal ulcer may be infectious, the hypopyon itself seldom contains organisms and is an example of the vascular response to acute inflammation.
Figure 2. Corneal Ulcer
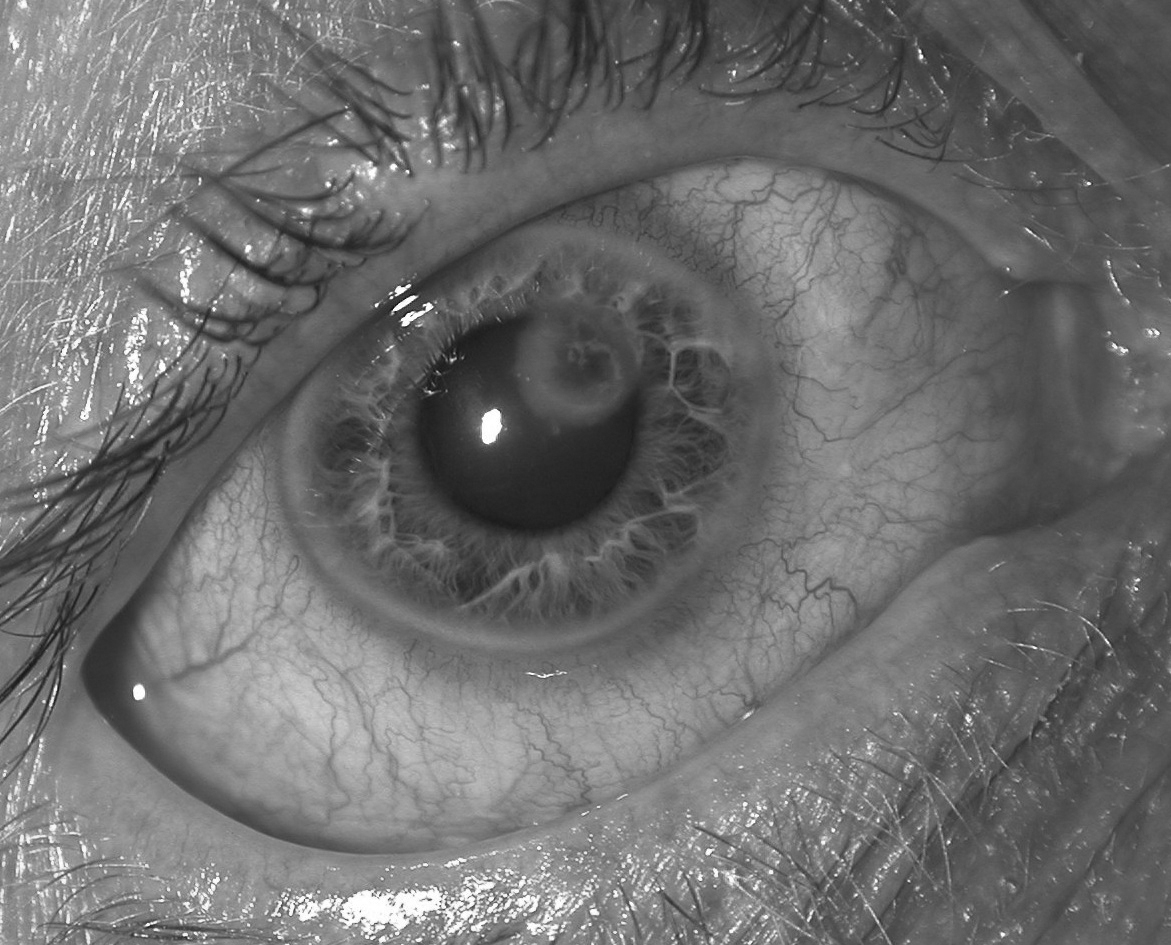
Image courtesy of J. Stephan Stapczynski, MD
Improper and overnight contact lens wear is the largest risk factor for ulcer formation, contributing to the local breakdown of the host defense mechanisms.22 Corneal ulcers, therefore, are seen most commonly in contact lens wearers. Although there are many organisms that can infect the cornea, Pseudomonas is a well-recognized pathogen. Pseudomonas rapidly destroys stromal tissue and leads to corneal perforation. While bacteria produce more severe and rapidly progressing corneal ulcers, fungal and herpetic infections are also potential causes. Thus, correct pathogen identification is important. Perforations and scarring from corneal ulcers are thought to cause approximately 10% of the blindness seen in the United States.23
On slit lamp examination, corneal ulcers are sharply demarcated infiltrates with a dense stromal inflammation.24 The corneal defect will stain with fluorescein. These patients need immediate consultation with, and follow-up referral to, ophthalmology.
Empiric treatment options include topical antibiotics, such as the fluoroquinolones, gatifloxacin, or moxifloxacin. Often, tobramycin is added to provide additional antibiotic coverage in case Pseudomonas is present. Frequency of dosing should be emphasized to patients because these drops need to be administered every hour for the first 48 hours.25
A secondary analysis of the Steroids for Corneal Ulcer Trial found that patients treated with topical prednisolone in conjunction with topical antibiotics had a better visual outcome.26 Patients treated with topical corticosteroids as adjunctive therapy within two to three days of antibiotic therapy had approximately one line better visual acuity at three months than did those given placebo, suggesting that the earlier the application of corticosteroids in corneal ulcers (but always after the initiation of antibiotics), the better preservation and improvement of vision long term.27
Preseptal/Periorbital Cellulitis. Inflammation of the lids and periorbital tissues without signs of orbital involvement, as suggested by signs such as proptosis or pain with extraocular movements, generally is referred to as preseptal or periorbital cellulitis and is a form of facial cellulitis. This is common in young children and may be caused by bacteremia, sinusitis, trauma, an infected wound, or an abscess of the lid or periorbital region, such as a hordeolum, conjunctivitis, dacryocystitis, or an insect bite.28 (See Figure 3.)
Figure 3. Preseptal Cellulitis
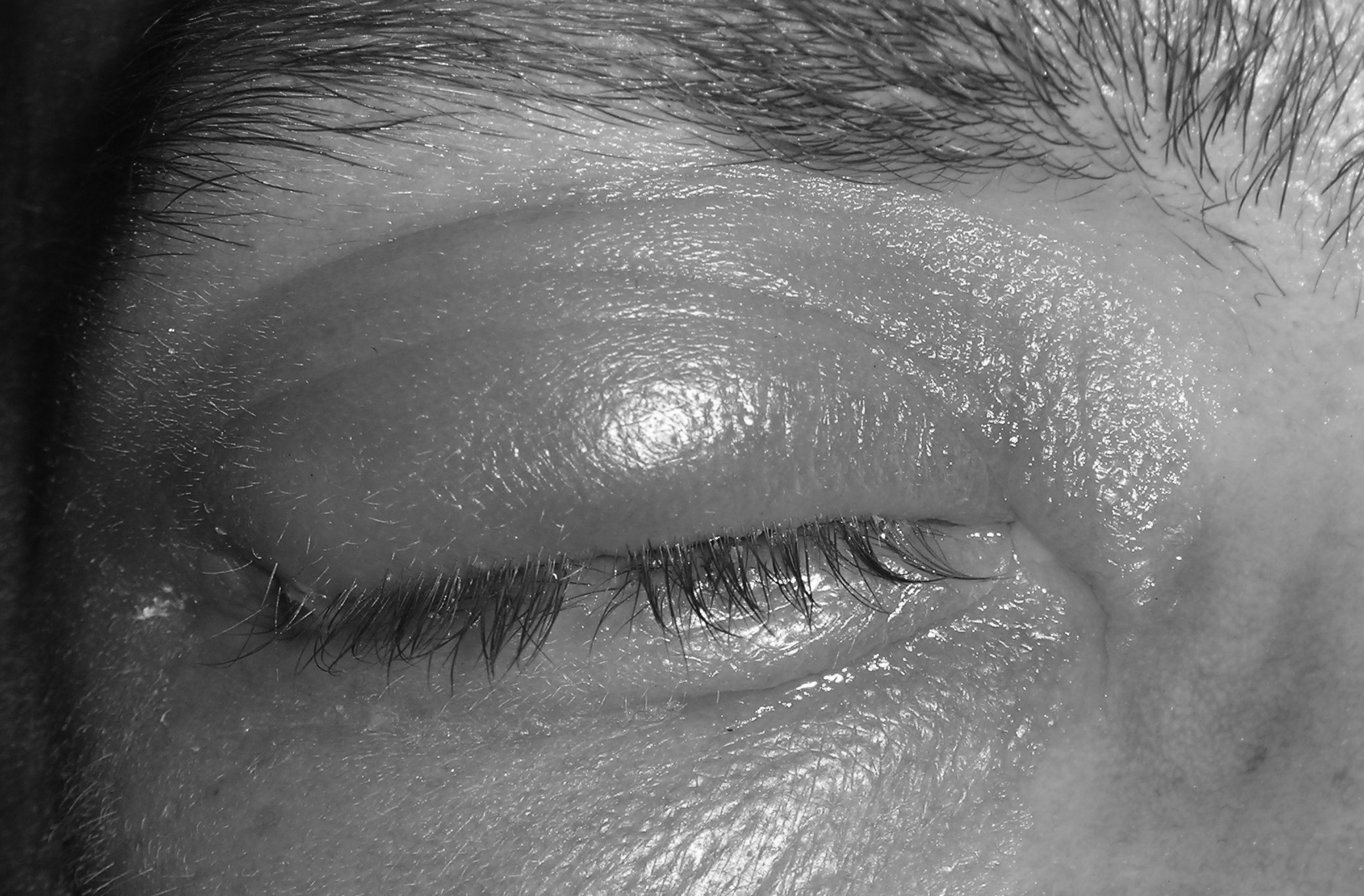
Image courtesy of J. Stephan Stapczynski, MD
Prior to the widespread use of the Haemophilus influenzae type b (Hib)immunization as part of the routine childhood vaccination schedule, H. influenzae was a serious potential cause of preseptal cellulitis in children, associated with bacteremia, pneumonia, and meningitis. Assessment and treatment regimens from that era included blood cultures, chest radiograph, and even lumbar puncture with admission for intravenous antibiotics. Since the Hib vaccine, H. influenzae type b has become a very uncommon cause of preseptal cellulitis in children. The current causes of preseptal cellulitis are S. pneumoniae, followed by S. aureus and Streptococcus pyogenes.
In nontoxic patients, effective treatment of preseptal cellulitis with oral antibiotics can be done as an outpatient. Antibiotic selection should include coverage for streptococcus and staphylococcus species. A typical choice would be amoxicillin/clavulanate 500/125 mg PO three times per day for 10-14 days in adults, and 40 mg/kg per day of the amoxicillin component divided into two or three times per day in children for 10-14 days.29 Alternatives for patients with penicillin allergy include cefpodoxime, cefdinir, and clindamycin.29
Hospitalization may be needed in patients with more severe disease, and IV forms of second- or third-generation cephalosporin, such as cefuroxime or ceftriaxone, are indicated.
CT imaging is not required in nontoxic patients when swelling does not impair assessment of the globe and its motility. If there is significant eyelid edema that interferes with assessment of the globe, then a CT scan is indicated to evaluate for orbital cellulitis. In preseptal cellulitis, the CT scan shows edema of the lids and subcutaneous tissues anterior to the septum, but the orbital contents are normal in appearance.
Postseptal/Orbital Cellulitis. Orbital cellulitis is an uncommon infection involving the ocular structures and adnexa posterior to the orbital septum. It may follow direct infection of the orbit from a wound, metastatic deposition of organisms during bacteremia, or, more often, direct extension or venous spread of infection from contiguous sites such as the lids, conjunctiva, globe, lacrimal gland, nasolacrimal sac, or, more commonly, the paranasal, particularly the ethmoid sinuses.29 (See Figure 4.)
Key features suggesting orbital rather than periorbital cellulitis include proptosis, impaired ocular motility, pain with eye movement, conjunctival chemosis, and toxic appearance.30 If any of these findings are present, orbital cellulitis or abscess should be expected, and the patient requires aggressive management with imaging studies.
Figure 4. Orbital Cellulitis
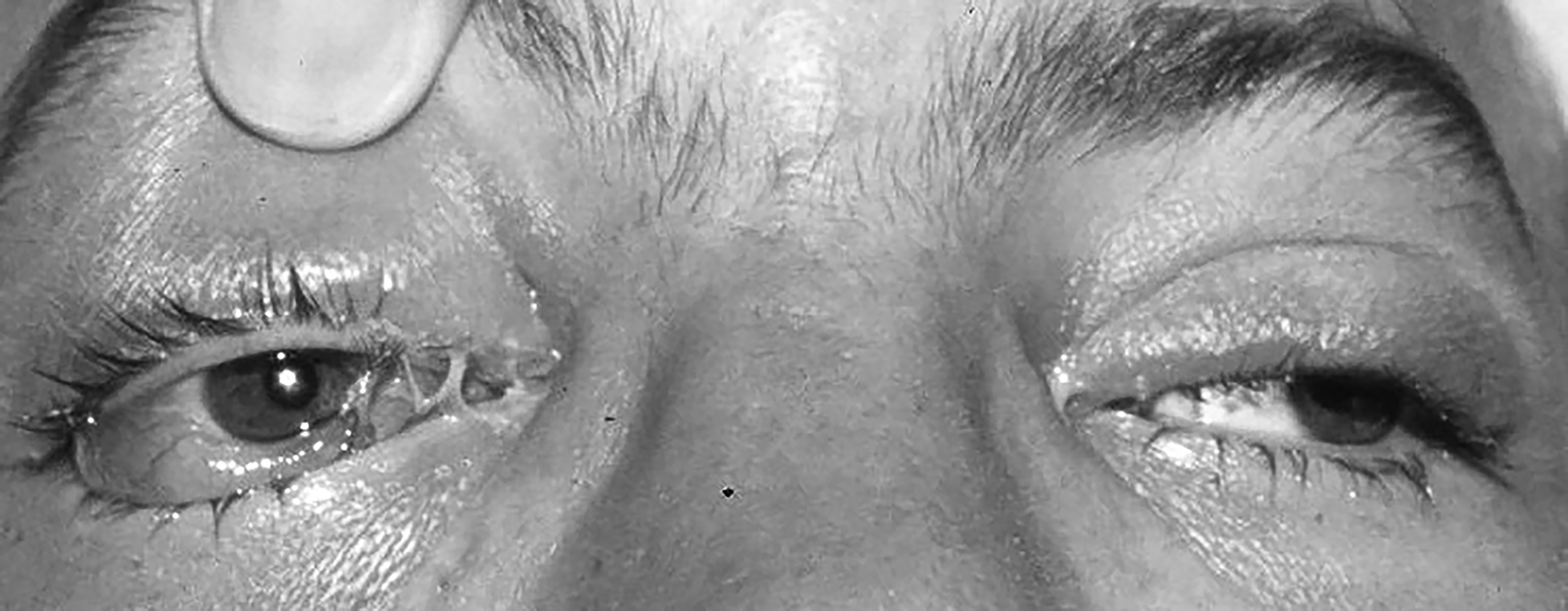
Image courtesy of J. Stephan Stapczynski, MD
All patients should have a CT of the brain and orbits, preferably with IV contrast to evaluate for any abscess, sinus diseases, or intracranial extension. These patients need hospitalization, as there is great potential for severe complications without proper treatment. Some of the concerning complications include vision loss due to an increase in orbital pressures, retinal artery occlusion, optic neuritis, meningitis, cavernous sinus thrombosis, and brain abscesses. Parenteral antibiotics are required and should begin with vancomycin and ceftriaxone, in addition to anaerobic coverage with metronidazole.31,32
Chemical and Radiation Burns
Chemical Burns
Eye contact with acids or alkalis requires emergent evaluation and treatment to prevent permanent vision loss. Burn severity depends on the pH of the agent involved, duration of exposure, and depth of penetration. On initial assessment, the severity of burns can be judged by the degree of corneal cloudiness and scleral whitening.
Alkaline substances typically cause more severe damage than acids because they saponify phospholipid membranes. This action leads to rapid epithelial cell death and caustic penetration into the eye. In severe cases of alkali exposure, the eye may appear white because of ischemia of the conjunctiva and scleral vessels. Concentrated ammonia is particularly dangerous and known to inflict severe injury to anterior ocular structures after less than one minute of exposure. Lye can cause deep eye injury within three to five minutes.
Acid burns usually are less devastating to the eye tissues and cause coagulation necrosis. The precipitation of proteins created in the process limits the depth of the injury. Contact may result in extensive corneal scarring and ulceration, possibly resulting in vision loss. Weak acids do not penetrate the anterior chamber easily, making prolonged irrigation generally unnecessary.
Detergents and aerosols, most commonly Mace®, typically cause conjunctival irritation only. More irritating chemicals can denude corneal epithelium and even cause anterior chamber inflammation.
Laundry detergent pods are of concern, as ocular pain or irritation is the most common presenting complaint to the ED after contact with these substances. It is thought that the increased likelihood of adverse events with these products is caused by their formulations, which are highly concentrated and include ethanolamine, alcohol ethoxylate, and benezenesulfonic acid, all of which can be ocular irritants.33 Thirty-three percent of children ages 5 years and younger who seek medical attention after exposure to laundry detergent pods do so for an ocular complaint.34
Management of chemical ocular exposure involves immediate copious irrigation with either water or isotonic saline. Topical anesthetics, such as tetracaine 0.5% one to two drops or proparacaine 0.5% one to two drops, may be required to ensure thorough irrigation, and repeat doses may be needed. Irrigation is needed until the pH of tear film is within the normal range, 7.0 to 7.3. Measurements of pH generally are taken at the fornix, the region between the conjunctiva and the lower eyelid. The pH should be checked again 10 minutes after cessation of irrigation.
Cyanoacrylate (Super Glue®) is also a common exposure. If the eye is sealed shut in normal anatomic position and cannot be opened with gentle traction, it may remain untouched and allow the glue to dissolve by physiologic mechanisms, which takes several days. Immediately after exposure, it might be possible to copiously irrigate the eye for 15 minutes to prevent adherence of the lids. However, the polymerization reaction of cyanoacrylate is so rapid that there is only a very short window of time for this to be initiated.
Gentle palpation of the closed lids can be performed to ensure that the globe moves freely and is not glued to the palpebral conjunctival surface.35 If the eyelids are bonded to the conjunctival surface, wash thoroughly with warm water and apply a gauze patch.
Discharge the patient with erythromycin ophthalmic ointment to apply every four hours to lubricate, soften, and help loosen the glue. Tetracaine drops will assist for analgesia, if needed, to help provide thorough examination. The cyanoacrylate often is mobile and does not adhere to the cornea, but if it has, do not attempt to forcefully scrape it off, as corneal abrasions will occur.36
Corneal abrasions are common from cyanoacrylate exposure, even if the lids were not adhered together, so erythromycin ophthalmic ointment every four hours for three days is recommended.37 If movement of the eye is compromised because the cornea or conjunctiva is glued to the inside of the lids, do not try to forcefully open the lids or remove the glue. Eye proteins in the tears will break down the inert chemical, usually over several hours, and the eye will regain mobility in one to four days.38,39
Radiation Burns
The toxic effects of acute high-dose ultraviolet radiation resulting in photochemical injury cause ultraviolet keratitis. The clinical syndrome presents with ocular pain, tearing, conjunctival chemosis, blepharospasm, and deterioration of vision, typically several hours after exposure and lasting up to three days. Mountaineers, skiers, and beach recreationalists are particularly at risk of ultraviolet keratitis, as the reflectivity in these environments is extremely high.40
Ultraviolet light damages the corneal epithelium. Patients usually have a latent period of six to 10 hours, and then develop photophobia, tearing, and intense pain. Use of topical anesthetics helps to enable physical examination, which usually reveals decreased visual acuity and injected conjunctiva. Fluorescein staining often reveals diffuse punctate corneal lesions.41
Management of radiation burns includes a short-acting cycloplegic, such as cyclopentolate, and typically a fluoroquinolone drop, such as moxifloxacin four times a day. A topical antibiotic ointment, such as erythromycin four times a day, is an acceptable alternative.40 One to three days of oral opioids may be needed for pain control.
While topical anethestics — tetracaine or proparacaine — are useful in the ED to control pain and facilitate examination, they are not prescribed commonly for home use because of the belief that such use will slow healing and lead to ulcer formation. This concept has been studied in a few small clinical trials of patients with corneal abrasions comparing topical local anesthetics with saline for one to three days after ED assessment.42 No difference in healing or persistent symptoms was observed, but also, there was no difference in pain scores. Thus, while topical anesthetics used at home for up to three days had no observed harm (at least for corneal abrasions), there was also no consistent benefit in pain control, although in one study, patients did rate topical tetracaine as globally better than saline.43
Vascular
Acute Closed-Angle Glaucoma
Glaucoma is the leading cause of irreversible blindness in the world, according to the World Health Organization. It is estimated that by 2020, about 5.9 million people will be blind because of primary angle closure glaucoma.44
This ocular emergency occurs most in patients who have anatomically small and shallow anterior chambers. This anatomic variation, particularly in Asians and Inuits, results in the iris being nearly in contact with the lens and results in resistance to aqueous humor flow from the posterior to the anterior chamber. The process is referred to as pupillary block. Episodes of closed-angle glaucoma are precipitated by pupillary dilatation.
Patients may present after being in a dimly lit room, after emotional upset, or while using various anticholinergic or sympathomimetic medications.1 Also, initiation of selective serotonin reuptake inhibitors is associated with a five-fold increase in the risk of acute closed angle glaucoma.47
When the pupil is dilated, this increases the degree of pupillary block and leads to an accumulation of aqueous humor in the posterior chamber, causing the iris to bulge forward. When the iris bulges forward, this seals off the angle between the cornea and the iris and obstructs the trabecular meshwork. This obstruction decreases outflow and leads to a rapid rise in intraocular pressure.
Symptoms are very abrupt in onset and are accompanied by severe eye pain, blurred vision, headache, nausea, and vomiting. Patients may report seeing a halo around lights. On physical exam, pupils appear cloudy in a midposition and are sluggish or fixed. (See Figure 5.)
Normal intraocular pressure is between 10-21 mmHg. Intraocular pressures in acute angle-closure glaucoma are markedly elevated, often 60-90 mmHg. If the attack has been prolonged, ischemia of the ciliary body reduces aqueous humor production with a resultant decrease in intraocular pressure. This process is especially important because the ultimate damage depends on the duration of the attack rather than the severity of the elevation in pressure.46
Figure 5. Acute Angle-Closure Glaucoma
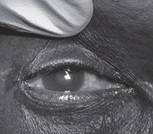
Hazy, fixed, and mid-dilated pupil. Note light shining into pupil without constriction.
Image courtesy of Chasi Skinner, DO
Treatment should be administered promptly. Therapy is geared toward decreasing aqueous production, increasing aqueous outflow, and reducing vitreous volume to lower IOP. If visual acuity is markedly reduced, a combination of topical glaucoma medications with IV acetazolamide should be used. If intraocular pressures are found to be less than 50 mmHg without significant visual acuity changes, the pathology can be managed without IV medications.
Topical timolol 0.5% will decrease intraocular pressure within 30-60 minutes. Pilocarpine 1-2% is a miotic agent to help decrease pressure and may be used every 15 minutes. An alpha-2 agonist, such as apraclonidine 1% for one dose, also is indicated. Topical steroids, such as prednisolone acetate 1% one to two drops, may be given every 15 minutes for four doses.
In attacks with severe visual deficits, a carbonic anhydrase inhibitor, such as acetazolamide 250-500 mg IV, should be given, and practitioners should consider the addition of mannitol 250-500 mg/kg IV over 45 minutes.1,48 Patients may need antiemetic therapies and sedatives, and emergent ophthalmologic consultation is warranted. Definitive therapy for angle-closure glaucoma is surgical with iridectomy.
Central Retinal Artery Occlusion
Central retinal artery occlusion (CRAO) causes an ischemic stroke of the retina, and is the ocular equivalent of an acute cerebral ischemic event. It is caused by partial or complete occlusion of the central retinal artery, most commonly by thromboembolus from the ipsilateral carotid artery, aortic arch, or heart. It presents as a profound painless monocular vision loss with potential for significant functional morbidity, with 80% of affected patients having final visual acuity of counting fingers or worse.49
The incidence is estimated to be roughly 1.9/100,000 in the United States.49 It most commonly occurs in patients between 50-70 years of age, and 45% have carotid artery disease. The risk factors for CRAO include hypertension, diabetes, collagen vascular disease, cardiac valvular abnormality, and sickle cell disease.50
Patients typically present with the complaint of a severe loss of vision that develops over seconds. Examination of the eye reveals markedly reduced visual acuity with the presence of an afferent pupillary defect. The fundoscopic examination will reveal a retina that is pale white and the fovea appears as a cherry-red spot, which is the pathognomonic finding.
Therapy should be initiated immediately and should be directed at dislodging the embolus, dilating the artery to promote blood flow, and reducing intraocular pressure to allow an increase in perfusion. Digital globe massage should be initiated immediately in the ED. This is achieved by applying direct digital pressure over a closed eyelid for 10-15 seconds followed by quick release. Applying timolol 0.5% topically may help preserve vision by decreasing intraocular pressure. Acetazolamide 500 mg IV is indicated, as it not only lowers intraocular pressure, but also increases retinal blood flow.
Emergent ophthalmologic consultation should be obtained, as anterior chamber paracentesis may be attempted in efforts to lower the intraocular pressure. Recent studies suggest more aggressive treatment with fibrinolytics (with tPA being the most studied) are beneficial for visual outcomes, especially if given within 4.5 hours of symptom onset, with a number needed to treat of four.51
Up to 24% of patients with acute retinal ischemia have concomitant cerebral infarctions on brain diffusion-weighted MRI and require additional evaluation.52 Thus, it is reasonable to obtain a diffusion-weighted MRI of the brain to look for concurrent cerebral ischemia, even in patients without other neurologic symptoms.
Central Retinal Vein Occlusion
Central retinal vein occlusion (CRVO) is another acute, painless loss of vision that develops from blockage of the central retinal vein due to edema, hemorrhage, or vascular leakage. This is often referred to as the “deep vein thrombosis of the eyes.” Central retinal vein occlusion usually affects patients older than 50 years of age and is associated commonly with hypertension, diabetes, or glaucoma. For young patients, central retinal vein occlusion occasionally is associated with acquired or hereditary hypercoagulable states or systemic inflammation, typically in patients with personal or family history of venous thromboembolism.53,54 Depending on the degree of obstruction and the presence of collateral flow, the loss of vision can range from minimal to severe.1
There are both non-ischemic and ischemic forms of CRVO. Non-ischemic forms involve mild fundus changes, do not have associated pupillary defect, and present with less severe forms of visual loss. Patients with ischemic CRVO have a marked decrease in visual acuity and usually are seen with afferent pupillary defects. Both types of CRVO, ischemic and non-ischemic, share similar findings: dilated retinal veins and retinal hemorrhages in all four quadrants. The retinal hemorrhages seen in CRVO can be very diffuse, producing what is termed a “blood and thunder” appearance. This process is usually unilateral in the affected eye. Bilateral retinal hemorrhages likely suggest hypertension, diabetes mellitus, or hyperviscosity syndrome.
The management of CRVO is very complex and includes, once again, lowering intraocular pressures, topical steroids, and photocoagulation. Prognosis, as seen with severity of symptoms, depends on the degree of the obstruction.54
Vitreous Hemorrhage
The incidence of vitreous hemorrhage is about seven in every 100,000 population, making it one of the most common causes of acute or subacute visual loss.55 The composition of the vitreous humor is 99% water and 1% collagen and hyaluronic acid. This gives it optical clarity and gelatinous characteristics.55 The vitreous space accounts for about 80% of ocular volume, and it is attached to the retina in three different places. The strongest attachment is at the vitreous base, followed by the optic nerve head and the retinal vasculature.
The mechanism of vitreous hemorrhage falls into the following three categories: abnormal vessels that are prone to bleeding, normal vessels that rupture under duress, and extension of blood from adjacent sources.55
A common eye symptom is floaters, which are opacities in the vitreous body that cast shadows onto the retina and appear to patients as small moving spots or specs in the visual field. They may appear as lines, circles, dots, cobwebs, clouds, or any other shape. Floaters move as the eye moves, but do not follow eye movements precisely. They are seen most commonly when looking at something bright like white paper, a plain white wall, or blue sky.56 The most common causes of floaters include diabetic retinopathy and retinal tears. Floaters also can be seen with branch vein occlusion, sickle cell disease, retinal detachment, trauma, macular degeneration, microaneurysms, and intraocular tumor.
Vitreous hemorrhage often is heralded with floaters or what patients may describe as “cobwebs.” Over the next few hours, as blood accumulates in the preretinal space or vitreous cavity, profound vision loss occurs without any eye pain. On funduscopic exam, details of the fundus will be very difficult to visualize because the hemorrhage can be distributed widely.
Ultrasound is an important diagnostic tool for the emergency physician and can help identify the cause of hemorrhage.58 Intravitreal hemorrhage produces easily detectable diffuse dots and blobs of vitreal echoes that correlate with the amount of blood present. Vitreous hemorrhage induces a vitreous gel liquefaction and separation from the retina, manifested by rapid, staccato movement of the vitreous echoes on ultrasound during eye movement. Point-of-care ultrasound in the ED by the emergency physician is quite effective in making the diagnosis and has a 92% sensitivity in identifying vitreous hemorrhage.59
Retinal Tears and Detachments
A tear in the retinal membrane may or may not lead to retinal detachment. The retina consists of two layers, the inner neuronal layer and the outer pigment epithelial layer, which can be separated by fluid accumulation. About five per 100,000 population develops retinal detachment yearly, with almost one in 300 chances of lifetime development.1
Detachment of the retina occurs from either rhegmatogenous, exudative, or tractional etiologies. Rhegmatogenous detachment is the most common type and it occurs as a result of a compromise of the retinal tissue or the neuronal layer, which allows sub-retinal fluid to accumulate and dissects the two layers (inner neuronal and outer pigment epithelial). This type of detachment is often seen in patients older than 45 years of age. It is more common in men than in women and can be induced by trauma. Patients with severe myopia are at the greatest risk.1
Exudative retinal detachment results when fluid or blood leakage from vessels collects in the retina. Exudative detachment is driven by hypertension, glomerulonephritis, papilledema, vasculitis, and pregnancy. Tractional detachment is seen from fibrous band formation in the vitreous and then the contraction of these bands.
Patients often will complain of flashes of light related to the traction on the retina, floaters from vitreal blood, or pigmented debris and loss of vision. For patients with acute onset of floaters and/or flashes who are self-referred or referred to an ophthalmologist, the prevalence of retinal tear is 14%.60 Visual loss is described as cloudy, filmy, or curtain-like. This is a painless condition. Visual acuity greatly varies from being mildly affected to severely affected. Visual field cuts correlate to the location of the retinal detachment. Retinal detachment cannot be ruled out by direct fundoscopy. Indirect ophthalmoscopy is needed to visualize anterior portions of the retina.
ED ultrasonography is helpful to determine presence of retinal detachment. Point-of-care ultrasonography by the emergency physician has a sensitivity of 91%.61 With ultrasonography, a detached retina appears as a highly reflective sheet-like tissue within the vitreous space. Small detachments often appear dome-like on imaging. The appearance of total retinal detachment, which anatomically is cone-shaped, varies depending upon the position of the examining probe. Axial images are funnel-shaped with attachment to the optic nerve head. Coronal images show a circular cross section of the cone. Acute and recent detachments have a characteristic undulating movement slower than that of the vitreous gel. Long-standing detachments appear stiff because of proliferation of scar tissue on the retinal surface.62 Additionally, it is prudent for the emergency physician to be aware of ultrasonographic mimics, such as posterior vitreous detachment and vitreous hemorrhage.
ED intervention includes stabilizing the patient and ensuring prompt ophthalmologic follow-up. It is imperative that the ED physician communicate with the ophthalmologist, provide the name and contact information of the ophthalmologist to the patient, document conversations, and inform the patient of the need to follow up promptly.
Neurologic
Optic Neuritis
Optic neuritis is an acute inflammation of the optic nerve and is referred to as “acute inflammatory optic neuropathy.” It often is manifested as the classic triad of decreased visual acuity, pain with extraocular movement, and dyschromatopsia (impairment of color vision). The initial age at presentation often ranges from 15 to 45 years.63 Typically, symptoms begin insidiously over several hours or days and are accompanied by ocular pain during eye movement, specifically retrobulbar pain. Pain with eye movements precedes the vision loss in approximately 90% of cases.63 Pain typically lasts three to five days, and if pain persists longer, other etiologies should be strongly considered.
There are several causes of optic neuritis, such as genetic, autoimmune, nutritional, infectious, compressive, and toxic and drug-induced. The role of the emergency physician is to identify the likely causes for which ED intervention would improve prognosis. The infectious causes to consider include: bacterial (tuberculosis, Lyme disease, syphilis, or meningitis), viral (measles, herpes, mononucleosis, encephalitis), and respiratory infections (mycoplasma pneumonia). The toxic and drug-induced causes to consider in the ED include: methanol, ethambutol, and isoniazid poisoning.
Cases of acute optic neuritis often present with unilateral visual field abnormalities. Bilateral optic neuritis should warrant evaluation for systemic inflammatory disorder, infection, or paraneoplastic disease. Visual acuity can range from 20/20 in 10% of patients to no light perception. Visual loss usually is central, and visual decline tends to reach its plateau within days to weeks and then improves over weeks to months.64
Evaluation and diagnosis of optic neuritis in the ED requires recognizing the classic triad. A detailed ophthalmologic exam with understanding of what to look for often is required. The presence of ipsilateral afferent pupillary defect or the so-called “Marcus Gunn pupil” with direct light to the pupil is the norm. Direct opthalmoscopic examination reveals a normal or, in up to one-third of patients, a swollen disk.
Roughly 30% of patients with optic neuritis will develop multiple sclerosis over the next five years.65 When suspected in the ED, high-dose methyl-prednisone should be initiated with ophthalmologist consultation. In studies, treatment with a three-day course of IV methylprednisolone reduced development of multiple sclerosis over a two-year period, but no significant differences were seen over a five-year period. Long-term visual outcome seems to be no different from treatment vs. observation alone.65
The majority of these patients will be admitted for at least a three-day course of IV methylprednisone in addition to a detailed workup that is beyond the purview of the ED.66
Acute Third Cranial Nerve Palsy
There are several causes of isolated third nerve palsy, including vasculitic infarction (e.g., giant cell arteritis), infection (e.g., herpes zoster ophthalmicus), chemotherapeutic toxicity, internal carotid artery dissection or occlusion, and posterior communicating (PCOM) artery aneurysm.67
The differential diagnosis of isolated third nerve palsy is not as lengthy as for fourth and sixth nerve palsies because of the many structures innervated by the third nerve and the characteristic findings. It is important to note that third cranial nerve palsy is associated more frequently with aneurysm than are fourth or sixth nerve palsies, hence the need for prompt and thorough evaluation of a potentially imminent life-threatening underlying etiology of the palsy.
A complete third cranial nerve palsy will manifest as an ipsilateral eye that is deviated inferiorly and laterally. The physician also may see ipsilateral absence of ocular elevation, depression, adduction, ipsilateral complete ptosis, or an ipsilateral enlarged and nonreactive pupil. (See Figure 6.) It is highly important to identify the presence of pupil involvement vs. pupil sparing with regard to potential lesion etiology. Pupillary fibers are located superomedially near the surface of the nerve and are particularly prone to compression by a PCOM aneurysm. Thus, pupillary dysfunction along with a third cranial nerve palsy supports the likelihood of a PCOM aneurysm.68
Figure 6. Right-sided Third Cranial Nerve Palsy Due to PCOM Aneurysm
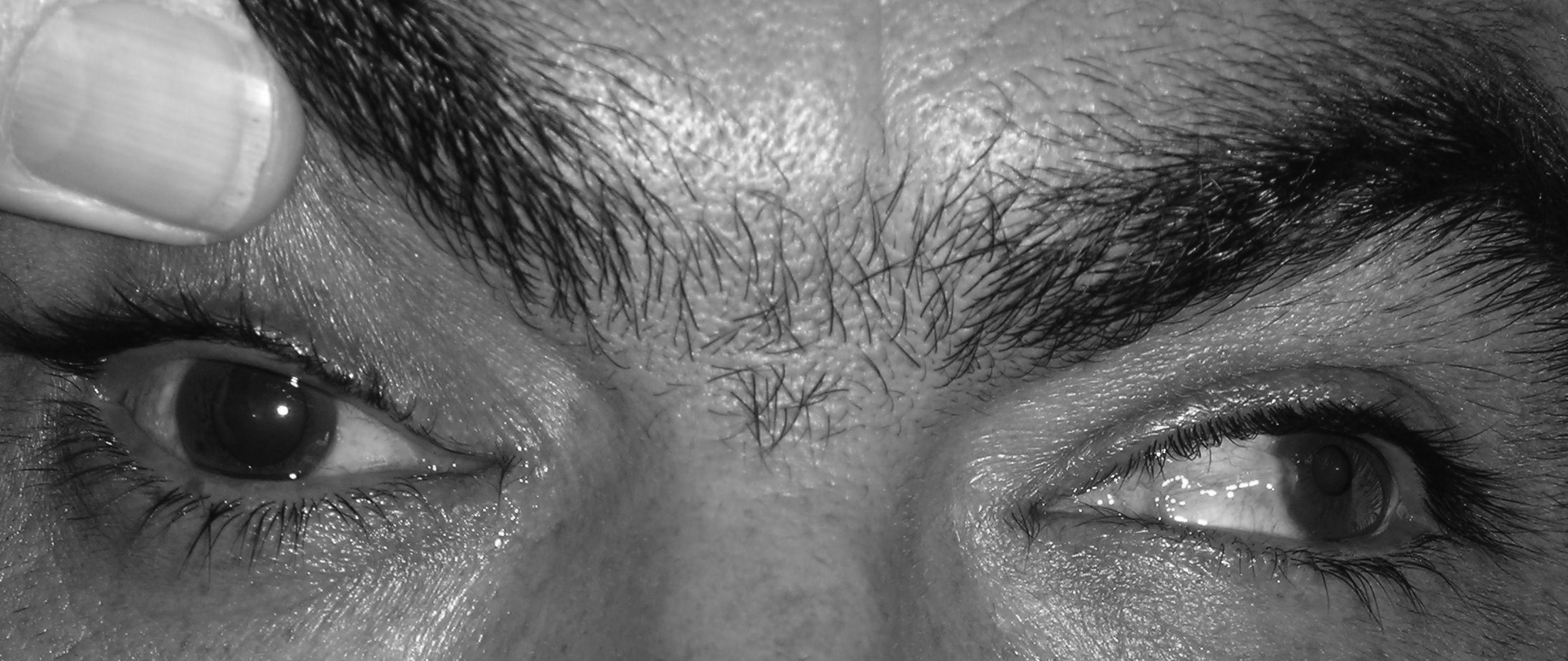
Image courtesy of J. Stephan Stapczynski, MD
In vasculitic third nerve palsy, pain often precedes the onset of ptosis or diplopia, the pupillary reaction is usually spared, and the pupil does not become enlarged. The pupil is spared because of the location of the infarction, which occurs in the center of the nerve, and the pupillary fibers are located in the nerve periphery where there is good collateral supply.69 A vasculitic third nerve palsy has a relatively benign course and recovery is expected over weeks to usually three months.
An ophthalmoplegic migraine refers to a syndrome that consists of migraine-type headache and a third nerve palsy; the pupil usually is involved. Most patients have an antecedent worsening in migraine severity that precedes the oculomotor paresis and is intense, continuous, and located in the orbital region.70 As the paralysis reaches its maximum, the headache begins to recede. The initial presentation is usually in childhood, multiple attacks may occur, and a family history of migraine often is present. The third nerve palsy may last from hours to weeks, and permanent deficits occur after repeated attacks.
Although aneurysmal compression must be excluded in isolated third cranial nerve palsies, other mass lesions also cause relative pupillary spared third nerve palsies.71 Therefore, in addition to MRA or CTA, standard MR or CT imaging is required. Compression of the third cranial nerve from an expanding aneurysm is a true neuro-ophthalmic emergency.
Summary
Ocular disorders seen in the ED can range from mild and self-limited to severe and vision-threatening. The standard ophthalmologic exam with selected ancillary tests enables the emergency physician to make appropriate diagnosis and initiate treatment in the majority of these patients.
REFERENCES
- Marx, JA, Pfaff J, Moore G, et al. Rosen’s Emergency Medicine. 8th ed., Ophthalmology, Philadelphia, Elsevier, 2014;71:1054-1077.
- Resnikoff S, Pascolini D, Etya’ale D, et al. Global data on visual impairment in the year 2002. Bull World Health Organ 2004;82:844-851.
- Channa R, Zafar NS, Canner JK, et al. Epidemiology of eye-related emergency department visits. JAMA Ophthalmol 2016;134:312-319.
- Azari AA, Barney NP. Conjunctivitis: A systematic review of diagnosis and treatment. JAMA 2013;310:1721.
- Sheikh A, Hurwitz B. Antibiotics for acute bacterial conjunctivitis: A systematic review. Clinical Science 2000;98:15P.3-15P.
- Mauer P, Pearson R. Infectious conjuncitivitis. In: First Consult. Philadelphia: Elsevier; 2013.
- Yanoff M, Duker JS. Herpes Simplex Keratitis. In: Ophthalmology, 1st ed. Pittsburgh: Elsevier/Saunders; 2014.
- Wilhelmus KR, Gee L, Hauck WW, et al. A controlled trial of oral acyclovir for the prevention of stromal keratitis or iritis in patients with herpes simplex virus epithelial keratitis. Arch Ophthalmol 1997;115:703.
- Cobo LM, Foulks GN, Liesegang T, et al. Oral acyclovir in the therapy of acute herpes zoster ophthalmicus. Ophthalmology 1985;92:1574-1583.
- Ragozzino MW, Melton LJ 3rd, Kurland LT, et al. Population-based study of herpes zoster and its sequelae. Medicine 1982;61:310-316.
- Mathur, Umang et al. Clinical efficacy of oral and topical acyclovir in herpes simplex virus stromal keratitis. Ind J Ophthalmol 2016;64:292.
- Deschenes J, Murray PI, Rao NA, et al; International Uveitis Study Group (IUSG). Clinical classification of uveitis. Ocul Immunol Inflamm 2008;16:1-2.
- Alexander KL, Dul MW, Lalle PA, et al. Optometric Clinical Practice Guideline: Care of the Patient with Anterior Uveitis. St Louis, MO: American Optometric Association; 1994.
- Hooper C, McCluskey P. Intraocular inflammation: Its causes and investigations. Curr Allergy Asthma Rep 2008;8:331-338.
- Butler F, Chalfin S. The eye in the wilderness. In: Auerbach PS, Cushing TA, Harris NS, eds. Auerbach’s Wildnerness Medicine. Philadelphia: Elsevier; 2017:1109-1127.
- Hoffman R. Conjunctivitis. In: Ferri F. Ferri’s Clinical Advisor 2017. Philadelphia: Elsevier; 2017.
- Naradzay J, Barish RA. Approach to ophthalmologic emergencies. Med Clin North Am 2006;90:305-328.
- Olitsky SE, Hug D. Ocular infections. In: Nelson Essentials Of Pediatrics. Philadelphia: Saunders; 2016:3041-3044.
- Ng JQ, Morlet N, Pearman JW, et al. Management and outcomes of postoperative endophthalmitis since the endophthalmitis vitrectomy study: The Endophthalmitis Population Study of Western Australia (EPSWA)’s fifth report. Ophthalmology 2005;112:1199-1206.
- Ciulla TA, Starr MB, Masket S. Bacterial endophthalmitis prophylaxis for cataract surgery: An evidence-based update. Ophthalmology 2002;109:13-24.
- Uveitis. In: Yanoff M, Duker JS. Ophthalmology: Expert Consult. St. Louis: Mosby Elsevier; 2012.
- Stryjewski TP, Chodosh J, Kim IK, et al. Severe corneal ulcer with progression to endophthalmitis and high-grade bacteremia. Am J Ophthalmol Case Rep 2017;6:30-32.
- Kassam F, Lee BE, Damji KF. Concurrent endophthalmitis and orbital cellulitis in a child with congenital glaucoma and a glaucoma drainage device. Digit J Ophthalmol 2011;17:58-61.
- Liesegang TJ. Contact lens-related microbial keratitis: Part I: Epidemiology. Cornea 1997;16:125-131.
- Farahani M, Patel R, Dwarakanathan S. Infectious corneal ulcers. Dis Mon 2017;63:33-37.
- Ray KJ, Srinivasan M, Mascarenhas J, et al. Early addition of topical corticosteroids in the treatment of bacterial keratitis. JAMA Ophthalmol 2014;132:737-741.
- Herretes S, Wang X, Reyes JM. Topical corticosteroids as adjunctive therapy for bacterial keratitis. Cochrane Database Syst Rev 2014;(10):CD005430.
- Marcdante KJ, Kliegman RM. Ocular infections. In: Nelson Essentials of Pediatrics. Philadelphia; Elsevier Saunders: 2015:386-389.
- Amin RM, Hussein FA, Idriss HF. Pathological, immunohistochemical and microbiological analysis of lacrimal sac biopsies in patients with chronic dacrocystitis. Int J Ophthalmol 2013;6:817-826.
- Olitsky SE, Hug D, Plummer L, et al. Orbital cellulitis. In: Kliegman RM, ed. Nelson Textbook of Pediatrics. Philadelphia: Elsevier; 2016:3062-3064.
- Takle LM, Bernardino RC. A rapidly emerging ocular adnexal villain. Rev Ophthalmology 2007;14:115-120.
- Nageswaran S, Woods CR, Benjamin DK, et al. Orbital cellulitis in children. Pediatr Infect Dis J 2006;25:695-699.
- American Academy of Ophthalmology. Preferred Practice Pattern Guidelines: Bacterial Keratitis – Limited Revision, 2011.
- American Association of Poison Control Centers. AAPCC and poison centers issue warning about concentrated packets of laundry detergent. American Association of Poison Control Centers, Alexandria, Virginia; May 17, 2012.
- Centers for Disease Control and Prevention. Health hazards associated with laundry detergent pods. Morb Mortal Wkly Rep 2012;61:825-829.
- Superglue Corp. Removing superglue. Available at: http://www.supergluecorp.com/?q=removingsuperglue.html. Accessed May 17, 2017.
- Cohen J. Super glued shut. Brown Emergency Medicine. April 12, 2017. Available at: http://brownemblog.com/blog-1/2017/4/12/super-glued-shut. Accessed May 17, 2017.
- Reddy SC. Superglue injuries of the eye. Int J Ophthalmol 2012;5:634-637.
- Terman SM. Treatment of ocular super glue instillation. J Trauma 2009;66:E70-77.
- Pavan-Langston D. Manual of Ocular Diagnosis and Therapy, 6th ed. Philadelphia; Lippincott Williams and Wilkins: 2007.
- Willmann G. Ultraviolet keratitis: From the pathophysiological basis to prevention and clinical management. High Alt Med Biol 2015;16:277-282.
- Puls HA, Cabrera D, Murad MH, et al. Safety and effectiveness of topical anesthetics in corneal abrasions: Systematic review and meta-analysis. J Emerg Med 2015;49:816.
- Waldman N, Densie IK, Herbison P. Topical tetracaine used for 24 hours is safe and rated highly effective by patients for the treatment of pain caused by corneal abrasions: A double-blind, randomized clinical trial. Acad Emerg Med 2014;21:374.
- See JL, Aquino MC, Aduan J, Chew PT. Management of angle closure glaucoma. Indian J Opthalmol 2011;59(Suppl1):S82-S87.
- Goldstein J, Edlow J. Headache. In: Adams JG, Barton ED, Collings J, et al. Emergency Medicine: Clinical Essentials. Philadelphia: Elsevier Saunders; 2013;881-889.
- Bhatia K, Sharma R. Eye emergencies. In: Adams JG, Barton ED, Collings J, et al. Emergency Medicine: Clinical Essentials. Philadelphia: Elsevier Saunders; 2013: 209-225.
- Chen HY, Lin CL, Lai SW, Kao CH. Association of selective serotonin reuptake inhibitor use and acute angle-closure glaucoma. J Clin Psychiatry 2016;77:e692.
- Seltz LB, Smith J, Durairaj VD, et al. Microbiology and antibiotic management of orbital cellulitis. Pediatrics 2011;127:e566-572.
- Cugati S, Varma DD, Chen CS, Lee AW. Treatment options for central retinal artery occlusion. Curr Treat Options Neurol 2013;15:63-77.
- Dattilo M, Biousse V, Newman N. Update on the management of central retinal artery occlusion. Neurol Clin 2017;35:83-100.
- Schrag M, Youn T, Schindler J, et al. Intravenous fibrinolytic therapy in central retinal artery occlusion: A patient-level meta-analysis. JAMA Neurol 2015;72:1148-1154.
- Biousse V, Newman N. Retinal and optic nerve ischemia. Continuum 2014;20:838-856.
- McIntosh RL, Rogers SL, Lim L, et al. Natural history of central retinal vein occlusion: An evidence-based systematic review. Ophthalmology 2010;117:1113-1123.
- Garoon R, Carvounis P. Central retinal vein occlusion with bilateral stenosis of the internal carotid arteries. Lancet 2015;387:914.
- Berdahl JP, Mruthyunjaya P. Vitreous hemorrhage: Diagnosis and treatment. EyeNet Magazine 2007; March.
- Cline D, Hofstetter HW, Griffin JR. Dictionary of Visual Science. Boston: Butterworth-Heinemann: 1997.
- Conart JB, Berrod JP. [Non-traumatic vitreous hemorrhage]. J Fr Ophthalmol 2016;39:219-225.
- Rabinowitz R, Yagev R, Shoham A, Lifshitz T. Comparison between clinical and ultrasound findings in patients with vitreous hemorrhage. Eye 2004;18:253-256.
- Parchand S, Singh R, Bhalekar S. Reliability of ocular ultrasonography findings for pre-surgical evaluation in various vitreo-retinal disorders. Semin Ophthalmol 2014;29:236-241.
- Hollands H, Johnson D, Brox AC, et al. Acute-onset floaters and flashes: Is this patient at risk for retinal detachment? JAMA 2009;302:2243-2249.
- Jacobsen B, Lahham S, Lahham S, et al. Retrospective review of ocular point-of-care ultrasound for detection of retinal detachment. West J Emerg Med 2016;17:196-200.
- Coleman DJ, Silverman RH, Lizzi FL, et al. Ultrasonography of the Eye and Orbit. Philadelphia: Lippincott Williams & Wilkins; 2006.
- [No authors listed.] Optic Neuritis Study Group. The clinical profile of optic neuritis. Experience of the Optic Neuritis Treatment Trial. Arch Ophthalmol 1991;109:1673-1678.
- Martin TJ, Corbett JJ. Neuroophthalmology. In: Winn H. Youmans and Winn Neurological Surgery. Philadelphia: Elsevier; 2017:1-28.
- Optic Neuritis Study Group. The 5-year risk of MS after optic neuritis. Experience of the optic neuritis treatment trial. Neurology 1997;49:1404-1513.
- Kedar S. Optic neuritis. In: Ferri F. Ferri’s Clinical Advisor. Philadelphia: Elsevier; 2017:889.
- Nizam A, Yacoub HA, McKinney JS. Internal carotid artery dissection heralded by an oculomotor nerve palsy: Case report and literature review. Neurologist 2011;17:333-337.
- Rucker JC, Thurtell M. Cranial neuropathies. In: Daroff RB, Jankovic J, Mazziotta JC, Pomeroy SL, eds. Bradley’s Neurology in Clinical Practice. Philadelphia: Elsevier; 2016:1720-1735.
- Akagi T, Miyamoto K, Kashii S, Yoshimura N. Cause and prognosis of neurologically isolated third, fourth, or sixth cranial nerve dysfunction in cases of oculomotor palsy. Jpn J Ophthalmol 2008;52:32-35.
- Lal V, Sahota P, Singh P, et al. Ophthalmoplegia with migraine in adults: Is it opthalmoplegic migraine? Headache 2009;49:838-850.
- Jacobson DM, Trobe JD. The emerging role of magnetic resonance angiography in the management of patients with third cranial nerve palsy. Am J Ophthalmol 1999;128:94-96.
Evaluating eye-related complaints in the ED requires a good understanding of the anatomy and potential implications of failure to treat. Although most ED presentations are nonemergent, it is important that the emergency physician identify the emergent presentations and manage them promptly to prevent potential vision loss.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
