Systolic and Diastolic Heart Failure
AUTHORS
Leslie Anne Juarbe Rivera, MD, MPH, Emergency Medicine Surgical Critical Care Fellowship, R Adams Cowley Shock Trauma Center, University of Maryland, Baltimore.
Jay Menaker, MD, Associate Professor of Surgery and Emergency Medicine, University of Maryland School of Medicine; Medical Director, Lung Rescue Unit; Medical Director, Critical Care Resuscitation Unit, R Adams Cowley Shock Trauma Center, Baltimore, MD.
PEER REVIEWER
Steven M. Winograd, MD, FACEP, Assistant Professor of Clinical Emergency Medicine, New York Institute of Technology College of Osteopathic Medicine, Old Westbury, NY; St. John’s Riverside, Yonkers, NY; CityMD, New York City.
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Farel (CME question reviewer) owns stock in Johnson & Johnson. Ms. Light (nurse planner) reports that she serves as a consultant for Bard Medical. Brian Hocum, PharmD, (pharmacist reviewer) reports that he is an employee of United Therapeutics. Dr. Schneider (editor), Dr. Stapczynski (editor), Dr. Juarbe Rivera (author), Dr. Menaker (author), Dr. Winograd (peer reviewer), Ms. Mark (executive editor), Ms. Coplin (executive editor), and Ms. Hatcher (AHC Media editorial group manager) report no financial relationships with companies related to the field of study covered by this CME activity.
Figure 1. B Lines (arrow) on Lung Ultrasound
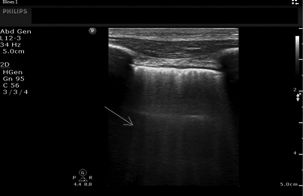
Image courtesy of Daniel Haase, MD, RDCS, RDMS, Assistant Professor of Emergency Medicine, Program in Trauma/Surgical Critical Care, Shock Trauma Critical Care Ultrasonography, University of Maryland School of Medicine and R Adams Cowley Shock Trauma Center.
Figure 2. B Lines (arrow) and Comet Tail on Lung Ultrasound
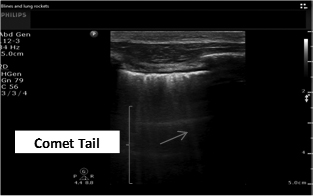
Image courtesy of Daniel Haase, MD, RDCS, RDMS, Assistant Professor of Emergency Medicine, Program in Trauma/Surgical Critical Care, Shock Trauma Critical Care Ultrasonography, University of Maryland School of Medicine and R Adams Cowley Shock Trauma Center.
Treatment for Systolic Heart Failure
The ideal outpatient treatment of systolic heart failure has three goals that should be followed simultaneously: control of risk factors for the development and progression of heart failure, treatment of heart failure symptoms, and education of patients.26
Control of risk factors involves treating hypertension, diabetes, and coronary artery disease, and eliminating the use of alcohol and tobacco. Symptoms of volume overload may be controlled with the use of diuretics and restricted dietary sodium. Diuretics, specifically furosemide, still continue to be the first-line drug in the treatment of heart failure and volume overload.8 While diuretics decrease venous return, reduce ventricular filling pressures, increase fluid loss, and decrease symptoms of pulmonary and systemic congestion and edema, no decrease in mortality has been observed with the use of these drugs.8
All patients with heart failure should be taking an angiotensin-converting enzyme (ACE) inhibitor or angiotensin-receptor blocker (ARB). ACE inhibitors work by decreasing both afterload and preload. In addition to decreasing systemic vascular resistance and myocardial oxygen consumption, ACE inhibitors also increase cardiac output. Significant evidence shows that ACE inhibitors reduce morbidity and mortality when used as a first-line therapy in patients who have heart failure with reduced ejection fraction.27
ACC/AHA guidelines recommend that ARBs be used in patients with heart failure if they develop an intolerance to ACE inhibitors. Some examples of ACE inhibitor side effects are cough, worsening kidney function, angioedema, or increasing blood potassium levels. Randomized, controlled trials have shown that ARBs can reduce morbidity and mortality, especially in patients who do not tolerate ACE inhibitors.27
Beta-blockers should be considered by physicians as an adjuvant to ACE-inhibitor therapy. Beta-blockers not only decrease the heart muscle’s oxygen requirement but also can help slow the progression of the disease.26 It is important to remember that beta-blockers are not a rescue drug for patients who have acute decompensated heart failure. Patients with heart failure and a history of prior myocardial infarction with or without asymptomatic low LV ejection fraction can benefit from a treatment regimen that includes both ACE inhibitors and beta-blockers.8,28 The reason behind this is that the combination of both medications can help patients reduce the incidence of new coronary events and heart failure.8
In patients with severe heart failure, spironolactone can be added to the treatment regimen. Digoxin comes into play when a patient continues to have symptoms despite already being treated with diuretics, ACE inhibitors, and beta-blockers.29 Digoxin also can be used with patients who have dyspnea at rest or a recent history of dyspnea at rest.29 It is important to state that digoxin therapy may reduce the probability of hospitalization but does not reduce mortality.30 One of the drawbacks of digoxin is that it must be monitored closely, with a target dosage level of 0.5 to 1.1 ng per mL.26
In patients with moderate to severe heart failure, a low left ventricular ejection fraction, and a QRS duration on the resting ECG of 120 ms or more, cardiac resynchronization therapy (CRT) has been shown to provide significant clinical improvement.8,31,32,33
The ultimate option for patients who have exhausted all medical management and/or device therapy is a heart transplant. Unfortunately, there are more patients with end-stage heart failure that are eligible for heart transplant than the current available number of donor hearts. In this circumstance, ventricular assist devices (VADs) can be used as a bridge pathway while the patient waits to make a decision, awaits recovery, or waits to be eligible.34 The main advantage of VADs and/or other mechanical circulatory support (MCS) is the improvement of organ perfusion and function, which in turns leads to better quality of life and increased survival. However, despite the benefits, VAD implantation is not without risk, and it is associated with the risk of serious complications, such as bleeding, infection, arrhythmias, blood clots, right ventricular failure, and cardiovascular events.
Treatment for Diastolic Heart Failure
The treatment of diastolic heart failure is less well defined than the treatment of systolic heart failure. When pharmacologic treatments of both diastolic and systolic heart failure are compared superficially, the impression is that they do not vary significantly. However, current recommendations for diastolic heart failure treatment are based on the disease pathophysiology, knowledge about other aspects of cardiovascular disease, data from small studies, and expert opinion. Unfortunately, to this author’s knowledge, none of the treatment recommendations have been validated by prospective randomized, controlled trials.
Currently, the best available treatment for diastolic decompensated heart failure is built around the concept of modifying the disease pathophysiology, such as relieving volume overload, decreasing heart rate, prolonging diastolic filling time, restoring atrial contraction, blocking the renin-angiotensin system, blocking aldosterone, controlling hypertension, and relieving ischemia.6
Diuretics and vasodilators often are used to reduce volume overload that presents as pulmonary congestion. However, aggressive diuresis can be dangerous; over-diuresis of the patient with decompensated heart failure will decrease the patient’s preload and stroke volume and impair renal function. Most patients with diastolic dysfunction are highly sensitive to volume changes and preload.
Beta-blockers are another option, with their ability to decrease heart rate, increase diastolic filling time, decrease oxygen consumption, lower blood pressure, and cause reversion of left ventricular hypertrophy. ACE inhibitors have some similarities to the benefits of betablockers, such as the ability to decrease blood pressure and cause regression of left ventricular hypertrophy. While ACE inhibitors are potentially beneficial, there have been few studies of patients with diastolic heart failure who use ACE inhibitors as part of their treatment regimen.35
It is theorized that calcium channel blockers may be more beneficial because these medications can also decrease oxygen demand (similar to beta-blockers) and dilate coronary arteries, in addition to decreasing blood pressure. However, there are not enough data available that measure patient-centric outcomes such as morbidity and mortality. Thus, calcium channel blockers should be used with caution in patients with both systolic and diastolic heart failure.
Management of Acute Heart Failure in the Emergency Department
The treatment for acute heart failure has not changed meaningfully in the last four decades. Medications such as loop diuretics and vasodilators still are used as the primary treatment for acute heart failure. However, one new addition to the management of heart failure is the use of noninvasive ventilation in cardiogenic pulmonary edema. Studies have shown that early use in these patients can prevent the need for endotracheal intubation.21,36
Initial treatment of acute heart failure is most influenced by the blood pressure. (See Figure 3.) About 50% or more of patients who arrive at the emergency department with acute heart failure have an elevated blood pressure, about 40% have a normal blood pressure, and only 5% have a low blood pressure.21,37,38 For intial treatment decisions, the three primary blood pressure divisions are: hypertensive (SBP > 140 mmHg), normotensive (SBP 139-90 mmHg), and hypotensive (SBP < 90 mmHg).21,39 It is important to stress that these cutoff points are intended as guidelines and are not absolute thresholds.21
Figure 3. General Outline for Treatment of Acute Heart Failure
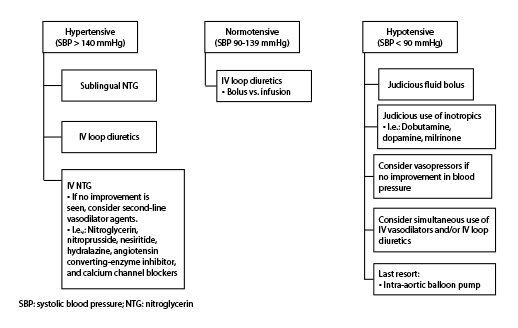
Patients who are hypertensive should be treated with the goal of vasodilation instead of concentrating on fluid removal. The symptoms of patients with hypertension are most likely related to volume re-distribution, as opposed to volume overload.21,40,41 Examples of medications used for initial vasodilation are nitroglycerin (most commonly used), nitroprusside, nesiritide, hydralazine, ACE inhibitors, and calcium channel blockers.21 (See Table 1.)
Table 1. Drugs Used to Treat Acute Heart Failure
Agent |
Mechanism |
Dosing |
Comments |
|
Nitroglycerin SL |
Direct vasodilator |
0.4 to 1.2 mg SL every 5 minutes |
Can be initiated while IV access is being obtained |
|
Nitroglycerin IV |
Direct vasodilator |
Start 10 mcg/min IV, increase by Max dose 200 mcg/min |
Typical dose is 100 mcg/min for resolution of dyspnea and control of blood pressure |
|
Nitroprusside |
Vasodilator |
Start 10 mcg/min IV, increase by 10 mcg every 10 minutes according to clinical response |
Typical dose is 200 to 300 mcg/min IV for control of dyspnea and blood pressure |
|
Nesiritide |
Recombinant form of BNP; vasodilator |
2 mcg/kg IV loading dose, followed by 0.01 mcg/kg per min IV infusion, increase by 0.005 mcg/kg per min increments every 3 hours to 0.03 mcg/kg per min |
Stop if hypotension develops |
|
Furosemide |
Loop diuretic |
40 mg IV bolus or 40 mg/h infusion over 1 hour |
May repeat in 1 hour |
|
Enalaprilat |
ACE inhibitor vasodilator |
0.625 to 1.25 mg IV over 5 minutes every 6 hours |
Consensus guidelines do not encourage early use in acute heart failure |
|
Hydralazine |
Direct vasodilator |
10 to 20 mg IV |
May repeat in 2 hours |
|
Dobutamine |
Beta-agonist inotropic |
Start infusion at 1 to 2 mcg/kg per min IV and increase according to clinical response Max dose 20 mcg/kg per min |
Fewer adverse side effects than milrinone |
|
Milrinone |
Type III phosphodiesterase inhibitor; vasodilator |
50 mcg/kg IV loading dose, followed by 0.375 mcg/kg per min IV infusion, increase according to clinical response Max dose 0.75 mcg/kg per min |
Consider in cases of acute heart failure associated with severe pulmonary hypertension |
Nitroglycerin has a rapid onset and clearance, in addition to being inexpensive. It can be used both sublingually and intravenously. Nitroglycerin can be used in higher dosages and still be both safe and effective.21,42,43 Furthermore, lower rates of intubation and myocardial infarction, as well as improvement in heart rate, respiratory rate, and oxygen saturation have been seen with high-dose nitrates when compared to lower doses.21,42,43
The normotensive patient is usually the one that has chronic heart failure with reduced ejection fraction that decompensates; however, the symptoms might not be as obvious as in the patient with hypertensive acute heart failure. Generally, these patients are maxed out on their outpatient oral diuretic therapy when they present with exacerbations. The goal for these patients should be to focus on decongestion through fluid removal via IV loop diuretics, which is the first-line treatment. There are debates regarding the most effective way to administer loop diuretics in acute heart failure exacerbation, either as a bolus or continuous infusion.21,44
Less common (< 5%) is the hypotensive patient. It is important to remember that patients without any previous history of heart failure who present with cardiogenic shock most likely have a life-threatening etiology, such as a myocardial infarction.21 In addition, it is important to assess the patient’s volume status. The most important goal in the treatment of this type of patient is to improve perfusion, not simply just to raise their blood pressure. Current guidelines recommend the use of inotropes such as dobutamine and milrinone only for those patients who truly require them, meaning patients who present with hypotension and acute heart failure. Inotropic therapy can help in the short term but is associated with worsening morbidity and mortality.21,45,46
Lifestyle Modification
Lifestyle modifications are important in the long-term management of patients with heart failure. And physicians should provide advice and encourage patients to modify their lifestyle in addition to prescribing appropriate medications. In some patients, lifestyle modifications and weight loss may be so effective as to enable reduction or even complete elimination of daily medication. This is why all patients with stable heart failure should be encouraged to begin and maintain a regular exercise program. Specifically, to patients classified at stage A of heart failure, according to the ACC/AHA Classification.5,8 In the 1970s, patients who complained of fatigue and/or shortness of breath related to exertion were not encouraged to exercise. This also applied to patients with heart failure or those who had suffered from a heart attack. However, in the late 1980s, it was understood that these patients suffered from deconditioning and muscle atrophy from the lack of activity.
Exercise in patients with heart failure has multiple benefits that include improvement in the muscle tolerance to exertion, neuro-hormonal changes that favor cardiac hemodynamics, and overall improvement of symptoms of heart failure.47 Moreover, it is proven that exercise not only improves symptoms but also decreases the number of admissions to the hospital and improves survival in patients with heart failure.48
Patients can control their fluid status and, consequently, their heart failure symptoms by simply restricting sodium intake to 2 g or less per day.26 There are no studies available regarding the effect in morbidity and mortality of dietary sodium restriction, either alone or in combination with pharmacological treatment. Moderate exercise (i.e., at 60% of maximum exercise capacity on a stationary bicycle for two to three hours per week) improves quality of life, decreases mortality, and decreases hospital readmissions for heart failure in patients with stable chronic heart failure.49
Conclusion
Heart failure is a major health issue and will continue to be so because of the growing elderly population. Diastolic heart failure can be diagnosed in a patient with clinical symptoms of heart failure and preserved systolic function, regardless of the absence of a laboratory and echocardiographic findings. The BNP measurement can increase the accuracy of diagnosis. Further randomized therapeutic studies are needed regarding the treatment of diastolic heart failure. Currenlty, physicians should focus on treating aspects of the pathophysiology that are modifiable, such as reducing volume overload, decreasing the heart rate, controlling hypertension, and relieving myocardial ischemia.20 Patients need to be educated on the importance of making good lifestyle choices, such as healthy eating and medication compliance.
REFERENCES
- Ahmed A, Allman RM, Fonarow GC, et al. Incident heart failure hospitalization and subsequent mortality in chronic heart failure: A propensity-matched study. J Card Fail 2008;14:211-218.
- Solomon SD, Dobson J, Pocock S, et al. Influence of nonfatal hospitalization for heart failure on subsequent mortality in patients with chronic heart failure. Circulation 2007;116:1482-1487.
- Hunt SA, Abraham WT, Chin MH, et al. ACC/AHA 2005 Guideline Update for the Diagnosis and Management of Chronic Heart Failure in the Adult: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Update the 2001 Guidelines for the Evaluation and Management of Heart Failure): Developed in collaboration with the American College of Chest Physicians and the International Society for Heart and Lung Transplantation: Endorsed by the Heart Rhythm Society. Circulation 2005;112:e154.
- American Heart Association. Heart Disease and Stroke Facts, 2006 Update. Dallas, Texas: AHA, 2006.
- Azad N, Lemay G. Management of chronic heart failure in the older population. J Geriatr Cardiol 2014;11:329-337.
- Angeja BG, Grossman W. Evaluation and management of diastolic heart failure. Circulation 2003;107:659-663.
- Roger VL. Epidemiology of heart failure. Circulation Research 2013;113:646-659.
- Aronow WS. Epidemiology, pathophysiology, prognosis, and treatment of systolic and diastolic heart failure. Cardiol Rev 2006;14:108-124.
- Chatterjee K. Primary diastolic heart failure. Am J Geriatric Cardiol 2002;11:178-189.
- Dauterman KW, Massie BM, Gheorghiade M. Heart failure associated with preserved systolic function: A common and costly clinical entity. Am Heart J 1998;135:S310-S319.
- Senni M, Tribouilloy CM, Rodeheffer RJ, et al. Congestive heart failure in the community: A study of all incident cases in Olmsted County, Minnesota, in 1991. Circulation 1998;98:2282-2289.
- Roger VL, Go AS, Lloyd-Jones DM, et al, for the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2011 update: A report from the American Heart Association. Circulation 2011;123:e18-e209.
- Dumitru I. Heart failure. Medscape. Jan. 11, 2016. Available at: http://emedicine.medscape.com/article/163062-overview#a5. Accessed May 1, 2017.
- O’Brien JF, Hunter CL. Chapter 81: Heart failure. In: Marx J, Hockberger R, Walls R, eds. Rosen’s Emergency Medicine – Concepts and Clinical Practice, 8th ed. Philadelphia, PA: Elsevier Saunders; 2013:1075.
- Chapter 53. Acute heart failure. In: Tintinalli JE, Stapczynski JS, Ma OJ, eds. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 7th ed. New York, NY: McGraw-Hill Education: 2011.
- Satpathy C, Mishra TK. Diagnosis and management of diastolic dysfunction and heart failure. Am Fam Physisican 2006;73:841-846.
- Mosterd A, Hoes AW. Clinical epidemiology of heart failure. Heart 2007;93:1137-1146.
- Morrison LK, Harrison A, Krishnaswamy P, et al. Utility of a rapid B-natriuretic peptide assay in differentiating congestive heart failure from lung disease in patients presenting with dyspnea. J Am Coll Cardiol 2002;39:202-209.
- Lubien E, DeMaria A, Krishnaswamy P, et al. Utility of B-natriuretic peptide in detecting diastolic dysfunction: Comparison with Doppler velocity recordings. Circulation 2002;105:595-601.
- Askoxylakis V, Thieke C, Pleger ST, et al. Long-term survival of cancer patients compared to heart failure and stroke: A systematic review. BMC Cancer 2010;10:105.
- Pang PS, Levy P, Shah SJ. Treatment of acute heart failure in the emergency department. Expert Rev Cardiovasc Ther 2013;11:
1195-1209. - Gheorghiade M, Filippatos G, De Luca L, Burnett J. Congestion in acute heart failure syndromes: An essential target of evaluation and treatment. Am J Med 2006;119(12Suppl1):S3-S10.
- Gheorghiade M, Pang PS. Acute heart failure syndromes. J Am Coll Cardiol 2009;53:557-573.
- Zehtabchi S, Brandler ES. Evidence-based emergency medicine/rational clinical examination abstract. Does this patient have congestive heart failure? Ann Emerg Med 2008;51:87-90.
- Aggarwal M, Gupta M, Vijan V, et al. Use of lung ultrasound for diagnosing acute heart failure in emergency department of southern India. J Clin Diagn Res 2016;10:TC05–TC08.
- McConaghy JR, Smith SR. Outpatient treatment of systolic heart failure. Am Fam Physician 2004;70:2157-2164.
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: A report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines. J Am Coll Cardiol 2013;62:e147-e239.
- Brophy JM, Joseph L, Rouleau JL. Beta-blockers in congestive heart failure. A Bayesian meta-analysis. Ann Intern Med 2001;134:550.
- Chavey WE 2nd, Blaum CS, Bleske BE, et al. Guideline for the management of heart failure caused by systolic dysfunction: Part II. Treatment. Am Fam Physician 2001;64:1045-1054.
- Ahmed A. Digoxin and reduction in mortality and hospitalization in geriatric heart failure: Importance of low doses and low serum concentrations. J Gerontol A Biol Sci Med Sci 2007;62:323-329.
- Aronow WS. CRT plus ICD in congestive heart failure. Use of cardiac resynchronization therapy and an implantable cardioverter–defibrillator in heart failure patients with abnormal left ventricular dysfunction. Geriatrics 2005;60:24-28.
- Young JB, Abraham WT, Smith AL, et al. Combined cardiac resynchronization and implantable cardioversion defibrillation in advanced chronic heart failure: The MIRACLE ICD Trial. JAMA 2003;289:2685-2694.
- Bristow MR, Saxon LA, Boehmer J, et al. Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure. N Engl J Med 2004;350:2140-2150.
- Szyguła-Jurkiewicz B, Szczurek W, Suliga K, et al. Mechanical circulatory support in heart failure. Kardiochir Torakochiururgia Pol 2016;13:130-134.
- Chavey WE 2nd, Blaum CS, Bleske BE, et al. Guideline for the management of heart failure caused by systolic dysfunction: Part II. Treatment. Am Fam Physician 2001;64:1045-1055.
- Vital FM, Saconato H, Ladeira MT, et al. Non-invasive positive pressure ventilation (CPAP or bilevel NPPV) for cardiogenic pulmonary edema. Cochrane Database Syst Rev 2008;(3):CD005351.
- Adams KF Jr, Fonarow GC, Emerman CL, et al. Characteristics and outcomes of patients hospitalized for heart failure in the United States: Rationale, design, and preliminary observations from the first 100,000 cases in the Acute Decompensated Heart Failure National Registry (ADHERE). Am Heart J 2005;149:209-216.
- Gheorghiade M, Abraham WT, Albert NM, et al. Systolic blood pressure at admission, clinical characteristics, and outcomes in patients hospitalized with acute heart failure. JAMA 2006;296:2217-2226.
- Collins S, Storrow AB, Kirk JD, et al. Beyond pulmonary edema: Diagnostic, risk stratification, and treatment challenges of acute heart failure management in the emergency department. Ann Emerg Med 2008;51:45-57.
- Cotter G, Felker GM, Adams KF, et al. The pathophysiology of acute heart failure — is it all about fluid accumulation? Am Heart J 2008;155:9-18.
- Cotter G, Metra M, Milo-Cotter O, et al. Fluid overload in acute heart failure — re-distribution and other mechanisms beyond fluid accumulation. Eur J Heart Fail 2008;10:165-169.
- Cotter G, Metzkor E, Kaluski E, et al. Randomised trial of high-dose isosorbide dinitrate plus low-dose furosemide versus high-dose furosemide plus low-dose isosorbide dinitrate in severe pulmonary oedema. Lancet 1998;351:389-393.
- Breidthardt T, Noveanu M, Potocki M, et al. Impact of a high-dose nitrate strategy on cardiac stress in acute heart failure: A pilot study.
J Intern Med 2010;267:322-330. - Felker GM, O’Connor CM, Braunwald E, et al.; Heart Failure Clinical Research Network Investigators. Loop diuretics in acute decompensated heart failure: Necessary? Evil? A necessary evil? Circ Heart Fail 2009;2:56-62.
- Hunt SA, Abraham WT, Chin MH, et al. 2009 Focused update incorporated into the ACC/AHA 2005 Guidelines for the diagnosis and management of heart failure in adults: A report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines developed in collaboration with the International Society for Heart and Lung Transplantation. J Am Coll Cardiol 2009;53:e1-e90.
- Felker GM, Benza RL, Chandler AB, et al. Heart failure etiology and response to milrinone in decompensated heart failure: Results from the OPTIME-CHF study. J Am Coll Cardiol 2003;41:997-1003.
- Working Group on Cardiac Rehabilitation & Exercice Physiology and Working Group on Heart Failure of the European Society of Cardiology. Recommendations for exercise training in chronic heart failure patients. Eur Heart J 2001;22:125-135.
- McKelvie RS, Teo KK, McCartney W, et al. Effects of exercise training in patients with congestive heart failure: A critical review. J Am Coll Cardiol 1995;25:789.
- Khand A, Gemmel I, Clark AL, Cleland JG. Is the prognosis of heart failure improving? J Am Coll Cardiol 2000;36:2284-2286.
Acute decompensated heart failure is a serious condition that presents in the emergency department and the intensive care unit. The causes of heart failure are multifactorial, making it, at times, difficult to diagnose and treat.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
