Stroke: Rehabilitation and Recovery
July 1, 2017
Reprints
AUTHOR
Joel Stein, MD, Simon Baruch Professor and Chair, Department of Rehabilitation and Regenerative Medicine, Columbia University College of Physicians and Surgeons; Professor and Chair, Department of Rehabilitation Medicine, Weill Cornell Medical College; and Physiatrist-in-Chief, New York-Presbyterian Hospital, New York
PEER REVIEWER
Glen D. Solomon, MD, Professor and Chair, Department of Internal Medicine, Professor and Interim Chair, Department of Neurology, Boonshoft School of Medicine, Wright State University, Dayton, OH
EXECUTIVE SUMMARY
- With millions of stroke survivors in the United States, primary care physicians often are involved in providing post-stroke rehabilitation care, including recognizing common treatable and reversible conditions such as depression.
- Key components of rehabilitation include assessment, goal setting, treatment of functional and psychosocial impairments, and prevention of complications.
- A coordinated, multidisciplinary rehabilitation evaluation and treatment approach during the early post-stroke period is associated with improved clinical outcomes.
- Muscle weakness and impaired motor control are managed with various modalities such as repetitive task training, constraint-induced movement therapy, mirror therapy, and functional electrical stimulation.
- Spasticity is addressed primarily through stretching and range of motion exercises, bracing, and botulinum toxin injections. Sedating medications should be avoided if possible.
- Post-stroke depression and fatigue are common and underdiagnosed. Standard antidepressant treatment including selective serotonin reuptake inhibitors, such as fluoxetine, have been found effective.
Rehabilitation is a critical component of stroke treatment, as most stroke survivors are left with significant neurological impairments and other sequelae, such as spasticity and pain. Approximately 40% of stroke patients are left with moderate functional impairment and 15%-30% with severe disability.1 Stroke rehabilitation aims to reverse these impairments to the extent possible, maximize functionality through the use of compensatory approaches, prevent complications, and manage comorbidities. This article reviews the basic principles of rehabilitation, current practices, and evidence supporting various aspects of stroke rehabilitation.
Role of Primary Care Physician
With millions of stroke survivors in the United States, primary care physicians often are faced with providing care to these individuals and, thus, need to be able to identify common post-stroke rehabilitation issues. Post-stroke depression can occur at any time after stroke, and it is important that primary care physicians recognize this as a treatable and reversible condition, rather than a “natural” consequence of disability after stroke. Monitoring range of motion to identify developing contracture and/or spasticity is important so that referral can be made to a rehabilitation physician if these occur. Lastly, addressing the stroke survivor’s mobility, ability to perform activities of daily living (ADL), and any deterioration in functional status are critical to making timely referrals to rehabilitation services when needed. When these issues are identified early, the primary care physician can prevent the progression of debilitating consequences by initiating treatment or referring to appropriate specialists for further evaluation and management.
Rehabilitation
Functional improvement in rehabilitation is accomplished through a combination of neurological recovery and adoption of compensatory techniques and equipment. Improved functional independence may be attained by reducing impairment directly (i.e., via neural recovery using cerebral plasticity to overcome neuronal loss) or through compensation for impairment by using remaining physical and cognitive abilities and strategies. Both recovery and compensation are crucial concepts in rehabilitation and key therapeutic approaches; interventions must be balanced to address patient goals and efficiently deploy the available rehabilitation resources.2
Over the last several decades, a significant amount of research has been conducted to further elucidate how best to provide comprehensive stroke rehabilitation. Currently, it is comprised of several key components, including assessment, goal setting, treatment of functional and psychosocial impairments, and prevention of complications. Patients are reassessed at regular intervals to evaluate progress, and treatment plans are adjusted accordingly. Ideally, rehabilitation begins immediately following stroke and often becomes a long-term element in the lives of these patients. Assessment and treatment should begin in the setting of acute hospitalization, and depending on the needs of the individual, continued rehabilitation may transition to the appropriate inpatient or outpatient setting.
National and international guidelines have been developed to provide resources for healthcare providers with the latest evidence-based practices, such as the American Heart Association (AHA) Guidelines published in 2016.3 The AHA grading system for level of evidence and strength of recommendation are referenced in this article where applicable.3 (See Table 1.) Adherence to stroke rehabilitation guidelines is associated with improved patient functional outcomes.4
Table 1. American Heart Association Evidence-based Scoring System |
Classification of Recommendations
|
Level of Evidence
|
In an effort by The Joint Commission and the AHA, certification has been developed using current guidelines and established standards of care to identify both Primary Stroke Centers and Comprehensive Stroke Centers. The rehabilitation requirements for Primary Stroke Center certification include the ability to assess for rehabilitation needs and refer for appropriate post-acute care. Comprehensive Stroke Centers also must have a rehabilitation service led by a physician with expertise in neurorehabilitation, and the service must include therapists, nurses, and social workers with an expertise in addressing the rehabilitation needs of stroke patients.5,6 Complete guidelines can be found on The Joint Commission’s website.5
Services and Settings
A coordinated, multidisciplinary rehabilitation evaluation and treatment during the early post-stroke period is associated with improved clinical outcomes (AHA Class 1, Evidence Level A).3 The multidisciplinary team typically consists of physicians, physical therapists, occupational therapists, speech therapists, recreational therapists, nurses, social workers, and psychologists who work closely with patients and their family members or caregivers to achieve rehabilitation goals.
Stroke patients should be assessed for rehabilitation needs, and rehabilitation should commence during the acute hospitalization. Upon discharge, rehabilitation (if required) may continue in either the inpatient, home, or outpatient setting. For patients who require inpatient rehabilitation prior to returning home, the two main options are acute inpatient rehabilitation (often referred to as Inpatient Rehabilitation Facilities, or IRFs) or subacute inpatient rehabilitation (also known as skilled nursing facilities). IRFs typically provide three or more hours of therapy per day, with subacute rehabilitation facilities providing a lesser (and more variable) amount. AHA guidelines advise that patients who qualify for and have access to IRF rehabilitation care after stroke receive this in preference to skilled nursing facility level rehabilitation (AHA Class I, Evidence Level B). Many patients cannot benefit from IRF care because of preexisting dementia or other disability, or are unable to tolerate rehabilitation of this intensity. For these patients, a skilled nursing facility may be the most appropriate choice for rehabilitation after stroke. For patients who can be safely discharged home, rehabilitation may continue either through homecare services or in an outpatient rehabilitation program. The choice of rehabilitation setting depends on the patient’s care needs, the team’s assessment, goals of care, available resources, and patient’s ability and willingness to participate in and tolerate a therapeutic exercise program.
Timing and Intensity
Initiation of rehabilitation during the acute hospitalization should occur once the patient is medically stable and safe to participate in therapy. This early rehabilitation helps prevent complications, such as venous thrombosis and contractures, and initiates the assessment process for post-acute services. Aggressive early mobilization within the first 24 hours after stroke was found to be associated with worse outcomes, and should be avoided (AHA Class III, Level of Evidence A), but more conventional mobilization, such as having a patient get out of bed and sit in a chair, remains appropriate.7
There is wide acceptance of the hypothesis that a higher intensity of therapy is more beneficial than a lower intensity;2,3,8-10 however, results of a recent clinical trial failed to confirm this hypothesis for an upper limb exercise intervention.11 Overall, there is insufficient evidence to make specific recommendations regarding the optimal level of intensity of rehabilitation services. Determining intensity level as well as duration of therapy is often greatly affected by the mental and physical tolerance of the patient to participate in therapy, and therefore, programs must be individualized based on the multidisciplinary team’s assessment and plan of care.
Motor Rehabilitation
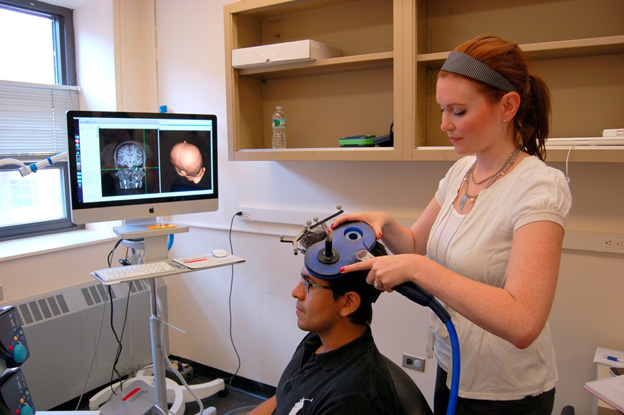
Many stroke patients suffer from muscle weakness and impaired motor control, with resultant functional deficits. Multiple therapeutic methods have been developed to aid stroke patients with improving motor function. These methods typically involve repeated practice of movements as a foundation, and vary greatly from simple task-specific training to more complex methods, using virtual reality or advanced robotics. Additional therapeutic options have emerged recently and are still currently being studied, including pharmacotherapy to facilitate plasticity and the use of non-invasive brain stimulation. (See Figure 1.) Even if ultimately adopted, these novel therapies are likely to serve an adjunctive role to conventional physical and occupational therapy, rather than obviate the need for traditional forms of rehabilitation.
Figure 1. Transcranial Magnetic Stimulation |
|
This technique is used both as a tool for investigating brain physiology during stroke recovery, and also as a potential therapy to enhance motor recovery.
|
|
Photo courtesy of Dr. Joel Stein. |
Conventional stroke rehabilitation includes repetitive task training; however, effects of specific interventions may generalize poorly to related tasks. Emphasis should be placed on task- or context-specific training, which has been found to be effective in improving upper and lower extremity motor function.12
Constraint-induced movement therapy (CIMT) is another type of repetitive task training, involving forced use of the affected limb by “constraining” use of the non-paretic limb. A randomized trial found benefit in upper limb use after CIMT training, and it is considered a “reasonable” treatment for suitable stroke survivors (AHA Class IIa, Level of Evidence A).13 The durability of this benefit is unknown, however, and additional research is required to define optimal dosing and timing of CIMT.14,15,16
In mirror therapy, a mirror is placed in the patient’s midsagittal plane while performing bilateral exercises, providing the patient with the visual illusion of successfully moving the affected limb. In a Cochrane review, mirror therapy was shown to improve upper extremity motor function, ADLs, and pain; however, data are limited.17
More than half of stroke patients are unable to walk independently during the acute phase of recovery and may benefit from intensive gait training, which can improve gait function significantly.18,19 Gait training may include assisted ambulation, treadmill training, and body-weight-supported treadmill training (BWSTT).20 A large, multicenter, randomized trial found that BWSTT compared to a home exercise program with physical therapy had no incremental benefit for BWSTT on walking speed, motor recovery, balance, and functional status, with patients participating in the home exercise program experiencing fewer falls.21
Functional electrical stimulation (FES) involves stimulating specific muscle groups to cause a muscle contraction. FES may lead to a short-term increase in motor strength and reduction in impairment severity, but without evidence of improved function.22,23 This method may be considered for use in ankle, knee, or wrist motor impairment, shoulder subluxation, and for gait training. Several FES devices have been designed to aid in ambulation for patients with foot drop, including the Bioness L300 (Bioness Inc., Valencia, CA) and the WalkAide (Innovative Neurotronics Inc., Austin, TX). Kluding et al compared use of the FES (Bioness L300) with a traditional ankle-foot orthosis (AFO) and found that use of both FES and AFO with physical therapy training significantly improved gait speed and functional outcomes, with no significant difference between groups.24
Electrical signals also can be detected in the muscle using surface electromyographic electrodes and translated into visual or auditory signals to provide biofeedback during therapeutic exercises.25,26 The benefits of this therapy remain unproven.
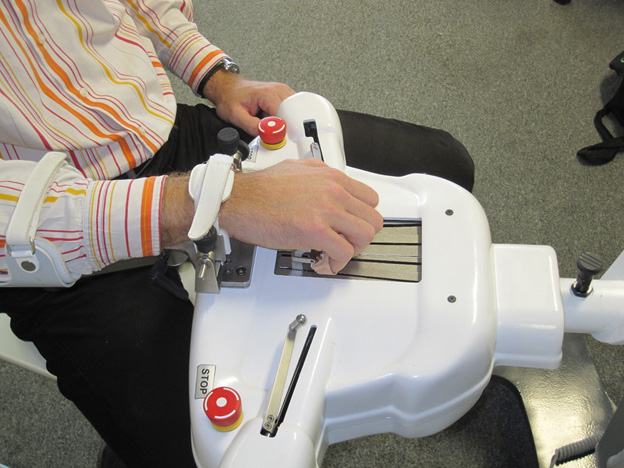
Robots have been used in stroke rehabilitation to provide reliable, reproducible, high-intensity exercise therapy.27,28,29 (See Figure 2.) A Cochrane review found that patients who received robot-assisted arm training were more likely to improve arm function and ADLs but not muscle strength,30 and AHA guidelines indicate that this treatment is reasonable to consider (AHA Class IIa, Level of Evidence A). Other systems, including computer games and virtual reality, have been studied as stand-alone therapies to provide guidance and a motivating environment as well as in combination with robot-assisted therapy.31,32 Although some benefit has been demonstrated, further research is needed to optimize these treatments and determine their clinical utility and cost-effectiveness.
Figure 2. Hand Robotic Device |
|
The Amadeo robot (Tyromotion Inc., Graz, Austria) is one of a growing number of robotic devices for providing exercise therapy for the upper limb after stroke. This device provides finger flexion and extension training. |
|
Photo courtesy of Dr. Joel Stein. |
Multiple drugs have been studied as potential facilitators of motor recovery. The Fluoxetine for Motor Recovery in Acute Ischemic Stroke (FLAME) study found that patients with moderate-to-severe motor deficits experienced enhanced motor recovery with combined fluoxetine and physical therapy after three months.33 However, larger, more definitive studies are needed before fluoxetine can be recommended routinely for this indication.
Non-invasive cortical stimulation currently is being studied to assess its effect on motor recovery, but it is not yet used routinely in clinical practice.34,35 The two main types of non-invasive cortical stimulation are repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS).36 In rTMS, electric current is induced in the underlying cortex using pulses of high-intensity magnetic fields. Depending on the stimulation parameters, rTMS can either augment or inhibit cortical excitability. In tDCS, surface electrodes on the scalp deliver direct current that similarly can modulate excitatory or inhibitory effects on the underlying cortex. A recent multicenter, randomized, controlled trial failed to demonstrate benefits for inhibitory rTMS delivered to the undamaged hemisphere combined with exercise therapy when compared with exercise therapy alone.37
Approximately one-third of stroke patients develop muscle spasticity, which may impair motor function and quality of life further. However, treatment of spasticity is not necessary unless it causes impaired function, pain, deforming contractures, or limits skin hygiene. Treatment options include stretching and range of motion exercises, bracing, oral medications, and intramuscular injections. The most frequently used oral medications include baclofen, tizanidine, benzodiazepines, and dantrolene. If possible, use of benzodiazepines should be avoided during the stroke recovery period because of possible deleterious effects on recovery.38 Moreover, many stroke survivors do not tolerate these medications well, with sedation a common complaint. Intramuscular injection is performed most commonly with botulinum toxins (AHA Class 1, Level of Evidence A) and less commonly with phenol or ethanol, and should be considered for disabling or painful spasticity, particularly if localized. Botulinum toxin injections are generally safe and well-tolerated, but with a duration of action of only three months, and therefore may need to repeated on a regular basis. Occasionally, patients may require more invasive treatments, such as intrathecal baclofen or surgical correction of contractures.
Assistive Devices
Stroke survivors often benefit from assistive devices as a compensatory approach to persistent weakness and difficulty with mobility. Many hemiparetic patients have absent or insufficient ankle dorsiflexion (“drop foot”) that interferes with gait. A polypropylene or carbon fiber AFO can improve gait efficiency and reduce the risk of falls. Despite the benefits of an AFO for patients who have drop foot, some patients are reluctant to use these because of discomfort or desire to manage without a medical device. If symptoms of drop foot are relatively mild, some individuals benefit from the use of a work boot or other supportive high-top shoe that provides additional support as a partial substitute for a brace. Another option may be the use of an electrical stimulation device that stimulates the peroneal (fibular) nerve, although these are more expensive than AFOs.
Many patients benefit from the use of a cane to help with walking after a stroke. Canes with multiple prongs (e.g., a four-pronged cane) provide a larger base of support and more stability, but are bulkier and more cumbersome than a conventional single-pronged cane. Many stroke survivors begin with more supportive devices and then transition to simpler devices over time.
While most stroke survivors can resume ambulation after a stroke, longer distance ambulation may be difficult for some, and wheelchairs can be useful for longer trips for this reason. For non-ambulatory stroke patients with well-preserved cognition and visual-spatial skills, a motorized wheelchair can be useful to maximize independent mobility.
Resting wrist/hand splints and ankle splints commonly are employed in the hospital or post-acute setting, despite limited evidence supporting their utility in preventing contracture or other complications. While these may be useful on a chronic basis for patients with moderate or severe spasticity, a careful review of the continued need for these devices in the home setting is appropriate. Stroke survivors with limited volitional movement of the limb(s) should be instructed in performing self-range of motion exercises themselves. Caregivers also should be educated about these exercises, as appropriate.
Pain
Pain can occur after stroke from a variety of etiologies. The most common pain syndrome after stroke is hemiplegic shoulder pain. Although this pain commonly is associated with shoulder subluxation (due to flaccidity of the shoulder girdle muscles that normally stabilize the shoulder), the precise pathophysiology of post-stroke shoulder pain remains uncertain. The use of shoulder slings or arm supports to reduce subluxation may be useful in some cases, but is not always effective in treating or preventing shoulder pain. The use of electrical stimulation is of uncertain value, with both surface and percutaneous stimulating systems available.
Another relatively common pain post-stroke is central pain. While classically seen after thalamic stroke (generally in conjunction with an area of sensory loss), central pain can be seen in some individuals with strokes in other locations. Medical therapy using anticonvulsant medications is helpful in some but not all individuals; trials of several medications may be necessary until an effective treatment is found.
Aerobic Exercise and Secondary Stroke Prevention
Stroke survivors may have difficulty in performing exercise, yet it remains an important aspect of overall fitness and of secondary prevention post-stroke. Stroke survivors should be encouraged to undertake regular exercise for this purpose — most commonly walking, swimming, or use of a stationary bicycle. More broadly, other lifestyle modifications, including smoking cessation, proper diet, weight loss, and medication compliance, are important aspects of post-stroke care to improve overall wellness and prevent another stroke.
Speech and Language Rehabilitation
Disorders of communication, which may occur in as many as 40% of stroke patients, most commonly include aphasia, dysarthria, or apraxia. Aphasia results from damage to the regions of the central nervous system that affect language reception, expression, or both. Apraxia is caused by impairment in the patient’s ability to plan and organize commands for muscle movement. Dysarthria results from neuromuscular damage, causing impairment in muscle movement and subsequent alterations in speech production. Speech and language rehabilitation involves careful assessment of the patient’s abilities and deficits, as many patients suffer from a complex combination of these communication disorders. Therapy includes working on the affected system (e.g., phonation, articulation, prosody, etc.) along with use of compensatory strategies and potential use of augmentative or alternative communication devices. Patient and caregiver education is a critical component of speech therapy. Speech therapy can aid in recovery and prevent ineffective or inappropriate compensatory behaviors (AHA Class 1, Level of Evidence A).39 Several novel therapeutic approaches are currently being investigated and include constraint-induced aphasia therapy, where use of compensatory strategies (such as communicating using gestures) is not permitted;40 use of non-invasive brain stimulation; and use of medications, such as memantine, an NMDA receptor antagonist.
Dysphagia
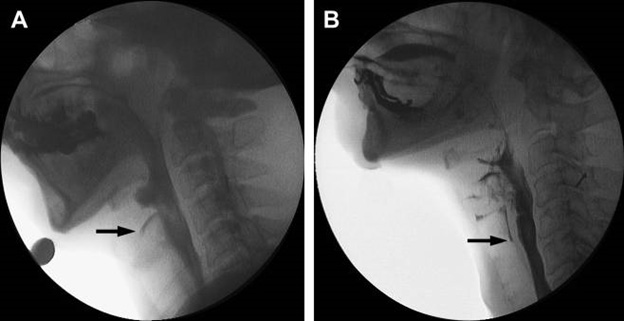
Dysphagia is seen in approximately 45% of hospitalized stroke patients; therefore, early assessment is critically important because of the risk of aspiration. Bedside dysphagia screening assessment should be performed before initiating oral intake for all stroke patients (AHA Class 1, Level of Evidence B).3 If the bedside screening assessment is abnormal, then a complete bedside swallow evaluation generally is performed (AHA Class IIa, Level of Evidence C). Additional measures, including modified barium swallow studies (see Figure 3) and flexible endoscopic evaluation of swallowing and sensory testing, also may be necessary in evaluating patients at high risk for aspiration (AHA Class IIa, Level of Evidence B). Treatment of dysphagia includes use of dietary texture modification, compensatory strategies, exercises, and postural advice.41,42,43 In some cases, a feeding tube may be needed temporarily or long-term to support a patient’s nutrition. Malnutrition affects approximately 30% of stroke patients during the first week of hospitalization, and nutritional status should be monitored carefully and appropriate supplementation provided when necessary.
Figure 3. Modified Barium Swallow Study |
|
Static images from modified barium swallow studies show laryngeal penetration (A) and aspiration of barium into the trachea (B)
|
|
Reprinted with permission from Elsevier. Matsuo K, Palmer JB. Anatomy and physiology of feeding and swallowing: Normal and abnormal. Phys Med Rehabil Clin N Am 2008;19:691-707, vii. |
Cognitive Rehabilitation
Cognitive deficits are very common after stroke, may be quite complex, and may affect multiple aspects of cognition such as attention, executive function, memory, and insight. Screening for alterations in cognition is important, as sustained cognitive deficits may result in poor outcomes. These deficits may affect a patient’s ability to perform therapeutic exercise, and may affect one’s ability to recognize one’s own impairments, known as anosognosia. When possible, sedating medications that may exacerbate cognitive difficulties, such as benzodiazepines, neuroleptics, barbiturates, and anticonvulsants, should be avoided. Severe cognitive impairments may pose significant obstacles in community and home reintegration.
Spatial neglect and visual deficits may result from damage to the non-dominant parietal lobe or to various areas of the visual system. Patients should be screened for visual deficits and spatial neglect. Use of prisms, education, functional training, compensatory strategies, and sensory stimulation may be employed in the treatment of spatial neglect or visual deficits.44,45,46
Psychological
Psychological disorders after stroke, including depression and fatigue, are very common and under-diagnosed, and can have a negative effect on rehabilitation. Screening for depression is advisable given its high prevalence in this population (AHA Class 1, Level of Evidence B). It is important to recognize that while some sadness is expected shortly after a stroke occurs as a normal grief reaction, persistent symptoms of depression represent a treatable and disabling condition, and are not a “normal” consequence of an acquired disability due to stroke. Signs or symptoms of depression frequently require treatment. Standard antidepressant therapeutic modalities, including psychotherapy and psychopharmacologic treatment (e.g., selective serotonin reuptake inhibitors [SSRIs], such as fluoxetine), have been found to be effective for post-stroke depression.47
Emotional lability, also known as pseudobulbar affect, is common after stroke, with minor stimuli (e.g., a television commercial) causing patients to laugh or cry due to inability to modulate their emotional response. Emotional lability is distinguished from depression by its highly transient nature, with normal mood in between episodes of lability. In many cases, reassurance and education is sufficient, but some individuals are distressed by this symptom and pharmacological therapy may be helpful. SSRIs or a combination of quinidine and dextromethorphan (marketed as Nuedexta®) generally are effective.
Fatigue is a common after effect of stroke, and can contribute to disability. While the mechanisms are not yet elucidated, treatment focuses on symptoms, with primarily behavioral (e.g., obtaining adequate sleep, planning for rest or nap periods), or occasionally pharmacologic (e.g., modafinil or stimulant medications) interventions.
Sexuality
Sexual dysfunction is common after stroke, with multiple potential contributors. Psychosocial considerations include anxiety about the risk of precipitating another stroke, and alterations to traditional roles as one partner may be disabled from a stroke and the other is involved in providing personal care to the stroke survivor. Reassurance regarding the safety of resuming sexual activity is important, and counselling may be useful to address the issues related to disability and caregiving.
Motor impairments can pose a barrier to resumption of sexual activity, but most couples can identify strategies to overcome any physical limitations without the need for any detailed clinician guidance.
Medical considerations include the frequency of comorbid conditions that may adversely affect sexual function, such as diabetes, hypertension, and depression. Moreover, the medications used to treat these conditions, such as SSRIs for depression and many anti-hypertensive medications, may iatrogenically contribute to altered sexual functioning as well. Men with erectile dysfunction may be candidates for phosphodiesterase 5 inhibitors, subject to the usual contraindications such as avoiding use while on nitrate medications. There are little data on the use of flibanserin (Addyi®) by women with stroke.
Return to Driving
Return to driving is an important concern of many stroke survivors once they return to the community. Driving ability may be adversely affected by visual field defects, seizure disorders, visual-spatial deficits, and communication difficulties (e.g., inability to read road signs). Perhaps surprisingly, hemiparesis is less often a limiting factor, and in the absence of other impairments, many hemiparetic individuals can resume driving with minor car modifications. Use of a car with an automatic transmission generally is required, and for individuals with right hemiparesis, a modification may be needed to shift the control pedals to the left. A “spinner knob” is useful to assist with steering for individuals who lack upper limb motor abilities.
The process of evaluating driving ability after stroke often begins with a determination from a physician that there are no overt medical contraindications or neurological deficits that would preclude driving, such as a poorly controlled seizure disorder or hemispatial neglect. Referral to an occupational therapist with expertise in pre-driving assessment often is useful to evaluate vision, perceptual abilities, and reaction time in a more detailed way. For individuals who perform well on this pre-driving assessment, referral to a driving school with expertise in working with individuals with disabilities is advisable for an on-the-road assessment, any vehicle modifications, and training. Finally, once ready, a driving test administered by the state motor vehicles agency is necessary prior to return to driving in the community.
State laws vary regarding physician obligations to report impaired drivers directly to the driving licensure agency, and local requirements should be reviewed by physicians caring for individuals who may seek to continue driving despite advice to the contrary from their physician.
Vocational Rehabilitation
While many stroke survivors are already retired prior to a stroke due to their age or other medical issues, there is a substantial minority of stroke survivors who seek to return to work after their stroke. Generally speaking, it is often easiest for stroke survivors to return to their prior employment, where their employers may be more motivated to support a valued employee who may require accommodations, and where discrimination against individuals with physical disabilities is likely to be countered by personal relationships. While there are legal protections against workplace discrimination against disabled individuals (most notably the Americans with Disabilities Act), the unfortunate reality remains that discrimination in the hiring process remains difficult to detect and likely widespread, and therefore it may be difficult for someone with a visible disability (e.g., hemiparesis) after stroke to obtain a new job.
Individuals with “white collar” (professional) jobs often have an easier time with return to work, whereas jobs that involve manual labor may be particularly problematic. Stroke survivors seeking to return to work should give themselves a sufficient amount of time to maximize their recovery, and consider neuropsychological assessment prior to attempting to return to work if they have any cognitive sequelae of the stroke. Vocational rehabilitation agencies exist in each state and can be helpful in return to work efforts.
Caregiver Stress and Other Psychosocial Issues
Rehabilitation not only should assess and treat motor, sensory, and cognitive deficits, but also should address issues of social support and caregiver stress early and at regular intervals. Living with disability after stroke is a lifelong challenge and adequate support from family and caregivers is critical to successful outcomes.
CONCLUSION
Stroke is one of the most common causes of acquired disability worldwide. The field of stroke rehabilitation is ever evolving to better meet the needs of stroke survivors. Novel techniques and technology are being developed and studied with the ultimate goal of improving patient function and quality of life.
REFERENCES
- Bates B, Choi JY, Duncan PW, et al. Veterans Affairs/Department of Defense Clinical Practice Guideline for the Management of Adult Stroke Rehabilitation Care: Executive summary. Stroke 2005;36:2049-2056.
- Kwakkel G, Kollen B, Lindeman E. Understanding the pattern of functional recovery after stroke: Facts and theories. Restor Neurol Neurosci 2004;22:281-299.
- Winstein CJ, Stein J, Arena R, et al. Guidelines for adult stroke rehabilitation and recovery. Stroke 2016;47:e98-169.
- Duncan PW, Horner RD, Reker DM, et al. Adherence to postacute rehabilitation guidelines is associated with functional recovery in stroke. Stroke 2002;33:167-177.
- The Joint Commission. Advanced Disease-Specific Care Certification Requirements for Comprehensive Stroke Center (CSC). 2013. Available at: http://www.jointcommission.org/assets/1/18/DSC_CSC_Chap.pdf. Accessed Sept. 10, 2013.
- Alberts MJ, Latchaw RE, Selman WR, et al. Recommendations for comprehensive stroke centers: A consensus statement from the Brain Attack Coalition. Stroke 2005;36:1597-1616.
- AVERT Trial Collaboration group. Efficacy and safety of very early mobilisation within 24 h of stroke onset (AVERT): A randomised controlled trial. Lancet 2015;386:46-55.
- European Stroke Organisation (ESO) Executive Committee; ESO Writing Committee. Guidelines for management of ischaemic stroke and transient ischaemic attack 2008. Cerebrovasc Dis 2008;25:457-507.
- French B, Thomas LH, Coupe J, et al. Repetitive task training for improving functional ability after stroke. Cochrane Database Syst Rev 2016;(4):CD006073.
- Langhorne P, Wagenaar R, Partridge C. Physiotherapy after stroke: More is better? Physiother Res Int 1996;1:75-88.
- Winstein CJ, Wolf SL, Dromerick AW, et al. Effect of a task-oriented rehabilitation program on upper extremity recovery following motor stroke: The ICARE randomized clinical trial. JAMA 2016;315:571-581.
- Arya KN, Verma R, Garg RK, et al. Meaningful task-specific training (MTST) for stroke rehabilitation: A randomized controlled trial. Top Stroke Rehabil 2012;19:193-211.
- Wolf SL, Winstein CJ, Miller JP, et al. Effect of constraint-induced movement therapy on upper extremity function 3 to 9 months after stroke: The EXCITE randomized clinical trial. JAMA 2006;296:2095-2104.
- Sirtori V, Corbetta D, Moja L, Gatti R. Constraint-induced movement therapy for upper extremities in patients with stroke. Cochrane Database Syst Rev 2009;(4):CD004433.
- Nijland R, Kwakkel G, Bakers J, van Wegen E. Constraint-induced movement therapy for the upper paretic limb in acute or sub-acute stroke: A systematic review. Int J Stroke 2011;6:425-433.
- Peurala SH, Kantanen MP, Sjogren T, et al. Effectiveness of constraint-induced movement therapy on activity and participation after stroke: A systematic review and meta-analysis of randomized controlled trials. Clin Rehabil 2012;26:209-223.
- Thieme H, Mehrholz J, Pohl M, et al. Mirror therapy for improving motor function after stroke. Stroke 2012;44:e1-e2.
- Visintin M, Barbeau H, Korner-Bitensky N, Mayo NE. A new approach to retrain gait in stroke patients through body weight support and treadmill stimulation. Stroke 1998;29:1122-1128.
- Peurala SH, Airaksinen O, Huuskonen P, et al. Effects of intensive therapy using gait trainer or floor walking exercises early after stroke. J Rehabil Med 2009;41:166-173.
- States RA, Salem Y, Pappas E. Overground gait training for individuals with chronic stroke: A Cochrane systematic review. J Neurol Phys Ther 2009;33:179-186.
- Duncan PW, Sullivan KJ, Behrman AL, et al. Body-weight-supported treadmill rehabilitation after stroke. N Engl J Med 2011;364:2026-2036.
- Glanz M, Klawansky S, Stason W, et al. Functional electrostimulation in poststroke rehabilitation: A meta-analysis of the randomized controlled trials. Arch Phys Med Rehabil 1996;77:549-553.
- Price CI, Pandyan AD. Electrical stimulation for preventing and treating post-stroke shoulder pain: A systematic Cochrane review. Clin Rehabil 2001;15:5-19.
- Kluding PM, Dunning K, O’Dell MW, et al. Foot drop stimulation versus ankle foot orthosis after stroke: 30-week outcomes. Stroke 2013;44:1660-1669.
- Woodford H, Price C. EMG biofeedback for the recovery of motor function after stroke. Cochrane Database Syst Rev 2007;(2):CD004585.
- Zijlstra A, Mancini M, Chiari L, Zijlstra W. Biofeedback for training balance and mobility tasks in older populations: A systematic review. J Neuroeng Rehabil 2010;7:58.
- Stein J. Motor recovery strategies after stroke. Top Stroke Rehabil 2004;11:12-22.
- Stein J. Robotics in rehabilitation: Technology as destiny. Am J Phys Med Rehabil 2012;91(11 Suppl 3):S199-S203.
- Klamroth-Marganska V, Blanco J, Campen K, et al. Three-dimensional, task-specific robot therapy of the arm after stroke: A multicentre, parallel-group randomised trial. Lancet Neurol 2014;13:159-166.
- Mehrholz J, Pohl M, Platz T, et al. Electromechanical and robot-assisted arm training for improving generic activities of daily living, arm function, and arm muscle strength after stroke. Cochrane Database Syst Rev 2012;6:CD006876.
- Saposnik G, Levin M; Outcome Research Canada (SORCan) Working Group. Virtual reality in stroke rehabilitation: A meta-analysis and implications for clinicians. Stroke 2011;42:1380-1386.
- Saposnik G, Cohen LG, Mamdani M, et al. Efficacy and safety of non-immersive virtual reality exercising in stroke rehabilitation (EVREST): A randomised, multicentre, single-blind, controlled trial. Lancet Neurol 2016;15:1019-1027.
- Chollet F, Tardy J, Albucher JF, et al. Fluoxetine for motor recovery after acute ischaemic stroke (FLAME): A randomised placebo-controlled trial. Lancet Neurol 2011;10:123-130.
- Sasaki N, Mizutani S, Kakuda W, Abo M. Comparison of the effects of high- and low-frequency repetitive transcranial magnetic stimulation on upper limb hemiparesis in the early phase of stroke. J Stroke Cerebrovasc Dis 2013;22:413-418.
- Lindenberg R, Renga V, Zhu LL, et al. Bihemispheric brain stimulation facilitates motor recovery in chronic stroke patients. Neurology 2010;75:2176-2184.
- Harvey RL, Liu C, Edwards D, et al. Navigated brain stimulation for upper limb recovery after stroke: A randomized sham controlled clinical trial of low frequency r-TMS to non-injured hemisphere combined with upper limb rehabilitation. Abstract presented at International Stroke Conference, Houston 2017. Stroke 2017;48:A104.
- Nitsche MA, Cohen LG, Wassermann EM, et al. Transcranial direct current stimulation: State of the art 2008. Brain Stimul 2008;1:206-223.
- Goldstein LB. Potential effects of common drugs on stroke recovery. Arch Neurol 1998;55:454-456.
- Robey RR. A meta-analysis of clinical outcomes in the treatment of aphasia. J Speech Lang Hear Res 1998;41:172-187.
- Berthier ML, Green C, Lara JP, et al. Memantine and constraint-induced aphasia therapy in chronic poststroke aphasia. Ann Neurol 2009;65:577-585.
- Bath PM, Bath FJ, Smithard DG. Interventions for dysphagia in acute stroke. Cochrane Database Syst Rev 2000;(2):CD000323.
- Foley N, Teasell R, Salter K, et al. Dysphagia treatment post stroke: A systematic review of randomised controlled trials. Age Ageing 2008;37:258-264.
- Matsuo K, Palmer JB. Anatomy and physiology of feeding and swallowing: Normal and abnormal. Phys Med Rehabil Clin N Am 2008;19:691-707, vii.
- Legg LA, Drummond AE, Langhorne P. Occupational therapy for patients with problems in activities of daily living after stroke. Cochrane Database Syst Rev 2006;(4):CD003585.
- Antonucci G, Guariglia C, Judica A, et al. Effectiveness of neglect rehabilitation in a randomized group study. J Clin Exp Neuropsychol 1995;17:383-389.
- Bowen A, Knapp P, Gillespie D, et al. Non-pharmacological interventions for perceptual disorders following stroke and other adult-acquired, non-progressive brain injury. Cochrane Database Syst Rev 2011;(4):CD007039.
- Hackett ML, Anderson CS, House A, Xia J. Interventions for treating depression after stroke. Cochrane Database Syst Rev 2008;(4):CD003437.
Rehabilitation is a critical component of stroke treatment, as most stroke survivors are left with significant neurological impairments and other sequelae, such as spasticity and pain. Stroke rehabilitation aims to reverse these impairments to the extent possible, maximize functionality through the use of compensatory approaches, prevent complications, and manage comorbidities. This article reviews the basic principles of rehabilitation, current practices, and evidence supporting various aspects of stroke rehabilitation.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.