Management of Pulmonary Embolism in the ICU
By Samuel Nadler, MD, PhD
Critical Care, Pulmonary Medicine, The Polyclinic Madison Center, Seattle; Clinical Instructor, University of Washington, Seattle
Dr. Nadler reports no financial relationships relevant to this field of study.
Pulmonary embolism (PE) is a common condition in the ICU. Estimates are that 350,000 to 600,000 individuals in the United States suffer from deep venous thrombosis (DVT) and PE, with at least 100,000 related deaths.1 Small, low-risk PE can be treated as an outpatient procedure or with heparin infusion alone, but higher-risk PE cases are managed properly in an ICU. With the introduction of newer modalities of treatment, appropriate risk stratification and the choice of treatment are increasingly complex.
RISK STRATIFICATION
Once pulmonary embolism has been identified, an assessment of risk is important for appropriate treatment and triage to the ICU, medical floor, or treatment at home. Commonly used tools include the simplified pulmonary embolism severity index (sPESI), the Bova score, and the European Society of Cardiology (ESC) model.2,3 The sPESI score evaluates risk based on simple clinical features, including age > 80 years, history of cancer, history of chronic cardiopulmonary disease, heart rate > 110 bpm, systolic blood pressure < 100 mmHg, and oxygen saturation (SpO2) < 90%. If none of these are present, this is considered low risk, and outpatient treatment is appropriate. If any conditions are met, it is considered higher risk. The ESC model uses markers for shock, right ventricle (RV) dysfunction, and myocardial injury. If none are present, it is low risk. If one or two are present, it is intermediate risk, and the presence of all three defines high risk. Similarly, the Bova score assigns points for variables to generate an overall risk. A systolic blood pressure < 90-100 mmHg, positive cardiac troponins, or right ventricular dysfunction by echocardiogram or CT receive 2 points each, while tachycardia (heart rate > 110 bpm) receives 1 point. Stage I is 0-2 points, stage II is 3-4 points, and stage III is > 4 points.
How do these scoring systems perform in predicting mortality? Two recent studies addressed this question. Lankeit et al compared the ability of the sPESI and ESC models to predict 30-day mortality, non-fatal recurrent thromboembolism, and non-fatal major bleeding.2 In this study, there were no deaths in patients with sPESI = 0 vs. 11.1% mortality with sPESI > 1. The ESC model had a mortality rate of 3.4% in low-risk patients vs. 10.3% with elevated risk. Fernandez et al validated the Bova risk score.3 The PE-related mortality was 3.1%, 6.8%, and 10% in stages I, II, and III, respectively. From these data, test characteristics for each score can be generated (see Table 1). sPESI = 0, BOVA stage I/II, and ESC low-risk categories had a high negative predictive value for mortality, but the positive predictive values were poor. The sPESI = 0 category seemed to predict low mortality best, while a high Bova score performed best in terms of predicting high mortality.
These systems suggest risk stratification into three categories. Low-risk patients demonstrate no hemodynamic instability, no evidence of right ventricular strain, or hypoxemia, and often are treated as outpatients (sPESI = 0, BOVA stage I, ESC low risk). High-risk patients or those with “massive PE” present with shock, RV heart strain, and hypoxemia (Bova stage III, ESC high risk). An intermediate-risk pool presents without hemodynamic compromise or hypoxemia, but with evidence of right ventricular strain and myocardial injury, referred to as “sub-massive PE.” Each risk category defines the appropriate treatment.
TREATMENT MODALITIES
Treatment options for PE include anticoagulation alone, catheter-directed thrombolysis, systemic thrombolysis, and surgical embolectomy. The International Cooperative Pulmonary Embolism Registry (ICOPER) was one of the first broad assessments of the outcomes of patients with PE.4 This study followed 2,454 patients with PE between 1995-1996. In this cohort, 97% of patients received heparin, 76% received oral anticoagulants, 13% received systemic thrombolysis, and catheter or surgical thrombo-embolectomy was performed in 15 patients. Two-week all-cause mortality was 11.4%, and three-month mortality was 17.4%. More recently, the experience of the Massachusetts General Hospital pulmonary embolism response team (PERT) was published.5 They followed 394 activations of the PERT system. Of these, 29% were classified low risk, 45.5% as intermediate, and 25.5% as massive PE. Overall, 69% were treated with anticoagulation, 9% received catheter-directed therapy, 9% received systemic thrombolytics, and 3% underwent surgical embolectomy. Although anticoagulation alone still represents the most common treatment for PE, often it is undertaken outside the ICU and, thus, will not be a focus of this review.
High-risk PE patients may present with syncope or sudden cardiac arrest. For these patients, systemic thrombolytics often are administered. However, the systemic administration of thrombolytics carries a substantial risk of bleeding. In the ICOPER registry, 3% of patients receiving thrombolysis had intracranial hemorrhage, and major bleeding occurred in 21.7%.4 Overall, three-month mortality was 23% in patients requiring thrombolysis vs. 12.9% in those who did not. More recently, creators of the PEITHO study examined the effect of thrombolysis with 100 mg of tissue plasminogen activator (tPA) in intermediate-risk PE.6 The rate of major extracranial bleeding was 6.3%, minor bleeding occurred in 32.6%, and major bleeding in 11.5%. Thrombolytics did not improve mortality but reduced the incidence of hemodynamic decompensation. This suggested a lower dose of thrombolytics may be more appropriate. Authors of the MOPETT trial used 50 mg of tPA for intermediate risk PE.7 In the thrombolysis arm of this trial, no bleeding events were noted. A meta-analysis of trials of thrombolytics in all risk categories of PE from 1973-2002 failed to demonstrate a reduction in mortality or recurrent PE.8 Additionally, thrombolytics were associated with major bleeding in 9.1%, non-major bleeding in 22.7%, and intracranial hemorrhage in 0.5%. More recently, a meta-analysis of thrombolytic therapy in intermediate-risk patients showed lower all-cause mortality (odds ratio [OR], 0.53) with thrombolytics, but a greater risk of major bleeding (OR, 2.73).9 This analysis showed a number needed to treat of 59, with a number needed to harm of 18. While thrombolytics often are administered to patients with high-risk and massive or submassive PE, the benefits are unproven, and there is a known risk of bleeding. Thus, this treatment is reserved for patients with the highest risk in which the known risk of complications does not exceed the likely benefit of thrombolytics. Surgical pulmonary embolectomy may be considered for patients with life-threatening PE. The SPEAR work group reported outcomes of 214 embolectomies performed between 1994 and 2014.10 These included both massive PE (17.8%) defined as pulselessness, bradycardia, or the requirement for inotropes, and submassive PE (82.2%). Overall mortality was 11.7%, with a rate of 23.7% in massive PE, and 9.1% in submassive PE. Rates of stroke were 7.9% and 2.8% in massive and submassive PE, respectively. Other complications reported include prolonged mechanical ventilation (42.1% and 25%), renal failure (13.2% and 10.8%), transfusion requirements (76.3% and 36.4%), and the need for re-exploration (15.8% and 6.8%). A non-randomized, prospective study of 78 patients with massive PE failed to demonstrate a statistically significant mortality benefit of surgical embolectomy over thrombolytics but observed improved hemodynamics with embolectomy.11
With the significant complication rates observed with systemic thrombolysis and surgical embolectomy, catheter-directed techniques (CDTs) were developed to deliver thrombolytics precisely to emboli and mechanically retrieve clots using endovascular procedures. These include catheter-directed, low-dose thrombolytic infusions, embolus fragmentation, vortex embolectomy, rheolytic embolectomy, rotational embolectomy, and catheter-directed extraction embolectomy.12 A meta-analysis of 35 studies between 1990 and 2008 of 594 patients with massive PE who were treated with catheter-directed therapy demonstrated an overall clinical success rate, defined as hemodynamic stabilization, resolution of hypoxia, and survival of 86.5% (95% confidence interval [CI], 82.2-90.2%).13 The risks of major and minor procedural complications were 2.4% and 7.9%, respectively. The most common technique studied was rotating pigtail fragmentation, but it also included rheolytic devices such as the AngioJet device that has been associated with a high rate of complications. These techniques are considered off label as they do not carry an official indication by the FDA for treatment of PE.
More recently, three studies have extended the use of CDTs to both high- and intermediate-risk PE. The authors of the ULTIMA trial randomized 59 patients to heparin alone vs. ultrasound-assisted, catheter-directed thrombolysis (USAT).14 This demonstrated improvement in right ventricle to left ventricle (RV/LV) size ratio and improvement in pulmonary artery (PA) pressures and cardiac index. The SEATTLE II trial was a prospective single-arm study of USAT in 150 patients.15 This trial similarly demonstrated a reduction in PA pressures and RV/LV ratio. The 30-day mortality was 2.7%, with major bleeding complications in 10%, but no intracranial hemorrhage. The PERFECT trial was a prospective registry of 101 patients with both massive and submassive PE undergoing CDT.16 The in-hospital mortality was 5.9% without any reported major bleeding or intracranial hemorrhage. Interestingly, 64.4% of these patients also received inferior vena cava (IVC) filters.
ADJUNCT THERAPIES
In addition to therapies directly treating pulmonary embolism, IVC filters and extracorporeal membrane oxygenation (ECMO) are used increasingly in patients with PE. IVC filters are intended to prevent additional thromboembolism and hemodynamic compromise in patients with PE. IVC filters are indicated in patients with IVC, iliac, or ileofemoral DVT who have contraindications to anticoagulation such as recent intracranial hemorrhage, gastrointestinal or retroperitoneal bleeding, or major surgical procedures. One of the first large studies of IVC filters for PE with associated DVT, the PREPIC study, demonstrated that IVC filters reduced the likelihood of PE at day 12 (1.1% vs. 4.8%) without differences in major bleeding or death.17 However, in the two-year follow-up period, there was an increased risk of recurrent DVT. An eight-year follow-up also demonstrated reduced rates of symptomatic PE, but increased rates of DVT such that the overall hazard ratio for symptomatic venous thromboembolism was 1.12 (95% CI, 0.7-1.62).18 Reported complications with IVC filters include IVC penetration (0-41%), IVC occlusion (2-30%), filter migration (0-18%), filter fracture (2-10%), and death (0.12%).19 This led to the development of removable IVC filters, but filter retrieval rates are only about 34%. In patients with circulatory collapse, ECMO has been considered for hemodynamic support while definitive therapy of the thrombus is undertaken. Although the ESC guidelines for PE state ECMO may be used for PE, there are no randomized, controlled trials of ECMO for the management of PE, although case reports exist.20 The 2016 American College of Chest Physicians guidelines do not mention ECMO. As a point of reference, in the CESAR trial of patients with severe acute respiratory distress syndrome placed on ECMO, the six-month mortality rate was 37%.21 In centers with expertise and capability, ECMO may be considered as adjunct therapy to support the delivery of other modalities of treatment.
DISCUSSION
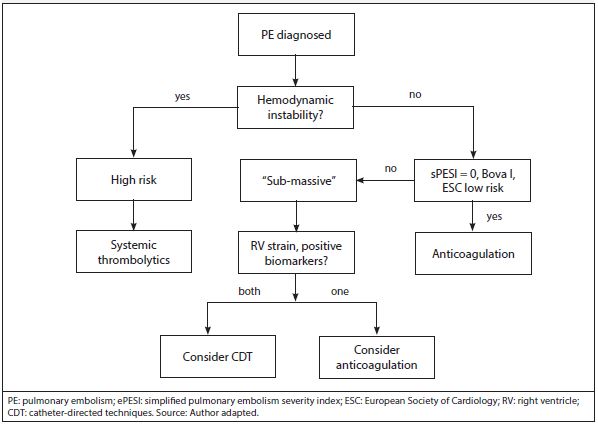
With many available risk stratification tools and treatment modalities, what is the optimal management for PE? Figure 1 presents a possible treatment algorithm. In a hemodynamically unstable patient with shock despite fluid administration, persistent vasopressor needs, or cardiac arrest, systemic thrombolytics may be administered. Although the data to support this approach are sparse, high-risk PE has a high associated mortality, and the risks of systemic thrombolytics probably are outweighed by the mortality risk. In contrast, in patients with PE who are low risk (sPESI = 0, BOVA stage I, ESC low risk), management with anticoagulation alone is appropriate and often is accomplished without hospitalization. Management of patients with submassive or intermediate-risk PE is more complex. Many treatment modalities with associated risks and benefits are available. In these cases, decision-making with a multidisciplinary approach, such as with a PERT program, may improve outcomes. Not all modalities are available within each center, and this availability may drive therapy. But when interventional approaches are available, one must consider the risk and benefits of each approach. In patients with submassive PE with both RV strain and positive biomarkers for cardiac injury, more interventional approaches may be appropriate. Recall the SEATTLE II and PERFECT trials reported mortality rates for patients with intermediate- and high-risk PE of 2.7% and 5.9%, respectively. This compares with an overall mortality of patients with sPESI > 1 of 11.1% and BOVA stage II of 6.8%. Mortality with surgical embolectomy for submassive PE in the SPEAR registry was 9.1%. In contrast, when only one of the findings of RV strain or cardiac injury is apparent, the risks may not justify such interventions. Recent trials of catheter-directed thrombolytics evaluated benefit by changes in RV/LV ratio and hemodynamics. If there is no RV strain, the benefit of CDT becomes less clear. IVC filters are indicated only when anticoagulation is contraindicated. The PREPIC2 trial demonstrated no benefit to IVC filters, even when large residual clot is present and patients are hemodynamically unstable.18 It is important to understand that most retrievable IVC filters are never removed, even under a controlled environment. The well-intentioned effort to temporarily protect patients with a “removable” IVC filter often results in the management of long-term complications of an irretrievable device. ECMO should be reserved for select patients with hemodynamic collapse in centers with considerable experience, as it remains an unproven intervention. Pulmonary embolism has variable presentations, from incidental findings on CT scan to cardiovascular collapse and sudden death. Many approaches to treatment are available, and matching the risk of mortality with the intervention to treat PE provides optimal care.
REFERENCES
- Office of the Surgeon General, National Heart, Blood, and Lung Institute. The Surgeon General’s Call to Action to Prevent Deep Vein Thrombosis and Pulmonary Embolism. Washington DC; 2008.
- Lankeit M, et al. A strategy combining imaging and laboratory biomarkers in comparison with a simplified clinical score for risk stratification of patients with acute pulmonary embolism. Chest 2012;141:916-922.
- Fernández C, et al. Validation of a model for identification of patients at intermediate to high risk for complications associated with acute symptomatic pulmonary embolism. Chest 2015;148:211-218.
- Goldhaber SZ, et al. Acute pulmonary embolism: Clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER). Lancet 1999;353:1386-1389.
- Kabrhel C, et al. A multidisciplinary pulmonary embolism response team: Initial 30-month experience with a novel approach to delivery of care to patients with submassive and massive pulmonary embolism. Chest 2016;150:384-393.
- Meyer G, et al. Fibrinolysis for patients with intermediate-risk pulmonary embolism. N Engl J Med 2014;370:1402-1411.
- Sharifi M, et al. Moderate pulmonary embolism treated with thrombolysis (from the “MOPETT” Trial). Am J Cardiol 2013;111:273-277.
- Wan S, et al. Thrombolysis compared with heparin for the initial treatment of pulmonary embolism. Circulation 2004;110:744-749.
- Chatterjee S, et al. Thrombolysis for pulmonary embolism and risk of all-cause mortality, major bleeding, and intracranial hemorrhage: A meta-analysis. JAMA 2014;311:2414-2421.
- Keeling WB, et al. Outcomes after surgical pulmonary embolectomy for acute pulmonary embolus: A multi-institutional study. Ann Thorac Surg 2016;102:1498-1502.
- Azari A, et al. Surgical embolectomy versus thrombolytic therapy in the management of acute massive pulmonary embolism: Short and long-term prognosis. Heart Lung 2015;44:335-339.
- Tapson VF, Jimenez D. Catheter-based approaches for the treatment of acute pulmonary embolism. Semin Respir Crit Care Med 2017;38:73-83.
- Kuo WT, et al. Catheter-directed therapy for the treatment of massive pulmonary embolism: Systematic review and meta-analysis of modern techniques. J Vasc Interv Radiol 2009;20:1431-1440.
- Kucher N, et al. Randomized, controlled trial of ultrasound-assisted catheter-directed thrombolysis for acute intermediate-risk pulmonary embolism. Circulation 2014;129:479-486.
- Piazza G, et al. A prospective, single-arm, multicenter trial of ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for acute massive and submassive pulmonary embolism: The SEATTLE II study. JACC Cardiovasc Interv 2015;8:1382-1392.
- Kuo WT, et al. Pulmonary Embolism Response to Fragmentation, Embolectomy, and Catheter Thrombolysis (PERFECT): Initial results from a prospective multicenter registry. Chest 2015;148:667-673.
- Decousus H, et al. A clinical trial of vena caval filters in the prevention of pulmonary embolism in patients with proximal deep-vein thrombosis. Prévention du Risque d’Embolie Pulmonaire par Interruption Cave Study Group. N Engl J Med 1998;338:409-415.
- PREPIC Study Group. Eight-year follow-up of patients with permanent vena cava filters in the prevention of pulmonary embolism: The PREPIC (Prevention du Risque d’Embolie Pulmonaire par Interruption Cave) randomized study. Circulation 2005;112:416-422.
- Moriarty JM, et al. Inferior vena cava filters: When to place and when to remove. Semin Respir Crit Care Med 2017;38:84-93.
- Weinberg A, et al. Massive pulmonary embolism: Extracorporeal membrane oxygenation and surgical pulmonary embolectomy. Semin Respir Crit Care Med 2017;38:66-72.
- Peek GJ, et al. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): A multicentre randomised controlled trial. Lancet 2009;374:1351-1363.
- Konstantinides SV, et al. 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J 2014;35:3033-3069.
Table 1: Test Characteristics for sPESI, ESC, and Bova Score |
||||||
|
Test |
Sensitivity |
Specificity |
PPV |
NPV |
+LR |
-LR |
|
sPESIa |
100 |
33.9 |
11.1 |
100 |
1.51 |
0 |
|
ESCb |
82.5 |
41.1 |
10.3 |
96.6 |
1.40 |
0.43 |
|
Bovac |
26 |
97 |
42 |
93 |
7.93 |
0.76 |
|
PPV = positive predictive value; NPV = negative predictive value; +LR = positive likelihood ratio; -LR = negative likelihood ratio. a 30-day mortality risk sPESI = 0 vs. sPESI > 1. b 30-day mortality risk ESC low vs. intermediate or high. c Risk of PE-related death, hemodynamic compromise, or recurrent non-fatal PE Bova stage I/II vs. stage III |
||||||
Figure 1. Treatment Algorithm for Acute Pulmonary Embolism22 |
 |
Small, low-risk pulmonary embolism can be treated as an outpatient procedure or with heparin infusion alone, but higher-risk pulmonary embolism cases are managed properly in an ICU. With the introduction of newer modalities of treatment, appropriate risk stratification and the choice of treatment are increasingly complex.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
