Hemodynamic Monitoring Modalities in the Emergency Department
October 15, 2017
Reprints
AUTHORS
Steven Field, DO, Assistant Professor, Department of Emergency Medicine, University of Texas Southwestern/Parkland Memorial Hospital, Dallas
Maxwell Hockstein, MD, University of Texas Southwestern/Parkland Memorial Hospital, Dallas
PEER REVIEWER
Boris Garber, DO, Attending Physician, MetroHealth Medical Center, Assistant Professor, Case Western Reserve University School of Medicine, Cleveland, OH
EXECUTIVE SUMMARY
- When a patient is in shock, one of the first assessments is whether to start fluid resuscitation. There are now several ways to assess this at the bedside, including waveform monitors and passive leg raise with ultrasound of the inferior vena cava.
- Passive leg raise relocates about 300 mL to 500 mL from the legs to the central circulation. Distension of the inferior vena cava on ultrasound after leg raise signals a positive response to fluid.
- Lung ultrasound can be used to determine early fluid overload as well as pneumonia or pneumothorax.
- An increase in quantitative end tidal CO2 indicates improved cardiac output.
Introduction
Hemodynamic monitoring involves the assessment, measurement, and detection of changes in the blood flow to body tissues. This includes blood volume, fluid balance, and how well the heart is pumping. Emergency physicians (EPs) commonly diagnose, stabilize, and care for critically ill patients in emergency departments (EDs). Hemodynamic monitoring is a very important component in effectively resuscitating critically ill patients. Various methods of hemodynamic monitoring not only give the physician an idea of the patient’s overall pathophysiology, but can alert the physician to acute changes. Hemodynamic monitoring can play a key role in the decision to start IV fluids, place a central venous catheter, start vasopressors, or initiate thrombolytics. Hemodynamic monitoring also can play a role in determining the appropriate resuscitative efforts by providing information on the response to treatment.1 Although time and resources typically are more restricted in the ED, the initial hours of a patient’s presentation to the ED are critical, often determining the patient’s ultimate outcome.
Cardiac Output
Monitoring of hemodynamics is essential in the ED and intensive care units. In addition to heart rate, blood pressure (both invasive and noninvasive), respiratory rate, oxygen saturations, and temperature, there are many other monitoring modalities that confer diagnostic and therapeutic benefit. Monitoring these parameters yields important resuscitation targets that serve as important care milestones. The job of professionals taking care of critical care patients is deceptively simple: to optimize cardiac output.
Cardiac output is a product of two values: heart rate and stroke volume (the amount of blood ejected with each heart beat). Heart rate is a function of autonomic tone and is regulated relatively easily when needed either pharmacologically or electrically. Stroke volume is a function of several more variables: preload, afterload, and contractility. The relationship between preload and stroke volume is articulated in the Frank-Starling mechanism.
Fluid Responsiveness
One of the first concerns during resuscitation is the assessment of volume status. One of the terms used frequently in the literature is “fluid responsiveness,” which can be defined broadly as any measurable increase in cardiac output in response to a fluid bolus (“challenge”). In the ED, there are an increasing number of options to determine the presence of fluid responsiveness and assessment of interventions aimed at increasing cardiac output.
Invasive Hemodynamic Monitoring
Central venous pressure (CVP) has been used for many years as a surrogate for volume status, providing a numerical value to guide volume resuscitation. CVP is an easily attainable value that reflects right atrial pressure but does require a central line and some special equipment not always available in the ED. Either an internal jugular or subclavian central venous access can be used. The general assumption is that CVP estimates right ventricular end-diastolic volume index.2 Right ventricular end-diastolic volume index is a measure of preload,3 which, when low, is assumed to be a marker of hypovolemia. Because ventricular compliance (defined as change in volume over change in pressure) is not a linear relationship, ventricular volumes do not predict fluid responsiveness.2
Arterial lines have the potential to provide more information than just blood pressure. The waveforms by themselves have the potential to disclose much about hemodynamic status. Several invasive monitoring devices use both arterial line contours in combination with proprietary technologies to measure additional variables (the most common are cardiac output and stroke volume). Several parameters also must be satisfied for the values to be interpreted correctly. However, in the right patient, these measurements can be used to determine fluid responsiveness.
The underlying concept between all waveform analysis devices (also called pulse contour devices) is that arterial waveform analysis can determine increases and decreases in cardiac output.4 Several products are available, but none have been proven to be superior to others. For the sake of an example, we will be discussing the FloTrac device (Edwards Lifesciences Corporation, California). Although there are differences in the individual proprietary technologies, many of the monitors work similarly.
The FloTrac calculates cardiac output, cardiac index, stroke volume, stroke volume variation, and stroke volume index. With appropriate central venous access, the device also can calculate SVR and central venous oxygen saturation. These devices were validated during cardiovascular surgery using thermodilution as the gold standard for determining cardiac output,5 and utility has been expanded to the critical care arenas. It has been suggested that minimally invasive cardiac output monitors may be useful in the ED (for example, in burn patients),6 but large-scale studies still are needed to demonstrate the clinical effect of the early implementation of these monitors.
Stroke volume variation can be an important parameter that is reported by most minimally invasive devices. Under normal conditions (spontaneous breathing), there is variation with respiration, decreasing blood pressure with inhalation — typically the systolic pressure does not decrease more than 5 mmHg.7 During inspiration, blood collects in the pulmonary vasculature, which decreases the LVEDV and, therefore, the stroke volume. Extreme variation is known as pulsus paradoxus, in which the pulse disappears during inspiration.
During mechanical ventilation, intrathoracic pressure dynamics are inverted, which leads to an increase in LVEDV because of an afterload reduction and, therefore, an increase in stroke volume. This phenomenon has many names, but the most common are paradoxical pulsus paradoxus, systolic pressure variation, and pulse pressure variation.7 Pulse pressure variation (PPV) is reported as a percentage. It is generally accepted that a pulse pressure variation less than 10% suggests that a patient will not be volume responsive.
PPV = [(Maximum pulse pressure - Minimum PP)/PP mean] × 100
The studies that have validated pulse pressure variation were performed in the intensive care unit and operating room on patients who were mechanically ventilated receiving > 8 mL/kg tidal volume8 and in patients who were not hemodynamically labile or unstable. Although likely variable per institution, one study demonstrated that only 7% of patients in the ICU met all the criteria that made PPV interpretable.9 Stroke volume variation is a natural extension of pulse pressure variation; however, the relationship between the two is not linear.10
Stroke volume variation is more difficult to calculate at the bedside because stroke volume is not a readily available measurement; however, it is calculated similarly to PPV, substituting SV for PP. The calculation of stroke volume is proprietary to many of the pulse contour devices, and the same exclusion criteria for its interpretation apply. Normal values for stroke volume variation (in the aforementioned patient population) are between 10-15% and, in general, the higher the variability, the more likely the patient is to be volume-responsive.
Noninvasive Hemodynamic Monitoring
Passive Leg Raise. Passive leg raise (PLR) can be used to determine fluid responsiveness. It is performed with the patient in a sitting position at a 45 degree angle. The patient then is reclined to a supine position, and the patient’s legs are raised to a 45-degree angle with the bed. This maneuver mobilizes 300 mL to 500 mL of blood volume from the lower limbs to the intrathoracic compartment, which in turn increases right ventricular preload and, thus, stroke volume.1,11-13 Stroke volume/cardiac output are measured before and after to determine if the patient may be fluid responsive. A 10% increase in stroke volume is consistent with fluid responsiveness.14 If a threshold level of improvement is reached, there is preload reserve.12,15 This also has been called an “auto-bolus,” as the maneuver mobilizes 300 mL to 500 mL of blood that mimics fluid administration without actually having to give an exogenous fluid bolus.12 In a recent systematic review and meta-analysis by Cherpanath et al, PLR was demonstrated to have high diagnostic performance with a sensitivity of 86% and specificity of 92%.16,17 The PLR is an easy, quick maneuver that requires minimal training. However, the PLR must be interpreted in conjunction with a method that detects changes in cardiac output, such as VTI, arterial Doppler, or end tidal CO2 (ETCO2).1,13
End Tidal CO2. ETCO2 involves the noninvasive measurement of the partial pressure of carbon dioxide (CO2) in an exhaled breath expressed as the CO2 concentration over time.18 It is represented as waveform on most standard machines as well as an absolute value. Quantitative end tidal capnography generally is available in many EDs in the United States. It has many common uses, including as a tool to monitor cardiac output during cardiopulmonary resuscitation (CPR), to monitor for apnea during procedural sedation, and to confirm endotracheal tube (ETT) placement after intubation, and it may be used in deciding whether or not a patient is fluid responsive. Thus, it is widely available and within our skill set to use in general practice as EPs.
The measurement of ETCO2 indicates changes in the elimination of CO2 from the lungs. Additionally, ETCO2 can indicate blood flow and tissue perfusion indirectly by demonstrating changes in CO2 production at the tissue level as well delivery of CO2 to the lungs by the circulatory system.19 Changes in the shape of the waveform as well as the absolute ETCO2 value can indicate a pathologic disease process as well as severity of disease. Additionally, ETCO2 may be monitored in the ED for changes from baseline as a hemodynamic monitoring device to indicate changes in the patient’s condition and need for intervention.
During CPR, measurement of ETCO2 varies directly with the cardiac output produced by chest compression and, thus, can be used as a hemodynamic monitoring device to guide compression quality and effectiveness. This can include effectiveness of compressions, when to consider return of spontaneous circulation (ROSC) with spike in ETCO2 during CPR, and the decision of when to terminate compressions.18-22 The 2013 American Heart Association (AHA) consensus statement recommends titrating CPR performance to a goal ETCO2 of greater than 20 mmHg while not excessively ventilating the patient.21 The 2010 AHA Guidelines for CPR and ECC recommend monitoring ETCO2 during CPR to assess blood flow in two ways: to keep ETCO2 above 10 mmHg during CPR as a reflection of adequate chest compression and to consider an abrupt sustained increase as an indication of return of spontaneous circulation. The failure to maintain ETCO2 above 10 mmHg during CPR is a poor prognostic indicator for survival.23,24
Verification and monitoring of ETT placement in the ED is an important part of intubation in the ED. For patients who are not in cardiac arrest, qualitative capnography has been shown to be highly accurate in determining ETT placement.25 Patients with normal lung function have prototypical rectangular waveforms. There are four phases: 1) inspiratory baseline; 2) expiratory upstroke; 3) alveolar plateau; 4) (also known as phase 0) inspiratory downstroke (beginning of next inspiration).25 When the esophagus is intubated, there is a gastric washout that occurs, and the ETCO2 plummets to zero.25
Monitoring in procedural sedation for analgesia/sedation-related apnea and hypopnea has long been a topic of interest in emergency medicine. ETCO2 monitoring is one modality that has been available to most EDs. ETCO2 values less than 30 mmHg (hypopneic hypoventilation) or greater than 50 mmHg (bradypneic hypoventilation) would give the EP cause for concern and would require intervention.26 The 2014 Clinical Policy: Procedural Sedation and Analgesia in the Emergency Department gives a level B recommendation regarding its use to decrease adverse respiratory events, such as serious patient-centered outcomes, aspiration, unplanned intubation, or cardiac arrest. The policy states that capnography can be used with pulse oximetry and clinical assessment to detect hypoventilation and apnea early in patients undergoing procedural sedation and analgesia.27 Use of ETCO2 during procedural sedation decreases the incidence of hypoxia and respiratory events;26 however, the jury is still out on whether ETCO2 monitoring reduces the incidence of serious adverse events during procedural sedation, for example neurologic injury caused by hypoxia, aspiration, or death.18,26,27
Lastly, ETCO2 monitoring also has been used in conjunction with a PLR to determine fluid responsiveness. A patient who is fluid responsive will increase stroke volume and thus cardiac output in response to a fluid bolus from PLR. As described above, it has been demonstrated that changes in ETCO2 correlate with changes in cardiac output. Thus, ETCO2 monitoring can assist in determining whether a patient may be fluid responsive.
In a recent prospective observational study, Toupin et al showed that a change in cardiac output by 15% induced by PLR correlates with a change in ETCO2 by 2 mmHg.28 Garcia et al demonstrated a sensitivity of 90.5% and a specificity of 93.7% of predictive performance of ETCO2 to predict fluid responsiveness.29 ETCO2 monitoring can be very limited in those patients with co-existing pulmonary disease. If a patient has a V/Q mismatch, then CO2 cannot be delivered to the lungs and ventilated where it then can be measured.28 Also, in spontaneously breathing patients, the unpredictable variations in minute ventilation could make the analysis of changes in ETCO2 more difficult.29 All in all, ETCO2 monitoring could a viable, noninvasive method for the assessment of fluid responsiveness and hemodynamic monitoring in the ED.
Point-of-Care Ultrasound. Point-of-care ultrasound (POCUS) is readily available in most EDs and currently is being used in bedside diagnostics, therapeutics, and procedural guidance. It has become part of the core curriculum in residency programs, and most graduates from emergency medicine programs have the basic skills required to use POCUS in hemodynamic monitoring.
POCUS has many distinct advantages that make it an ideal noninvasive tool for hemodynamic monitoring and assessment, including being inexpensive, readily available, safe, free of radiation, and usable at the bedside. There are various ultrasound modalities that can be used in hemodynamic monitoring of patients. This article will focus on inferior vena cava (IVC) ultrasound, echocardiography, lung ultrasound, and velocity time integral of the left ventricular outflow tract for cardiac output.
The IVC is a thin-walled, large, compliant blood vessel that empties into the right atrium. Because the IVC is so compliant, its respiratory variability can indicate intravascular volume status and fluid responsiveness. In a spontaneously breathing patient, during inspiration, intrathoracic pressure decreases and the IVC collapses as blood is transferred to the right atrium. Conversely, in patients who are undergoing positive pressure ventilation (either patients on noninvasive ventilation or those who are intubated), intrathoracic pressure increases on inspiration, the blood is pushed back into the IVC from the right atrium, and the IVC distends.
IVC variation has been shown to correlate with central venous pressure (CVP).30,31 Although the utility of CVP is limited, low and high values of CVP can have good positive predictive value and negative predictive value, respectively, for fluid responsiveness.32,33 Furthermore, IVC ultrasound can be more advantageous than standard CVP monitoring because, comparatively, it is noninvasive and quickly accessible. It does not require an invasive procedure, unlike placement of a central line, which is a requirement for CVP catheters.31,34 Collapse of the IVC greater or less than 50% is noted 2 cm distal to where the IVC joins the right atrium.11,15,30,34,35,36 An IVC that collapses greater than 50% correlates with a CVP of 0-5 mmHg, and an IVC that collapses less than 50% correlates to greater than 10.34,36
IVC distensibility in a ventilated patient is demonstrated by IVC distension greater than 18% after a fluid bolus or passive leg raise.15 Zhang et al concluded in a meta-analysis in 2014 that the diagnostic accuracy of change in the IVC diameter did predict fluid responsiveness particularly well in patients who were on mechanical ventilation. They showed a sensitivity and specificity overall of 76% and 86%, respectively. Additionally, they showed in the ventilated subgroup sensitivity and specificity of 81% and 87%, respectively.30,36
IVC ultrasound does have limitations. First, it is specific but not as sensitive. If the IVC does not collapse/distend, then this may be falsely negative for fluid responsiveness.11,33 Respiratory effort in spontaneously breathing patients can affect IVC diameter. Patients who are breathing harder may have greater intrathoracic pressure changes with respiration independent of fluid responsiveness. Likewise, patients who have decreased amplitude of intrathoracic pressure changes with respiration (such as patients with respiratory depression or hypopnea) will decrease respiratory variability in IVC diameter independent of fluid responsiveness.31,32,33
The IVC ultrasound view is obtained with a phase array or curvilinear probe in the subxiphoid area, in the longitudinal plane. The IVC is identified entering the right atrium, and collapse/distension is noted 2 cm distal to the junction of the IVC and right atrium.
Figure 1. Inferior Vena Cava View
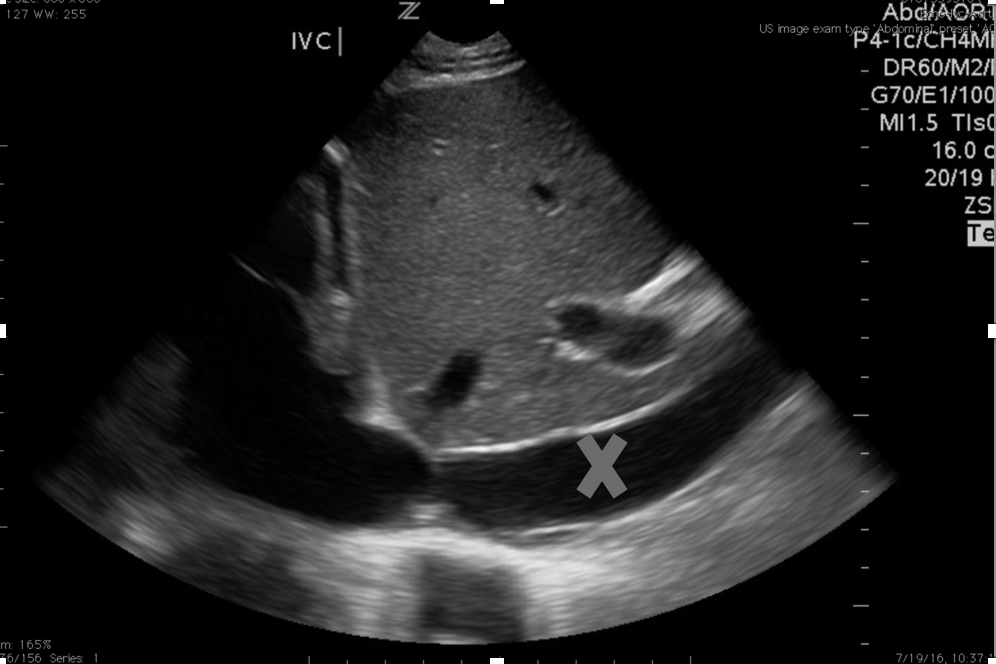
Appropriate place for IVC visual estimation
Echocardiography. Echocardiography is a great bedside tool for hemodynamic assessment as it allows real-time anatomical and functional cardiac assessment in many different pathophysiologic situations.37 There are many views and calculations that may be obtained by cardiologists and echocardiographers during a full evaluation echocardiogram. At the bedside in the hemodynamically unstable patient, limited echocardiography is used in just two ways: to measure cardiac output and cardiac function.15 It has an additional diagnostic advantage by allowing for visualization of the pericardium, the ventricles, and the valves.38 Point-of-care echocardiography is called bedside limited echocardiogram or a focused cardiac ultrasound (FOCUS). FOCUS typically includes four basic views: a parasternal long view, parasternal short view, subxiphoid view, and apical four-chamber view. The three key findings that a clinician should assess are left ventricular (LV)39 contractility, right ventricular dilation, and the presence of a pericardial effusion/cardiac tamponade.15,40,41
Figure 2. Subxiphoid View of Echocardiogram
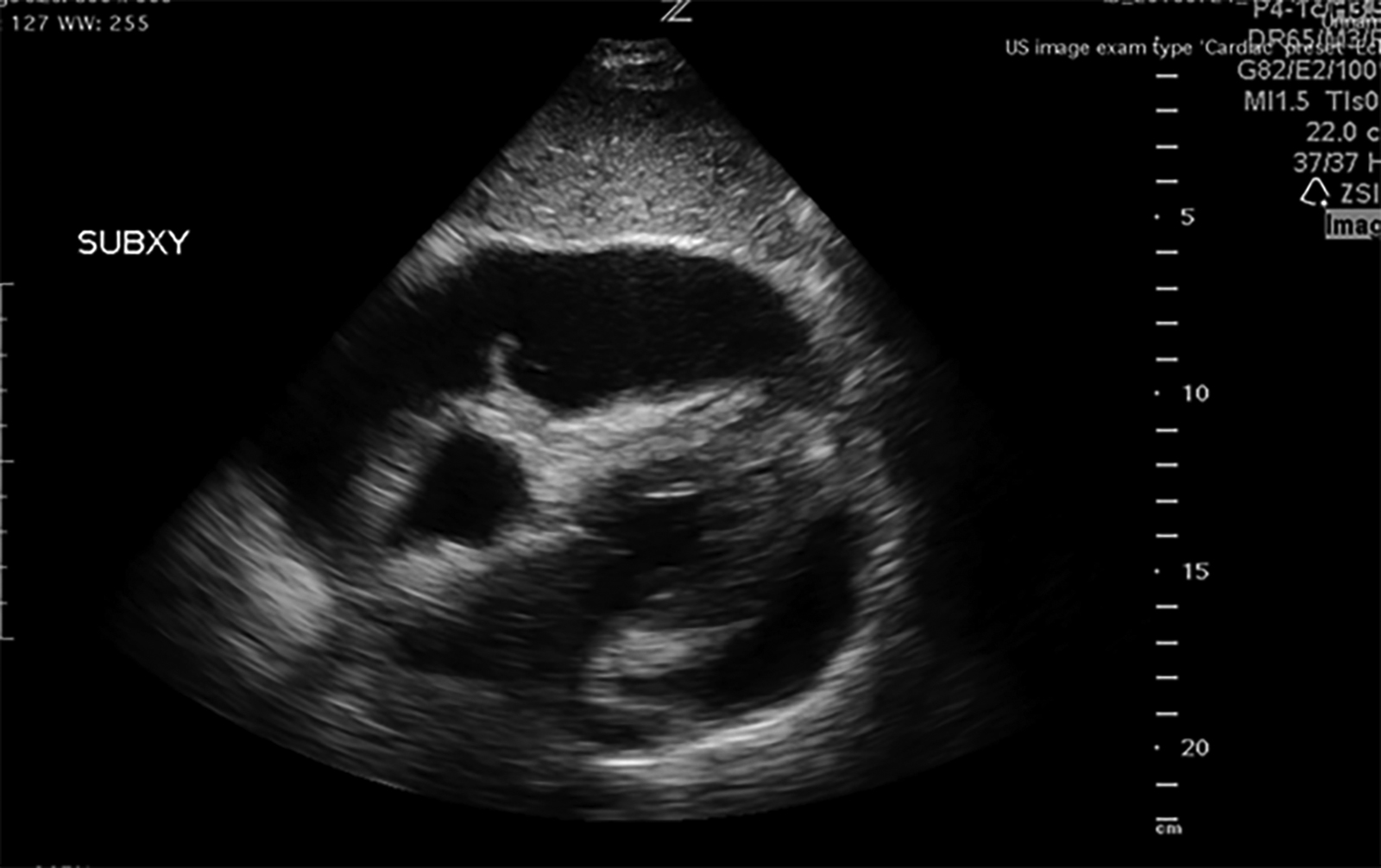
Diastolic right ventricular collapse consistent with cardiac tamponade
Echocardiographic monitoring plays a fundamental role in patients presenting with pericardial effusion to the ED. Specifically, determining if there is a pericardial effusion present is important in the setting of hemodynamic instability to determine if the patient has cardiac tamponade. FOCUS can play a critical role in decision making for patients presenting with significant pericardial effusion, guiding diagnosis, monitoring, procedural guidance, and postpericardiocentesis management.42
Cardiac tamponade occurs when the presence of a pericardial effusion causes equal increases in atrial and pericardial pressures. This prevents diastolic filling, decreases SV, and thus decreases cardiac output, resulting in hypotension and shock. The amount of fluid is not as important as the speed of fluid accumulation. FOCUS is the test of choice for the diagnosis of pericardial effusion, and has good sensitivity and specificity.41,43 Identification of a pericardial effusion can be performed on any of the aforementioned views and is noted by an anechoic space between the parietal and visceral pericardium.
Assessment of LV function is essential in hemodynamic monitoring of unstable patients. LV systolic function often is used as a surrogate for myocardial contractility, the most common method of measurement is the ejection fraction (EF).43 There are many methods to calculate EF. The most common is the Teichholz method. However, there is a significant amount of data to suggest that visual estimation of EF is just as accurate. In a prospective cross sectional study performed by Unluer et al, 133 patients had an EF measurement that was performed by visual estimation by an EP which was compared to a formal echocardiogram by an experienced cardiologist who performed a full evaluation of left ventricular systolic function. The EP’s visual estimation of EF agreed with the comprehensive echocardiogram by the cardiologist and could be performed at the bedside in a shorter amount of time.44
EF can be simplified into three different classifications: severely depressed (EF < 30%), mild-moderately depressed (EF = 30-55%), and normal (EF > 55%). EF greater than 70% is considered hyperdynamic, which may appear as near obliteration of the ventricular cavity during systole.34 For example, a hyperdynamic LV may indicate vasodilatory shock in the setting of sepsis, prompting intravenous fluid administration.41
Assessment of right ventricular function can give a great deal of information in the setting of clinical concern for right heart dysfunction, i.e., RV infarct, acute pulmonary embolism, or acute cor pulmonale.34,41 The right side of the heart typically is a lower pressure system in comparison to the left side of the heart and is very sensitive to acute elevations in pressure. With very small changes in pressure, there can be large shifts in volume, and right ventricular dilation (RVD) can develop. Typically the RV:LV ratio is 0.6:1.0. When the RV is 1:1 with the LV, it is moderately dilated, and when the RV is larger than the LV, the RV is severely dilated.34 RVD in patients with pulmonary embolism can be evidence of right-heart dysfunction, which can significantly increase morbidity and mortality. Bedside echocardiography can be used to risk-stratify and to better manage these patients.34,41 Specifically, identifying RVD on bedside ultrasound can play a significant role in making therapeutic decisions in patients with pulmonary embolism and can facilitate a change in management by identifying those at high risk who might otherwise be treated with less aggressive therapies.36,42 Furthermore, EPs are good at making this bedside determination and thus acting upon the results. Rutz et al found that EPs can diagnose RVD on focused cardiac ultrasound with good agreement with cardiology.45
FOCUS also can be used to calculate cardiac output. Determining the cardiac output can be a main driver in the decision to initiate a particular treatment therapy, such as crystalloid fluids, vasopressors, inotropes, etc. Cardiac output is defined by the product of the stroke volume (SV) and the heart rate (HR). On FOCUS, SV can be calculated as the column of blood ejected out of the left ventricular outflow tract (LVOT) through the aortic valve. The cross-sectional area of the LVOT is multiplied by the velocity time integral (VTI) of flow through aortic valve to get the SV. In practice, the LVOT diameter is measured at the level of the aortic valve leaflet insertion while the heart is in systole from the parasternal long-axis view. The area is calculated by dividing the diameter by 2 to get the radius, then using pi × radius squared to obtain the area. The VTI of LVOT blood velocity is obtained using pulse wave Doppler from a transthoracic apical five-chamber view. This requires the pulse wave gate to be placed at the aortic valve in the same place where the LVOT was measured.34,42,43
Figure 3. Apical Four-chamber View of Echocardiogram
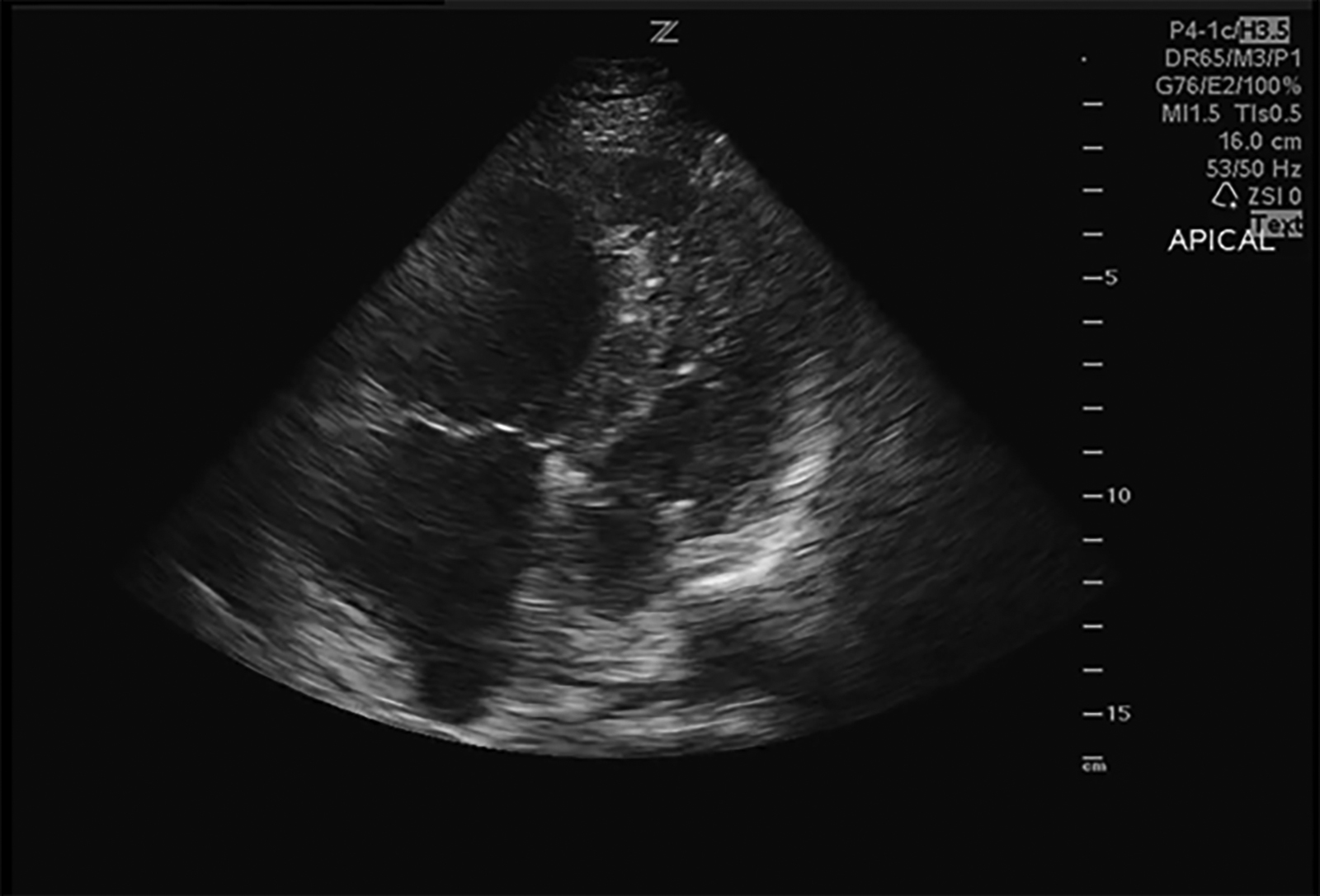
Right ventricular dilation with septal bowing into the left ventricle
After calculation of cardiac output, the percent change before and after a PLR is a specific indicator of fluid responsiveness. Percent change is calculated by [(CO after PLR – CO before PLR)/CO after PLR] × 100%. An increase in cardiac output of greater than 10% predicts fluid volume responsiveness and then is a good indicator for which patients will respond to a fluid bolus.11 Additionally, EPs are capable of determining cardiac output using this method with good reliability.35 Dinh et al demonstrated that EPs could obtain accurate cardiac index (CI) measurements by performing a bedside focused cardiac ultrasound examination with cardiac LVOT/VTI.46
For completeness, a brief mention of transesophageal echocardiography (TEE) is essential. In the past, this mainly has been a procedure performed in the operating room by anesthesiology, in the echo lab by cardiology, and at the bedside in the ICU. However, in recent years, TEE in the ED has gained popularity and has become implemented sporadically in EDs. It not only uses a skill set that EPs are familiar with on a day-to-day basis, but it can be a vital tool to answer clinical questions during cardiac arrest and circulatory failure.
TEE offers many distinct advantages over transthoracic echocardiogram, including more reliable acoustic windows, better image quality, and better reproducibility. Additionally, TEE may be used unrestricted during a cardiac arrest while chest compressions are ongoing without losing image quality.47
Lung Ultrasound. Another cutting edge test for the ED is the lung ultrasound. Not only is it a great tool at the bedside for diagnostic purposes, but it also can be a significant hemodynamic monitoring tool. Lung ultrasound can be used for initial evaluation of a patient with hypotension and shock as well as for monitoring those patients undergoing fluid resuscitation in early demonstration of fluid overload.48,49 Additionally, lung ultrasound can be used to rapidly assess and monitor patients in respiratory failure, differentiating between pneumothorax, pneumonia, pleural effusion, and alveolar/interstitial diseases.49,59
When scanning the lung, it is important to orient oneself in the longitudinal plane. The sonographer should use a low-frequency probe (2-6 MHz) to ensure the ability to see deeper. The sonographer can orient himself or herself by using the rib shadows as a landmark for pleural line identification.34,51
Once the pleural line is identified, the sonographer sees (in normal lung tissue) movement of the parietal on the visceral pleura called “lung slide.” This indicates there is no pneumothorax. In the right clinical setting, the absence of lung slide may indicate pneumothorax. When looking deeper than the pleural line, it is also important to remember that air does not transfer sound well, but instead scatters it so that the sound does not return to the probe in an organized fashion.34
In normal lung tissue, ultrasound travels deep to the pleural line and is scattered, never returning back to the probe. Instead, ultrasound waves bounce back and forth within the pleura, creating a horizontal reverberation artifact that is called an A-line.34
As density of the lung tissue increases, for example with fluid accumulation, the lung behaves more like a solid organ. Ultrasound waves are able to permeate the lung, reflecting many times, creating bright white, vertical, laser-like lines that originate from the pleura and transmit the full depth of the screen. These lines are called B-lines.34,51
Figure 4. Normal Thoracic View
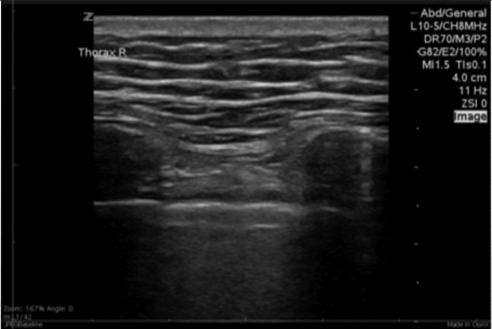
Figure 5. Thoracic View With Multiple B-Lines
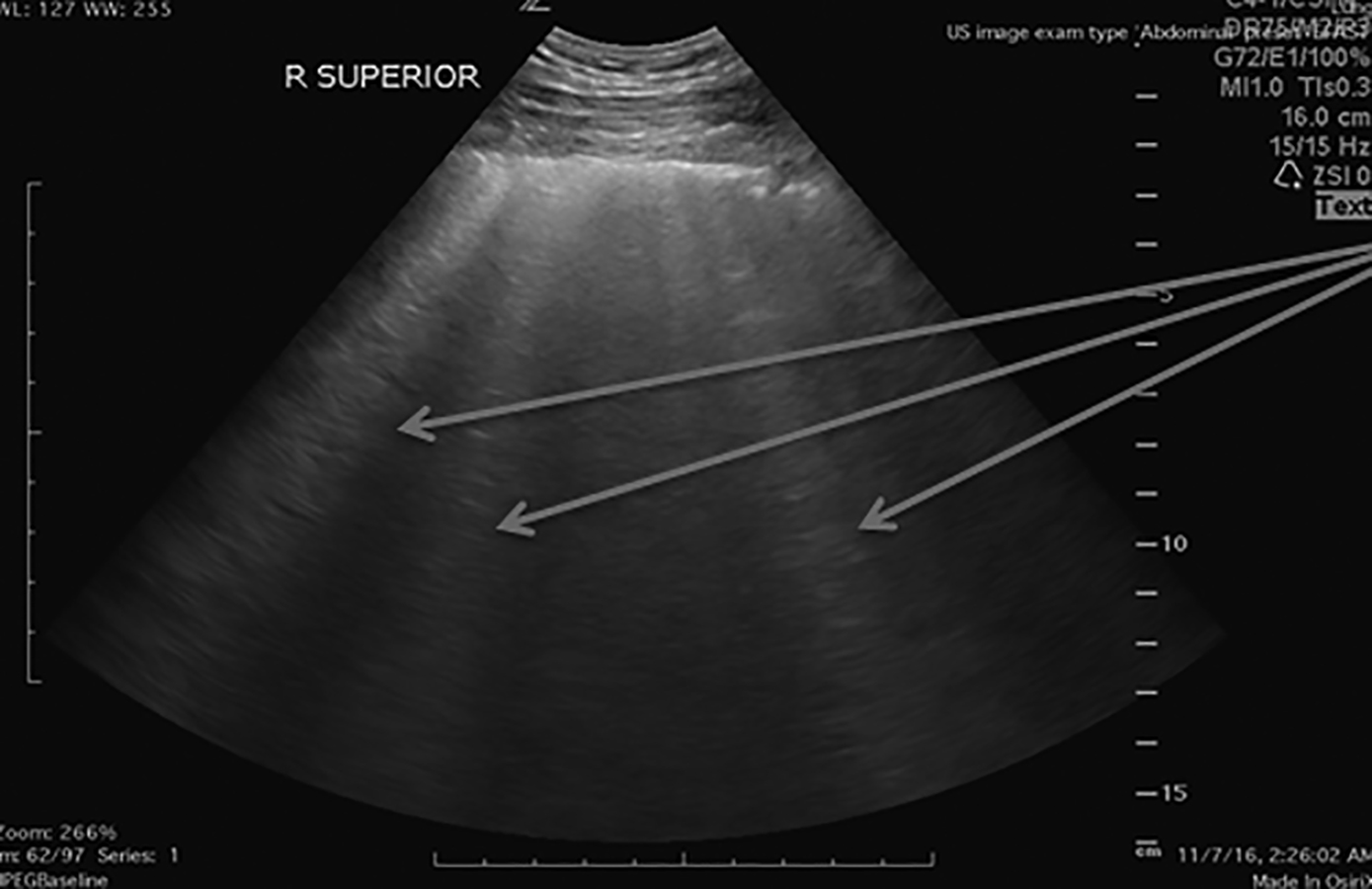
When scanning the lungs, it is important to scan eight different zones between both sides. This is to ensure that the sonographer is not missing any key areas of pathology as well as to get a good sense of distribution of disease.34
As previously stated, lung ultrasound also can be a great tool in assessment and monitoring in those patients with hypotension and shock states. In a patient identified as being hypotensive or in shock, the FALLS protocol developed by Daniel Lichtenstein can be a quick, efficient way to diagnose the etiology of shock as well as to guide the resuscitation in intravenous fluid (IVF) administration.
The diagram in Figure 6 is a quick approach to the hypotensive patient wherein the type of shock can be identified and treated rapidly. This may be used many times during the resuscitation to monitor the patient and his or her progress and trajectory. After quickly identifying the type of shock, if appropriate, fluid may be administered. The improvement of the patient’s perfusion during the FALLS protocol, while A-lines still remain unchanged, also may indicate hypovolemic shock and/or septic shock. This may be indicative of more room for fluid administration. If the signs of shock persist despite fluid therapy, fluid overload can be generated. This can be detected immediately on lung ultrasound because B-lines begin to replace A-lines. Fluid resuscitation should be stopped and other forms of pressure support started if necessary.34,48,49,51
Figure 6. FALLS Protocol
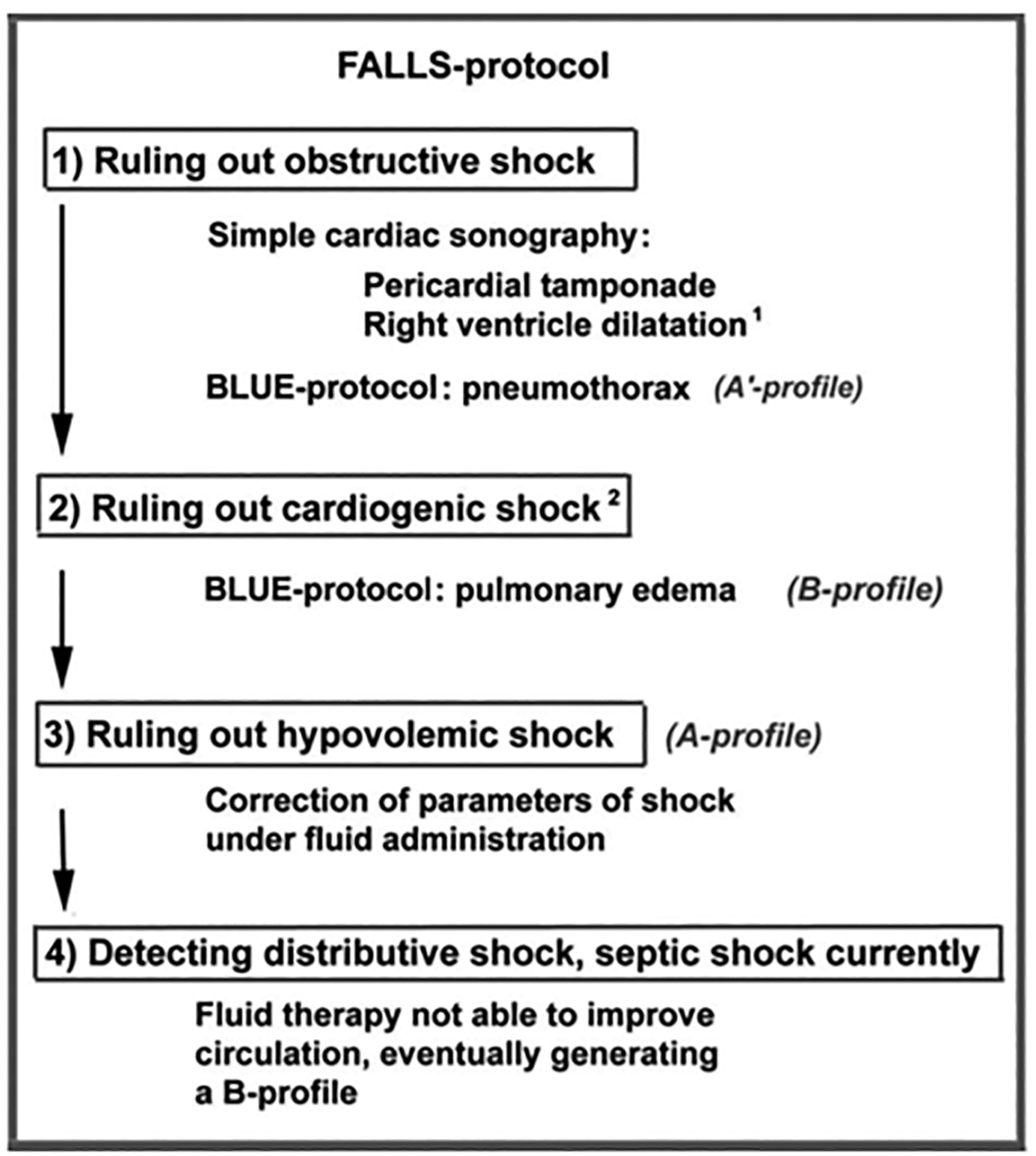
Reprinted with permission from: Lichtenstein DA. BLUE-Protocol and FALLS-Protocol: Two applications of lung ultrasound in the critically ill. Chest 2015;147:1659-1670.
The development of more than three B-lines in a particular lung zone is considered pathologic. In the setting of fluid overload and interstitial disease, this generally develops bilaterally, indicating the need to stop fluid administration.34 Anile et al demonstrated that detecting more than three B-lines in a particular view on lung ultrasound correlates reliably with the increase in extravascular lung water (EVLW) and, in turn, the evolution of cardiogenic pulmonary edema, acute lung injury, and acute respiratory distress syndrome (ARDS).52 This is one of the advantages of using lung ultrasound in monitoring the patient in shock. Changes in fluid shifts can be measured in real time by the sonographer, making this a useful emergency medicine hemodynamic monitoring tool. Additionally, it requires no ionizing radiation, no patient transport, and no lag time between image acquisition and interpretation for a trained clinician sonographer.34
An additional advantage and use of lung ultrasound is the bedside evaluation and continued monitoring of acute respiratory failure. EPs using bedside lung ultrasound can differentiate reliably between patients with cardiogenic pulmonary edema, pneumonia, ARDS, pneumothorax, and diaphragmatic dysfunction.50 Decompensated heart failure/cardiogenic pulmonary edema may be differentiated accurately from noncardiogenic causes of dyspnea with sensitivity and specificity of 94% and 92%, respectively.53,54 In a systematic review, the diagnostic performance of lung ultrasound was better than any other test that was studied, with a positive likelihood ratio of 7.4 and a negative likelihood ratio of 0.16.55
Additionally, lung ultrasound can be used to monitor patients who have cardiogenic pulmonary edema. It has been demonstrated that using lung ultrasound, physicians can monitor a patient’s response to therapy (i.e., noninvasive ventilation, diuretics, etc.) by assessing the clearance of B-lines. This can be done in the early hours of treatment, further demonstrating its usefulness as a hemodynamic monitoring tool in the ED to guide therapy titration.56
Lung ultrasound also has been shown to have high sensitivity, positive predictive value, and negative predictive value compared to chest X-ray for the diagnosis of acute pneumonia.57 A recent meta-analysis demonstrated 94% sensitivity and 96% specificity.58 Lung ultrasound has long been shown to have superior accuracy in diagnosing pneumothorax, with 91% sensitivity and 98% specificity.48,49,50,53,54,55,59,60
Bedside ultrasound can be a great adjunctive noninvasive hemodynamic monitoring tool. Individually, all of the above modalities have been described in detail. However, when a clinician becomes more facile with bedside ultrasound, these modalities may be used in concert with each other (IVC, FOCUS, lung ultrasound, VTI, etc.). At this point, physicians can monitor their patients noninvasively as well as have better understanding of the pathophysiology particular to their patient. This can aid in hemodynamic monitoring and diagnostics, as well as in guiding therapeutic interventions.
Conclusion
Hemodynamic monitoring is of vital importance in managing and caring for critically ill patients in the ED. During the initial clinical evaluation of the patient in most EDs, it is commonplace to have frequent monitoring of heart rate, blood pressure (both invasively and noninvasively), respiratory rate, oxygen saturations, and temperature. Understanding the multitude of different, current hemodynamic monitoring options to guide appropriate therapy is paramount in managing critically ill patients presenting to the ED.
A common initial approach is one that includes clinical assessment, basic bedside monitoring, and point-of-care ultrasonography coupled with a PLR maneuver and/or a fluid bolus trial. However, in today’s practice, it is vital for the ED clinician to be knowledgeable with regard to the breadth of advanced and complex hemodynamic monitoring techniques, which may be available and even necessary in patients with complex presentations as well as in EDs where boarding critically ill patients is standard practice.1
REFERENCES
- Laher AE, Watermeyer MJ, Buchanan SK, et al. A review of hemodynamic monitoring techniques, methods and devices for the emergency physician. Am J Emerg Med 2017 Mar 18. [Epub ahead of print.]
- Marik PE, Cavallazzi R. Does the central venous pressure predict fluid responsiveness? An updated meta-analysis and a plea for some common sense. Crit Care Med 2013;41:1774-1781.
- Siniscalchi A, Pavesi M, Priaccini E, et al. Right ventricular end-diastolic volume index as a predictor of preload status in patients with low right ventricular ejection fraction during orthotopic liver transplantation. Transplant Proc 2005;37:2541-2543.
- Argueta E, Berdine G, Pena C, Nugent KM. FloTrac® monitoring system: What are its uses in critically III medical patients? Am J Medical Sciences 2015;349:352-356.
- Button D, Weibel L, Reuthebuch O, et al. Clinical evaluation of the FloTrac/Vigileo system and two established continuous cardiac output monitoring devices in patients undergoing cardiac surgery. Br J Anaesth 2007;99:329-336.
- Reid RD, Jayamaha J. The use of a cardiac output monitor to guide the initial fluid resuscitation in a patient with burns. Emerg Med J 2007;24:e32.
- Michard F. Changes in arterial pressure during mechanical ventilation. Anesthesiology 2005;103:419-428.
- Yang X, Du B. Does pulse pressure variation predict fluid responsiveness in critically ill patients? A systematic review and meta-analysis. Crit Care 2014;18:1-23.
- Delannoy B, Wallet F, Maucort-Boulch D, et al. Applicability of pulse pressure variation during unstable hemodynamic events in the intensive care unit: A five-day prospective multicenter study. Crit Care Res Pract 2016 [Epub 2016 Mar 31].
- Bighamian R, Hahn JO. Relationship between stroke volume and pulse pressure during blood volume perturbation: A mathematical analysis. BioMed Res Int 2014;2014;459269.
- Evans D, Ferraioli G, Snellings J, Levitov A. Volume responsiveness in critically ill patients: Use of sonography to guide management. J Ultrasound Med 2014;33:3-7.
- Mackenzie DC, Nobel VE. Assessing volume status and fluid responsiveness in the emergency department. Clin Exp Emerg Med 2014;1:67-77.
- Marik PE, Monnet X, Teboul JL. Hemodynamic parameters to guide fluid therapy. Ann Intensive Care 2011;1:1-9.
- Miller J, Ho CX, Tang J, et al. Assessing fluid responsiveness in spontaneously breathing patients. Acad Emerg Med 2016;23:186-190.
- Beigel R, Cercek B, Arsanjani R, Siegel RJ. Echocardiography in the use of noninvasive hemodynamic monitoring. J Crit Care 2014;29:184.e1-8.
- Cherpanath TG, Hirsch A, Geerts BF, et al. Predicting fluid responsiveness by passive leg raising: A systematic review and meta-analysis of 23 clinical trials. Crit Care Med 2016;44:981-991.
- Bentzer P, Griesdale DE, Boyd J, et al. Will this hemodynamically unstable patient respond to a bolus of intravenous fluids? JAMA 2016;316:1298-1309.
- Krauss B, Falk JL. Carbon dioxide monitoring (capnography). Up To Date. Available at: http://bit.ly/2v8Ix0k. Accessed July 4, 2017.
- Galvagno SM Jr, Kodali BS. Use of capnography in emergency medicine and prehospital critical care. Available at: http://www.capnography.com/new/outside-or/emergency-medicine. Accessed July 26, 2017.
- Radwine Z. Capnography in the ED. emdocs.net. Aug. 26, 2014. Available at: http://www.emdocs.net/capnography-ed/. Accessed July 5, 2017.
- Meaney PA, Bobrow BJ, Mancini ME; CPR Quality Summit Investigators, the American Heart Association Emergency Cardiovascular Care Committee, and the Council on Cardiopulmonary Critical Care, Perioperative and Resuscitation. Cardiopulmonary resuscitation quality: Improving cardiac outcomes both inside and outside the hospital: A consensus statement from the American Heart Association. Circulation 2013;128:417-435.
- Neumar RW, Otto CW, Link MS, et al. Part 8: Adult advanced cardiovascular life support: 2010 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 2010;122(18 Suppl 3):S729-S767.
- Kodali BS, Urman RD. Capnography during cardiopulmonary resuscitation: Current evidence and future directions. J Emerg Trauma Shock 2014;7:332-340.
- Hartmann SM, Farris RW, Di Gennaro JL, Roberts JS. Systematic review and meta-analysis of end-tidal carbon dioxide values associated with return of spontaneous circulation during cardiopulmonary resuscitation. J Intensive Care Med 2015;30:426-435.
- Kodali BS. Capnography outside the operating rooms. Anesthesiology 2013;118:192-201.
- Krauss BS, Andolfatto G, Krauss BA, et al. Characteristics of and predictors for apnea and clinical interventions during procedural sedation. Ann Emerg Med 2016;68:564-573.
- Godwin SA, Burton JH, Gerardo CJ, et al; American College of Emergency Physicians. Clinical Policy: Procedural sedation and analgesia in the emergency department. Ann Emerg Med 2014;63:247-258.
- Toupin F, Clairoux A, Deschamps A, et al. Assessment of fluid responsiveness with end-tidal carbon dioxide using a simplified passive leg raising maneuver: A prospective observational study. Can J Anesth 2016;63:1033-1041.
- Monge García MI, Gil Cano A, Gracia Romero M, et al. Non-invasive assessment of fluid responsiveness by changes in partial end-tidal CO2 pressure during a passive leg-raising maneuver. Ann Intensive Care 2012;2:9.
- Zhang Z, Xu X, Ye S, Xu L. Ultrasonographic measurement of the respiratory variation in the inferior vena cava diameter is predictive of fluid responsiveness in critically ill patients: Systematic review and meta-analysis. Ultrasound Med Biol 2014;40:845-853.
- Eskesen TG, Wetterslev M, Perner A. Systematic review including re-analyses of 1148 individual data sets of central venous pressure as a predictor of fluid responsiveness. Intensive Care Med 2016;42:324-332.
- Corl KA, George NR, Romanoff J, et al. Inferior vena cava collapsibility detects fluid responsiveness among spontaneously breathing critically-ill patients. J Crit Care 2017;12:130-137.
- Long E, Oakley E, Duke T, Babl FE; Paediatric Research in Emergency Departments International Collaborative (PREDICT). Does respiratory variation in inferior vena cava diameter predict fluid responsiveness: A systematic review and meta-analysis. Shock 2017;47:550-559.
- Dawson M, Mallin MD. (2012). Introduction to Bedside Ultrasound: Volume 1. Lexington, KY; Emergency Medicine Solutions: 2012.
- Mikkelsen ME, Gaieski DF, Johnson NJ. Novel tools for hemodynamic monitoring in critically ill patients with shock. from UpToDate. Dec. 20, 2016. Available at: http://bit.ly/2wpTfNn. Accessed June 15, 2017.
- Middleton PM, Davies SR. Noninvasive hemodynamic monitoring in the emergency department. Curr Opin Crit Care 2011;17:342-350.
- Au SM, Vieillard-Baron A. Bedside echocardiography in critically ill patients: A true hemodynamic monitoring tool. J Clin Monit Comput 2012;26:355-360.
- Mok KL. Make it SIMPLE: Enhanced shock management by focused cardiac ultrasound. J Intensive Care 2016;4:51.
- Au SM, Vieillard-Baron A. A. Bedside echocardiography in critically ill patients: A true hemodynamic monitoring tool.
J Clin Monit Comput 2012;26:355-360. - Au SM, Vieillard-Baron A. Bedside echocardiography in critically ill patients: A true hemodynamic monitoring tool. J Clin Monit Comput 2012;26:355-360.
- Arntfield RT, Millington SJ. Point of care cardiac ultrasound applications in the emergency department and intensive care unit — a review. Curr Cardiol Rev 2012;8:98-108.
- Porter TR, Shillcutt SK, Adams MS, et al. Guidelines for the use of echocardiography as a monitor for therapeutic intervention in adults: A report from the American Society of Echocardiography. J Am Soc Echocardiogr 2015;28:40-56.
- Noritomi DT, V. M. Echocardiography for hemodynamic evaluation in the intensive care unit. Shock 2010;34 Suppl 1:59-62.
- Ünlüer EE, KaragÖz A, Akoğlu H, Bayata S. Visual estimation of bedside echocardiographic ejection fraction by emergency physicians. West J Emerg Med 2014;15:221-226.
- Rutz MA, Clary JM, Kline JA, Russell FM. Emergency physicians are able to detect right ventricular dilation with good agreement compared to cardiology. Acad Emerg Med 2017;24:867-874.
- Dinh VA, Ko HS, Rao R, et al. Measuring cardiac index with a focused cardiac ultrasound examination in the ED. Am J Emerg Med 2012;30:1845-1851.
- Arntfield R, Pace J, Hewak M, Thompson D. Focused transesophageal echocardiography by emergency physicians is feasible and clinically influential: Observational results from a novel ultrasound program. J Emerg Med 2016;50:286-294.
- Lichtenstein D, Karakitsos D. Integrating lung ultrasound in the hemodynamic evaluation of acute circulatory failure (the fluid administration limited by lung sonography protocol). J Crit Care 2012;27:533.e11-9.
- Lichtenstein DA. BLUE-protocol and FALLS-protocol: Two applications of lung ultrasound in the critically ill. Chest 2015;147:1659-1670.
- Whitson MR, Mayo PH. Ultrasonography in the emergency department. Crit Care 2016;20:227.
- Ma OJ, Mateer JR, Reardon RF, Joing SA. Ma and Mateer’s Emergency Ultrasound, third ed. McGraw-Hill Education; 2013.
- Anile A, Russo J, Castiglione G, Volpicelli G. A simplifed lung ultrasound approach to detect increased extravascular lung water in critically ill patients. Crit Ultrasound J 2017;9:1-7.
- Al Deeb M, Barbic S, Featherstone R, et al. Point-of-care ultrasonography for the diagnosis of acute cardiogenic pulmonary edema in patients presenting with acute dyspnea: A systematic review and meta-analysis. Acad Emerg Med 2014;21:843-852.
- Chiem AT, Chan CH, Ander DS, et al. Comparison of expert and novice sonographers’ performance in focused lung ultrasonography in dyspnea (FLUID) to diagnose patients with acute heart failure syndrome. Acad Emerg Med 2015;22:564-573.
- Martindale JL, Wakai A, Collins SP, et al. Diagnosing acute heart failure in the emergency department: A systematic review and meta-analysis. Acad Emerg Med 2016;23:223-242.
- Cortellaro F, Ceriani E, Spinelli M, et al. Lung ultrasound for monitoring cardiogenic pulmonary edema. Intern Emerg Med 2016 Jul 29. [Epub ahead of print.]
- Pagano A, Numis FG, Visone G, et al. Lung ultrasound for diagnosis of pneumonia in emergency department. Intern Emerg Med 2015;10:851-854.
- Chavez MA, Shams N, Ellington LE, et al. Lung ultrasound for the diagnosis of pneumonia in adults: A systematic review and meta-analysis. Respir Res 2014;15:50.
- Pivetta E, Goffi A, Lupia E, et al; SIMEU Group for Lung Ultrasound in the Emergency Department in Piedmont. Lung ultrasound-implemented diagnosis of acute decompensated heart failure in the ED: A SIMEU multicenter study. Chest 2015;148:202-210.
- Alrajhi K, Woo MY, Vaillancourt C. Test characteristics of ultrasonography for the detection of pneumothorax: A systematic review and meta-analysis. Chest 2012;141:703-708.
Hemodynamic monitoring is a very important component in effectively resuscitating critically ill patients. Various methods of hemodynamic monitoring not only give the physician an idea of the patient’s overall pathophysiology, but can alert the physician to acute changes.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
