Management of Skin and Soft Tissue Infections
November 1, 2017
Reprints
AUTHORS
Raquel M. Schears, MD, MPH, MBA, FACEP, Brandeis University, The Heller School for Social Policy and Management, Executive MBA for Physicians Program, Waltham, MA
Catherine A. Marco, MD, FACEP, Professor, Department of Emergency Medicine, Wright State University Boonshoft School of Medicine, Dayton, OH
PEER REVIEWER
Steven M. Winograd, MD, FACEP, Queens Hospital Center, Jamaica, NY; Core Faculty, Attending Emergency Physician
EXECUTIVE SUMMARY
Soft tissue infections, ranging from uncomplicated cellulitis, impetigo, folliculitis, erysipelas, and focal abscesses to necrotizing fasciitis, commonly are encountered in ambulatory practices.
- About 650,000 patients are hospitalized each year, with 14.5 million cases annually treated as outpatients, accounting for $3.7 billion in ambulatory care costs.
- Blood cultures and skin cultures have a low rate of positivity and low impact on treatment decisions. Tissue cultures have low yield and little effect on clinical decision-making.
- Empiric treatment of primary cellulitis typically is initiated with antibiotics effective against the two most common pathogens: beta-hemolytic streptococci and methicillin-sensitive Staphylococcus aureus.
- Infectious Diseases Society of America guidelines guide empiric therapy. Purulent cellulitis should be treated with 5-10 days of antibiotics, such as clindamycin, trimethoprim-sulfamethoxazole, doxycycline, minocycline, or linezolid, to cover methicillin-resistant Staphylococcus aureus. Oral therapy is just as effective as intravenous treatment for uncomplicated cellulitis.
Skin and soft tissue infections (SSTIs) are encountered commonly in primary care practices, presenting as a range of disorders, from uncomplicated cellulitis, impetigo, folliculitis, erysipelas, and focal abscesses to necrotizing fasciitis. Each year between 1998-2006 in the United States, there were 650,000 hospital admissions for cellulitis, with estimates of 14.5 million cases annually treated as outpatients, accounting for $3.7 billion in ambulatory care costs.1,2
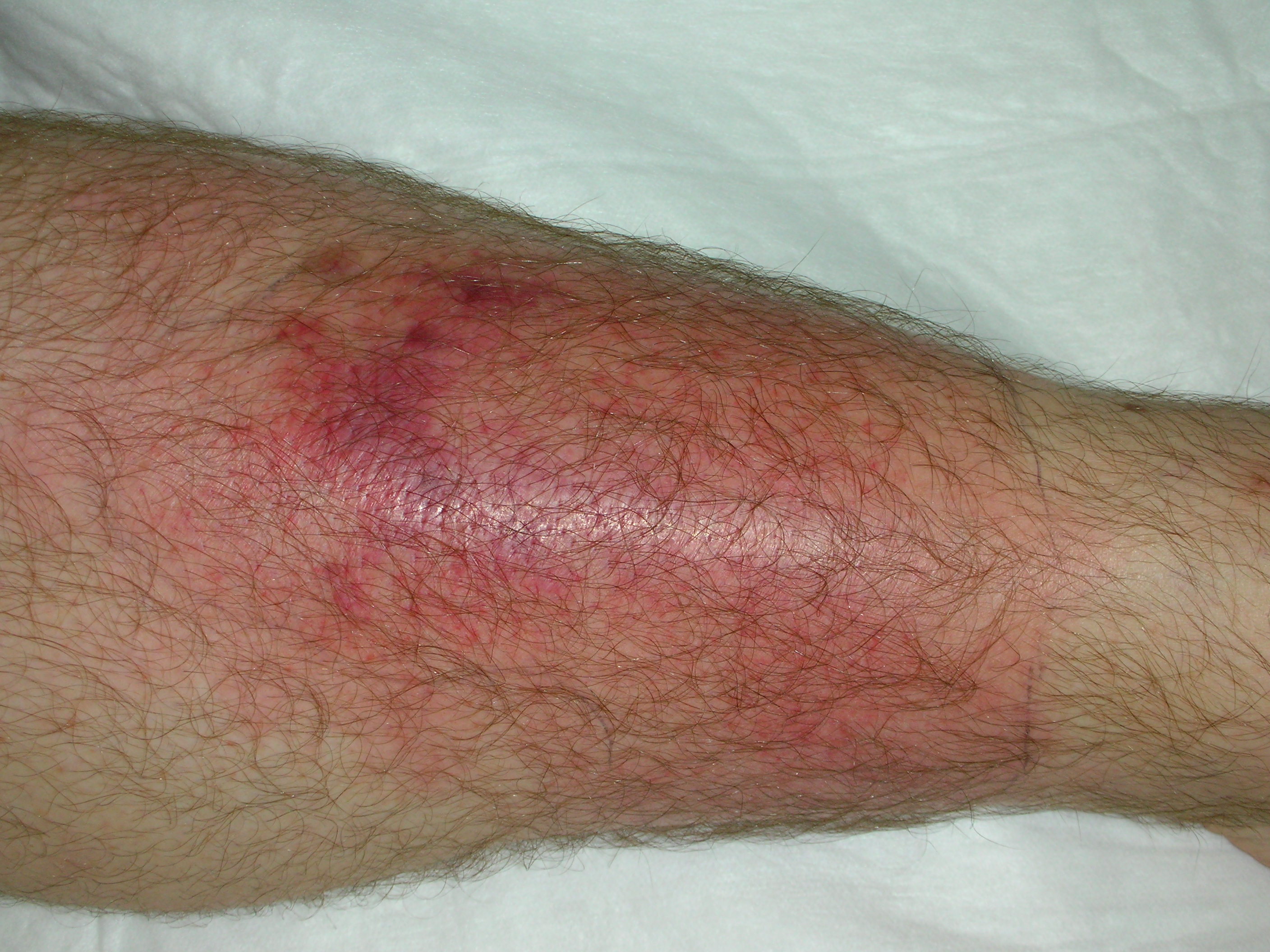
Patients with simple cellulitis typically present with an area of expanding erythema that is warm and tender to touch. (See Figure 1.) Other features may include swelling and fluid drainage. The diagnosis of cellulitis is made primarily by history and physical findings, and differentiated from other skin and soft tissue findings by appearance. (See Table 1.) Diagnostic modalities may be indicated to reliably discern cellulitis from other more serious infections or a true emergency, such as necrotizing cellulitis. Once the diagnosis of cellulitis is made, empiric treatment should be instituted based on common pathogens. The usual pathogenic organisms include beta-hemolytic streptococci and methicillin-sensitive Staphylococcus aureus (MSSA).1 Failure to respond to appropriate first-line antibiotics within five days of initiating therapy may indicate resistant organisms, polymicrobial infection, pseudocellulitic mimicry, underlying immunosuppression, or a deep space infection not treatable with oral antibiotics.
Figure 1. Cellulitis |
 |
|
Image courtesy of J. Stephan Stapczynski, MD. |
Table 1. Clinical Findings of Skin Infections |
|
|
Skin Infection |
Clinical Findings |
|
Cellulitis |
Localized erythema, induration, warmth |
|
Impetigo |
Erythema, yellow or brown crusting; possible bullae |
|
Erysipelas |
Well-demarcated erythema with raised border, often facial; may have fever and systemic illness |
|
Ecthyma |
Erythema with dermal and epidermal erosions |
|
Folliculitis |
Papules, pustules associated with hair follicles |
|
Furuncle |
Erythema, fluctuance, pustules associated with hair follicles |
|
Abscess |
Erythema, warmth, fluctuance |
Although generally of little concern for patients with cellulitis and other superficial skin infections, the initial evaluation of patients presenting for care and treatment of skin infections consists of assessment and stabilization of life and limb threats that begins with assessment and management of airway, breathing, and circulation. In all patients with skin and soft tissue infections, assessment of circulation in the involved region is important. The capillary refill should be checked in the involved area and, if the infection is on a limb, the arterial pulses above and below the area should be checked. If pulses cannot be palpated, Doppler assessment of flow is advised. Absent palpable pulses and Doppler flow indicate potential limb-threatening ischemia. A vascular surgeon should be consulted and additional imaging studies, such as computed tomography (CT) angiography, typically are done.
After assessing arterial inflow, look for signs suggestive of impaired venous outflow, such as dependent edema, stasis dermatitis, and prominent varicose veins on the legs. Venous insufficiency may mimic cellulitis and impair the response to antibiotic therapy.
Physicians should consider diseases and injuries that are the most serious and treatable. Because cellulitis can be challenging to diagnose properly, hospital admission and further clinical deliberation may be prudent in cases of uncertain diagnosis.3 Because cellulitis generally is more responsive to treatment than other similar conditions (drug reactions, venous stasis, which may give a clue in their bilaterality), physicians often err on the side of initiating empiric treatment even when the diagnosis is uncertain.4 They also may admit patients with cellulitis to the hospital to monitor for worsening of the condition. However, the practice of hospitalizing patients provisionally diagnosed with cellulitis to validate the diagnosis and observe the response to empiric treatment has been questioned recently.5,6
Empiric treatment of primary cellulitis starts with antibiotics effective against the two most common pathogens: beta-hemolytic streptococci and MSSA. Patients who have recurrent cellulitis or do not respond to initial therapy should be treated with antibiotics that have an expanded spectrum to include methicillin-resistant Staphylococcus aureus (MRSA). Consider expanded antibiotic spectrum to include MRSA in patients at risk for this pathogen, such as athletes, children, military recruits, intravenous drug abusers, prisoners, men who have sex with men, nursing home residents, and those with prior MRSA exposure.1,8,14
Epidemiology and Risk Factors
Impetigo occurs in about 20/1,000 people per year, and erysipelas occurs in about 1/1,000 people per year.9 Despite this high frequency, there are limited data on the frequency with which specific bacteria are associated with these infections.
Impetigo is a skin infection that is ubiquitous worldwide. It occurs most frequently among economically disadvantaged children in tropical or subtropical regions, but also is common in northern climates during summer months, and all epidemics show this seasonal proclivity.9 The incidence of impetigo is highest in children younger than 5 years of age, followed by children 5 to 14 years of age; the incidence then decreases rapidly in those between 14 and 44 years of age, and is at a minimum among the elderly (> 65 years of age).
Erysipelas affects mostly adults in their 60s and 70s and involves the lower extremities in more than 80% of cases.10 The incidence of lower-extremity cellulitis in Olmsted County, MN, was estimated at 2/1,000 people per year when both erysipelas and cellulitis affecting the leg were considered.11 Predisposing factors are well identified for erysipelas or cellulitis of the leg, including disruption of the cutaneous barrier (leg ulcer, wound, toe-web intertrigo, pressure ulcer), lymphedema, chronic edema, or local surgical operations. Toe-web intertrigo appears to be the main portal of entry with or without dermatophytes being coincident. Lymphedema is a very strong risk factor, but any type of edema is both a risk factor and a consequence of the disease.12,13 Other comorbidities, such as obesity or diabetes, and prior history of cellulitis are surprisingly less compelling as risk factors.
To date there are no consistent data on the prevalence or incidence of folliculitis and furunculosis in the community. However, there are reports of some individuals with repeated attacks of furunculosis. For most of these patients, the only identifiable predisposing factor is the carriage of S. aureus in the anterior nares. Twenty to forty percent of the general population harbors staphylococcal colonies in the nares.
There is literature on longitudinal outbreaks of furunculosis caused by MSSA as well as by MRSA.21,14 Risk factors for outbreaks include being part of a small community (i.e., family groups) and other settings involving close personal contact (i.e., athletics), inadequate hygiene, and exposure to others having furuncles.
Researchers in Chicago looked at community-acquired (CA)-MRSA infections, which mainly manifested clinically as abscesses. These investigators identified risk factors including incarceration, African-American race/ethnicity, and residing in the close quarters of geocentric public housing complexes, and found an inverse correlation among the elderly residents of these urban settings.15
As noted above, outbreaks of epidemic furuncles due to CA-MRSA occur in young otherwise healthy people. In many U.S. cities, MRSA has become the most common pathogen isolated in the ED from patients with SSTIs.16,20 Bacterial endocarditis in patients with CA-MRSA furunculosis has emerged as a complication, but unfortunately there are no large, community-based prospective studies to assess the risk of endocarditis from MRSA SSTIs.
Clinical Findings of Cellulitis
Of the common bacterial skin infections, impetigo is the most common in children worldwide. It consists of superficial, nonfollicular, discrete, purulent lesions that are caused by S. aureus or beta-hemolytic streptococci and may have a honey-crusted, pustular, or cornflake-like appearance.
Boils (furuncles) are infections of the hair follicle and often are caused by S. aureus. Therefore, they may have suppuration that extends to the deep dermis, where small abscess pockets can develop. Furuncles differ from folliculitis, which is more superficial as pus remains within the epidermis.
Erysipelas is an acute, superficial, non-necrotizing, dermal/deep dermal infection that is caused mainly by beta-hemolytic streptococci of group A, but also group B, C, and G, and rarely by staphylococci.16 The clinical diagnosis is based on findings of sharply demarcated, shiny erythematous plaques that appear suddenly and are tender to the touch, with associated local swelling and fever.
Cellulitis is a skin infection that extends deeper into the subcutaneous tissues than erysipelas. These cellulitis infections may have associated regional lymphadenopathy and red streaking, and are more prone to local complications (abscess formation, necrosis) than is seen with erysipelas. With more of the skin loft (depth) involved, petechiae, ecchymoses, and bullae develop in the inflamed skin, which may result in a hemorrhagic appearance.17 Bulla formation is a frequent local complication of the disease and may be indicative of greater severity of infection.
If untreated, these superficial SSTIs can lead to regional and systemic complications. Consider the presentation of the toxic adolescent patient describing what he perceives to be superficial severe skin pain overlying a limb but not involving a nearby joint. He explains that he plays hockey and a month ago sustained an injury to his knee area from an opponent’s skate that cut through his knee pad. The wound hardly bled and healed by secondary intention without additional medical attention. On exam, the skin surface appears undamaged and completely normal, yet the slightest touch causes significant pain. This exam is consistent with pain out of proportion to the exam, and other etiologies should be considered, such as thrombophlebitis, osteomyelitis, or deep space infection. There are well-described cases of septic thrombophlebitis with acute osteomyelitis19 and endocarditis that can complicate the aftermath of SSTIs.18
Other bacterial associations with skin infection include Pasteurella multocida following cat or dog bites, Vibrio species after salt water exposure, Aeromonas hydrophila following immersion in fresh water, Haemophilus influenzae in periorbital cellulitis in children, or, rarely, epiglottitis in adults.
As another example, natural disasters such as the massive tsunami that struck southern Thailand on December 26, 2004, provide perhaps the greatest experience in managing acute SSTIs following aquatic injuries. Soon after the wave decimated the coastline, Hiransuthikul et al reported acute SSTIs in 515 of 777 (66.3%) patients with crush injuries to the legs transferred to four regional referral hospitals in Bangkok.19 Wounds and/or pus were cultured from 396 (76.9%) of these patients. The majority of specimens were polymicrobial (71.8%), and the most common organisms isolated were Gram-negative bacteria, including Aeromonas species (22.6%), Escherichia coli (18.1%), Klebsiella pneumoniae (14.5%), Pseudomonas aeruginosa (12.0%), and Proteus species (7.3%). Only 4.5% of the isolates were Gram-positive bacteria, most commonly staphylococcal species.19
Another group of investigators reported on a subset of European tsunami survivors who developed late-onset (median 60 days) chronic post-traumatic SSTI caused by rapidly growing marine mycobacteria.20 These infections occurred in undamaged skin located near sutured traumatic wounds or skin grafts. These patients required antimycobacterial therapy, and healing was protracted, occurring within a year in most cases.
The forgoing cases demonstrate that a history of injury should be elicited from the patient, and the possibility of interval healing must be considered. Physicians need to think about the possibility of a retained foreign body, slow-growing organisms, and the potential for deeper tissue contamination dating back to an index event even a year out. In addition, the emerging problem of CA-MRSA, which is responsible for fully half of the cases of cellulitis with purulent exudates, also causes infections in patients lacking typical risk factors (hospitalization, long-term care factors).21
Necrotizing fasciitis represents a severe soft tissue infection with associated high morbidity and mortality.22 Risk factors include diabetes mellitus, injection drug use, hypertension, and obesity. Necrotizing fasciitis may present with significant pain, erythema, ecchymosis, purulence, and/or crepitus caused by subcutaneous air. Systemic signs of sepsis may be present. The most commonly infected sites include the perineum, lower limbs, upper limbs, and axillary region.23
Prompt recognition of necrotizing fasciitis is essential to ensure timely administration of broad-spectrum antibiotics and surgical debridement.24 Several studies have identified predictors of mortality, which may include advanced age, female gender, signs of sepsis or septic shock, obesity, diabetes, chronic heart disease, cirrhosis, skin necrosis, tachycardia, serum creatinine, hypertension, multifocal infection, severe peripheral vascular disease, hospital-acquired infection, and polymicrobial or Gram-negative infections.25,26,27,28
Diagnostic Tests
SSTIs are diagnosed primarily by clinical findings. Cellulitis typically is a clinical diagnosis, supported by the clinical findings of erythema, warmth, edema, and pain. An abscess is supported by the findings of localized erythema, swelling, and fluctuance. Unilateral findings are most suggestive of cellulitis as compared to venous stasis, which often is bilateral.
Individual laboratory tests have limited value in establishing the diagnosis of cellulitis. Multifactorial prediction tools incorporating laboratory tests may be more useful. Such prediction tools have been developed for lower extremity cellulitis and necrotizing fasciitis.
Researchers recently developed a diagnostic prediction tool for lower extremity cellulitis from 259 patients admitted from the ED for presumed cellulitis.29 (See Table 2.) Ultimately, 180 (69.5%) of the patients were diagnosed with cellulitis, while 79 (30.5%) were diagnosed with other disorders, termed pseudocellulitis. The derived decision tool, named ALT-70, had a score range from 0 to 7. The higher the score, the more likely the patient had cellulitis. The authors divided the patients into three categories based on the score, along with a recommended course of action. Patients with a score of 0 to 2 had a negative predictive value of 83%, so the recommended course of action was reassess. Patients with scores of 3 and 4 had indeterminant predictive values, so the recommended course of action was to consult a specialist, such as a dermatologist, who could perform skin biopsy and other tests. Patients with scores of 5 to 7 had a positive predictive value > 82%, so the recommended course of action was to treat for cellulitis with antibiotics.
Table 2. ALT-70 Prediction Tool for Lower Extremity Cellulitis |
|
|
Factor |
Score |
|
Asymmetry: unilateral leg involvement |
3 |
|
Leukocytosis: white blood cell count ≥ 10,000 |
2 |
|
Tachycardia: heart rate ≥ 90 beats per minute |
2 |
|
Age ≥ 70 years |
2 |
|
With an ALT-70 score of 0 to 2, the positive likelihood ratio (LR+) is 1.21 and the negative likelihood ratio (LR-) is 0.09. With an ALT-70 score of 5 to 7, the LR+ is 2.10 and the LR- is 0.55. |
|
Elevated C-reactive protein (CRP) levels may be seen in necrotizing fasciitis.30 The Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) is a score that may be used to identify patients at higher risk for necrotizing fasciitis. The LRINEC is comprised of serum sodium, glucose, creatinine, CRP, leukocyte count, and hemoglobin.31 LRINEC scores range from 0 to 13, with higher scores associated with increased risk of necrotizing fasciitis. The authors who developed the score proposed threshold values of 6 for moderate (50% to 75%) risk of necrotizing fasciitis and a score 8 for high (> 75%) risk of necrotizing fasciitis. (See Table 3.)
Table 3. LRINEC Prediction Tool for Necrotizing Fasciitis |
||
|
Factor |
Value |
Score |
|
Serum sodium |
≥ 135 mEq/L |
0 |
|
< 135 mEq/L |
2 |
|
|
Serum glucose |
> 180 mg/dL |
1 |
|
≤ 180 mg/dL |
0 |
|
|
Serum creatinine |
> 1.6 mg/dL |
2 |
|
≤ 1.6 mg/dL |
0 |
|
|
C-reactive protein |
< 150 |
0 |
|
≥ 150 |
4 |
|
|
Leukocyte count |
< 15,000 |
0 |
|
15,000 to 25,000 |
1 |
|
|
> 25,000 |
2 |
|
|
Hemoglobin |
> 13.5 g/dL |
0 |
|
11.0 to 13.5 g/dL |
1 |
|
|
< 11.0 g/dL |
2 |
|
A systematic review of 16 studies with a total of 846 patients found the mean LRINEC score was 6.06 for patients with necrotizing fasciitis and 2.45 for patients without.31 The receiver operating characteristic (ROC) curve was calculated using LRINEC scores; the fitted ROC area was 0.927, indicating the LRINEC was an excellent test to discriminate patients with necrotizing fasciitis from those without.
The LRINEC has been criticized as lacking adequate sensitivity for excluding patients with low scores. Both prospective and retrospective studies have found that about 20% to 30% of patients with necrotizing fasciitis will have LRINEC scores of 5 or less.31,32
Blood cultures have a low rate of positivity, a low rate of impact on clinical decision making, and a high false-positive rate, and are not recommended routinely for the treatment of cellulitis, even in febrile patients.33,34,35,36,37 Similarly, cultures of the soft tissues are low yield and of limited usefulness in clinical decision making.38,39
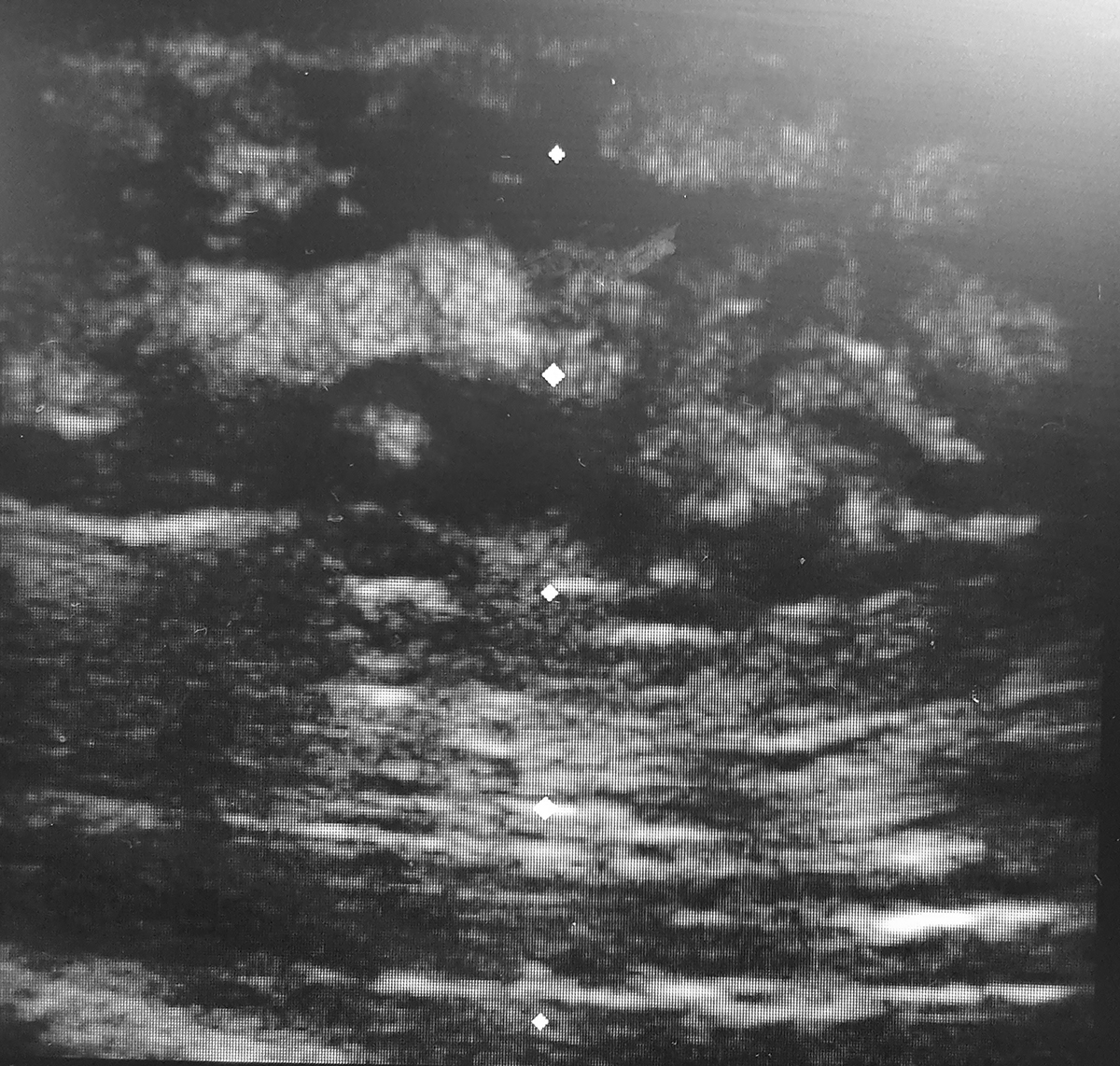
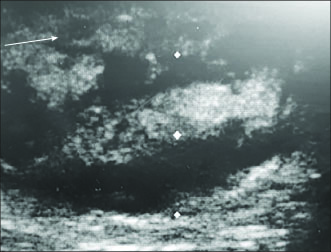
Several recent studies have demonstrated the utility of bedside ultrasound in clinical decision making, with findings that may result in a change in management.40,41 Ultrasound is of value in differentiating cellulitis from abscess.42,43 (See Figures 2 and 3.) Ultrasound also may be of particular use in the evaluation of hand infections44 and may help to identify foreign bodies. Sonographic evidence of necrotizing fasciitis may include subcutaneous air, abscess, edematous soft tissues, and/or muscle necrosis.45
Figure 2. Ultrasonographic Appearance of Subcutaneous Abscess |
 |
|
Note sonographic appearance of fluid-filled sac (dark appearance). |
Figure 3. Bedside Ultrasound Appearance of Cellulitis Demonstrating “Cobblestoning” Appearance of Subcutaneous Edema |
 |
CT or magnetic resonance imaging (MRI) may be indicated for anatomic areas of concern for vascular or other anatomic involvement, such as the face, neck, pelvis, or perineum, or if necrotizing fasciitis or other deep space infection is suspected.46,47,48 Plain radiographs are of limited utility unless there is a history of trauma.49 Plain radiographs may be used to screen for radiopaque foreign bodies, osteomyelitis, or soft tissue gas suggestive of suspected deep space necrotizing infections. However, radiographs lack sufficient sensitivity to be the sole test for any of these conditions.
Differential Diagnosis
Several conditions may mimic cellulitis. Venous stasis may present with edema and erythema, but most commonly is bilateral and may be associated with chronic skin changes, such as hyperpigmentation, lichenification, or ulcerations. Deep venous thrombosis may present with unilateral pain, swelling, and erythema, and may be differentiated from cellulitis by ultrasonography of the lower extremities. An abscess may be distinguished from cellulitis by ultrasonographic appearance.
Empiric Treatment
Empiric treatment is indicated for cellulitis, as the precise pathogen rarely is known. Tetanus immunization should be updated if not current.
Guidelines from the Infectious Diseases Society of America may be used to guide empiric therapy. (See Table 4.) Purulent cellulitis should be treated with 5-10 days of antibiotics, such as clindamycin, trimethoprim-sulfamethoxazole, doxycycline, minocycline, or linezolid, to cover MRSA. Nonpurulent cellulitis should be treated with 5-10 days of antibiotics, such as clindamycin, or linezolid, or combination therapy with doxycycline or minocycline, plus a beta-lactam/beta-lactamase inhibitor combination such as amoxicillin/clavulanate, to cover beta-hemolytic streptococci.50 Oral therapy is as efficacious as intravenous therapy for uncomplicated cellulitis.51 (See Table 5.)
Table 4. Empiric Treatment of Skin and Soft Tissue Infections |
||
|
Conditions |
Suggested Antibiotic |
Duration |
|
Purulent cellulitis |
Clindamycin, trimethoprim-sulfamethoxazole, doxycycline, minocycline, or linezolid |
5-10 days |
|
Nonpurulent cellulitis |
Clindamycin or linezolid, or combination therapy with doxycycline or minocycline, plus a beta-lactam, such as amoxicillin-clavulanate |
5-10 days |
|
Complicated cellulitis in hospitalized patients |
Intravenous vancomycin, oral or intravenous linezolid, intravenous daptomycin, intravenous telavancin, or intravenous or oral clindamycin |
7-10 days |
|
Impetigo |
Topical mupirocin or retapamulin, or oral therapy to include dicloxacillin or cephalexin |
5 days |
|
Ecthyma |
Dicloxacillin or cephalexin |
7 days |
|
Necrotizing fasciitis |
Vancomycin or linezolid plus piperacillin-tazobactam or a carbapenem Alternatives may include ceftolozane-tazobactam, ceftazidime-avibactam in association with an anti-anaerobic agent (metronidazole or clindamycin) |
|
Table 5. Antibiotic Doses for Outpatient Treatment |
|||
|
Drug |
Adult Dose |
Pediatric Dose |
Comments |
|
Penicillin VK |
250-500 mg QID PO |
25-75 mg/kg per day in 4 divided doses PO |
For nonpurulent cellulitis |
|
Amoxicillin-clavulanate |
875/125 mg BID PO |
25 mg/kg per day of the amoxicillin component in 2 divided doses PO |
For nonpurulent cellulitis and impetigo |
|
Cephalexin |
250-500 mg QID PO |
25-50 mg/kg per day in 4 divided doses PO |
Use in penicillin-allergic patients except those with immediate hypersensitivity reactions |
|
Dicloxacillin |
250-500 mg QID PO |
25-50 mg/kg per day in 4 divided doses PO |
Agent of choice in adults with methicillin-susceptible Staphylococcus aureus (MSSA) |
|
Clindamycin |
300-450 mg QID PO |
25-30 mg/kg per day in 3 divided doses PO |
Important option for methicillin-resistant Staphylococcus aureus (MRSA) treatment children |
|
Doxycycline |
200 mg once PO, followed by 100 mg BID PO |
Not recommended for age |
Limited recent clinical experience |
|
Minocycline |
200 mg once PO, followed by 100 mg BID PO |
Not recommended for age |
Limited recent clinical experience |
|
Trimethoprim-sulfamethoxazole |
1-2 double strength tablets BID PO Each tablet: |
8-12 mg/kg per day based on trimethoprim component in |
For purulent cellulitis |
|
Linezolid |
600 mg BID PO |
< 5 years old: 30 mg/kg per day in 3 divided doses PO 5-11 years old: 20 mg/kg per day in 2 divided doses PO ≥ 12 years old: 600 mg BID PO |
Use for MRSA and complicated purulent skin infection Retail price for 600 mg # 28 tablets about $3,000-4,000, but significant discounts available |
|
Mupirocin 2% ointment |
Apply to lesions BID |
Apply to lesions BID |
Retail price for 22 gram tube about $52 |
|
Retapamulin 1% ointment |
Apply to lesions BID |
Apply to lesions BID |
Retail price for 15 gram tube about $250 |
Risk factors for failure of outpatient therapy include fever, chronic leg ulcers, chronic edema or lymphedema, prior cellulitis in the same area, and cellulitis of a wound site.52
Complicated cellulitis in hospitalized patients should be treated with broad-spectrum antibiotics, including empiric therapy for MRSA. Antibiotic choices may include intravenous vancomycin, oral or intravenous linezolid, intravenous daptomycin, intravenous telavancin, or intravenous or oral clindamycin. Therapy should be given for 7 to 14 days.53
Recurrent infections should be managed with appropriate antibiotic therapy, as well as environmental management, including frequent hand washing, covering wounds, and environmental hygiene of household surfaces. For patients with recurrent infections despite appropriate therapy and environmental hygiene, decolonization may be considered with a regimen including nasal mupirocin and a topical antiseptic solution, such as chlorhexidine or dilute household bleach.
Impetigo should be treated with topical mupirocin or retapamulin, or with oral therapy to include dicloxacillin or cephalexin. Similarly, ecthyma may be treated with oral therapy, such as dicloxacillin or cephalexin.53
If necrotizing fasciitis is suspected, prompt administration of broad-spectrum antibiotics and surgical consultation for emergent debridement are indicated.54 Antibiotic regimens may include vancomycin or linezolid plus piperacillin-tazobactam or a carbapenem.53 Alternatives may include ceftolozane-tazobactam, ceftazidime-avibactam in association with an anti-anaerobic agent (metronidazole or clindamycin). Other newer treatment alternatives may include ceftaroline, ceftobiprole, oritavancin, or dalbavancin.55
Disposition
Disposition of patients with cellulitis should be determined based on severity of disease, presence of systemic symptoms, ability to ambulate, ability to care for self, and ability to comply with outpatient medical therapy and follow-up. Uncomplicated cellulitis may be treated with outpatient oral antibiotic therapy. Complicated or severe infections require inpatient parenteral therapy. Outpatient parenteral therapy may be considered for select cases.56,57 For patients who may require a single dose of intravenous therapy, this may be given in the ED or an ambulatory setting, followed by outpatient antibiotics.58
REFERENCES
- Raff AB, Kroshinsky D. Cellulitis: A review. JAMA 2016;316:325-337.
- Christensen KL, Holman RC, Steiner CA, et al. Infectious disease hospitalizations in the United States. Clin Infect Dis 2009;49:1025-1035.
- Mills AM, Chen EH. Are blood cultures necessary in adults with cellulitis? Ann Emerg Med 2005;45:549.
- Tracey EH, Modi B, Micheletti RG. Pemetrexed-induced pseudocellulitis reaction with eosinophilic infiltrate on skin biopsy. AM J Dermatopathol 2017;39:e1-e2.
- Moran GJ,Talan DA. Cellulitis: Commonly misdiagnosed or just misunderstood? JAMA 2017;317:760-761.
- Weng QY, Raff AB, Cohen JM, et al. Cost and consequences associated with misdiagnosed lower extremity cellulitis. JAMA Dermatol 2016 Nov 2. [Epub ahead of print.]
- Levell NJ, Wingfield CG, Garioch JJ. Severe lower limb cellulitis is best diagnosed by dermatologists and managed with shared care between primary and secondary care. Br J Dermatol 2011;164:1326-1328.
- Talan DA, Salhi BA, Moran GJ, et al. Factors associated with decision to hospitalize emergency department patients with skin and soft tissue infection. West J Emerg Med 2015;16:89-97.
- Elliot AJ, Cross KW, Smith GE, et al. The association between impetigo, insect bites and air temperature: A retrospective 5-year study (1999-2003) using morbidity data collected from a sentinel general practice network database. Fam Pract 2006;23:490-496.
- Bernard P. Management of common bacterial infections of the skin. Curr Opin Infect Dis 2008;21:122-128.
- McNamara DR, Tleyieh IM, Berbari EF, et al. Incidence of lower-extremity cellulitis: A population-based study in Olmsted county, Minnesota. Mayo Clin Proc 2007;82:817-821.
- Dupuy A, Benchikhi H, Roujeau JC, et al. Risk factors for erysipelas of the leg (cellulitis): Case-control study. BMJ 1999;318:1591-1594.
- Cox NH. Oedema as a risk factor for multiple episodes of cellulitis/erysipelas of the lower leg: A series with community follow-up. Br J Dermatol 2006;155:947-950.
- Wiese-Posselt M, Heuck D, Draeger A, et al. Successful termination of a furunculosis outbreak due to lukS-lukF-positive, methicillin-susceptible Staphylococcus aureus in a German village by stringent decolonization, 2002-2005. Clin Infect Dis 2007;44:e88-e95.
- Hota B, Ellenbogen C, Hayden BK, et al. Community-associated methicillin-resistant Staphylococcus aureus skin and soft tissue infections at a public hospital: Do public housing and incarceration amplify transmission? Arch Intern Med 2007;167:1026-1033.
- Moran GJ, Krishnadasan A, Gorwitz RJ, et al. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med 2006;355:666-674.
- Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the management of skin and soft tissue infections. Clin Infect Dis 2005;41:1373-1406.
- LePage AA, Hess EP, Schears RM. Septic thrombophlebitis with acute osteomyelitis in adolescent children: A report of two cases and review of the literature. Int J Emerg Med 2008;1:155-159.
- Hiransuthikul N, Tantisiriwat W, Lertutsahakul K, et al. Skin and soft-tissue infections among tsunami survivors in southern Thailand. Clin Infect Dis 2005;41:e93-e96.
- Appelgren P, Farnebo F, Dotevall L, et al. Late-onset posttraumatic skin and soft-tissue infections caused by rapid-growing mycobacteria in tsunami survivors. Clin Infect Dis 2008;47:e11-e16.
- Zetola N, Francis JS, Nuermberger EL, Bishai WR. Community-acquired methicillin-resistant Staphylococcus aureus: An emerging threat. Lancet Infect Dis 2005;5:275-286.
- Faraklas I, Yang D, Eggerstedt M, et al. A multi-center review of care patterns and outcomes in necrotizing soft tissue infections. Surg Infect (Larchmt) 2016;17:773-778.
- Misiakos EP, Bagias G, Papadopoulos I, et al. Early diagnosis and surgical treatment for necrotizing fasciitis: A multicenter study. Front Surg 2017;4:5.
- Hadeed GJ, Smith J, O’Keeffe T, et al. Early surgical intervention and its impact on patients presenting with necrotizing soft tissue infections: A single academic center experience. J Emerg Trauma Shock 2016;9:22-27.
- Moore A, Levy BH, Prematilake C, Dissanaike S. The prediction predicament: Rethinking necrotizing soft tissue infections mortality. Surg Infect 2015;16:813-821.
- Jabbour G, El-Menyar A, Peralta R, et al. Pattern and predictors of mortality in necrotizing fasciitis patients in a single tertiary hospital. World J Emerg Surg 2016;11:40.
- Hua C, Sbidian E, Hemery F, et al. Prognostic factors in necrotizing soft-tissue infections (NSTI): A cohort study. J Am Acad Dermatol 2015;73:1006-12.e8.
- Khamnuan P, Chongruksut W, Jearwattanakanok K, et al. Necrotizing fasciitis: Risk factors of mortality. Risk Manag Healthc Policy 2015;8:1-7.
- Raff AB, Weng QY, Cohen JM, et al. A predictive model for diagnosis of lower extremity cellulitis: A cross-sectional study. J Am Acad Dermatol 2017;76:618-625.e2.
- Borschitz T, Schlicht S, Siegel E, et al. Improvement of a clinical score for necrotizing fasciitis: ‘Pain out of proportion’ and high CRP levels aid the diagnosis. PLoS One 2015;10:e0132775.
- Bechar J, Sepehripour S, Hardwicke J, Filobbos G. Laboratory risk indicator for necrotising fasciitis (LRINEC) score for the assessment of early necrotising fasciitis: A systematic review of the literature. Ann R Coll Surg Engl 2017;99:341-346.
- Burner E, Henderson SO, Burke G, et al. Inadequate sensitivity of laboratory risk indicator to rule out necrotizing fasciitis in the emergency department. West J Emerg Med 2016;17:333-336.
- Long B, Koyfman A. Best clinical practice: Blood culture utility in the emergency department. J Emerg Med 2016;51:529-539.
- Paolo WF, Poreda AR, Grant W, et al. Blood culture results do not affect treatment in complicated cellulitis. J Emerg Med 2013;45:163-167.
- van Daalen FV, Kallen MC, van den Bosch CMA, et al. Clinical condition and comorbidity as determinants for blood culture positivity in patients with skin and soft-tissue infections. Eur J Clin Microbiol Infect Dis 2017; Jun 7. [Epub ahead of print].
- Bauer S, Aubert CE, Richli M, Chuard C. Blood cultures in the evaluation of uncomplicated cellulitis. Eur J Intern Med 2016;36:50-56.
- Gunderson CG, Martinello RA. A systematic review of bacteremias in cellulitis and erysipelas. J Infect 2012;64:148-155.
- Pallin DJ, Bry L, Dwyer RC, et al. Toward an objective diagnostic test for bacterial cellulitis. PLoS One 2016;11:e0162947.
- Piso RJ, Pop R, Wieland M, et al. Low sensitivity of needle aspiration cultures in patients with cellulitis/erysipelas. Springerplus 2016;5:1578.
- Iverson K, Haritos D, Thomas R, Kannikeswaran N. The effect of bedside ultrasound on diagnosis and management of soft tissue infections in a pediatric ED. Am J Emerg Med 2012;30:1347-1351.
- Alsaawi A, Alrajhi K, Alshehri A, et al. Ultrasonography for the diagnosis of patients with clinically suspected skin and soft tissue infections: A systematic review of the literature. Eur J Emerg Med 2015; Oct 19. [Epub ahead of print].
- Subramaniam S, Bober J, Chao J, Zehtabchi S. Point-of-care ultrasound for diagnosis of abscess in skin and soft tissue infections. Acad Emerg Med 2016;23:1298-1306.
- Barbic D, Chenkin J, Cho DD, et al. In patients presenting to the emergency department with skin and soft tissue infections what is the diagnostic accuracy of point-of-care ultrasonography for the diagnosis of abscess compared to the current standard of care? A systematic review and meta-analysis. BMJ Open 2017;7:e013688.
- Marvel BA, Budhram GR. Bedside ultrasound in the diagnosis of complex hand infections: A case series. J Emerg Med 2015;48:63-68.
- Shyy W, Knight RS, Goldstein R, et al. Sonographic findings in necrotizing findings in necrotizing fasciitis: Two ends of the spectrum. J Ultrasound Med 2016;35:2273-2277.
- Maroldi R, Farina D, Ravanelli M, et al. Emergency imaging assessment of deep neck space infections. Semin Ultrasound CT MR 2012;33:432-442.
- Hayeri MR, Ziai P, Shehata ML, et al. Soft-tissue infections and their imaging mimics: From cellulitis to necrotizing fasciitis. Radiographics 2016;36:1888-1910.
- Carbonetti F, Cremona A, Carusi V, et al. The role of contrast enhanced computed tomography in the diagnosis of necrotizing fasciitis and comparison with the laboratory risk indicator for necrotizing fasciitis (LRINEC). Radiol Med 2016;121:106-121.
- Stranix JT, Lee ZH, Bellamy J, et al. Indications for plain radiographs in uncomplicated lower extremity cellulitis. Acad Radiol 2015;22:1439-1442.
- Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis 2011;52:e18-e55.
- Aboltins CA, Hutchinson AF, Sinnappu RN, et al. Oral versus parenteral antimicrobials for the treatment of cellulitis: A randomized non-inferiority trial. J Antimicrob Chemother 2015;70:581-586.
- Peterson D, McLeod S, Woolfrey K, McRae A. Predictors of failure of empiric outpatient antibiotic therapy in emergency department patients with uncomplicated cellulitis. Acad Emerg Med 2014;21:526-531.
- Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis 2014;59:e10-e52.
- Leiblein M, Marzi I, Sander AL, et al. Necrotizing fasciitis: Treatment concepts and clinical results. Eur J Trauma Emerg Surg 2017; May 8. [Epub ahead of print].
- Menichetti F, Giuliano S, Fortunato S. Are there any reasons to change our behavior in necrotizing fasciitis with the advent of new antibiotics? Curr Opin Infect Dis 2017;30:172-179.
- Rentala M, Andrews S, Tiberio A, et al. Intravenous home infusion therapy instituted from a 24-hour clinical decision unit for patients with cellulitis. Am J Emerg Med 2016;34:1273-1275.
- Hodgson KA, Huynh J, Ibrahim LF, et al. The use, appropriateness and outcomes of outpatient parenteral antimicrobial therapy. Arch Dis Child 2016;101:886-893.
- Claeys KC, Lagnf AM, Patel TB, et al. Acute bacterial skin and skin structure infections treated with intravenous antibiotics in the emergency department or observational unit: Experience at the Detroit Medical Center. Infect Dis Ther 2015;4:173-186.
Skin and soft tissue infections are encountered commonly in primary care practices, presenting as a range of disorders, from uncomplicated cellulitis, impetigo, folliculitis, erysipelas, and focal abscesses to necrotizing fasciitis. Each year between 1998-2006 in the United States, there were 650,000 hospital admissions for cellulitis, with estimates of 14.5 million cases annually treated as outpatients, accounting for $3.7 billion in ambulatory care costs.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
