Soft Tissue Disorders of the Neck in Children
January 1, 2018
Reprints
AUTHORS
Stephen John Cico, MD, MEd, Assistant Dean for Educational Affairs & Faculty Development, Associate Professor of Clinical Emergency Medicine & Pediatrics, Fellowship Director for Pediatric Emergency Medicine, Division of Emergency Pediatrics, Department of Emergency Medicine, Indiana University School of Medicine, Riley Hospital for Children at IU Health, Indianapolis
Derya Caglar, MD, Associate Professor of Pediatrics, Associate Fellowship Director for Pediatric Emergency Medicine, Division of Emergency Medicine, Department of Pediatrics, University of Washington School of Medicine, Seattle Children’s Hospital, Seattle
PEER REVIEWER
Charles Nozicka, DO, FAAP, FACEP, Division Director, Pediatric Emergency Medicine, Advocate Children’s Hospital, Clinical Professor of Emergency Medicine, Rosalind Franklin University, Libertyville, IL
EXECUTIVE SUMMARY
- Acute unilateral cervical lymphadenitis is caused by streptococcal or staphylococcal infection in 40-80% of cases. The initial drug of choice is amoxicillin/clavulanate potassium (Augmentin), although clindamycin is preferred where methicillin-resistant Staphylococcus aureus has high prevalence.
- Subacute and chronic lymphadenitis typically is due to less common bacterial etiologies, and the provider should consider cat scratch disease, atypical mycobacteria, and toxoplasmosis.
- Nontuberculous mycobacterial (NTM) infections have been rising in children. The majority of patients with NTM lymphadenitis are younger than 5 years of age, contrasting with tuberculous lymphadenitis, which most commonly affects teenagers and young adults.
- Epstein-Barr virus often begins with malaise, fevers, and headaches before development of exudative pharyngitis. It often is associated with posterior cervical lymphadenopathy, myalgias, hepatomegaly, and splenomegaly.
- Adenovirus pharyngitis frequently is associated with conjunctivitis, laryngotracheitis, bronchitis, or pneumonia. Fever, malaise, headache, myalgias, and abdominal pain are common.
- Several clinical prediction rules to guide in testing and treatment have been created to identify cases of group A streptococcal (GAS) pharyngitis, and most are modifications of the original Centor criteria. However, these criteria have not been shown to be effective in predicting GAS in the pediatric population. Thus, rapid antigen strep testing is recommended in those patients in whom GAS pharyngitis is suspected.
- In patients 15 to 30 years of age with an acute sore throat, Fusobacterium necrophorum may be causative in 10%-20.5% of cases. It also is associated with development of retropharyngeal abscesses in younger patients and in Lemierre’s syndrome. F. necrophorum is susceptible to penicillin and first-generation cephalosporins.
- A thyroglossal duct cyst is a benign cystic mass that is found at the base of the tongue and is the most common congenital lesion of the head and neck. The lesion, located most commonly infrahyoid (inferior to the hyoid bone), has a hallmark of physical exam finding of moving upward with extrusion of the tongue.
Pediatric patients frequently present to the emergency department (ED) with issues affecting the soft tissues of the neck. Emergency physicians must be able to recognize and treat these disease processes quickly and accurately. Infections can range from benign, self-limited, viral, or reactive etiologies to potentially life-threatening, emergent, deep tissue necrosis from serious bacterial disease leading to sepsis and death. In addition, providers should be aware of a variety of noninfectious disorders that can present in the soft tissues of the neck that may resolve with supportive care or may require subspecialty evaluation and treatment. This article will focus on the assessment, evaluation, differential, and treatment of both superficial and deep neck soft tissue disorders that clinicians should consider in the emergency setting.
— Ann Dietrich, MD, FAAP, FACEP, Editor
Infectious Etiologies
Cervical Lymphadenitis
Cervical lymphadenitis is very common in childhood and is defined by enlarged, inflamed, and tender lymph node(s) of the neck. Cervical lymph nodes track along the carotid sheath and drain the tonsils, tongue, nose, teeth, and the back of the head and neck. These nodes often become inflamed and may suppurate from upstream infections in any of these areas. However, it is difficult to discern actual incidence, as most cases are caused by viral illnesses and resolve spontaneously.
Acute bilateral cervical lymphadenitis is the most common clinical presentation and often is associated with a self-limited viral infection. Patients will present with current or recent upper respiratory infection (URI) symptoms, including runny nose, sore throat, nasal congestion, and/or cough. Those with Epstein-Barr virus (EBV), cytomegalovirus (CMV), or adenovirus may present with erythematous, enlarged, occasionally exudative pharyngitis and fever associated with enlarged cervical nodes. Herpes-simplex gingivostomatitis can cause painful ulcers on the buccal mucosa, leading to decreased oral intake, fevers, and enlarged cervical lymph nodes. Lymph nodes typically are small, mobile, and mildly tender to palpation without significant swelling or overlying erythema or induration. In most cases, the precipitating illness will resolve with supportive care alone, but lymphadenopathy may persist for months.
Bacteria from nearby infections in the head and neck can invade the cervical lymph node chain and lead to suppuration of the node itself. Acute unilateral cervical lymphadenitis is caused by streptococcal or staphylococcal infection in 40-80% of cases.1 Pyogenic lesions have a unilateral, visible, tender, and discrete swelling, and may progress to generalized erythema, swelling, and tenderness with an area of fluctuance. Typically, patients are febrile and have significant pain on neck movement, which can mimic meningismus. Soft tissue neck ultrasound or CT may be helpful in defining the extent of abscess or surrounding cellulitis. (See Figures 1 and 2.) Empirical treatment of acute bacterial cervical lymphadenitis should provide adequate coverage for both Staphylococcus aureus, including community-associated methicillin-resistant S. aureus (CA-MRSA) if indicated by local microbiology patterns, and group A beta hemolytic strep.1,2,3,4 The initial drug of choice is amoxicillin/clavulanate potassium (Augmentin), although clindamycin is preferred where MRSA has high prevalence.3,4 Nodes with abscess formation may need incision and drainage if they show no improvement despite 48 hours of antibiotic treatment. Acute group A streptococcal (GAS) pharyngitis also may present with bilateral cervical lymphadenitis and is discussed below.
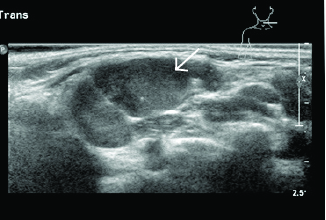
Figure 1. Enlarged Node in Cervical Lymphadenitis |
 |
|
Image courtesy of Derya Caglar, MD. |
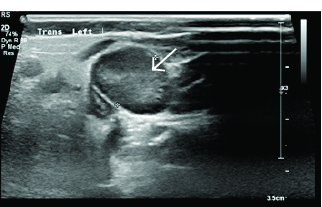
Figure 2. Enlarged Node in Cervical Lymphadenitis, Measuring > 2 cm |
 |
|
Image courtesy of Derya Caglar, MD. |
Subacute and chronic lymphadenitis typically is due to less common bacterial etiologies, and the provider should consider cat scratch disease, atypical mycobacteria, and toxoplasmosis. Cat scratch disease presents in the vast majority of children as a localized skin and lymph node disorder near the site of inoculation approximately one to two weeks later. In some individuals, the organisms disseminate and infect the liver, spleen, eye, or central nervous system. Patients with localized disease generally have a self-limited illness, whereas those with dissemination can have life-
threatening complications. Although lymphadenopathy is most commonly axillary and supraclavicular, patients can present with cervical and submandibular lymph nodes that are enlarged, tender, and have overlying erythema. Bartonella henselae, a pleomorphic Gram-negative bacterium, is the etiologic agent. The patient should be evaluated for any signs of systemic disease and need for parenteral antibiotics, but typically can be discharged with supportive care in mild, isolated disease or treated with azithromycin for five days in moderate disease.5
Atypical organisms often have a more insidious and prolonged course. Nontuberculous mycobacterial (NTM) infections have been rising in children.6 The majority of patients with NTM lymphadenitis are younger than 5 years of age, contrasting with tuberculous lymphadenitis, which most commonly affects teenagers and young adults. Patients often will present as otherwise healthy with unilateral, nontender progressively enlarging cervical lymphadenopathy over weeks to months that has failed to improve on standard antibiotics. Diagnosis can be challenging. Some studies have reported that 30-60% of children with NTM disease have positive tuberculin test results; however, tissue biopsy of the affected node remains the standard of care.6 Main treatment options include surgical excision, treatment with anti-mycobacterial antibiotics, and/or observation without intervention. Cure rates are highest with surgical incision, but there is a risk of facial nerve injury depending on node location and fistula formation. For this reason, incision and drainage and fine needle aspiration are not recommended in cases in which NTM is suspected.6
Toxoplasmosis is associated most frequently with congenital infections but occasionally can present as cervical lymphadenitis associated with flu-like symptoms in an otherwise healthy child. Most patients recover with supportive care alone, although patients with immunodeficiencies may develop systemic disease and need antiparasitic therapy to avoid complications.
Children also may present with enlarged posterior cervical chain nodes, particularly with any scalp infection or irritation. Somewhat large and tender nodes may arise from tinea capitis or kerion formation. This often will require several weeks of systemic antifungal treatment with griseofulvin, terbinafine, itraconazole, or fluconazole for resolution of the infection, with related lymphadenopathy lasting much longer.7
Pharyngitis
Acute pharyngitis commonly is associated with tonsillitis resulting from inflammation of the throat and tonsils. It accounts for 1-2% of all visits to outpatient clinics and EDs, resulting in 7.3 million annual visits for children.8
Symptoms associated with acute pharyngitis include sore throat, odynophagia, fever, headaches, cough, coryza, arthralgias, and fatigue. Physical exam may reveal pharyngeal and tonsillar erythema and/or exudates, palatal petechiae, anterior cervical lymphadenitis, rash, conjunctivitis, anterior stomatitis, and ulcerative lesions. It can be difficult to distinguish between viral and bacterial causes of pharyngitis based on physical exam alone. This ambiguity often results in overdiagnosis of bacterial etiology and unnecessary antibiotic treatment. Most viral infections are self-limited and require only symptomatic treatment.
Many viruses can lead to pharyngitis.9 EBV often begins with malaise, fevers, and headaches before development of exudative pharyngitis. It often is associated with posterior cervical lymphadenopathy, myalgias, hepatomegaly, and splenomegaly. Patients mistakenly treated for bacterial pharyngitis with amoxicillin or ampicillin may develop a characteristic erythematous, maculopapular rash that may be confused with an allergic reaction.
Diagnosis is often clinical. A heterophile antibody (monospot) test can aid in diagnosis, but has frequent false-negative results early in the disease and in children younger than 6 years of age. Thus, a negative test does not exclude the diagnosis of EBV pharyngitis. Testing for EBV immunoglobulin M and immunoglobulin G is more sensitive and specific but results are not available immediately, and often provide little help in guiding treatment, which is largely supportive. Patients may have elevated transaminases, with very rare cases progressing to liver failure. Splenomegaly can persist for weeks, and patients should be counseled to avoid contact sports until resolution to decrease risk of splenic rupture. Patients who have significant pain and dehydration from pharyngitis may benefit from intravenous (IV) hydration and pain control. There is no definitive evidence of the efficacy of steroids on pain control; however, they may be considered to reduce tonsillar swelling when swallowing and respiratory symptoms are a concern.10
CMV also can cause acute pharyngitis and often mimics EBV. Patients with EBV-like symptoms with a negative heterophile test should be considered for CMV. Often, diagnosis is confirmed only with testing for CMV antibodies. Again, treatment is largely supportive.
Adenovirus is another common cause of acute pharyngitis. It leads to 5-10% of all febrile illnesses in infants and young children, and most will have serologic evidence of prior adenoviral infection by the age of 10 years. Adenovirus pharyngitis frequently is associated with conjunctivitis, laryngotracheitis, bronchitis, or pneumonia. Fever, malaise, headache, myalgias, and abdominal pain are common. Physical exam reveals exudative tonsillitis and cervical lymphadenopathy, a syndrome that can be clinically indistinguishable from bacterial infection. Again, supportive care is the mainstay of treatment.
Bacterial pathogens account for approximately 30% of pediatric pharyngitis, with GAS infection being the most common.8 The primary concern for pharyngitis in children aged 3 years or older is that untreated GAS pharyngitis subsequently may cause rheumatic fever or poststreptococcal glomerulonephritis. This is most common in children 5 to 12 years of age. Symptoms in school-age children typically include sore throat, fever, and headache associated with vague abdominal pain. Often, patients do not endorse URI symptoms commonly seen with viral illnesses.8 The exam reveals exudative pharyngitis with associated palatal petechia and bilateral cervical lymphadenitis that is difficult to distinguish from viral causes. Nodes typically are enlarged, mobile, and tender to palpation. A scarlatiniform rash (scarlet fever) may or may not accompany other symptoms.9
Several clinical prediction rules to guide in testing and treatment have been created to identify cases of GAS pharyngitis, and most are modifications of the original Centor criteria.10 (See Table 1.) However, these criteria have not been shown to be effective in predicting GAS in the pediatric population. Thus, rapid antigen strep testing is recommended in those patients in whom GAS pharyngitis is suspected.13 Although children younger than 3 years of age may have GAS infection, the incidence and classic presentation of GAS pharyngitis are uncommon and subsequent systemic complications are fairly rare, so regular testing in this age group is not recommended.10,11,12,13,14 Rapid antigen detection assays for GAS are diagnostic if positive because the specificity of such tests is 98-99% (i.e., 1-2% false-positive results); however, their sensitivity is only 70% (i.e., 30% false-negative results), necessitating follow-up cultures for negative results in pediatric patients.3 Amoxicillin 50 mg/kg given once a day for 10 days or benzathine penicillin 600,000-1,200,000 units given intramuscularly (IM) once is adequate treatment and will prevent systemic sequelae like rheumatic disease, but will not prevent post-streptococcal glomerulonephritis.10,14
Table 1. Centor Criteria |
|
Clinical Feature |
Score |
|
Fever |
+1 |
|
Tonsillar exudates |
+1 |
|
Absence of cough |
+1 |
|
Tender anterior cervical lymphadenopathy |
+1 |
|
Age < 15 years* |
+1 |
|
Age > 44 years* |
-1 |
|
* Included in the Modified Centor Criteria 1 point: 5-10% likelihood of strep, no further testing, no antibiotics 2 points: 11-17% likelihood of strep, optional rapid strep testing/culture, antibiotics if positive 3 points: 28-33% likelihood of strep, consider rapid strep testing/culture, antibiotics if positive 4-5 points: 51-53% likelihood of strep, consider rapid strep testing/culture/antibiotics if positive Note: IDSA and ASIM no longer recommend empiric treatment for GAS. |
|
An increasingly significant bacterial cause of pharyngitis seen predominantly in adolescents is Arcanobacterium haemolyticum. Clinical features overlap with those of GAS and include fever, exudative pharyngitis, and often a rash that occurs in approximately one-half of patients. It may be missed on routine cultures and is more readily detected on human-blood agar plates. A. haemolyticum is unresponsive to penicillin; erythromycin or azithromycin are the treatments of choice.16 This organism should be considered in older adolescent patients who present with pharyngitis that does not respond to appropriate treatment.
Mycoplasma pneumonia also may cause pharyngitis in adolescents and is associated with mucositis and upper respiratory tract illness with or without associated pneumonia.17 Patients often will present with intraoral lesions and nonproductive to mildly productive cough. They may have wheeze but rarely have significant respiratory distress. There actually may be no findings on chest auscultation, even if pneumonia is present early in the course of disease. Treatment includes pain control, hydration, and azithromycin.
Fusobacterium necrophorum also should be considered in older adolescents presenting with pharyngitis. In patients 15 to 30 years of age with an acute sore throat, F. necrophorum may be causative in 10%-20.5% of cases.18 It also is associated with development of retropharyngeal abscesses in younger patients and in Lemierre’s syndrome.19 F. necrophorum is susceptible to penicillin and first-generation cephalosporins.
Diphtheria
Diphtheria is uncommon in developed countries because of immunizations, but it is important to consider in patients from endemic areas or in patients without vaccinations. Corynebacterium diphtheriae produces an endotoxin that causes localized necrosis of the respiratory mucosa and can lead to neurologic (seizures) and cardiac (cardiomyopathy) sequelae. In contrast with GAS pharyngitis, which has acute onset, the onset of symptoms in diphtheria usually is gradual, beginning with discomfort from pharyngeal erythema and inflammation. The hallmark of diphtheria is the formation of a tightly adhering gray membrane in the nares and throat, known as a pseudomembrane, that can lead to airway obstruction and may cause bleeding when dislodged. Penicillin and erythromycin are the antibiotics of choice, although prevention is achieved through effective vaccination programs.
Peritonsillar Abscess
Peritonsillar abscess (PTA) is the most common deep neck infection in pediatric patients.20 This typically is a suppurative complication of acute tonsillitis and cervical lymphadenitis with extension into the peritonsillar space, although hematogenous spread, infected branchial cleft and thyroglossal duct cysts, and odontogenic infections also have been implicated.20 These abscesses can form in patients of all ages but are most common among young adults between the ages of 15 and 30 years. Infections often begin as a palatal or tonsillar cellulitis and progress to abscess formation, typically only on one side. Patients present with high fever, odynophagia, unilateral worsening sore throat, and jaw or ear pain. Classic signs include a muffled “hot-potato” voice, trismus, deviation of the uvula toward the unaffected side, and fullness to the soft palate due to edema. The oral airway may be compromised in significant disease and can present as drooling and shortness of breath.20 The diagnosis of PTA can be made clinically with the presence of medial displacement of the tonsil and uvular deviation. Laboratory data or imaging often is not necessary. Blood work may show an elevated white count and inflammatory markers. Imaging is not necessary to make the diagnosis but may be necessary to differentiate PTA from peritonsillar cellulitis, deep neck space infections (e.g., retropharyngeal or parapharyngeal abscess), or epiglottitis.
Treatment for PTA includes hydration, pain control, IV antibiotics, drainage, and/or tonsillectomy. PTAs often are polymicrobial. The predominant bacterial species are Streptococcus pyogenes (GAS), S. aureus (including MRSA), and oral anaerobes. Current parenteral treatment recommendations include clindamycin at 30 mg/kg/day divided every eight hours or ampicillin-sulbactam 150 mg/kg/day divided every six hours in the acute disease phase.21,22 PTA usually requires drainage through needle aspiration, incision and drainage, or tonsillectomy — procedures that typically are performed by a pediatric otolaryngologist. Drainage with any of these procedures, in combination with antimicrobial therapy and hydration, results in resolution in more than 90% of cases.23,24
There is debate as to whether corticosteroids are helpful in the treatment of PTA. Although some studies have shown benefit in older adolescents and adults, others have not shown a clear benefit. Additional studies are needed to determine benefits of treatment.
Parapharyngeal Abscess
Parapharyngeal abscess may occur with extension of peritonsillar abscesses, although dental abscesses are the leading cause.20 These infections are potentially life-threatening because of the possibility of involving the carotid sheath and its vital contents (e.g., common carotid artery, internal jugular vein, vagus nerve), propensity for airway impingement, and increased likelihood of bacteremia. Carotid artery erosion and suppurative jugular thrombophlebitis (Lemierre’s syndrome) both carry very high mortality rates, so prompt diagnosis and treatment are key for good patient outcomes.19,25
Patients with parapharyngeal abscesses often will look ill with fevers and complain of throat pain and difficulty swallowing. With significant disease, dyspnea may be seen with swelling near the epiglottis or larynx. Exam findings of significance include trismus and medial bulging of the posterior pharyngeal wall. However, abscess can progress through deep neck spaces quickly into the mediastinum without localizing exam findings. Diagnosis is best made by CT with IV contrast.
Patients diagnosed with parapharyngeal abscesses should be hospitalized and managed in consultation with otolaryngology. Initial treatment depends on respiratory status and location of abscess. For those cases arising from an oral or odontogenic source, ampicillin-sulbactam and clindamycin are antibiotics of choice. In toxic-appearing patients or those with MRSA risk factors, treatment should begin with vancomycin.25
Lemierre’s Syndrome
Lemierre’s disease is a rare infection caused by F. necrophorum18 or mixed anaerobic flora, and is associated with internal jugular venous thrombophlebitis and septic emboli. Infection spreads from the oropharynx to the parapharyngeal or lateral pharyngeal space, extending into the carotid sheath and vessels. Patients often are adolescents and young adults (16-30 years of age) and previously healthy. They usually will appear ill and present with high fevers, rigors, and severe sore throat and neck pain. Clinical examination of the oropharynx may demonstrate erythema, ulceration, a pseudomembrane, or posterior pharyngeal swelling. They often will have tachycardia, tachypnea, and decreased oxygen saturations.
Septic emboli to the lungs is a common finding in a majority of cases.18 Chest radiography may demonstrate infiltrates, which can cavitate to form lung abscesses and empyema. Patients may develop septic arthritis and/or osteomyelitis. Complications include a mortality rate as high as 10%, especially when antibiotic treatment is delayed.19
Patients should be admitted for close monitoring of sepsis and respiratory distress and initiated on broad-spectrum parenteral antibiotics. Anticoagulation is a controversial issue. Based on the current evidence, it is not clear whether anticoagulation therapy is beneficial. There were no apparent differences in mortality or clinical course in those who received anticoagulants and those who did not. Unfortunately, no randomized studies on this controversy are available.19
Retropharyngeal Abscess
Retropharyngeal abscesses (RPA) are serious suppurative infections of the neck occurring with a peak age between 2 and 4 years.26 Infection often occurs as bacteria in the pharynx spread via lymphatics to the retropharyngeal nodes where they suppurate. Oral trauma, spread from bacteremia, and direct extension from vertebral osteomyelitis are other rare causes. It is unusual to see RPA in older children as the retropharyngeal nodes typically involute around 5 to 6 years of age.
Children with RPAs often appear moderately ill with fever and present with difficulty or pain with swallowing, drooling, stiff neck, or torticollis. The clinical picture may be confused with meningitis, although these children often have more difficulty with neck extension rather than flexion. Parents may report a change in vocal quality. Patients also may have significant respiratory distress with tachypnea or stridor as the disease progresses. Many, but not all, will have associated neck swelling or lymphadenopathy. The physical exam often is limited if the child is unable to open his or her mouth because of pain. If there is any concern for epiglottitis, imaging or exam under anesthesia may be necessary. If the posterior pharynx is visualized, a mass or swelling may be appreciated. Tender, enlarged anterior cervical lymphadenopathy is common.
These infections often are polymicrobial, including S. pyogenes, S. aureus (including MRSA), F. necrophorum, and respiratory anaerobes.18,23
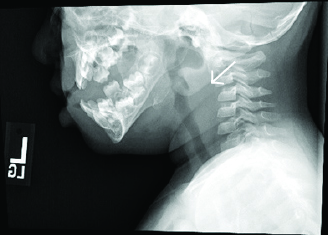
Initial evaluation should include complete blood count and blood culture, although this may be postponed to the operating room if the child’s airway is thought to be tenuous. The white count often will be elevated with bandemia. Blood cultures rarely are positive but are helpful in determining a pathogen. Imaging often begins with a lateral neck radiograph. When a retropharyngeal infection is present, the prevertebral space will be widened. Prevertebral soft tissues at the level of C3 should measure less than two-thirds of the anteroposterior width of the body of the third cervical vertebra. Alternatively, the retropharyngeal space is considered pathologically widened if it is greater than 7 mm at C2 or 14 mm at C6 in children.26 There often will be loss of the normal cervical lordosis due to spasm of inflammation. In rare cases, an air-fluid level or gas may be seen in the pre-vertebral area. (See Figure 3.)
Figure 3. Soft Tissue Neck X-ray in Child With RPA Shows Widening of the Prevertebral Space |
 |
|
Image courtesy of Derya Caglar, MD. |
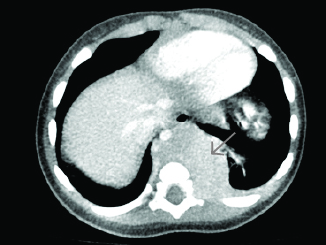
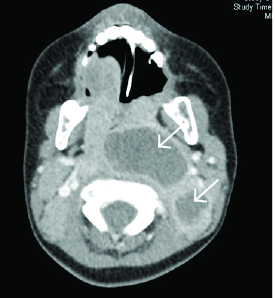
CT of the neck with IV contrast is the best study to identify abscesses in the retropharyngeal space. It can demonstrate extension of an abscess into contiguous spaces in the neck or chest and help differentiate between abscess and cellulitis. CT also can help identify the position of critical vessels in the neck when planning surgical intervention. (See Figure 4.)
Figure 4. CT Neck With Contrast Reveals Two Retropharyngeal Abscesses in an Intubated Patient |
 |
|
Image courtesy of Derya Caglar, MD. |
Children found to have an RPA should be hospitalized and managed in consultation with otolaryngology. Initial treatment depends on respiratory status and extension of abscess. In early disease, IV antibiotics may prevent progress and should be initiated as soon as possible. Empirical therapy should cover likely organisms (GAS, Staph, and respiratory anaerobes). Clindamycin is the drug of choice for initial treatment, given increasing rates of MRSA.22 With larger lesions and those patients in respiratory distress, immediate surgical drainage is necessary.
Ludwig’s Angina
Ludwig’s angina is an infection of the submandibular space, including the floor of the mouth, the sublingual space, and the submylohyoid (also known as submaxillary) space. This infection most commonly arises from dental infections and abscesses. It is an aggressive, rapidly spreading cellulitis and potentially can lead to airway obstruction.27
Ludwig’s angina is typically polymicrobial and reflects oral flora. The most common organisms include viridans streptococci. Oral anaerobes, including Peptostreptococcus species, Fusobacterium nucleatum, Bacteroides, and Actinomyces spp. also frequently are implicated.
Patients typically present with ill appearance, fever, chills, mouth pain, and stiff neck. Patients often will drool and complain of dysphagia. They may lean forward to maximize the airway diameter. They may have a muffled voice or be unable to speak. Trismus usually is absent unless there is spread into the parapharyngeal space. With more progressive disease, patients may develop respiratory distress, stridor, and cyanosis. On physical examination, patients have tender, symmetric induration in the submandibular area. There is typically no lymphadenopathy. The floor of the oropharynx usually is elevated and erythematous, and is tender to palpation.27
Patients should be started immediately on broad-spectrum antibiotics with activity against beta-lactamase-producing aerobes, anaerobes, and S. aureus, including in some cases, MRSA. Ampicillin-sulbactam, clindamycin, and vancomycin all would provide appropriate coverage, with the latter antibiotics offering MRSA coverage if suspected. Patients should be admitted and monitored closely for worsening pain and development of respiratory distress.
Congenital Etiologies
Thyroglossal Duct Cyst
A thyroglossal duct cyst is a benign cystic mass that is found at the base of the tongue and is the most common congenital lesion of the head and neck. There is a 5-10% prevalence in the general population, and it is the most common congenital abnormality in the pediatric population.25,29 These lesions occur equally between the sexes, except in a rare autosomal dominant form that has a predilection for women.28 It often presents as a painless midline neck mass, typically before 10 years of age. The mass often varies in size, and can increase after viral illnesses of the head and neck region. Rarely, thyroglossal duct cysts are large enough to cause airway compromise in neonates and present with symptoms of upper airway obstruction.28,29 The lesion, located most commonly infrahyoid (inferior to the hyoid bone), has a hallmark of physical exam finding of moving upward with extrusion of the tongue.28 Thyroglossal duct cysts are located midline in 75% of patients, and just off midline in the remaining patient population.28 The diagnostic modality of choice is ultrasound, although CT or MRI also can be used. Complete surgical excision is the definitive treatment of thyroglossal duct cysts,28,30,31 since partial resection has a recurrence rate of more than one-third.28 About 1% of thyroglossal duct cysts are associated with papillary carcinoma of the thyroid, and should be considered when the lesion is cystic or has areas of calcification.28,29
Branchial Cleft Abnormalities
Abnormalities of the branchial clefts are caused by incomplete involution of the branchial cleft apparatus, with the second branchial arch being the most common and representing 95% of lesions.28 These lesions are found lateral to the midline of the submandibular region and superior neck anteriorly, and are the second most common congenital abnormality of the neck in the pediatric population. Abnormalities related to the first branchial cleft are the second most common congenital abnormality of the branchial cleft apparatus, occurring in 1-4% of patients. These can present as lesions from the ear region to the angle of the jaw. Clinically they may appear as areas of swelling (cysts), sinuses, or fistulas, may involve the parotid gland, and often are confused as preauricular pits or sinuses.28,30,32 They are associated closely with the external auditory canal, tympanic membrane, angle of the mandible, and/or facial nerve. Less common are abnormalities of the remnants of the third and fourth branchial arches, which can be difficult to differentiate on examination. Surgically, those arising from the third branchial cleft are located superior to the superior laryngeal nerve, whereas those from the fourth are below the superior laryngeal nerve.29,33
Preauricular Pits, Cysts, and Sinuses
Appearing either unilaterally or bilaterally, preauricular pits, cysts, and sinuses are harmless unless they become prone to recurrent infections. This is the typical indication for excision, and without complete resection of the tract, they are prone to recurrence.32
Cystic Hygromas
Usually found posteriorly in the neck, cystic hygromas are the most common lymphangioma, and often are associated with chromosomal abnormalities (Turner’s syndrome; trisomy 13, 18, 21; Noonan syndrome).28,29 More than three-quarters of cystic hygromas involve the neck and lower portion of the face, and they represent 5% of all benign tumors of infants and children.28 Because these lesions can cross tissue planes, they can be invasive and cause significant mass effect on surrounding structures. They can be slow-growing or they can increase in size suddenly after infections, trauma, or spontaneous hemorrhage into the lesion. They present as large, cystic lesions, and can lead to airway compromise. Ultrasound often can demonstrate increased blood flow within the cystic hygroma, and they can be delineated further by both CT and MRI.28,29
Dermoid Cysts
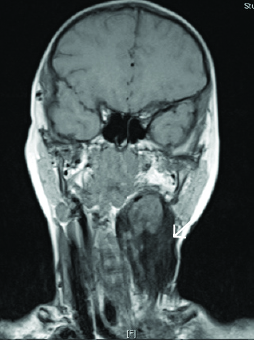
Dermoid cysts are classified by their histologic composition as epidermoid, dermoid, and teratoid cysts. (See Figure 5.) Collectively referred to as dermoid cysts, they can appear in a similar location to thyroglossal duct cysts, although they typically are found in a suprahyoid location rather than the infrahyoid location of thyroglossal duct cysts. However, dermoid cysts tend to form later, appearing more commonly in late childhood or early adulthood, as a result of their slow-growing nature;28 pregnancy is associated with increased growth of dermoid cysts.30 Unlike the thyroglossal duct cyst, dermoid cysts do not move when the tongue is protruded.
Figure 5. Patient With Large Left-sided Dermoid Cyst on MRI |
 |
|
Image courtesy of Derya Caglar, MD. |
Thymic Cysts
The thymus originates as part of the third branchial arches, and if tissue descent into the upper anterior mediastinum is disrupted during development, remnants of thymic tissue can become cystic over time. Although rare, two-thirds are found because of swelling in the first 10 years of life, with the remaining most commonly identified over the next two decades. This correlates to the ages at which the thymus is most active. Although much less common, respiratory problems, dysphagia, hoarseness, and stridor all can be presenting symptoms of thymic cysts in young infants.28,30
Thyroid Nodules/Cysts
Thyroid nodules and cysts are common in the pediatric population, being found in an estimated 0.05-1.8% of the population.32 However, these nodules are malignant in approximately 25% of patients, which is significantly higher than in the adult population,34,35,36,37,38 in whom the range for malignancy is between 5-15%. The majority of pediatric patients present as euthyroid, with a small minority of patients presenting as hyperthyroid.40 The majority of cases of cancer of the thyroid are found in solitary nodules, although cancer can be found in multinodular lesions as well.41,42 Malignant lesions are discussed later. More than 80% of thyroid nodules in children are found by palpation on examination, and the remaining present with goiter and subsequently are found to have thyroid nodules identified on imaging studies, most commonly ultrasonography.41 Thyroid cysts may undergo aspiration, but are likely to recur. Large cysts or nodules should be referred for nonemergent excision by an otolaryngologist or surgeon.
Malignant Etiologies
Neuroblastoma
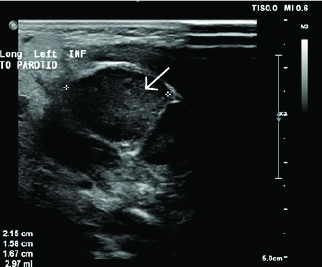
Although the neck is a rare location for presentation, neuroblastoma tumors presenting as neck masses carry a better prognosis than neuroblastoma presenting at other primary sites in the body.39 They can present as simple areas of swelling/mass, or they can present with secondary symptoms such as stridor, dysphagia, Horner syndrome, or voice changes. Physical examination shows a solid mass (rather than fluctuant) that is relatively immobile, typically non-tender, and may demonstrate surrounding lymphadenopathy. Ultrasound, CT, and MRI all can help make the diagnosis.28 (See Figure 6.)
Figure 6. Ultrasound and Computed Tomography of Neuroblastoma |
|
Ultrasound of patient presenting with neck swelling found to have neuroblastoma. Ultrasound shows matted, enlarged lymph nodes with calcifications
|
|
CT chest and abdomen shows the primary intrathoracic and abdominal mass
|
|
Image courtesy of Derya Caglar, MD. |
Rhabdomyosarcoma
Rhabdomyosarcoma is an aggressive malignancy of the soft tissues in the pediatric population, and presentation in the head and neck region represents approximately 40% of primary tumor sites. Because it is an invasive tumor, surrounding structures can be infiltrated, so presentation can range from a solid, non-mobile mass to symptoms secondary to infiltration of surrounding structures. Although typically a solid-type tumor, increased vascularity can be seen on Doppler ultrasound. CT and MRI also can aid in the diagnosis.29
Thyroid Cancer
Thyroid malignancy, of which papillary thyroid cancer is the most common subtype, can be associated with 1% of thyroglossal duct cysts. Calcifications may be seen on ultrasound or CT imaging, and surrounding lymphadenopathy may be secondary to lymphangitic spread of the tumor.28,29 Other potential malignancies of the thyroid gland seen in the pediatric population include follicular and medullary carcinomas, Hurthle cell adenomas, and teratomas, although all of these are more rare than papillary thyroid cancer.
Leukemia and Lymphoma
Non-Hodgkin’s lymphoma, which presents most often in younger children, and Hodgkin’s lymphoma, which presents in older children and teenagers, can present as neck masses. Hodgkin’s lymphoma often presents with constitutional symptoms, the so-called “B-symptoms” of lymphoma. These include fever, night sweats, and weight loss of 10% or greater; however, itching and fatigue also can be symptoms of lymphoma. Depending on the size and location of the enlarged nodes, patients can present with hoarseness, dysphagia, stridor, cranial nerve palsies, or upper airway obstruction. Evaluation for the presence of hepatosplenomegaly and other areas of lymphadenopathy can increase a physician’s suspicion for this diagnosis. Tissue biopsy makes the definitive diagnosis of lymphoma.29,34
Leukemia, the most common pediatric cancer, can present as lymphadenopathy in the neck region. Acute lymphoblastic leukemia is the most common form of leukemia in children. Besides lymphadenopathy in the cervical and peritonsillar region, hepatosplenomegaly also may be present. A complete blood count with differential showing blasts on the peripheral smear is diagnostic, although blasts sometimes are present only on bone marrow aspirate. Infectious diseases, as with EBV or CMV, or aplastic anemia can mimic the diagnosis of leukemia and should be considered in the differential.28,33,34
General Approach to Neck Masses in Pediatric Patients
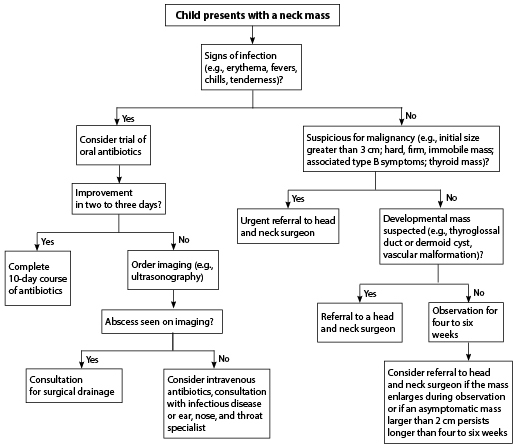
When a child presents to the ED with a neck mass, initial assessment should include a thorough history, including family history of head and neck cancers; familial endocrine disorders that predispose to cancer; weight loss; difficulty swallowing; time frame of appearance of the lesion; recurrence or previous lesion; drainage from the lesion; or infectious symptoms, such as fever, erythema, or purulent drainage. If there are typical signs of infection without airway compromise or swallowing difficulties, a course of appropriate antibiotics should be considered. If there is no improvement or continued worsening after 48 hours, further imaging may be warranted, and either IV antibiotics or surgical consultation should be considered.
If there is no evidence of infectious etiology, the next consideration should be whether there are features concerning for malignancy. If there are, referral to a qualified surgeon and appropriate imaging are the next steps. For those in whom malignancy is less likely, congenital abnormalities become a greater possibility. For these patients, a period of observation is appropriate, although referral to a surgeon and imaging also can be considered. If a lesion persists or enlarges during a four- to six-week period of observation, referral to a surgeon is indicated. Figure 7 outlines this general approach to the evaluation and treatment of pediatric neck masses.
Figure 7. Algorithm for the Treatment of Children With Neck Masses |
 |
|
Reprinted with permission from: Meier JD, Grimmer JF. Evaluation and management of neck masses in children. Am Fam Physician 2014;89:353-358. |
REFERENCES
- Leung AK, Davies HD. Cervical lymphadenitis: Etiology, diagnosis, and treatment. Curr Infect Dis Rep 2009;11:183-189.
- van Driel ML, De Sutter AI, Keber N, et al. Different antibiotic treatments for group A streptococcal pharyngitis. Cochrane Database Syst Rev 2013;(4):CD004406.
- Naseri I, Jerris RC, Sobol SE. Nationwide trends in pediatric Staphylococcus aureus head and neck infections. Arch Otolaryngol Head Neck Surg 2009;135:14-16.
- Walker PC, Karnell LH, Ziebold C, Kacmarynski DS. Changing microbiology of pediatric neck abscesses in Iowa 2000-2010. Laryngoscope 2013;123:249-252.
- American Academy of Pediatrics. Kimberlin DW, Brady MT, Jackson MA, Long SS, eds. Red Book: 2015 Report of the Committee on Infectious Diseases. Elk Grove Village, IL: American Academy of Pediatrics; 2015.
- Zimmermann P, Curtis N, Tebreugge M. Nontuberculous mycobacterial disease in childhood — update on diagnostic approaches and treatment. J Infect 2017;74:s136-s142.
- Chen X, Jiang X, Yang M, et al. Systemic antifungal therapy for tinea capitis in children. Cochrane Database Syst Rev 2016;(5):CD004685.
- Wessels MR. Clinical practice. Streptococcal pharyngitis. N Engl J Med 2011;364:648-655.
- Kociolek LK, Shulman ST. In the clinic. Pharyngitis. Ann Intern Med 2013;157:ITC3-1.
- Centor R, Witherspoon J, Dalton HP, et al. The diagnosis of strep throat in adults in the emergency room. Med Decis Making 1981;1:239-246.
- Lean WL, Arnup S, Danchin M, Steer AC. Rapid diagnostic tests for group A streptococcal pharyngitis: A meta-analysis. Pediatrics 2014;134:771-781.
- Shulman ST, Bisno AL, Clegg HW, et al. Clinical practice guideline for the diagnosis and management of group A streptococcal pharyngitis: 2012 update by the Infectious Diseases Society of America. Clin Infect Dis 2012;55:1279-1282.
- Roggen I, van Berlaer G, Gordts F, et al. Centor criteria in a paediatric emergency department: For what it’s worth. BMJ Open 2013; Apr 22. doi:10.1136/bmjopen-2013-002712. Print 2013.
- American Academy of Pediatrics Committee on Infectious Diseases. Red Book, 28th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2009
- Shulman S, Bisno A, Clegg HW, et al. Clinical practice guideline for the diagnosis and management of group A streptoccoal pharyngitis: 2012 update by the IDSA. Clin Infect Dis 2012; 55:86-102.
- Mitchell M, Sorrentino A, Centor RM. Adolescent pharyngitis: A review of bacterial causes. Clin Pediatr 2011;50:1091-1095.
- Ravin KA , Rappaport LD , Zuckerbraun NS , et al. Mycoplasma pneumoniae and atypical Stevens-Johnson syndrome: A case series. Pediatrics 2007;119:e1002-1005.
- Centor RM, Atkinson TP, Ratliff AE, et al. The clinical presentation of Fusobacterium-positive and streptococcal-positive pharyngitis in a university health clinic. Ann Intern Med 2015;162:241-247.
- Johannesen KM, Bodtger U. Lemierre’s syndrome: Current perspectives on diagnosis and management. Infect Drug Resist 2016;9:221-227.
- Conrad D, Parikh S. Deep neck infections. Infect Disord Drug Targets 2012;12:286-290.
- Baldassari C, Shah RK. Pediatric peritonsillar abscess: An overview. Infect Disord Drug Targets 2012;12:277-280.
- Abdel-Haq N, Quezada M, Asmar BI. Retropharyngeal abscess in children: The rising incidence of methicillin-resistant Staphylococcus aureus. Pediatr Infect Dis J 2012;31:696-699.
- Tebruegge M, Curtis N. Infections related to the upper and middle airways. In: Principles and Practice of Pediatric Infectious Diseases, 4th ed. Long SS, Pickering LK, Prober CG, eds. New York: Elsevier Saunders; 2012: 205.
- Chang BA, Thamboo A, Burton MJ, et al. Needle aspiration versus incision and drainage for the treatment of peritonsillar abscess. Cochrane Database Syst Rev 2016;(12):CD006287.
- Goldstein N, Hammersclag M. Peritonsillar, retropharyngeal, and parapharyngeal abscesses. In: Textbook of Pediatric Infectious Diseases. 6th ed. Philadelphia: Saunders; 2009: 177.
- Abdel-Haq N, Quezada M, Asmar BI. Retropharyngeal abscess in children: The rising incidence of methicillin-resistant Staphylococcus aureus. Pediatr Infect Dis J 2012;31:696-699.
- Reynolds SC, Chow AW. Life-threatening infections of the peripharyngeal and deep fascial spaces of the head and neck. Infect Dis Clin North Am 2007;21:557-576.
- LaPlante JK, Pierson NS, Hedlund GL. Common pediatric head and neck congenital/developmental anomalies. Radiol Clin North Am 2015;53:181-196.
- Brown R, Harave S. Diagnostic imaging of benign and malignant neck masses in children—a pictorial review. Quant Imaging Med Surg 2016;6:591-604.
- Koeller KK, Alamo L, Adair CF, Smirniotopoulos JG. Congenital cystic masses of the neck: Radiologic-pathologic correlation. Radiographics 1999;19:121-146.
- Kim MH, Chung JH. Failure of sclerotherapy in the treatment of thyroglossal duct cyst in children: 2 case reports and review of the literature. J Pediatr Surg 2012;47:E37-40.
- LaRiviere CA, Waldhausen JH. Congenital cervical cysts, sinuses, and fistulae in pediatric surgery. Surg Clin North Am 2012;92:583-597, viii.
- Prosser JD, Myer CM 3rd. Branchial cleft anomalies and thymic cysts. Otolaryngol Clin North Am 2015;48:1-14.
- Niedziela M. Pathogenesis, diagnosis and management of thyroid nodules in children. Endocr Relat Cancer 2006;13:427-453.
- Wiersinga WM. Management of thyroid nodules in children and adolescents. Hormones (Athens) 2007;6:194-199.
- Huang SA. Thyromegaly. In: Lifshitz F, ed. Pediatric Endocrinology, 5th ed. New York: Informa Healthcare USA; 2007: 446-447.
- Dinauer CA, Breuer C, Rivkees SA. Differentiated thyroid cancer in children: Diagnosis and management. Curr Opin Oncol 2008;20:59-65.
- Dinauer C, Francis GL. Thyroid cancer in children. Endocrinol Metab Clin North Am 2007;36(3):779-806, vii.
- Young G, Toretsky J, Campbell A, Eskenazi A. Recognition of common childhood malignancies. Am Fam Physician 2000;61:2144-2154.
- Guille JT. Evaluation and management of pediatric thyroid nodule. Oncologist 2015;20:19-27.
- Corrias A, Mussa A, Baronio F, et al. Diagnostic features of thyroid nodules in pediatrics. Arch Pediatr Adolesc Med 2010;164:714-719.
- Corrias A, Einaudi S, Chlorboli E, et al. Accuracy of fine needle aspiration biopsy of thyroid nodules in detecting malignancy in childhood: Comparison with conventional, clinical, laboratory, and imaging approaches. J Clin Endocrinol Metab 2001;86:4644-4648.
Pediatric patients frequently present to the emergency department with issues affecting the soft tissues of the neck. This article will focus on the assessment, evaluation, differential, and treatment of both superficial and deep neck soft tissue disorders that clinicians should consider in the emergency setting.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.