Advanced Cardiac Life Support Updates
AUTHORS
Alan A. Dupre, MD, FACEP, Assistant Professor, Emergency Medicine, Boonshoft School of Medicine, Wright State University, Kettering, OH
Mikel Elias Borup, DO, Department of Emergency Medicine, Wright State University, Kettering, OH
Jamie Siesel, DO, Department of Emergency Medicine, Wright State University, Kettering, OH
PEER REVIEWER
William Brady, MD, FACEP, FAAEM, Professor of Emergency Medicine & The David A. Harrison Distinguished Educator, University of Virginia School of Medicine, Charlottesville
Introduction
Heart disease is the leading cause of death worldwide. Death can be reduced when practitioners provide evidence-based care, especially during acute resuscitations. The American Heart Association (AHA) Advanced Cardiac Life Support (ACLS) guidelines are recognized as the authoritative reference for acute cardiopulmonary resuscitation. Emergency medicine providers should be thoroughly familiar with the AHA ACLS guidelines.
ACLS guidelines were established first in 1974 and have been periodically updated since.1 In 2015, the AHA introduced a web-based format for the latest guideline update, which allows them to be updated continuously.2 This article will discuss the newest AHA evidence-based guidelines and recommendations. The major updates include an emphasis on the quality of cardiopulmonary resuscitation (CPR), a change in layperson CPR, changes in cardiac arrest medication, and renewed emphasis on post-cardiac arrest management.
Quality of Cardiopulmonary Resuscitation
Chest Compressions
Uninterrupted chest compressions and defibrillation remain the most important components for resuscitation of cardiac arrest. For that reason, the AHA has reorganized the importance of sequential tasks for cardiac arrest from “ABC” to “CAB.” Coronary perfusion pressure (CPP) is dependent on continuous chest compressions at an adequate rate and depth. CPP is one of the most important factors to achieving a return of spontaneous circulation (ROSC). Chest compressions are the first priority for both adult and pediatric patients, and interruptions in compressions must be minimized before and after defibrillation, or if halted to perform other interventions.
Inadequate compression rate and depth are the most common opportunities for improvement in resuscitation.2 The 2010 guidelines stated that a rate of > 100 was appropriate. However, the 2015 guidelines have instituted an upper limit of 120 compressions per minute, making the new guideline compression rate 100 to 120 compressions per minute.3 An upper limit was instituted after a large study demonstrated that compression rate above 120/minute was associated with an increased likelihood of inadequate compression depth, thus decreasing coronary blood flow and cardiac output.4 The minimum adult chest compression depth is 2 inches (5 cm), established by the 2010 guidelines. The 2015 guidelines, similarly, have added a maximum depth of 2.4 inches (6 cm) because data suggested potential for significant injury from excessive chest compression depth.3,5
Compression Devices
Given the increased popularity of automatic compression devices, the 2015 ACLS guidelines also address their use. Automatic compression devices have been shown to have no greater success rate than manual CPR; but conversely, there are also no worsened outcomes when compression devices are used by trained personnel. In some cases, using these devices can increase safety and allow providers to focus on other elements of resuscitation, but manual chest compressions remain the preferred delivery method. Some newer studies are showing that both rate and compression depth are improved during manual chest compressions with feedback devices, and this may be the best option for CPR.6,7,8
Airway/Breathing
Chest compressions should not be interrupted during the early phase of resuscitation to place an advanced airway. Placement of a definitive airway is a frequent cause of paused compressions, with data showing that 25% of all interruptions in chest compressions were due to placement of an advanced airway.9 The 2015 guidelines state that it is reasonable to delay positive-pressure ventilation if compressions are being performed and passive oxygenation is applied. AHA guidelines from 2010 recommend supraglottic airway devices as an appropriate alternative to endotracheal intubation during the initial phase of resuscitation, and this recommendation remains in the most recent update.10
If an advanced airway is indicated (patient is difficult to bag mask ventilate or more than three rounds of CPR have been completed with passive oxygenation), cricoid pressure is not recommended during intubation. For confirmation of endotracheal tube placement, continuous waveform capnography remains the gold standard. This is a class I recommendation. (See Table 1.) The newest guidelines also mention ultrasound as an additional method to confirm endotracheal tube placement.11 After placement of an advanced airway, chest compressions should be performed continuously with one breath being delivered every six seconds while chest compressions are being performed. This simplifies the algorithm and, again, decreases pauses in chest compressions.3
Table 1. Strength of Evidence
Class |
Strength |
Recommendation |
|
I |
Strong: benefit much greater than risk |
Recommended Indicated |
|
IIa |
Moderate: benefit greater than risk |
Is reasonable Can be useful |
|
IIb |
Weak: benefit greater than or equal to risk |
May be reasonable Might be considered |
|
III |
No benefit: benefit about equal to risk |
Not recommended Not indicated |
ECPR or ECMO
Extracorporeal CPR (ECPR) and extracorporeal membrane oxygenation (ECMO) to resuscitate the arrested heart has attracted increasing interest. The recent guidelines state that ECMO can be an appropriate option in select cases of refractory cardiac arrest with potentially reversible causes, such as a large pulmonary embolism (PE) or myocardial infarction. There have been two recent large studies, one in the United States and one in Australia, that have shown ECMO to be effective in select cases.12,13
Medications During Cardiac Arrest
Vasopressors in Cardiac Arrest
Epinephrine. Epinephrine is a naturally occurring sympathomimetic hormone released from the adrenal medulla. It acts on both alpha- and beta-adrenergic receptors to cause both beneficial and harmful effects during a cardiac arrest. Stimulating alpha receptors on vascular smooth muscle causes vasoconstriction, which can increase both coronary and cerebral perfusion pressure. Some of the harmful effects come from actions on beta-adrenergic receptors, which can cause increased myocardial work and oxygen demand.
Studies of epinephrine in human cardiac arrest have shown mixed results. In general, epinephrine increases the rate of ROSC, but does not increase the rate of hospital discharge or favorable neurologic outcome, in both out-of-hospital and in-hospital cardiac arrests.14,15 Despite its limitations, epinephrine remains the vasopressor of choice for resuscitation of cardiac arrest, given the lack of a more beneficial alternative medication.
Epinephrine Dose. There have been multiple studies and clinical trials evaluating the short-term and long-term outcomes of standard dose epinephrine (1 mg intravenous [IV]/intraosseous [IO] every 3-5 minutes). These studies have shown no clear long-term, neurologically favorable survival benefit. Currently, the recommendation (class IIb) is that standard-dose epinephrine “may be reasonable” for a patient in cardiac arrest.11
Some studies have looked at high-dose epinephrine (0.1 to 0.2 mg/kg), which theoretically may increase coronary perfusion and increase ROSC and long-term survival.11,16 It is thought that the acidosis during cardiac arrest may diminish the response to adrenergic agents, and high doses may be needed. However, there is concern that the increased dose may have detrimental outcomes in the post-arrest period, including demand ischemia from tachycardia, hypertension, and myocardial dysfunction.14 Studies investigating high-dose epinephrine have shown short-term survival benefit, but have failed to show any improvement in survival to hospital admission, survival to hospital discharge, or survival with good neurologic recovery.17 Based on these findings, high-dose epinephrine is not recommended for routine use in cardiac arrest.11
Epinephrine Timing. Several large observational studies have looked at the timing of epinephrine administration in out-of-hospital cardiac arrest (OHCA), in-hospital cardiac arrest (IHCA), in both shockable rhythms and non-shockable rhythms. In non-shockable rhythms, the studies evaluating both OHCA and IHCA found that administering epinephrine as soon as possible after the onset of cardiac arrest resulted in improvement in all measured outcomes (ROSC, survival to discharge, and improved neurologically favorable outcomes).18 In shockable rhythms, with clear benefit from defibrillation, the question is when epinephrine should be administered in relation to defibrillation. Epinephrine timing in shockable rhythms has not been well studied, and the available studies have had mixed results.
The studies reviewed as part of the 2015 guideline update show varied benefit with early administration of epinephrine, but worsened outcomes with late administration. Based on this, the AHA states that there is currently insufficient evidence to make a recommendation on the optimal timing of epinephrine administration in relation to defibrillation.11
Studies published after the 2015 guidelines were written noted as much as a 4% decrease in the likelihood of ROSC with each minute that passes with 911 activation and administration of vasopressors.19,20 Further studies are needed to evaluate the time-dependency of vasopressor administration on ROSC, especially in shockable rhythms, but it is reasonable to attempt defibrillation as soon as possible, followed by early administration of epinephrine. If the ability to defibrillate is delayed, resuscitation should proceed with administration of epinephrine. More recent studies have compared less frequent average epinephrine dosing (every 6 to < 7 min/dose rather than standard 4 to < 5 min/dose) and showed improved survival to hospital discharge.21,22
Vasopressin. Vasopressin is a non-adrenergic neuroleptic hormone that acts via V1 receptors on smooth muscle cells, causing constriction of the peripheral, coronary, and renal vasculature. In comparison to epinephrine, vasopressin has a gradual onset and longer half-life.14 Vasopressin had been included in previous guidelines as an alternative to the first or second dose of epinephrine.
Studies have demonstrated no benefit to the use of vasopressin over epinephrine for ROSC or survival to hospital discharge, regardless of neurological outcome.11,23 Additional studies evaluated the combination of epinephrine and vasopressin and also showed no benefit to survival to hospital discharge or ROSC compared to epinephrine alone.24 Given that vasopressin has been shown to offer no advantage when used in substitution or in addition to epinephrine, it has been removed from the Adult Cardiac Arrest Algorithm.11
Steroids
Cortisol levels have been found to be low both during and after CPR. The response to cardiac arrest and ROSC is an increase in cytokines, endotoxin release, coagulopathy, and adrenal insufficiency, which all contribute to post-resuscitation shock.25,26 It is theorized that corticosteroids during and after CPR may result in better post-ROSC hemodynamics.
Two studies from Greece have looked at a vasopressin-steroids-epinephrine (VSE) combination during cardiac arrest, followed by a steroid supplementation afterward. The doses used in these studies were 20 IU of vasopressin plus the standard 1 mg dose of epinephrine per cycle of CPR for five cycles, followed by epinephrine alone. In addition, a one-time dose of methylprednisolone (40 mg) during the first cycle was added. Four hours after ROSC, in patients who continued to have shock, a stress-dose of hydrocortisone (300 mg daily for seven days max with taper) was given. To reduce the effects of steroids on infarct healing, a shorter course of three days or less was used in patients with evidence of acute myocardial infarction.26,27
These VSE studies showed improved outcomes, including survival to hospital discharge with good neurological outcome, when compared to epinephrine alone and placebo.27 Of note, these studies only examined IHCA. Investigation of OHCA, with steroids as a sole treatment, rather than the VSE bundle, reveals mixed results and currently is of uncertain benefit.27,28
Based on this data, the current recommendations state that there are not sufficient data to recommend for or against steroids alone (class IIb recommendation). A VSE bundle followed by post-arrest hydrocortisone may be considered, but further studies are needed before recommendations can be made for routine use.11
Antiarrhythmic Drugs During and After Cardiac Arrest
Currently, there are no recommendations or indications for antiarrhythmic drugs in asystole or pulseless electrical activity (PEA). Although there are indications for antiarrhythmic medications in other arrhythmias, discussion here will be limited to the use of these medications in cardiac arrest (ventricular fibrillation and pulseless ventricular tachycardia).
Refractory ventricular fibrillation (VF) or pulseless ventricular tachycardia (pVT) has been defined variably, but will be defined here as VF or pVT that either recurs or persists after one or more defibrillation attempts, as this is the threshold for administering antiarrhythmic medication during a cardiac arrest. The goal is not to pharmacologically convert the rhythm, but rather to assist in the electrical termination of VF/pVT and to restore an organized, perfusing rhythm.
None of the antiarrhythmic medications have yet to show an improvement in long-term survival or survival with good neurological outcome, although some studies show increased rates of ROSC and survival to hospital admission.29,30,31 Due to these findings, the current guidelines emphasize that during CPR, attempts to obtain vascular access and to administer medications should not interfere with high-quality CPR or timely defibrillation, both of which have been proven to improve survival.
Amiodarone. Amiodarone acts on potassium, calcium, and sodium channels and also possesses some alpha- and beta-blocking properties.14 IV amiodarone is available in two formulations in the United States. The traditional formulation of amiodarone contains polysorbate 80, which is a vasoactive solvent and can cause unwanted hypotension. A new formulation has been introduced that contains Captisol and has no vascular effects.11,14,32
Based on current studies, the AHA continues to recommend amiodarone (300 mg IV/IO initially, followed by supplemental 150 mg IV/IO if needed) in a patient with VF/pVT and not resuscitated by CPR, defibrillation, and epinephrine (class IIb).11 In the studies of amiodarone, patients were considered refractory after at least three failed defibrillation attempts. These studies only were able to demonstrate improved ROSC and survival to hospital admission with amiodarone administration.30,31,33 Thus far, studies have failed to show any increased long-term survival or improved neurologic outcomes in the VF/pVT population.30
Lidocaine. Lidocaine acts as an antiarrhythmic agent by blocking voltage-gated sodium channels to increase the depolarization threshold.34 Recent reviews have shown that both amiodarone and lidocaine are the only agents studied to date that are associated with improved survival to hospitalization after OHCA.29 In a randomized clinical trial, lidocaine was shown to be less effective than amiodarone in survival to hospital admission, but no difference between the two medications was identified in survival to hospital discharge.33 The 2015 guidelines state that lidocaine may be considered as an alternative to amiodarone in refractory VF/pVT as a bolus of 1-1.5 mg/kg IV/IO (class IIb recommendation).11,29
Magnesium. Magnesium acts as a vasodilator and also helps regulate sodium, potassium, and calcium transport across cell membranes.35 The use of IV magnesium has been proven to be effective at terminating polymorphic VT.14 Most cases of polymorphic VT are self-terminating, but in the unusual case of sustained polymorphic VT, defibrillation is indicated, followed by consideration of IV magnesium sulfate (bolus of 1 to 2 g IV). The routine use of magnesium for VF/pVT is not currently recommended.11
New Medical Therapies
Despite advances in resuscitation science, mortality and survival with neurologically favorable outcomes from OHCA have remained stable over the past 30 years. New research is beginning to target alternative pathways to improve survival from cardiac arrest. These studies are evaluating treatments to reduce injury caused by ischemia and reperfusion. Novel therapies include use of beta-blockers, vasodilator therapy, mitochondrial protective therapies, and therapies aimed at decreasing calcium overload.36 Further studies are needed in these areas, but these therapies potentially could lead to improved cardiac arrest outcomes.
New Recommendations for Lay Rescuers
The newest guidelines put an emphasis on instruction for lay rescuers. Perhaps the most significant change is that rescue breaths are no longer recommended for the lay rescuer. The AHA recommends continuous chest compressions or hands-only CPR. This change is based on two findings. First, the lay public often did not perform CPR because they were fearful of contracting a disease with mouth-to-mouth resuscitation. Second, mouth-to-mouth breath provides less than 21% FIO2 delivered to the patient, as a fraction of the atmospheric oxygen is consumed by the rescuer during inhalation to subsequently deliver the mouth-to-mouth breath. Studies have shown that passive ventilation with room air with chest compressions provides approximately 21% FIO2, thus this method provides superior oxygenation when compared to mouth-to-mouth ventilation.2
The AHA also recommends that bystanders be trained in administering naloxone to those with suspected opioid overdose.37 This is especially important in areas with high opioid overdose rates and even more specifically to those involved in any type of addiction rehabilitation care.
In an unusual step, the AHA made a recommendation on a specific means of contacting emergency services providers. The AHA recommends that citizens be able to contact EMS through social media to promote greater access to rapid emergency medical care. A survey conducted by the American Red Cross in 2012 showed that 44% of people would contact EMS through social media if they could not call 911. Social media applications such as Facebook and Twitter now have enabled the ability to contact 911 through their app or online.2
Special Circumstances
The AHA resuscitation guidelines from 2010 addressed 15 special circumstances of cardiac arrest, and a few of these were updated in 2015. These special circumstances are cardiac arrest in pregnancy, cardiac arrest due to PE, cardiac arrest associated with opioid overdose, cardiac arrest due to poisoning, and cardiac arrest during percutaneous coronary intervention (PCI).38
Cardiac Arrest Associated With Pregnancy
There are very few high-quality studies that address cardiac arrest during pregnancy; therefore, the guidelines provided by the AHA are based on physiologic principles and a few small observational studies. One focus of acute resuscitation in pregnancy is patient positioning. Research has shown that gravid patients greater than 20 weeks can have a decrease in cardiac output of up to 30% due to aortocaval compression from the gravid uterus, thus patient positioning is extremely important.39,40,41 The left lateral decubitus position is preferred for patients who do not require compressions.
However, studies have shown that compressions are more likely to be inadequate when the patient is in this position; therefore, when compressions are being performed, manual left lateral displacement of the uterus, rather than left lateral decubitus positioning, is the optimal method.42,43 Perimortem cesarean delivery should be performed by an appropriately trained provider within four minutes of the onset of cardiac arrest if ROSC is not achieved.38
Cardiac Arrest Associated With Pulmonary Embolism
In patients with a confirmed PE leading to cardiac arrest, thrombolysis, surgical embolectomy, and mechanical embolectomy are reasonable emergency treatment options (class IIa recommendation). There are not enough data to recommend one method over the other. In patients with suspected pulmonary embolism, thrombolysis may be considered (class IIb recommendation). Surgical embolectomy and mechanical embolectomy have insufficient evidence in cases of suspected PE.38
Cardiac or Respiratory Arrest Due to Opioid Overdose
It is recommended to give intranasal (IN), intramuscular (IM), or IV naloxone to cardiac arrest patients suspected of having an opioid overdose. EMS providers should not delay transport of the patient while waiting for a response to naloxone (class I recommendation). Any patient who receives naloxone should be transported to a facility with advanced healthcare services (class I recommendation).38
Cardiac Arrest Due to Local Anesthetic Poisoning
A systematic review of human case reports showed that the majority of patients in cardiac arrest due to local anesthetic toxicity who received IV lipid emulsion had significant improvement.44 This was specifically true for bupivacaine toxicity where intravenous lipid emulsion increased the rate of ROSC, increased BP during hypotension, resolved dysrhythmias, and improved mental status. The most common dose consisted of 20% emulsion of long-chain triglycerides given in an initial bolus of 1.5 mL/kg lean body mass over one minute, followed by an infusion of 0.25 mL/kg per minute for 30-60 minutes. Because of such findings, the AHA has made giving IV lipid emulsion in patients with local anesthetic systemic toxicity a class IIb recommendation.38,45
Cardiac Arrest During Percutaneous Coronary Intervention
The 2015 AHA guidelines for cardiac arrest during PCI state that ECPR may be a viable option in patients suffering a cardiac arrest during PCI because the arrest was due to a potentially reversible cause (class IIb recommendation). The AHA recommends implementation and adherence to institutional guidelines for the appropriate selection of patients for mechanical support devices to ensure that these devices are used as a bridge to recovery, surgery or transplant, or other treatment (class I recommendation).38
Possible Future Updates
Pulseless Electrical Activity
For years, the AHA guidelines have emphasized, and continue to emphasize, the “Hs and Ts” when it comes to PEA management (see Table 2); however, recent studies are showing improved outcomes from a more simplified approach.46,47,48,49 In these studies, other possible algorithms for wide vs. narrow QRS PEA are evaluated. (See Figure 1.)
Table 2. The Hs and Ts: Common Causes of Cardiac Arrest
6 Hs |
6 Ts |
|
|
Figure 1. Pulseless Electrical Activity Management
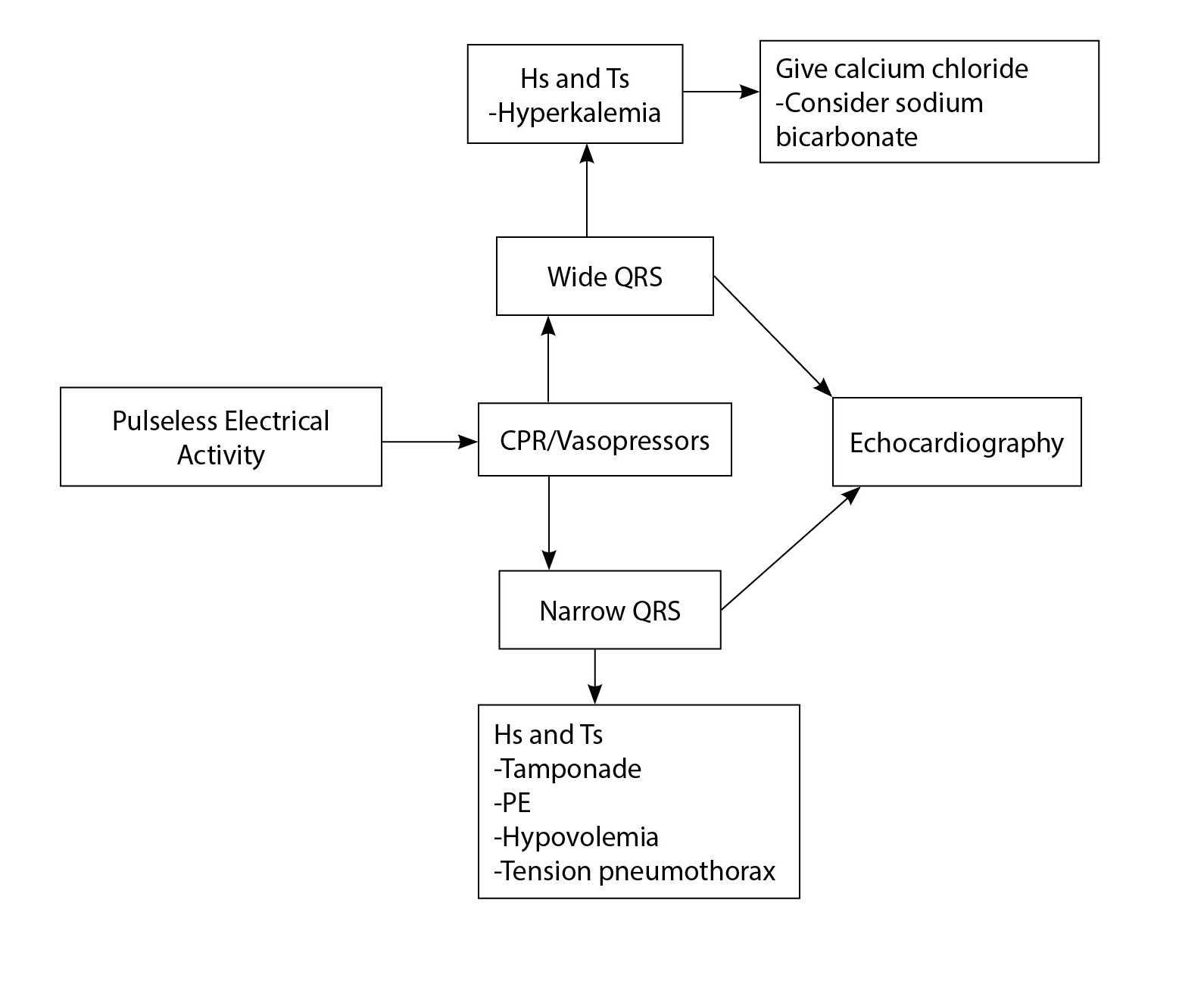
Initially, management is the same: CPR and vasopressors, specifically epinephrine. However, depending on the width of the QRS complex, management changes. For wide complex PEA, these studies suggest giving calcium chloride initially, and consider sodium bicarbonate. This will help if PEA is due to hyperkalemia or sodium channel blockers. For narrow complex PEA, initial action is fluid resuscitation and then consider PE, hypovolemia, tamponade, or tension pneumothorax. Interventions for both wide and narrow complex PEA should be followed by bedside echocardiography.
If there is no cardiac activity after these initial steps, these protocols recommend terminating CPR. If a large right ventricle and a small left ventricle are found, consider TPA for PE, needle decompression for pneumothorax, or disconnect the ventilator for possible hyperinflation. A small right ventricle with a small left ventricle could indicate hypovolemia or distributive shock; fluids, transfusions (if bleeding), and vasopressors should be initiated. With a pericardial effusion, tamponade physiology should be considered, and pericardiocentesis should be performed. Finally, if a hypocontractile left ventricle is seen on echocardiogram, vasopressors and inotropic agents should be continued with consideration for mechanical support.46,47,48,49
Double Sequential Defibrillation
Double sequential defibrillation (DSD) has been studied since 1986. The first study was conducted on dogs and showed promising outcomes.50 In this study, the authors showed that providing two defibrillations delivered one after the other using different pad placements to generate nonidentical current pathways through the heart was able to terminate experimental ventricular fibrillation in these animals with both reduced peak voltage and total energy. See Figure 2 for suggested double sequential defibrillation pad placement (see the online issue for this image in color).
Figure 2. Double Sequential Defibrillation Pad Placement
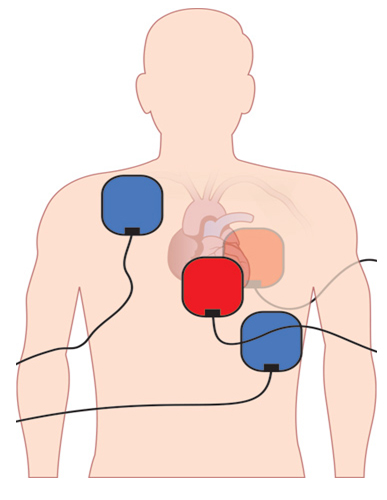
Image used with permission from: Journal of Emergency Medical Services (JEMS)
The first human study was conducted in 1994. In this study, five patients with refractory ventricular fibrillation received seven to 20 shocks at 200-360 joules before receiving DSD. In each of these refractory cases, the patient was defibrillated on the first attempt of DSD. The investigators used the anterior-posterior and apex-sternum orientations to deliver the shocks.51 Recognizing the limitation of this observational study, it showed that DSD does have potential in patients with refractory ventricular fibrillation.
In 2015, an EMS service published a study with 10 patients with refractory ventricular fibrillation after at least five failed attempts at single defibrillation with epinephrine and an antiarrhythmic medication having been delivered. DSD was successful in defibrillating seven patients, and three of those obtained ROSC; however, none of those patients survived.52
Although these studies have been limited, they do suggest the ability to terminate refractory ventricular fibrillation using DSD, which may be included in future recommendations as further studies are conducted and evidence is strengthened.
Post-Cardiac Arrest Care
Cardiac arrest management does not end with ROSC. Just as important for patient outcome is the implementation of post-cardiac arrest care to address both the cause of the arrest and the resulting physiologic abnormalities. Post-cardiac arrest syndrome (PCAS) consists of an ischemic/reperfusion response with brain injury and myocardial dysfunction.53 The majority of post-arrest patients are comatose and unable to provide a history. Good use of available resources, including EMS, family members, and the medical chart, is important. Pertinent factors to document are any known prodromal symptoms, CPR details (witnessed vs. unwitnessed arrest, bystander CPR, length of CPR, and initial rhythm), and any medications administered.54 Post-arrest management will be discussed by system below. See Table 3 for a systems-based management checklist.
Table 3. Post-resuscitation Checklist
Post-arrest Issues |
Management |
|
• Ischemia-reperfusion brain injury • Epileptiform activity • Possible intracranial process |
|
|
• Hypoxia/hypercarbia • Airway management • Pulmonary cause of arrest |
|
|
• Acute coronary syndrome • Hypotension • Arrhythmia |
|
|
• Hyperglycemia • Adrenal insufficiency |
|
|
• Electrolyte abnormalities • Ischemia-reperfusion acute kidney injury |
|
|
• Bacteremia, pneumonia, possible pre-arrest infections |
|
|
• Anemia • Uncontrolled bleeding |
|
Cardiovascular Care
Coronary Angiography. As soon as possible after ROSC is obtained, a 12-lead electrocardiogram (ECG) should be performed to look for any evidence of ST-segment elevation. This will help determine whether coronary angiography should be performed immediately.55 (See Figure 3.)
Figure 3. Post-ROSC ECG Algorithm
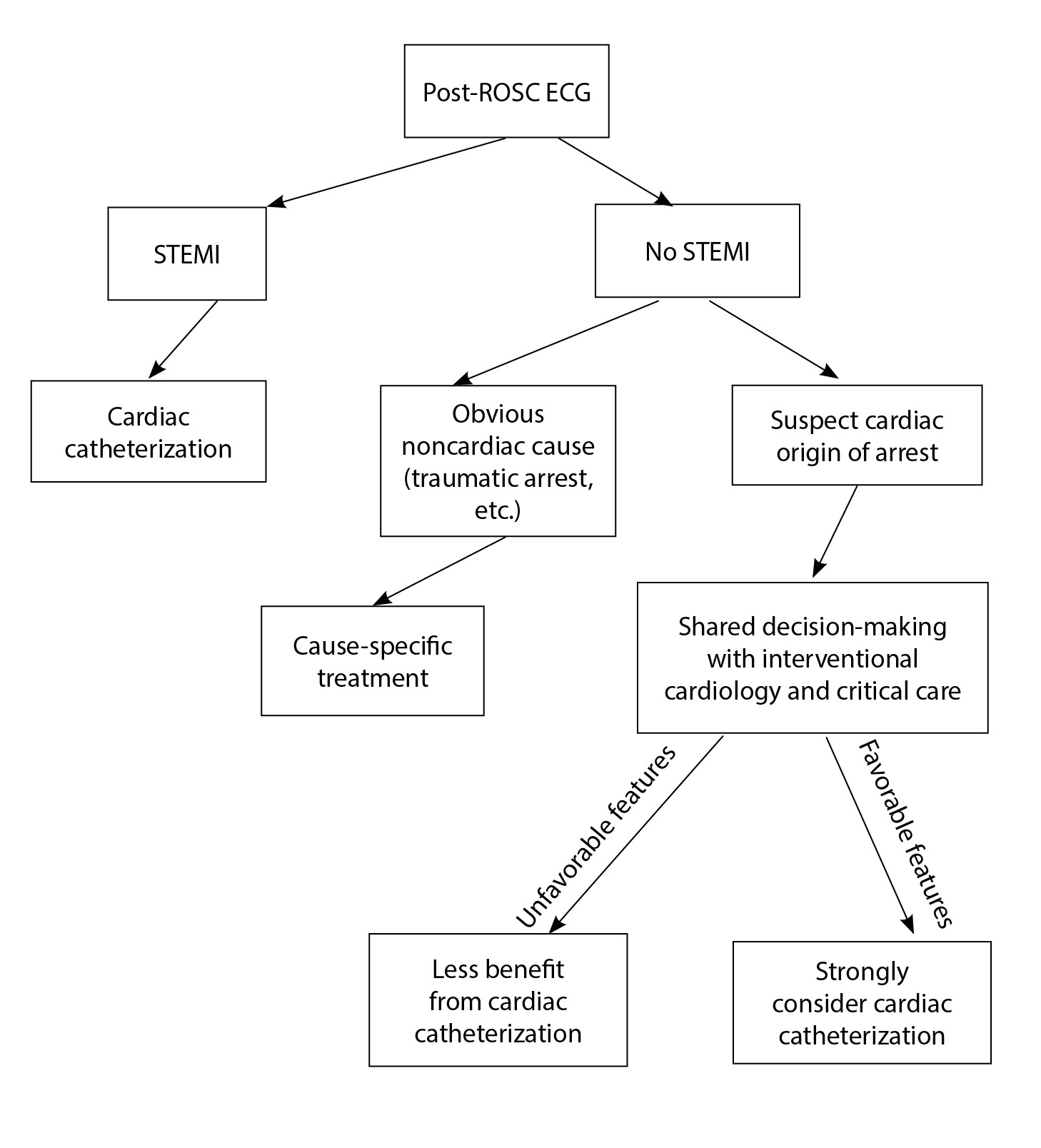
Unchanged from previous recommendations, patients with ST-elevation myocardial infarction (STEMI) should undergo emergent coronary angiography and PCI.55 Thus far, only observational studies have been published concerning post-resuscitation PCI, but these studies show improved survival to hospital discharge and improved neurological outcomes associated with emergent coronary angiography in post-arrest STEMI patients. These interventions are recommended regardless of the patient’s mental status or cooling status. The decision for cardiovascular intervention should be made without immediate determination of neurologic prognosis, as neurologic recovery cannot be predicted reliably in the initial post-arrest period.54,55
In patients with no STEMI on ECG, a culprit vessel is found in 33% of patients, with about 70% of these vessels being occluded.56 Thus, emergent catheterization is reasonable in patients suspected of having a cardiovascular cause of arrest, even in the absence of STEMI.55 The American College of Cardiology’s Interventional Council developed an algorithm to risk stratify OHCA patients with ROSC who remain comatose should undergo PCI, although it has not been prospectively validated yet.
In patients with no STEMI on post-arrest ECG, the AHA guidelines recommend assessing for unfavorable resuscitation features, including unwitnessed arrest, initial rhythm non-VF, no bystander CPR, > 30 minutes to ROSC, ongoing CPR, pH < 7.2, lactate > 7, age > 85 years, end-stage renal disease, or non-cardiac causes (e.g., traumatic arrest). Patients with multiple unfavorable resuscitation features are less likely to benefit from cardiac catheterization, but consultation with interventional cardiology still is strongly recommended.56
In a cohort study from France, cardiac catheterization was performed on all ROSC patients without an obvious non-cardiac cause of OHCA at admission (regardless of ST-segment elevation), with subsequent PCI when appropriate. This study found a nearly two-fold increase in the rate of favorable neurologic outcome (cerebral performance category 1 to 2 at discharge).57,58 More studies are needed, but emergent angiography is reasonable after OHCA of suspected cardiac origin but without ST-segment elevation on ECG.55
Hemodynamic Goals. Post-arrest patients often have hemodynamic instability. This often is multifactorial with contributions from the underlying cause of the arrest, along with subsequent ischemia-reperfusion injury, causing myocardial stunning and systemic inflammatory response. There is a lack of adequate data to establish optimal hemodynamic goals or interventional strategies to achieve them.55
It is thought that patients are relatively hypovolemic post-arrest due to venous pooling and capillary dysfunction, so initial fluid boluses of 1-2 liters should be considered, followed by additional fluids based on typical resuscitation measures.59 Following optimization of preload, vasopressors may be required.
Multiple observational studies have found that hypotension in the post-arrest period has been associated with higher mortality and worsening functional recovery. These same studies have shown that greater mean arterial pressure (MAP) is associated with better neurological recovery and survival.55,59,60 A specific MAP or systolic blood pressure (SBP) goal was not identified consistently in the studies, but published protocols target MAPs of > 65-80 mmHg.55,59
Based on these findings, the guidelines state that hypotension, defined as MAP < 65 mmHg or SBP < 90 mmHg, should be avoided and immediately corrected when present.61 It is recognized that optimal blood pressure will vary by patient due to baseline blood pressure and the underlying etiology of the patient’s cardiac arrest. Recommendations for maintaining blood pressure include first optimizing preload (fluids), followed by inotropes and vasopressors if hemodynamic goals are not met.
Arrhythmias. Antiarrhythmic medications should be used as needed.55 If patients continue to experience electrical instability, coronary angiography should be considered.60 Evaluation and correction of electrolyte abnormalities (calcium, potassium, magnesium, and phosphate) to the upper ends of normal also should be considered. If the patient is being cooled, consider rewarming to 36° C if the patient is unstable.
Neurological Care
A baseline neurologic exam should be performed on all patients achieving ROSC. Not only does this help determine the need for targeted temperature management (TTM), but it also helps determine possible causes of the arrest while providing a baseline for evaluating the future clinical course. It is important to remember that the exam must be performed not under the influence of paralytics or sedation, and the exam should be delayed if those medications were used prior to ROSC.
Temperature Control
Previously, it was recommended that post-arrest patients be treated with a therapeutic hypothermia protocol. Recently, data suggest that patients maintained in strict normothermia have no worsened outcome when compared to those treated with hypothermia. As a result, the AHA has adopted the term TTM to reflect close temperature maintenance within the normothermic to hypothermic range.
Several studies have evaluated the outcomes associated with TTM in OHCA with VF/pVT, OHCA with non-shockable rhythms, and after IHCA. There have been mixed results on TTM in non-shockable rhythms, although recent studies have shown more favorable outcomes.59 Current AHA recommendations are that comatose (lack of meaningful or purposeful response to verbal commands) patients with ROSC have TTM regardless of the initial rhythms.55 No ideal temperature or timing has been well studied to date.53,55,59,62 Reflecting the recent data that suggest more normothermic temperatures may be as beneficial as hypothermia, temperatures between 32° C and 36° C are recommended. It is also recommended that the temperature be maintained for at least 24 hours after the target is achieved.
When initiating TTM in the ED, it is important to include adequate sedation and management of shivering through both pharmacologic and nonpharmacologic methods. Typical sedatives used in ventilated patients are appropriate. Nonsteroidal anti-inflammatory drugs, acetaminophen, buspirone, or meperidine also can be used to control shivering. Skin counter-warming with warming blankets also can be effective. A last resort may include neuromuscular blockade if shivering is preventing target temperature from being obtained.53,55,63
Several trials have investigated pre-hospital hypothermia and found no direct benefit and some potential harm. Current recommendations are against routine prehospital cooling of patients after ROSC.55
Hyperthermia is associated with worsened neurologic outcomes in comatose patients for other conditions, but has not been well studied in post-arrest patients. Extrapolating these data to post-arrest patients, it is reasonable to actively prevent fever/hyperthermia in comatose patients. This is based on expert opinion and the thought that fever/hyperthermia prevention is relatively benign.53,55
Seizure Management
Another important consideration in the comatose post-arrest patient is seizure identification and management. The prevalence of convulsions, nonconvulsive status epilepticus, and other seizure activity in comatose patients after arrest is estimated to be between 12-22%.55,64 Some studies indicate that seizures most commonly occur in the first eight hours after ROSC, but they may be difficult to identify, as seizures often are masked by neuromuscular blockade as well.53 Although prophylactic administration of anticonvulsants is not recommended, it is recommended that an electroencephalogram (EEG) be obtained promptly in comatose patients after ROSC. Typical anticonvulsant treatments for status epilepticus should be used.55
Respiratory Care
Ventilator settings in the post-arrest patient should be individualized, and patient response should be monitored using end-tidal CO2, pulse oximetry, or arterial blood gases. In terms of ventilation, hypocapnia has been associated with worse outcomes, including neurological outcomes in some studies.53,65 This is likely due to cerebral vasoconstriction associated with hypocapnia. Normocapnia is a reasonable goal (end-tidal CO2 30-40 mmHg or PaCO2 35-45 mmHg), but as stated, adjustments must be made for individual patients. Permissive hypercapnia may be considered for lung protective ventilator settings.53,55
Similarly, management of oxygenation in the post-arrest period is important to optimize patient outcomes. Both hyperoxemia and hypoxemia are detrimental in this period. Hyperoxemia after ROSC contributes to free radical production, which can further damage already injured tissue. Hypoxemia is defined as arterial oxygen saturation of < 94%, and hypoxia as PaO2 < 60 mmHg.66 The causes of hypoxemia vary but can be related to lung contusions or pneumothorax from chest compressions, atelectasis, ventilator-associated lung injury, or from the original cause of the arrest (PE, acute respiratory distress syndrome [ARDS], etc.).53,55 Studies on hyperoxia have shown mixed results compared to definitive studies showing poor outcomes with low oxygen saturation.
Based on this, it is recommended to use the highest available oxygen concentration until a reliable assessment of oxygenation can be performed. When oxygen saturation is found to be 100%, it is best to titrate the FiO2 down while maintaining an oxygen saturation of 94% or greater.53,55,66
Endocrine Care
The two major areas to improve endocrine function in post-arrest patients are glycemic control and possible pituitary-adrenal insufficiency. The use of steroids was discussed in the medication section. Glycemic control in critically ill patients is highly controversial, although studies have shown moderate control is superior to tight control.67 Early hyperglycemia after cardiac arrest has been associated with unfavorable neurologic outcomes in several studies.53 There is no evidence that a specific blood glucose range has improved outcomes in the post-arrest population, but avoiding extremes on either side is reasonable.53,55
Renal Care
Resuscitated patients typically develop severe metabolic and electrolyte abnormalities. This is related to both the period of resuscitation and the ischemia-reperfusion injury in the post-arrest period.68 All serum electrolytes should be monitored closely, but it seems that potassium and magnesium have the biggest potential for impact.54 More studies are needed to determine whether attempting to correct pH with bicarbonate or ventilator management is helpful. Secondary renal injuries also are common. These typically are transient, but care must be taken to avoid nephrotoxic drugs if acute kidney injury is present.54,68
Infectious Disease Care
Infection and sepsis may play a role in both pre- and post-arrest periods. Studies have found that pneumonia is present in close to half of OHCA patients undergoing TTM.69,70 In addition, bacteremia was seen in 38% of patients presenting with OHCA in one study.71 Although the AHA has not made any recommendations on the use of empiric broad-spectrum antibiotics in the post-arrest period, it may be an area of interest in the future. Consideration should be given to obtaining blood cultures on post-arrest patients. Aggressive respiratory care to prevent pneumonia also should be initiated in the ED and continued in the critical care setting, including elevation of the head of the bed and standard pulmonary toilet.72
Hematologic Care
Blood counts and coagulation studies should be checked in all post-arrest patients. Anemia may have contributed to arrest and should be corrected with blood products as needed, along with attempts to determine the source of blood loss.54 Patients with active bleeding that is non-compressible in nature should not undergo therapeutic hypothermia, as hypothermia causes coagulopathy. This is most prevalent at temperatures below 35° C, when clotting and platelet function are impaired.73 If the patient is already cooled, it may be appropriate to raise the temperature to 36° C, while maintaining a TTM protocol to prevent hyperthermia.54,55
Conclusion
Resuscitation during a cardiac arrest and post-resuscitation management are complex processes. The recommendations and algorithms developed by the AHA are meant to serve as a guide to ensure the best possible patient outcomes based on the most up-to-date evidence. Although there are opportunities for further research, evidence-based management of the cardiac arrest patient is most likely to improve patient survival and neurologically favorable outcomes.
REFERENCES
- Hermreck AS. The history of cardiopulmonary resuscitation. Am J Surg 1988;156:430-436.
- Neumar RW, et al. Part 1: Executive Summary: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015;132(18 Suppl 2):S315-S367.
- Kleinman ME, et al. Part 5: Adult Basic Life Support and Cardiopulmonary Resuscitation Quality: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015;132(18 Suppl 2):S414-S435.
- Idris AH. The sweet spot: Chest compressions between 100-120/minute optimize successful resuscitation from cardiac rest. JEMS 2012;37:4-9.
- Hellevuo H, et al. Deeper chest compression — more complications for cardiac arrest patients? Resuscitation 2013;84:760-765.
- Brooks SC, et al. Part 6: Alternative Techniques and Ancillary Devices for Cardiopulmonary Resuscitation: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015;132(18 Suppl 2):S436-S443.
- Cave DM, et al. Part 7: CPR techniques and devices: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2010;122(18 Suppl 3):S720-S728.
- Halperin H. Through the years. A brief history of mechanical CPR devices. JEMS 2009;Suppl:suppl 12-13.
- Wang HE, et al. Interruptions in cardiopulmonary resuscitation from paramedic endotracheal intubation. Ann Emerg Med 2009;54:645-652.e1.
- Benoit JL, et al. Endotracheal intubation versus supraglottic airway placement in out-of-hospital cardiac arrest: A meta-analysis. Resuscitation 2015;93:20-26.
- Link MS, et al. Part 7: Adult Advanced Cardiovascular Life Support: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015;132(18 Suppl 2):S444-S464.
- Dennis M, et al. Extracorporeal cardiopulmonary resuscitation for refractory cardiac arrest: A multicentre experience. Int J Cardiol 2017;231:131-136.
- Maekawa K, et al. Extracorporeal cardiopulmonary resuscitation for patients with out-of-hospital cardiac arrest of cardiac origin: A propensity-matched study and predictor analysis. Crit Care Med 2013;41:1186-1196.
- Beavers CJ, Pandya KA. Pharmacotherapy considerations for the management of advanced cardiac life support. Nurs Clin North Am 2016;51:69-82.
- Atiksawedparit P, et al. Effects of prehospital adrenaline administration on out-of-hospital cardiac arrest outcomes: A systematic review and meta-analysis. Crit Care 2014;18:463.
- Vandycke C, Martens P. High dose versus standard dose epinephrine in cardiac arrest — a meta-analysis. Resuscitation 2000;45:161-166.
- Larabee TM, et al. Vasopressors in cardiac arrest: A systematic review. Resuscitation 2012;83:932-939.
- Donnino MW, et al. Time to administration of epinephrine and outcome after in-hospital cardiac arrest with non-shockable rhythms: Retrospective analysis of large in-hospital data registry. BMJ 2014;348:g3028.
- Hubble MW, et al. Probability of return of spontaneous circulation as a function of timing of vasopressor administration in out-of-hospital cardiac arrest. Prehosp Emerg Care 2015;19:457-463.
- Hubble MW, Tyson C. Impact of early vasopressor administration on neurological outcomes after prolonged out-of-hospital cardiac arrest. Prehosp Disaster Med 2017;32:297-304.
- Hoyme DB, et al. Epinephrine dosing interval and survival outcomes during pediatric in-hospital cardiac arrest. Resuscitation 2017;117:18-23.
- Warren SA, et al. Adrenaline (epinephrine) dosing period and survival after in-hospital cardiac arrest: A retrospective review of prospectively collected data. Resuscitation 2014;85:350-358.
- Mentzelopoulos SD, et al. Vasopressin for cardiac arrest: Meta-analysis of randomized controlled trials. Resuscitation 2012;83:32-39.
- Gueugniaud PY, et al. Vasopressin and epinephrine vs. epinephrine alone in cardiopulmonary resuscitation. N Engl J Med 2008;359:21-30.
- Mentzelopoulos SD, et al. Vasopressin, epinephrine, and corticosteroids for in-hospital cardiac arrest. Arch Intern Med 2009;169:15-24.
- Mentzelopoulos SD, et al. Vasopressin, steroids, and epinephrine and neurologically favorable survival after in-hospital cardiac arrest: A randomized clinical trial. JAMA 2013;310:270-279.
- Botnaru T, et al. Improved neurologic outcomes after cardiac arrest with combined administration of vasopressin, steroids, and epinephrine compared to epinephrine alone. CJEM 2015;17:202-205.
- Tsai MS, et al. The effect of hydrocortisone on the outcome of out-of-hospital cardiac arrest patients: A pilot study. Am J Emerg Med 2007;25:318-325.
- McLeod SL, et al. Comparative effectiveness of antiarrhythmics for out-of-hospital cardiac arrest: A systematic review and network meta-analysis. Resuscitation 2017;121:90-97.
- Laina A, et al. Amiodarone and cardiac arrest: Systematic review and meta-analysis. Int J Cardiol 2016;221:780-788.
- Huang Y, et al. Antiarrhythmia drugs for cardiac arrest: A systemic review and meta-analysis. Crit Care 2013;17:R173.
- Captisol® and Nexterone®: Captisol. Available at: http://www.captisol.com/partnerships/case-studies/captisol-and-nexterone. Accessed Oct. 22, 2017.
- Dorian P, et al. Amiodarone as compared with lidocaine for shock-resistant ventricular fibrillation. N Engl J Med 2002;346:884-890.
- Dager WE, et al. Pharmacotherapy considerations in advanced cardiac life support. Pharmacotherapy 2006;26:1703-1729.
- Panahi Y, et al. The role of magnesium sulfate in the intensive care unit. EXCLI J 2017;16:464-482.
- Bartos JA, Yannopoulos D. Novelties in pharmacological management of cardiopulmonary resuscitation. Curr Opin Crit Care 2013;19:417-423.
- Correction to: Part 10: Special Circumstances of Resuscitation: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2016;134:e122.
- Lavonas EJ, et al. Part 10: Special Circumstances of Resuscitation: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015;132(18 Suppl 2):S501-S518.
- Bamber JH, Dresner M. Aortocaval compression in pregnancy: The effect of changing the degree and direction of lateral tilt on maternal cardiac output. Anesth Analg 2003;97:256-258, table of contents.
- Abengochea A, et al. Effect of lateral tilt angle on the volume of the abdominal aorta and inferior vena cava in pregnant and nonpregnant women determined by magnetic resonance imaging. Anesthesiology 2015;123:733-734.
- Kundra P, et al. Effect of positioning from supine and left lateral positions to left lateral tilt on maternal blood flow velocities and waveforms in full-term parturients. Anaesthesia 2012;67:889-893.
- Kim S, et al. Quality of chest compressions performed by inexperienced rescuers in simulated cardiac arrest associated with pregnancy. Resuscitation 2013;84:98-102.
- Butcher M, et al. Efficacy of cardiopulmonary resuscitation in the supine position with manual displacement of the uterus vs lateral tilt using a firm wedge: A manikin study. Anaesthesia 2014;69:868-871.
- Hoegberg LC, et al. Systematic review of the effect of intravenous lipid emulsion therapy for local anesthetic toxicity. Clin Toxicol (Phila) 2016;54:167-93
- Ciechanowicz S, Patil V. Lipid emulsion for local anesthetic systemic toxicity. Anesthesiol Res Pract 2012;2012:1-11.
- Myerburg RJ, et al. Pulseless electric activity: Definition, causes, mechanisms, management, and research priorities for the next decade: Report from a National Heart, Lung, and Blood Institute workshop. Circulation 2013;128:2532-2541.
- Littmann L, et al. A simplified and structured teaching tool for the evaluation and management of pulseless electrical activity. Med Princ Pract 2014;23:1-6.
- Desbiens NA. Simplifying the diagnosis and management of pulseless electrical activity in adults: A qualitative review. Crit Care Med 2008;36:391-396.
- Mehta C, Brady W. Pulseless electrical activity in cardiac arrest: Electrocardiographic presentations and management considerations based on the electrocardiogram. Am J Emerg Med 2012;30:236-239.
- Chang MS, et al. Double and triple sequential shocks reduce ventricular defibrillation threshold in dogs with and without myocardial infarction. J Am Coll Cardiol 1986;8:1393-1405.
- Hoch DH, et al. Double sequential external shocks for refractory ventricular fibrillation. J Am Coll Cardiol 1994;23:1141-1145.
- Cabañas JG, et al. Double sequential external defibrillation in out-of-hospital refractory ventricular fibrillation: A report of ten cases. Prehosp Emerg Care 2015;19:126-130.
- Nakashima R, et al. Critical care management focused on optimizing brain function after cardiac arrest. Circ J 2017;81:427-439.
- Rittenberger JC, et al; Post Cardiac Arrest Service. Postcardiac arrest management. Emerg Med Clin North Am 2015;33:691-712.
- Callaway CW, et al. Part 8: Post-Cardiac Arrest Care: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015;132(18 Suppl 2):S465-S482.
- Rab T, et al. Cardiac arrest: A treatment algorithm for emergent invasive cardiac procedures in the resuscitated comatose patient. J Am Coll Cardiol 2015;66:62-73.
- Booth CM, et al. Is this patient dead, vegetative, or severely neurologically impaired? Assessing outcome for comatose survivors of cardiac arrest. JAMA 2004;291:870.
- Dumas F, et al. Emergency percutaneous coronary intervention in post-cardiac arrest patients without ST-segment elevation pattern: Insights from the PROCAT II registry. JACC Cardiovasc Interv 2016;9:1011-1018.
- Jentzer JC, et al. Recent developments in the management of patients resuscitated from cardiac arrest. J Crit Care 2017;39:97-107.
- Jentzer JC, et al. Improving survival from cardiac arrest: A review of contemporary practice and challenges. Ann Emerg Med 2016;68:678-689.
- Pellis T, et al. The optimal hemodynamics management of post-cardiac arrest shock. Best Pract Res Clin Anaesthesiol 2015;29:485-495.
- Rabinstein AA. How cool it is: Targeted temperature management for brain protection post–cardiac arrest. Semin Respir Crit Care Med 2016;37:34-41.
- Randhawa VK, et al. Out-of-hospital cardiac arrest and acute coronary syndromes: Reviewing post-resuscitation care strategies. Can J Cardiol 2015;31:1477-1480.
- Rittenberger JC, et al. Frequency and timing of nonconvulsive status epilepticus in comatose post-cardiac arrest subjects treated with hypothermia. Neurocrit Care 2012;16:114-122.
- Eastwood GM, et al. Arterial carbon dioxide tension and outcome in patients admitted to the intensive care unit after cardiac arrest (reply). Resuscitation 2013;84:e105.
- Sutherasan Y, et al. Ventilation and gas exchange management after cardiac arrest. Best Pract Res Clin Anaesthesiol 2015;29:413-424.
- Ling Y, et al. Intensive versus conventional glucose control in critically ill patients: A meta-analysis of randomized controlled trials. Eur J Intern Med 2012;23:564-574.
- Bellomo R, et al. Metabolic and electrolyte disturbance after cardiac arrest: How to deal with it. Best Pract Res Clin Anaesthesiol 2015;29:471-484.
- Nielsen N, et al. Adverse events and their relation to mortality in out-of-hospital cardiac arrest patients treated with therapeutic hypothermia. Crit Care Med 2011;39:57-64.
- Mongardon N, et al. Infectious complications in out-of-hospital cardiac arrest patients in the therapeutic hypothermia era. Crit Care Med 2011;39:1359-1364.
- Coba V, et al. The incidence and significance of bacteremia in out of hospital cardiac arrest. Resuscitation 2014;85:196-202.
- Davies KJ, et al. Early antibiotics improve survival following out-of hospital cardiac arrest. Resuscitation 2013;84:616-619.
- Michelson AD, et al. Reversible inhibition of human platelet activation by hypothermia in vivo and in vitro. Thromb Haemost 1994;71:633-640.
The American Heart Association Advanced Cardiac Life Support guidelines are recognized as the authoritative reference for acute cardiopulmonary resuscitation. Emergency medicine providers should be thoroughly familiar with these guidelines.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
