Atrial Fibrillation in Sepsis: Should We Worry?
By Betty Tran, MD, MSc
Assistant Professor of Medicine, Pulmonary and Critical Care Medicine, Rush University Medical Center, Chicago
Dr. Tran reports no financial relationships relevant to this field of study.
New-onset atrial fibrillation (AF) is a common occurrence in the ICU. Over the past decade, there is increasing interest in its epidemiology, specifically in the population of critically ill patients with sepsis. Recent literature suggests that far from a transient complication of sepsis, new-onset AF is associated with worse short- and long-term outcomes. As such, exploring its potential causes and evaluating its overall management is warranted in hopes of discovering ways to prevent and treat AF with the goal of improving outcomes for patients with sepsis. What follows is a review of the incidence, risk factors, and outcomes of new-onset AF in sepsis and a discussion on AF management in this population.
INCIDENCE
Based on a systematic review of multiple studies published up to 2013, the incidence of new-onset AF in patients with sepsis is around 5% (range 3-10%).1 However, other investigators recently reported incidences of up to 35-40%.2,3 A more detailed analysis shows that the incidence of new-onset AF in patients with sepsis probably varies in studies depending on the severity of disease, with the mean incidence 8% (range 0-14%), 10% (range 4-23%), and 23% (range 6-46%) in patients with sepsis, severe sepsis, and septic shock, respectively.1 These findings are observed across several medical centers and ICU types (medical, surgical), and in both retrospective and prospective studies. In a study using a representative 5% sample of Medicare beneficiaries with sepsis, the authors estimated that 20,000-50,000 of them experience newly diagnosed AF during a hospitalization for sepsis annually.4
RISK FACTORS
Sepsis itself is a strong risk factor for new-onset AF in the critical care setting. Using administrative claims data from the California State Inpatient Database for 3,144,787 hospitalized adults, Walkey et al found that compared to those without severe sepsis, patients with severe sepsis exhibited a significantly increased risk of new-onset AF (odds ratio [OR], 6.82; 95% confidence interval [CI], 6.54-7.11; P < 0.001).5
Risk factors for new-onset AF in the context of sepsis can differ from traditional risk factors associated with chronic AF (i.e., AF occurring prior to or outside the context of sepsis). Although advanced age and even race6 have been reported as risk factors for AF in general, risk factors for new-onset AF in sepsis also include conditions that are not related to chronic cardiovascular disease, such as increased number of acute organ failures/dysfunction, mechanical ventilation, increased comorbidities, and use of pulmonary artery catheterization.1,3,4,5,7 These observations lend insight into the potential mechanisms by which sepsis can predispose to the development of AF.
OUTCOMES
New-onset AF in the setting of sepsis has been associated with increased length of stay (LOS) in multiple studies. In a retrospective observational study of patients admitted to a mixed medical-surgical ICU, the average ICU LOS for septic patients with AF who survived their ICU stay was almost triple that of septic survivors without AF (27.8 vs. 7.6 days; P = 0.0001), although this was a small study overall (n = 274; 159 ICU survivors).7 A prospective study of non-cardiac surgical patients with sepsis found similar results for ICU LOS for new-onset AF vs. non-AF patients (30 vs. 17 days; P = 0.017).8 The authors of a large, prospective cohort study of critically ill patients with sepsis at two tertiary centers in the Netherlands also found that median ICU LOS in patients with new-onset AF was significantly longer compared to those who never developed AF (7.5 vs. 4.1 days; P < 0.001).3
Another short-term outcome associated with new-onset AF during sepsis is in-hospital ischemic stroke. In a large, retrospective, population-based cohort of California inpatient administrative claims data, patients with severe sepsis and new-onset AF demonstrated a higher risk of in-hospital stroke compared to septic patients without new-onset AF (2.6% vs. 0.6%; OR, 2.70; 95% CI, 2.05-3.57; P < 0.001) and when compared to septic patients with pre-existing AF (OR, 3.63; 95% CI, 2.51-5.25; P < 0.001).5 Researchers did not observe this association when comparing septic patients with pre-existing AF to septic patients without AF. Overall, new-onset AF in critically ill patients in general (not necessarily with sepsis) is associated with increased mortality. A prospective study of 1,770 patients admitted to the medical and surgical ICUs at Vanderbilt University found that critically ill patients with any form of AF (new-onset or recurrent/prior AF) exhibited an increased risk of hospital death compared to no AF (OR, 1.62; 95% CI, 1.14-2.29; P = 0.007). However, this effect was magnified in the subpopulation of patients with sepsis (OR, 2.92; 95% CI, 1.52-5.60; P = 0.001).9
The authors of other prospective and large database studies have reported similarly increased risks.3,5 Interestingly, a retrospective cohort of admissions to the surgical and medical ICUs at a tertiary care center found that even subclinical cases of new-onset AF (diagnosed via 500 randomly selected 30-minute segments of continuous ECG recordings without an accompanying diagnosis code and confirmed on 12-lead ECG) were associated with two-fold higher hospital mortality compared to admission without AF.10
Extending findings from the acute care setting to longer-term outcomes, Walkey et al used claims data from a 5% sample of Medicare beneficiaries to identify 138,722 survivors of a hospitalization for sepsis.11 They reported that compared to patients without AF, those with new-onset AF during a hospitalization for sepsis demonstrated a higher five-year risk of hospitalization for heart failure (OR, 11.2% vs. 8.2%; hazard ratio [HR], 1.25; 95% CI, 1.16-1.34), ischemic stroke (5.3% vs. 4.7%; HR, 1.22; 95% CI, 1.15-1.47), and death (74.8% vs. 72.1%; HR, 1.04; 95% CI, 1.01-1.07).11 These findings challenge the notion that new-onset AF is a transient issue that disappears with resolution of sepsis.
POTENTIAL MECHANISMS
Whether new-onset AF acts as a surrogate marker for increased illness severity and subsequently poor prognosis in sepsis or whether it directly contributes to mortality and poor outcomes is not entirely clear. Compared to patients with chronic/recurrent AF or no AF, those with new-onset AF more frequently exhibit hypotension, experience organ failure, and often require more fluids and/or vasopressors.2,4,9 However, it is difficult to distinguish between cause and effect in the studies presented here given their observational nature and/or the use of administrative data. The sepsis state itself, in addition to other acute factors such as organ failures and use of mechanical ventilation, certainly can trigger AF via mechanisms of acute inflammation, rises in catecholamines, and myocardial injury/ischemia/dysfunction. It is conceivable that the poor outcomes seen with AF may be a result of a confluence of these other factors. Even though many studies referenced here adjusted for severity of illness via multiple variables, residual confounding still may exist. Furthermore, the interventions that often are used to treat severe sepsis, namely intravenous fluids and vasopressors, also can increase the risk of developing AF through atrial stretch and chronotropic effects.
On the other hand, theoretically, AF could effect worse outcomes directly. A rapid ventricular rate can contribute to hypotension in already hemodynamically unstable patients. New-onset AF could be a potential source for future cardioembolic stroke and contribute to sepsis-induced cardiac dysfunction, thereby accounting for some of the longer-term effects on post-discharge mortality and hospitalizations for heart failure and stroke described previously.11 It is highly plausible that some, if not all, of these factors play a role in the development of new-onset AF, with some factors carrying more weight depending on the patient and clinical scenario.
MANAGEMENT
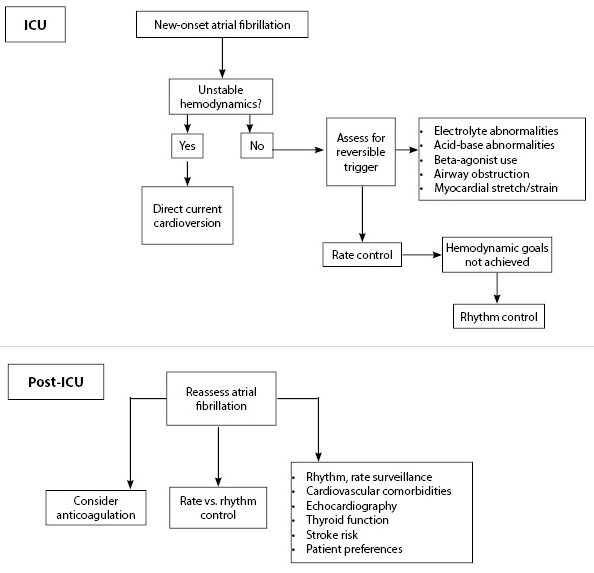
Little evidence is available to guide management of new-onset AF in the critical care setting. Direct current cardioversion (DCCV) should be employed for patients with hemodynamic instability related to the arrhythmia, even though the frequency of remaining in sinus rhythm may be low. Failure to remain in sinus rhythm also may signal a poor prognosis.8 For patients who are not acutely unstable, consideration should be given to reversible factors (e.g., electrolyte disturbances, airway obstruction, myocardial strain/stretch).
Beyond this, large-scale, direct comparisons among AF therapies in critically ill patients are lacking. Common options include beta-blockers, nondihydropyridine calcium channel blockers, digoxin, and amiodarone. A retrospective cohort study using a database representative of 20% of hospitalized patients in nonfederal U.S. hospitals found that there was wide variability in selection of an initial AF agent.12 Calcium channel blockers were the most commonly prescribed drug, although beta-blockers were associated with improved hospital mortality. However, unmeasured confounding may be responsible for this observed association, as more severe hypotension associated with sepsis may narrow provider options to either digoxin and amiodarone, thereby accounting for the worse outcomes observed with these medications compared to beta-blockers.12
Finally, the long-term poor outcomes associated with new-onset AF during acute illness suggest that close follow-up with outpatient providers for survivors will be important — namely, to optimize stroke prevention, as often-competing bleeding risks in the acute setting can limit initiation of therapeutic anticoagulation. Additionally, a discussion of the risks/benefits of rate vs. rhythm control is necessary. Figure 1 provides an overview to approaching management of new-onset AF developing during critical illness.
SUMMARY
New-onset AF in sepsis is common and associated with a higher inpatient LOS and overall mortality. The sepsis milieu provides several potential AF triggers, including systemic inflammation, endogenous and exogenous catecholamines, and direct and indirect effects on the myocardium. It is plausible that the more critical the patient (e.g., more organ failure, need for mechanical ventilation, etc.), the higher the risk of developing AF, and, subsequently, the higher overall morbidity and mortality.
On the other hand, new-onset AF in sepsis also is associated with poorer long-term outcomes, such as five-year risk of hospitalization for heart failure and stroke, and mortality, suggesting that its effects extend beyond the sepsis episode. Therefore, management of new-onset AF does not end with sepsis resolution, but should extend beyond discharge from the ICU to improve overall outcomes for sepsis survivors.
Figure 1. Approach to Short- and Long-term Management of New-onset Atrial Fibrillation in the ICU |
 |
|
Adapted from: Walkey AJ, Hogarth DK, Lip GYH. Optimizing atrial fibrillation management: From ICU and beyond. Chest 2015;148:859-864. |
REFERENCES
- Kuipers S, Klein Klouwenberg PM, Cremer OL. Incidence, risk factors and outcomes of new-onset atrial fibrillation in patients with sepsis: A systematic review. Crit Care 2014;18:688.
- Liu WC, Lin WY, Lin CS, et al. Prognostic impact of restored sinus rhythm in patients with sepsis and new-onset atrial fibrillation. Crit Care 2016;20:373.
- Klein Klouwenberg PM, Frencken JF, Kuipers S, et al. Incidence, predictors, and outcomes of new-onset atrial fibrillation in critically ill patients with sepsis: A cohort study. Am J Respir Crit Care Med 2017;195:205-211.
- Walkey AJ, Greiner MA, Heckbert SR, et al. Atrial fibrillation among Medicare beneficiaries hospitalized with sepsis: Incidence and risk factors. Am Heart J 2013;165:949-955.
- Walkey AJ, Wiener RS, Ghobrial JM, et al. Incident stroke and mortality associated with new-onset atrial fibrillation in patients hospitalized with severe sepsis. JAMA 2011;306:2248-2255.
- Gbadebo TD, Okafor H, Darbar D. Differential impact of race and risk factors on incidence of atrial fibrillation. Am Heart J 2011;162:31-37.
- Christian SA, Schorr C, Ferchau L, et al. Clinical characteristics and outcomes of septic patients with new-onset atrial fibrillation. J Crit Care 2008;23:532-536.
- Meierhenrich R, Steinhilber E, Eggermann C, et al. Incidence and prognostic impact of new-onset atrial fibrillation in patients with septic shock: A prospective observational study. Crit Care 2010;14:R108.
- Shaver CM, Chen W, Janz DR, et al. Atrial fibrillation is an independent predictor of mortality in critically ill patients. Crit Care Med 2015;43:2104-2111.
- Moss TJ, Calland JF, Enfield KB, et al. New-onset atrial fibrillation in the critically ill. Crit Care Med 2017;45:790-797.
- Walkey AJ, Hammill BG, Curtis LH, Benjamin EJ. Long-term outcomes following development of new-onset atrial fibrillation during sepsis. Chest 2014;146:1187-1195.
- Walkey AJ, Evans SR, Winter MR, Benjamin EJ. Practice patterns and outcomes of treatments for atrial fibrillation during sepsis. Chest 2016;149:74-83.
- Walkey AJ, Hogarth DK, Lip GYH. Optimizing atrial fibrillation management: From ICU and beyond. Chest 2015;148:859-864.
New-onset atrial fibrillation (AF) is a common occurrence in the ICU. Over the past decade, there is increasing interest in its epidemiology, specifically in the population of critically ill patients with sepsis. Recent literature suggests that far from a transient complication of sepsis, new-onset AF is associated with worse short- and long-term outcomes. As such, exploring its potential causes and evaluating its overall management is warranted in hopes of discovering ways to prevent and treat AF with the goal of improving outcomes for patients with sepsis.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
