Hemorrhage Control: Advances in Trauma Care
March 1, 2018
Reprints
AUTHORS
Creagh Boulger, MD, Associate Professor, Department of Emergency Medicine, The Ohio State University Wexner Medical Center, Columbus
Betty Yang, MD, Clinical Instructor, House Staff, Department of Emergency Medicine, The Ohio State University Wexner Medical Center, Columbus
PEER REVIEWER
Ademola Adewale, MD, Director of Research and Simulation, Florida Hospital Emergency Medicine Residency, Orlando
EXECUTIVE SUMMARY
- In the guidelines from the Eastern Association for the Surgery of Trauma (EAST), a lower threshold for trauma activation and a high level of care are recommended for geriatric trauma.
- Damage control resuscitation is a multifaceted method for hemorrhage control that includes mechanical hemostasis, limited crystalloid and artificial colloids, permissive hypotension, and balanced resuscitation.
- Recent updates include 1:1 ratios of packed red blood cells to fresh frozen plasma and platelets. This appears to reduce the risk of trauma-induced coagulopathy.
- Tourniquets can provide simple, cost-effective, lifesaving care; however, consideration should be given to certain rare potential complications, such as nerve palsies and ischemia.
- When placing a tourniquet, it is important to document a neurovascular exam prior to placement, as well as the time at which the tourniquet was placed.
- Some military studies suggest a limit of two hours, with take down of the device and assessment every two hours.
- Combat Gauze, Celox Guaze, and Chito Gauze are deemed safe and efficacious and, thus, are considered first-line recommended hemostatic agents by the Committee on Tactical Combat and Casualty Care (CoTCCC). These agents should be packed into the wound, and the provider should apply three to five minutes minimum of direct pressure, followed by a pressure dressing.
- Pelvic fractures can be life-threatening, with mortality reported as ranging between 7.6% and 50% for open pelvic fractures. When an unstable pelvic fracture is suspected or diagnosed, Advanced Trauma Life Support guidelines advise the use of a pelvic circumferential compression device as a temporizing measure until definitive fixation can be achieved.
- Tranexamic acid (TXA) is a synthetic lysine analogue and competitive inhibitor of plasminogen. The EAST guidelines for damage control resuscitation conditionally recommend early TXA for the severely hemorrhaging trauma patient.
Massive hemorrhage is a devastating scenario. Early identification is essential to allow for prompt, potentially lifesaving interventions with the goal of rapid control of exsanguination. Over the last two decades, advances in hemorrhage control and damage control resuscitation have facilitated minimizing blood loss pending definitive surgical intervention. Understanding the utilization of these interventions and procedures may result in a significant improvement in morbidity and mortality for the patient with hemorrhage.
— Ann M. Dietrich, MD, Editor
Case Study
You are working in a single coverage, rural emergency department when a 70-year-old male presents after a motor vehicle collision with HR 103, BP 80/p, evident bruising to his flank, and an unstable pelvis. A second patient presents from the same accident. She is a 20-year-old female with a crush injury, with HR 130, BP 70/p, and a partial right upper extremity amputation. What injuries should be considered? What is the best way to stabilize these patients? What are current advances, pharmacologic and nonpharmacologic, to stabilize these patients?
This article will discuss management strategies for hemorrhage control.
Introduction
Epidemiology
Trauma is a significant global health issue, leading to more than 4.6 million deaths annually worldwide.1 In the United States, injury is the primary cause of death in people ages 1 to 44 years, and the fourth leading cause of death overall.2 Death from hemorrhage often occurs rapidly, with a median time of two to three hours after presentation, and about 50% of these deaths occur within the first 24 hours.3,4,5
Many of these deaths are preventable. Early recognition of subtle but potentially significant hemorrhage, an understanding of the underlying physiology, and timely and appropriate utilization of resources can decrease morbidity and mortality. This has led to the development of a multimodal approach, called damage control resuscitation (DCR), with the goal of minimizing blood loss until definitive hemostasis may be achieved.6
Hemorrhage control in trauma has changed drastically over the last two decades, with better understanding of the physiologic changes from trauma and improved approaches to resuscitation.6 Crucial management begins in the prehospital setting.
Patient Population
Trauma is a unique disease process, as it often affects the young and otherwise healthy individual. However, trauma does not discriminate. Patients with a significant mechanism of injury, at the extremes of life (pediatrics and geriatrics), and with medical co-morbidities and polypharmacy are at higher risk for hemorrhage, shock, and mortality.
Mechanism of Injury
The mechanism of injury is a useful screening tool to determine which patients may be at risk for significant hemorrhage. Advanced Trauma Life Support (ATLS) guidelines describe significant mechanisms of injury as falls from a critical height of 6 m (20 ft),7 high-energy deceleration impacts, and penetrating wounds.8 It is important to have a higher index of suspicion to facilitate the early identification and management of potential injuries.
Blunt trauma can lead to occult injuries. A deceleration injury involves a difference in speed between fixed and non-fixed parts of the body, causing injuries such as liver and spleen lacerations and bowel damage. The most commonly injured organs in blunt trauma injuries are the spleen (40-55%), liver (35-50%), and small bowel (5-15%), with the occult injury of retroperitoneal hematoma.8
Penetrating trauma leads to different types of injuries. Stab wounds and low-velocity gunshot wounds cause direct tissue damage with laceration and cutting. Stab wounds most commonly cause injury to the liver (40%), small bowel (30%), diaphragm (20%), and colon (15%).8 Gunshot wounds can be of low velocity or high velocity. High-velocity gunshot wounds transfer more kinetic energy and cause more damage around the track of trajectory. Explosive devices can cause combined blunt and penetrating damage.
Elderly Patients
The geriatric population is a high-risk group for trauma. In the guidelines from the Eastern Association for the Surgery of Trauma (EAST), a lower threshold for trauma activation and a high level of care are recommended for geriatric trauma patients. This population is at increased risk for adverse outcomes after injury due to comorbidities, limited cardiovascular reserve, and general frailty. In a study by Calland et al, those patients with a base deficit of -6 mEq/L or less correlated with severe injury and significant mortality, up to 60% risk, compared to those with -5 mEq/L and with less than 23% mortality.9 Elderly patients frequently take anticoagulation and antiplatelet agents that increase the risk of hemorrhage; these iatrogenic coagulopathies should be recognized, evaluated, and corrected.9
Complications of Trauma
Trauma is a complex process. The injuries themselves often pose a significant risk to the patient. However, the complications associated with trauma can carry a significant increase in morbidity and mortality as well.
Hemorrhagic Shock. Early detection and management of hemorrhagic shock is vital to survival. Multiple studies have been performed to determine early signs of hemorrhagic shock and predict the requirement of blood products.
In severely injured patients, the lethal triad of acidosis, coagulopathy, and hypothermia worsen morbidity and mortality.
Trauma-induced Coagulopathy. One-third of patients with bleeding from trauma show signs of coagulopathy on admission to hospitals.5,10 Trauma-induced coagulopathy has been associated with blood product transfusion recommendations, with significant associated morbidity and mortality. Recent updates to existing protocols have been made to include 1:1 ratios of packed red blood cells to fresh frozen plasma and platelets. This appears to reduce the risk of trauma-induced coagulopathy.5,10
Diagnostics
Hemodynamics: ATLS Classification of Blood Loss
The ATLS classification of hemorrhagic shock is a useful guide to quantify blood loss, but, interestingly, it has not been validated.11 (See Table 1.) Blood loss may be obvious in some patients, but even in these patients, vital signs and visualization of blood loss are not good estimates of hemorrhagic shock.12,13 New scoring systems to predict the extent of shock and injury have been developed. However, many of these are not practical for application in a timely fashion and none have taken hold as a new standard of care.
Table 1. Classes of Hemorrhagic Shock
I |
II |
III |
IV |
|
|
Adapted from: American College of Surgeons Committee on Trauma. ATLS® Student Manual, 9th ed. Chicago, IL: American College of Surgeons; 2012 |
||||
|
Blood loss |
Up to 750 mL |
750 to 1,500 mL |
1,500 to 2,000 mL |
> 2,000 mL |
|
Blood loss (% blood volume) |
Up to 15% |
15% to 30% |
30% to 40% |
> 40% |
|
Pulse rate (per minute) |
< 100 |
100 to 120 |
120 to 140 |
> 140 |
|
Blood pressure |
Normal |
Normal |
Decreased |
Decreased |
|
Pulse pressure (mmHg) |
Normal or increased |
Decreased |
Decreased |
Decreased |
|
Respiratory rate (per minute) |
14 to 20 |
20 to 30 |
30 to 40 |
> 35 |
|
Urine output (mL/hour) |
> 30 |
20 to 30 |
5 to 15 |
Negligible |
|
Central nervous system/mental status |
Slightly anxious |
Mildly anxious |
Anxious, confused |
Confused, lethargic |
In many disease processes, laboratory studies can be helpful in diagnosing and risk-stratifying patients. Unfortunately, because of the close proximity of most traumatic events to hospital presentation, laboratory studies and other diagnostics generally are not helpful in risk-stratifying this population. The following sections briefly discuss a few studies and their utility and pitfalls.
End Tidal CO2
Low end tidal CO2 (ETCO2), CO2 < 35, may be a novel early predictor for hemorrhagic shock and the need for massive transfusion. A study by Caputo et al demonstrated that nasal cannula ETCO2 had a negative association with lactate and was predictive of operative intervention in penetrating trauma patients.14 Low ETCO2 had strong association with standard indicators for shock and was predictive of patients meeting CAT+ (≥ 3 units of packed red blood cells/h) criteria in the first six hours after admission.
Hemoglobin and Hematocrit
In nontraumatic causes of hemorrhagic shock, hemoglobin and hematocrit often are used to assess severity of blood loss, ongoing hemorrhage, and response to treatment. However, in trauma patients, these values often are normal on initial presentation and may take hours to drop.15,16 Serial measurements of hematocrit have a high sensitivity for detecting ongoing blood loss.17 In a study by Bruns et al, an initial hemoglobin below 10 g/dL in the first 30 minutes of patient arrival was associated with a three times higher need for intervention.18
Base Deficit and Lactate
Base deficit and lactate are measures of perfusion. Both of these studies have been shown to correlate with mortality and adequacy of resuscitation.19,20 Elevated lactate levels at presentation have been shown to correlate to mortality in trauma patients.21 In addition, failure to clear lactate in the first 24 hours was shown to be a strong negative prognostic marker after injury.21
Coagulation Markers
Standard laboratory evaluation of coagulation is the following: prothrombin time (PT), activated partial thromboplastin time, platelet counts, and fibrinogen. However, these tests only monitor the initiation of clot production and may appear normal despite the patient being coagulopathic. Viscoelastic hemostatic assays (VHA), such as thromboelastography (TEG) and rotational thromboelastometry (ROTEM), which promptly evaluate the entire clotting process, are relatively new and may be used to direct blood product resuscitation.22,23,24 Early amplitudes in the VHA, TEG, and ROTEM provide fast results, which is critical in the resuscitation of bleeding patients. Introducing the use of early amplitudes can reduce time to diagnosis of coagulopathy and may be used in TEG monitoring of trauma patients.25 The VHAs appear to show promise in tailoring resuscitation to the individual patient as long as they can have quick turnaround times and be cost effective.
Management
Damage control resuscitation (DCR) is a multifaceted method for hemorrhage control. This approach includes mechanical hemostasis, limited crystalloid and artificial colloids, permissive hypotension, and balanced resuscitation.6,26
This is a drastic change from 20 years ago, when trauma teams would infuse 20 to 30 liters of crystalloid, transfuse 10 units of red blood cells, and stay in the operating room to repair all injuries and close the fascia on all patients.27 Mortality was high, and survivors suffered from coagulopathy, renal failure, abdominal compartment syndrome, wound complications, and acute respiratory distress syndrome.26,27 Many studies in the combat setting have advanced the literature about optimal trauma resuscitation and management. The main principles are described in Table 2, adapted from the EAST guidelines on DCR.6
Table 2. Principles of Damage Control Resuscitation
- Avoid/reverse hypothermia.
- Minimize blood loss and apply early hemorrhage control measures.
- Target low-normal blood pressures before definitive hemostasis.
- Minimize crystalloid administration.
- Use massive transfusion protocol to ensure blood product is given in pre-specified ratio.
- Avoid delays to surgical and/or angiographic hemostasis.
- Obtain functional laboratory measures to guide resuscitation.
- Give pharmacologic adjuncts to safely promote hemostasis.
Adapted from EAST guidelines. Cannon JW, Khan MA, Raja AS, et al. Damage control resuscitation in patients with severe traumatic hemorrhage. J Trauma Acute Care Surg 2017;82:605-661.
Prehospital
Hemorrhage in the field remains the leading preventable cause of death in combat and the second leading cause of death in the civilian setting.3,4,5 Patients with traumatic injuries are on the clock. While there is minimal evidence behind the historical “golden hour,” we know that patients with active hemorrhage who achieve hemostasis and receive definitive care sooner have better outcomes.28,29 Many of the principles described in the EAST guidelines, such as normothermia, hemorrhage control, and conservative fluid administration, can and should be initiated in the field to optimize conditions for the patient. The subsequent section will discuss means of hemostasis that can be started in the field.
Direct Pressure and Pressure Dressings
Direct pressure and pressure dressings often are the first line of defense in hemorrhage. This technique requires little to no resources and has been shown to be an effective and safe means of hemostasis for smaller wounds.30 Successful direct pressure usually involves irrigating the wound, identifying the bleeding vessel, and applying direct one- to two-finger manual compression of the region uninterrupted for five to 15 minutes.31 The downside of this technique is that the source is not always easily visualized, the method requires personnel available to attend only to that wound, and the technique is useful only for smaller wounds with a clear, single source.
This technique can be combined with a pressure dressing, with or without hemostatic agents, and possible temporary closure. When applied, pressure dressings should not be bulky and should be tight enough to provide hemostasis while maintaining distal perfusion and sensation. If distal neurovascular status is compromised, the pressure dressing now begins to function as a tourniquet. Some commercial products are available for temporary wound hemostasis. Wound closure clamps essentially grab the wound edges, pull them together, and lock in place. Wound closure clips are placed on the wound margin, typically the scalp, to provide compression on the edges. Both of these devices typically can be applied in a minute or less. The drawbacks of such devices are cost and availability.
Temporizing Mechanical Compression Devices
Multiple hemorrhage control devices exist, including truncal, junctional, extremity, intravascular, and intraperitoneal. These are temporary adjuncts, primarily for the prehospital setting and initial hospital assessment phase, until definitive hemostasis can be achieved.
Extremities Trauma: Tourniquets
Exsanguination from extremity trauma is a preventable cause of death.32,33 Tourniquets have been well studied on the battlefield and found to have significant association with lives saved, leading to a policy of all military personnel carrying tourniquets.33
In 2009, Kragh et al published a prospective study that was conducted in Baghdad over seven months in 2006. They evaluated 2,838 injuries and admitted civilian and military casualties with major limb trauma. A total of 232 patients had 428 tourniquets applied to 309 limbs. Early tourniquet placement, prior to the onset of shock, was associated strongly with survival.33
Tourniquets serve as the standard of care for control of severe external hemorrhage in the military setting, even though civilian data remain limited.34,35 Both the American College of Surgeons Committee on Trauma (ACS-COT) and the American College of Emergency Physicians (ACEP) recommend tourniquets in the setting of life-threatening extremity hemorrhage.36,37
If indicated, placement of a tourniquet before extrication and transport is advised whenever possible. Tourniquets have a few varieties. The common categories are open or closed, and manual or pneumatic. Open tourniquets do not require the provider to fully cinch around a limb prior to engaging the tourniquet, and they come apart completely for placement around the limb. Pneumatic tourniquets tend to be more expensive than the manual versions. Despite these differences, most studies demonstrate similar hemostasis among the various models when applied properly.38,39 Multiple commercial tourniquets are available. Common items, such as belts, sheets, and other cords, may be used, but they are less efficient.40 Tourniquet placement should be maintained until definitive surgical management can be accomplished. 26,37,41
Tourniquets can provide simple, cost-effective, lifesaving care; however, consideration should be given to certain rare potential complications, such as nerve palsies and ischemia.33 When placing a tourniquet, it is important to document a neurovascular exam prior to placement, as well as the time at which the tourniquet was placed. Some military studies suggest a limit of two hours, with take down of the device and assessment every two hours.42 Other studies have shown limb viability up to six hours with tourniquet use.41 Additional tourniquets can be applied if the first did not achieve hemostasis. Once hemostasis has been achieved, the tourniquet should not be removed until definitive surgical management is possible.
Junctional Hemorrhage and Other Sites of Significant Blood Loss
Junctional hemorrhage is bleeding that occurs at the junction of an extremity with the abdomen and pelvis that cannot be controlled adequately by an extremity tourniquet.43 Four mechanical devices have been FDA-approved for junctional hemorrhage, but they are not well studied. The Committee on Tactical Combat Casualty Care recommends three of the four devices.43 Another type of junctional hemorrhage control device also has been approved for axillary use.
Meusnier et al evaluated the Combat Ready Clamp (CRoC) compared to the SAM Junctional Tourniquet (SJT) for ease of use and complete interruption of popliteal flow. Both devices were equally effective in arterial occlusion, but the SJT was preferred by users for ease of application and had quicker “hemostasis” in this small study.44 In a study by Rall et al, the Abdominal Aortic and Junctional Tourniquet (AAJT) was able to increase mean arterial pressure (MAP) in hemorrhagic swine without any clear adverse consequences.45 These devices have been used and studied primarily in simulation and military settings. Many of these devices require assembly and can be very bulky to store and transport. Their broad applicability to the civilian setting has yet to be demonstrated.46
Hemostatic Agents
Since direct manual pressure and tourniquet application are not feasible for all types of injuries, multiple topical hemostatic agents have been developed. These hemostatic dressings can be classified by their mechanism of action: factor concentrators, mucoadhesives, and procoagulant supplementers.47 The FDA has approved a number of these agents.47 These agents are used widely, but have not been validated or well-studied.
The benefit of these products is that they typically are compact and lightweight, making them easy to store and transport for both in-hospital and out-of-hospital use. Most of these agents have similar efficacies; however, they may be caustic to surrounding tissue. Combat Gauze, Celox Guaze, and Chito Gauze are the only ones deemed safe and efficacious and, thus, considered first-line recommended hemostatic agents by the Committee on Tactical Combat and Casualty Care (CoTCCC).48,49,50 These agents should be packed into the wound, and the provider should apply three to five minutes minimum of direct pressure, followed by a pressure dressing.
In 2016, the CoTCCC also proposed the addition of XStat to its hemorrhage control guidelines. XStat is a radiopaque hemostatic sponge system that can be deployed into deep tracks of wounds. Currently it cannot be used on the neck or torso. It is in early phases of use and is fairly costly, but it could reduce the need for extremity tourniquets.51,52
Unstable Pelvic Injuries: Binders
Pelvic fractures can be life-threatening, with mortality reported as ranging between 7.6% and 50% for open pelvic fractures.53 Pelvic injuries can be overlooked easily in normotensive trauma patients with distracting injuries. A retrospective cohort study that examined blunt trauma between August and December 2011 found a high proportion of missed fractures (31%).53
If pelvic instability exists or if the patient is persistently hypotensive without a clear source, pelvic binding with a pelvic circumferential compression device (PCCD) should be considered. Pelvic circumferential compression can provide tamponade by “closing” the pelvis and reducing the potential volume of the pelvis from nearly 6 L of potential space to 1-2 L.54 (See Figures 1 and 2 for X-rays of pelvis before and after placement of a pelvic binder.) When an unstable pelvic fracture is suspected or diagnosed, ATLS guidelines advise the use of a PCCD as a temporizing measure until definitive fixation can be achieved.8 Several commercial products exist for these purposes, or a simple bedsheet can be used.
Figure 1. Patient With an APC2 Fracture
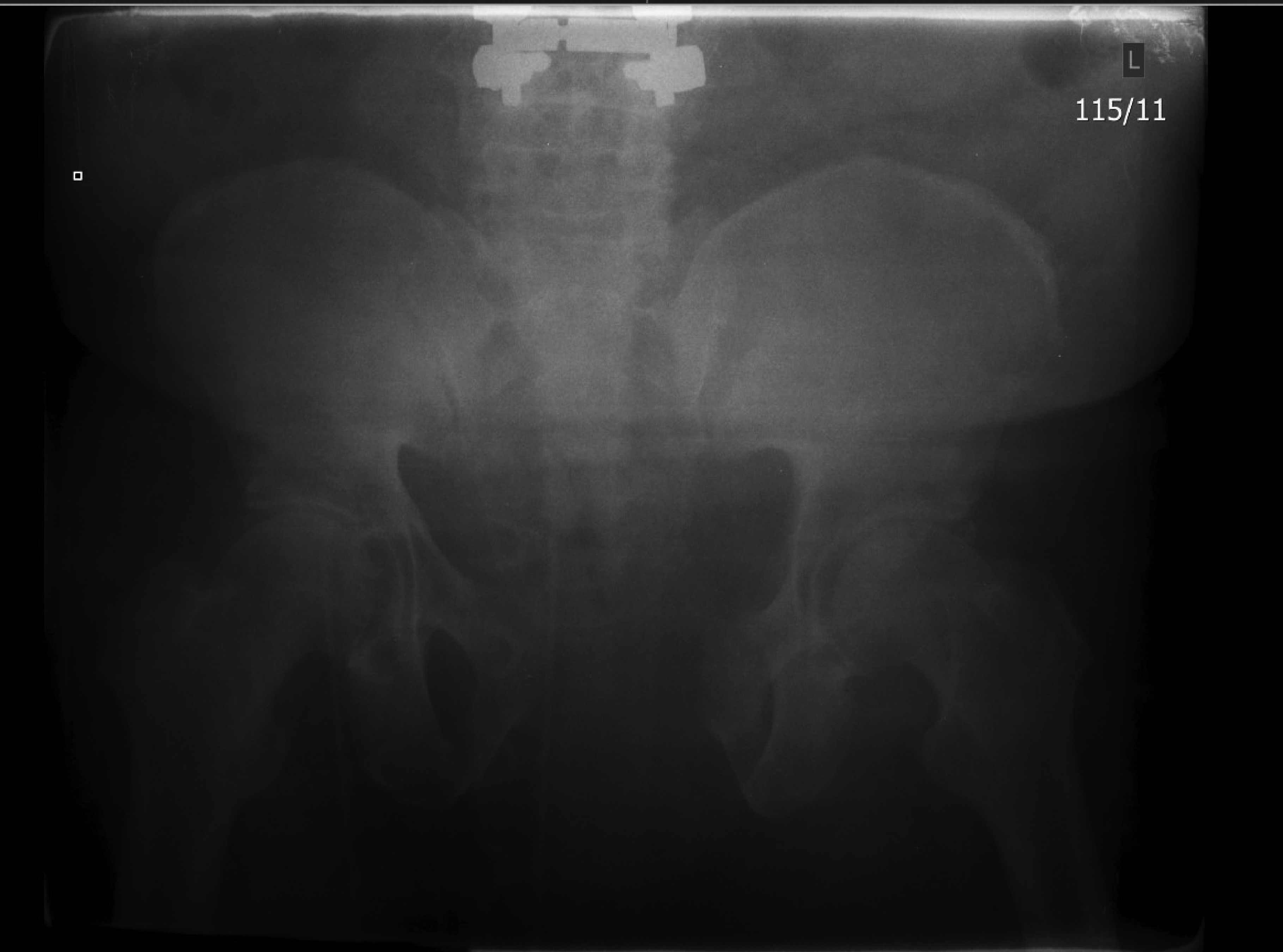
There is pubic diastasis of more than 2.5 cm and there is anterior widening of the SI joint.
Source: Uzcategui M, Menaker J. Blunt pelvic trauma. Trauma Reports 2014;15:6.
Figure 2. Patient With APC2 Fracture After Placement of a Pelvic Binder
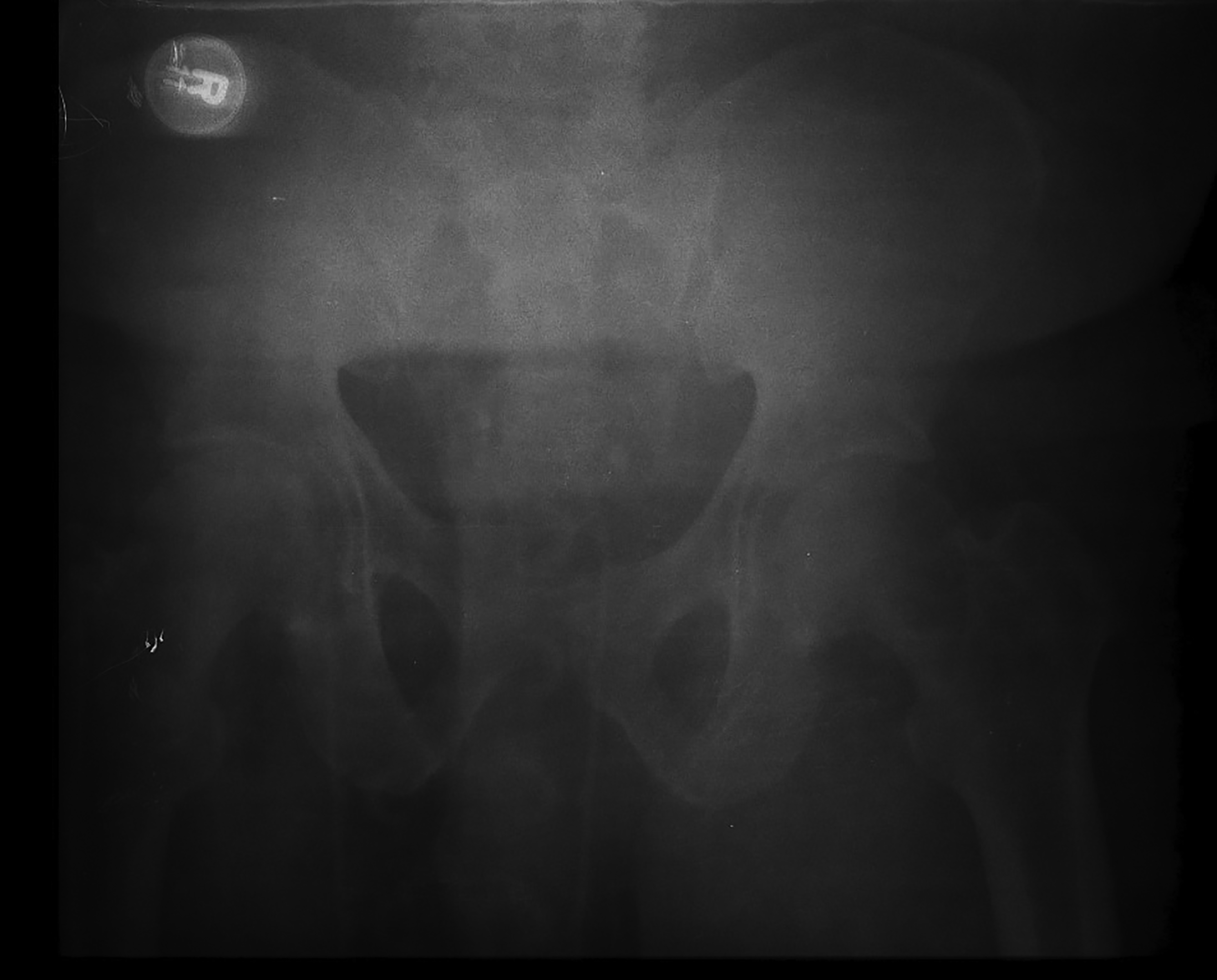
Placement of the pelvic binder with approximation and stabilization of the anterior and posterior elements, resulting in decreased pelvic volume.
Source: Uzcategui M, Menaker J. Blunt pelvic trauma. Trauma Reports 2014;15:6.
Many studies indicate that when placed correctly, these various modalities have similar efficacies.55-59 Appropriate placement is crucial to efficacy. The PCCD should be placed at the level of the greater trochanter, not the abdomen, to optimize compression. In addition, efforts should be made to minimize friction and pressure points on the skin, as this can lead to skin breakdown and ulcerations. Binders should be used only as temporizing measures until definitive hemostasis can be achieved, as prolonged use can cause skin breakdown and nerve damage.60
For pelvic injuries that continue to be hemodynamically unstable despite stabilization and closure, further invasive intervention should be implemented, such as external fixation, open fixation, or embolization. (See Figure 3.)
Figure 3. Internal Fixation of Pelvic Fracture
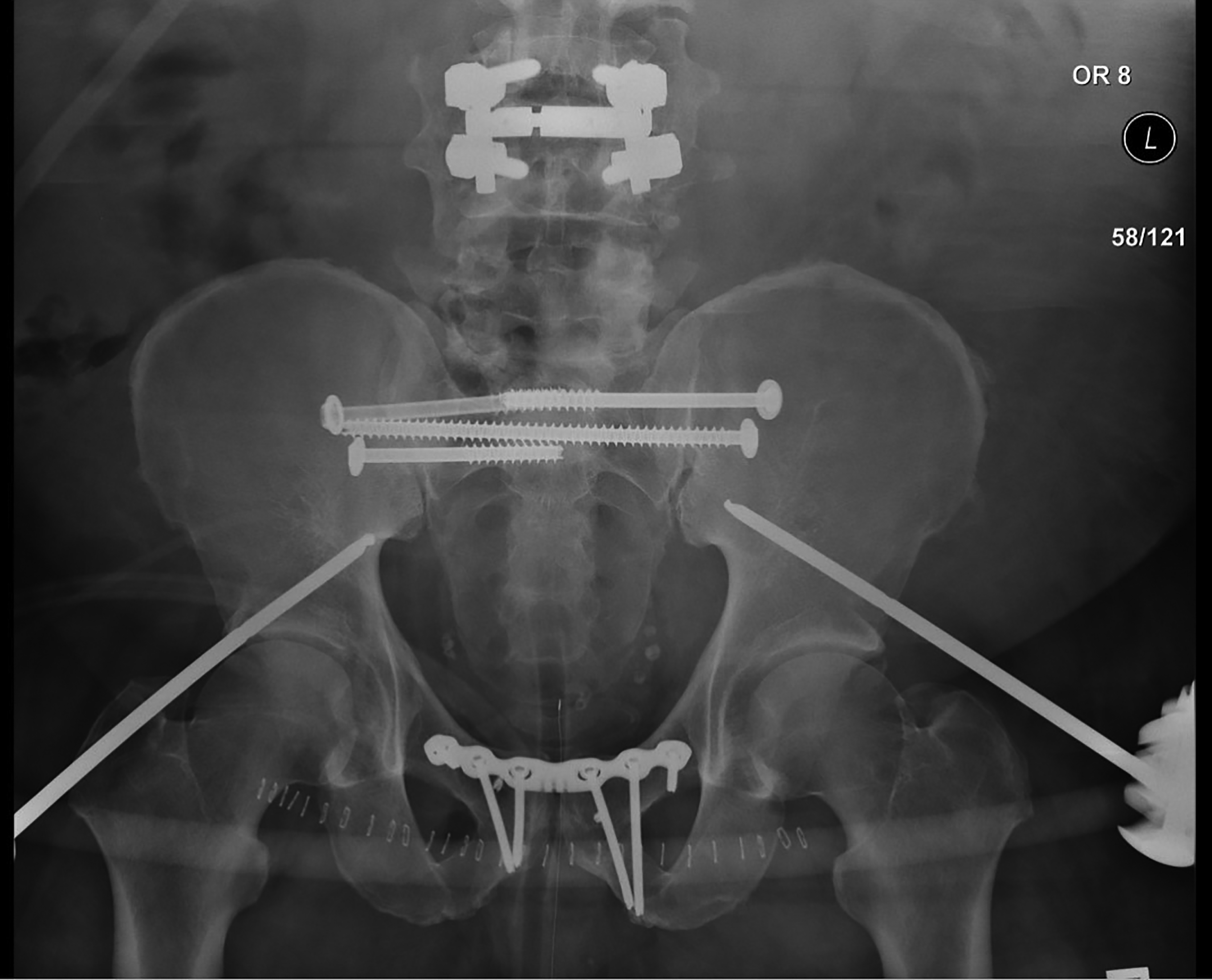
Patient originally stabilized with external fixation was bridged to definitive stabilization of the anterior and posterior elements with internal fixation.
Source: Uzcategui M, Menaker J. Blunt pelvic trauma. Trauma Reports 2014;15:7.
Definitive Hemostasis
To optimize care, patients should be transported to a hospital with trauma capabilities as soon as possible. The authors of a systematic review in 2006 showed a 15% reduction in mortality for patients treated in a trauma system, with a reported 50% decrease in preventable deaths.61 In addition to the aforementioned methods of hemorrhage control, which also can be applied in-hospital, once in the hospital, efforts should be made to expedite definitive surgical or angiographic management of bleeding.
In a study by Clarke et al, in hypotensive patients with isolated intra-abdominal injuries, the degree of hypotension and duration in the emergency department increased mortality by 1% for every three minutes in the emergency department. Meizoso et al studied hypotensive gunshot wound patients, and found that delays greater than 10 minutes increased the likelihood of death by threefold, highlighting the crucial push to the operating room.62 To improve morbidity and mortality in the trauma patient, definitive surgery or angiography should not be delayed.
Non-compressible Truncal Hemorrhage
Patients with non-compressible truncal hemorrhage (NCTH) have high mortality rates, from 18% to 50%, and are known to be at risk of imminent decompensation.63 These patients have limited bedside options for stabilization. One option is aortic occlusion (AO), which remains controversial for traumatic shock. The goal of AO is to preserve cerebral perfusion and coronary filling in life-threatening hypotension and hypovolemia due to hemorrhage in the chest.64 AO can be obtained in the more traditional method via a resuscitative thoracotomy or endovascularly. Resuscitative thoracotomy is achieved with a large open incision with aortic cross clamping. This technique typically is reserved for patients who have lost vital signs. This dramatic procedure has a poor survival rate of 0% to 15%.63
Comparing open approach, endovascular approach, and resuscitative thoracotomy, Chang et al conducted a multicenter, retrospective study of patients with NCTH. A total of 543 patients with NCTH were treated with open management (n = 309, 31%), endovascular management (n = 166, 31%), and resuscitative thoracotomy (n = 68, 12%). Mortality was 20%, 15%, and 79%, respectively.65 Endovascular management is recommended to be reserved for more hemodynamically stable patients, whereas open hemorrhage control is recommended for the unstable patient.
Resuscitative endovascular balloon occlusion of the aorta (REBOA) offers a less invasive alternative to open aortic occlusion.64,66,67,68 A retrospective case series by Moore et al between October 2011 and September 2015 examined 31 patients who underwent REBOA. A return to spontaneous circulation was observed in 60% of patients who had arrested prior to insertion (n = 10). After REBOA placement and balloon inflation, a significant increase in systolic blood pressure was observed in all patients with vital signs.63 The survival rate in this study was 32%.
Currently, the American Association for the Surgery of Trauma (AAST) Aortic Occlusion for Resuscitation in Trauma and Acute Care Surgery (AORTA) registry is prospectively observing patients with AO in traumatic shock at eight Level I Trauma Centers. In one study by Dubose et al, data comparing REBOA and open AO were collected between November 2013 and February 2015. Overall, hemodynamic improvement was observed after AO in 62.3%. Overall survival was 21%, with no significant difference between REBOA and open AO. Further evidence is needed to understand the true potential of these two methods.64
As with open AO, REBOA is not without risk. Rare REBOA complications have been reported, including pseudoaneurysm, embolism, and limb ischemia.64 A recent Japanese case series found three cases of vascular injury or ischemia in the placement of 24 REBOA for blunt trauma.63
Adjunctive Agents
Tranexamic Acid (TXA). The Clinical Randomization of Antifibrinolytic Therapy in Significant Haemorrhage (CRASH-2) trial was a randomized, controlled trial that examined the administration of TXA in 20,211 adult trauma patients.69,70 TXA is a synthetic lysine analogue and competitive inhibitor of plasminogen. At four weeks after injury, all-cause mortality was reduced by 1.5%. When TXA was administered between one and three hours of injury, mortality was reduced by 1.3%, whereas TXA administered after three hours increased risk of death by 1.3%.69,70 The EAST guidelines for DCR conditionally recommend early TXA for the severely hemorrhaging trauma patient.6
Conclusion
Early identification of massive hemorrhage is essential. Simple yet lifesaving interventions, such as good vascular access, tourniquet placement, and pelvic binding should be used. Imaging and laboratory studies are of little use in the initial evaluation and management of the trauma patient in hemorrhagic shock.
Hemorrhage is a significant cause of death in trauma patients. Many advances have changed the way trauma patients are managed, especially those in hemorrhagic shock. We have come full circle and returned to a more damage control-based system. While much of the research on newer techniques, such as MTP, TXA, REBOA, ROTEM, is still in its early phases, several of these methods show promise in improving outcomes for trauma patients.
Case Conclusions
The 70-year-old male presents after a motor vehicle collision with HR 103, BP 80/p, evident bruising to flank, and unstable pelvis.
What injuries should be considered? What is the best way to stabilize this patient?
His airway, breathing, and pulses in all four extremities are intact. The vital signs, tachycardia, and hypotension suggest significant hemorrhage, even though his heart rate is not extremely high. The unstable pelvis suggests a pelvic fracture. A pelvic binder should be placed to tamponade the pelvis and reduce the potential 6 L of blood loss down to 1-2 L. The flank bruising suggests concern for a retroperitoneal bleed. If there is blood available immediately, this patient would benefit from trauma blood. If there is no blood, minimal crystalloid intravenous fluid should be given with the goal of systolic blood pressure of 80-90s.
In the elderly patient population, it important to ask about past medical problems, specifically if they are taking blood thinners. Reversal agents can be given to facilitate hemostasis.
Once in the hospital, there can be further evaluation of hemorrhage. In this case, the patient is fluid responsive. The FAST exam reveals no free fluid in the abdomen. A CT scan shows continued extravasation into the pelvis and retroperitoneal hemorrhage. Angiography is performed to stop the bleeding.
The second patient from the same accident is a 20-year-old female with a crush injury, with HR 130, BP 70/p, and a partial right upper extremity amputation.
What injuries should be considered? What is the best way to stabilize this patient?
Her airway and breathing were intact at the scene. Her vital signs indicate significant hemorrhage. A tourniquet was placed proximal to the right upper extremity amputation site, but bleeding continued. Another tourniquet was placed with successful hemostasis. The time the tourniquet was placed was documented. This may be the source of her significant blood loss, but the mechanism and crush injury are concerning for internal hemorrhage. Beyond the tourniquet and initial fluid or blood resuscitation, her transfer to a trauma center should be expedited.
Her vital signs remain unstable on arrival. Despite initial blood and fluid, she remains hypotensive. MTP is activated, with blood products being given in a 1:1:1 ratio of packed red blood cells to fresh frozen plasma to platelets through two large-bore IVs. The crush injury with bruising to her chest and abdomen is concerning for intraperitoneal hemorrhage. FAST reveals significant intraabdominal fluid in the right upper quadrant and left upper quadrant. With her persistent tachycardia and hypotension, she is taken emergently to the operating room for exploratory laparotomy for definitive management. A partial hepatectomy and splenectomy are performed, which stops the bleeding.
REFERENCES
- GBD 2016 Causes of Death Collaborators. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017;390:1151-1210.
- Centers for Disease Control and Prevention. 10 Leading Causes of Death by Age Group, United States – 2015.
- Geeraedts LM Jr, Kaasjager HA, van Vugt AB, Frölke JP. Exsanguination in trauma: A review of diagnostics and treatment options. Injury 2009;40:11-20.
- Paull B. Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs a 1:1:2 ratio and mortality in patients with severe trauma: The PROPPR randomized clinical trial. J Emerg Med 2015;49:122.
- Chang R, Cardenas JC, Wade CE, Holcomb JB. Advances in the understanding of trauma-induced coagulopathy. Blood 2016;128:1043-1049.
- Cannon JW, Khan MA, Raja AS, et al. Damage control resuscitation in patients with severe traumatic hemorrhage. J Trauma Acute Care Surg 2017;82:605-617.
- Liu CC, Wang CY, Shih HC, et al. Prognostic factors for mortality following falls from height. Injury 2009;40:595-597.
- American College of Surgeons Committee on Trauma. Advanced Trauma Life Support (ATLS®) Student Manual, 9th ed. Chicago, IL: American College of Surgeons; 2012.
- Calland JF, Ingraham AM, Martin N, et al. Evaluation and management of geriatric trauma. J Trauma Acute Care Surg 2012;73(5 Suppl 4):S345-S350.
- Cohen MJ, Kutcher M, Redick B, et al. Clinical and mechanistic drivers of acute traumatic coagulopathy. J Trauma Acute Care Surg 2013;75(1 Suppl 1):S40-S47.
- Lawton LD, Roncal S, Leonard E, et al. The utility of Advanced Trauma Life Support (ATLS) clinical shock grading in assessment of trauma. Emerg Med J 2014;31:384-389.
- Mutschler M, Nienaber U, Brockamp T, et al. A critical reappraisal of the ATLS classification of hypovolaemic shock: Does it really reflect clinical reality? Resuscitation 2013;84:309-313.
- Guly HR, Bouamra O, Spiers M, et al. Vital signs and estimated blood loss in patients with major trauma: Testing the validity of the ATLS classification of hypovolaemic shock. Resuscitation 2011;82:556-559.
- Stone ME Jr, Kalata S, Liveris A, et al. End-tidal CO2 on admission is associated with hemorrhagic shock and predicts the need for massive transfusion as defined by the critical administration threshold: A pilot study. Injury 2017;48:51-57.
- Lamm RL, Coopersmith CM. Chapter 10. Hypovolemic and hemorrhagic shock. In: Roberts PR, Todd SR, eds. Comprehensive Critical Care: Adult. Society of Critical Care Medicine; 2012:183-198.
- Thorson CM, Van Haren RM Van, Ryan ML, et al. Admission hematocrit and transfusion requirements after trauma. J Am Coll Surg 2013;216:65-73.
- Zehtabchi S, Sinert R, Goldman M. Diagnostic performance of serial haematocrit measurements in identifying major injury in adult trauma patients. Injury 2006;37:46-52.
- Bruns B, Lindsey M, Rowe K. Hemoglobin drops within minutes of injuries and predicts need for an intervention to stop hemorrhage. J Trauma 2007;63:312-315.
- Davis JE, Mackersie RV, Holbrook TL, Hoyt DB. Base deficit as an indicator of significant abdominal injury. Ann Emerg Med 1991;20:842-844.
- Rutherford EJ, Morris JA Jr, Reed GW, Hall KS. Base deficit stratifies mortality and determines therapy. J Trauma 1992;33:417-423.
- Dezman ZD, Comer AC, Smith GS, et al. Failure to clear elevated lactate predicts 24-hour mortality in trauma patients. J Trauma Acute Care Surg 2015;79:580-585.
- Davenport R, Manson J, De’Ath H, et al. Functional definition and characterisation of acute traumatic coagulopathy. Crit Care Med 2011;39:2652-2658.
- Prat NJ, Meyer AD, Ingalls NK, et al. Rotational thromboelastometry significantly optimizes transfusion practices for damage control resuscitation in combat casualties. J Trauma Acute Care Surg 2017;83:373-380.
- Baksaas-Aasen K, Gall L, Eaglestone S, et al. iTACTIC - implementing Treatment Algorithms for the Correction of Trauma-Induced Coagulopathy: Study protocol for a multicentre, randomised controlled trial. Trials 2017;18:486.
- Laursen TH1, Meyer MAS, Meyer ASP. Thrombelastography early amplitudes in bleeding and coagulopathic trauma patients: Results from a multicenter study. J Trauma Acute Care Surg 2018;84:334-341.
- Rossaint R, Bouillon B, Cerny V, et al. The European guideline on management of major bleeding and coagulopathy following trauma: Fourth edition. Crit Care 2016;20:100.
- Holcomb JB. Major scientific lessons learned in the trauma field over the last two decades. PLoS Med 2017;14:4-7.
- Kauvar DS, Lefering R, Wade CE. Impact of hemorrhage on trauma outcome: An overview of epidemiology, clinical presentations, and therapeutic considerations. J Trauma 2006;60(6 Suppl):S3-S11.
- Malone DL, Dunne J, Tracy JK, et al. Blood transfusion, independent of shock severity, is associated with worse outcome in trauma. J Trauma 2003;54:898-907.
- Sharif MA, Wyatt MG. Vascular trauma. Surgery (Oxford International Edition) 2012;30:399-404.
- Park MJ, Gans I, Lin I, et al. Timing of forearm arterial repair in the well-perfused limb. Orthopedics 2014;37:e582-e586.
- Dorlac WC, DeBakey ME, Holcomb JB, et al. Mortality from isolated civilian penetrating extremity injury. J Trauma 2005;59:217-222.
- Kragh JF Jr, Walters TJ, Baer DG, et al. Survival with emergency tourniquet use to stop bleeding in major limb trauma. Ann Surg 2009;249:1-7.
- Schroll R, Smith A, McSwain NE Jr, et al. A multi-institutional analysis of prehospital tourniquet use. J Trauma Acute Care Surg 2015;79:10-14.
- Inaba K, Siboni S, Resnick S, et al. Tourniquet use for civilian extremity trauma. J Trauma Acute Care Surg 2015;79:232-237.
- Bulger EM, Snyder D, Schoelles K, et al. An evidence-based prehospital guideline for external hemorrhage control: American College of Surgeons Committee on Trauma. Prehosp Emerg Care 2014;18:163-173.
- American College of Emergency Physicians. Out-of-Hospital Severe Hemorrhage Control. Policy statement. Ann Emerg Med 2015;66:693.
- Gibson R, Housler GJ, Rush SC, et al. Preliminary comparison of new and established tactical tourniquets in a manikin hemorrhage model. J Spec Oper Med 2016;16:29-35.
- Gibson R, Aden JK 3rd, Dubick MA, Kragh JF Jr. Preliminary comparison of pneumatic models of tourniquet for prehospital control of limb bleeding in a manikin model. J Spec Oper Med 2016;16:21-27.
- Bequette BW, Kragh JF Jr, Aden JK Rd, Dubick MA. Belts evaluated as limb tourniquets: BELT study comparing trouser supporters used as medical devices in a manikin model of wound bleeding. Wilderness Environ Med 2017;28:84-93.
- Beekley AC, Sebesta JA, Blackbourne LH, et al. Prehospital tourniquet use in Operation Iraqi Freedom: Effect on hemorrhage control and outcomes. J Trauma 2008;64(2 Suppl):S28-S37.
- Dayan L, Zinmann C, Stahl S, Norman D. Complications associated with prolonged tourniquet application on the battlefield. Mil Med 2008;173:63-66.
- Kotwal RS, Butler FK Jr. Junctional hemorrhage control for tactical combat casualty care. Wilderness Environ Med 2017;28:S33-S38.
- Meusnier JG, Dewar C, Mavrovi E, et al. Evaluation of two junctional tourniquets used on the battlefield. J Spec Oper Med 2016;16:41-46.
- Rall JM, Ross JD, Clemens MS, et al. Hemodynamic effects of the Abdominal Aortic and Junctional Tourniquet in a hemorrhagic swine model. J Surg Res 2017;212:159-166.
- van Oostendorp SE, Tan EC, Geeraedts LM Jr. Prehospital control of life-threatening truncal and junctional haemorrhage is the ultimate challenge in optimizing trauma care; a review of treatment options and their applicability in the civilian trauma setting. Scand J Trauma Resusc Emerg Med 2016;24:110.
- Khoshmohabat H, Paydar S, Kazemi HM, Dalfardi B. Overview of agents used for emergency hemostasis. Trauma Mon 2016;21:e26023.
- Bennett BL, Littlejohn LF, Kheirabadi BS, et al. Management of external hemorrhage in tactical combat casualty care: Chitosan-based hemostatic gauze dressings—TCCC Guidelines-Change 13-05. J Spec Oper Med 2014;14:40-57.
- Butler FK, Blackbourne LH, Gross K. The Combat Medic Aid Bag: 2025. CoTCCC Top 10 Recommended Battlefield Trauma Care Research, Development, and Evaluation Priorities for 2015. J Spec Oper Med 2015;15:7-19.
- Holcomb JB, Butler FK, Rhee P. Hemorrhage control devices: Tourniquets and hemostatic dressings. J Spec Oper Med 2015;15:153-156.
- Sims K, Montgomery HR, Dituro P, et al. Management of external hemorrhage in tactical combat casualty care: The adjunctive use of XStatTM compressed hemostatic sponges: TCCC Guidelines Change 15-03. J Spec Oper Med 2016;16:19-28.
- Kragh JF Jr, Aden JK, Steinbaugh J, et al. Gauze vs XSTAT in wound packing for hemorrhage control. Am J Emerg Med 2015;33:974-976.
- Yong E, Vasireddy A, Pavitt A, et al. Pre-hospital pelvic girdle injury: Improving diagnostic accuracy in a physician-led trauma service. Injury 2016;47:383-388.
- Ghaemmaghami V, Sperry J, Gunst M, et al. Effects of early use of external pelvic compression on transfusion requirements and mortality in pelvic fractures. Am J Surg 2007;194:720-723.
- Vaidya R, Roth M, Zarling B, et al. Application of circumferential compression device (binder) in pelvic injuries: Room for improvement. West J Emerg Med 2016;17:766-774.
- Chesser TJS, Cross AM, Ward AJ. The use of pelvic binders in the emergent management of potential pelvic trauma. Injury 2012;43:667-669.
- Bonner TJ, Eardley WG, Newell N, et al. Accurate placement of a pelvic binder improves reduction of unstable fractures of the pelvic ring. J Bone Joint Surg Br 2011;93:1524-1528.
- Mallinson T. Alternative improvised pelvic binder. African J Emerg Med 2013;3:195-196.
- Pizanis A, Pohlemann T, Burkhardt M, et al. Emergency stabilization of the pelvic ring: Clinical comparison between three different techniques. Injury 2013;44:1760-1764.
- Jain S, Bleibleh S, Marciniak J, Pace A. A national survey of United Kingdom trauma units on the use of pelvic binders. Int Orthop 2013;37:1335-1339.
- Celso B, Tepas J, Langland-Orban B, et al. A systematic review and meta-analysis comparing outcome of severely injured patients treated in trauma centers following the establishment of trauma systems. J Trauma 2006;60:371-378.
- Meizoso JP, Ray JJ, Karcutskie CA 4th, et al. Effect of time to operation on mortality for hypotensive patients with gunshot wounds to the torso: The golden 10 minutes. J Trauma Acute Care Surg 2016;81:685-691.
- Moore LJ, Martin CD, Harvin JA, et al. Resuscitative endovascular balloon occlusion of the aorta for control of noncompressible truncal hemorrhage in the abdomen and pelvis. Am J Surg 2016;212:1222-1230.
- Dubose JJ, Scalea TM, Brenner M, et al. The AAST prospective Aortic Occlusion for Resuscitation in Trauma and Acute Care Surgery (AORTA) registry: Data on contemporary utilization and outcomes of aortic occlusion and resuscitative balloon occlusion of the aorta (REBOA). J Trauma Acute Care Surg 2016;81:409-419.
- Chang R, Fox EE, Greene TJ, et al. Multicenter retrospective study of noncompressible torso hemorrhage: Anatomic locations of bleeding and comparison of endovascular versus open approach. J Trauma Acute Care Surg 2017;83:11-18.
- Napolitano LM. Resuscitative endovascular balloon occlusion of the aorta: Indications, outcomes, and training. Crit Care Clin 2017;33:55-70.
- Brenner M, Teeter W, Hoehn M, et al. Use of resuscitative endovascular balloon occlusion of the aorta for proximal aortic control in patients with severe hemorrhage and arrest. JAMA Surg 2017;21201:1-6.
- Aso S, Matsui H, Fushimi K, Yasunaga H. Resuscitative endovascular balloon occlusion of the aorta or resuscitative thoracotomy with aortic clamping for noncompressible torso hemorrhage: A retrospective nationwide study. J Trauma Acute Care Surg 2017;82:910-914.
- Roberts I, Shakur H, Coats T, et al. The CRASH-2 trial: A randomised controlled trial and economic evaluation of the effects of tranexamic acid on death, vascular occlusive events and transfusion requirement in bleeding trauma patients. Health Technol Assess 2013;17:1-80.
- CRASH-2 collaborators; Roberts I, Shakur H, Afolabi A, et al. The importance of early treatment with tranexamic acid in bleeding trauma patients: An exploratory analysis of the CRASH-2 randomised controlled trial. Lancet 2011;377:1096-1101, 1101.e1-1101.e2.
Massive hemorrhage is a devastating scenario. Early identification is essential to allow for prompt, potentially lifesaving interventions with the goal of rapid control of exsanguination.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
