Medical Cannabis and Chronic Pain
April 1, 2018
Reprints
Adjunct Faculty, Bastyr University, Seattle; Owner, Naturopathic Doctor, Harbor Integrative Medicine, Bellingham, WA
Dr. Pantuso reports no financial relationships relevant to this field of study.
SUMMARY POINTS
- Cannabis plants contain two primary physiologically active compounds: tetrahydrocannabinol and cannabidiol.
- Between 45% and 80% of individuals who use medical cannabis do so for pain management.
- Insufficient evidence exists to make a good recommendation regarding the use of medical cannabis for the treatment of pain.
SYNOPSIS: The authors of this systematic review investigating the effects of medical cannabis (MC) on chronic pain in adults found that there is limited evidence to support the use of MC in treating neuropathic pain and that there is insufficient evidence that MC improves other types of chronic pain.
SOURCE: Nugent SM, Morasco BJ, O’Neil ME, et al. The effects of cannabis among adults with chronic pain and an overview of general harms: A systematic review. Ann Intern Med 2017;167:319-332.
Cannabis sativa and Cannabis indica increasingly are becoming available for both medical and recreational use in the United States, with chronic pain one of the most cited reasons for medical use.1 In 1999, California was the only state in which medical cannabis (MC) was legal, and by 2018, MC was legal in 29 states and recreational cannabis was legal in nine states (Alaska, California, Colorado, Oregon, Massachusetts, Maine, Nevada, Vermont, and Washington).2 Although several states have deregulated cannabis use, the federal government has not followed suit and cannabis remains a Schedule 1 (no known legal medicinal use) substance. Individuals suffering from chronic pain conditions increasingly present to their healthcare providers wanting to discuss using MC to treat their pain. A recent study investigating U.S. physician attitudes toward MC found that 89.5% of surveyed residents and fellows did not feel prepared to prescribe MC and only 35.3% reported readiness to answer questions regarding MC.3 Only 9% of U.S. medical schools reported clinically relevant MC content in their curriculum.3
To further elucidate whether there is evidence to recommend MC in the treatment of chronic pain and whether there are harms associated with its use, Nugent et al further examined the evidence in a systematic review. This study included English-language studies investigating the effect of plant-based cannabis or whole plant extracts on chronic pain in non-pregnant adults. The authors defined plant-based cannabis preparations to include any preparation of the cannabis plant or extracts to capture the variety of products available in U.S. dispensaries. (See Table 1.)
Table 1: Cannabis Types Studied and Available in U.S. Dispensaries |
|
Type of Medical Cannabis |
Number
|
|
Nabiximols |
12 |
|
Smoked THC |
5 |
|
THC/CBD capsules |
3 |
|
Vaporized THC |
3 |
|
Orally ingested, standardized THC product |
2 |
|
Sublingual spray |
1 |
|
Oral mucosal spray |
1 |
|
THC: tetrahydrocannabinol; CBD: cannabidiol |
|
The authors also included studies of nabiximols, which is a cannabis extract that is standardized to deliver 2.7 mg of tetrahydrocannabinol (THC) and 2.5 mg of cannabidiol (CBD) through an oral mucosal spray. The authors identified potential studies to be included in this systematic review through MEDLINE, Embase, PubMed, PsycINFO, Evidence-Based Medicine Reviews, and gray literature sources from database inception until February 2016. The authors then updated this search in March 2017 to identify any new randomized, controlled trials and other systematic reviews. The authors also identified ongoing studies through ClinicalTrials.gov, the International Clinical Trials Registry Platform, the National Institutes of Health RePORTER, and the Agency for Healthcare Research and Quality Grants On-Line Database.
A total of 13,674 studies were identified; 12,460 studies were excluded because they were not relevant to this review and another 1,139 studies did not meet inclusion criteria. (See Figure 1.) Of the 75 publications included in this systematic review, 32 studies were included in the chronic pain analysis and 43 studies were included in the harms analysis. Two investigators abstracted details of study design, setting, patient population, intervention, and follow-up, as well as important co-interactions, health outcomes, healthcare use, and harms. Each trial was assessed as having low, high, or unclear risk of bias (ROB) for the pain outcomes by two reviewers using an outcome tool developed by the Cochrane Collaboration. Disagreements were resolved by consensus. Statistical heterogeneity was assessed using the Cochran chi-square test. All analyses were performed using Stata/IC version 13.1. For the subgroup of neuropathic pain, a meta-analysis was performed and the profile-likelihood random-effects model was used to perform the risk ratios. A meta-analysis on the other subgroups could not be performed because of heterogeneity, variation in outcomes reported, and the small number of trials, so these were reported qualitatively. The authors discussed the evidence for these subgroups and classified the overall strength of evidence for each outcome as high, moderate, low, or insufficient based on the consistency, coherence, and applicability of the body of evidence as well as the internal validity of individual studies.
Figure 1: Data from Systematic Review of Cannabis |
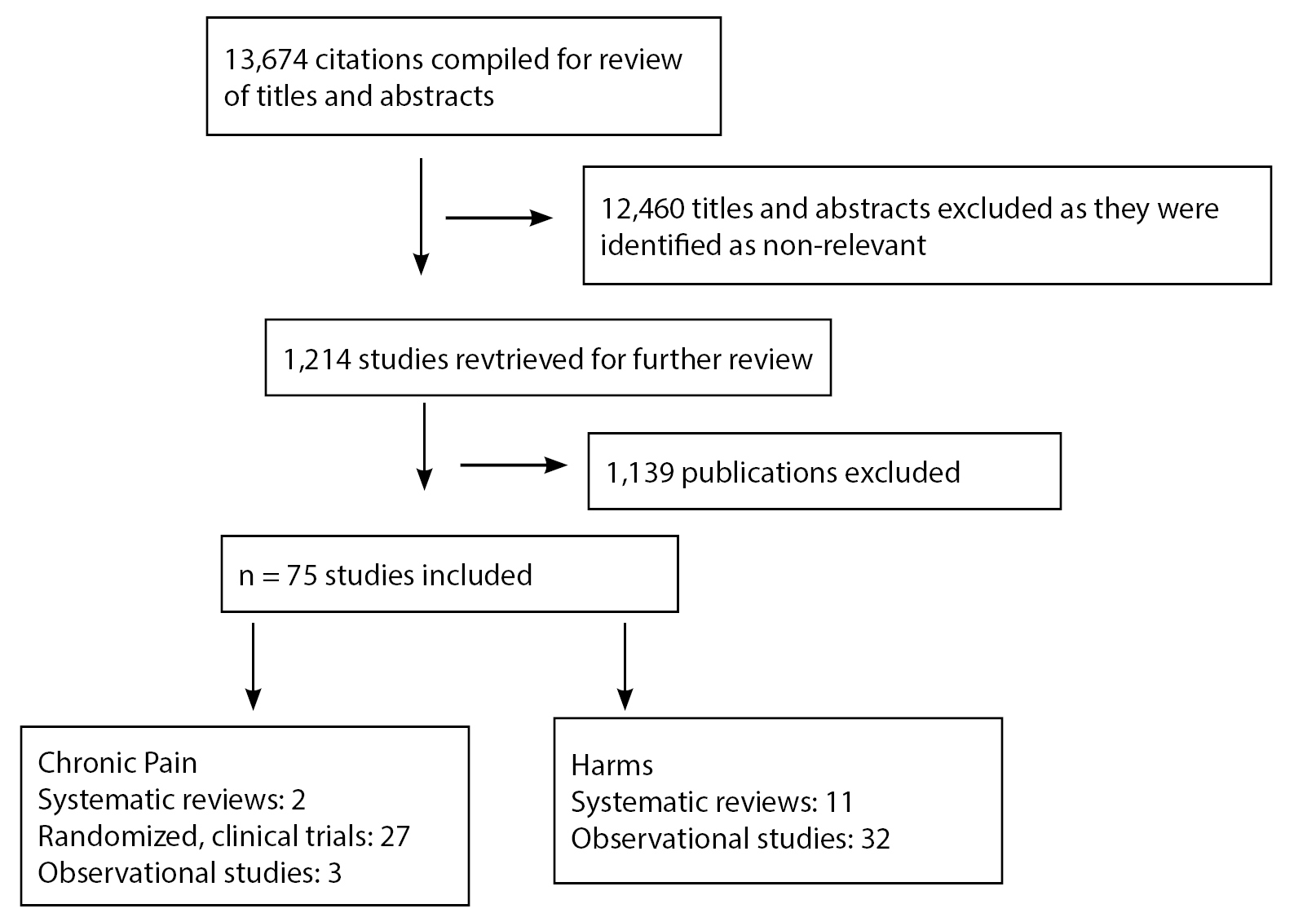 |
|
Adapted from: Nugent SM, Morasco BJ, O’Neil ME, et al. The effects of cannabis among adults with chronic pain and an overview of general harms: A systematic review. Ann Intern Med 2017;167:319-332. |
Neuropathic Pain
Thirteen studies investigated the effects of MC preparations on neuropathic pain. Eleven were considered to have low ROB, one had unclear ROB, and the other had high ROB. The authors found low strength of evidence that MC may alleviate neuropathic pain in some patients. In nine studies, patients using cannabis to treat neuropathic pain were more likely to report at least a 30% improvement in pain (risk ratio, 1.43; 95% confidence interval [CI], 1.16-1.88; I2 = 38.6; P = 0.111), although these results were not statistically significant.
Multiple Sclerosis
There were nine trials investigating the effects of cannabis on pain among patients with multiple sclerosis (MS). Three were considered to have low ROB, five had unclear ROB, and one had high ROB. The authors determined that they were unable to find sufficient evidence to characterize the effects of MC on pain in patients with MS. Among the three low ROB studies, one demonstrated no difference in outcome with cannabis, another found a small but clinically nonsignificant alleviation of pain at five weeks, and the authors of a trial with more participants found that more patients in the MC group reported decreased pain at 12 weeks compared to the control group (28.0% vs. 18.7%; P = 0.028).
Cancer Pain
Three trials (n = 547) investigated the effects of cannabis extracts on pain in cancer patients. The authors concluded that there was insufficient evidence because of the small number of studies and their methodological limitations.
Other or Mixed Pain Conditions
The authors of two trials and three cohort studies concluded that there was insufficient evidence because of methodologically flawed studies.
Harms of Cannabis Use
Adverse effects reported in two systematic reviews of cannabis use in chronic pain patients included dizziness and light-headedness, while other more serious side effects included suicide attempts, paranoia, and agitation. A prospective observational study did not find a difference between the cannabis group (12.5% ± 1.5% THC, 2.5 g/d) and the control group (adjusted incidence rate ratio for event, 1.08; 95% CI, 0.57-2.04).4
In the general population, two well-designed cohort studies indicated that low levels of cannabis smoking did not affect lung function adversely over about 20 years in young adults.5,6 In eight studies and one systematic review, researchers found an association between the development of psychotic symptoms and cannabis use. These psychotic symptoms were related to the strength of the THC content. The risk of developing psychosis with MC use is similar for the general population and those with a prior history of psychological conditions. The authors of a recent meta-analysis that included 21 multinational observational studies suggested that acute cannabis intoxication was associated with a moderate increase in motor vehicle collision risk (odds ratio [OR], 1.35; 95% CI, 1.15-1.61).7
Cannabinoid hyperemesis syndrome is a severe form of cyclic vomiting that has been associated with long-term cannabis use and acute cannabinoid toxicity. Aspergillosis and tuberculosis also have been associated with smoking cannabis. There is mixed evidence about whether cannabis has an effect on violent behavior. Cannabis use disorder, which is defined in the Diagnostic and Statistical Manual of Mental Disorders 5 as a problematic pattern of cannabis use, is associated with cannabis use (adjusted OR, 9.5; 95% CI, 6.4-14.1) in a prospective cohort study (n = 34,653).8 In a cross-sectional study of daily opioid therapy for chronic pain and cannabis use disorder, the prevalence was 2.4%, and 13.2% of patients reported using cannabis in the previous 30 days.9
COMMENTARY
An increasing number of patients are using MC to treat chronic pain. In states in which recreational cannabis is legal, patients do not require a recommendation from their healthcare provider. However, patients ask questions regarding the use of MC to treat their chronic pain, and because of insufficient evidence, healthcare providers often are unable to make evidence-based recommendations. Nugent et al sought to provide further clarification. Unfortunately, because of the quality of the original research studies, the authors only could perform a meta-analysis on the treatment of neuropathic pain with MC, which showed a reduction in pain and was not significant. Adverse side effects also were investigated, and cannabis use was significantly associated with cannabis use disorder. More research needs to be conducted to further understand the risk of adverse side effects from use MC.
This study was part of a larger investigation commissioned by the Veteran’s Health Administration. A protocol was posted to a public website prior to the study initiation. The strengths of this systematic review are the selection of studies to be included in this analysis. Although the authors’ review did not demonstrate a significant effect in using cannabis preparations for chronic pain, a report released by the National Academy of Sciences (NAS) differs in its level of significance.2 The NAS report concluded that cannabis use to treat chronic pain has conclusive or substantial evidence of effect, with plant-derived cannabinoids 40% more likely to reduce pain than the control agent (odds ratio, 1.41; 95% CI, 0.99-2.00); of note, this is marginally statistically significant. Both the NAS and Nugent et al reviews included essentially the same trials, but the NAS report found a more favorable effect of MC on chronic pain.2 It is unclear how to interpret these disparate results other than to say that even the experts disagree.
There are many limitations to this systematic review, including the lack of methodologically sound studies, lack of studies in general, heterogeneity, and increased levels of ROB. Another important variable that may have influenced the results greatly are the cannabis preparations that were used.10 Of the 27 RCTs included in the analysis of effects of cannabis for treating chronic pain, 12 used the cannabis extract nabiximols, five used smoked THC, three used THC/CBD capsules, three used vaporized THC, two used an orally ingested standardized THC product, one used a pump-action sublingual spray, and one used an oral mucosal spray with 2.7 mg of THC. There also was a wide range of study length, from one day to 15 weeks. The variation in cannabis preparations and method of administration may have a large effect on the results of the individual studies.10
Much of the research on the therapeutic effects of cannabis have used cannabis preparations that are pharmaceutical cannabinoids — dronabinol, nabilone, and nabiximols.1,2,10 This is a major barrier to extrapolating the conclusion of these studies to patients, as many patients are interested in using or currently using cannabis products obtained from dispensaries. (See Table 2.) For example, CBD oil is becoming increasingly popular, but it is difficult to know whether studies on pharmaceutical-grade products or whole cannabis leaf products equate with what is available as a dietary supplement.
Table 2: Cannabis Products Available at Dispensaries and Route of Administration |
||
Route of Administration |
Method |
Types of Cannabis Products |
|
Inhaled |
Smoking, vaporizing, vape pens |
Cannabis flower, oil, wax, resins, concentrates |
|
Oral |
Ingestion Sublingual and submucosal application |
Candy, chocolate, baked goods, flavored soda, encapsulated extracts, tinctures, infused food stuffs (honey, butter, oil, etc.) |
|
Transdermal |
Topical application on skin |
Oils, creams, ointments |
|
Other |
Suppositories |
Encapsulated oil |
Furthermore, because cannabis is a Schedule 1 substance, it is difficult to obtain to use in research studies and this is one barrier to cannabis research.1,2,3 Another important factor in cannabis research is that the DEA and FDA have only allowed researchers to use cannabis that was from the University of Mississippi; it was not until 2016 that researchers were allowed to begin to use other sources of cannabis. Although there is a lack of quality evidence to support MC use in chronic pain, one should not equate this lack of evidence to trial data suggesting no effects at this time.
Bottom Line
Currently, there is limited evidence to support the use of cannabis in chronic pain management. However, it is still in the early stages of research, and more methodologically sound trials need to be conducted before we can make good evidence-based recommendations and understand the risks and benefits of use.
Until such evidence arises, clinicians should understand that patients are counseled about which MC products to use by employees who work in the MC or recreational cannabis stores, and that these employees may or may not have received adequate training. Because of the extensive choices available, it is important that healthcare providers be involved in this decision-making process.10
Adverse events have been demonstrated in patients who consume higher amounts of THC, especially if they have never used or are infrequent users of cannabis. There is a wide range of MC products, and some contain large amounts of THC, which patients should be counseled against using. Patients should be counseled that when inhaling through smoking or vaporizing, the effects are noticed within five to 10 minutes, but when ingesting orally it may take up to one to three hours for the MC to take effect.
As with all treatment recommendations, patients should be counseled about their chronic pain treatment options, including discussion about strength of the evidence, risks, and benefits. An additional consideration of which patients may not be aware is that since many employers still require employees to undergo drug testing, if they test positive for MC their position could be terminated. Shared decision-making techniques should be used in the discussion of whether patients should consider incorporating MC into their treatment plan. If patients want to use MC, providers should counsel them to choose a product that is lower in THC and contains CBD in as close to a 1:1 ratio or a CBD predominant chemovarietal product.10 There is a great variation in individual sensitivities to the THC level in MC, and patients should be advised when ingesting edibles to start at a lower dose, such as 2.5 mg or less, and gradually increase the dosage over days to weeks to the lowest dose that produces a therapeutic effect.10 There is a large variation in the effects between different cannabis plants and preparations, which may be a result of both the types of CBD and the concentrations in the products.10 Informing patients of these differences is helpful so that they can make an informed decision about MC use.
REFERENCES
- Bridgeman MB, Abazia DT. Medicinal cannabis: History, pharmacology, and implications for the acute care setting. P T 2017;42:180-188.
- Abrams DI. The therapeutic effects of cannabis and cannabinoids: An update from the National Academies of Sciences, Engineering and Medicine report. Eur J Intern Med 2018;49:7-11.
- Evanoff AB, Quan T, Dufalt C, et al. Physicians-in-training are not prepared to prescribe medical marijuana. Drug Alcohol Depend 2017;180:151-155.
- Ware MA, Wang T, Shapiro S, et al. Cannabis for the management of pain: Assessment of safety study (COMPASS). J Pain 2015;16:1233-1242.
- Hancox RJ, Poulton, Ely M, et al. Effects of cannabis on lung function: A population-based cohort study. Eur Respir J 2010;35:42-47.
- Pletcher MJ, Vittinghoff E, Kalhan R, et al. Association between marijuana exposure and pulmonary function over 20 years. JAMA 2012;307:173-181.
- Rogeberg O, Elvik R. The effects of cannabis intoxication on motor vehicle collision revisited and revised. Addiction 2016;111:1348-1359.
- Blaco C, Hasin DS, Wall MM, et al. Cannabis use and risk of psychiatric disorders: Prospective evidence from a US national longitudinal study. JAMA Psychiatry 2016;73:388-395.
- Fleming MF, Balousek SL, Klessig CL, et al. Substance use disorders in a primary care sample receiving daily opioid therapy. J Pain 2007;8:573-582.
- MacCallum C, Russo EB. Practical considerations in medical cannabis administration and dosing. Eur J Intern Med 2018;49:12-19.
The authors of this systematic review investigating the effects of medical cannabis (MC) on chronic pain in adults found that there is limited evidence to support the use of MC in treating neuropathic pain and that there is insufficient evidence that MC improves other types of chronic pain.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
