Infectious Meningitis: A Focused Review
May 1, 2018
Reprints
AUTHOR
Trahern W. Jones, MD, Department of Pediatrics, University of Arizona, Tucson
PEER REVIEWER
Steven M. Winograd, MD, FACEP, Mt. Sinai Queens Hospital Center, Jamaica Queens, NY; Assistant Clinical Profesor of Emergency Medicine, Mt. Sinai Medical School, New York City; Assistant Clinical Professor of Emergency Medicine, NYiTCOM, Old Westbury, NY
Executive Summary
- Despite the general success of vaccinations, Haemophilus influenzae, Streptococcus pneumoniae, and Neisseria meningitidis remain the predominant causes of bacterial meningitis globally.
- Other important etiologies of meningitis include Group B streptococcus (GBS), Listeria monocytogenes, Enterobacteriaceae species (especially Escherichia coli in neonates, and non-typhoidal Salmonella species globally), Staphylococcus aureus, Streptococcus suis in Southeast Asia, and Mycobacterium tuberculosis.
- Low birth weight and very low birth weight infants are at much higher risk for meningitis, but additional maternal risk factors include intrapartum fever, prolonged rupture of membranes, and colonization status with GBS. Since the institution of universal screening and treatment of mothers with positive GBS, there has been a substantial decline in the incidence of early-onset GBS disease in the United States. Nevertheless, GBS remains the leading infectious cause of morbidity and mortality in U.S. neonates.
- More than 75% of all pediatric tuberculosis (TB) infections in the United States are thought to be related to a foreign contact by the child, family member, or other close contact. Other risk factors in pediatric tuberculous meningitis include concurrent thoracic TB infection, family member or close contact with TB infection, lower socioeconomic status, immigrants, refugees, international adoptees, and HIV-positive status or immunosuppression.
- Among all cases of fungal meningitis in pediatric and adult patients in the United States, Cryptococcus spp. prevails (70.1% of all cases), followed by Coccidioides spp. (16.4%), Candida spp. (7.6%), and Histoplasma capsulatum (6.0%).
- Neonates and younger infants are unable to engage the neuromuscular control and strength necessary to resist an examiner’s passive flexion of the neck despite the pain they experience, and, thus, “neck stiffness” is found less commonly in infants younger than 12 months. Instead, neonates and infants may demonstrate “paradoxical irritability” with meningitis, whereby the pain of inflamed meninges irritated by an examiner’s handling will cause the infant to cry more while being held vs. when lying still on a flat surface.
- Ultimately, the accuracy of any physical exam maneuver in distinguishing meningitis is unacceptably poor and, thus, lumbar puncture and cerebrospinal fluid analysis remain the gold standards for diagnosis.
- The Infectious Diseases Society of America has provided recommendations for computed tomography of the head before lumbar puncture. These include history of prior central nervous system disease, new-onset seizure, papilledema, immunocompromise, altered consciousness, or focal neurologic deficit.
- In infants younger than 1 month of age, Group B streptococcal infection or E. coli predominate. Adequate coverage will be obtained with ampicillin and cefotaxime, administered parenterally at meningitic doses. Ampicillin plus gentamicin is also an acceptable choice. In pediatric patients older than 1 month, vancomycin and a third-generation cephalosporin (usually ceftriaxone) should be administered parenterally at meningitic doses.
The effectiveness of the current vaccine schedule has led to a significant decline in the incidence of bacterial meningitis. Delays in recognition and antibiotic administration result in increased morbidity and mortality; therefore, clinicians must maintain a high degree of vigilance for the subtle findings of meningitis, particularly in infants. Current standards for selective imaging, diagnostic testing, and empiric antibiotics are discussed.
— Ann Dietrich, MD, FAAP, FACEP, Editor
Definition
The term meningitis specifically refers to an inflammatory process affecting the pia mater, arachnoid mater, and the cerebrospinal fluid (CSF) contained within the arachnoid space.1 Although this process is nonspecific and may be driven by a variety of disease processes, the primary concern for pediatric emergency medicine physicians is that of a life-threatening infectious process, for which a wide variety of agents may be responsible. (See Table 1.)
Table 1. Etiologic Agents Reported in Cases of Pediatric Meningitis and Meningoencephalitis, by Category |
|
Viral |
Bacterial |
|
Enteroviruses Parechovirus Adenoviruses Herpes simplex 1&2 Varicella zoster Mumps Measles Rubella Variola West Nile virus La Crosse virus Eastern equine virus Western equine virus Coronaviruses Rhinoviruses Parvovirus B19 Cytomegalovirus Epstein-Barr virus Human herpesvirus–6&7 HIV Human T-cell lymphotropic virus Influenza A and B Lymphocytic choriomeningitis Parainfluenza Rotavirus |
Streptococcus pneumoniae Haemophilus influenzae Neisseria meningitidis Group B Streptococcus Escherichia coli Listeria monocytogenes Staphylococcus aureus Coagulase-negative Staphylococcus spp. Salmonella spp. Bartonella spp. Brucella spp. Borrelia spp. Leptospira spp. Nocardia spp. Anaplasma phagocytophila Coxiella burnetii Ehrlichia chaffeensis Rickettsia spp. Mycoplasma spp. Chlamydia pneumoniae Chlamydia psittaci Ureaplasma urealyticum |
Fungal |
Mycobacterial |
|
Cryptococcus spp. Candida spp. Histoplasma capsulatum Coccidioides spp. Blastomyces dermatitidis |
Mycobacterium tuberculosis |
Protozoal/Helminthic |
Noninfectious |
|
Naegleria fowleri Acanthamoeba Balamuthia mandrillaris Angiostrongylus spp. Gnathostoma spp. Baylisascaris spp. Strongyloides stercoralis Trichinella spiralis Toxocara canis Paragonimus westermani Schistosoma spp. |
Kawasaki disease Behçet disease Systemic lupus erythematosus Sarcoidosis Tumors and neoplasms Leukemia Vaccine-related Medication-induced Intrathecal injections Foreign bodies Heavy metal poisoning |
Epidemiology by Category of Etiologic Agent
Viral Causes
A broad array of viral infections may lead to a clinical diagnosis of meningitis in children. (See Table 1.) Viral etiologies often are labeled as “aseptic meningitis,” although properly speaking, this term refers only to clinical diagnoses of meningitis for which an organism cannot be grown in any readily available laboratory media.2 Indeed, while this term is inclusive of viral etiologies, it also covers autoimmune causes, idiopathic or unspecified cases, as well as bacterial agents that cannot be cultured on traditional media. Thus, for the purposes of this review, we will use the term “viral meningitis” only for clarity.
The epidemiology of viral meningitis has changed substantially in the last several decades. In part, this is due to modern immunization practices, such as the introduction of vaccination against mumps, which likely was the most common cause of pediatric viral meningitis prior to 1990.3 Other previously common causes, including poliovirus, varicella, and measles, are now relatively rare. In their absence, non-poliovirus enteroviruses (consisting of coxsackieviruses and echoviruses, among others) have become the predominant cause of viral meningitis.4
Estimates for annual U.S. emergency department visits for adult and pediatric viral meningitis range as high as 60,000 per year,5 while other estimates for hospitalizations due to viral meningitis have been placed at 36,000 per year.6 Such viral and “unspecified” cases constitute the majority of all meningitis diagnoses in the United States.5,6
Among pediatric cases for which a viral agent is identified, non-polio enteroviruses constitute 47% to 52% of cases.3,7 Seasonal peaks generally occur in summer and fall months in the Northern hemisphere, following the typical seasonal variation of enterovirus infections.8,9 Other agents that may be responsible for viral meningitis and viral meningoencephalitis, such as arthropod-borne viruses like West Nile virus, La Crosse virus, St. Louis encephalitis virus, and Eastern and Western equine encephalitis viruses, also seem to demonstrate seasonal variation, likewise favoring illness in the summer and fall months when mosquito activity tends to be highest.2,10
Bacterial Causes
Bacterial causes of meningitis are truly the province of the pediatric emergency physician, in that prompt identification and treatment may be lifesaving; therefore, an understanding of the prevalence and presentation of such agents is essential. By some estimates, between 4,100 and 5,000 cases of bacterial meningitis occur in the United States annually, with upwards of 500 deaths per year.5,11
As with viral etiologies, modern immunization practices have altered the epidemiological landscape of bacterial meningitis drastically. The widespread introduction of vaccines against Haemophilus influenzae type b and the invasive serovars of Streptococcus pneumoniae has decreased the incidence of each as causative agents of bacterial meningitis since the late 1980s,12-15 and, moreover, has decreased the burden of bacterial meningitis in North American and European countries substantially.11,16 However, despite the general success of these vaccinations, H. influenzae, S. pneumoniae, and Neisseria meningitidis remain as the predominant causes of bacterial meningitis globally.17 Other important agents include Group B streptococcus (GBS), Listeria monocytogenes, Enterobacteriaceae species (especially Escherichia coli in neonates, and non-typhoidal Salmonella species globally), Staphylococcus aureus, Streptococcus suis in Southeast Asia, and Mycobacterium tuberculosis.17,18
The epidemiology of various bacterial organisms in pediatric meningitis cases varies by age and organism-specific risk factors. For example, bacterial meningitis in the neonatal period usually is due to GBS, E. coli, or less commonly to L. monocytogenes.1 These organisms constitute the majority of cases because of acquisition during labor and delivery from infected or colonized mothers, and also because of a greater predisposition to meningeal invasion.19 Meningitis occurs more frequently in infants 1 month of age or younger than in any other age group. Low birth weight and very low birth weight infants are at much higher risk, but additional maternal risk factors include intrapartum fever, prolonged rupture of membranes, and colonization status with GBS.19-21 Since the institution of universal screening and treatment of mothers with positive GBS, there has been a substantial decline in the incidence of early-onset GBS disease in the United States. Nevertheless, GBS remains the leading infectious cause of morbidity and mortality in U.S. neonates.20,21 Practitioners must be mindful that screening and intrapartum antibiotics do not eliminate the possibility of late-onset GBS meningitis, and many cases remain of late-onset disease in mothers who tested negative during pregnancy, potentially because of interval colonization after screening but before labor.21,22
Causative agents of bacterial meningitis differ among older infants, children, and adolescents. In the United States, S. pneumoniae is the leading cause of pediatric bacterial meningitis, although N. meningitidis predominates among children and adolescents 11-17 years of age.1,11 H. influenzae also is represented among younger age groups, with many cases due to non-typeable or non-type b serovars. While pneumococcal disease has been noted generally to occur sporadically, meningococcal disease may occur in outbreaks. Household contacts of patients with N. meningitidis disease may have a 1,000-fold greater risk of disease.1
Children at higher risk of meningitis include those with anatomic or immunologic abnormalities that predispose to invasion of the subarachnoid space or decreased ability to eliminate pathogenic organisms. (See Table 2.) Splenectomy, congenital asplenia, polysplenia, and functional asplenia long have been known to predispose patients to overwhelming infection, particularly meningitis with encapsulated organisms like S. pneumoniae, N. meningitidis, and H. influenzae.23 Practitioners should maintain a high index of suspicion for bacterial meningitis among children with any immunocompromising condition or iatrogenic immunosuppression who may be at risk for unusual pathogens or more severe manifestations of disease.24,25 Anatomic abnormalities (CSF fistula), surgical procedures (cochlear implant, intraventricular devices, etc.), or traumatic injuries (basilar skull fracture) also should alert the pediatric provider to a child at increased risk of subarachnoid space invasion, not only due to classic organisms like those listed above, but also S. aureus, coagulase-negative staphylococci, or gram-negative infections.24-31
Table 2. Predisposing Conditions to Pediatric Meningitis 1,27-31,51-53 |
|
Immunodeficiency, Immunosuppression, and Hematologic |
Disruption of Normal Anatomy |
|
Functional and anatomic asplenia Neutropenia Hematologic malignancy Chemotherapy recipients Hematopoietic transplant recipients Solid organ transplant recipients HIV/AIDS Congenital immunodeficiencies (e.g., severe Sickle cell anemia |
Cerebrospinal fluid fistula Mastoiditis Otogenic intracranial abscess Orbital cellulitis and abscess Dental abscess |
Neurosurgical/Device Related |
Trauma |
|
Ventriculoperitoneal shunt External ventricular drain Ommaya reservoir Cochlear implants Indwelling intraventricular catheters Lumbar puncture |
Basilar skull fracture Open skull fracture |
|
SOURCE: Author created. |
|
Mycobacterial Causes
Mycobacterial causes of meningitis predominantly are represented by M. tuberculosis. The risk of the disease is significant globally; the incidence of M. tuberculosis worldwide is estimated at 140 cases per 100,000 persons.32 Untreated active pulmonary infections may lead to extrathoracic spread, including tuberculous meningitis. Progression to tuberculous meningitis is estimated to occur in up to 30% of all untreated, active tuberculosis (TB) infections in children younger than 2 years of age.33,34 More than 75% of all pediatric TB infections in the United States are thought to be related to a foreign contact by the child, family member, or other close contact.35 Other risk factors in pediatric tuberculous meningitis include concurrent thoracic TB infection, family member or close contact with TB infection, lower socioeconomic status, immigrants, refugees, international adoptees, and HIV-positive status or immunosuppression.35-38 The Bacille Calmette et Guérin (BCG) vaccine has been used in various countries outside the United States since the 1920s, with significant successes in preventing pediatric disseminated forms of TB, including pediatric tuberculous meningitis, although it appears much less successful at preventing latent TB infection or later pulmonary infection.39
Fungal Causes
Fungal meningitis typically is diagnosed in patients with predisposing risk factors, especially immunosuppression. Among all cases of fungal meningitis in pediatric and adult patients in the United States, Cryptococcus spp. prevails (70.1% of all cases), followed by Coccidioides spp. (16.4%), Candida spp. (7.6%), and Histoplasma capsulatum (6.0%).40 Pediatric cryptococcal meningitis usually affects the immunocompromised, especially those infected by HIV, although immunocompetent patients occasionally are affected.41,42 Candidal meningitis remains an important consideration in neonates and premature infants, although more often this is a problem of the neonatal intensive care unit than the emergency department.43,44 The risk for meningitis due to geographically endemic fungi like coccidioidomycosis and histoplasmosis usually is dictated by exposure history. Thus, the provider should be alert to any recent travel to affected regions.45,46
Helminthic and Protozoal Causes
Although rare, helminthic and protozoal causes of meningitis and meningoencephalitis constitute characteristic syndromes associated with certain risk factors and environmental exposures. The severe, progressive, and typically fatal meningoencephalitis due to the amebic protozoa Naegleria fowleri commonly is associated with warm freshwater exposure.47 Other parasitic and protozoal causes of meningitis and meningoencephalitis (e.g., Balamuthia mandrillaris, gnathostomiasis, angiostrongyliasis, etc.) typically are so rare and specialized as to fall outside the scope of this review.48,49,50
Pathophysiology and Natural History
Infective organisms from any of the previous categories may find their way to the human meninges based on several routes of inoculation or risk factors. By far, the most important is hematogenous spread, often from a distant nidus of infection (e.g., nasopharyngeal colonization leading to dissemination and eventual infection through the cerebral capillaries).51,52 This process likely depends on the magnitude of bacteremia, which may be achieved with certain pathogenic organisms capable of evading host defenses.52 Other potential routes of infection include direct extension through nasal, sinus, dental, and mastoid spaces; disruption of normal anatomical barriers via skull base fractures; or direct inoculation from surgical trauma or implanted material (e.g., cochlear implants, ventriculitis from intraventricular catheters, etc.).27-31,53
Different pathologic changes may be observed depending on the infective agent. In bacterial meningitis, direct damage to the cerebral cortex may occur through hypoxia, vascular occlusion or thrombi, toxic encephalopathy, and bacterial and inflammatory factors.54 The host immune response typically is characterized by migration of polymorphonuclear leukocytes across the blood barrier and into the subarachnoid space.51,55 Subdural effusions often may be seen as concomitant processes due to the severe degree of meningeal inflammation and extravasation of fluids from inflamed subdural veins and dural capillary beds.1 Patients may experience increased intracerebral pressure due to alterations in cerebral blood flow, changes in the permeability of cerebral vasculature, and inadequate cerebrospinal fluid circulation.54 The classic drop in cerebrospinal fluid glucose, otherwise known as hypoglycorrhachia, occurs because of changes in glucose transport across the inflamed blood-brain barrier rather than because of bacterial consumption, as sometimes is suggested.1,56
In severe cases, neutrophilic infiltrates with vascular and parenchymatous changes may occur, accompanied also by cortical vein and venous sinus thrombosis.54 Hemorrhage or necrosis of cerebral cortex has been observed.57 Rarely, patients may develop subdural empyema.58 Cerebral herniation due to edema in fulminant bacterial and viral meningitis has been reported.54,59,60 Because of the severity of the pathophysiologic changes described above, the natural history of untreated bacterial meningitis is eventual coma and death. Late presentations may not necessarily be rescued, even with appropriate therapy.
For those patients who survive, the destructive processes and ensuing fibrosis may lead to complications ranging from sensorineural hearing loss, cerebral salt wasting, the syndrome of inappropriate antidiuretic hormone secretion, hydrocephalus from adhesive thickening of the arachnoid mater around the cisterns, or obstruction of the foramina of Magendie and Luschka.1,61
Other causes of meningitis may present a different course, with a variety of manifestations depending on the etiologic organism. Viral meningitis is described classically as mounting a less exuberant host immunologic response, comprised primarily of a mononuclear pleocytosis and without hypoglycorrhachia. However, the practitioner should be aware that studies have demonstrated significant overlap with polymorphonuclear pleocytosis and decreased CSF glucose between viral and bacterial meningitis and that such CSF findings are not fully reliable methods of distinguishing categories of etiologic agent.62,63 Viral meningitis also may accompanied by encephalitis, depending on the infective agent and host predisposition. However, for most agents, the natural history of viral meningitis holds a more favorable prognosis with minimal morbidity and mortality.64,65
Mycobacterial and fungal meningitis more often may present subacutely, and may manifest classically as basilar meningitis with lymphocytic pleocytosis,42,66 although neutrophilic predominance is also possible.67 Hydrocephalus may develop classically in mycobacterial and certain fungal meningitis syndromes because of obstruction of CSF circulation and reabsorption.66,68
Finally, meningitis characterized by eosinophilic predominance to the CSF pleocytosis is associated classically with a variety of atypical organisms in the differential diagnosis, including fungal, protozoan, and helminthic species.48-50,69
Clinical Presentation and Physical Findings
Although the signs and symptoms of meningitis may vary depending on the age of the patient (see Table 3), certain hallmarks remain constant. Viral and bacterial meningitis present acutely, progressing over the course of several hours to one or two days. In one study of bacterial and aseptic meningitis in pediatric patients, fever was seen in 93% of cases, followed by headache (84%), vomiting (71%), nausea (62%), and nuchal rigidity and pain (65%).70 Photophobia was described as highly specific (88%) but poorly sensitive (28%). A similar profile of symptoms has been noted in adult bacterial meningitis.71 Additional findings also may include seizures, altered mental status, irritability, and neurologic deficits. Findings more specific to infants include poor feeding, increased tone, bulging fontanelle, lethargy or drowsiness, “toxic appearance,” and altered cry.70,72 Of note, the presence of petechial rash in any age group is highly concerning for meningococcemia.
Table 3. Clinical Signs in Pediatric Meningitis 70,72,77,79 |
|||
Clinical Sign or Symptom |
Age Group |
Sensitivity |
Specificity |
|
Fever (> 40° C) |
All |
19% |
93% |
|
Fever not otherwise specified |
All |
76% |
34% |
|
Headache |
Children, adolescents |
76% |
53% |
|
Nausea/vomiting |
All |
71% |
62% |
|
Photophobia |
Children, adolescents |
28% |
88% |
|
Nuchal rigidity |
Children, adolescents |
39-65% |
54-89% |
|
Bulging fontanelle |
Neonates and infants |
36-50% |
62-90% |
|
Abnormal or high-pitched cry |
Neonates and infants |
84% |
52% |
|
Paradoxical irritability |
Neonates and infants |
— |
— |
|
Brudzinski’s neck sign |
Children, adolescents |
11-66% |
74-80% |
|
Brudzinski’s cheek sign |
Children, adolescents |
— |
— |
|
Brudzinski’s symphyseal sign |
Children, adolescents |
— |
— |
|
Kernig’s sign |
Children, adolescents |
14-53% |
85-95% |
|
Jolt accentuation headache |
Children, adolescents |
7-97% |
60-99% |
|
SOURCE: Author created. |
|||
Subtle signs may accompany mycobacterial and fungal meningitis, and a greater index of suspicion is required on the part of the clinician to make a diagnosis. The presentation typically is insidious in onset, sometimes progressing over months.42,46,66,68 Patients may complain of headache, with or without fever, vision changes, nausea, vomiting, and photophobia, progressing to altered mental status and coma. Notably, nuchal rigidity may be absent. Focal neurologic deficits have been described with basilar meningitis, more commonly including diplopia.42,66 Papilledema may accompany many subacute to chronic forms of meningitis, especially such fungal causes as cryptococcosis and coccidioidomycosis; thus, a proper fundoscopic exam should be performed.
In all forms of meningitis among older children and adolescents, it is thought that the pain due to irritation of inflamed meninges leads to the patient’s resistance to flexion of the neck, thus constituting nuchal rigidity. However, neonates and younger infants are unable to engage the neuromuscular control and strength necessary to resist an examiner’s passive flexion of the neck despite the pain they experience, and thus “neck stiffness” is found less commonly in infants younger than 12 months.73 Instead, neonates and infants may demonstrate “paradoxical irritability” with meningitis, whereby the pain of inflamed meninges irritated by an examiner’s handling will cause the infant to cry more while being held vs. when lying still on a flat surface.74 As many parents will attest, this is the opposite of what is expected with any baby or neonate, who typically would be comforted with holding and swaying movements. However, there is relatively little evidence besides anecdotal experience to confirm this finding.
Among neonates and infants with meningitis, a higher-pitched cry may be noted. This symptom, confirmed with acoustic analysis, appears to be a nonspecific response to intracranial insult.75 An association with hydrocephalus, brain damage, asphyxia, trisomies, and other CNS disease processes also has been demonstrated.
Examiners should assess infants for a bulging fontanelle. This constitutes an outward demonstration of increased intracranial pressure or inflammation in the meninges, the presence of which increases the likelihood of meningitis by almost eight-fold.72 However, this sign is poorly sensitive (36-50%) when correlated to CSF pleocytosis.70,72
Among older children, different maneuvers may be attempted to elicit classic signs of meningismus. Brudzinski’s neck sign may be elicited by attempting passive flexion of the child’s neck; to resist the resultant stretching of irritated meninges along the spinal canal and irritation of associated nerve roots, the child will respond by flexing the hips.76 (See Figure 1.) Likewise, Kernig’s sign may be demonstrated by passively flexing one hip to a 90-degree angle and attempting to passively extend the knee. (See Figure 2.) Pain due to stretching of the irritated meninges along the spinal canal and sciatic nerve roots will lead to resistance. Although these maneuvers remain time-tested indicators of meningitis, most studies to date show them to be of questionable sensitivity; Brudzinski’s neck sign has been found to have a sensitivity of 11% to 66%, and Kernig’s sign 14% to 53%.72,77 Therefore, such maneuvers cannot rule out meningitis. Their specificity may be more useful (74-80% for Brudzinski’s neck sign, and 85-95% for Kernig’s sign), and, thus, their presence should alert the clinician to the need for lumbar puncture and empiric therapy.
Figure 1. Brudzinski’s Neck Sign |
|
Brudzinski’s neck sign may be elicited by attempting passive flexion of the patient’s neck (A); in order to resist the resultant stretching of the irritated meninges along the spinal canal and irritation of associated nerve roots, the patient will respond by flexing the hips (B). |
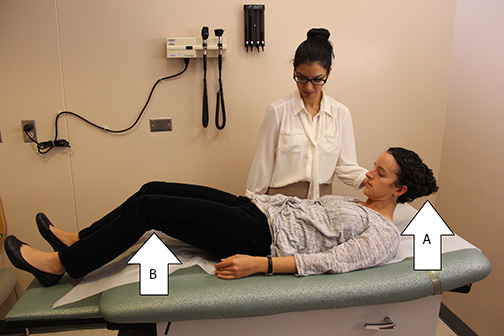 |
Figure 2. Kernig’s Sign |
|
Kernig’s sign may be demonstrated by passively flexing one hip to a 90-degree angle (A) and attempting to passively extend the knee (B). Pain due to stretching of the irritated meninges along the spinal canal and sciatic nerve roots will lead to resistance. |
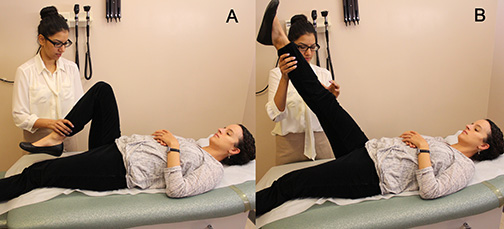 |
Additionally, Brudzinski’s eponym may be applied to further, lesser-known signs, including the symphyseal sign, in which pressure on the pubic symphysis leads to reflexive hip abduction and flexion with ipsilateral knee flexion, and the cheek sign, in which pressure on the child’s cheek below the zygomatic arch may lead to reflexive ipsilateral forearm flexion.78 The sensitivity of these signs is unknown, but their presence, as before, should alert the examiner that meningitis is highly suspected.
Additional descriptions have been made of a “Jolt Accentuation of Headache” or JAH sign, in which a conscious, cooperative patient is asked to quickly move his or her head side to side in a horizontal plane two to three times per second, and a concomitant increase in headache is considered positive.79 Although this maneuver initially was suggested to be far more sensitive (97%) in predicting CSF pleocytosis than the Kernig and Brudzinski signs, others have indicated its sensitivity is extremely variable (7% to 84%).80,81 Ultimately, the accuracy of any physical exam maneuver in distinguishing meningitis is unacceptably poor,80,82 and thus lumbar puncture and CSF analysis remain the gold standard for diagnosis.
The Lumbar Puncture and CSF Analysis
Fundamentally, meningitis of any etiology cannot be ruled out by history or physical exam alone. In any situation in which meningitis is suspected, the lumbar puncture with CSF analysis, Gram stain, and culture is important. Viral meningitis never can be distinguished from bacterial meningitis from a history and physical exam alone; obtaining CSF is critical for diagnosis.
Clinicians should be extremely wary of avoiding the lumbar puncture because of specious arguments regarding the likelihood of bacterial vs. viral meningitis.
The practitioner should bear in mind that certain contraindications to lumbar puncture must be appreciated. These include infections at or near the site of lumbar puncture.83 Anatomic abnormalities, such as spina bifida occulta, also will complicate the procedure, and the lumbar puncture should not be undertaken without further discussion with a neurosurgical service. Among the most important considerations is that of a possible space-occupying lesion or other source of increased intracranial pressure, such as a brain abscess or tumor.83 Undertaking lumbar puncture in the presence of an intracranial mass may risk disrupting the equilibrium of the cisternal pressure, and a tentorial or cerebellar “pressure cone” may form.83,84 This is followed by downward cerebral herniation, accompanied by seizures, obtundation, coma, and cardiorespiratory arrest.
Neuroimaging will assist the practitioner in identifying those at risk for cerebral herniation after lumbar puncture. To streamline the decision-making process, the Infectious Diseases Society of America has provided recommendations for computed tomography (CT) of the head before lumbar puncture. These include history of prior central nervous system disease, new-onset seizure, papilledema, immunocompromise, altered consciousness, or a focal neurologic deficit.85 Although these recommendations originally were targeted for adults, they may be extrapolated to the majority of pediatric patients. The practitioner should note that CT imaging in childhood may increase the later risk of cancer.86 However, if the child has a possible intracranial lesion, prudence dictates that the patient undergo rapid head imaging, even if it poses the risk of exposure to ionizing radiation.
After addressing any potential contraindications and reviewing any relevant neuroimaging, the practitioner may move forward with performing the lumbar puncture. Traditionally, the lumbar puncture is guided by anatomic landmarks.83 However, this may be a challenging procedure in pediatric patients because of the smaller size of the anatomic space at hand. Subsequent attempts after an initial failed lumbar puncture may be complicated by thecal displacement due to epidural or subdural hematoma formation.87 A high degree of success may be achieved with practice and training in the procedure. Ultrasound in the pediatric emergency department can be a useful adjunct.88 Ultrasound compared to the fluoroscopic approach has advantages, including speed and decreased exposure to ionizing radiation.89
An opening pressure should be obtained at the time of lumbar puncture, if possible. Measurement of the opening pressure may be difficult to achieve in an agitated neonate or infant. Notably, movement and crying may falsely elevate CSF opening pressure. The child must be in a lateral decubitus position with the legs extended and relaxed. Flexion of the legs in this position may cause minor false elevations in CSF opening pressure.90-92 An opening pressure greater than the upper limit of normal (28 cm H2O in children older than 1 year of age) is suspicious for meningitis or other pathologic processes, such as hydrocephalus or idiopathic intracranial hypertension.93,94
After accessing the subarachnoid space and obtaining adequate specimens, the CSF analysis must be undertaken. This analysis always should include quantification of glucose and protein, along with a cell count with differential, Gram stain, and culture.
The CSF glucose should be obtained simultaneously with a serum glucose. This is done to ensure that if CSF glucose is low, it can be correlated with serum levels to be distinguished from mere hypoglycemia vs. hypoglycorrhachia secondary to an infectious process.56,83,95 Hyperglycemia also potentially could mask hypoglycorrhachia; in this situation, measurement of the ratio of CSF glucose to blood glucose should be performed, and a value < 0.3 should be considered abnormal. Most commonly, meningitis with low CSF glucose is associated with bacterial etiologies. CSF glucose may be normal in viral meningitis, while in mycobacterial and fungal etiologies, the CSF glucose may range from low to normal, but hypoglycorrhachia has been found in meningitis due to almost any infectious etiology.95 Thus, the CSF glucose is not a reliable method for distinguishing etiologic agents of meningitis.
As a nonspecific marker, CSF protein typically is more concerning when it is elevated, which may be indicative of meningitis from any of the possible bacterial, mycobacterial, viral, or fungal etiologies, among other causes.83
Normal CSF should be clear and colorless, although pathologic processes still may be present with a normal-appearing specimen.83,96 Purulent or cloudy CSF is concerning for bacterial meningitis. Xanthochromia may be indicative of subarachnoid hemorrhage.
The cell count is possibly the most important laboratory value when diagnosing meningitis. Elevations in CSF white blood cells (WBC) are indicative of an inflammatory process, most likely an infectious one. However, such elevations must be measured against known normal values for CSF white blood cell counts, which may vary considerably with age.93 Notably, newborn infants up to one month of age may have a normal CSF WBC count of up to 19/µL, while the upper limit of normal for an older child is lower, between 6-9/µL.93
As mentioned previously, the character of this CSF pleocytosis also is potentially indicative (but not pathognomonic) of the category of pathogenic organism that is responsible for the patient’s infection. In general, neutrophilic predominance is thought to be suggestive of bacterial etiologies, while lymphocytic predominance may be seen in viral, mycobacterial, and fungal meningitis.96 However, the degree of overlap between these entities may seriously confound the diagnosis; in one cohort, the majority of patients with CSF neutrophilic predominance were found to have viral meningitis.97 Likewise, lymphocytic predominance in the CSF in bacterial meningitis has been reported as more common than previously believed.98,99 Immature neutrophils, previously thought to increase the likelihood of bacterial meningitis, have been found to be nonspecific in distinguishing bacterial from viral etiologies.100 Finally, the presence of eosinophils in CSF pleocytosis may lead the practitioner to consider a more unique differential diagnosis, which includes fungal and parasitic causes.69
Differences in any of these parameters may lead the practitioner to consider various etiologies for meningitis and expand empiric coverage as necessary. Other tests to order include nucleic acid-based tests. The use of enterovirus polymerase chain reaction (PCR) tests, if available, may help to quickly confirm an etiologic cause for viral meningitis, leading to decreased time on empiric antibiotics and reduced hospital stay.101-104 Some institutions use a PCR-based assay for a variety of known pathogenic organisms in pediatric meningitis and encephalitis to identify an etiologic organism quickly.104,105 A herpes simplex virus (HSV) PCR test always should be considered in febrile neonates or in older children with findings concerning for encephalitis.106
Ordering a CSF Gram stain and culture is an important step in diagnosing bacterial meningitis. The true utility of the Gram stain lies in helping the practitioner expand coverage as needed, rather than narrowing it. This is because interpretation of a Gram-stained slide is dependent on the expertise and experience of the individual reading the slide.85 In patients with bacterial meningitis, CSF cultures obtained prior to initiation of antibiotic therapy are positive in 70-85% of cases, although a period of up to 48 hours may be necessary for organism identification.85 Thus, patients usually will require hospitalization during this time for intravenous empiric antibiotics until cultures may be reliably deemed negative, although “culture-negative” bacterial meningitis still may be a possibility for certain organisms. (See Table 1.)
Ideally, the lumbar puncture with CSF analysis, Gram stain, and culture will be obtained quickly before initiating empiric therapy. However, the limitations of a busy emergency department, difficult-to-obtain specimens or failed lumbar punctures, and subsequent transfers of patient care from regional emergency departments to tertiary centers sometimes fall short of the ideal in practice. Given the mortality and morbidity of possible bacterial meningitis and the inconsistent rapidity with which patients may deteriorate, empiric antibiotic treatment should not be delayed longer than absolutely necessary.107 If the first lumbar puncture attempt is not successful, then antibiotic therapy must be initiated despite the lack of a “pre-treatment” CSF sample. This complicates the physician’s approach, because CSF may be sterilized by empiric parenteral antibiotics within two to four hours of initiating therapy.108 Nevertheless, a CSF sample still is critical to obtain in such situations, as the presence and character of pleocytosis in a sample still may inform subsequent therapy.107 Indeed, even with effective antibiotic therapy for bacterial meningitis, pleocytosis may persist for more than 10 days.109,110
Another factor that may obfuscate the diagnosis of meningitis is the “traumatic” or “bloody” lumbar puncture. In this situation, the CSF sample is contaminated with venous blood from a local vessel that has been penetrated inadvertently. This may lead to falsely elevated counts in the CSF analysis, with red blood cells (RBCs) numbering in the several thousands, and the concern that any WBC count is simply due to leukocytes from the patient’s contaminating venous bleed. Thus, the provider cannot say whether CSF pleocytosis truly is present, and the diagnosis of meningitis is obscured.
There is debate as to whether “correction” factors may be used to determine the presence of pleocytosis from a CSF sample with blood contamination. These include the calculation of a ratio of WBC:RBC counts from the blood vs. the CSF.111 Other methods simply subtract 1 CSF WBC for every 500 to 1,000 CSF RBCs, and any excess beyond this factor constitutes pleocytosis.112 However, it must be emphasized that although such practices are common, they are not universally accepted and may lead to underrecognition of pleocytosis.
Differential Diagnosis
When presented with the signs and symptoms discussed above, the pediatric provider must consider infectious meningitis at the top of the differential. Patients with bacterial meningitis have a limited amount of time before the disease progresses to coma and death, potentially within hours. Therefore, the provider must perform a lumbar puncture and CSF analysis quickly, with prompt initiation of empiric antibiotic therapy.
Fever, headache, neurologic deficits, seizures, and altered mental status are additionally concerning for encephalitis or meningoencephalitis, in which the parenchyma of the brain may be involved in the infective process. HSV is a frequent culprit, and empiric antiviral therapy must be undertaken if this is a consideration.106 Likewise, with certain bacterial agents and host characteristics, syndromes like endocarditis with septic thromboemboli or brain abscess also may be suspected. The practitioner must be aware that encephalitis and brain abscesses may not lead to significant or worrisome CSF changes; indeed, pleocytosis even may be absent.106,113 Thus, a high degree of suspicion is required to make the diagnosis in such cases, typically using magnetic resonance imaging.
Clinicians should recall that with any invasive bacterial infection, infants and neonates may present with subtle clinical signs, rather than those typical of older children and adolescents. This is particularly true for very young infants or neonates. Thus, any fever in a child younger than 90 days old, especially among the subset of those younger than 30 days of age, demands consideration of meningitis or another invasive bacterial infection.114 Numerous guidelines and clinical rules have been researched to provide recommendations on pursuing a full septic evaluation in such situations, including lumbar puncture with CSF studies and cultures, blood cultures, and empiric antibiotic therapy.115-122
Fever, seizures, and encephalopathy demand high suspicion for HSV encephalitis.106 Among neonates with HSV disease, only about 20-40% of mothers report active or known genital herpes during or before the pregnancy, and neonates with encephalitis frequently have no skin or mucous membrane findings.123 Therefore, a high index of suspicion is essential to the diagnosis.
Encephalopathy and fever in a child recently returned from a malaria-endemic region should be highly concerning for cerebral malaria, which may progress to coma and death in more than a third of patients.124
Other CNS disease processes, including subarachnoid hemorrhage, CNS tumor, or hydrocephalus of non-infectious cause, can be excluded with neuroimaging.
Management
In cases of suspected meningitis, practitioners first should assess the cardiopulmonary status and airway of the patient, and undertake basic life support or pediatric advanced life support measures if necessary.125 Upon establishing suspicion of meningitis, blood samples for cultures and lumbar puncture should be performed immediately.85 As mentioned previously, practitioners should assess the need for CT scan prior to lumbar puncture.
Until bacterial meningitis can be reliably ruled out with CSF analysis and culture, empiric antibiotic therapy is indicated to prevent further progression of disease. Although CSF culture is most accurate prior to initiating antibiotics, practitioners should not delay appropriate therapy, even if obtaining the lumbar puncture must be delayed for any reason. Acting quickly when such a case presents, practitioners should simultaneously attempt to obtain the lumbar puncture while antibiotics are procured. In the case of a failed lumbar puncture, antibiotics simply should be administered as soon as possible before re-attempting the procedure. As previously noted, although empiric antibiotic treatment may render CSF sterile in the context of bacterial meningitis, the practitioner still will have the ability to make the diagnosis of meningitis based on CSF pleocytosis for possibly several days.109,110
Antibiotics should be chosen based on the most likely etiologic agents for the child’s age group and risk factors. (See Table 4.) The chosen empiric regimen also should achieve adequate CNS penetration. In infants younger than 1 month of age, Group B streptococcal infection or E. coli predominate.85 Adequate coverage will be achieved with ampicillin and cefotaxime, administered parenterally at meningitic doses. Ampicillin plus gentamicin is also an acceptable choice.85
Table 4. Empiric Antibiotic Choices by Age Group 85,129 |
|
Age Group |
Antibiotic Choice and Dosing |
|
Neonates 0-7 days of age |
Ampicillin IV (200 to 300 mg/kg/day divided into Q8H dosing) PLUS Cefotaxime IV (100 to 150 mg/kg/day divided into Q8H or Q12H dosing) OR gentamicin IV (4 mg/kg/day divided into Q24H dosing) |
|
Neonates 8-28 days of age |
Ampicillin IV (300 mg/kg/day divided into Q6H dosing) PLUS Cefotaxime IV (150 to 200 mg/kg/day divided into Q8H dosing) OR gentamicin IV (4 mg/kg/day divided into Q12H dosing) |
|
Infants >1 month of age, children, and adolescents |
Ceftriaxone IV (100 mg/kg/day divided into Q12H dosing) PLUS Vancomycin IV (20 mg/kg given Q6H) |
|
Adults |
Ceftriaxone IV (2,000 mg given Q12H) PLUS Vancomycin IV (20 mg/kg given Q8H or Q12H) |
|
SOURCE: Author created. |
|
Older infants, children, and adolescents will benefit from broader coverage. Agents that must be considered and treated empirically in this age group include S. pneumoniae and N. meningitidis. Thus, in pediatric patients older than 1 month, vancomycin and a third-generation cephalosporin (usually ceftriaxone) should be administered parenterally at meningitic doses.85 Children and adolescents with recent neurosurgery and CSF shunts are at risk for a broader array of organisms, and therefore broader coverage with a fourth-generation cephalosporin (i.e., cefepime) may be used in place of a third-generation cephalosporin.85 Children with neurosurgical risk factors, such as ventriculoperitoneal shunts, will benefit from empiric
coverage from vancomycin against coagulase-negative Staphylococcus and S. aureus.
Adjunctive dexamethasone often is suggested for infants and children with bacterial meningitis. Adjunctive dexamethasone has been shown to prevent hearing loss in pediatric patients with meningitis due to H. influenzae type b.126 A more recent meta-analysis in the vaccination era found that dexamethasone did not reduce death or neurologic disability in children with bacterial meningitis.127 At this time, the Infectious Diseases Society of America only recommends the use of adjunctive dexamethasone in cases of H. influenzae type b.85 In cases of S. pneumoniae meningitis, adjunctive dexamethasone may be considered, but the benefit of this therapy is not well established.
Empiric treatment for most viral causes of meningitis are not available. However, if HSV encephalitis is a consideration within the differential diagnosis, acyclovir should be administered parenterally until adequate CSF analysis is performed with HSV PCR to rule out this entity.106 Because the sensitivity of this test may be low in the early stages of disease, patients with a high suspicion of HSV encephalitis should have a repeat lumbar puncture and CSF HSV PCR performed three to seven days later.106
Fungal, mycobacterial, and parasitic causes of meningitis are more difficult to distinguish in the emergency department, and often such empiric coverage is delayed until further laboratory results are available. Antifungal, antituberculous, and antiparasitic medications usually are chosen based on the specific etiologic agent.
After appropriate stabilization, diagnostic testing, and initiation of empiric antibiotics, children with evidence or suspicion of meningitis should be admitted for ongoing observation and intravenous empiric antimicrobial therapy. Although data from CSF analysis is suggestive in distinguishing various etiologies of meningitis, a negative CSF culture is far more reassuring against bacterial meningitis, and therefore patients should be admitted for at least 48 hours for empiric therapy. Patients with high suspicion of bacterial meningitis, altered mental status, or focal neurologic findings warrant intensive care unit admission because of their higher risk for neurologic complications, such as seizures or cerebral edema. In the case of proven bacterial meningitis, intravenous antibiotic therapy may be narrowed based on cultures and susceptibilities, and thereafter continued for 7 to 21 days, depending on the etiologic agent and complications.85
Close contacts of patients with N. meningitidis should be prescribed chemoprophylaxis with rifampin, ceftriaxone, ciprofloxacin, or azithromycin within 24 hours.128 Those at high risk who warrant chemoprophylaxis include those who may share a patient’s oral secretions through close personal contact (e.g., kissing, sharing eating utensils), child care or preschool contacts within seven days before onset of disease, people who sleep within the same household as the index case, passengers seated next to an index case on an airplane for eight hours or more, and healthcare personnel who may have undertaken high-risk interventions like mouth-to-mouth resuscitation, intubation, or suctioning in patients less than 24 hours before initiating antibiotic therapy.128 Immunization status with any of the meningococcal vaccines does not eliminate the need for chemoprophylaxis for these high-risk groups.
Conclusion
Infectious meningitis in pediatric patients may arise due to viral, bacterial, mycobacterial, fungal, protozoan, or helminthic etiologic agents. Practitioners must be alert to those infections that carry a high risk for morbidity and mortality. There are no symptoms, physical findings, or exam maneuvers that may reliably rule out the possibility of bacterial meningitis, and the clinical picture may be unclear or obfuscated in infants and neonates. Therefore, the lumbar puncture with CSF analysis, Gram stain, and culture is the most critical path to diagnosing or ruling out pediatric meningitis. Nucleic acid testing for viral and bacterial pathogens is helpful in establishing a diagnosis. Certain patients at risk of cerebral herniation should undergo CT neuroimaging prior to attempting lumbar puncture. In situations in which CSF cannot be obtained or the CSF analysis is complicated by contamination with venous blood, the practitioner should err on the side of empiric antibiotic therapy. Re-attempting the lumbar puncture with CSF analysis, even after initiation of therapy, is still a useful adjunct to diagnosis.
Empiric therapy should be chosen based on patient age and risk factors. Dexamethasone is known to benefit patients with meningitis due to
H. influenzae type b, but there is no established benefit for pediatric patients with other bacterial causes of meningitis. Patients with suspected meningitis should be admitted for empiric therapy and observation until bacterial causes can be ruled out from CSF cultures. Those with high suspicion for bacterial meningitis, altered mental status, or focal neurologic findings should be admitted to an intensive care unit. Therapy then should be narrowed according to culture results and susceptibilities and continued based on the identified etiologic agent. Close contacts of patients infected with N. meningitidis, as well as household members, day care or preschool contacts, and healthcare providers at risk of direct contact with oral secretions, should undertake chemoprophylaxis.
The author would like to acknowledge Dr. Kareem Shehab for his guidance in reviewing the accuracy of the manuscript, and Dr. Rachel Manzo and Dr. Maria Khan for their assistance in demonstrating the clinical signs of meningitis.
REFERENCES
- Kim KS. Bacterial Meningitis Beyond the Neonatal Period. In: Cherry J, Harrison GJ, Kaplan SL, et al, eds. Feigin and Cherry’s Textbook of Pediatric Infectious Diseases. 8th ed. Philadelphia, PA: Elsevier; 2018.
- Bronstein DE, Glaser CA. Aseptic Meningitis and Viral Meningitis. In: Cherry J, Harrison GJ, Kaplan SL, et al, eds. Feigin and Cherry’s Textbook of Pediatric Infectious Diseases. 8th ed. Philadelphia, PA: Elsevier; 2018.
- Martin NG, Iro MA, Sadarangani M, et al. Hospital admissions for viral meningitis in children in England over five decades: A population-based observational study. Lancet Infect Dis 2016;16:1279-1287.
- Davison KL, Ramsay ME. The epidemiology of acute meningitis in children in England and Wales. Arch Dis Child 2003;88:662-664.
- Takhar SS, Ting SA, Camargo CA Jr, Pallin DJ. U.S. emergency department visits for meningitis, 1993-2008. Acad Emerg Med 2012;19:632-639.
- Khetsuriani N, Quiroz ES, Holman RC, Anderson LJ. Viral meningitis-associated hospitalizations in the United States, 1988-1999. Neuroepidemiology 2003;22:345-352.
- Kadambari S, Okike I, Ribeiro S, et al. Seven-fold increase in viral meningo-encephalitis reports in England and Wales during 2004-2013. J Infect 2014;68:326-332.
- Maguire HC, Atkinson P, Sharland M, Bendig J. Enterovirus infections in England and Wales: Laboratory surveillance data: 1975 to 1994. Commun Dis Public Health 1999;2:122-125.
- Khetsuriani N, Lamonte-Fowlkes A, Oberst S, Pallansch MA, Centers for Disease Control and Prevention. Enterovirus surveillance — United States, 1970-2005. MMWR Surveill Summ 2006;55:1-20.
- Romero JR, Newland JG. Viral meningitis and encephalitis: Traditional and emerging viral agents. Semin Pediatr Infect Dis 2003;14:72-82.
- Thigpen MC, Whitney CG, Messonnier NE, et al. Bacterial meningitis in the United States, 1998-2007. N Engl J Med 2011;364:2016-2025.
- Shuchat A, Robinson K, Wenger JD, et al. Bacterial meningitis in the United States in 1995. N Engl J Med 1997;337:970-976.
- Whitney CG, Farley MM, Hadler J, et al. Decline in invasive pneumococcal disease after the introduction of protein-polysaccharide conjugate vaccine. N Engl J Med 2003;348:1737-1746.
- Kaplan SL, Mason EO, Wald ER, et al. Decrease of invasive pneumococcal infections among 8 children’s hospitals in the United States after the introduction of the 7-valent pneumococcal conjugate vaccine. Pediatrics 2004;113:443-449.
- Martin NG, Sadarangani M, Pollard AJ, Goldacre MJ. Hospital admission rates for meningitis and septicaemia caused by Haemophilus influenzae, Neisseria meningitidis, and Streptococcus pneumoniae in children in England over five decades: A population-based observational study. Lancet Infect Dis 2014;14:397-405.
- Brouwer MC, van de Beek D. Epidemiology of community-acquired bacterial meningitis. Curr Opin Infect Dis 2018;31:78-84.
- McIntyre PB, O’Brien KL, Greenwood B, van de Beek D. Effect of vaccines on bacterial meningitis worldwide. Lancet 2012;380:1703-1711.
- Bottomley MJ, Serruto D, Sáfadi MA, Klugman KP. Future challenges in the elimination of bacterial meningitis. Vaccine 2012;30(Suppl 2):B78-86.
- Nizet V, Klein JO. Bacterial Sepsis and Meningitis. In: Wilson CB, Nizet V, Maldonado YA, et al, eds. Remington and Klein Infectious Diseases of the Fetus and Newborn Infant. 8th ed. Philadelphia, PA: Elsevier; 2015.
- Centers for Disease Control and Prevention. Prevention of Perinatal Group B Streptococcal Disease Revised Guidelines from CDC, 2010. MMWR Morb Mortal Wkly Rep 2010;59:1-32.
- Phares CR, Lynfield R, Farley MM, et al. Epidemiology of invasive group B streptococcal disease in the United States, 1999-2005. JAMA 2008;299:2056-2065.
- Van Dyke MK, Phares CR, Lynfield R, et al. Evaluation of universal antenatal screening for group B streptococcus. N Engl J Med 2009;360:2626-2636.
- Eraklis AJ, Kevy SV, Diamond LK, Gross RE. Hazard of overwhelming infection after splenectomy in childhood. N Engl J Med 1967;276:1225-1229.
- Sommers LM, Hawkins DS. Meningitis in pediatric cancer patients: A review of forty cases from a single institution. Pediatr Infect Dis J 1999;18:902-907.
- Chao YN, Chiu NC, Huang FY. Clinical features and prognostic factors in childhood pneumococcal meningitis. J Microbiol Immunol Infect 2008;41:48-53.
- Hénaff F, Levy C, Cohen R, et al. Risk factors in children older than 5 years with pneumococcal meningitis: Data from a national network. Pediatr Infect Dis J 2017;36:457-461.
- Farinetti A, Ben Gharbia D, Mancini J, et al. Cochlear implant complications in 403 patients: Comparative study of adults and children and review of the literature. Eur Ann Otorhinolaryngol Head Neck Dis 2014;131:177-182.
- Wei BP, Shepherd RK, Robins-Browne RM, et al. Pneumococcal meningitis post-cochlear implantation: Potential routes of infection and pathophysiology. Otolaryngol Head Neck Surg 2010;143:S15-23.
- Chamberlain MC, Kormanik PA, Barba D. Complications associated with intraventricular chemotherapy in patients with leptomeningeal metastases. J Neurosurg 1997;87:694-699.
- Scheithauer S, Bürgel U, Ryang YM, et al. Prospective surveillance of drain associated meningitis/ventriculitis in a neurosurgery and neurological intensive care unit. J Neurol Neurosurg Psychiatry 2009;80:1381-1385.
- McCutcheon BA, Orosco RK, Chang DC, et al. Outcomes of isolated basilar skull fracture: Readmission, meningitis, and cerebrospinal fluid leak. Otolaryngol Head Neck Surg 2013;149:931-939.
- World Health Organization. Global Tuberculosis Report 2017. Available at: http://www.who.int/tb/publications/global_report/gtbr2017_annex4.pdf?ua=1. Accessed April 9, 2018.
- Jaffe IP. Tuberculous meningitis in childhood. Lancet 1982;1:738.
- Marais BJ, Gie RP, Schaaf HS, et al. The natural history of childhood intra-thoracic tuberculosis: A critical review of literature from the pre-chemotherapy era. Int J Tuberc Lung Dis 2004;8:392-402.
- Tuberculosis. In: Kimberlin DW, Brady MT, Jackson MA, Long SS, eds. Red Book 2015 Report of the Committee on Infectious Diseases. 30th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2015.
- Doerr CA, Starke JR, Ong LT. Clinical and public health aspects of tuberculous meningitis in children. J Pediatr 1995;127:27-33.
- Yaramis A, Gurkan F, Elevli M, et al. Central nervous system tuberculosis in children: A review of 214 cases. Pediatrics 1998;102:E49.
- Rowe JS, Shah SS, Marais BJ, Steenhoff AP. Diagnosis and management of tuberculous meningitis in HIV-infected pediatric patients. Pediatr Infect Dis J 2009;28:147-148.
- Moliva JI, Turner J, Torrelles JB. Prospects in Mycobacterium bovis Bacille Calmette et Guérin (BCG) vaccine diversity and delivery: Why does BCG fail to protect against tuberculosis? Vaccine 2015;33:5035-5041.
- Charalambous LT, Premji A, Tybout C, et al. Prevalence, healthcare resource utilization and overall burden of fungal meningitis in the United States. J Med Microbiol 2018;67:215-227.
- Joshi NS, Fisher BT, Prasad PA, Zaoutis TE. Epidemiology of cryptococcal infection in hospitalized children. Pediatr Infect Dis J 2010;29:e91-95.
- Luo FL, Tao YH, Wang YM, Li H. Clinical study of 23 pediatric patients with cryptococcosis. Eur Rev Med Pharmacol Sci 2015;19:3801-3810.
- Yu Y, Du L, Yuan T, et al. Risk factors and clinical analysis for invasive fungal infection in neonatal intensive care unit patients. Am J Perinatol 2013;30:589-594.
- Fernandez M, Moylett EH, Noyola DE, Baker CJ. Candidal meningitis in neonates: A 10-year review. Clin Infect Dis 2000;31:458-463.
- Brown J, Benedict K, Park BJ, Thompson GR 3rd. Coccidioidomycosis: Epidemiology. Clin Epidemiol 2013;5:185-197.
- Saccente M. Central nervous system histoplasmosis. Curr Treat Options Neurol 2008;10:161-167.
- Stowe RC, Pehlivan D, Friederich KE, et al. Primary amebic meningoencephalitis in children: A report of two fatal cases and review of the literature. Pediatr Neurol 2017;70:75-79.
- Trabelsi H, Dendana F, Sellami A, et al. Pathogenic free-living amoebae: Epidemiology and clinical review. Pathol Biol (Paris) 2012;60:399-405.
- Sawanyawisuth K, Chindaprasirt J, Senthong V, et al. Clinical manifestations of Eosinophilic meningitis due to infection with Angiostrongylus cantonensis in children. Korean J Parasitol 2013;51:735-738.
- Shah I, Barot S, Madvariya M. Eosinophilic meningitis: A case series and review of literature of Angiostrongylus cantonensis and Gnathostoma spinigerum. Indian J Med Microbiol 2015;33:154-158.
- Kim KS. Pathogenesis of bacterial meningitis: From bacteraemia to neuronal injury. Nat Rev Neurosci 2003;4:376-385.
- Kim KS. Mechanisms of microbial traversal of the blood-brain barrier. Nat Rev Microbiol 2008;6:625-634.
- Isaacson B, Mirabal C, Kutz JW Jr, et al. Pediatric otogenic intracranial abscesses. Otolaryngol Head Neck Surg 2010;142:434-437.
- Gerber J, Nau R. Mechanisms of injury in bacterial meningitis. Curr Opin Neurol 2010;23:312-318.
- Koedel U, Klein M, Pfister HW. New understandings on the pathophysiology of bacterial meningitis. Curr Opin Infect Dis 2010;23:217-223.
- Viola GM. Extreme hypoglycorrhachia: Not always bacterial meningitis. Nat Rev Neurol 2010;6:637-641.
- Engelen-Lee JY, Brouwer MC, Aronica E, van de Beek D. Pneumococcal meningitis: Clinical-pathological correlations (MeninGene-Path). Acta Neuropathol Commun 2016;4:26.
- Jim KK, Brouwer MC, van der Ende A, van de Beek D. Subdural empyema in bacterial meningitis. Neurology 2012;79:2133-2139.
- Muralidharan R, Rabinstein AA, Wijdicks EF. Cervicomedullary injury after pneumococcal meningitis with brain edema. Arch Neurol 2011;68:513-516.
- Krous HF, Chadwick AE, Miller DC, et al. Sudden death in toddlers with viral meningitis, massive cerebral edema, and neurogenic pulmonary edema and hemorrhage: Report of two cases. Pediatr Dev Pathol 2007;10:463-469.
- Dodge PR, Swartz MN. Bacterial meningitis — A review of selected aspects. II. Special neurologic problems, postmeningitic complications and clinicopathological correlations. N Engl J Med 1965;272:1003-1010.
- Negrini B, Kelleher KJ, Wald ER. Cerebrospinal fluid findings in aseptic versus bacterial meningitis. Pediatrics 2000;105:316-319.
- Michos AG, Syriopoulou VP, Hadjichristodoulou C, et al. Aseptic meningitis in children: Analysis of 506 cases. PLoS One 2007;2:e674.
- Nigrovic LE, Fine AM, Monuteaux MC, et al. Trends in the management of viral meningitis at United States children’s hospitals. Pediatrics 2013;131:670-676.
- Lee BE, Chawla R, Langley JM, et al. Paediatric Investigators Collaborative Network on Infections in Canada (PICNIC) study of aseptic meningitis. BMC Infect Dis 2006;6:68.
- Romero JR, Jacobs RF. Fungal Meningitis. In: Cherry J, Harrison GJ, Kaplan SL, et al, eds. Feigin and Cherry’s Textbook of Pediatric Infectious Diseases. 8th ed. Philadelphia, PA: Elsevier; 2018.
- Zou Y, He J, Guo L, et al. Prediction of cerebrospinal fluid parameters for tuberculous meningitis. Diagn Cytopathol 2015;43:701-704.
- Raut T, Garg RK, Jain A, et al. Hydrocephalus in tuberculous meningitis: Incidence, its predictive factors and impact on the prognosis. J Infect 2013;66:330-337.
- Weatherhead J, Mejia R. Eosinophilic Meningitis. In: Cherry J, Harrison GJ, Kaplan SL, et al, eds. Feigin and Cherry’s Textbook of Pediatric Infectious Diseases. 8th ed. Philadelphia, PA: Elsevier; 2018.
- Amarilyo G, Alper A, Ben-Tov A, Grisaru-Soen G. Diagnostic accuracy of clinical symptoms and signs in children with meningitis. Pediatr Emerg Care 2011;27:196-199.
- Durand ML, Calderwood SB, Weber DJ, et al. Acute bacterial meningitis in adults. A review of 493 episodes. N Engl J Med 1993;328:21-28.
- Curtis S, Stobart K, Vandermeer B, et al. Clinical features suggestive of meningitis in children: A systematic review of prospective data. Pediatrics 2010;126:952-960.
- Walsh-Kelly C, Nelson DB, Smith DS, Losek JD, et al. Clinical predictors of bacterial versus aseptic meningitis in childhood. Ann Emerg Med 1992;21:910-914.
- Rabbitt AL. Irritable Infant. In: Kliegman RM, Lye PS, Bordini BJ, et al, eds. Nelson Pediatric Symptom-Based Diagnosis. 1st ed. Philadelphia, PA: Elsevier; 2017.
- LaGasse LL, Neal AR, Lester BM. Assessment of infant cry: Acoustic cry analysis and parental perception. Ment Retard Dev Disabil Res Rev 2005;11:83-93.
- Ward MA, Greenwood TM, Kumar DR, et al. Josef Brudzinski and Vladimir Mikhailovich Kernig: Signs for diagnosing meningitis. Clin Med Res 2010;8:13-17.
- Bilavsky E, Leibovitz E, Elkon-Tamir E, et al. The diagnostic accuracy of the ‘classic meningeal signs’ in children with suspected bacterial meningitis. Eur J Emerg Med 2013;20:361-363.
- Campbell WW. Clinical Signs in Neurology: A Compendium. 1st ed. Philadelphia: Wolters Kluwer; 2016.
- Uchihara T, Tsukagoshi H. Jolt accentuation of headache: The most sensitive sign of CSF pleocytosis. Headache 1991;31:167-171.
- Nakao JH, Jafri FN, Shah K, Newman DH. Jolt accentuation of headache and other clinical signs: Poor predictors of meningitis in adults. Am J Emerg Med 2014;32:24-28.
- Ala A, Rahmani F, Abdollahi S, Parsian A. Accuracy of neck stiffness, Kernig, Brudzinski, and Jolt Accentuation of Headache signs in early detection of meningitis. Emerg (Tehran) 2018;6(1):e8.
- Waghdhare S, Kalantri A, Joshi R, Kalantri S. Accuracy of physical signs for detecting meningitis: A hospital-based diagnostic accuracy study. Clin Neurol Neurosurg 2010;112:752-757.
- Euerle BD. Spinal Puncture and Cerebrospinal Fluid Examination. In: James RR, Custalow CB, Thomsen TW, eds. Roberts and Hedges’ Clinical Procedures in Emergency Medicine and Acute Care. 7th ed. Philadelphia, PA: Elsevier; 2018.
- Joffe AR. Lumbar puncture and brain herniation in acute bacterial meningitis: A review. J Intensive Care Med 2007;22:194-207.
- Tunkel AR, Hartman BJ, Kaplan SL, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis 2004;39:1267-1284.
- Pearce MS, Salotti JA, Little MP, et al. Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: A retrospective cohort study. Lancet 2012;380:499-505.
- Kim S, Adler DK. Ultrasound-assisted lumbar puncture in pediatric emergency medicine. J Emerg Med 2014;47:59-64.
- Muthusami P, Robinson AJ, Shroff MM. Ultrasound guidance for difficult lumbar puncture in children: Pearls and pitfalls. Pediatr Radiol 2017;47:822-830.
- Pierce DB, Shivaram G, Koo KSH, et al. Ultrasound-guided lumbar puncture in pediatric patients: Technical success and safety. Pediatr Radiol 2018; Feb 3. doi: 10.1007/s00247-018-4091-2. [Epub ahead of print].
- Ellis R 3rd. Lumbar cerebrospinal fluid opening pressure measured in a flexed lateral decubitus position in children. Pediatrics 1994;93:622-623.
- Avery RA. Interpretation of lumbar puncture opening pressure measurements in children. J Neuroophthalmol 2014;34:284-287.
- Avery RA, Mistry RD, Shah SS, et al. Patient position during lumbar puncture has no meaningful effect on cerebrospinal fluid opening pressure in children. J Child Neurol 2010;25:616-619.
- Blood chemistries and body fluids. In: Hughes HK, Kahl LK, eds. The Harriet Lane Handbook. 21st ed. Philadelphia, PA: Elsevier; 2017.
- Avery RA, Shah SS, Licht DJ, et al. Reference range for cerebrospinal fluid opening pressure in children. N Engl J Med 2010;363:891-893.
- Silver TS, Todd JK. Hypoglycorrhachia in pediatric patients. Pediatrics 1976;58:67-71.
- Lehman RK, Schor NF. Neurologic Evaluation. In: Kliegman RM, Stanton BF, St Geme JW, Schor NF, eds. Nelson Textbook of Pediatrics. 20th ed. Philadelphia, PA: Elsevier; 2015.
- Jaijakul S, Salazar L, Wootton SH, et al. The clinical significance of neutrophilic pleocytosis in cerebrospinal fluid in patients with viral central nervous system infections. Int J Infect Dis 2017;59:77-81.
- Samakoses R, Suwanpakdee D, Watanaveeradej V, Kerdpanich P. Cerebrospinal fluid lymphocytosis in an infant with acute Streptococcus pnuemoniae meningitis: A case report. J Med Assoc Thai 2010;93(Suppl 5):S49-52.
- Powers WJ. Cerebrospinal fluid lymphocytosis in acute bacterial meningitis. Am J Med 1985;79:216-220.
- Kanegaye JT, Nigrovic LE, Malley R, et al. Diagnostic value of immature neutrophils (bands) in the cerebrospinal fluid of children with cerebrospinal fluid pleocytosis. Pediatrics 2009;123:e967-e971.
- Huizing KM, Swanink CM, Landstra AM, et al. Rapid enterovirus molecular testing in cerebrospinal fluid reduces length of hospitalization and duration of antibiotic therapy in children with aseptic meningitis. Pediatr Infect Dis J 2011;30:1107-1109.
- King RL, Lorch SA, Cohen DM, et al. Routine cerebrospinal fluid enterovirus polymerase chain reaction testing reduces hospitalization and antibiotic use for infants 90 days of age or younger. Pediatrics 2007;120:489-496.
- Lyons TW, McAdam AJ, Cohn KA, et al. Impact of in-hospital enteroviral polymerase chain reaction testing on the clinical management of children with meningitis. J Hosp Med 2012;7:517-520.
- Polage CR, Cohen SH. State-of-the-art microbiologic testing for community-acquired meningitis and encephalitis. J Clin Microbiol 2016;54:1197-1202.
- Leber AL, Everhart K, Balada-Llasat JM, et al. Multicenter evaluation of BioFire FilmArray meningitis/encephalitis panel for detection of bacteria, viruses, and yeast in cerebrospinal fluid specimens. J Clin Microbiol 2016;54:2251-2261.
- Tunkel AR, Glaser CA, Bloch KC, et al. The management of encephalitis: Clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis 2008;47:303-327.
- Talan DA, Hoffman JR, Yoshikawa TT, Overturf GD. Role of empiric parenteral antibiotics prior to lumbar puncture in suspected bacterial meningitis: State of the art. Rev Infect Dis 1988;10:365-376.
- Kanegaye JT, Soliemanzadeh P, Bradley JS. Lumbar puncture in pediatric bacterial meningitis: Defining the time interval for recovery of cerebrospinal fluid pathogens after parenteral antibiotic pretreatment. Pediatrics 2001;108:1169-1174.
- Chartrand SA, Cho CT. Persistent pleocytosis in bacterial meningitis. J Pediatr 1976;88:424-426.
- Connolly KD. Lumbar punctures, meningitis, and persisting pleocytosis. Arch Dis Child 1979;54:792-793.
- Mazor SS, McNulty JE, Roosevelt GE. Interpretation of traumatic lumbar punctures: Who can go home? Pediatrics 2003;111:525-528.
- Lyons TW, Cruz AT, Freedman SB, et al. Interpretation of cerebrospinal fluid white blood cell counts in young infants with a traumatic lumbar puncture. Ann Emerg Med 2017;69:622-631.
- Brouwer MC, Tunkel AR, McKhann GM 2nd, van de Beek D. Brain abscess. N Engl J Med 2014;371:447-456.
- Jain S, Cheng J, Alpern ER, et al. Management of febrile neonates in US pediatric emergency departments. Pediatrics 2014;133:187-195.
- Baker MD, Avner JR, Bell LM. Failure of infant observation scales in detecting serious illness in febrile, 4- to 8-week-old infants. Pediatrics 1990;85:1040-1043.
- Bonadio WA, Hennes H, Smith D, et al. Reliability of observation variables in distinguishing infectious outcome of febrile young infants. Pediatr Infect Dis J 1993;12:111-114.
- Bachur RG, Harper MB. Predictive model for serious bacterial infections among infants younger than 3 months of age. Pediatrics 2001;108:311-316.
- Gómez B, Mintegi S, Benito J, et al. Blood culture and bacteremia predictors in infants less than three months of age with fever without source. Pediatr Infect Dis J 2010;29:43-47.
- Gomez B, Mintegi S, Bressan S, et al. Validation of the “Step-by-Step” approach in the management of young febrile infants. Pediatrics 2016;138: pii: e20154381.
- Turner D, Leibovitz E, Aran A, et al. Acute otitis media in infants younger than two months of age: Microbiology, clinical presentation and therapeutic approach. Pediatr Infect Dis J 2002;21:669-674.
- Jaskiewicz JA, McCarthy CA, Richardson AC, et al. Febrile infants at low risk for serious bacterial infection — an appraisal of the Rochester criteria and implications for management. Febrile Infant Collaborative Study Group. Pediatrics 1994;94:390-396.
- Dagan R, Powell KR, Hall CB, Menegus MA. Identification of infants unlikely to have serious bacterial infection although hospitalized for suspected sepsis. J Pediatr 1985;107:855-860.
- Kimberlin DW, Gutierrez KM. Herpes Simplex Virus Infections. In: Wilson WB, Nizet V, Maldonado YA, et al, eds. Remington and Klein Infectious Diseases of the Fetus and Newborn Infant. 8th ed. Philadelphia, PA: Elsevier; 2019.
- Mallewa M, Wilmshurst JM. Overview of the effect and epidemiology of parasitic central nervous system infections in African children. Semin Pediatr Neurol 2014;21:19-25.
- de Caen AR, Maconochie IK, Aickin R, et al. Part 6: Pediatric Basic Life Support and Pediatric Advanced Life Support: 2015 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science With Treatment Recommendations. Circulation 2015;132(16 Suppl 1):S177-S203.
- McIntyre PB, Berkey CS, King SM, et al. Dexamethasone as adjunctive therapy in bacterial meningitis. A meta-analysis of randomized clinical trials since 1988. JAMA 1997;278:925-931.
- van de Beek D, Farrar JJ, de Gans J, et al. Adjunctive dexamethasone in bacterial meningitis: A meta-analysis of individual patient data. Lancet Neurol 2010;9:254-263.
- Meningococcal Infections. In: Kimberlin DW, Brady MT, Jackson MA, eds. Red Book 2015 Report of the Committee on Infectious Diseases. 30th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2015.
- Lee CK. Drug Dosages. In: Hughes HK, Kahl LK, eds. The Harriet Lane Handbook. 21st ed. Philadelphia, PA: Elsevier; 2018.
The effectiveness of the current vaccine schedule has led to a significant decline in the incidence of bacterial meningitis. Delays in recognition and antibiotic administration result in increased morbidity and mortality; therefore, clinicians must maintain a high degree of vigilance for the subtle findings of meningitis, particularly in infants. Current standards for selective imaging, diagnostic testing, and empiric antibiotics are discussed.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
