Diabetic Retinopathy, Associated Comorbidities, and Treatment Considerations
June 1, 2018
Reprints
AUTHOR
Todd Lang, OD, Diplomate, American Board of Optometry; Diplomate, American College of Lifestyle Medicine, President, Bayside Eye Center, Punta Gorda, FL
PEER REVIEWER
Kim Pfotenhauer, DO, Assistant Professor, Touro University California College of Osteopathic Medicine (TUCCOM), Vallejo, CA
Executive Summary
Diabetic retinopathy is a catastrophic complication of diabetes, with microvascular disease being the number one cause for loss of vision in those 20-74 years of age. Primary care physicians are charged with not only assisting patients in achieving optimal blood sugar control but also documenting that all patients have yearly dilated retinal exams.
- The HEDIS Eye Exam criteria require a retinal or dilated eye exam by an eye care professional (optometrist or ophthalmologist) completed every year OR a negative retinal exam (no evidence of retinopathy) by an eye care professional in the year prior OR bilateral eye enucleation anytime during the patient’s history. Practitioners who are not eye care professionals may indicate a low risk for retinopathy because of a negative retinal exam the year prior by using CPT II code 3072F.
- Primary care physicians also should be responsible for identifying prediabetes in their patients, as 84 million Americans are affected and should be managed actively. Up to 70% of these individuals develop diabetes mellitus with the risk of all the known macrovascular and microvascular complications.
- Tight control of blood sugar has resulted in reduced incidence of maculopathy and takes issue with the recent American College of Physician Guidance Statement #3, suggesting physicians deintensify pharmacologic therapy in patients with type 2 diabetes who achieve HbA1C levels less than 6.5%.
- Attention to fostering lifestyle changes in patients with prediabetes and diabetes takes time and can be frustrating for primary care physicians, but success is rewarding.
Diabetic retinopathy is a complex disorder of the microvascular and neuronal structure of the retina. Although poorly understood, the problem does not start in the retina, but rather begins through systemic metabolic dysfunction, which causes damage to retina vessels. The metabolic dysfunction and subsequent reduced circulation create ischemia and damage to adjacent neurons. This article will review the structure of the retina, the systemic pathophysiology of diabetes, the resulting damage to retinal structure and function, comorbidities, traditional treatments, and prevention and treatment for diabetic retinopathy and its comorbidities.
Anatomy of the Eye
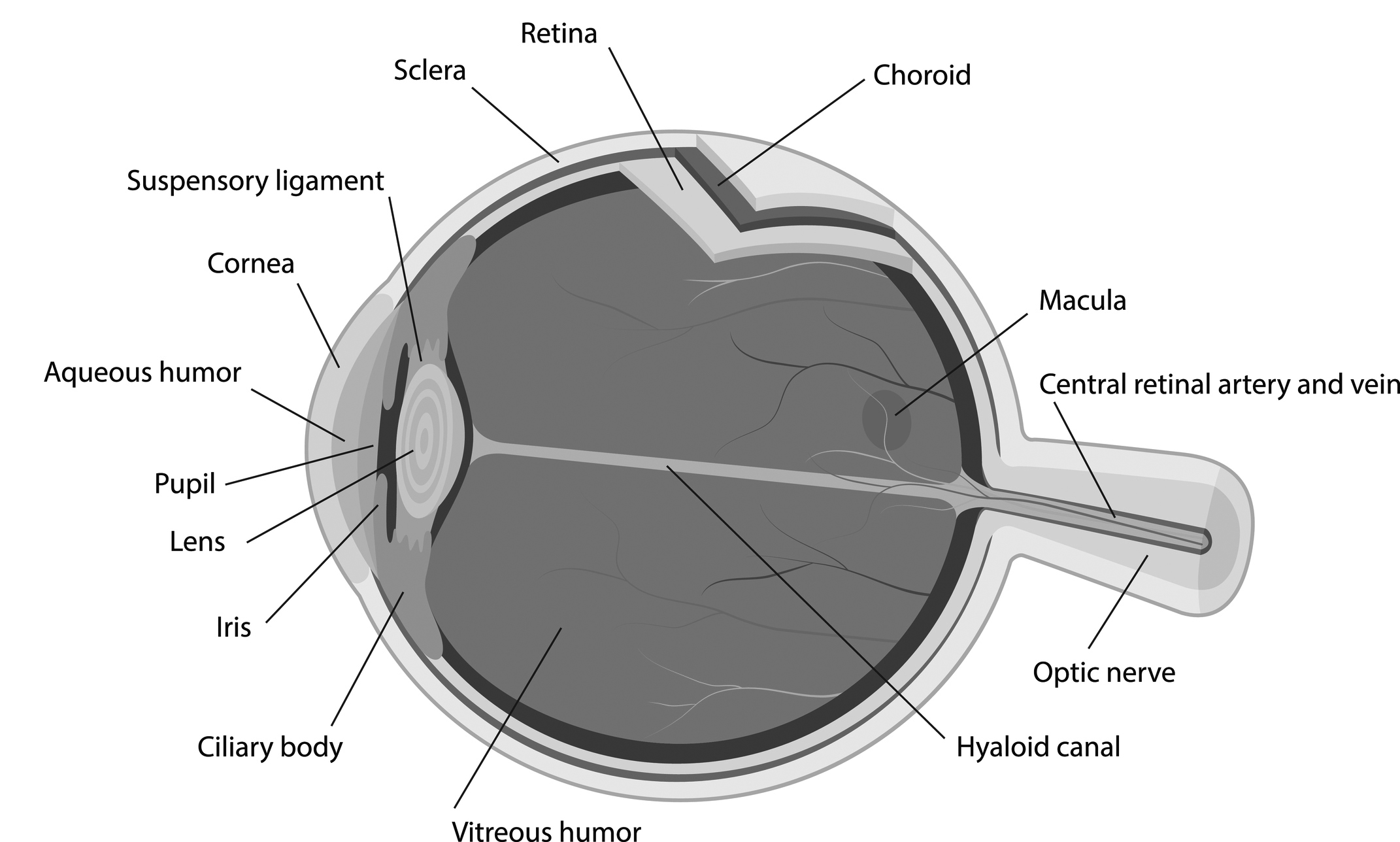
The eye functions much like a camera. There are two lenses of the eye critical to quality vision. Light entering the eye at the cornea refracts and transmits through the anterior chamber, the pupil, and the crystalline lens, through the posterior chamber to the retina with maximum focus at the macula. The photoreceptors, called rods and cones, have their highest density centrally. The cones, known best for their reception of clarity and color, are most concentrated at the fovea centralis, and the rods are most heavily concentrated approximately 10-15 degrees temporal and nasal to the fovea. The rods are most important for receiving and using light in the vision process. The rods and cones cover the entire retina, but their numbers gradually diminish as the focus migrates past the equator to the ora serrata. (See Figure 1.)
Figure 1. Anatomy of the Eye |
 |
|
Source: Getty Images |
The retina is composed of 10 layers from the inside out: 1) the internal limiting membrane; 2) the nerve fiber layer; 3) the ganglion cell layer; 4) the inner plexiform layer; 5) the inner nuclear layer; 6) the outer plexiform layer; 7) the outer nuclear layer; 8) the outer limiting membrane; 9) the inner and outer segments of rods and cones; and 10) the retinal pigment epithelium.
The retinal vasculature involves several interconnected vascular plexuses of vessels and capillaries that feed the various layers of neural structures that accept light. Through chemical transition into electrical impulses, the light transmits through the optic nerve, optic chiasma, lateral geniculate body, and temporal optic radiations to the visual cortex in the occipital lobe of the brain.
Diabetic Retinopathy
Diabetic retinopathy is a known sequela of diabetes. Vascular dysfunction occurs. In the eye, the blood retinal barrier breaks down. This allows cells to leak out of the once tight junctions of the blood vessel walls. In early diabetic retinopathy, researchers believe that pericyte loss is part of the pathophysiology of microaneurysm development and progression. In patients with proliferative diabetic retinopathy, researchers have identified higher levels of inflammatory cytokines in collected vitreous samples. Serum blood samples also have shown higher C-reactive proteins in patients with proliferative diabetic retinopathy as compared to controls.1,2
Essentially, diabetic retinopathy has four levels of severity. In the early stages, patients may not have any symptoms. During the late stages, patients often have symptoms secondary to blood in the posterior chamber, retinal detachment, vitreoretinal traction, or diabetic cystoid macular edema. The stages are identified with clinical findings as indicated in Table 1.
Table 1. Diabetic Retinopathy Stages |
Background Diabetic Retinopathy (BDR)Mild Pre-proliferative Diabetic Retinopathy
|
Moderate Pre-proliferative Diabetic Retinopathy (ModPPDR)Diabetic Retinopathy (MPDR)
|
Severe Pre-proliferative Diabetic Retinopathy (SPPDR)(ModPPDR) +
|
Proliferative Diabetic Retinopathy (PDR)SPPDR+
|
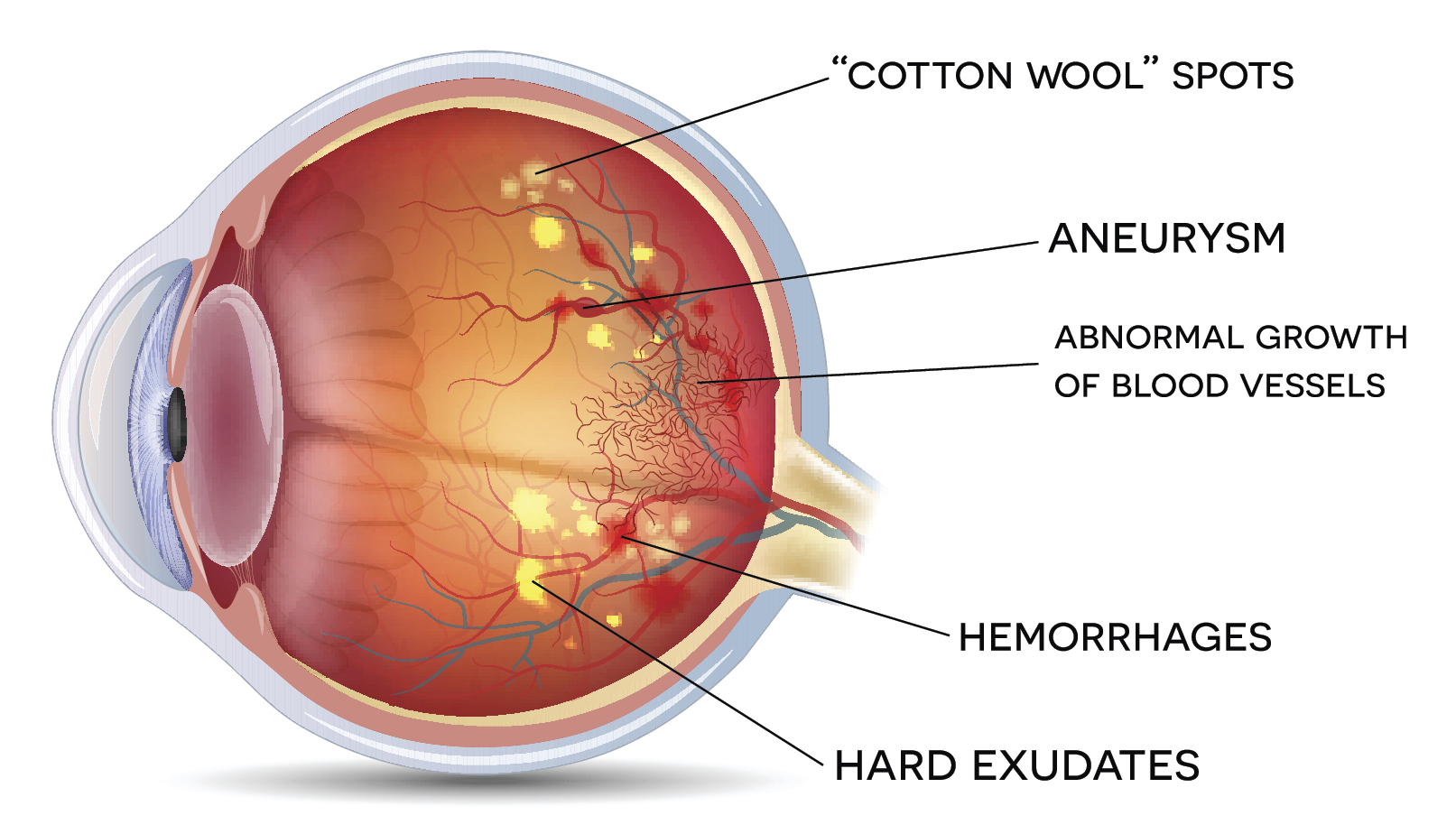
Diabetic retinopathy is the early presentation of blood vessel damage in retinal disease. This is considered a mild non-proliferative or pre-proliferative stage and is identified by one or a few microaneurysms. Some exudates or nerve fiber layer hemorrhages may be present. The moderate pre-proliferative stage is identified when microaneurysms, nerve fiber layer hemorrhages (also called flame-shaped hemorrhages), a few cotton wool spots, and/or exudates are present. Severe pre-proliferative diabetic retinopathy is present when patients have multiple cotton wool spots, all of the moderate pre-proliferative findings, venous beading in two or more quadrants, and/or intra-retinal microvascular abnormalities (IRMA). The most severe diabetic retinopathy is the proliferative stage. Identifying signs of this stage in the clinical exam include vitreous hemorrhage, tractional retinal detachment, pre-retinal hemorrhage, neovascularization at the optic disc (NVD), and neovascularization elsewhere (NVE). Sometimes, neovascularization can be difficult to decipher from IRMA. Fluorescein angiography will help to identify this differential diagnosis, as the blood vessels will leak in neovascularization but will not leak with IRMA. Fluorescein angiography is helpful in finding areas of capillary non-perfusion important in the pathogenesis of diabetic retinopathy and vessel leakage in the macula.1-4 (See Figure 2.)
Figure 2. Diabetic Retinopathy |
 |
|
Source: Getty Images |
Physical Exam Findings
Microaneurysms: Essentially, microaneurysms are outpouchings or ballooning of capillary walls thought to be a result of mural cell loss. Mural cells are part of a neurovascular unit made up of endothelial cells, neurons, and astrocytes. These are found in the superficial vascular plexus of the retina.
Venous Dilation and Beading: Venous dilation and beading is a relatively common finding that presents with varying calibers of the retinal veins. They can appear substantially larger than usual and may appear to have periodic banding that creates the appearance of “beads” along the vessel longitudinally. The veins also can become very circuitous, bending, or tortuous.
Flame-shaped Hemorrhages: These very common retinal signs of local pathology are found in the superficial nerve fiber layer of the retina. They are called flame-shaped hemorrhages because of their appearance.
Dot-blot Hemorrhages: Typically, these are found in some of the inner layers, such as the ganglion cell layer, inner nuclear layer, and the inner plexiform layer.
Cotton Wool Spots: These are areas of ischemia that have swelling or edema of the nerve fiber layer. They appear like an irregular, mildly elevated, white blotch on the retinal surface.
Intra-retinal Microvascular Abnormalities: IRMAs are shunt vessels that form in the most severe form of pre-proliferative diabetic retinopathy where ischemia and vessel damage are most severe but do not leak.
Neovascularization in the Retina: The most ominous finding for a patient with diabetes, NVE occurs when ischemic status has been present for an extended period. These new vessels are small, fragile, and leaky and are presumed to be the body’s response to ischemia.
Neovascularization at the Optic Disc: NVD presents as lacy microvascular proliferation at the optic nerve.
Neovascularization at the Iris: The most obvious identification for neovascularization at the iris is the lacy vessel formation at the pupillary margin. Sometimes, it may be seen only in the anterior chamber angle. If neovascularization is found at either place, this is considered a high-risk sign for neovascular glaucoma.
Vitreous Hemorrhage: Vitreous hemorrhage involves blood that has leaked out of the neovascularization vessels into the vitreous space. The blood cells create scarring and vitreous traction. There is significant risk for secondary retinal detachment to occur.
Diabetic Macular Edema (DME): DME is diagnosed when there is retinal thickening within 2 disc diameters (DD) of the macular center. It does not have to be treated at this stage.
Clinically Significant Macular Edema: This includes hard exudates within 500 microns (one-third DD) or less of the fovea with retinal thickening or 1 DD of thickening within 1 DD of the fovea. The Early Treatment Diabetic Retinopathy Study (ETDRS) set these parameters for grid laser treatment. These guidelines still are considered useful for non-center involving diabetic macular edema. When the fovea is involved (center involving DME), then anti-vascular endothelial growth factor (VEGF) intra-vitreal injections are the treatment of choice.5,6
The Diabetic Eye Exam
When evaluating a patient with diabetes, optometrists, ophthalmologists, and primary care physicians need to know the onset of diabetes and the current treatment regimen. Duration of disease is important for the assessment of the risk for retinopathy. Current research reveals that patients with type 1 diabetes have a higher incidence of retinopathy over time, but it is less prevalent at early stages of the disease.7 Maintaining blood pressure in the normal range is critical for best management of retinopathy. The hemoglobin A1c (HbA1c) is critical to understanding how well the patient’s disease is controlled and is helpful for evaluating the patient’s expected risk for developing diabetic retinopathy and/or the risk for worsening of existent retinopathy. An annual dilated retinal exam is standard protocol for all patients with diabetes. The retina is the best place in the body to see an unobstructed view of blood vessels and the effects of arteriosclerotic and other vascular changes. It is an excellent place to evaluate diabetic complications and to determine the risk for kidney disease.
Primary care physicians should use Snellen acuity charts to check the vision of patients with diabetes at least once a year for screening purposes. In addition, the standard of care is an annual comprehensive eye exam, including biomicroscopic, neuro-opthalmic, and dilated retinal exam, by an optometrist or ophthalmologist.
Traditionally, diabetic retinopathy has been diagnosed with a binocular retinal ophthalmoscope and/or a biomicroscope. It is helpful to use some of the latest technology to evaluate both the anatomy and function of the retina. The spectral domain optical coherence tomographer (OCT) is invaluable for analyzing the topography as well as the various layers of the macula. The resolution of the OCT is measured in microns. Electrodiagnostic testing with the electroretinogram can provide a helpful objective analysis of the retinal function and is useful to monitor disease progression.1 Although the electroretinogram can help provide earlier diagnosis of the disease and monitor disease progression, it is not used widely at this time.
Eye care physicians often will use a wide angle retinal diagnostic laser technology. This can photograph approximately 82% (or 200 degrees) of the retina with one exposure. This device does not replace a careful dilated retinal exam because it does not photograph the inferior retina well, but it augments the exam. Some studies reveal up to a 30% increase in pathologic discovery when the device is used.
Retinopathy
Microvascular disease in the retina is the number one cause for loss of vision in people ages 20 to 74 years. This is not surprising, as the number of patients with prediabetes and diabetes has increased dramatically during the last two decades. Multiple studies reveal that retinopathy presents to physician offices in those with prediabetes 8-13% of time, depending on the research results, locale, and design.8
Researchers have found that patients with hypertension and elevated HbA1c showed a notable higher incidence of retinopathy than patients who were normotensive with normal HbA1c. In a 2017 case study, Zaleska-Zmijewska et al found anatomical changes occurring at the microvascular level in the retinal arterioles.9 They demonstrated that abnormal anatomical changes of the blood vessel walls can occur as a result of hyperglycemia in a patient with prediabetes. This change occurs before conversion to diabetes and before retinopathy is present. The researchers demonstrated through multivariate regression that body mass index (BMI) and total cholesterol are significant correlates to the vessel wall changes.9
In another study, De Clerck et al performed multivariate regression to assess the relationship between the level of glycemia and macular thickness in 1,838 participants (1,087 controls, 279 subjects with prediabetes, and 472 subjects with diabetes without retinopathy).10 Using the macular subfields designed in the ETDRS and the spectral domain OCT to measure macular thickness, they found a substantial reduction in macular thickness in a somewhat linear relationship to hyperglycemia levels. They estimated that 50% of the thickness reduction already was present in participants with prediabetes.10
Yazgan et al compared 53 subjects with prediabetes to 53 age- and sex-matched controls.11 They used the spectral domain OCT and scanned the macula centrally, then at 1 mm, 2 mm, and 3 mm distances from the center in the nasal and temporal perimacular areas. They also scanned and measured choroidal thickness at eight points in the peripapillary area. System and laboratory tests were performed to categorize BMI, fasting blood glucose, and HbA1c. They found a positive correlation of increased macular choroidal thickness associated with increased BMI, fasting blood glucose, and HbA1c. This method needs further investigation,11 as it could be an affordable option for identifying patients at risk for retinopathy. Sakata et al studied the relationship of small vessel circulation in the para-macular area and retinal thickness to best corrected visual acuity in diabetics with macular edema. They concluded that the retinal thickness had the greatest effect on visual acuity.12
Diabetes, the underlying systemic disease causing diabetic retinopathy, is caused by insulin resistance and/or decreased insulin production. As pathogenesis continues, many researchers believe that as hyperglycemia increases, there is oxidative stress, inflammation, an increase in cytokines, and endothelial growth factor that damage the pericytes. The pericytes are part of a neurovascular unit, in that there is a one-to-one ratio between the endothelial cells of the retinal vascular plexuses that perfuse the ganglion cells and the photorecepters necessary for light transmission through the visual pathway to the visual cortex at the posterior occipital lobe.1,4
Diabetic Retinopathy in Pregnancy
Diabetes is present in about 1% of pregnant women. Pregnant patients with diabetes need to be followed closely for retinopathy before and after pregnancy. Researchers have found that if early pre-proliferative retinopathy was diagnosed prior to pregnancy, the patient had a 78% chance that the diabetic retinopathy would worsen. In addition, there was a 23% increase in the risk of proliferative retinopathy. Non-proliferative diabetic retinopathy has a high rate of regression over time after childbirth.13
Prediabetes
Prediabetes is defined as fasting plasma glucose = 100-125 mg/dL, HbA1c = 5.7-6.4%, and oral glucose tolerance = 140-199 mg/dL. Currently, approximately 30.3 million Americans have diabetes, according to the American Diabetes Association (ADA) and the Centers for Disease Control and Prevention.14,15 An estimated 10 million people do not know they have diabetes. The ADA recently reported the cost of diabetes as $327 billion, which includes $237 billion in direct medical treatment costs and $90 billion in lost production. An even larger concern lies ahead: It is estimated that 5-10% of patients with prediabetes will develop diabetes each year.16 This could lead to an additional $100 billion in healthcare costs to an already financially fragile healthcare system within three years. At the current trajectory, the cost of diabetes alone could top more than $1 trillion per year in 15 years.
Obesity
Obesity is epidemic in America, with 35-40% of the population falling into this category (defined by BMI > 30 kg/m2). Obesity is a significant risk factor for diabetes, yet only 11% of the population have diabetes. There is much yet to learn regarding the connection between weight gain, beta cell malfunction, and glycemic absorption at skeletal muscles. Being overweight is one of the top risk factors for having prediabetes and ultimately diabetes. If obese people were tested with HbA1c and glucose tolerance test instead of only a fasting blood glucose, many of those in this weight category would be found to have intermediate glycemia.17,18
Kidney Disease
Kidney disease is a known comorbidity with diabetes, but is less known as it relates to prediabetes. In 2012, Okada et al studied 99,140 people ages 20 to 89 years in Aichi Prefecture, Japan.19 The subjects underwent health checks and were categorized into one of four categories: no prediabetes, stage 1 prediabetes, stage 2 prediabetes, and diabetes. Subjects also were categorized for no prehypertension, stage 1 prehypertension, stage 2 prehypertension, and hypertension. The authors concluded that the prevalence of glomerular hyperfiltration increased with increasing stages of prediabetes and prehypertension, and they recommended that providers monitor kidney function in patients with prediabetes or prehypertension. In subjects with hyperfiltration, they suggested that earlier treatment of hyperglycemia and high blood pressure may help prevent the development of kidney damage.19
In a large study in China using HOMA-IR and logistic regression in 7,801 participants, Gu et al suggested that insulin resistance was a common independent risk factor for both chronic kidney disease and prediabetes.20 They acknowledged that chronic kidney disease and prediabetes are major public health problems in China and that insulin resistance may be a risk factor.
Using data from the 1999-2006 National Health and Nutrition Examination Survey, Plantinga et al reviewed fasting plasma glucose, serum creatinine, and urinary albumin-creatinine ratio among 8,188 participants. They confirmed that kidney disease commonly is associated with diabetes, with 39.6% of patients known to have diabetes and 41.7% of patients who did not know they had diabetes (diagnosed in the study) having chronic kidney disease. Patients with prediabetes had a 17.7% prevalence of kidney disease, and those with no diabetes or prediabetes had a 10.6% prevalence.21
Tapp et al used an oral glucose tolerance test (OGTT) and urinalysis simultaneously with OGTT to determine correlational progression of increased impaired glucose tolerance to increased albuminuria in 11,247 adults. They noted that patients with prediabetes as classified in OGTT had about twice the incidence of albuminuria as those with normal glucose tolerance.22
Cardiovascular Disease
Cardiovascular (macrovascular) disease has several causative factors, including diabetes. Could the more benign prediabetes show evidence of functional decline before damage occurs? Askin et al investigated this question in a study of 80 participants with prediabetes and 80 age-matched controls. Using the myocardial performance index and noninvasive Doppler of global ventricular function, they concluded that prediabetes is a high-risk condition for type 2 diabetes mellitus and that prediabetes is closely associated with subclinical left ventricular systolic and diastolic dysfunction.23
Patscheider et al assessed subclinical changes in the right ventricular volumes and function in controls, patients with prediabetes, and patients with diabetes without a history of cardiovascular disease. The 337 participants underwent 3-T whole-body magnetic resonance imaging using short-axis and four-chamber views. The authors demonstrated a subclinical association of reduced right ventricular volume in men with diabetes and prediabetes. This finding was not evident in women, and subclinical reduced right ventricular ejection fraction was not found in men or women.24
Xing et al studied 2,340 participants in the Dallas Heart Study 2 who did not have cardiovascular disease. They used typical blood tests to identify those with prediabetes, and measured carotid wall thickness, coronary artery calcium measures, estimated glomerular filtration rate, and urinary albumin-creatinine ratio. To remain in the study, the participants could not have diabetes or cardiovascular disease. The results revealed that participants had abnormalities in the presence of prediabetes, which was consistent with expected findings. HbA1c and fasting plasma glucose had loose association with markers for microvascular and macrovascular disease. However, when the multivariable adjustment was applied, the theories did not hold true. Xing et al concluded that characteristics other than HbA1c and fasting plasma glucose in patients with prediabetes have a relationship with the vascular disease process.25
In 2016, Abdul-Ghani et al conducted a literature review to determine if impaired fasting glucose and impaired glucose tolerance test are related to possible prediabetes and diabetes. It appears that suspicion of cardiovascular disease comes with impaired fasting glucose. However, the association or relationship to cardiovascular disease does not occur until there is impaired glucose tolerance. Impaired glucose tolerance and a decrease in beta cell function of the pancreas are considered biomarkers for future cardiovascular disease. They noted that once this is identified, intervention should be initiated to prevent progression to type 2 diabetes and cardiovascular disease.26
Ford et al performed a systematic review to determine if there was a connection between prediabetes and cardiovascular disease. They reviewed 18 papers between 1997 and 2008, and found a modest increase in cardiovascular risk associated with impaired glucose tolerance, impaired fasting glucose, insulin resistance, and free fatty acid metabolism.27,28
Neuropathy
Microvascular disease is a notable concern with intermediate glycemia and hyperglycemia. Although the pathologic process from the signs is not fully understood, they are common and are the result of an aberrant metabolism, most commonly believed to start with obesity and intermediate glucose tolerance and pancreatic beta cell dysfunction. Fat deposition is believed to cause insulin resistance, which is the early evidence of the metabolic process gone awry.
Zeng et al studied the relationships between tumor necrosis factor-alpha (inflammation), interleukin-10 (anti-inflammation), and neuropathy in the distal most sensory nerves of subjects’ toes. The study included 55 patients with prediabetes, 55 patients with diabetes, and 48 controls, and the researchers performed electrophysiology nerve conduction tests of the dorsal/medial plantar sensory nerves. They found a high enough correlation to suggest a prediabetes/diabetes causative factor for the symptomatic neuropathy.29
Rajabally performed a systematic review of 23 papers.30 The auathors of 14 papers hypothesized and investigated the presence of neuropathy in the presence of intermediate glucose tolerance (IGT). The authors of nine papers tested for IGT in the presence of chronic idiopathic axonal polyneuropathy. The findings suggest a probable strong association with IGT, especially for the small fiber painful type.30,31
Nephropathy
Intermediate hyperglycemia or prediabetes often has no symptoms or few symptoms that the general population would consider pertinent or that could put them at risk for developing diabetes. Multiple researchers have identified a statistically significant increase in microalbuminuria in patients with prediabetes. Australian researchers demonstrated twice the incidence of microalbuminuria in patients with prediabetes compared to normal controls.22 Researchers in India found a 19% incidence of microalbuminuria in patients with prediabetes.32
Many researchers have proposed theories regarding the causative factors for kidney disease, ranging from macrovascular disease or coronary artery disease, hypertension, microvascular disease, inflammation, oxidative stress, hyperlipidemia, and genetics. The consistent observation is a correlation between impaired fasting glucose and IGT to elevated HbA1c, showing a relationship to increased microalbuminuria. The definition of microalbuminuria seems to vary.
In the United States and in most studies, the standard definition of microalbuminuria is 30-300 mg/g creatinine. Some researchers have used 22-220 mg/g for men and 31-220 mg/g for women. Regardless of the definition used, the correlation was seen. Bahar et al conducted a literature review and found that BMI and hypertension seem to be unrelated to microalbuminuria, and that there is a high correlation of microalbuminuria in IGT especially.33
Metabolic Syndrome
Metabolic syndrome is increasing at epidemic proportions. Symptoms of metabolic syndrome include a large waist line or abdominal adiposity, low high-density lipoprotein levels, hypertension, high fasting plasma glucose, and high triglycerides in the blood. The presence of three or more of these signs results in a diagnosis of metabolic syndrome. Metabolic syndrome is one of the precursors for common chronic diseases, including obesity, diabetes, prediabetes, hypertension, cardiovascular disease, and stroke. Researchers have worked diligently for years to determine why there has been such a rapid growth of obesity in the United States and other developed countries worldwide. Theories include lack of exercise, increased stress, improper sleep patterns, dietary choices, genetics, sedentary hobbies, and sedentary work environments.
Researchers also have evaluated how food additives may play a role in human dietary behaviors. The most common involves monosodium glutamate (MSG). Multiple researchers are studying whether MSG is healthy. Ironically, most of the studies concluding that MSG is healthy were funded by the food industry.34 MSG has been found to have an obesogenic effect in both rat and human studies.35-38 The majority of processed food in the United States includes MSG hidden within covert names, such as natural flavoring.
Alzheimer’s Disease and Senile Dementia
Neurodegenerative disease generally is associated with genetics and/or aging. Some researchers have called this diabetes of the brain or type 3 diabetes.39 There appears to be an association with insulin resistance, which may contribute to the pathogenesis of Alzheimer’s disease and senile dementia. In a well-designed meta-analysis, Cheng et al found that diabetes is a significant risk factor in Alzheimer’s disease, senile dementia, and mild cognitive decline. This is not surprising, since the brain accounts for roughly 25% of energy resource demand, and insulin receptors and proper function are critical to optimal cognitive function. In addition, most chronic diseases are presumed to be metabolic in nature, and insulin resistance appears to be germane to their pathogenic process.28,40,41
Glaucoma
Glaucoma is a chronic progressive multifactorial disease of the optic nerve causing damage with a resulting loss of vision. Intraocular pressure is important in determining risk for damage or progression. However, 30% of glaucoma patients will have a normal pressure at the time of their eye exam. As a result, clinicians no longer include high intraocular pressure in the definition of glaucoma, but it still is important in treating glaucoma.
People who have diabetes are twice as likely to develop glaucoma as those without diabetes; in fact, some researchers call glaucoma “diabetes of the brain.”42 There are some similarities, in that circulation compromise and neuropathy occur in both.42 Hypertension is a risk factor as well. Atherogenic and arteriosclerotic factors associated with chronic diseases play a role in the group of ocular disorders that culminate in glaucomatous loss of vision. At least 10% of glaucoma patients never have an intraocular pressure above normal (> 21), and at least 20% or more have a normal intraocular pressure at the time of their eye exam but have a pressure above normal at some other time of day. Furthermore, many patients continue to have progression of disease even in the presence of a normal treated intraocular pressure.
Research indicates that eye pressure is highest in the middle of the night while sleeping in the prone or supine position. Some studies indicate as much as a 6 mmHg increase in nocturnal intraocular pressure. Other research has been conducted related to ocular perfusion pressure. Researchers believe low oxygen perfusion pressure may play a role in progression of glaucoma damage in patients who appear to have ocular pressure that is well controlled. In one study, women were affected more than men, and ethnicity was believed to be a factor.43 The results of this study provide further evidence that a vascular mechanism is a causative factor in glaucoma. There are no symptoms until late in pathogenesis of primary open-angle glaucoma, the most common form of the disease. This is another reason it is important for people to get yearly eye exams.
Sometimes, cranial nerve palsy is the first sign of diabetes. Microvascular changes create ischemia to innervation for the extraocular muscles, causing double vision or diplopia. Sixth nerve palsy would present with horizontal diplopia, fourth nerve palsy would present as vertical diplopia, and third nerve could present as vertical or horizontal diplopia but may have pupil involvement or lid drooping (ptosis). Fortunately, when the pathogenesis of diplopia is related to diabetes, it has an excellent prognosis in most cases.
Lajmi et al found that the sixth cranial nerve palsy, which causes horizontal diplopia, showed a 50% incidence. In addition, 75% of the time diabetes was poorly controlled, and there was diabetic retinopathic comorbidity more than 50% of the time in study participants with sixth nerve palsy. Symptoms typically resolved in six to 12 weeks with normalization of the hyperglycemia.44,45
More than 50% of patients with diabetes have dry eyes and should be treated as any other patient with dry eye syndrome.46 Some studies indicate there is reduced tear production in patients with diabetes. Other studies reveal eyelid disease and corneal denervation. Often, patients will be given artificial tears to treat burning or dry eye symptoms.47 This may be appropriate for some patients with diabetes, but providers do not know what they are treating when basing it on symptoms alone. Patients with diabetes have a relatively high prevalence of both evaporative as well as aqueous deficiency dry eye. Patients with diabetes should be examined by an eye doctor under biomicroscope to determine which type of dry eye is present.This will help to achieve a more focused and appropriate treatment.
Treatment for Diabetic Retinopathy and Diabetic Maculopathy
The majority of patients with diabetes do not have retinopathy early in their disease. However, they still should have a comprehensive eye exam with dilation of the retina by an ophthalmologist or an optometrist who is properly equipped with at least a spectral domain OCT. Patients with diabetes who do not have retinopathy or who have mild pre-proliferative diabetic retinopathy should be monitored regularly, with a report sent to their primary care physician. This reporting is important for HEDIS scoring requirements. When moderate pre-proliferative retinopathy is present, patients should be seen at three- to six-month intervals. More aggressive discussion of compliance and optimal management of the disease is imperative to reduce the risk of blindness.
When severe pre-proliferative retinopathy is present with IRMA, patients should see a retina consultant for a fluorescein angiogram to determine if the blood vessels are leaking or if there is capillary non-perfusion. This is necessary to determine whether patients have proliferative disease, as well as what treatment is needed and where laser would be applied, if applicable. Patients have a significant risk for blindness and should be more diligent in managing their systemic disease.
Proliferative retinopathy is diagnosed when neovascularization is present. The neovascularization may be at the optic disc (NVD) or elsewhere (NVE). These are very small, lacy, leaky vessels and are considered pathognomonic in the diagnosis of proliferative stage disease. Additional findings in more severe proliferative retinopathy are vitreous hemorrhage, tractional retinal detachment, and preretinal hemorrhage. Patients who have proliferative stage disease, especially with vitreous hemorrhage and/or retinal detachment, should avoid any heavy lifting in their exercise or daily activities.
The current therapeutic regimen for patients with proliferative retinopathy is pan-retinal photocoagulation. It is believed that the eye has become so ischemic that laser cauterization of the retina will reduce the metabolic demand. Furthermore, it is thought that when the demand for new blood vessels goes away, then so will the existing neovascularization as well as future neovascular growth. This is seen clinically. The neovascularization certainly can return. The return depends on sufficient treatment and how well systemic disease is managed.
Although it would appear that the peripheral vision is reduced substantially, such that someone would be at high risk for losing driving privileges, this turns out not to be true, especially in a binocular patient. The studies show that 80-92% of people undergoing this procedure can expect to still be able to drive. 48,49
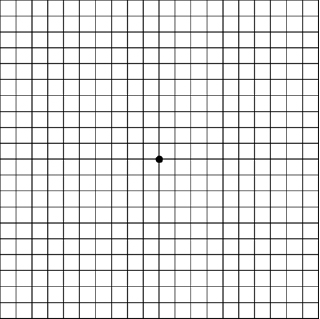
When diabetic maculopathy is present but is not clinically significant, the patient can be monitored with serial macular scans with spectral domain OCT every three to six months. The patient also should monitor at home with an Amsler grid. (See Figure 3.) This device provides a means by which the patient can identify change or distortion. Patients should perform self-monitoring by evaluating one eye at a time while wearing prescription eyewear. If they identify notable distortion or wavy line changes via this means between office visits, they should return to the clinic as soon as possible.
Figure 3. Amsler Grid |
|
The Amsler Grid is a home testing device to monitor for increasing macular disease. |
 |
Clinically significant macular edema is a pathologic development with a statistically increased risk for vision loss and must be treated. Ten to 15 years ago, it was treated primarily with grid argon laser using the ETDRS parameters. Over the last five years, the use of injected anti-VEGF treatments has changed the treatment substantially. Previously, laser and vitreous steroid injections were the only treatments available. Central visual acuity often had to be sacrificed to manage the macular edema with laser. There is about a 27-40% incidence of increased intraocular pressure for up to 100 days following treatment with steroids.
With the introduction of vitreous injected anti-VEGF, retina specialists often use one of three treatment protocols that have a substantially lower risk of intraocular pressure elevations. These treatment protocols involve anti-VEGF injections used monthly, when necessary (PRN), or treat-and-extend. These have been tested in multiple clinical trials, and all protocols demonstrate significant improvements to diabetic retinopathy severity measures. Aderman and Garg concluded the treat-and-extend method can provide both anatomical and visual improvements similar to monthly and PRN methods at less cost to the patient and insurance company.5 Argon grid laser and intravitreal steroid injections still are important for some patients who do not respond to anti-VEGF.5,6,50
Discussion
Diabetic retinopathy is secondary to the metabolic disorder of diabetes. The pathogenesis occurs chronically over many years for the majority of patients with type 1 and type 2 diabetes. Further, the National Institute of Diabetes and Digestive and Kidney Diseases Diabetes Prevention Program showed that lifestyle modification as treatment is almost twice as efficacious as metformin. The results are even better among patients older than 60 years of age. The Diabetes Prevention Program Outcomes Study (DPPOS) revealed that lifestyle treatment compared to metformin reduced diabetes development over 15 years. The researchers also noted there were no overall differences in the microvascular outcomes between the two treatments at the end of 15 years. As expected, those participants who did not develop diabetes had fewer microvascular complications than those who had developed diabetes.51-53
There is some concern among practitioners regarding aggressive treatment of diabetes and early worsening of diabetic retinopathy. The Diabetes Control and Complications Trial (DCCT) demonstrated that retinopathy worsens early. However, the long-term progression of the retinal disease is better with aggressive treatment than moderate treatment.54
A central characteristic of prediabetes and its comorbidities is insulin resistance. Whether the insulin resistance comes from oxidative stress, inflammation, lipid dysfunction, obesity, and/ or a poor diet is not fully understood. Insulin resistance appears to be central to or closely related to early pathologic development of many chronic diseases prominent today. Prediabetes leads to diabetes up to 70% of the time.55,56
A recent American College of Physicians (ACP) guidance on HbA1c treatment levels57 is of some concern as it relates to diabetic retinopathy. The guidance suggests that it is acceptable for most patients with type 2 diabetes to maintain an HbA1c of 7-8%. However, this range does not seem reasonable. In 2009, Cheng et al found an approximately 400% increase in retinopathy prevalence from the prediabetic HbA1c of 5.5% to diabetic HbA1c of 8%,58 which provides credence to tighter control than what is being advocated with the ACP guidance.
The research regarding common comorbidities of prediabetes is important because multi-organ damage is occurring even in the prediabetic stage. In addition, there is a multi-system benefit in lifestyle modification as treatment. A whole food, plant-based diet has been shown to reduce lipids, decrease hypertension, and lower plasma glucose.59-61 In addition, macrovascular disease responds positively to lifestyle modification.62 More longitudinal and observational studies are needed to determine what occurs anatomically and functionally to the neurovascular units in the diabetic retina when patients follow a lifestyle that includes primarily a whole food, plant-based diet and regular exercise.
Conclusion
Diabetic retinopathy, a complication of diabetes, is the leading cause of blindness in people 25 to 74 years of age and is a serious sequela of hyperglycemia. Microvascular and macrovascular changes often begin in the prediabetic phase. Any treatment that reduces the systemic disease of diabetes and its related comorbidities reduces the risk of blindness for patients.
REFERENCES
- Heping X, Curtis T, Stitt A. Pathophysiology and pathogenesis of diabetic retinopathy. Diapedia: The living textbook of diabetes. Available at: https://www.diapedia.org/acute-and-chronic-complications-of-diabetes/7104343513/pathophysiology-of-diabetic-retinopathy. Accessed May 1, 2018.
- Atchison E, Barkmeier A. The role of systemic risk factors in diabetic retinopathy. Curr Ophthalmol Rep 2016;4:84-89.
- Bhavsar AR, Khardoro R. Diabetic Retinopathy. Medscape. Available at: https://bit.ly/2GkwEFP. Accessed May 1, 2018.
- Tarr JM, Kaul K, Chopra M, et al. Pathophysiology of diabetic retinopathy. ISRN Ophthalmol 2013;2013:343560.
- Aderman CM, Garg SJ. Intravitreal anti-VEGF injection treatment algorithms for DME. Retina Today July/August 2017. Available at: http://retinatoday.com/2017/08/intravitreal-anti-vegf-injection-treatment-algorithms-for-dme. Accessed May 1, 2018.
- Blinder KJ, Dugel PU, Chen S, et al. Anti-VEGF treatment of diabetic macular edema in clinical practice: Effectiveness and patterns of use (ECHO Study Report 1). Clin Ophthalmol 2017;11:393-401.
- Romero-Aroca P, Navarro-Gil R, Valls-Mateu A, et al. Difference in incidence of diabetic retinopathy between type 1 and 2 diabetes mellitus: A nine-year follow-up study. Br J Ophthalmol 2017;101:1346-1351.
- Diabetes Prevention Program Research Group. The prevalence of retinopathy in impaired glucose tolerance and recent-onset diabetes in the Diabetes Prevention Program. Diabet Med 2007;24:137-144.
- Zaleska-Zmijewska A, Piatkiewicz P, Smigielska B, et al. Retinal photoreceptors and microvascular changes in prediabetes measured with adaptive optics (rtx1™): A case-control study. J Diabetes Res 2017;2017:4174292.
- De Clerck EEB, Schouten JSAG, Berendschot TTJM, et al. Macular thinning in prediabetes or type 2 diabetes without diabetic retinopathy: The Maastricht Study. Acta Ophthalmol 2018;96:174-182.
- Yazgan S, Arpaci D, Celik HU, et al. Macular choroidal thickness may be the earliest determiner to detect the onset of diabetic retinopathy in patients with prediabetes: A prospective and comparative study. Curr Eye Res 2017;42:1039-1047.
- Sakata K, Funatsu H, Harino S, et al. Relationship of macular microcirculation and retinal thickness with visual acuity in diabetic macular edema. Ophthalmology 2007;114:2061-2069.
- Wykoff CC, Brown DM. The effect of pregnancy on diabetic retinopathy. Retina Today. Jan/Feb 2012. Available at: http://retinatoday.com/2012/02/the-effect-of-pregnancy-on-diabetic-retinopathy/. Accessed May 1, 2018.
- Centers for Disease Control and Prevention. Prediabetes. Available at: https://www.cdc.gov/diabetes/basics/prediabetes.html. Accessed May 1, 2018.
- American Diabetes Association. Economic costs of diabetes in the U.S. in 2017. (March 22, 2018). Available at: http://care.diabetesjournals.org/content/early/2018/03/20/dci18-0007. Accessed May 1, 2018.
- Tabak AG, Herder C, Rathmann W, et al. Prediabetes: A high-risk state for diabetes development. Lancet 2012;379:2279-2290.
- Eckel RH, Kahn SE, Ferrannini E, et al. Obesity and type 2 diabetes: What can be unified and what needs to be individualized? J Clin Endocrinol Metab 2011;96:1654-1663.
- Grundy SM. Pre-diabetes, metabolic syndrome and cardiovascular risk. J Am Coll Cardiol 2012;59:635-643.
- Okada R, Yasuda Y, Tsushita K, et al. Glomerular hyperfiltration in prediabetes and prehypertension. Nephrol Dial Transplant 2012;27:1821-1825.
- Gu DF, Shi YL, Chen YM, et al. Prevalence of chronic kidney disease and prediabetes and association risk factors: A community based screening in Zhuhai, Southern China. Chin Med J (Engl) 2013;126:1213-1219.
- Plantinga LC, Crews D, Coresh J, et al; CDC CKD surveillance Team. Prevalence of chronic kidney disease in US adults with undiagnosed diabetes or prediabetes. Clin J Am Soc Nephrol 2010;5:673-682.
- Tapp RJ, Shaw JE, Zimmet PZ, et al. Albuminuria is evident in the early stages of diabetes onset: Results from the Australian Diabetes, Obesity and Lifestyle Study. (AusDiab) Am J Kidney Dis 2004;44:792-798.
- Askin L, Cetin M, Tasolar H, Akturk E. Left ventricular myocardial performance index in prediabetic patients without coronary artery disease. Echocardiography 2018;35:445-449.
- Patscheider H, Lorbeer R, Auweter S, et al. Subclinical changes in MRI-determined right ventricular volumes and function in subjects with prediabetes and diabetes. Eur Radiol 2018; Feb. 8. doi: 10.1007/s00330-017-5185-1. [Epub ahead of print].
- Xing FY, Neeland IJ, Gore MO, et al. Association of prediabetes by fasting glucose and/or haemoglobin A1c levels with subclinical atherosclerosis and impaired renal function: Observations from the Dallas Heart Study. Diab Vasc Dis Res 2014;11:11-18.
- Abdul-Ghani M, DeFronzo RA, Jayyousi A. Prediabetes and risk of diabetes and association complications: Impaired fasting glucose versus impaired glucose tolerance: Does it matter? Curr Opin Clin Nutr Metab Care 2016; Jul 6. [Epub ahead of print].
- Ford ES, Zhao G, Li C. Pre-Diabetes and the risk for cardiovascular disease: A systematic review of the evidence. J Am Coll Cardiol 2010;55:1310-1317.
- Cheng G, Huang C, Deng H, Wang H. Diabetes as a risk factor for dementia and mild cognitive impairment: A meta-analysis of longitudinal studies. Inter Med J 2012;42:484-491.
- Zeng J, Xu Y, Shi Y, Jiang C. Inflammation role in sensory neuropathy in Chinese patients with diabetes/prediabetes. Clin Neurol Neurosurg 2018;166:136-140.
- Rajabally YA. Neuropathy and impaired glucose tolerance: An updated review of the evidence. Acta Neurol Scand 2011;124:1-8.
- Cortez M, Singleton JR, Smith AG. Glucose intolerance, metabolic syndrome and neuropathy. Handb Clin Neurol 2014;126:109-122.
- Rao BP, Bai GRL, Kennedy GRS, Satyanarayana KVV. Study of the prevalence of microalbuminuria and retinopathy in prediabetes in a tertiary care hospital. J Evid Based Med Hlthcare 2015;2015:608-614.
- Bahar A, Makhlough A, Yousefi A, et al. Correlation between prediabetes conditions and microalbuminuria. Nephrourol Mon 2013;5:741-744.
- Magerowski G, Giacona G, Patriarca L, et al. Neurocognitive effects of umami: Association with eating behavior and food choice. Neuropsychopharmacology 2018; Mar 30. doi: 10.1038/s41386-018-0044-6. [Epub ahead of print].
- Shannon M, Green B, Willars G, et al. The endrocrine disrupting potential of monosodium glutamate (MSG) on secretion of the glucagon-like peptide-1 (GLP-1) gut hormone and GLP-1 receptor interaction. Toxicol Lett 2017;256:97-105.
- Lopez-Miranda V, Soto-Montenegro ML, Urango-Ocio JA, et al. Effects of chronic dietary exposure to monosodium glutamate on feeding behavior, adiposity, gastrointestinal motility, and cardiovascular function in healthy adult rats. Neurogastroenterol Motil 2015;27:1559-1570.
- He K, Du S, Xun P, et al. Consumption of monosodium glutamate in relation to incidence of overweight in Chinese adults: China Health and Nutrition Survey (CHNS). Am J Clin Nutr 2011;93:1328-1336.
- Insawang T, Selmi C, Cha’on U, et al. Monosodium glutamate (MSG) intake is associated with prevalence of metabolic syndrome in a rural Thai population. Nutr Metab 2012;9:50.
- De la Monte SM, Wands JR. Alzheimer's disease is type 3 diabetes — evidence reviewed. J 2008;2:1101-
- Bitra VR, Rapaka D, Akula A. Prediabetes and Alzheimers’s disease. Indian J Pharm Sci 2015;77:511-514.
- Tang J, Pei Y, Zhou G. When aging-onset diabetes is coming across with Alzheimer disease: Comparable pathogenesis and therapy. Exp Gerontol 2013;48:744-750.
- Faiq MA, Dada R. Saluja D, Dada T. Glaucoma — diabetes of the brain: A radical hypothesis about its nature and pathogenesis. Med Hypotheses 2014;82:535-546.
- Zheng Y, Wong TY, Mitchell P, et al. Distribution of ocular perfusion pressure and its relationship with open angle glaucoma: The Singapore Malay Eye study. Invest Ophthalmol Vis Sci 2010;51:3399-3404.
- Lajmi H, Hmaied W, Ben Jalel W, et al. Oculomotor palsy in diabetics. J Fr Ophtalmol 2018;41:45-49.
- Urso DL, Formaro L, Scattarella L, et al. [Sixth cranial nerve palsy associated with diabetes mellitus: A case report]. [Article in Italian]. Recentri Prog Med 2011;102:20-22.
- Manaviat MR, Rashidi M, Afkhami-Ardekani M, Shoja MR. Prevalence of dry eye syndrome and diabetic retinopathy in type 2 diabetic patients. BMC Ophtalmol 2008;8:10.
- Hom MM. Diabetes and dry eye: The forgotten connection. Rev Optometry Available at: https://www.reviewsce.com/. Accessed May 10, 2018.
- Subash M, Comyn O, Samy A, et al. The effect of multisport laser panretinal photocoagulation of retinal sensitivity and driving eligibility in patients with diabetic retinopathy. JAMA Ophthalmol 2016;134:666-672.
- Pearson AR, Tanner V, Keightley SJ, Casswell AG. What effect does laser photocoagulation have on driving visual fields in diabetics? Eye (Lond) 1998;12:64-68.
- Noecker RJ. Managing intraocular pressure elevation following intravitreal steroid injection. Retinal Physician. July 1, 2006. Available at: https://www.retinalphysician.com/issues/2006/july-aug/managing-intraocular-pressure-elevation-following. Accessed May 1, 2018.
- Diabetes Prevention Program Research Group. Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: The Diabetes Prevention Program Outcomes Study. Lancet Diabetes Endocrinol 2015;3:866-875.
- National Institutes of Health. A decade later, lifestyle changes or metformin still lower type 2 diabetes risk. Available at: https://www.nih.gov/news-events/news-releases/decade-later-lifestyle-changes-or-metformin-still-lower-type-2-diabetes-risk. Accessed May 10, 2018.
- Diabetes Prevention Program (DPP) Research Group. The Diabetes Prevention Program (DPP): Description of lifestyle intervention. Diabetes Care 2002;25:2165-2171.
- The Diabetes Control and Complications Trial Research Group. Early worsening of diabetic retinopathy in the Diabetes Control and Complications Trial. Arch Ophthalmol 1998;116:874-886.
- Kandula NR, Moran MR, Tang JW, O’Brien MJ. Preventing Diabetes in primary care: Providers’ persepectives about diagnosing and treating prediabetes. Clin Diabetes 2018;3659-3666.
- StatPearls Publishing. PreDiabetes. Oct. 6, 2017. Available at: https://www.ncbi.nlm.nih.gov/books/NBK459332/. Accessed May 1, 2018.
- Qaseem A, Wilt TJ, Kansagara D, et al. Hemoglobin A1C targets for glycemic control with pharmacologic therapy for nonpregnant adults with type 2 diabetes mellitus: A guidance statement update from the American College of Physicains. Ann Intern Med 2018;168:569-576.
- Cheng YJ, Gregg EW, Geiss LS, et al. Association of A1C and fasting plasma glucose levels with diabetic retinopathy prevalence in the US population: Implications for diabetes diagnostic thresholds. Diabetes Care 2009;32:2027-2032.
- McMacken M, Shah S. A plant-based diet for the prevention and treatment of type 2 diabetes. J Geriatr Cardiol 2017;14:342-354.
- Tonstad S, Butler T, Yan R, Fraser GE. Type of vegetarian diet, body weight, and prevalence of type 2 diabetes. Diabetes Care 2009;32:791-796.
- Lee Y, Park K. Adherence to a vegetarian diet and diabetes risk: A systematic review and meta-analysis of observational studies. Nutrients 2017;9: pii:E603. doi: 10.3390/nu9060603.
- Ornish D, Scherwitz LW, Billings JH, et al. Intensive lifestyle changes for reversal of coronary heart disease. JAMA 1998;280:2001-2007.
Diabetic retinopathy is a complex disorder of the microvascular and neuronal structure of the retina. Although poorly understood, the problem does not start in the retina, but rather begins through systemic metabolic dysfunction, which causes damage to retina vessels. This article will review the structure of the retina, the systemic pathophysiology of diabetes, the resulting damage to retinal structure and function, comorbidities, traditional treatments, and prevention and treatment for diabetic retinopathy and its comorbidities.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
