New Concepts of Ultrasound in the Emergency Department: Focused Cardiac Ultrasound in Cardiac Arrest
June 15, 2018
Reprints
AUTHORS
Michael I. Prats, MD, Assistant Director of Ultrasound, Assistant Professor of Emergency Medicine, The Ohio State University Wexner Medical Center, Columbus
Carolyn L. Martinez, MD, Department of Emergency Medicine, PGY-2, The Ohio State University Wexner Medical Center, Columbus
PEER REVIEWER
Carl Mitchell, MD, RDMS, RMSK, Director, Emergency Ultrasound Division, Clinical Assistant Professor, University of Arizona College of Medicine, Phoenix
EXECUTIVE SUMMARY
- Point-of-care cardiac ultrasound (POCUS) can identify three potentially reversible causes of cardiac arrest in victims with non-shockable rhythms: cardiac tamponade, hypovolemia, and pulmonary embolus.
- During cardiac resuscitation, visualization of organized cardiac activity using POCUS suggests that continuous IV adrenergic agents be used instead of bolus therapy to enhance recovery of spontaneous circulation (ROSC).
- A protocol should be used with transthoracic echocardiography (TTE) to minimize interruptions in chest compression during cardiac resuscitation.
- Transesophageal echocardiography (TEE) has four main advantages over TTE during cardiac resuscitation: a more reliable sonographic window of the heart, the ability to continuously image the heart during resuscitation, elimination of pauses in chest compressions to visualize the heart, and the potential for higher resolution to detect small abnormalities.
Focused cardiac ultrasound has been used by emergency physicians to evaluate patients in shock and cardiac arrest, but, until recently, there has been limited evidence to support its utility. New studies provide data regarding the diagnostic and prognostic values of this modality. Furthermore, transesophageal echocardiography (TEE) performed by the emergency physician presents an intriguing new application with the potential to solve many difficulties encountered in standard transthoracic echocardiography (TTE).
Introduction
During the past 20 years, practitioner-performed ultrasound has become an integral aspect of caring for patients in the emergency department (ED). Real-time use of this imaging modality at the bedside allows practitioners a hands-on approach to the clinical evaluation of patients. Ultrasound adds anatomical, functional, and physiological information that cannot be obtained by traditional exam alone. Ultrasound helps guide resuscitation, determine or manage therapies, and monitor patients from the moment they enter the ED to final disposition. The effect of emergency ultrasound within the United States includes improvement in diagnostic accuracy, reduced time to appropriate treatment, improved safety of procedures, and reduction of costs.1 The American College of Emergency Physicians (ACEP) has outlined core applications for ultrasound in the emergency setting that are highly focused and goal-directed toward answering a select set of questions. Applications now include trauma, pregnancy, cardiac, aorta, thoracic, biliary, urinary tract, deep vein thrombosis (DVT), musculoskeletal, ocular, bowel, and procedural guidance.1,2 Point-of-care ultrasound (POCUS) has particular benefit in the critically ill patient for whom rapid information for decision-making is essential. Given this benefit, there is clear application for POCUS in the patient in cardiac arrest.
In general, indications for echocardiography include the patient with chest pain, dyspnea, hypotension, syncope, and cardiac arrest. With this noninvasive imaging modality, the provider can visualize left ventricular abnormalities, right ventricular strain, pericardial effusion, and occasionally intimal flaps within the ascending aorta. This information facilitates rapid diagnosis of many of the causes of shock, including cardiac failure, acute myocardial ischemia, massive pulmonary embolus, and cardiac tamponade. In the absence of these findings, but with findings of a hyperdynamic heart and collapsible inferior vena cava (IVC) indicating volume responsiveness, the imaging can aid in the diagnosis and treatment of hypovolemic, hemorrhagic, or septic shock. Likewise, many of these uses extend to the patient in cardiac arrest. Here, ultrasound can help guide management by detecting reversible causes of the arrest, visualizing organized cardiac activity, and offering guidance or feedback on the quality of chest compressions.3
Epidemiology
Heart disease continues to rank as the number one cause of death in the United States. In 2016, there were more than 350,000 out-of-hospital cardiac arrests and an additional 209,000 in-hospital arrests. Survival rates range from 12-24.8%.4,5 Along with cardiopulmonary resuscitation (CPR) guided according to the principles of Advanced Cardiac Life Support (ACLS), patients in cardiac arrest also deserve rapid consideration of potential causes for the arrest, specifically those that potentially may be treatable or reversible. Adding ultrasound to this algorithm can give the provider a greatly increased ability to determine etiology and perform life-saving bedside procedures.6
Of the thousands of patients with out-of-hospital cardiac arrests annually in the United States, almost 80% are transported to an ED, but less than 8% survive to discharge. It is well documented that factors associated with survival after a cardiac arrest include presence of a shockable rhythm with early defibrillation and early effective CPR.7 The survival for patients presenting with a shockable rhythm is 40%, while pulseless electrical activity (PEA) holds a much poorer prognosis, with survival rates of 1.3-2.4%.8,9 When a reversible cause is identified in a PEA or asystole arrest, survival rates increase dramatically. Cardiac tamponade with subsequent pericardiocentesis raises survival above other causes of PEA to 15.4% and intervention on massive pulmonary embolism raises it to 6.7%.9 For this reason, management strategies continue to focus on finding, and ideally reversing, the underlying cause of a non-shockable arrest.
Determining and correcting the underlying cause of these arrests is so important that many ultrasound ACLS protocols have been dedicated to the topic and practical application.10,11 The most frequent causes of PEA arrest include hypoxia, hypovolemia, tension pneumothorax, pericardial tamponade, and pulmonary embolism. The etiologies on this list often are reversible, but the required treatments can be invasive and detrimental if applied incorrectly. With the growing use of ultrasound in the ED and increasing numbers of trained providers, ultrasound as a diagnostic tool in cardiac arrest is expanding. Ultrasound can be used alongside the ACLS protocol without interrupting the resuscitation process to provide information otherwise unavailable from the history and and physical exam.12
Reversible Causes With Cardiac Ultrasound
To use ultrasound successfully during a cardiac arrest, emergency physicians first must understand potential etiologies and recognize these findings on echocardiography. These include cardiac tamponade, hypovolemia, and pulmonary embolism. In addition to determining diagnosis, there also has been a recent push to distinguish PEA from pseudo-PEA. In other words, it is important to differentiate true electrical mechanical dissociation from electrical activity with some retained cardiac kinetic motion. By visualizing the presence or absence of myocardial activity, ultrasound also may be of special utility to this population.
Evaluation of cardiac tamponade on bedside ultrasound is a proven, highly accurate, reliable means of diagnosis. Long et al described an overall greater than 96% sensitivity and 98% specificity.12 Gardner et al found cardiac tamponade to cause arrest in 4-15% of patients, with a higher survival rate than among other PEA arrests.8
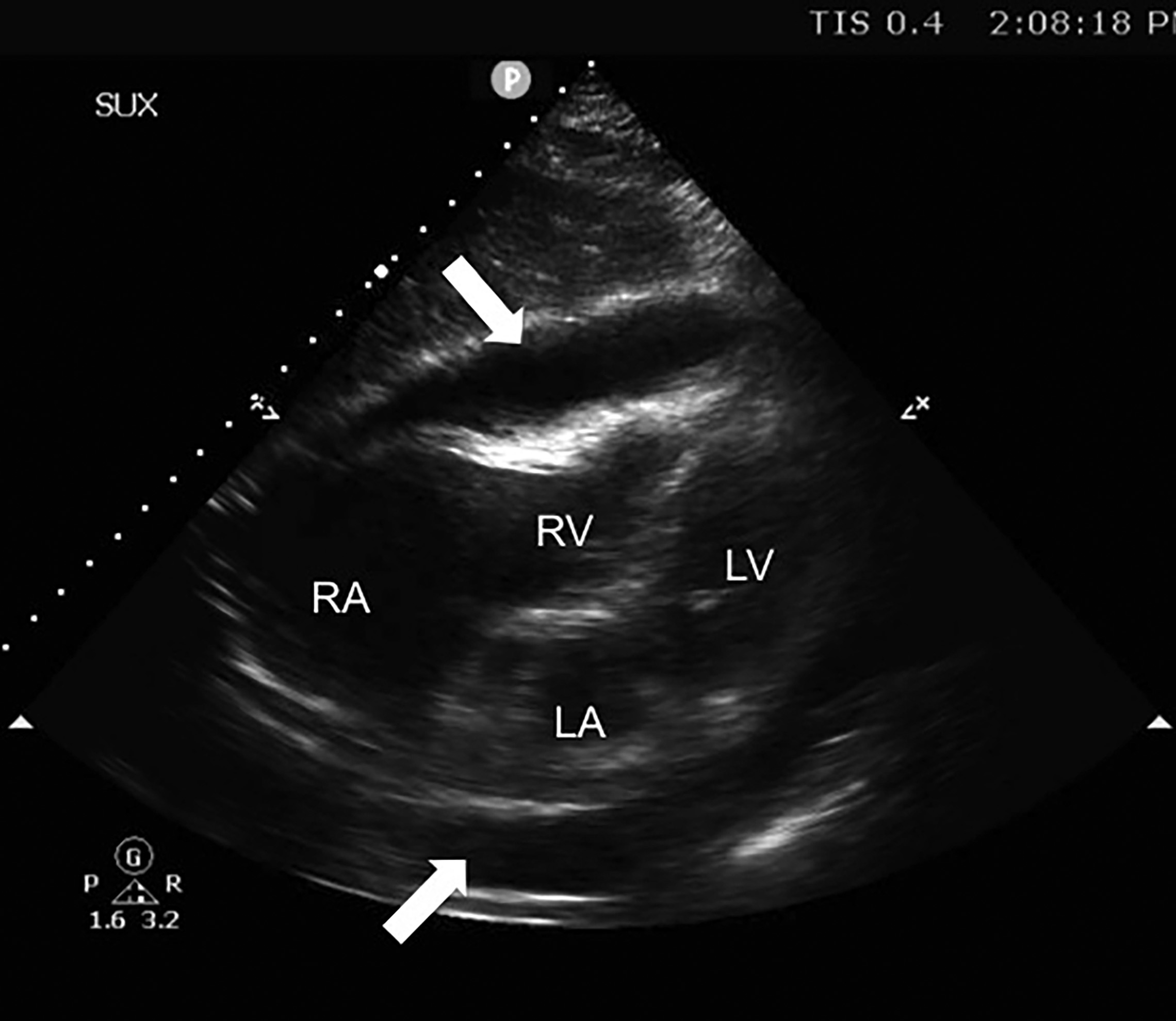
Ultrasound findings of tamponade include pericardial effusion with evidence of hemodynamic compromise. This is seen most commonly on ultrasound as right atrial systolic collapse or right ventricular diastolic collapse. These findings can be seen in Figure 1 and Figure 2.
Figure 1. TTE Image in the Subxiphoid View |
 |
|
The right atrium (RA), right ventricle (RV), left atrium (LA), and left ventricle (LV) are all labeled. The two large arrows indicate a circumferential pericardial effusion. |
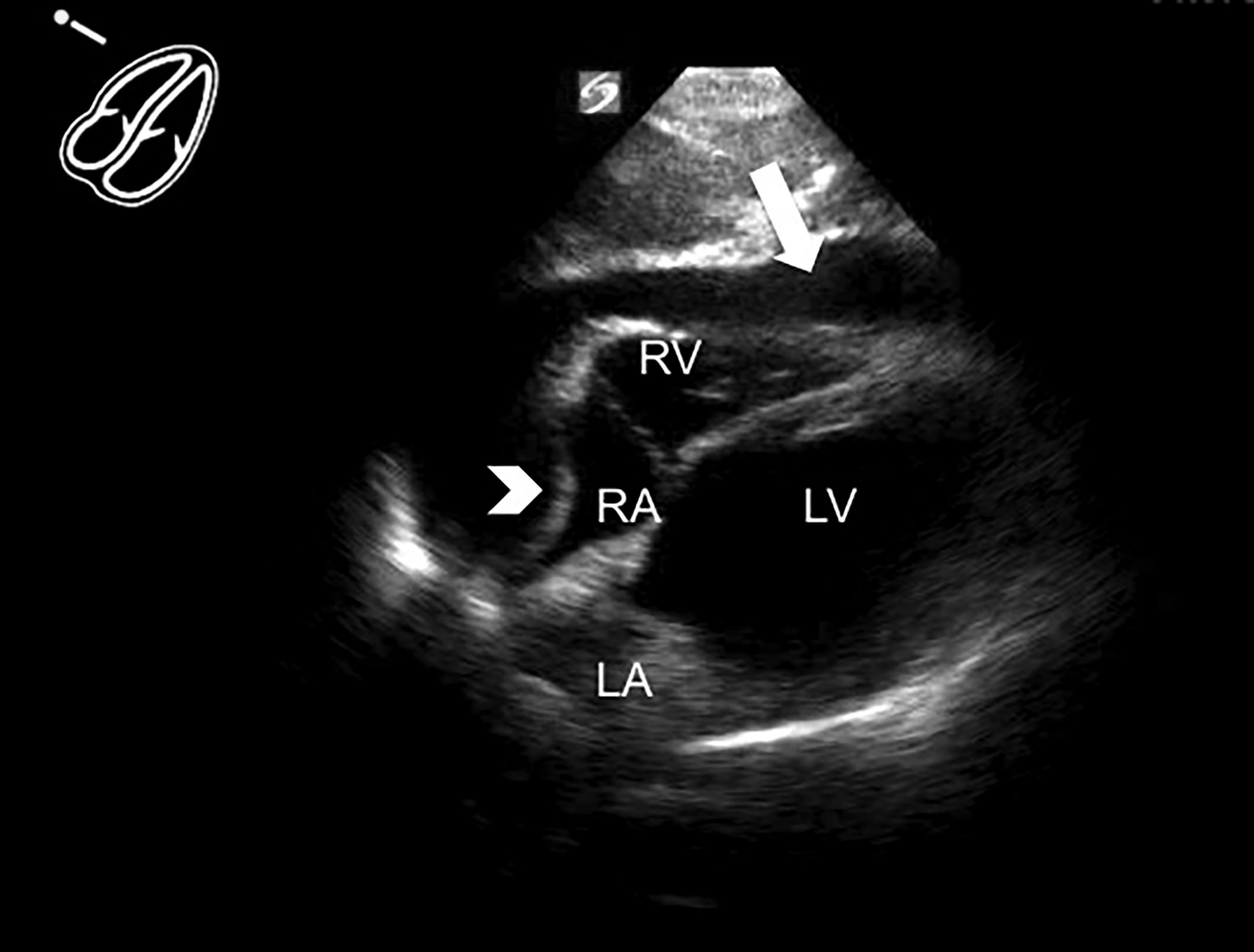
Figure 2. TTE Image in the Subxiphoid View |
 |
|
The right atrium (RA), right ventricle (RV), left atrium (LA), and left ventricle (LV) are all labeled. The large arrow shows a pericardial effusion. The arrowhead indicates bowing of the right atrial wall indicating right atrial collapse and is concerning for pericardial tamponade physiology. |
Ultrasound-guided pericardiocentesis has a success rate of 90% with relatively few complications.12 Providers must be cautious of pleural effusions and epicardial fat pads, as these can be mistaken for pericardial effusions and may result in harm to the patient if unnecessary drainage procedures are performed.10,11
Recognize that cardiac tamponade remains a clinical diagnosis, and the sonographic findings often can precede the hypotension associated with this pathophysiology. In this case, these findings are useful in expediting definitive managment.
Assessment of volume responsiveness with POCUS via IVC collapsibility is slightly controversial and is difficult during a cardiac arrest with positive pressure ventilation, lack of cardiac contractility, ongoing chest compressions, and venous pooling. However, visualization of a small, underfilled left ventricle can be an indication of hypovolemia. Ultrasound images show a hyperdynamic heart with touching of the left ventricular walls causing obliteration of the left ventricular volume during systole. Fluid resuscitation is a common practice in cardiac arrest, but these sonographic findings can lead to the discovery of hypovolemia and, therefore, direct potential treatment of this cause.8,11,12
Estimates indicate pulmonary embolism as a cause of roughly 5% of cardiac arrests, and the detection of a pulmonary embolus as the cause of cardiac arrest offers the potential for survival via treatment with fibrinolytics or extra-corporeal circulatory support. Acute right heart strain refers to right heart failure as a result of a sudden increase in right heart pressures. This diagnosis can be made on POCUS, as the practitioner can visualize right ventricular (RV) dilation and dysfunction. RV dilation typically is visualized in multiple different POCUS views, which can been seen in Figure 3 and Figure 4.
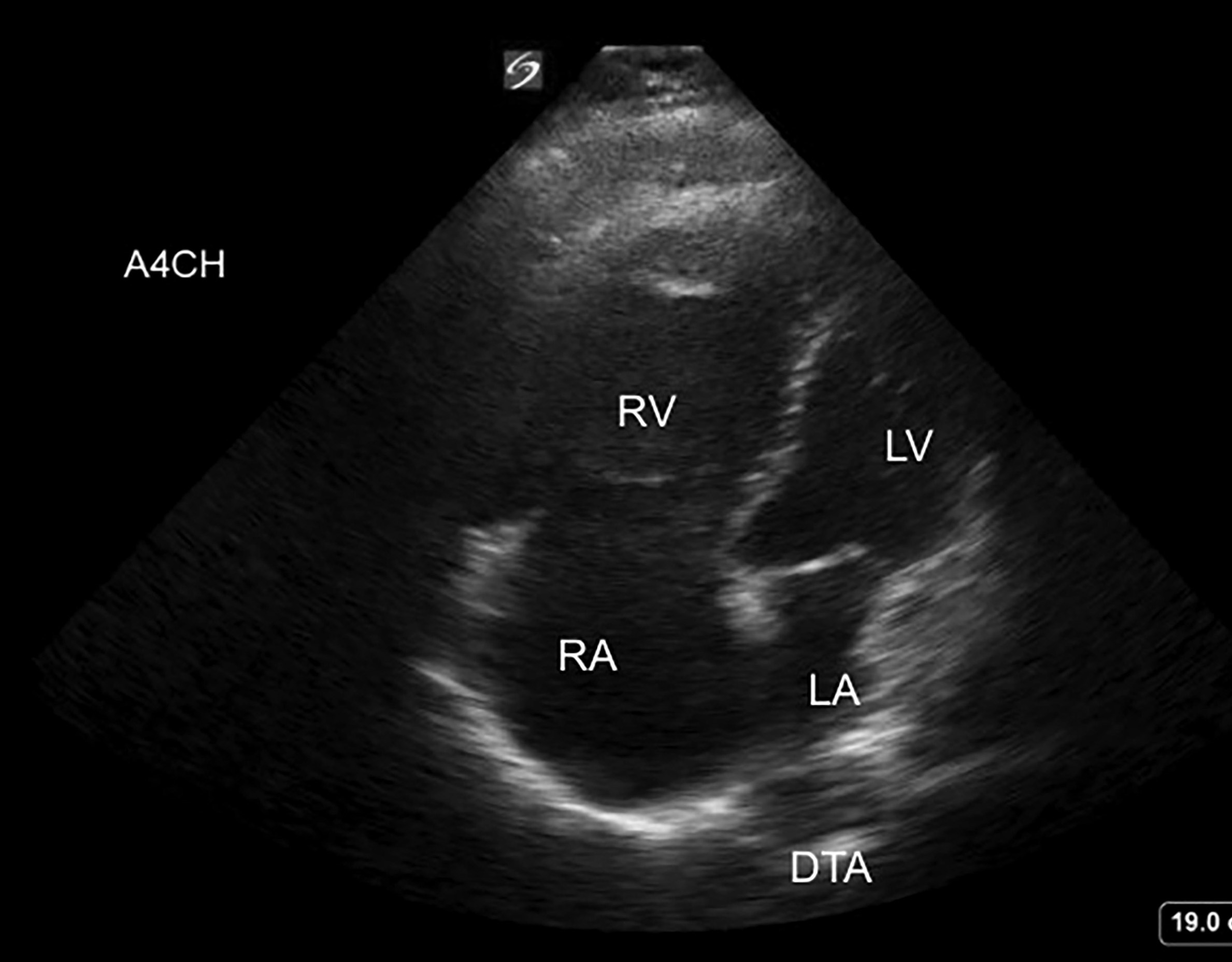
Figure 3. TTE Image in the Apical Four-chamber View |
 |
|
The right atrium (RA), right ventricle (RV), left atrium (LA), and left ventricle (LV) are labeled. The descending thoracic aorta (DTA) is labeled at the bottom of the image as well. In this image, the right ventricle is dilated, assessed via comparison to the left ventricle. The right atrium is dilated as well. These findings are concerning for right heart strain. |
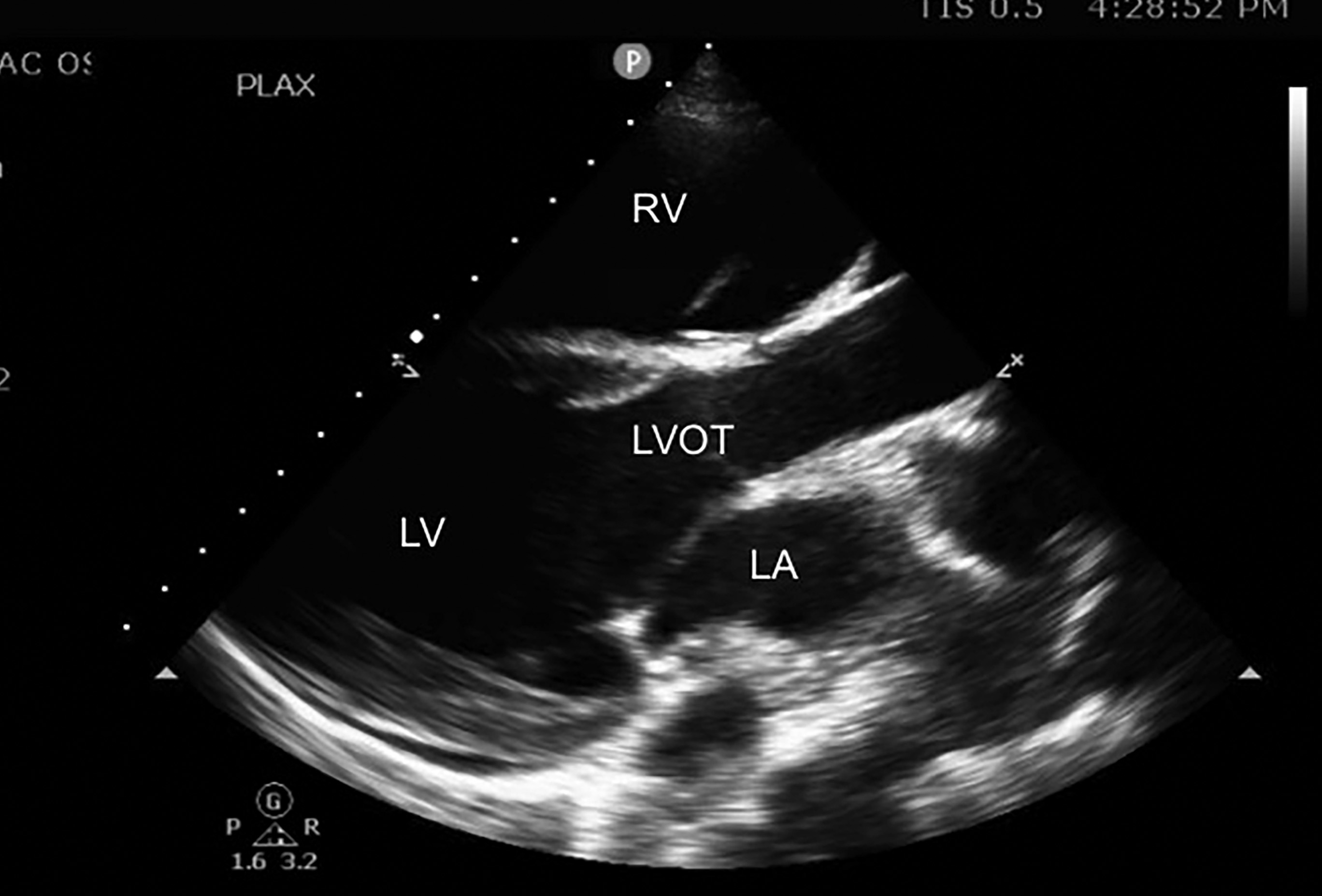
Figure 4. TTE Image in the Parasternal Long Axis View |
 |
|
The right ventricle (RV), left atrium (LA), left ventricle (LV), and left ventricular outflow tract (LVOT) are labeled. The right ventricle is dilated in this image, which can be determined by comparison to the LVOT and LA. In this view, all three should be approximately equal in diameter. |
Severe RV dilation can impose pressure on the left ventricle and cause bowing or flattening of the intraventricular septum; this can be seen as the “D sign” in Figure 5. In the setting of cardiac arrest, this finding often is assumed to be indicative of massive pulmonary embolism.3,8,11 However, there are some risks with this assumption. Chronic causes of pulmonary hypertension can have nearly identical findings on ultrasound. Furthermore, there is some evidence that right heart dilation also is found in untreated ventricular fibrillation and severe hypoxia, two other common etiologies of cardiac arrest. This was observed recently in a porcine model by Aagaard et al, who showed significant RV dilation in all three of the above clinical scenarios.13 While the RV was more dilated from pulmonary embolism, it was very difficult to discriminate between “severely dilated” and “moderately dilated” ventricles.
Figure 5. TTE Image in the Parasternal Short Axis View |
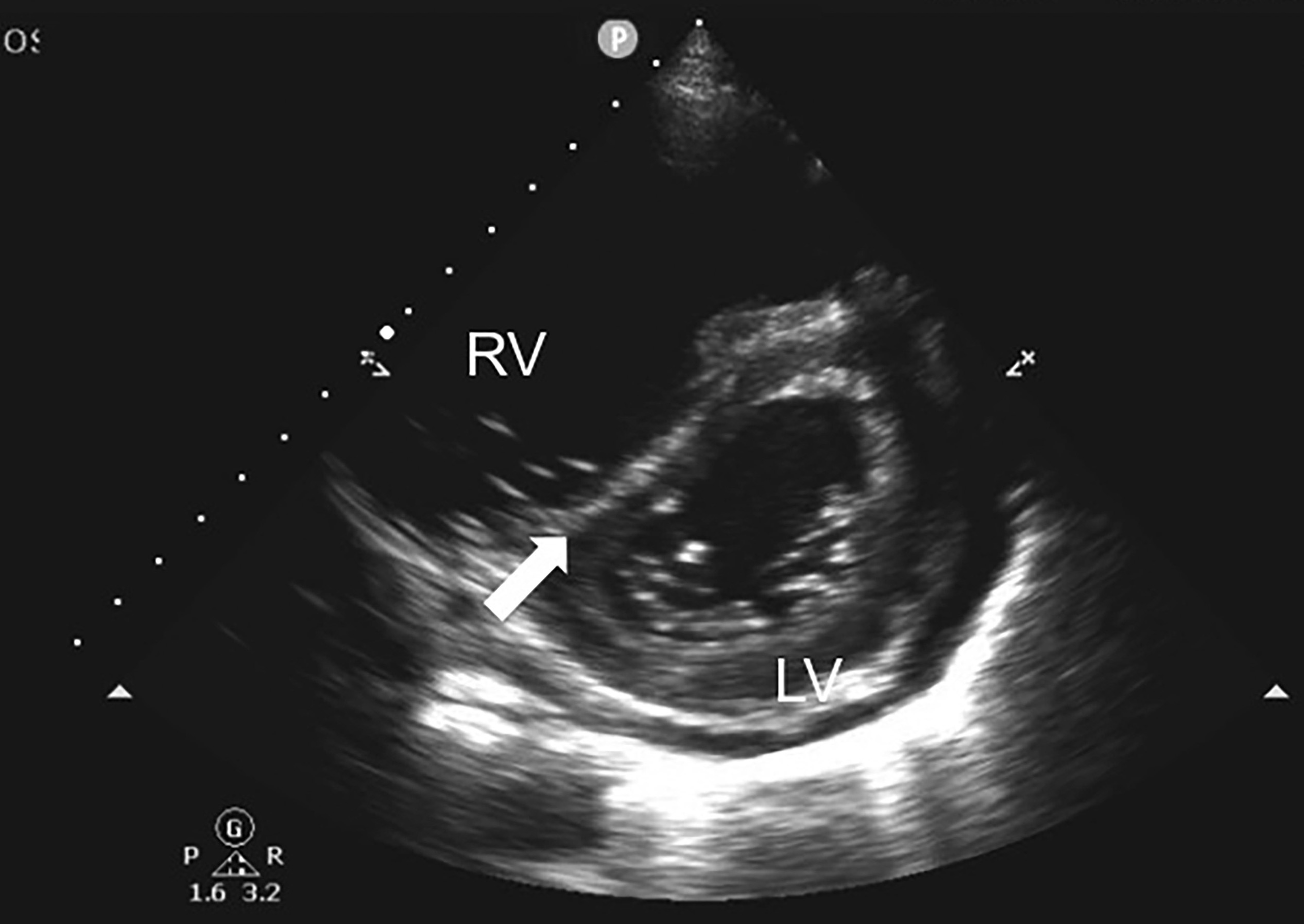 |
|
The right ventricle (RV) and left ventricle (LV) are labeled. The RV is dilated in this image. The large arrow points to the intraventricular septum, which is flattened, causing the left ventricle to lose its normal circular shape. Often this is referred to as the “D sign” to describe the shape of the flattened left ventricle. |
Findings on POCUS may not be enough to definitively diagnose or exclude PE, and images should be interpreted with caution. Until further studies can determine more accurate sonographic criteria, it is reasonable for clinicians to initiate treatment for pulmonary embolus in cardiac arrest patients who have clinical characteristics (initial rhythm is PEA, witnessed arrest, age younger than 65 years, history of venous thromboembolism) and POCUS criteria (RV dilation) suggestive of an acute pulmonary embolus.
Cardiac Standstill
With a video obtained at the initiation of ACLS and ongoing new visual data throughout the resuscitation, the emergency physician receives new information to guide resuscitation. In addition to physical exam and electrical activity, providers now can have information on the myocardial contractions within the heart itself. In general, a binary determination of the presence or absence of meaningful contraction is helpful; however, this can be delineated further. Cardiac activity on ultrasound can be characterized as organized or disorganized. Organized cardiac activity is defined by coordinated contractions of the myocardium resulting in a change in the volume of the ventricle. Disorganized cardiac activity is any other motion of the myocardium not meeting the definition of organized. Cardiac standstill is the absence of any intrinsic contraction of the myocardium.5 (See Figure 6.)
Figure 6. TTE Image in the Subxiphoid View |
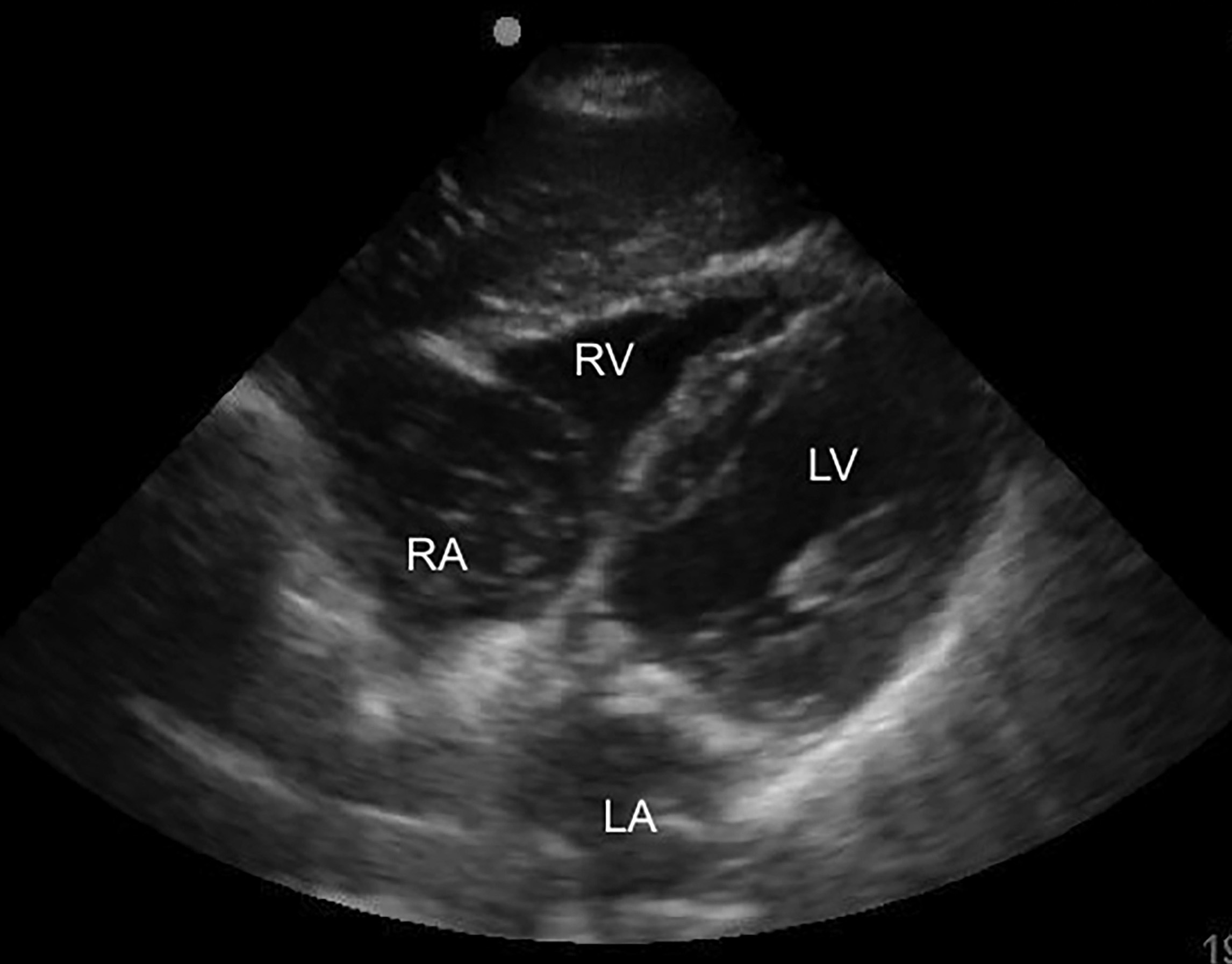 |
|
The right atrium (RA), right ventricle (RV), left atrium (LA), and left ventricle (LV) are labeled. Although better visualized in video form, this image is of a patient in cardiac arrest with complete cardiac standstill. In this image, the right atrium contains heterogenous material indicating blood pooling or stasis. |
PEA arrest warrants special attention with regard to ultrasound findings. PEA indicates electrical activity is present within the heart, but without sufficient contraction to generate a pulse. Many of the aforementioned etiologies of arrest generally cause a PEA arrest. While the lack of pulse indicates cardiac arrest, it does not provide any additional information about possible underlying heart motion. In these patients, ineffective contractions of the heart still may exist. If this is the case, ultrasound potentially can help classify this subset of patients. The next question is if making this determination has benefit in changing the treatment strategy or informing the prognosis. Fortunately, multiple recent studies outline the predictive value of cardiac activity evaluated by ultrasound as an early prognostic indicator in cardiac arrest.14 The following section will briefly review the history of the literature on this topic before examining the most recent evidence.
In 2001, Blaivas et al found that cardiac arrest patients determined to be in complete cardiac standstill on ultrasound did not survive to leave the ED.15 In this early study, all patients arriving with CPR in progress had immediate cardiac ultrasound performed. The researchers found that regardless of the initial electrical rhythm or length of downtime, cardiac standstill had a positive predictive value of 100% for death in the ED. The study brought forth the possibility of using ultrasound as an additional marker for cessation of resuscitative efforts and seemingly was the evidence base for this practice for many years.
In a systematic review performed in 2012, Blyth et al attempted to determine if presence or absence of cardiac activity visualized during echocardiography in cardiac arrest could be extrapolated to predict the likelihood of achieving return of spontaneous circulation (ROSC).16 These data included all initial presenting rhythms and traumatic and atraumatic arrests. Data analysis showed that absence of cardiac activity was a predictor of poor prognosis and may be used to support decisions to terminate resuscitation efforts. The authors concluded that the presence of activity may encourage practitioners to continue more aggressive treatments. Patients with cardiac motion achieved ROSC in 51.6% of cases. On the other hand, of patients who had no motion on echocardiography, 2.4% achieved ROSC. Cardiac activity held a negative likelihood ratio for not obtaining ROSC of 0.18 and, therefore, would be considered a fairly effective test of predicting death. However, the conclusions appropriately state that echocardiography was not reliable as an independent prognostic indicator, and there is insufficient evidence to use it as the sole decision point in continuing or stopping a resuscitation. However, ultrasound does provide further information to assist emergency physicians in making this exceedingly difficult decision with more confidence.
As POCUS became increasingly used alongside ACLS, Gaspari et al conducted a prospective protocol-driven observational study in multiple sites across the United States and Canada to determine whether cardiac activity on ultrasound during ACLS was associated with improved survival.9 This first REASON (Real-time Assessment and Evaluation with Sonography Outcomes Network) study, published in 2016, used POCUS at both the beginning and end of ACLS. The primary outcome of the study was survival to admission, with secondary outcomes of survival to discharge and achieving ROSC. A total of 793 patients were enrolled. Patients were enrolled if they experienced out-of-hospital or ED arrest and had an initial rhythm of PEA or asystole. Cardiac activity was defined as movement of the myocardium or isolated valve movement.
The results showed that if cardiac motion was detected on initial echocardiogram, survival to admission was 28.9%. This number was reduced to 7.2% with no motion, for an odds ratio of 3.6. The secondary outcome of achieving ROSC had similar values, with cardiac motion at 51.0% and no motion 14.3%. Survival to discharge with cardiac activity was 3.8% and without was 0.6%. Subanalysis also was done categorizing patients in PEA and asystole and then by cardiac activity. Patients presented in PEA 52.2% of the time, and 54.4% of these had cardiac activity.9 Asystole was the presenting cardiac rhythm in 47.8% of patients, and only 10.0% of these patients had ultrasound visualized cardiac activity. Cardiac activity on initial ultrasound had the strongest association with survival at every endpoint studied. The study demonstrated a strong association between lack of cardiac activity and non-survival. The subgroup analysis showed that lack of cardiac activity and initial presentation in asystole provided an even stronger association with non-survival. Furthermore, patients who had pericardial effusion identified and underwent pericardiocentesis (n = 13) had a much higher surival rate of 15.4%.
However, although the association exists, the performance of ultrasound in diagnosing survival is less than ideal for such an important determination. In asystole, the ultrasound exam was 90% sensitive and 17% specific for survival to admission. In asystole, it was 53% sensitive. A further criticism of this study is that the authors did not assess for neurologic outcomes of the survivors, a more meaningful outcome than absolute survival. The authors suggest that patients without cardiac activity may be a population in which survival is exceedingly small and additional ACLS interventions may not provide measurable benefit. However, as with the aforementioned systematic review, there were patients with no cardiac activity who survived (three patients, 0.6%).9,12 This emphasizes the importance of using the ultrasound exam in combination with the other information available to assist in decision making.
Gaspari et al performed a secondary analysis on the initial REASON study, with an interest in identifying a subset of patients who might benefit from additional interventions during resuscitation.17 This analysis used the imaging collected in the first study from cardiac arrest patients who presented in PEA arrest with initial echocardiography showing cardiac activity. The researchers analyzed these clips to determine if the cardiac activity was organized or disorganized. Disorganized cardiac activity was defined as “agonal twitching,” and organized was defined as “contractions with changes in ventricular dimensions.” The study further categorized each patient based on which of two treatment strategies they had received: standard ACLS or other interventions beyond ACLS. Standard ACLS treatment included bolus medications that were part of a cardiac arrest algorithm. Non-standard ACLS included any patient who received alternative resuscitation medications, mainly continuous adrenergic agents. As in the initial REASON study, the primary outcome included survival to hospital admission with secondary outcomes of ROSC and survival to discharge.
Patients with organized cardiac activity showed an improved survival rate compared to disorganized activity. In the subgroup with standard ACLS medications and organized cardiac activity, 54.7% achieved ROSC, with 37.7% surviving to hospital admission. Among those with disorganized cardiac activity and standard ACLS medications, 37.2% achieved ROSC, with 17.9% surviving to hospital admission.16 Comparing groups based on medications received showed that continuous IV adrenergic agents in replacement of standard ACLS bolus doses had higher ROSC for those with organized cardiac activity (90.9% compared to 54.7%). The same improvement was not seen consistently at the endpoint in patients with disorganized cardiac activity.
The authors concluded that bedside ultrasound may be able to identify a group of patients that responds differently to interventions, information that fundamentally may change resuscitation of these patients. However, there are several limitations to consider when interpreting these data. The first is that this was a retrospective secondary analysis, allowing for unknown confounding variables. Secondly, there was considerable overlap of the confidence intervals in many of the comparisons made. Lastly, there was no significant difference in survival to hospital discharge, regardless of the groups compared. Just as in the original REASON study, there was also no assessment of neurologic outcome of these patients.
With these limitations in mind, this study is still significant in that it provides some data to suggest a benefit by distinguishing between intact myocardial activity and absent myocardial activity. This is a concept that has been suggested before, as some believe that there are two distinct phenotypes of “pulseless electrical activity” — those who are pulseless because of profound hypotension (also known as “pseudo-PEA”) and those who are pulseless because of the absence of any cardiac activity (true PEA).18,19 The difference in pathophysiology may necessitate different treatment strategies.
Although a previous trial from Tehran showed no difference in outcomes when pseudo-PEA patients were identified by POCUS and treated differently,20 the second REASON trial analysis was much larger.17 Others have shown similar findings that using ultrasound to modify treatment of a patient in PEA can result in improved outcomes.21,22 In a sense, this use is similar to the diagnostic aspect of ultrasound in arrest discussed previously. In both cases, POCUS potentially can identify a subset of cardiac arrest patients who would benefit from a more tailored therapy.
Additional studies have shown similar results of cardiac motion and the absence of cardiac motion providing useful prognostic data for patients in cardiac arrest.5,7,9,14 A meta-analysis of these studies performed by Tsou et al in 2017 showed a combined sensitivity of 90% and specificity of 80% in spontaneous cardiac movement prediction of ROSC.5 These data included all presenting rhythms and potentially reversible causes of arrest and produced a positive likelihood ratio of 4.8 and negative likelihood ratio of 0.06. In patients with a low pretest probability for ROSC, the absence of cardiac motion predicts low likelihood for survival. An additional meta-analysis performed by Wu et al in 2018, focusing on patients with initial rhythm of PEA, outlined the predictive ability of ultrasound to show which patients would achieve ROSC.7 Analysis showed that patients with cardiac activity were more likely to obtain ROSC than those with cardiac standstill with a relative risk of 4.35.7 In these studies, the authors concluded that POCUS may be effective, but not definitive, for predicting ROSC and it may be used as a tool to assist physicians in the decision to terminate resuscitation.
The results of this collection of research must be viewed with potential weaknesses in mind. There are very little data within these studies that focuse directly on long-term patient outcomes. Most studies focus on a primary outcome of obtaining ROSC, but do not continue to measure neurological outcomes or long-term survival. Long-term morbidity and mortality is complicated and compounded by several factors, including comorbid conditions, medications, treatments, and complications, and, therefore, is difficult to predict based on initial echocardiographic findings.16
There is also a large discrepancy among the studies of calculated sensitivity and specificity for ROSC. This likely is due to multiple factors: inclusion of traumatic and nontraumatic arrest, inclusion of different presenting rhythms, stringency in criteria of detected cardiac movement, and training of ultrasonographers. Even considering these weaknesses, there now is a substantial body of literature on the topic of POCUS in cardiac arrest. These trials are the best evidence to date on the topic. Overall, these studies offer evidence that supports the use of POCUS in cardiac arrest as both a diagnostic and prognostic tool.
Feasibility During ACLS and Issues With Transthoracic Echocardiography
The 2015 American Heart Association (AHA) ACLS update included recommendations for the use of ultrasound as an adjunct to CPR. The report states that ultrasound may be considered during the management of cardiac arrest assuming a qualified sonographer is present. Use of ultrasound comes with the express understanding that it should not interfere with the standard treatment protocol.23,24 Performing POCUS successfully during cardiac arrest can be challenging. Patient factors, such as body habitus, inflated stomach from bag-valve-mask ventilation, emphysema, and subcutaneous air, complicate image acquisition. In addition to these factors, ongoing CPR poses a great challenge to collecting useful ultrasound images. Because of the latter, many emergency providers limit POCUS evaluation to occur during the expected pulse checks.25
To maximize resuscitative efforts, ultrasound should be integrated seamlessly into ACLS algorithms to avoid interruption of chest compressions. A major factor affecting survival during arrests is maintenance of adequate arterial pressure and, therefore, perfusion to coronary arteries. This allows for oxygenation and clearance of metabolic waste, increasing the likelihood of obtaining ROSC. Perfusion builds gradually during the first few compressions and drops substantially anytime compressions are interrupted.26 The AHA guidelines specifically include concern for pulse checks taking an extended period and instruct healthcare providers to take no more than 10 seconds.24 Rhythm checks have the propensity to become excessive solely because of the difficulty in determining if a pulse is present. For this reason, pulse checks are limited to minimize the pause in compressions and the default is to resume if any doubt is present.26 With the initial introduction of ultrasound into ACLS, there was concern for an unintentional significant extension of the pulse check time.
The practical application of integrating ultrasound into the cardiac arrest algorithm presents a challenge. The use of POCUS should conform to the structure of ACLS without causing undue disruption. The initial concept and basic protocol for POCUS in arrest was well outlined by Breitkreutz et al in 2007.11 The protocol is structured for execution simultaneously with ACLS, fitting into the already planned CPR cycles, reducing unwanted increase in interruptions. Preparation for ultrasound image acquisition happens in parallel to CPR, ensuring the ultrasound is prepared and tested, and the probe is loaded with gel. Image acquisition is started while compressions are ongoing, attempting to find a good acoustic window in the subxiphoid view. During the subsequent five-second pause, a video is collected, with initial “eyeball” recommendations provided to the code leader. Additional evaluation of the saved clip is performed while CPR is resumed and ACLS continued. If additional findings are clinically significant, they can be relayed to the code leader. There are other protocols described in the literature with the same underlying goal and overarching idea, to obtain a useful image within a five- to 10-second pulse check and limit the time without compressions. These include the recent CAUSE (Cardiac Arrest Ultrasound Exam), CASA (Cardiac Arrest Sonographic Assessment), and SHoC (Sonography in Hypotension and Cardiac Arrest) presented at the International Federation of Emergency Medicine.8,10,25,26,27
Even with a clearly outlined and practiced protocol, the risk of expanding pauses within CPR is high and is described in the literature. In a prospective trial, Huis et al measured the length of time for 123 pulse checks over the course of 23 cardiac arrests.28 The researchers found the mean duration of pulse checks and interruption in chest compressions when POCUS was performed was 21 seconds, an eight second increase from the duration measured without ultrasound of 13 seconds. A similar result was obtained by Clattenburg et al, also via a prospective trial, in which a five-second difference was found between pauses with and without POCUS.29 CPR pauses when POCUS was involved lasted an average of 19.3 seconds and without POCUS an average of 14.2 seconds. However, the authors also reported a trend toward shorter pauses when ultrasound fellowship-trained providers were present and performing image acquisition. These initial trials raise a significant amount of concern for potential harm from extended time without compressions. However, the previously discussed REASON study, which included 793 patients with POCUS exams at 20 different sites, measured an average time recording ultrasound images during pulse checks of 4.9 seconds, which was not statistically significant from the 4.4 seconds measured without POCUS.9
There is no denying that a delay in chest compressions is a potential consequence of performing ultrasound during cardiac arrest. This should be acknowledged and efforts should be made to mitigate this risk. However, when performed correctly as described in the protocol above, most experts agree that it is feasible to perform this examination without significant delays.
An additional concern for integrating ultrasound into ACLS and cardiac arrest management lies not just in the implementation of POCUS but in the interpretation of images. With such focus on cardiac activity in the setting of PEA, the definitions of “cardiac motion” and “cardiac standstill” become very important.17 As increasing data show prognostic value in these exam findings, the importance of correct classification only will increase. As discussed previously, cardiac standstill is used to guide termination or continuation of resuscitative efforts and even is being used in the prehospital setting to assist with prehospital efforts.30 This makes it critically important to reduce variability in interpretation, create precise definitions, and to provide as objective a measure of prognosis as possible. With these concerns in mind, Hu et al created a survey-based study to determine the degree of variability in the definition of cardiac standstill among physicians who perform POCUS.31 Among the 127 physician sonographers surveyed, only moderate inter-rater agreement was appreciated, highlighting concerning disagreement in the interpretation of clips of cardiac arrest echocardiography. Clips with the most variability included valve flutter, movement from mechanical ventilation, weak myocardial contraction, and profound bradycardia.31 Decreasing this variability of interpretation is key prior to obtaining confidence in ultrasound as a prognostic indicator.
Transesophageal Echocardiography
The studies discussed thus far have used TTE exclusively. This body of evidence demonstrates that TTE during cardiac arrest has several benefits; however, there are many potential impediments to performing this exam. As mentioned, performing transthoracic ultrasound often is challenging based on patient body habitus, air in the stomach, defibrillator pads on the chest, thoracic trauma, or ongoing chest compressions. Unlike when using TEE on an awake patient, the patient in cardiac arrest is unable to assist with positioning or breathing changes to optimize images. Broken ribs that occurred prior to resuscitation or because of chest compressions can lead to subcutaneous emphysema impeding transthoracic views of the heart. Although it is possible, maintaining a cardiac view during ongoing compressions is difficult, limiting sonographic information to the periods without chest compressions. When this is the case, there has been a risk of ultrasound leading to delays in chest compressions.28,29
TEE offers a solution to many of these problems. Performed for several decades by cardiologists and anesthesiologists, TEE obtains high-quality images of various cardiac diseases and is used for perioperative management.32-35 This experience has noted TEE can be a useful tool for assessing critically ill and arresting patients.36 Although these examinations are performed occasionally in the ED, only recently has the idea of a focused TEE exam performed by an emergency physician been described. TEE in cardiac arrest in the ED was reported first in 2008.37
TEE offers multiple potential benefits over TTE. First, by providing a more reliable sonographic window of the heart, TEE is feasible in cases in which TTE is difficult or impossible during the resuscitation. For most images, the TEE probe sits in the esophagus directly posterior to the left atrium. This allows for consistent sonographic windows regardless of a patient’s body habitus or body mass index. Furthermore, the TEE transducer can be left in place without requiring constant manipulation, allowing for continuous monitoring during resuscitation. This includes constant imaging during ongoing chest compressions or other interventions and the ability to monitor response. Use of TEE potentially can minimize pauses during cardiac rhythm and pulse checks when compared to TTE. In addition, the range of the TEE transducer frequency often extends to higher frequencies, providing a potential for higher resolution images.
With TEE, the provider can diagnose all the pathologies evaluated by TTE, often with an added degree of accuracy.34,35 Use of TEE can identify aortic dissection (see Figure 7), cardiac tamponade (see Figure 8), myocardial dysfunction, arrhythmias, and right heart failure suggestive of a pulmonary embolism during cardiac arrest and alter the acute management.38 Compared to TTE, TEE more readily demonstrates valvular pathology and pathology of the aortic arch. Given that these conditions can lead to cardiac arrest, diagnosis can aid in management during resuscitation.
Figure 7. TEE Image in the Midesophageal Long Axis View |
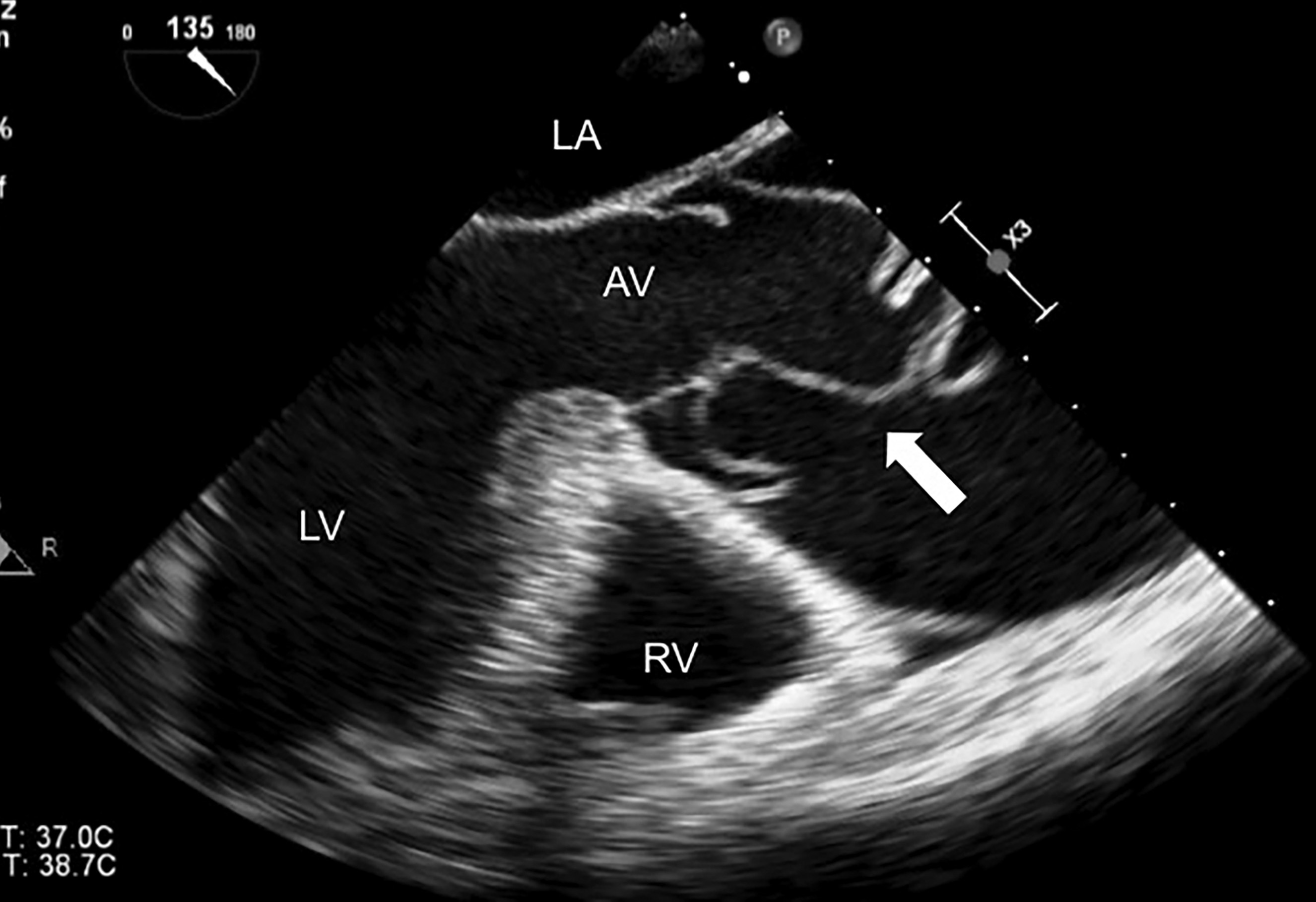 |
|
The right ventricle (RV), left atrium (LA), left ventricle (LV), and aortic valve (AV) are labeled. The ascending aorta is severely dilated and a dissection flap can be seen, indicated by the large arrow. |
Figure 8. TEE Image in the Transgastric Axis View |
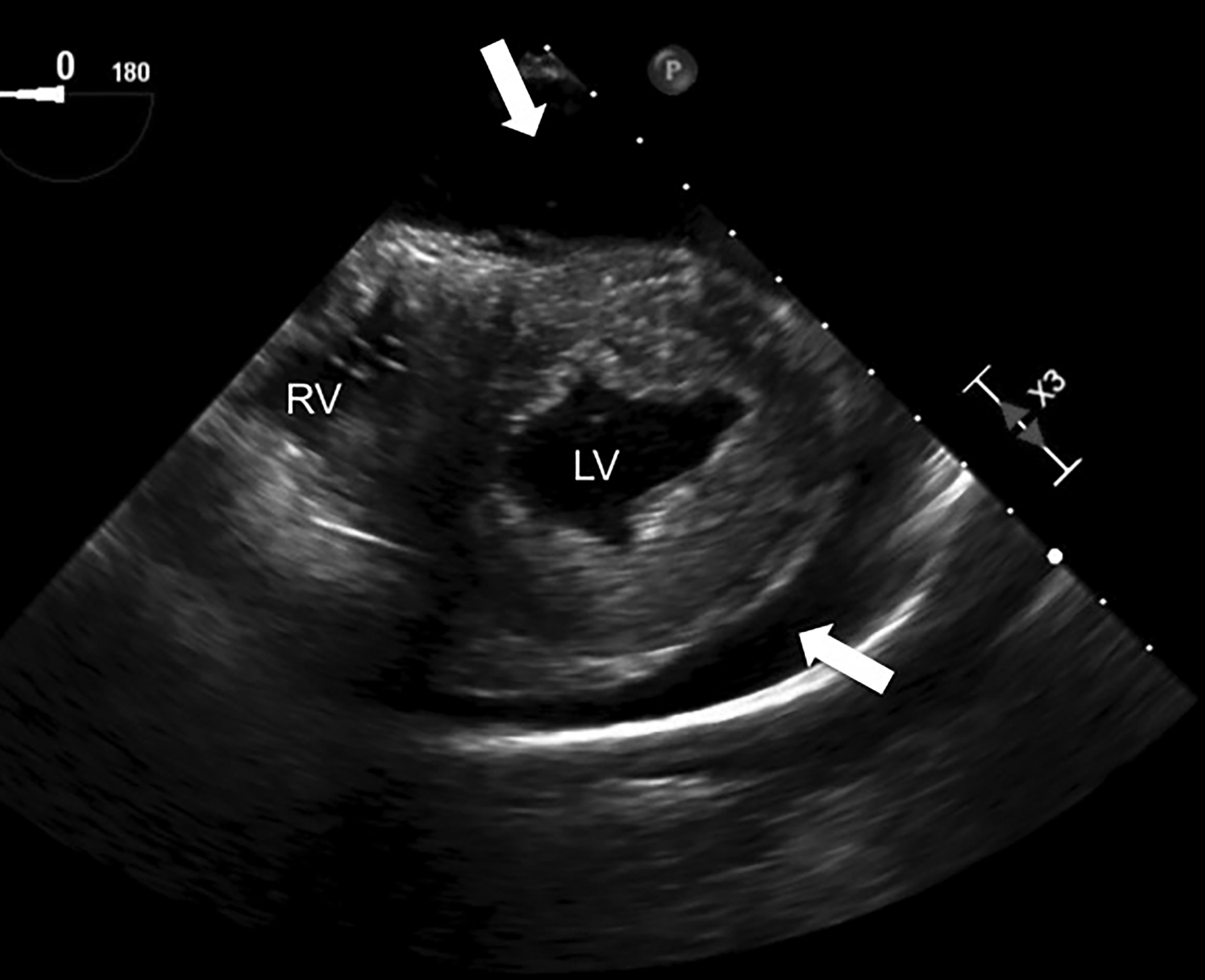 |
|
The right ventricle (RV) and left ventricle (LV) are labeled. A large circumferential pericardial effusion can be seen, indicated by both large arrows. |
Furthermore, TEE can provide a constant evaluation of the efficacy of ongoing chest compressions. This is important because there is evidence that landmark-based standard hand position during chest compressions mistakenly can compress the left ventricular outflow tract, potentially decreasing perfusion.39 Porcine models have shown that this can lead to reduced return of spontaneous circulation.40 TEE provides for real-time guidance of chest compression, allowing for adjustment of hand position to optimize the compression of the left ventricle. Lastly, TEE also can be beneficial for procedural guidance, such as placing a transvenous pacemaker or extracorporeal life support cannulation.41
TEE Training and Feasibility
It is clear, then, that focused TEE can offer many advantages when performed in the ED, especially in the setting of cardiac arrest. From there, it must be established that this practice is feasible, both from a training and from a logistical standpoint. The first study showing that emergency physicians could be trained effectively in this modality was published by Arntfield et al in 2015.42 These authors described a simulation-based training workshop at a single center wherein emergency physicians and residents participated in a four-hour didactic and hands-on course. From the many views that are obtained in comprehensive TEE, this article introduced and taught four views that are considered to represent a focused TEE exam. These views are the midesophageal four chamber, midesophageal long axis, transgastric short axis, and bicaval views. These views are discussed further in a subsequent section. This study showed that after the brief training intervention, 85.7% of the participants felt confident in their ability to use TEE and 100% were able to obtain an acceptable midesophageal four-chamber view both directly post-workshop and at six weeks afterward.42 All participants also were able to identify pathology correctly based on saved TEE video clips. In addition, by the six-week retention examination, 64.3% had begun using TEE in clinical practice.
In a more recent study, 40 emergency medicine residents were trained in a two-view focused protocol.43 Participants were tested with a simulated cardiac arrest scenario in which they performed TEE on a simulator and were asked to identify pathology including asystole, cardiac tamponade, ventricular fibrillation, and acute myocardial infarction. The results showed 100% success in obtaining the focused views on a simulator. There was a 98% sensitivity and 99% specificity for diagnosing pathology based on images obtained.43
The main criticism is that both studies used a TEE simulator, and therefore the results may not correlate directly with performance of TEE in an actual live patient in cardiac arrest in the ED. However, these studies do demonstrate that training is feasible and can be effective, especially for those with prior experience with TTE.
Regarding the feasibility in clinical practice, Arntfield et al reported a review of the TEE examinations performed in the ED over a two-year period.38 This study represents the best data to date regarding TEE use in the ED. In this retrospective review, the authors analyzed the TEE exams performed at a single center, assessing the indications, feasibility, safety, and clinical impact. Of the 54 exams performed, all of the patients who received TEE had an endotracheal tube in place. The majority of the studies were performed for cardiac arrest or post-arrest management (69%). The remainder were performed for hypotension (both medical and traumatic) and in one case, to assess for an aortic dissection. There was 100% success with probe insertion, 83% on the first attempt.38 In all but a single patient, the TEE exam produced interpretable images. There were no documented cases of esophageal perforation or other complication from the TEE examinations in the population that survived to hospital admission.
Most impressive were the findings on clinical effect. After performance of each TEE examination, physicians prospectively recorded their actions based on the TEE results. TEE was reported to be diagnostically helpful in 76% of the cases. The most common use was excluding a cardiac etiology for the cardiac arrest (43%). Other diagnoses made by these examinations were depressed left ventricular function, hypovolemia, aortic dissection, and determination of the cardiac rhythm. Therapeutically, the TEE was influential in 67% of the cases. In this case, 42% of the time it led to changing cardiac compressions, either the placement, depth, duration, or identifying return of cardiac activity. TEE also was used to help determine futility in resuscitation, guide hemodynamic support, and provide procedural guidance. An analysis was performed to estimate what proportion of these findings may have been possible to discern using only TTE (assuming there were adequate windows for imaging), and the authors determined that 55.6% of the exams had a finding that was identifiable with a TEE.38
TEE Guidelines and Protocol
Most recently, ED-focused TEE guidelines have been developed and endorsed by ACEP that describe the use of TEE in the ED for patients in cardiac arrest.44,45 These guidelines include recommendations for scope of practice, training and credentialing, and equipment maintenance. The sole indication for ED TEE noted in this guideline is cardiac arrest, although the accompanying articles noted that this feasibly could be expanded to include concern for other pathologies in critically ill patients. Important contraindications are known esophageal injury or stricture and the lack of a definitive airway. The latter is important because this may differ from many scenarios encountered by cardiologists and anesthesiologists when an exam under sedation without a definitive airway may be reasonable.
The focused TEE exam described includes the midesophageal four chamber, midesophageal long axis, and the transgastric short axis view. There are five focused questions specified: identification of cardiac activity (or the absence of activity), identification of cardiac rhythm, evaluation of left ventricular function, evaluation of the right ventricular function, and identification of pericardial effusion and cardiac tamponade. The training recommended is a minimum of two to four hours of TEE-specific training, 10 proctored TEE examinations (including probe insertion), and a standardized assessment by a credentialed TEE provider. Also discussed in these guidelines are the many considerations for beginning an ED TEE program, including cost, compatibility with existing equipment, high-level disinfection, storage, continuing education, and collaboration with other departments performing TEE.
The transesophageal transducer is distinct from other transducers commonly used in the ED.46 Prior to using TEE, it is important to understand probe control, how to introduce the probe into the esophagus, and how to obtain the focused views mentioned previously. The TEE probe has additional movements not possible with other probes. Rotating the probe clockwise or counterclockwise also can allow for visualization of structures in a 360-degree plane perpendicular to the probe. The large wheel on the handle can cause anteflexion and retroflexion; the smaller wheel causes left and right flexion. The most unique function of the TEE probe is the multiplane button. This allows for rotation of the plane of imaging from 0 to 180 degrees. This allows the image to transect the heart through various planes.
Probe insertion usually will take place in a patient with an endotracheal tube, which makes the possibility of endotracheal placement less likely. If there is difficulty with placement, do not force the probe, as this can cause injury to the aerodigestive tract. Neck flexion may facilitate placement, and, in some cases, a laryngoscope is needed to guide placement.38
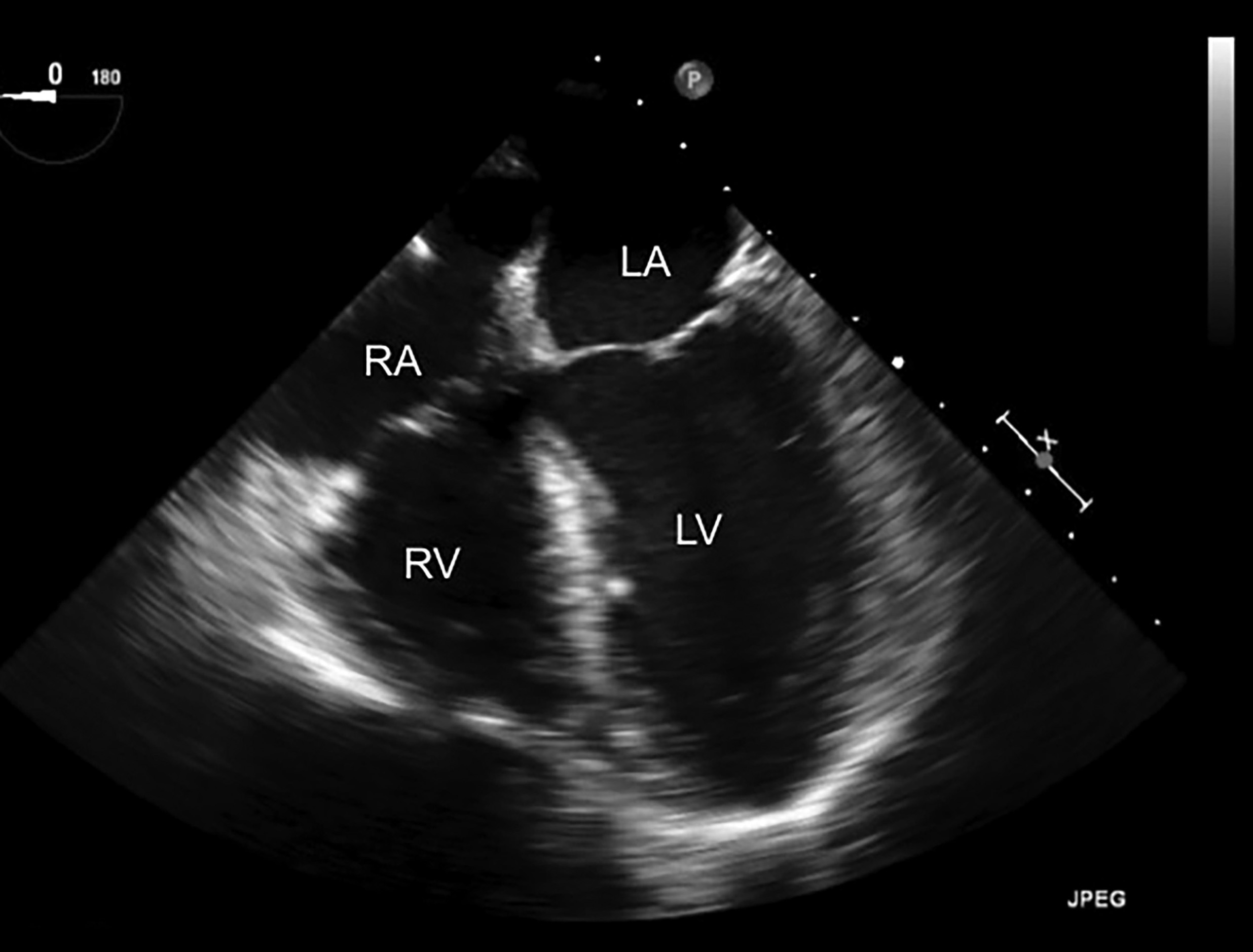
The midesophageal four-chamber view will be the first focused view visible after placement. (See Figure 9.) The multiplane will be 0-20 degrees. Slight retroflexion of the probe may be needed. This view looks similar to a transthoracic apical four chamber except that the near field is focused over the left atrium. Remembering that the esophagus is posterior to the left atrium will help with orientation. This view shows all four chambers and can be used to assess ventricular function as well as to compare the relative sizes of the ventricles.
Figure 9. TEE Image in the Midesophageal Four Chamber View |
 |
|
The right atrium (RA), right ventricle (RV), left atrium (LA), and left ventricle (LV) are labeled. This image represents normal cardiac anatomy. |
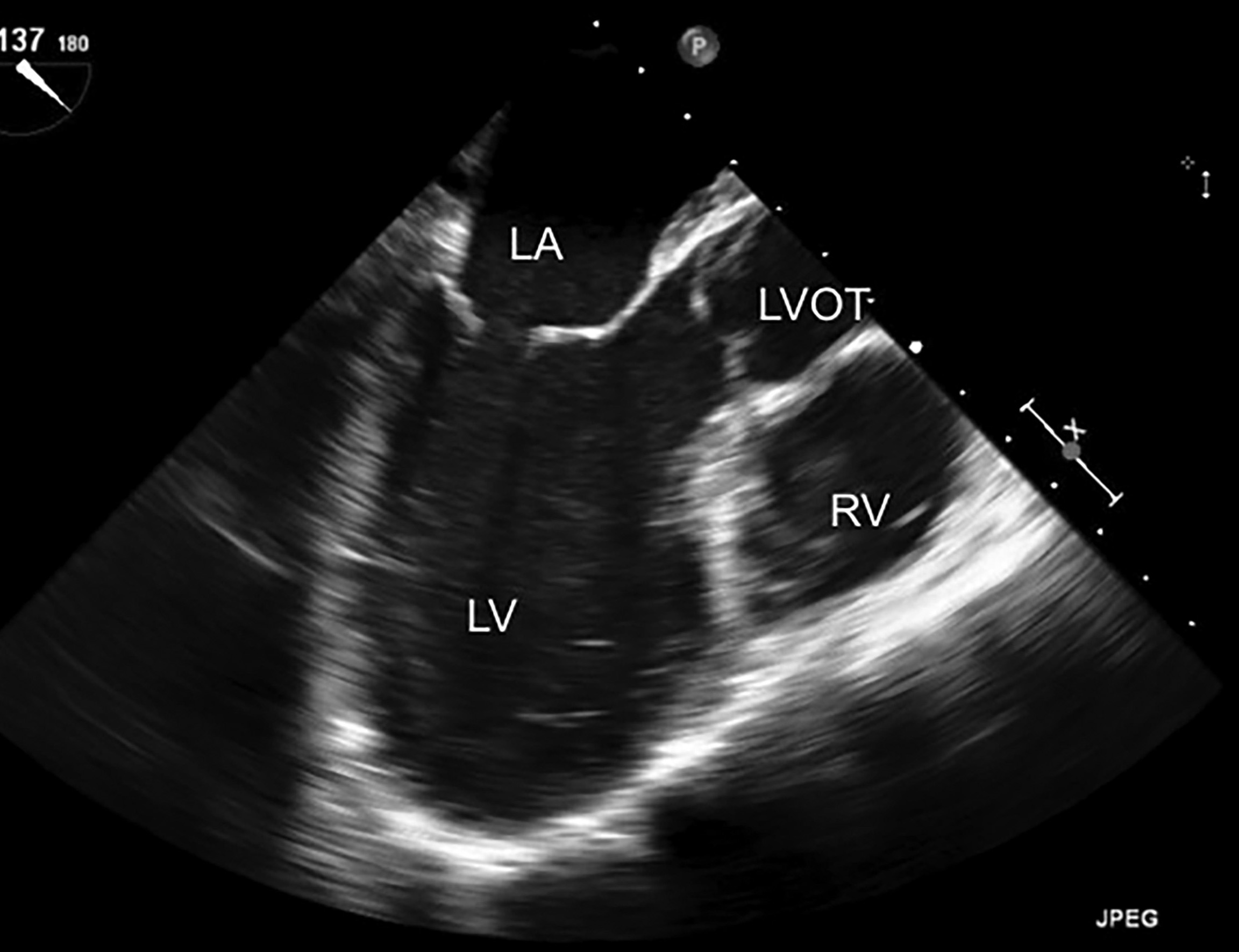
By rotating the multiplane to 110-160 degrees, the midesophageal long axis view will be obtained, analogous to the parasternal long axis view in TTE. (See Figure 10.) This view can be used to evaluate left ventricular function and also the efficacy of compressions during chest compressions.
Figure 10. TEE Image in the Midesophageal Long Axis View |
 |
|
The right ventricle (RV), left atrium (LA), left ventricle (LV), and left ventricular outflow tract (LVOT) are labeled. The mitral valve and aortic valve are both visualized. This image represents normal cardiac anatomy. |
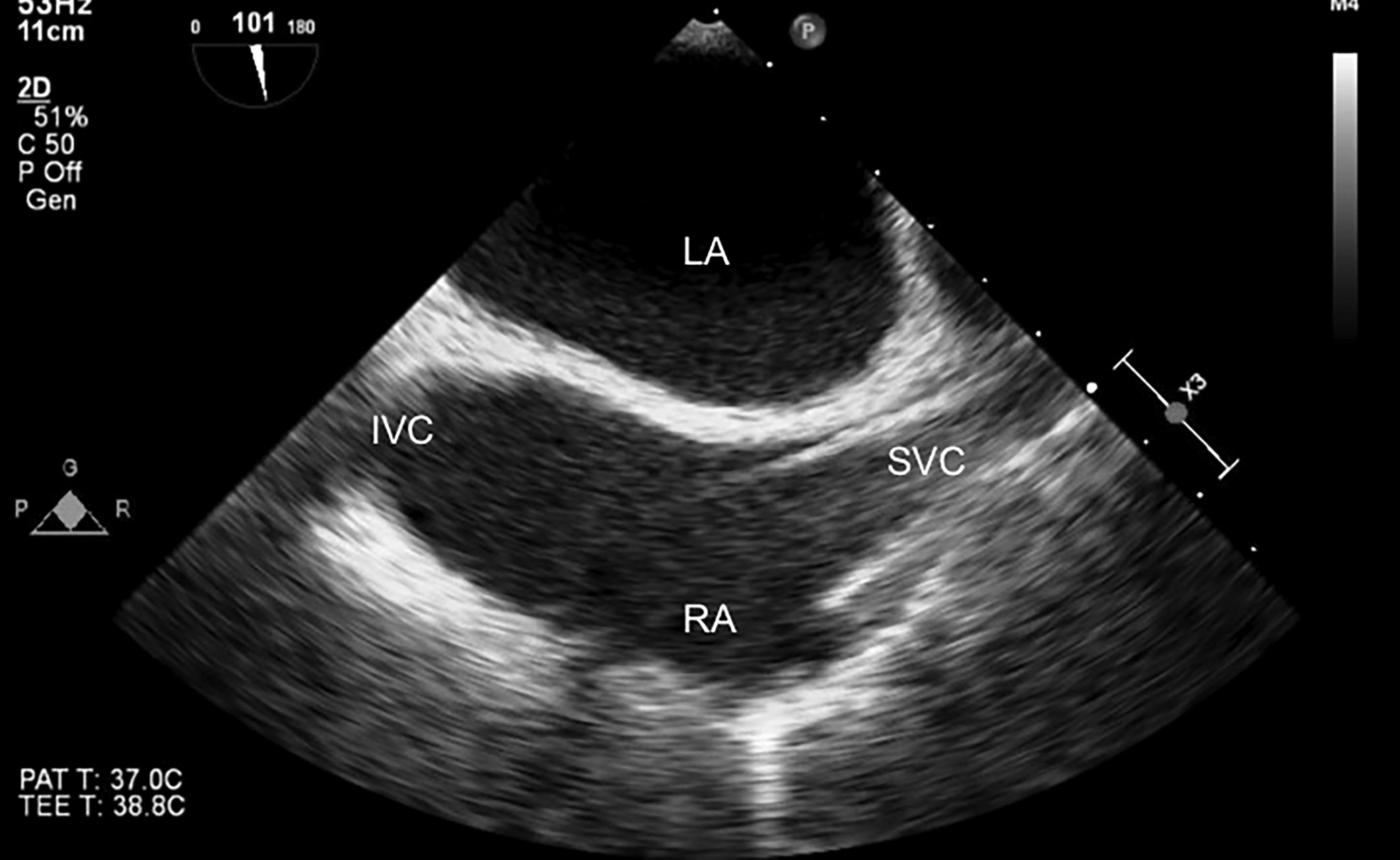
The bicaval view shows both the inferior and superior vena cava entering the right atrium. (See Figure 11.) This view also is obtained from the midesophageal position. The probe is rotated clockwise toward the right side of the heart with the multiplane at 90-100 degrees without flexion. This view can help with estimating fluid responsiveness and guiding procedures.
Figure 11. TEE Image in the Bicaval View |
 |
|
The left atrium (LA), right atrium (RA), superior vena cava (SVC), and inferior vena cava (IVC) are labeled. This image represents normal cardiac anatomy. |
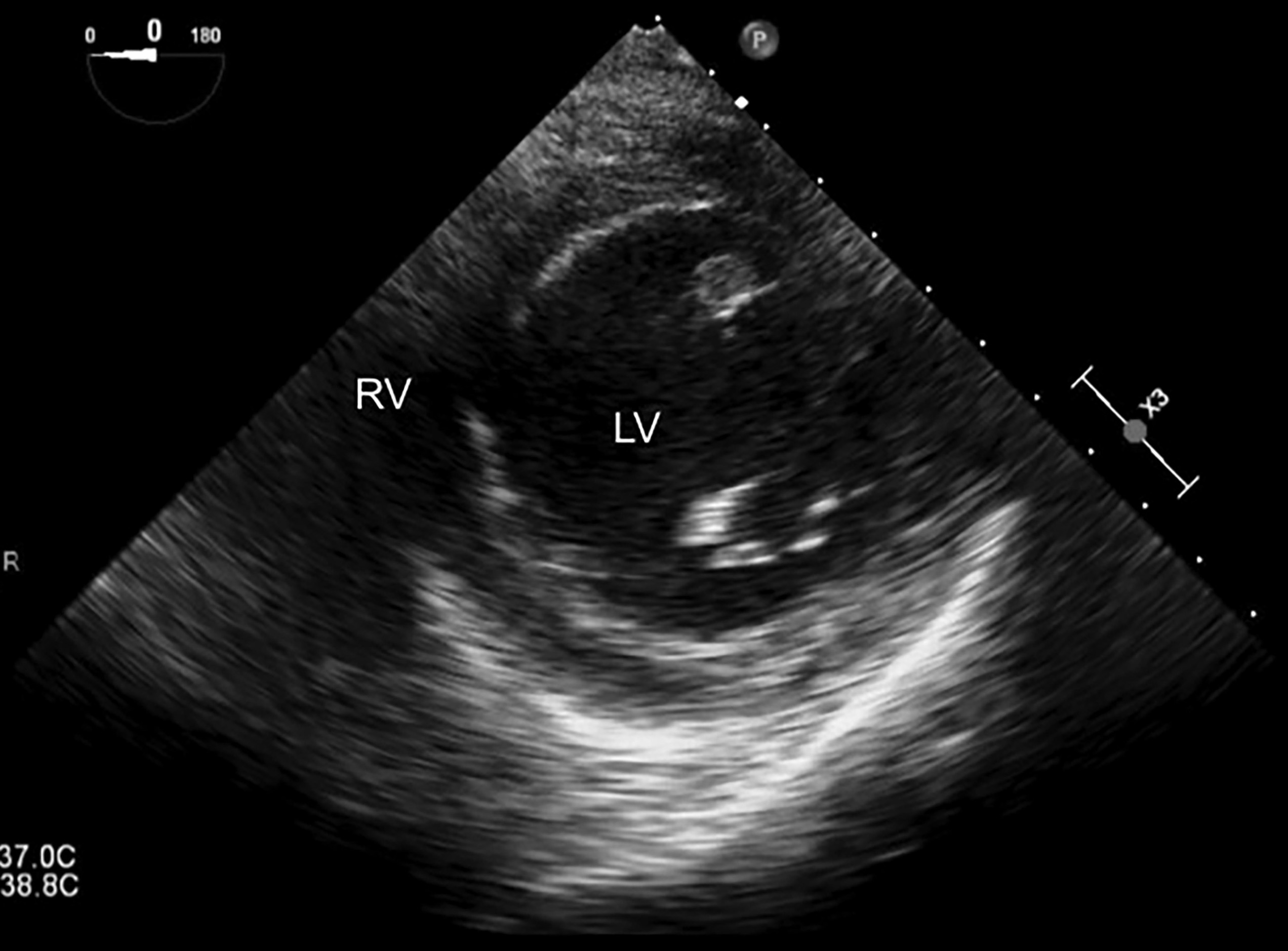
Advancing the probe into the stomach and anteflexing with the multiplane at 0 degrees will obtain the transgastric short axis view. (See Figure 12.) This view is analogous to the parasternal short axis view. The views are almost identical, with the important exception that in TEE the inferior wall of the left ventricle is found in the near field. This view can be used to assess for circumferential left ventricular function and regional wall motion abnormalities.45
Figure 12. TEE Image in the Transgastric Short Axis View |
 |
|
The right ventricle (RV) and left ventricle (LV) are labeled. This image represents normal cardiac anatomy. |
Not all views need to be obtained during each exam. In the aforementioned article by Arntfield and colleagues, the midesophageal four-chamber view was obtained 98% of the time, compared to the significantly lower frequency of obtaining the other views.38 This likely is because of both the ease and rapidity of obtaining this view and the many questions that can be answered from this vantage. The bicaval view was used 47% of the time, perhaps explaining why this view was not included as a recommended view in the TEE guidelines.38,45 It is possible that in the setting of cardiac arrest, one or two views may provide all of the information needed.44
Considerations Prior to Implementation of TEE
Despite this early evidence showing potential benefit for TEE in the ED, several issues must be considered. The first would be the significant investment required. TEE transducers have substantial cost, both the initial purchase (generally at least $40,000)38 and the necessary upkeep and storage. It is important to ensure that there is appropriate storage and a system for sanitation in place prior to purchase. Beginning an emergency department TEE program takes not only a financial investment, but also many hours of faculty time for training and systems management. There should be discussion and collaboration with other departments that routinely perform TEE. There may be initial concern for lost revenue opportunity; however, most will agree that in the emergent case of cardiac arrest, it is neither practical nor feasible to expect a consultant to present to the patient’s bedside 24 hours a day expeditiously enough to provide this service. Relationships with these departments are opportunities for continued training, education, and research.
Regarding the clinical challenges of TEE in the ED, it is clear that appropriate indications and contraindications will help lead to a successful program. Many fear the serious complications of TEE, such as esophageal perforation, bleeding, pulmonary compromise, or cardiac arrhythmias, despite the fact that these collectively have been reported to occur in only 0.18% of cases.47 Considering the fact that the ED cardiac arrest population will have a definitive airway and by the nature of their cardiac arrest generally have a poor prognosis, the risk-to-benefit ratio of this procedure is a favorable one. Nonetheless, it is important to ensure prior to an exam that the individual patient does not have contraindications.
Perhaps even more important is to ensure that each patient has the appropriate indication. Recall that even within the impressive results of the Arntfield et al study, 24% of the cases had no diagnostic benefit.38 A study performed in the medical intensive care unit in 2017 shows that expanding the indications will lead to a lower yield on the exam.48 In this study, TEE could be performed for shock state, guiding therapy, or concern for endocarditis. With these broader indications, the researchers found 38% change in clinical management, much less than the 67% seen in the Arntfield et al study. Regardless, it is still too early to evaluate whether patients would benefit from other indications in the ED, but “scope creep” is an obvious risk. Potential applications for future use include patients with shock, patients with atrial fibrillation in whom a cardiac thrombus must be ruled out, procedural guidance, evaluation for endocarditis, or evaluation of other valvular pathology.44 Although many emergency POCUS experts continue to blur the lines between focused and comprehensive exams, the majority of these applications are outside of the scope of practice endorsed by ACEP.1
Although TEE offers an exciting new development with great potential, it is paramount to realize that the data for this modality in the ED still are quite limited. The existing data, to our knowledge all of which have been reviewed above, are limited to small observational or retrospective studies. In fact, there is concern that ACEP endorsed these guidelines prematurely. In a letter to the editor, authors cited concerns that TEE would lead to more intubation with possible deleterious effects.49 They also worried that many views potentially could be obtained with TTE. Although this latter concern was somewhat addressed by Arntfield et al, there is not sufficient evidence to incontestably support the superiority of TEE at this time. Another concern not mentioned, but important from an administrative standpoint, is the fact that there has not been any cost analysis of supporting a TEE program in the ED.
Although these concerns are valid, the greater concern, also brought forth by these authors, is the lack of patient-centered outcomes within the current data. There have been no prospective trials performed within the ED, and the majority of the existing data focus on provider-oriented outcomes, such as change in diagnosis or treatment. What still is unknown is whether performing TEE in ED has a benefit to patients, such as improved time to treatment or mortality benefit.
There are many challenges to performing this type of research. The incidence of cardiac arrest leads to long periods of time needed in enrollment for sufficient sample size. The poor outcomes and low survival of this population overall may make finding a patient-centered benefit difficult. Furthermore, in EDs in which TEE is performed routinely in cardiac arrest, it already may be considered unethical to withhold this modality. Therefore, it may take years to build an evidence base for this practice. Practitioners likely will need to decide based on the limited data available, whether this investment is worthwhile for the potential benefits offered.
In summary, although there is limited evidence to support TEE in cardiac arrest in the ED, the promising preliminary data and the intuitive advantages offered in this select population lead to a risk-to-benefit ratio that favors adoption of this tool. This advancement offers the opportunity to improve the operations and outcomes in a critically ill population. Emergency physicians need to be aware that this modality has the potential to provide assistance with management beyond that possible with TTE during a cardiac arrest and with little established risk. At this time, the available data and national guidelines would caution for a close adherence to the appropriate indications and a deliberate coordination for training and quality assurance.
CONCLUSION
The recent multicenter trials have offered evidence that ultrasound in cardiac arrest provides important prognostic information and can identify etiologies of cardiac arrest that may change management.9,14 Furthermore, there is preliminary evidence that ultrasound can identify a population of PEA arrest that would benefit from a deviation from standard cardiac arrest treatment.17,21 Along with potential benefits, there are important concerns about possible harm associated with POCUS during cardiac resuscitation.28,29,31 Care should be taken not to delay chest compressions when performing ultrasound during cardiac arrest. The lack of agreement between providers regarding what constitutes cardiac standstill calls for careful training in this area. Transesophageal echocardiography offers potential benefits over transthoracic echocardiography in the population of patients in cardiac arrest.
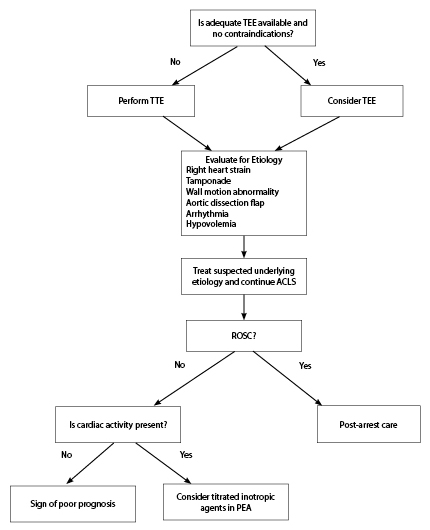
Based on our review of the literature, we have created an algorithm detailing a workflow for using ultrasound in cardiac arrest. Figure 13 includes indications, considerations, and suggested management based on ultrasound findings during cardiac arrest. Undoubtedly, this will be an area of continued research over the next years. By maintaining a practice of evidence-based sonology, we can ensure that this modality is used to its full potential and provide the best care possible to our patients.
Figure 13. Proposed Algorithm for Workflow Using Ultrasound in Cardiac Arrest |
 |
|
This includes standard ACLS guidelines and the potential for non-standard, patient-specific treatments in the setting of PEA arrest with cardiac activity. In the setting of cardiac arrest without ultrasound-visualized cardiac activity, the practitioner should incorporate this information into the clinical scenario as a prognostic tool. |
REFERENCES
- [No authors listed.] Ultrasound Guidelines: Emergency, Point-of-Care and Clinical Ultrasound Guidelines in Medicine. Ann Emerg Med 2017;69:e27-e54.
- Michalke JA. An overview of emergency ultrasound in the United States. World J Emerg Med 2012;3:85-90.
- Herbst MK, O’Rourke MC. Cardiac Ultrasound. Updated 2017 Nov 21]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018.
- Heart Disease and Stroke Statistics. American Heart Association. 2018. Available at: http://cpr.heart.org/AHAECC/CPRAndECC/General/UCM_477263_Cardiac-Arrest-Statistics.jsp. Accessed April 29, 2018.
- Tsou PY, Kurbedin J, Chen YS, et al. Accuracy of point-of-care focused echocardiography in predicting outcome of resuscitation in cardiac arrest patients: A systematic review and meta-analysis. Resuscitation 2017;114:92-99.
- Labovitz AJ, Noble VE, Bierig M, et al. Focused cardiac ultrasound in the emergency setting: A consensus statement of the American Society of Echocardiography and American College of Emergency Physicians. American Society of Echocardiography. 2010. Available at: https://bit.ly/2J5i6PE. Accessed May 23, 2018.
- Wu C, Zheng Z, Jiang L. The predictive value of bedside ultrasound to restore spontaneous circulation in patients with pulseless electrical activity: A systematic review and meta-analysis. PLoS One 2018;13:e0191636.
- Gardner KF, Clattenburg EJ, Wroe P, et al. The Cardiac Arrest Sonographic Assessment (CASA) exam — A standardized approach to the use of ultrasound in PEA. Am J Emerg Med 2018;36:729-731.
- Gaspari R, Weekes A, Adhikari S, et al. Emergency department point-of-care ultrasound in out-of-hospital and in-ED cardiac arrest. Resuscitation 2016;109:33-39.
- Hernandez C, Shuler K, Hannan H, et al. C.A.U.S.E.: Cardiac arrest ultrasound exam — a better approach to managing patients in primary non-arrhythmogenic cardiac arrest. Resuscitation 2008;76:198-206.
- Breitkreutz R, Walcher F, Seeger FH. Focused echocardiographic evaluation in resuscitation management: Concept of an advanced life support-conformed algorithm. Crit Care Med 2007;35(5 Suppl):S150-S161.
- Long B, Alerhand S, Maliel K, Koyfman A. Echocardiography in cardiac arrest: An emergency medicine review. Am J Emerg Med 2018;36:488-493.
- Aagaard R, Caap P, Hansson NC, et al. Detection of pulmonary embolism during cardiac arrest-ultrasonographic findings should be interpreted with caution. Crit Care Med 2017;45:e695-e702.
- Bolvardi E, Pouryaghobi SM, Farzane R, et al. The prognostic value of using ultrasonography in cardiac resuscitation of patients with cardiac arrest. Int J Biomed Sci 2016;12:110-114.
- Blaivas M, Fox JC. Outcome in cardiac arrest patients found to have cardiac standstill on the bedside emergency department echocardiogram. Acad Emerg Med 2001;8:616-621.
- Blyth L, Atkinson P, Gadd K, Lang E. Bedside focused echocardiography as predictor of survival in cardiac arrest patients: A systematic review. Acad Emerg Med 2012;19:1119-1126.
- Gaspari R, Weekes A, Adhikari S, et al. A retrospective study of pulseless electrical activity, bedside ultrasound identifies interventions during resuscitation associated with improved survival to hospital admission. A REASON Study. Resuscitation 2017;120:103-107.
- Littmann L, Bustin DJ, Haley MW. A simplified and structured teaching tool for the evaluation and management of pulseless electrical activity. Med Princ Pract 2014;23:1-6.
- Flato UAP, Paiva EF, Carballo MT, et al. Echocardiography for prognostication during the resuscitation of intensive care unit patients with non-shockable rhythm cardiac arrest. Resuscitation 2015;92:1-6. doi:10.1016/j.resuscitation.2015.03.024.
- Chardoli M, Heidari F, Rabiee H, et al. Echocardiography integrated ACLS protocol versus conventional cardiopulmonary resuscitation in patients with pulseless electrical activity cardiac arrest. Chin J Traumatol 2012;15:284-287.
- Prosen G, Križmarić M, Završnik J, Grmec Š. Impact of modified treatment in echocardiographically confirmed pseudo-pulseless electrical activity in out-of-hospital cardiac arrest patients with constant end-tidal carbon dioxide pressure during compression pauses. J Int Med Res 2010;38:1458-1467.
- Paradis NA, Halperin HR, Zviman M, et al. Coronary perfusion pressure during external chest compression in pseudo-EMD, comparison of systolic versus diastolic synchronization. Resuscitation 2012;83:1287-1291.
- Cunningham LM, Mattu A, O’Connor RE, Brady WJ. Cardiopulmonary resuscitation for cardiac arrest: The importance of uninterrupted chest compressions in cardiac arrest resuscitation. Am J Emerg Med 2012;30:1630-1638.
- American Heart Association. American Heart Association Guidelines for CPR and Emergency Cardiovascular Care. Updated 2017. Available at: https://eccguidelines.heart.org/index.php/circulation/cpr-ecc-guidelines-2/. Accessed April 26, 2018.
- Testa A, Cibinel GA, Portale G, et al. The proposal of an integrated ultrasonographic approach into the ALS algorithm for cardiac arrest: The PEA protocol. Eur Rev Med Pharmacol Sci 2010;14:77-88.
- Atkinson P, Bowra J, Milne J, et al. International Federation for Emergency Medicine Consensus Statement: Sonography in hypotension and cardiac arrest (SHoC): An international consensus on the use of point of care ultrasound for undifferentiated hypotension and during cardiac arrest. CJEM 2017;19:459-470.
- Damjanovic D, Schröder T, Breitkreutz R. The acronym of resuscitation ultrasound: RCC – Resume chest compressions! Resuscitation 2018 March 13; doi:10.1016/j.resuscitation.2018.03.014.
- Huis In ‘t Veld MA, Allison MG, Bostick DS, et al. Ultrasound use during cardiopulmonary resuscitation is associated with delays in chest compressions. Resuscitation 2017;119:95-98.
- Clattenburg EJ, Wroe P, Brown S, et al. Point-of-care ultrasound use in patients with cardiac arrest is associated prolonged cardiopulmonary resuscitation pauses: A prospective cohort study. Resuscitation 2018;122:65-68.
- Reed MJ, Gibson L, Dewar A, et al. Introduction of paramedic led Echo in Life Support into the pre-hospital environment: The PUCA study. Resuscitation 2017;112:65-69.
- Hu K, Gupta N, Teran F, et al. Variability in interpretation of cardiac standstill among physician sonographers. Ann Emerg Med 2018;71:193-198.
- Hahn RT, Abraham T, Adams MS, et al. Guidelines for performing a comprehensive transesophageal echocardiographic examination: Recommendations from the American Society of Echocardiography and the Society of Cardiovascular Anesthesiologists. Anesth Analg 2014;118:21-68.
- American Society of Anesthesiologists and Society of Cardiovascular Anesthesiologists Task Force on Transesophageal Echocardiography. Practice guidelines for perioperative transesophageal echocardiography. An updated report by the American Society of Anesthesiologists and the Society of Cardiovascular Anesthesiologists Task Force on Transesophageal Echocardiography. Anesthesiology 2010;112:1084-1096.
- Sengupta PP, Khandheria BK. Transoesophageal echocardiography. Heart 2005;91:541-547.
- O’Rourke MC, Mendenhall BR. Transesophageal Echocardiogram (TEE). Updated June 12, 2017. Treasure Island (FL): StatPearls Publishing; 2018 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK442026/?report=classic. Accessed April 26, 2018.
- Memtsoudis SG, Rosenberger P, Loffler M, et al. The usefulness of transesophageal echocardiography during intraoperative cardiac arrest in noncardiac surgery. Anesth Analg 2006;102:1653-1657.
- Blaivas M. Transesophageal echocardiography during cardiopulmonary arrest in the emergency department. Resuscitation 2008;78:135-140.
- Arntfield R, Pace J, Hewak M, Thompson D. Focused transesophageal echocardiography by emergency physicians is feasible and clinically influential: Observational results from a novel ultrasound program. J Emerg Med 2016;50:286-294.
- Hwang SO, Zhao PG, Choi HJ, et al. Compression of the left ventricular outflow tract during cardiopulmonary resuscitation. Acad Emerg Med 2009;16:928-933.
- Anderson KL, Fiala KC, Castaneda MG, et al. Left ventricular compressions improve return of spontaneous circulation and hemodynamics in a swine model of traumatic cardiopulmonary arrest. J Trauma Acute Care Surg 2018 Apr. 2: doi:10.1097/ta.0000000000001901.
- Fair J, Tonna J, Ockerse P, et al. Emergency physician-performed transesophageal echocardiography for extracorporeal life support vascular cannula placement. Am J Emerg Med 2016;34:1637-1639.
- Arntfield R, Pace J, McLeod S, et al. Focused transesophageal echocardiography for emergency physicians-description and results from simulation training of a structured four-view examination. Crit Ultrasound J 2015;7:27.
- Byars DV, Tozer J, Joyce JM, et al. Emergency physician-performed transesophageal echocardiography in simulated cardiac arrest. West J Emerg Med 2017;18:830-834.
- Fair J, Mallin M, Mallemat H, et al. Transesophageal echocardiography: Guidelines for point-of-care applications in cardiac arrest resuscitation. Ann Emerg Med 2018;71:201-207.
- [No authors listed.] Guidelines for the use of transesophageal echocardiography (TEE) in the ED for cardiac arrest. Ann Emerg Med 2017;70:442-445.
- Prabhu M, Raju D, Pauli H. Transesophageal echocardiography: Instrumentation and system controls. Ann Card Anaesth 2012;15:144-155.
- Daniel WG, Erbel R, Kasper W, et al. Safety of transesophageal echocardiography. A multicenter survey of 10,419 examinations. Circulation 1991;83:817-821.
- Garcia YA, Quintero L, Singh K, et al. Feasibility, safety, and utility of advanced critical care transesophageal echocardiography performed by pulmonary/critical care fellows in a medical ICU. Chest 2017;152:736-741.
- Ehrman RR, Favot MJ, Sullivan AN. Putting the guidelines ahead of the evidence for the use of transesophageal echocardiography by emergency physicians. Ann Emerg Med 2018;71:655-656.
Ultrasound is an integral aspect of caring for patients in the emergency department, and real-time use of this imaging modality at the bedside allows practitioners a hands-on approach to the clinical evaluation of patients. Point-of-care ultrasound (POCUS) has particular benefit in the critically ill patient for whom rapid information for decision-making is essential. Given this benefit, there is clear application for POCUS in the patient in cardiac arrest.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
