Pediatric Abdominal Ultrasound: A Comprehensive Guide to Making the Diagnosis — Part I
July 1, 2018
Reprints
AUTHORS
Kimberly M. Fender, MD, Emergency Medicine Resident, PGY 1, Department of Emergency Medicine, University of North Carolina, Chapel Hill
Daniel B. Park, MD, Associate Medical Director, Pediatric Emergency Medicine; Director, Pediatric Emergency Ultrasound; Assistant Professor, Departments of Pediatrics and Emergency Medicine, University of North Carolina School of Medicine, Chapel Hill
Daniel Migliaccio, MD, Clinical Assistant Professor, Department of Emergency Medicine, University of North Carolina, Chapel Hill
PEER REVIEWER
Aaron Leetch, MD, Assistant Professor, Departments of Emergency Medicine and Pediatrics; Program Director, Combined Emergency Medicine and Pediatrics Residency, University of Arizona, Tucson
EXECUTIVE SUMMARY
- Infantile hypertrophic pyloric stenosis is the most common cause of vomiting in infants requiring surgical intervention. The disease is caused by a thickening and elongation of the pylorus, which leads to a pathologic narrowing of the pyloric channel with a resultant gastric outlet obstruction. Physical exam findings (visible peristalsis and a pathognomonic palpable “olive-shaped mass”) are not present, and ultrasound is necessary to confirm the diagnosis (a pyloric muscle thickness > 3 mm and a pyloric length > 15 mm).
- Acute appendicitis is one of the most common indications for surgery in the pediatric population. The diagnosis is clinically challenging, as the presenting symptoms often are nonspecific or atypical. Proficiency in the diagnosis of appendicitis with point-of-care ultrasound is a useful skill for a rapid acute appendicitis diagnosis.
- Intussusception is the main source of bowel obstruction in the pediatric population and usually occurs at the junction between the ileum and the cecum where a segment of small bowel telescopes into the adjacent colon resulting in an ileocecal intussusception. Point-of-care ultrasound can be diagnostic and can decrease length of stay in the ED. Air reduction enema is the initial treatment of choice, with only a small subset of pediatric cases of intussusception requiring operative management.
- The role of the Focused Assessment With Sonography for Trauma (FAST) exam in the pediatric population is controversial, with variable use among institutions. Patients with a true positive FAST exam are likely to have an abnormal abdominal examination, with many having a seatbelt sign or other significant abrasion or contusion to the abdomen. In contrast to adults, the presence of free fluid on an abdominal CT scan in children does not necessitate an exploratory laparotomy. The results of a negative FAST exam in a child with blunt abdominal trauma should be interpreted carefully. A negative FAST exam is insufficient evidence to preclude the presence of serious intra-abdominal injury given its low sensitivity. Further imaging and diagnostic tests are warranted if there is a high clinical suspicion for injury.
Ultrasound is rapidly evolving as the ideal imaging modality for many common pediatric complaints. This two-part series reviews the select applications of ultrasound, reviewing the basic techniques, indications, and limitations for each exam.
— Ann M. Dietrich, MD, FAAP, FACEP, Editor
To avoid ionizing radiation in support of the ALARA (as low as reasonably achievable) concept in pediatric imaging, ultrasound frequently is the preferred imaging modality in the pediatric population.1 Ultrasound is noninvasive, cost-effective, easy to use, portable, and requires no sedation; however, the reliability of ultrasound is highly operator dependent. Point-of-care ultrasound performed and interpreted in the emergency department (ED) can reduce the time to disposition and expedite definitive patient care.2-4 This two-part series reviews the applications of ultrasound to diagnose common pediatric abdominal pathologies, as well as discusses the basic technique for each ultrasound exam. Part I will include discussion of pyloric stenosis, acute appendicitis, intussusception, the Pediatric Focused Assessment with Sonography for Trauma (FAST) exam, and imaging of the inferior vena cava (IVC) and aorta. Part II will include renal, testicular, ovarian, and biliary ultrasound focusing on diagnosing obstructive uropathy, ovarian torsion, testicular torsion, small bowel obstruction, and cholecystitis. Emergency medicine physicians should be proficient in point-of-care pediatric ultrasound while having a thorough understanding of the limitations and common pitfalls.
Pyloric Stenosis: The Nonbilious Enemy
A 4-week-old infant presents to the ED with a three-day history of progressively worsening projectile, nonbilious emesis. The infant appears well, but has intermittent emesis. The physical exam is unremarkable. The presence of infantile hypertrophic pyloric stenosis (IHPS) is highly suspect as the cause of the infant’s vomiting, but a diagnostic ultrasound must be performed to confirm the diagnosis. Emergency medicine physicians trained in the evaluation of IHPS can make the diagnosis accurately using point-of-care ultrasound.5
IHPS is the most common cause of vomiting in infants requiring surgical intervention.6-8 Although the etiology is not understood fully, the disease is caused by a thickening and elongation of the pylorus. This leads to a pathologic narrowing of the pyloric channel with a resultant gastric outlet obstruction.9 Pyloric stenosis often is observed in infants between 2-8 weeks of age, with the highest prevalence between 3-5 weeks of age.8 Between two to five infants in 1,000 live births will be affected, with the disease occurring four to five times more frequently in males than females.10 Often, physical exam findings (visible peristalsis and a pathognomonic palpable “olive-shaped mass”) are not present, and ultrasound becomes necessary to confirm the diagnosis.11 Ultrasound is the diagnostic method of choice for pyloric stenosis.12
To perform the exam, place the patient in the supine or right lateral decubitus position. The high-frequency linear array transducer (12-5 MHz) should be used for the exam. In the transverse oblique plane, place the transducer in the right upper quadrant parallel to the right costal margin and just below the xiphoid process.13,14 Identify the gastric antrum and then follow the stomach lumen caudally to locate the pylorus in its longitudinal axis. The pylorus will be found between the fluid-filled antrum and the duodenal bulb with the liver visualized ventrally and the pancreas dorsally.15 Alternatively, in the longitudinal plane, the pylorus can be identified in its short axis medial and slightly posterior to the gallbladder and lateral to the stomach.6,11 It may be helpful to administer 2 to 4 ounces of fluid orally to the patient to enhance the delineation of structures by creating an acoustic window if initial visualization is difficult.7,14,16,17 Conversely, it is important to consider that one of the most common reasons for a false-negative result is gastric overdistension.13
Pyloric measurements reported in the literature include pyloric muscle thickness, length, diameter, and volume; however, pyloric muscle thickness is the most commonly used diagnostic measurement to confirm pyloric stenosis.18 Although there is some variability in the literature and among institutions, the generally accepted thresholds to make the diagnosis of IHPS include a pyloric muscle thickness > 3 mm and pyloric length > 15 mm (long-axis measurement).7,11,15,16 (See Table 1.) To measure the pyloric muscle thickness, place the calipers at the superficial and deep borders of the anterior single layer of the pyloric muscle.13 If gastric emptying occurs through the relaxed pylorus during the exam, IHPS is ruled out.7,19 Furthermore, it is important to observe the pylorus for approximately five to 10 minutes to confirm a persistent abnormal thickening and to avoid confusion with a pylorospasm that may lead to a false-positive result.20 The sensitivity and specificity of ultrasound imaging in the diagnosis of IHPS is 98% and 100%, respectively, with a positive predictive value of 100% and negative predictive value of 90%.19 (See Figure 1.)
Table 1. Findings on Point-of-care Ultrasound |
||
Pathology |
Findings |
Comments |
|
Pyloric stenosis |
Pyloric muscle > 3 mm Pyloric length > 14 mm |
Diagnostic study of choice |
|
Appendicitis |
Dilated > 6 mm blind ended tubular structure, noncompressible, hyperemia, free fluid Appendicolith |
Ask patient where it hurts the most and start the exam there |
|
Intussusception |
Pseudokidney, donut sign, mass ~3-5 cm in diameter (commonly on right side of abdomen) |
Ileocecal most common |
Figure 1. Longitudinal and Transverse Views of the Pylorus |
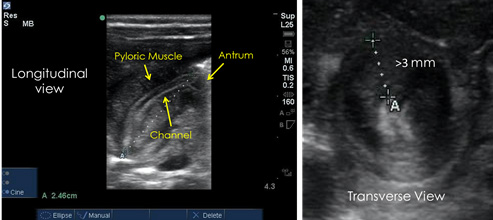 |
A helpful marker to guide the diagnosis in the long-axis view is the “antral nipple” sign, also known as the “cervix” sign, that is produced from the mucosa of the hypertrophic pylorus extending back into the gastric antrum.7,13,21 Additionally, the characteristic “donut” sign formed by the outer hypertrophied pyloric muscle and the inner more echogenic mucosa and submucosa can be recognized in the short-axis view.11,14 Several studies have shown that pyloric muscle thickness varies with age and weight.16,22 Mean pyloric muscle thickness was found to be 0.42 cm (range, 0.18 to 0.86 cm), and mean pyloric length was 1.89 cm (range, 0.8 to 2.8 cm).16 Patients may have IHPS despite not meeting the ultrasound diagnostic measurements. Repeat ultrasound exams may be useful to monitor the progression of the disease and for patients with initial equivocal ultrasounds but with a high pretest probability of IHPS.16,23
Acute Appendicitis: To See or Not to See the Appendix? That Is the Question
An 11-year-old boy with an antalgic gait presents to the children’s ED. He reports a gradual onset of dull, constant periumbilical pain that began several hours earlier. The pain, now sharp in quality, has migrated to his right lower quadrant since onset. He had one episode of nonbilious emesis. He is febrile, but otherwise appears nontoxic. His exam is significant only for right lower quadrant tenderness to palpation. The immediate concern is evaluating the child for acute appendicitis (AA).
AA is one of the most common indications for surgery in the pediatric population.24 Frequently an appendicolith, impacted feces, or lymphoid follicle hyperplasia obstructs the lumen of the appendix causing inflammation.25 The peak incidence occurs in the second decade of life with the median age of diagnosis between 10 and 11 years.26 The disease is more common in males, and prevalence increases in the summer months.25,26 The diagnosis of AA is clinically challenging, as the presenting symptoms often are nonspecific or atypical. The classic signs (e.g., psoas sign) have not been well studied in children and lack sensitivity.27 Proficiency in the diagnosis of appendicitis with point-of-care ultrasound is a useful skill for a rapid AA diagnosis. (See Figure 2.)
Figure 2. Findings Indicative of Appendicitis |
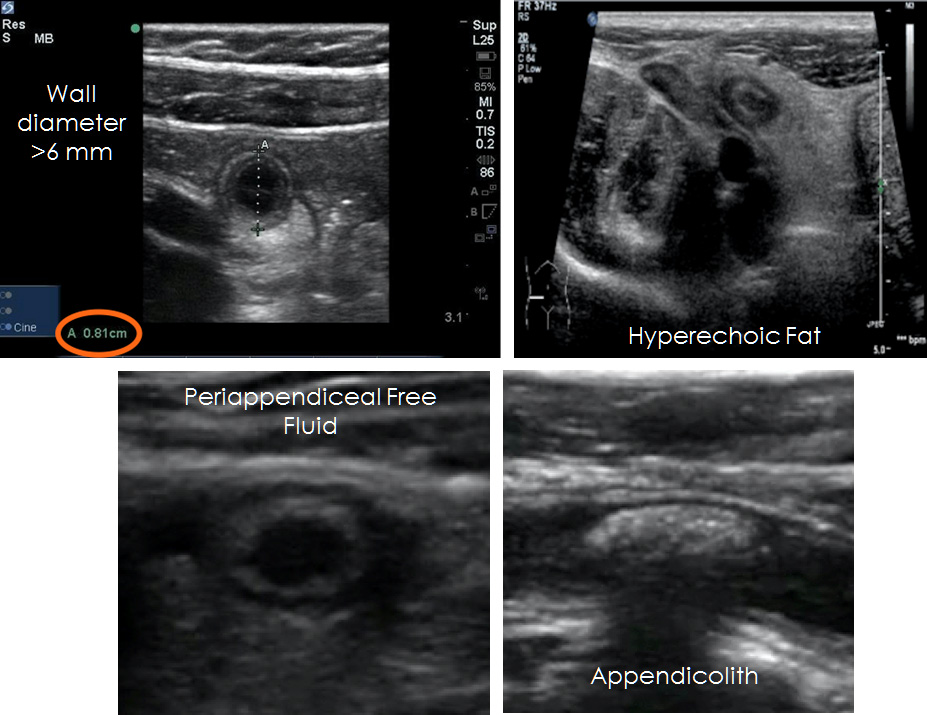 |
With appropriate training, emergency medicine physicians can diagnose appendicitis accurately in the pediatric population using point-of-care ultrasound.28,29 Although computed tomography (CT) is considered the gold standard for diagnosis, ultrasound is the preferred initial imaging modality in the diagnosis of AA in the pediatric population to reduce radiation exposure.30,31 CT often is reserved for children in whom the diagnosis of AA still remains uncertain after a nondiagnostic ultrasound and a high clinical suspicion persists, usually after consulting with a pediatric surgeon. The use of ultrasound preferentially over CT has been shown to reduce costs associated with imaging without leading to higher rates of missed cases or negative appendectomies.32 Recently, CT utilization has decreased significantly, while ultrasound use has continued to rise in AA diagnosis.33 The sensitivity and specificity of ultrasound varies in the literature from 86-98% and 81-97%, respectively.34-36 In comparison, a CT scan has a sensitivity of 94% and specificity of 95%.35,36
Several factors, including female patients, after-hours unavailability of an ultrasonographer specialized in pediatrics, older children, operator inexperience, and obesity, have been associated with a nondiagnostic ultrasound.37,38 In contrast, researchers in one study found that body mass index (BMI) did not affect the likelihood of a nondiagnostic scan.29 Alternatively, a recent retrospective review found that the positive predictive value of ultrasound in obese and overweight children was high, but the sensitivity and negative predictive value were poor.39 Selecting ultrasound as the initial imaging study in suspected appendicitis does not increase the risk of perforation compared to children who only received CT imaging.40
To perform the exam, place the patient in the supine position while using a curvilinear or linear transducer. To improve visualization of the appendix, apply continuous, graded compression during expiration to displace overlying bowel gas and reduce the distance between the probe and the appendix.41,42 If the patient’s pain is well localized, starting the exam at the point of maximal tenderness can be helpful. Use of pain medication to improve the patient’s tolerance of the exam may be beneficial.
Start the exam in the transverse plane at the level of the umbilicus. Move the probe laterally toward the abdominal wall until the ascending colon comes into view. Proximally trace the ascending colon to the end of the cecum in the right iliac fossa. Then, move the probe medially. Key landmarks to identify are the psoas muscle and iliac vessels; the appendix should be seen anterior and medial to the psoas muscle and lateral to the iliac vessels. Sweep the probe into the pelvis then back toward the umbilicus to assist with visualization. If the appendix has not yet been identified, rotate the probe to the sagittal plane and fan across the cecum and psoas muscle.28
The normal appendix should appear as a non-peristaltic, compressible, and blind-ending tubular structure. A normal appendiceal wall diameter measures less than 6 mm.43-45 In children younger than 6 years of age, a single wall thickness of 3 mm is considered normal.46 The “gut signature” (ultrasound term used to describe the appearance of the gastrointestinal wall) of the appendix is composed of five distinct layers. These layers, including the outermost echogenic serosa to the innermost hyperechoic mucosal surface, can be visualized on ultrasound and may assist in locating the appendix.43
A non-compressible appendix with an outer-to-outer wall diameter of > 6 mm, along with a single wall thickness of ≥ 3 mm, is considered diagnostic for AA in most cases.28,47 (See Table 1.) Researchers in one study found that a non-compressible appendix was 96% sensitive and specific for AA.48 Authors of several studies have challenged the traditional cutoff diameter and proposed developing a diameter-based stratification scheme or alternatively increasing or eliminating the cutoff diameter for the diagnosis of AA.49-52
Increased vascularity of the wall of the appendix, visualized by color Doppler, is suggestive of inflammation but has low sensitivity.48 Thickening and hyperechogenicity of the periappendiceal mesenteric fat and loss of the normal appendiceal wall signature also can be helpful in the diagnosis of AA.47,53 Secondary signs that may be present on ultrasound include mesenteric lymphadenopathy and free fluid in the abdomen and pelvis.47 It is important to consider that recent studies have questioned whether the aforementioned markers remain applicable given conflicting evidence in the literature.44,53
Recognizing perforation of the appendix can be challenging diagnostically. Markers associated with perforation include abscess formation, periappendiceal fluid, intraluminal appendicolith, and loss of the submucosal layer of the appendiceal wall.54 An abscess typically appears on ultrasound as a walled-off fluid collection with internal echoes and surrounding inflammation. The appendix may become compressible after perforation occurs.55
Many algorithms and scoring systems have been developed to guide clinical decision-making in children with nondiagnostic ultrasounds, with a primary aim to reduce CT scans and hospital admissions.37,56,57 Authors of one study found that children with a nondiagnostic ultrasound concurrent with a white blood count < 11 x 103/µL and C-reactive protein < 5 mg/dL had a low likelihood of having AA.37 Alternatively, other researchers have suggested pursuing CT after a nondiagnostic ultrasound if the child met at least two clinical predictors, including right lower tenderness, peritoneal signs, and a WBC > 10,000 in mm3.58 Authors of another study suggested the initial use of ultrasound along with the Pediatric Appendicitis Score as exclusionary means, followed by CT for inconclusive cases.57 Regardless, it is important for hospital systems to develop an evidenced-based algorithm for these equivocal cases to streamline care and avoid unnecessary radiation.
Intussusception: What Do You Have Against Jelly?
A smiling 8-month-old male, previously healthy and up-to-date on immunizations, arrives at the pediatric ED. His parents report that throughout the day he has had episodes of playfulness followed by periods of discomfort and irritability. He has had one episode of bloody stool. His abdominal exam is significant for a “sausage-shaped” mass. The classic triad of vomiting, “currant-jelly” stools, and a palpable mass is not seen commonly in intussusception. Ultrasound is the diagnostic method of choice.59 Point-of-care ultrasound, performed by trained pediatric emergency medicine physicians, can be diagnostic and can decrease length of stay in the ED.60,61 (See Figure 3.)
Figure 3. Intussusception Signs |
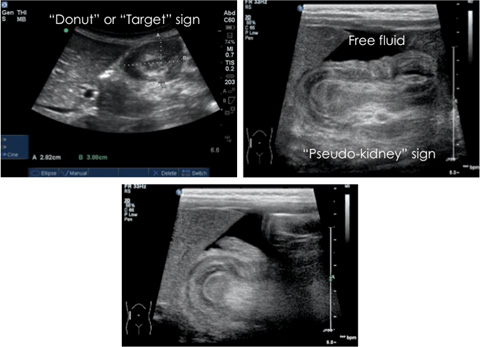 |
Intussusception is the main source of bowel obstruction in the pediatric population.17 It occurs primarily in children between the ages of 6 months and 2 years.14 Intussusception usually occurs at the junction between the ileum and the cecum where a segment of small bowel telescopes into the adjacent colon, resulting in an ileocecal intussusception. Most cases of intussusception are idiopathic in etiology; however, a percentage of cases are produced by a lead point such as a Meckel’s diverticulum or Henoch–Schönlein purpura.17 High morbidity and mortality may occur if the diagnosis is not made in a timely manner, including reduced enema reduction success, bowel necrosis, peritonitis, sepsis, and bowel perforation.61 Air reduction enema is the initial treatment of choice, with only a small subset of pediatric cases of intussusception requiring operative management.62
Most commonly, intussusception is found on the right side of the abdomen measuring as a mass 3-5 cm in diameter.63 For the examination, the patient should be placed in the supine position, and a linear, high-frequency transducer selected. Multiple techniques exist to identify intussusception. Two similar approaches recommend either starting the exam at the epigastric region or hepatic flexure (the transition between the ascending and transverse colon) and following either the transverse and/or the ascending colon downward toward the cecum in the right lower quadrant.64,65 An alternative method initiates the exam in the right lower quadrant with the psoas muscle as a starting landmark. From the right lower abdominal quadrant, the colon is traced to the right upper quadrant then to the left upper quadrant, and subsequently to the left lower quadrant, rotating the probe to either the transverse or longitudinal orientation with each turn of the colon.61 Particular attention should be paid to the right upper quadrant, as upward of 20% of cases may be overlooked if an inadequate exam is performed.64 Another method suggests a “lawn mower” approach in which the entire abdomen is scanned thoroughly in both the longitudinal and horizontal directions, comparable to mowing a lawn.64
In the longitudinal view, the characteristic “pseudokidney” sign can be recognized. Likewise in the transverse plane, a “donut” or “target” sign can be observed.64 (See Table 1.) If there is a lack of color flow on Doppler and fluid is present between the layers of the intussusception, a bowel ischemia is strongly indicated and the likelihood of a successful reduction with air enema decreases.17 Haustral markings, larger size, and lack of peristalsis are helpful markers to differentiate the colon from the small bowel.65 Using ultrasound to diagnose intussusception has a high negative predictive value of 99.7%, with a sensitivity and specificity of 97.9% and 97.8%, respectively.66
The Pediatric Focused Assessment With Sonography for Trauma (FAST): Not So Fast
Trauma is one of the leading causes of death in the pediatric population.67 The FAST exam is a well-accepted, noninvasive method to rapidly assess for free fluid (presumably blood in the setting of trauma) in adults and is an integral part of the Advanced Trauma Life Support algorithm.68 The role of the FAST exam in the pediatric population is controversial, with variable use among institutions. Authors of one study found that 63% of pediatric emergency medicine fellowship programs used the FAST exam “almost always or most of the time” compared to 35% of programs that used the FAST exam “occasionally or rarely.”59 Importantly, children are less likely to have free intraperitoneal fluid with blunt abdominal trauma (BAT) compared to adults.69 (See Figure 4.)
Figure 4. Free Fluid Images |
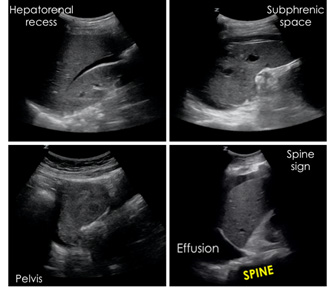 |
Authors of a 2007 meta-analysis found that the FAST exam had a sensitivity of 80%, a specificity of 96%, a positive likelihood ratio of 22.9, and a negative likelihood ratio of 0.2 for detecting hemoperitoneum in children. However, the sensitivity and specificity of the FAST exam decreased to 66% (95% confidence interval [CI], 56%-75%) and 95% (95% CI, 93%-97%), respectively, when only the most methodologically rigorous studies (Level I and II evidence) were included in the analysis.70 Holmes et al found that the sensitivity of the FAST exam for hemoperitoneum, secondary to blunt trauma, improved in pediatric patients presenting with hypotension, while another study found no significant difference.71 Sola et al suggested that a negative FAST exam combined with liver enzymes < 100 IU/L was an effective screening tool for intra-abdominal injury (IAI) and recommended observation over CT scan.72 Karam et al developed a scoring system to stratify the risk of IAI following BAT and found that patients with a blunt abdominal trauma in children (BATiC) score ≤ 7 who were hemodynamically stable with a negative FAST exam were unlikely to have IAI.73
In a 2017 randomized, controlled trial (RCT), Holmes et al compared the clinical outcomes of hemodynamically stable pediatric patients in a BAT setting who received a FAST exam with those who did not. The authors found no significant difference between rates of abdominal CT scans, missed IAI, length of stay in the ED, and hospital charges. Overall, the findings of this RCT did not support the routine use of the FAST exam in this patient population.74 In another recent multi-institutional analysis, the FAST exam rarely determined clinical management, as no child underwent a surgical intervention without a prior CT scan, and performance of the FAST exam did not correspond with a decreased use of CT imaging.75
Patients with a true positive FAST exam are likely to have an abnormal abdominal examination, with many having a seatbelt sign or other significant abrasion or contusion to the abdomen.75 In contrast to adults, the presence of free fluid on an abdominal CT scan in children does not necessitate an exploratory laparotomy.76 Holmes et al found that pediatric patients with isolated free fluid on CT imaging, in conjunction with a normal level of consciousness and benign abdominal exam, were at low risk for subsequently developing identifiable IAI.69 In another retrospective study, researchers found that the presence of abdominal tenderness on initial physical exam and the quantity of free fluid on CT scan predicted the need for future operative intervention.77 Many children with benign abdominal exams can be managed nonoperatively with observation and serial abdominal exams.76,77
The results of a negative FAST exam in a child with BAT should be interpreted carefully. A negative FAST exam is insufficient evidence to preclude the presence of serious IAI given its low sensitivity. Further imaging and diagnostic tests are warranted if there is a high clinical suspicion for injury. A negative FAST exam may provide additional reassurance in the setting of a low mechanism of injury, reinforcing physical exam, and low clinical suspicion for solid organ injury. A negative FAST exam must not be the sole determinant tool to dictate pediatric care in BAT cases.
Despite the controversy over the role of the FAST exam in the pediatric population, emergency medicine physicians should be proficient in the exam, as some patients still may benefit with low risk of harm to others. There are nine potential spaces in which free fluid may be present on the FAST exam, including the hepatorenal recess (Morison’s pouch), subphrenic recesses, caudal tips of the liver and spleen, caudal tips of the inferior poles of the kidneys, splenorenal recess, and pelvis. In the adult population, the caudal tip of the liver in the right upper quadrant is the most sensitive area for the accumulation of free fluid.78 In contrast, authors of a large retrospective review found that free fluid in the pediatric population is most likely to be visualized in the pelvis; the caudal tip of the liver is the next most common site.79 In the context of trauma, free fluid likely represents hemoperitoneum, but it is important to consider other sources of free fluid, including bile, bowel contents, ascites, and urine.14 The amount of free fluid that can be detected by the FAST exam varies from less than 100 mL in the pelvis to more than 600 mL in supine patients in the right upper quadrant.80-82
For this exam, the patient is placed in the supine position. Use a curvilinear or phase-array transducer to perform the scan. The phase-array transducer provides a smaller footprint offering easier scanning between the rib spaces. The FAST exam does not have to be completed in any particular order; however, a systematic approach, using a prescribed order, is recommended to avoid neglecting a view. Furthermore, the mechanism of injury or indications from the physical assessment may guide the initial starting location of the exam.
Place the probe in the right mid-axillary line between the eighth and 11th ribs83 with the probe indicator toward the patient’s head. Position the probe either in the coronal or oblique plane. The optimal view includes the liver with the caudal tip, Morison’s pouch, the right kidney, diaphragm, pleural space, and spine. The diaphragm will appear as a thin hyperechoic line separating the abdominal and thoracic cavity. Manipulate the probe as needed to acquire the view while attempting to avoid overlying rib and bowel gas shadowing. It is critical to fan the ultrasound probe through the entirety of the image. A single negative still-clip of an image is insufficient information to assume a negative FAST exam, as free fluid in the abdomen may be missed on a single view. Free fluid most often will appear hypoechoic or anechoic. Capture the caudal tip of the liver in the right upper quadrant. Often, all of the structures may not be visualized readily in a single image. In this scenario, start with the ultrasound probe more cephalad and subsequently slide the probe more caudally to capture each potential space where free fluid may collect.
Next, move the ultrasound probe to the left posterior axillary space between the eighth and 11th ribs. The left kidney, spleen, diaphragm, and pleural space should be identified. Ensure that both of the renal and splenic tips are seen. Often, this view is harder to obtain than the right upper quadrant for a variety of reasons, including the higher abdominal location of the kidney, overlying bowel or stomach gas, and the smaller acoustic window of the spleen compared to the liver. If the spleen and left kidney are difficult to visualize, asking the patient to take a deep inspiration may bring these organs into view.83 In the left upper quadrant, free fluid will collect in the splenodiaphragmatic space, splenorenal recess, and paracolic gutter. The most sensitive area in the left quadrant is the splenodiaphragmatic space.78 Beware of falsely identifying fluid in the stomach as free fluid. Although not the primary goal, an opportunistic use of the FAST exam is to detect hepatic and splenic parenchymal injuries.
Subsequently, move the ultrasound probe to the pelvis. A full bladder provides an optimal acoustic window to view the pelvic structures. To identify the pelvic space and the bladder, place the probe midline in the longitudinal plane approximately 2 cm superior to the pubic symphysis and aim the probe caudally.83 The probe indicator should be toward the patient’s head. The bladder will be a well-circumscribed, hyperechoic structure filled with anechoic fluid. With the bladder and the pelvic space in view, fan the probe left to right, capturing the entirety of the bladder. Free fluid will be present in the anterior pelvis, cul-de-sac (pouch of Douglas), or laterally in the paravesicular space. Rotate the transducer 90 degrees counterclockwise. The probe indicator should be oriented toward the patient’s right. Visualize the entire bladder in the transverse view. Located posterior to the bladder, the uterus in females and the prostate and seminal vesicles in males will be visualized. If not already present, placing a Foley catheter can help differentiate the bladder from free fluid.83 Doppler color imaging can assist in identification of the iliac vessels. Trace physiologic fluid may be present in the pelvis, particularly in menarchal females, making it sometimes difficult to discern from pathologic free fluid in the trauma setting.
The prompt diagnosis of a pericardial effusion in the trauma patient can be lifesaving. The diagnosis of a pericardial effusion can be made in multiple cardiac views; however, the subxiphoid approach is used most commonly. Place the ultrasound probe midline in the subxiphoid space with the probe indicator toward the patient’s right. It may be helpful to angle the probe indicator toward the patient’s left shoulder to take full advantage of the acoustic window provided by the liver. A four-chamber view of the heart should be visualized in the coronal plane. The pericardium is a hyperechoic line surrounding the heart. If a pericardial effusion is present, hypoechoic or anechoic fluid will be present between the pericardium and epicardium.
The extended-FAST exam (E-FAST), which includes the thoracic examination, is outside of the scope of this article; however, it is important to mention the mirror artifact and spine sign that may be present during the right upper quadrant portion of the FAST exam. In a normal scan, the mirror artifact should be observed. The mirror artifact occurs when an image (the liver) is duplicated on the opposite side of a strong reflector (the diaphragm).14 If the image on the opposite side of the diaphragm does not appear similar to the liver, thoracic pathology likely is present. Another characteristic sign is called the “spine” sign. In a normal scan, the spine will stop at the level of the diaphragm. If a clinically significant pleural effusion is present, the fluid functions as a medium that allows the visualization of the spine beyond the level of the diaphragm into the thoracic cavity.84,85
The Pediatric Inferior Vena Cava and Aorta: A Discourse on Dehydration
A 15-month-old boy arrives at the ED with a two-day history of multiple episodes of vomiting and diarrhea. His parents are concerned that he is dehydrated based on a reduction in oral fluid intake and the number of wet diapers. Vitals are significant for mild tachycardia. On exam, his mucus membranes appear dry, but his capillary refill is less than 2 seconds. Gastroenteritis, pyrexia, respiratory diseases, and other illnesses that cause vomiting, diarrhea, and poor oral intake predispose children to dehydration.
According to the World Health Organization, one in 10 childhood deaths is attributed to diarrheal illness.86 Early identification of dehydration can prevent morbidity and mortality such as shock and death. Assessing volume status in children is challenging clinically. Many of the existing clinical dehydration scales, as well as signs and symptoms historically associated with volume depletion, are suboptimal at identifying children with dehydration because of a lack of sensitivity.87-90 The percent weight change between a child’s baseline and acute illness weight is considered an accepted method for assessing the extent of dehydration in children.89-91 However, in the emergent setting, a pre-illness weight rarely is available. An accurate and efficient method to assess dehydration is vital to guide fluid resuscitation in critically ill children.
Multiple researchers have assessed and found varying results as to whether IVC measurements can act as a proxy for clinical dehydration and response to fluid replenishment in children. Several indices have been studied, including the inferior vena caval index (IVCI), aorta/IVC cross-sectional area index (Ao/IVCA), and aorta/IVC diameter index (Ao/IVCD).92 The IVC/Ao index, inverse of the Ao/IVCD, one of the most commonly used metrics, is calculated using the maximal anterior-posterior diameter of the IVC during maximal expiration and the aorta during systole in the transverse plane.93 (See Figure 5.) Although the IVC diameter varies with hydration status and respiration, the aortic diameter does not change significantly.93-95
Figure 5. Transverse Views of the IVC and Aorta |
|
“V” indicates the echogenic vertebral body. The arrow and the arrowhead indicate the superior mesenteric artery and the splenic vein, respectively. |
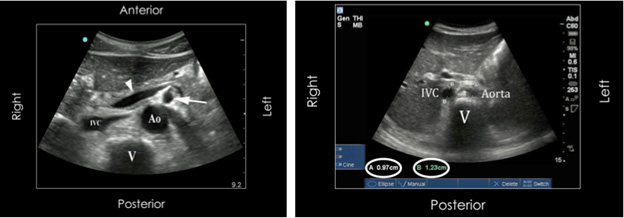 |
The IVC/Ao ratio was found to be relatively consistent for euvolemic children, regardless of age and weight, but was reduced significantly in patients who were considered clinically dehydrated, and subsequently increased with IV hydration.93 The IVC/Ao cutoff ratio of 0.8 was shown to predict the severity of dehydration based on a ≥ 5% weight loss, with a sensitivity of 86% and a specificity of 56% and a positive predictive value of 56% and negative predictive value of 86%.91
The Buddy Study and others validated the IVC/Ao ratio as a marker for dehydration status, while inspiratory IVC collapse and clinical assessment were found to be inadequate.94,95 In resource-limited settings, an aorta-to-IVC ratio ≥ 2 predicted severe dehydration (> 9% of percent weight changed after fluids) in children younger than 5 years of age with an acute diarrheal illness with a sensitivity of 67%, specificity of 49%, negative predictive value of 92%, and positive predictive value of 14%. For a one-point increase in aorta-to-IVC ratio, the percent dehydration increased by 0.8%.96 A prospective observational study found that Ao/IVCD had a greater correlation with fluid administration compared to IVCI and Ao/IVCA, which were found to be less reflective of intravascular volume.92 Central venous pressure does not correlate with the diameter of the IVC or aorta.97 The clinical dehydration score was found to correlate with Ao/IVCA, but no association was found with Ao/IVCD.98 However, in a 2015 meta-analysis, Freedman et al found insufficient evidence to support the routine use of ultrasound in determination of dehydration severity.87
Other measurements reported in the literature include the sole use the IVC and aorta cross-sectional area separately. The IVC cross section has an elliptical geometry whose area can be determined by π × (.5 × major axis diameter) × (.5 × minor axis diameter). The aorta has a circular geometry whose area is given by π × (.5 × diameter)2.
Perform the exam with the patient in the supine position using either the curvilinear or phase-array probe. Place the probe midline caudal to the xiphoid process with the probe indicator toward the patient’s right to obtain a transverse view of the IVC and aorta. Measure the maximum anterior-posterior diameter of the aorta during cardiac systole and the IVC during expiration.
In the longitudinal axis with the probe indicator toward the patient’s head, obtain a view of the IVC as it empties into the right atrium of the heart ensuring that the anterior and posterior walls of the IVC are parallel. Mark the IVC either 2 cm distal to either the confluence of the hepatic veins emptying into the IVC or right atrial-caval junction.94,95 Using M-mode on the ultrasound, measure the maximal and minimal width of the IVC; these measurements correspond to the expiration and inspiration phases of respiration, respectively.94 (See Figure 6.) From these measurements, the percentage of IVC inspiratory collapse can be calculated using [(IVC expiratory diameter – IVC inspiratory diameter) ÷ IVC expiratory diameter] × 100.
Figure 6. Longitudinal Views of the RA/IVC Junction |
|
Note the near complete collapse of the IVC with spontaneous inspiration in a dehydrated patient demonstrated by M-mode. |
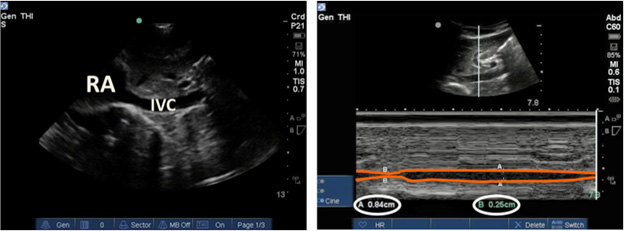 |
Ultrasound should not be used as the sole predictor of dehydration status in children presenting with acute diarrhea, but it serves as a helpful adjunct.87,96 Given that the IVC size increases with hydration in volume-depleted children, serial IVC ultrasounds may be a beneficial tool to track fluid resuscitation.93,96
CONCLUSION
Ultrasound is instrumental in the diagnosis of many pediatric abdominal diseases and guides patient care. Emergency medicine physicians should have a foundation in the basics of point-of-care ultrasound. Being adept at using ultrasound is an invaluable skill in expediting patient care by providing a quick diagnosis and disposition while avoiding ionizing radiation. While ultrasound is a very effective and efficient means to diagnose pediatric abdominal pathologies, there are cautions and limitations for each exam to which providers must be attentive to. While the diagnostic parameters in this article are widely accepted in the literature, it is important to verify the values used with the guidelines of each institution’s radiology department.
REFERENCES
- The ALARA (as low as reasonably achievable) concept in pediatric CT intelligent dose reduction. Multidisciplinary conference organized by the Society of Pediatric Radiology. August 18-19, 2001. Pediatr Radiol 2002;32:217-313.
- Thamburaj R, Sivitz A. Does the use of bedside pelvic ultrasound decrease length of stay in the emergency department? Pediatr Emerg Care 2013;29:67-70.
- Hall MK, Taylor RA, Luty S, et al. Impact of point-of-care ultrasonography on ED time to disposition for patients with nontraumatic shock. Am J Emerg Med 2016;34:1022-1030.
- Park YH, Jung RB, Lee YG, et al. Does the use of bedside ultrasonography reduce emergency department length of stay for patients with renal colic?: A pilot study. Clin Exp Emerg Med 2016;3:197-203.
- Sivitz AB, Tejani C, Cohen SG. Evaluation of hypertrophic pyloric stenosis by pediatric emergency physician sonography. Acad Emerg Med 2013;20:646-651.
- Costa Dias S, Swinson S, Torrao H, et al. Hypertrophic pyloric stenosis: Tips and tricks for ultrasound diagnosis. Insights Imaging 2012;3:247-250.
- Arys B, Mandelstam S, Rao P, et al. Sonography of the pediatric gastrointestinal system. Ultrasound Q 2014;30:101-117.
- Panteli C. New insights into the pathogenesis of infantile pyloric stenosis. Pediatr Surg Int 2009;251043-1052.
- Ranells JD, Carver JD, Kirby RS. Infantile hypertrophic pyloric stenosis: Epidemiology, genetics, and clinical update. Adv Pediatr 2011;58:195-206.
- MacMahon B. The continuing enigma of pyloric stenosis of infancy: A review. Epidemiology 2006;17:195-201.
- Malcom GE 3rd, Raio CC, Del Rios M, et al. Feasibility of emergency physician diagnosis of hypertrophic pyloric stenosis using point-of-care ultrasound: A multi-center case series. J Emerg Med 2009;37:283-286.
- Hiorns MP. Gastrointestinal tract imaging in children: Current techniques. Pediatr Radiol 2011;41:42-54.
- Cogley JR, O’Connor SC, Houshyar R, Al Dulaimy K. Emergent pediatric US: What every radiologist should know. Radiographics 2012;32:651-665.
- Hagen-Ansert SL. Textbook of Diagnostic Sonography. St. Louis: Elsevier Health Sciences; 2017.
- Rohrschneider WK, Mittnacht H, Darge K, Troger J. Pyloric muscle in asymptomatic infants: Sonographic evaluation and discrimination from idiopathic hypertrophic pyloric stenosis. Pediatr Radiol 1998;28:429-434.
- Said M, Shaul DB, Fujimoto M, et al. Ultrasound measurements in hypertrophic pyloric stenosis: Don’t let the numbers fool you. Perm J 2012;16:25-27.
- Gale HI, Gee MS, Westra SJ, Nimkin K. Abdominal ultrasonography of the pediatric gastrointestinal tract. World J Radiol 2016;8:656-667.
- Blumhagen JD, Maclin L, Krauter D, et al. Sonographic diagnosis of hypertrophic pyloric stenosis. AJR Am J Roentgenol 1988;150:1367-1370.
- Niedzielski J, Kobielski A, Sokal J, Krakos M. Accuracy of sonographic criteria in the decision for surgical treatment in infantile hypertrophic pyloric stenosis. Arch Med Sci 2011;7:508-511.
- Blumer SL, Zucconi WB, Cohen HL, et al. The vomiting neonate: A review of the ACR appropriateness criteria and ultrasound’s role in the workup of such patients. Ultrasound Q 2004;20:79-89.
- Fonio P, Coppolino F, Russo A, et al. Ultrasonography (US) in the assessment of pediatric non-traumatic gastrointestinal emergencies. Crit Ultrasound J 2013;5(Suppl 1):S12.
- Iqbal CW, Rivard DC, Mortellaro VE, et al. Evaluation of ultrasonographic parameters in the diagnosis of pyloric stenosis relative to patient age and size. J Pediatr Surg 2012;47:1542-1547.
- Hernanz-Schulman M. Pyloric stenosis: Role of imaging. Pediatr Radiol 2009;39(Suppl 2):S134-139.
- Fleischman RJ. Acute Abdominal Pain in Infants and Children. In: Tintinalli JE, Stapczynski JS, Ma OJ, et al, eds. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide. 8th ed. New York: McGraw-Hill Education; 2016.
- Bajaj L. Appendicitis. In: Schafermeyer R, Tenenbein M, Macias CG, et al, eds. Strange and Schafermeyer’s Pediatric Emergency Medicine. 4th ed. New York: McGraw-Hill Education; 2015.
- Pepper VK, Stanfill AB, Pearl RH. Diagnosis and management of pediatric appendicitis, intussusception, and Meckel diverticulum. Surg Clin North Am 2012;92:505-526, vii.
- Mayer TA, Krupa A, Mobley B. Chapter 45. Pediatric Emergency Medicine: Diversification in the ED. In: Strauss RW, Mayer TA, eds. Strauss & Mayer’s Emergency Department Management. New York: The McGraw-Hill Companies; 2014.
- Sivitz AB, Cohen SG, Tejani C. Evaluation of acute appendicitis by pediatric emergency physician sonography. Ann Emerg Med 2014;64:358-364.e354.
- Doniger SJ, Kornblith A. Point-of-care ultrasound integrated into a staged diagnostic algorithm for pediatric appendicitis. Pediatr Emerg Care 2018;34:109-115.
- Smith MP, Katz DS, Lalani T, et al. ACR Appropriateness criteria right lower quadrant pain — suspected appendicitis. Ultrasound Q 2015;31:85-91.
- Cundy TP, Gent R, Frauenfelder C, et al. Benchmarking the value of ultrasound for acute appendicitis in children. J Pediatr Surg 2016;51:1939-1943.
- Kharbanda AB, Christensen EW, Dudley NC, et al. Economic analysis of diagnostic imaging in pediatric patients with suspected appendicitis. Acad Emerg Med 2018; Feb 10. doi: 10.1111/acem.13387.
- Otero HJ, Crowder L. Imaging utilization for the diagnosis of appendicitis in stand-alone children’s hospitals in the United States: Trends and costs. J Am Coll Radiol 2017;14:603-608.
- Dibble EH, Swenson DW, Cartagena C, et al. Effectiveness of a staged US and unenhanced MR imaging algorithm in the diagnosis of pediatric appendicitis. Radiology 2018;286:1022-1029.
- Doria AS, Moineddin R, Kellenberger CJ, et al. US or CT for diagnosis of appendicitis in children and adults? A meta-analysis. Radiology 2006;241:83-94.
- Terasawa T, Blackmore CC, Bent S, Kohlwes RJ. Systematic review: Computed tomography and ultrasonography to detect acute appendicitis in adults and adolescents. Ann Intern Med 2004;141:537-546.
- Sola R Jr, Wormer BA, Anderson WE, et al. Predictors and outcomes of nondiagnostic ultrasound for acute appendicitis in children. Am Surg 2017;83:1357-1362.
- Keller C, Wang NE, Imler DL, et al. Predictors of nondiagnostic ultrasound for appendicitis. J Emerg Med 2017;52:318-323.
- Love BE, Camelo M, Nouri S, et al. Ultrasound accuracy in diagnosing appendicitis in obese pediatric patients. Am Surg 2017;83:1063-1067.
- Alerhand S, Meltzer J, Tay ET. Evaluating the risk of appendiceal perforation when using ultrasound as the initial diagnostic imaging modality in children with suspected appendicitis. Ultrasound 2017;25:166-172.
- Puylaert JB. Acute appendicitis: US evaluation using graded compression. Radiology 1986;158:355-360.
- Lee JH, Jeong YK, Park KB, et al. Operator-dependent techniques for graded compression sonography to detect the appendix and diagnose acute appendicitis. AJR Am J Roentgenol 2005;184:91-97.
- Gongidi P, Bellah RD. Ultrasound of the pediatric appendix. Pediatr Radiol 2017;47:1091-1100.
- Sanchez TR, Corwin MT, Davoodian A, Stein-Wexler R. Sonography of abdominal pain in children: Appendicitis and its common mimics. J Ultrasound Med 2016;35:627-635.
- Jeffrey RB Jr, Laing FC, Townsend RR. Acute appendicitis: Sonographic criteria based on 250 cases. Radiology 1988;167:327-329.
- Simonovsky V. Normal appendix: Is there any significant difference in the maximal mural thickness at US between pediatric and adult populations? Radiology 2002;224:333-337.
- Kwon LM, Lee K, Min SK, et al. Ultrasound features of secondary appendicitis in pediatric patients. Ultrasonography 2017; Aug 25. doi: 10.14366/usg.17029.
- Kessler N, Cyteval C, Gallix B, et al. Appendicitis: Evaluation of sensitivity, specificity, and predictive values of US, Doppler US, and laboratory findings. Radiology 2004;230:472-478.
- Trout AT, Towbin AJ, Fierke SR, et al. Appendiceal diameter as a predictor of appendicitis in children: Improved diagnosis with three diagnostic categories derived from a logistic predictive model. Eur Radiol 2015;25:2231-2238.
- Goldin AB, Khanna P, Thapa M, et al. Revised ultrasound criteria for appendicitis in children improve diagnostic accuracy. Pediatr Radiol 2011;41:993-999.
- Chicaiza HP, Malia L, Mulvey CH, Smith SR. Revisiting the appendiceal diameter via ultrasound for the diagnosis of acute appendicitis. Pediatr Emerg Care 2017; Oct. 2. doi: 10.1097/PEC.0000000000001278. .
- Trout AT, Sanchez R, Ladino-Torres MF, et al. A critical evaluation of US for the diagnosis of pediatric acute appendicitis in a real-life setting: How can we improve the diagnostic value of sonography? Pediatr Radiol 2012;42:813-823.
- Trout AT, Sanchez R, Ladino-Torres MF. Reevaluating the sonographic criteria for acute appendicitis in children: A review of the literature and a retrospective analysis of 246 cases. Acad Radiol 2012;19:1382-1394.
- Blumfield E, Nayak G, Srinivasan R, et al. Ultrasound for differentiation between perforated and nonperforated appendicitis in pediatric patients. AJR Am J Roentgenol 2013;200:957-962.
- Park NH, Oh HE, Park HJ, Park JY. Ultrasonography of normal and abnormal appendix in children. World J Radiol 2011;3:85-91.
- Ramarajan N, Krishnamoorthi R, Gharahbaghian L, et al. Clinical correlation needed: What do emergency physicians do after an equivocal ultrasound for pediatric acute appendicitis? J Clin Ultrasound 2014;42:385-394.
- Sayed AO, Zeidan NS, Fahmy DM, Ibrahim HA. Diagnostic reliability of pediatric appendicitis score, ultrasound and low-dose computed tomography scan in children with suspected acute appendicitis. Ther Clin Risk Manag 2017;13:847-854.
- Nishizawa T, Maeda S, Goldman RD, Hayashi H. Predicting need for additional CT scan in children with a non-diagnostic ultrasound for appendicitis in the emergency department. Am J Emerg Med 2018;36:49-55.
- Marin JR, Zuckerbraun NS, Kahn JM. Use of emergency ultrasound in United States pediatric emergency medicine fellowship programs in 2011. J Ultrasound Med 2012;31:1357-1363.
- Chang YJ, Hsia SH, Chao HC. Emergency medicine physicians performed ultrasound for pediatric intussusceptions. Biomed J 2013;36:175-178.
- Riera A, Hsiao AL, Langhan ML, et al. Diagnosis of intussusception by physician novice sonographers in the emergency department. Ann Emerg Med 2012;60:264-268.
- Vo A, Levin TL, Taragin B, Khine H. Management of intussusception in the pediatric emergency department: Risk factors for recurrence. Pediatr Emerg Care 2017; Dec 11. doi: 10.1097/PEC.0000000000001382.
- Daneman A, Navarro O. Intussusception. Part 1: A review of diagnostic approaches. Pediatr Radiol 2003;33:79-85.
- Doniger SJ, Salmon M, Lewiss RE. Point-of-care ultrasonography for the rapid diagnosis of intussusception: A case series. Pediatr Emerg Care 2016;32:340-342.
- Fischer JW, Sivitz AB, Abo AM. Chapter 20. Pediatric Applications. In: Ma OJ, Mateer JR, Reardon RF, Joing SA, eds. Ma and Mateer’s Emergency Ultrasound. 3rd ed. New York: The McGraw-Hill Companies; 2014.
- Hryhorczuk AL, Strouse PJ. Validation of US as a first-line diagnostic test for assessment of pediatric ileocolic intussusception. Pediatr Radiol 2009;39:1075-1079.
- Ballesteros MF, Williams DD, Mack KA, et al. The epidemiology of unintentional and violence-related injury morbidity and mortality among children and adolescents in the United States. Int J Environ Res Public Health 2018;15: pii: E616. doi: 10.3390/ijerph15040616.
- ATLS Subcommittee; American College of Surgeons’ Committee on Trauma; International ATLS working group. Advanced trauma life support (ATLS): The ninth edition. J Trauma Acute Care Surg 2013;74:1363-1366.
- Holmes JF, London KL, Brant WE, Kuppermann N. Isolated intraperitoneal fluid on abdominal computed tomography in children with blunt trauma. Acad Emerg Med 2000;7:335-341.
- Holmes JF, Gladman A, Chang CH. Performance of abdominal ultrasonography in pediatric blunt trauma patients: A meta-analysis. J Pediatr Surg 2007;42:1588-1594.
- Holmes JF, Brant WE, Bond WF, et al. Emergency department ultrasonography in the evaluation of hypotensive and normotensive children with blunt abdominal trauma. J Pediatr Surg 2001;36:968-973.
- Sola JE, Cheung MC, Yang R, et al. Pediatric FAST and elevated liver transaminases: An effective screening tool in blunt abdominal trauma. J Surg Res 2009;157:103-107.
- Karam O, Sanchez O, Chardot C, La Scala G. Blunt abdominal trauma in children: A score to predict the absence of organ injury. J Pediatr 2009;154:912-917.
- Holmes JF, Kelley KM, Wootton-Gorges SL, et al. Effect of abdominal ultrasound on clinical care, outcomes, and resource use among children with blunt torso trauma: A randomized clinical trial. JAMA 2017;317:2290-2296.
- Calder BW, Vogel AM, Zhang J, et al. Focused assessment with sonography for trauma in children after blunt abdominal trauma: A multi-institutional analysis. J Trauma Acute Care Surg 2017;83:218-224.
- Beierle EA, Chen MK, Whalen TV, Doolin EJ. Free fluid on abdominal computed tomography scan after blunt trauma does not mandate exploratory laparotomy in children. J Pediatr Surg 2000;35:990-992; discussion 993.
- Christiano JG, Tummers M, Kennedy A. Clinical significance of isolated intraperitoneal fluid on computed tomography in pediatric blunt abdominal trauma. J Pediatr Surg 2009;44:1242-1248.
- Lobo V, Hunter-Behrend M, Cullnan E, et al. Caudal edge of the liver in the right upper auadrant (RUQ) view is the most sensitive area for free fluid on the FAST exam. West J Emerg Med 2017;18:270-280.
- Brenkert TE, Adams C, Vieira RL, Rempell RG. Peritoneal fluid localization on FAST examination in the pediatric trauma patient. Am J Emerg Med 2017;35:1497-1499.
- Branney SW, Wolfe RE, Moore EE, et al. Quantitative sensitivity of ultrasound in detecting free intraperitoneal fluid. J Trauma 1995;39:375-380.
- Abrams BJ, Sukumvanich P, Seibel R, et al. Ultrasound for the detection of intraperitoneal fluid: The role of Trendelenburg positioning. Am J Emerg Med 1999;17:117-120.
- Von Kuenssberg Jehle D, Stiller G, Wagner D. Sensitivity in detecting free intraperitoneal fluid with the pelvic views of the FAST exam. Am J Emerg Med 2003;21:476-478.
- Ma OJ, Mateer JR, Kirkpatrick AW. Chapter 5. Trauma. In: Ma OJ, Mateer JR, Reardon RF, Joing SA, eds. Ma and Mateer’s Emergency Ultrasound. 3rd ed. New York: The McGraw-Hill Companies; 2014.
- Dickman E, Terentiev V, Likourezos A, et al. Extension of the thoracic spine sign: A new sonographic marker of pleural effusion. J Ultrasound Med 2015;34:1555-1561.
- Ahmed AA, Martin JA, Saul T, Lewiss RE. The thoracic spine sign in bedside ultrasound. Three cases report. Med Ultrason 2014;16:179-181.
- Unicef. One Is Too Many: Ending Child Deaths from Pneumonia and Diarrhea. United Nations Children’s Fund, The (UNICEF); 2016.
- Freedman SB, Vandermeer B, Milne A, Hartling L, Pediatric Emergency Research Canada Gastroenteritis Study Group. Diagnosing clinically significant dehydration in children with acute gastroenteritis using noninvasive methods: A meta-analysis. J Pediatr 2015;166:908-916.e901-906.
- Falszewska A, Szajewska H, Dziechciarz P. Diagnostic accuracy of three clinical dehydration scales: A systematic review. Arch Dis Child 2018;103:383-388.
- Gorelick MH, Shaw KN, Murphy KO. Validity and reliability of clinical signs in the diagnosis of dehydration in children. Pediatrics 1997;99:E6.
- Steiner MJ, DeWalt DA, Byerley JS. Is this child dehydrated? JAMA 2004;291:2746-2754.
- Chen L, Hsiao A, Langhan M, et al. Use of bedside ultrasound to assess degree of dehydration in children with gastroenteritis. Acad Emerg Med 2010;17:1042-1047.
- Choi YA, Kwon H, Lee JH, et al. Comparison of sonographic inferior vena cava and aorta indexes during fluid administered in children. Am J Emerg Med 2018; Jan 4. pii: S0735-6757(18)30009-3. doi: 10.1016/j.ajem.2018.01.010.
- Chen L, Kim Y, Santucci KA. Use of ultrasound measurement of the inferior vena cava diameter as an objective tool in the assessment of children with clinical dehydration. Acad Emerg Med 2007;14:841-845.
- Levine AC, Shah SP, Umulisa I, et al. Ultrasound assessment of severe dehydration in children with diarrhea and vomiting. Acad Emerg Med 2010;17:1035-1041.
- Jauregui J, Nelson D, Choo E, et al. The BUDDY (Bedside Ultrasound to Detect Dehydration in Youth) study. Crit Ultrasound J 2014;6:15.
- Modi P, Glavis-Bloom J, Nasrin S, et al. Accuracy of inferior vena cava ultrasound for predicting dehydration in children with acute diarrhea in resource-limited settings. PLoS One 2016;11:e0146859.
- Ng L, Khine H, Taragin BH, et al. Does bedside sonographic measurement of the inferior vena cava diameter correlate with central venous pressure in the assessment of intravascular volume in children? Pediatr Emerg Care 2013;29:337-341.
- Kwon H, Jung JY, Lee JH, et al. Sonographic aorta/IVC cross-sectional area index for evaluation of dehydration in children. Am J Emerg Med 2016;34:1840-1844.
Ultrasound is rapidly evolving as the ideal imaging modality for many common pediatric complaints. This two-part series reviews the select applications of ultrasound, reviewing the basic techniques, indications, and limitations for each exam.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
