Limb Ischemia and Gangrene
November 15, 2018
Reprints
AUTHORS
Judith Toski Welsh, MD, FACEP, Cleveland Clinic Emergency Services Institute, Assistant Professor, Medicine, Cleveland Clinic Lerner College of Medicine, Cleveland
Todd S. Welsh, MD, FACEP, Cleveland Clinic Emergency Services Institute, Clinical Assistant Professor, Medicine, Cleveland Clinic Lerner College of Medicine, Cleveland
PEER REVIEWER
Steven M. Winograd, MD, FACEP, Assistant Clinical Professor of Emergency Medicine, Mt. Sinai, NY
EXECUTIVE SUMMARY
- Blue toe syndrome involves the appearance of cyanotic toes with normal pedal pulses and is caused by emboli.
- Acute limb ischemia presents with sudden pain and cyanosis of the limb. Pulses are not palpable. Risk factors include smoking, diabetes, hyperlipidemia, hypertension, and male sex, and the risk increases with age.
- The diagnosis of limb ischemia is largely clinical. Extensive testing delays treatment that might either salvage the limb or reduce complications. Heparin should be started.
- Wet gangrene indicates infection, and patients should receive broad-spectrum antibiotics. These patients require emergent surgery.
Extremity pain is a common complaint in the emergency department (ED). Although many cases of extremity pain are the result of mild, self-limited issues, ischemia and gangrene are catastrophic causes of pain that initially can present with nondescript findings. To limit tissue loss and optimize patient outcomes, emergency physicians must be able to distinguish benign limb pain from the earliest stages of high-risk, life- and limb-threatening disease.
Introduction
Acute limb ischemia is a sudden decrease in limb perfusion that is a threat to the limb’s viability. Occlusion of a vessel leads to a reduction in blood flow that compromises oxygen and nutrient delivery to tissues. Peripheral nerves and skeletal muscles are sensitive to ischemia, and permanent damage can occur within six hours. By contrast, onset of symptoms of more than two weeks’ duration is considered chronic limb ischemia. When chronic limb ischemia leads to rest pain or tissue loss, it is referred to as critical limb ischemia. Despite advances in detection and treatment of limb ischemia, morbidity is high. Ulceration and infection are complications associated with the low-oxygen state of ischemic tissues. Early diagnosis and treatment are critical to ensure limb salvage and a functional outcome for the patient.
Case Presentation
A 55-year-old man with a history of hypertension, poorly controlled type 2 diabetes, tobacco use, and high cholesterol comes to the ED complaining of left foot and leg pain. He initially only had mild discomfort on and off for a few weeks, but the pain has become progressively more intense over the past 24 hours. The pain has kept him awake all night, radiates from the sole of his foot to his leg, and is now 10/10. He also reports black spots on several of his toes. In the past, he had a non-healing ulcer on his other foot that resulted in several surgeries and, ultimately, an amputation below the knee. The year before the amputation, he had a femoral-popliteal bypass.
About a week ago, he was seen in your ED for mild swelling and discomfort of the leg. He reports that he had a deep vein thrombosis study at that time that was unremarkable and he was started on furosemide.
Vitals: Blood pressure 189/83 mmHg, heart rate 84 beats per minute, respiratory rate 16, pulse ox 94% on room air, temperature 98.1° F.
Examination: A chronically ill-appearing man is in mild distress. His lungs are clear bilaterally and his heartbeat is regular. Examination of his left lower extremity reveals a pale left leg and foot; toes appear dusky, and the patient is unable to move them. He is able to dorsiflex and plantarflex weakly at the ankle. The dorsalis pedis and posterior tibial pulses are not palpable. Capillary refill is > 5 seconds at the toes and through the entire foot. A mottled, blue-gray color extends the length of the entire lower extremity to the knee. He has sensation only to pinprick from the knee down.
The right leg has a clean, dry amputation site below the knee and is well-perfused. A chart review reveals that his previous amputation was related to an ulcer that was the result of a chronic perfusion deficit. The remainder of his exam is unremarkable.
Case Resolution: In this male patient with multiple historical risk factors as well as a previous ischemic event, there is a very high risk of recurrent acute limb ischemia.
Considering the Diagnosis of Limb Ischemia
Ischemia should be considered in any patient with a complaint of limb pain. (See Table 1.) A brief assessment of perfusion and pulse presence is all that is needed in most patients in the absence of risk factors. (See Table 2.) The clinician should obtain a detailed history, including review of previous ischemic events, such as acute myocardial infarction (MI), mesenteric ischemia, or limb ischemia. A history of atrial fibrillation, coagulopathy, or vasculopathy all increase the risk of limb ischemia. Identification of ischemia is important since the mortality rate is 6-12% — the same as the acute MI inpatient mortality rate.1 The high risk of death and limb loss associated with limb ischemia makes it critical to identify the condition early in the ED course. The amputation rate is 6% if revascularization is performed within 12 hours of onset and 20% if performed more than 24 hours later.2,3
Table 1. Differential Diagnosis of Acute Limb Ischemia |
|
|
Diagnosis |
How It Differs From Acute Arterial Occlusion |
|
Osteoarthritis |
• Worse at the end of the day • No pallor |
|
Claudication |
• Worse after walking, better with elevation • History of edema/venous stasis |
|
Spinal stenosis |
• Pain and weakness, worse with standing |
|
Radiculopathy |
• Sharp, often radiating pain from proximal to distal with standing or walking, history of back problems |
|
Chronic compartment syndrome |
• In athletes with large calf muscles; pain is worse with exercise |
|
Neuropathy |
• Usually bilateral, can be difficult to differentiate due to decreased pulses and skin discoloration |
|
Acute compartment syndrome |
• Pain worse with muscle palpation or passive stretch and usually accompanied by leg edema |
|
Raynaud phenomenon |
• Patient usually has history of the disease • Proximal pulses usually present; symmetric extremity involvement |
|
Extensive deep vein thrombosis (phlegmasia cerulea dolens) |
• Venous pressure increased so severely that perfusion is impaired to extremity • Leg swelling and cyanosis generally present |
|
Aortic dissection |
• Pulselessness and loss of sensation of one or more limbs associated with trunk pain (chest or back) |
Table 2. Full Vascular Exam |
|
|
Upper Limb Pulse Check Locations |
Lower Limb Pulse Check Locations |
|
Radial Ulnar Brachial Axillary |
Dorsalis pedis Posterior tibial Popliteal Femoral |
Epidemiology
The risk factors for acute limb ischemia include male sex, smoking, diabetes, hyperlipidemia, hypertension, and age > 60 years.4
Smoking and diabetes are the two most important risk factors for developing acute limb ischemia. Individuals with these conditions are at higher risk at a much younger age than patients without risk factors. The majority of patients with acute limb ischemia also have concurrent coronary or cerebrovascular disease.
Although arterial disease occurs most frequently in the legs, it also can affect the arms. In these instances, one should consider vasculitis in the differential. A classic example would be Takayasu’s arteritis, which typically presents with a pulseless upper extremity in women 20 to 30 years of age.
Etiology
Acute limb ischemia can occur in several ways. A thrombus can occlude a native vessel, graft, or stent. An embolus can dislodge from a more proximal vessel. Trauma to an artery, arterial dissection, or artery stenosis from chronic vasculitis can lead to acute limb ischemia.
Thrombotic occlusion is the most common cause of acute extremity ischemia. In a native vessel, atherosclerotic plaques are the sites of most thrombi. In a bypassed vessel, thrombi tend to develop around areas of kinks and other irregularities. In prosthetic vessels, it can occur even if there is no abnormality. Additionally, thrombophilic disease (i.e., malignancy, hyperhomocysteinemia, and coagulopathies) may lead to the development of thrombosis in otherwise normal limbs.
The most common locations for thrombotic occlusions are:
- common femoral artery;
- popliteal artery;
- any bypass graft.
Thrombotic disease frequently presents as intermittent claudication. In the lower limbs, thrombosis is the underlying disease process in more than 80% of cases. In the upper limbs, thrombosis is the cause of acute ischemia only 50% of the time.
Ischemia that occurs in a limb affected by atherosclerosis usually is less severe than in a limb subject to embolic disease, since limbs with chronically narrowed vessels generally develop collateral circulation over time.2
Arterial dissections occur when the inner layer or layers of a weakened artery tear and blood gets in between them, causing aneurysmal dilatation of the weakened remaining layers. This can lead to thrombus formation and stenosis of the artery. The resulting thrombus also can be a nidus for emboli. Dissections can be the result of prolonged untreated hypertension or connective tissue disorders, such as Marfan syndrome.
Trauma can occur as a complication of vascular or cardiac diagnostic and interventional procedures. Less than 1% of acute cardiac catheterization procedures result in trauma to a vessel, but almost all require surgical consultation.5 Blunt or penetrating injury to a limb can cause acute ischemia. Additionally, arterial injection can occur either intentionally or accidentally as a consequence of medical treatment (e.g., arterial blood gas) or by those who misuse intravenous drugs. Injection into an artery can cause vasospasm, aneurysm formation, thrombosis, or infectious arteritis. Components of the injected substance can crystallize within the vessel and can lead to obstruction and gangrene.
Emboli causing limb ischemia usually originate in the heart as a consequence of atrial fibrillation, mural thrombus, or debris from valves. Embolic occlusions most commonly present with a sudden onset of pain in a specific territory of arterial flow. The incidence of embolic limb occlusions has decreased with the decreasing incidence of rheumatic disease. A common finding in embolic disease is blue toe syndrome, which involves the sudden appearance of painful cyanotic plantar foot and/or toe(s). The pedal pulses are normal in this condition. The underlying cause is digital arterial emboli from a more proximal vessel. This can be the presenting complaint in the case of popliteal artery aneurysm or atherosclerosis.
Vasculitis syndromes, such as Takayasu’s arteritis, cause arterial occlusion and subsequent limb ischemia through inflammation and subsequent fibrosis of arteries.6
Clinical Features
Patients with acute ischemia of a limb present with a constellation of symptoms referred to as “the six Ps”:
- pain;
- pallor;
- paresthesias;
- paralysis;
- pulselessness; and
- poikilothermia or “polar” (limb feels cool/cold).
Pain frequently is one of the first signs of ischemia. In the case of chronic peripheral vascular disease, pain may be intermittent and worse with ambulation (intermittent claudication). Pallor often is the first change in the skin. Mottling and blotchiness soon follow; then, petechiae and blisters develop as the oxygen tension in the skin surface decreases and cell death occurs.
Changes in sensation occur as ischemic injury to the nerves progresses. Increased or decreased sensation and paresthesias occur as a result of nerve injury. Sensation to light touch and proprioception usually disappear before deep sensation. The presence of light touch or pinprick is a positive prognostic sign and suggests viability of a limb. Sudden loss of a pulse in a previously normal extremity suggests an acute embolic source. Limbs with peripheral vascular disease may be chronically pulseless, but the abnormalities of skin color, temperature (poikilothermia/polar), and sensation may suggest an acute-on-chronic ischemic event. Paralysis of muscle is one of the last signs to occur and often is coupled with a complete loss of sensation. When these symptoms occur, they are indicative of prolonged or severe ischemia and nonviability.
Communication With the Surgeon
It is critical to involve a surgeon early in the management of a patient with suspected limb ischemia to assist in the coordination of care. Clearly expressing concern for the diagnosis, describing the patient’s examination findings, and communicating the level of concern with language commonly used among surgeons will help provide effective care for the patient. Common pitfalls in the management of patients with acute limb ischemia include failure to involve the surgical team soon enough and failure to complete a full exam that supports the diagnosis.
The Rutherford classification categorizes the severity of acute limb ischemia and helps guide decision-making about testing and treatment.7 Stage I limbs are viable limbs that are not immediately threatened. There is no sensory or motor function loss, and both arterial and venous Doppler signals are audible. Stage IIa limbs are threatened marginally and are salvageable with prompt treatment. Sensory loss, if present, is limited to the toes, and there is no loss of motor function. Arterial Doppler signals are weak to inaudible. Stage IIa limbs still have some perfusion present and usually can be managed with endovascular therapies, generally catheter-directed thrombolysis. The catheter is placed and the thrombolytic is infused over 24-48 hours, so patients with severe ischemia related to a complete blockage are not candidates for this treatment.8 Stage IIb limbs are threatened immediately but potentially are salvageable with immediate revascularization. These patients are the most dependent on time to reperfusion. Sensory loss is present beyond the toes, and weakness is present. There is no audible Doppler arterial pulse present, but a venous pulse is heard. These patients generally have to be managed in the operating room. Stage III limbs are irreversibly damaged, with presence of major tissue loss, profound anesthesia, and paralysis of the limb. These limbs are irreversibly damaged and require amputation.
Clearly messaging the stage of disease and the urgent need for consultation is necessary. Often, consultations are requested only after extensive testing and diagnostic confirmation is performed. In the case of an acute limb ischemia event, time is limb. Consultation should be requested as soon as the complete focused exam is performed to reduce the risk of tissue loss and resulting loss of function.
ED Evaluation
The history and physical examination are critical to making the diagnosis of acute limb ischemia. Critical exam findings include:
- Cardiac: examine for atrial fibrillation and acute murmurs;
- Neurological: sensory and motor exam should be performed on all extremities;
- Extremities: search for signs of peripheral artery disease (i.e., shiny skin, hair loss, skin ulceration, skin/soft tissue necrosis, atrophy of muscle, and decreased pulse strength compared to the other limb).
Frequently, a pulse will be palpable in an unaffected extremity and absent in the affected limb. If the pulse is not palpable, a Doppler device should be used to determine if one is present. If no pulse is present and the patient has any signs or symptoms of ischemia, the patient should be referred immediately to vascular surgery. However, if arterial blood flow is present on Doppler, an ankle-brachial index or wrist-brachial index should be obtained as per current American Heart Association (AHA)/American College of Cardiology (ACC) guidelines.9
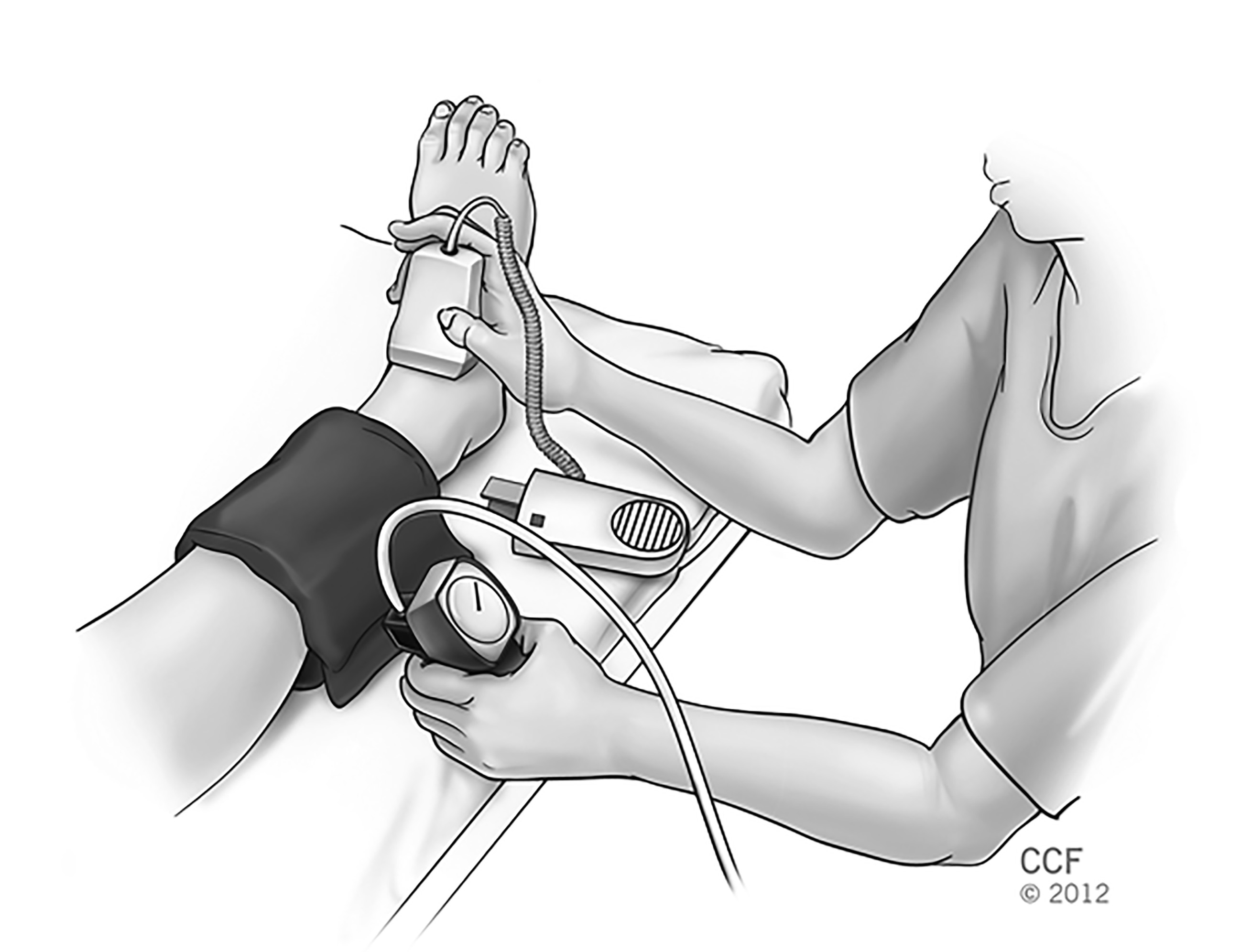
How to Measure Ankle-brachial Index and Wrist-brachial Index
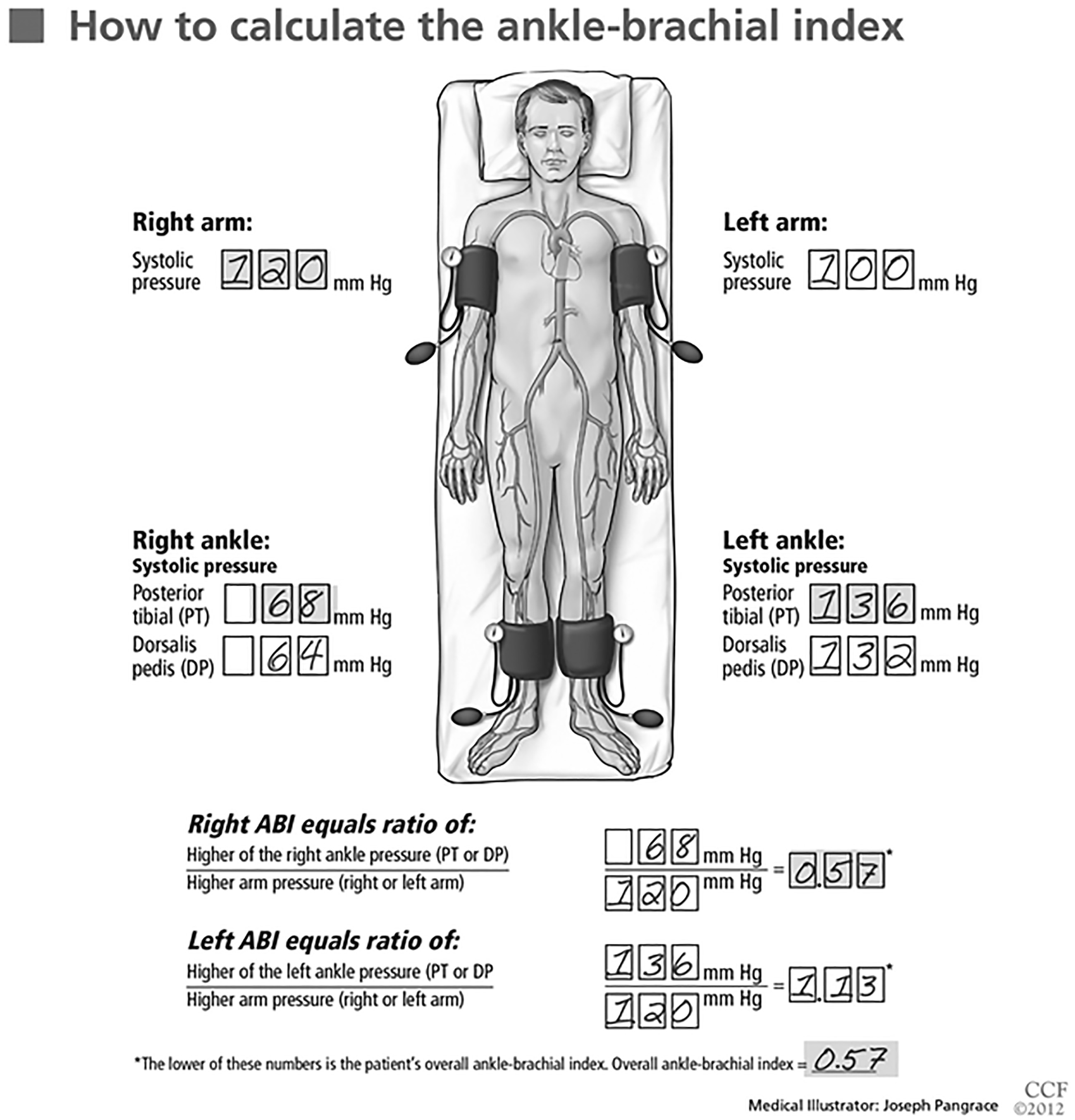
The ankle-brachial index is the ratio of the systolic blood pressure of the foot to the highest systolic pressure in either arm. The cuff should be placed above the elbow to measure the arm pressure and above the malleolus to measure the foot pressure. (See Figure 1.) Blood pressure measurements are made in the supine position after a period of five minutes of rest. The brachial pulse and the dorsalis pedis pulse are used for the ankle-brachial index, whereas the radial pulse or the axillary pulse and brachial pulse are used for the wrist-brachial index. A normal ratio rules out occlusive arterial disease in the extremity of interest. (See Table 3.) The ankle-brachial index is associated closely with cardiovascular risk factors and outcomes. Patients with an ankle-brachial index < 0.9 are at a higher risk of claudication and mortality. Artificially high ankle-brachial indexes are associated with medial calcinosis of the artery, which can be seen in patients with diabetes.10 (See Figure 2.)
Figure 1. Obtaining Ankle-brachial Index at the Dorsalis Pedis Pulse Point |
 |
|
Reprinted with permission, Cleveland Clinic Center for Medical Art & Photography © 2012-2018. All Rights Reserved. |
Table 3. Ankle-brachial Index Ratio |
|
|
Ankle-brachial Index Ratio |
Diagnosis |
|
< 0.9 |
Abnormal |
|
0.91 to 1.0 |
Borderline |
|
1.0 to 1.39 |
Normal |
|
> 1.4 |
Noncompressible vessel |
Figure 2. Calculation of Ankle-brachial Index |
 |
|
Reprinted with permission, Cleveland Clinic Center for Medical Art & Photography © 2012-2018. All Rights Reserved. |
Approach to Diagnostic Testing in the ED
The most useful diagnostic tool for determining presence of acute limb ischemia is the clinical evaluation. The current AHA/ACC guidelines recognize that imaging and extensive testing is unnecessary and may harm the patient. A focused exam and handheld Doppler study at the bedside are sufficient to make the diagnosis in most cases.9
If the diagnosis is not clear, multiple factors may drive the decision to undergo confirmatory testing. The decision to order testing depends of the severity and location of the occlusion, the patient’s underlying comorbidities, the availability of vascular surgery or need for transfer, and the recommendations of the consulting surgeon. Some vascular surgeons may prefer to have the patient transferred immediately to their receiving facility without additional imaging, especially if the patient is in a free-standing ED or hospital without vascular surgery coverage. With Rutherford stage IIb or III ischemic injury, delays in treatment may worsen patient outcomes. Therefore, it is critical to involve a surgeon early in the course of treatment with obviously threatened extremities.
Laboratory Testing
Although laboratory tests generally will not confirm the diagnosis, they can be helpful in preparing for operative intervention, for determining whether an embolic event took place, and for evaluating risk assessment post-operatively. Orders for the following tests should be considered:
- Complete blood count, coagulation studies: This test is necessary to prepare for aspirin and heparin administration.
- Chemistry and renal panel: Many patients have coexisting renal disease, and angiography is associated with exposure to contrast agents; hyperkalemia can present following revascularization.
- Creatine phosphokinase and myoglobin: Rhabdomyolysis is a risk with myonecrosis and revascularization. As the products of cell death are released into the bloodstream following reperfusion of a threatened limb, severe critical illness can result. About one-third of acute limb ischemia-associated deaths are due to hyperkalemia and acidosis, both of which are complications of revascularization.11
Imaging
Common ED diagnostic testing options include duplex ultrasound, computed tomography angiography (CTA), and magnetic resonance angiography (MRA).
- Duplex ultrasound: The advantages of duplex ultrasound are that it is inexpensive, is noninvasive, does not require nephrotoxic contrast agents, and is used widely as a first-line examination. Ultrasound can determine the level of occlusion, patency of the distal vessel, echogenicity of the thrombus, and quality of the vessel wall at the level of the occlusion. In very experienced hands, duplex ultrasound may provide imaging data that is as useful for planning surgery as data coming from angiography.12 It has a specificity of 95% and sensitivity of 88% for hemodynamically significant lesions (> 50% stenosis/occlusion). It also has a high sensitivity for upper extremity occlusive disease.11 The disadvantages of duplex ultrasound are that it is operator-dependent, provides incomplete information about vessels, and may miss occlusions present in structures below the knee.
- Computed tomography angiography (CTA): CTA has high sensitivity (94%) and specificity (100%) in most studies. It can identify atherosclerotic plaques, collateral arteries, and abnormities of vessels (aneurysms, dissections). It can be used safely for patients with pacemakers and claustrophobia.
- The disadvantages of CTA include the fact that use of intravenous (IV) contrast can be associated with nephrotoxicity or allergic reactions.13
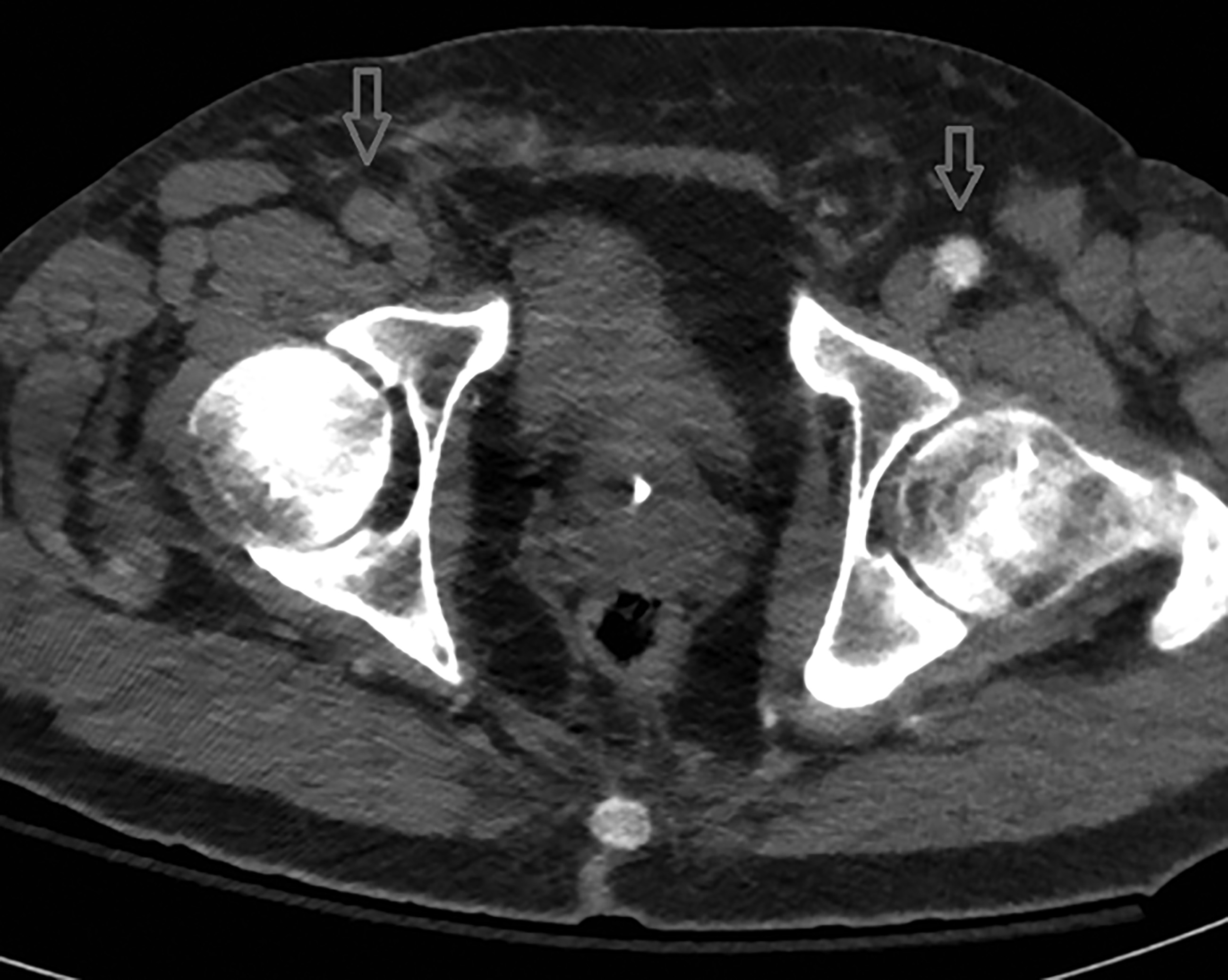
- Magnetic resonance angiography: MR angiography has no iodinated contrast or radiation risks, but sensitivity and specificity are very high, similar to CTA. (See Figure 3.)
The disadvantages of MRA are that it has limited availability in many centers and it is a time-consuming study that may cause delays in definitive care. Patients with pacemakers/defibrillators not compatible with MRA cannot be imaged using this modality.
Figure 3. CT Angiography: Common Femoral Artery Occlusion Present on Right |
 |
|
Source: Image used with permission from: Paul Schoenhagen, MD, Cleveland Clinic Robert and Suzanne Tomsich Department of Cardiovascular Medicine |
Comorbid Disease States
Since other vascular and nonvascular comorbid diseases generally are present in patients with an acute limb ischemia event, laboratory and radiographic testing may be needed in addition to those required for anticoagulation and surgery. Consider these diagnoses in any patient with suspected or proven acute limb ischemia:
- infection/gangrene;
- abnormal glucose levels;
- myocardial infarction;
- arrhythmia;
- renal disease;
- dehydration and low-flow states;
- congestive heart failure.
Treatment
Treatment options for acute limb ischemia depend on the extent of disease, the location of the lesion, how long it has been present, and the appropriateness of surgery for a given patient. In addition to patient-specific factors, treatment also may be predicated upon local resources. According to the 2012 American College of Chest Physicians guidelines, once the diagnosis of acute limb ischemia has been made by history and physical examination, anticoagulant therapy should be started with a heparin bolus and infusion.9 Heparin prevents the thrombus from growing larger and reduces the chance that an additional clot will form in areas of relatively stagnant flow. The decision to start heparin should not be delayed by obtaining imaging or other diagnostics.
The goals of therapy in the ED are:
- Maintain a reasonable temperature for the affected limb. The lack of sensation in an ischemic limb may make a patient susceptible to extremes of cold or heat. Keep the limb covered in a blanket without heat or ice in close contact with the limb.
- Initiate heparin: 80 units/kg bolus followed by 18 Units/hour infusion.
- Administer aspirin: 325 mg oral dose.
- Provide pain control: to ensure patient comfort and compliance with treatment regimen.
As soon as an ischemic limb is suspected, critical tasks include:
- determining the Rutherford classification/severity of disease;
- communicating the urgency of the patient’s issue to the vascular surgeon;
- arranging transfer to a facility capable of definitive care.
Operative Interventions
Often, multiple techniques will be used to achieve resumption of blood flow. The revascularization strategy employed depends on the degree and location of the occlusion, the presence of collaterals, the patient’s comorbid illnesses and risk of surgery, and the local availability of treatment options. Although the emergency physician usually will not determine the treatment plan, knowledge of the options is helpful to determine appropriateness of transfer and to inform patients of their options.
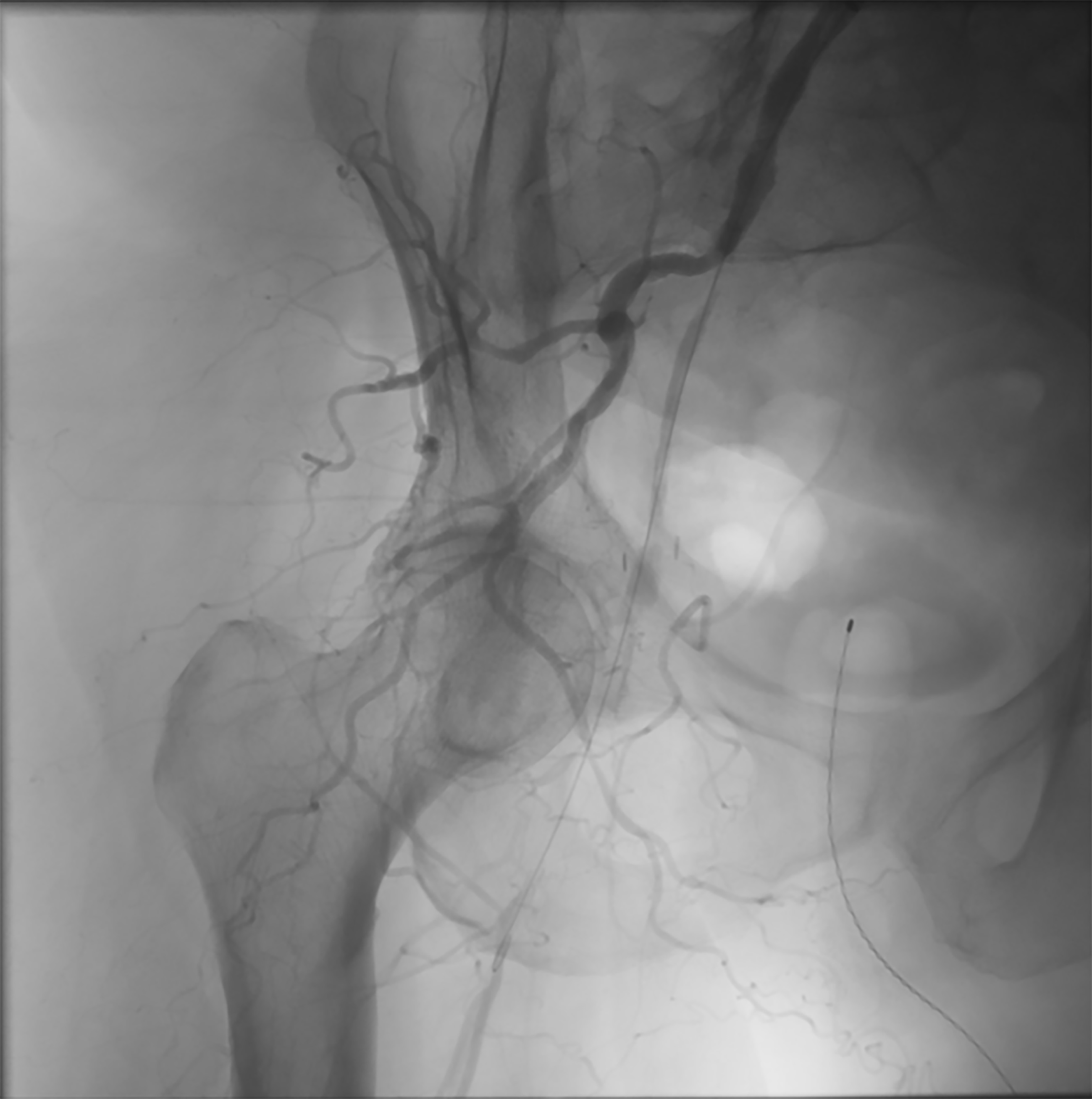
Catheter-directed Thrombolysis: Landmark Studies (See Figure 4.)
In 1994, the researchers for the STILE (Surgery versus Thrombolysis for Ischemia of the Lower Extremity) study compared traditional revascularization to catheter-directed thrombolytic therapy in patients with occlusion of a bypass graft or native artery. They found that patients with acute occlusion (< 14 days) treated with thrombolysis had modestly improved amputation-free survival and shorter hospitalizations, but that chronically ischemic limbs were better treated with surgical revascularization.14
Figure 4. Intraoperative Angiogram Showing Occlusion of Distal Right External Iliac Artery |
 |
In the 1998 TOPAS (Thrombolysis or Peripheral Artery Surgery) study, investigators demonstrated that performing thrombolysis as part of an initial strategy to revascularize an acutely ischemic limb reduced the need for surgery without increasing mortality.15 The TOPAS trial researchers demonstrated the increased risk of hemorrhage with urokinase, a risk that was more than doubled when heparin was added.
The 2016 AHA/ACC PAD guidelines recommend catheter-based thrombolysis for patients with acute limb ischemia and a salvageable limb.9
Percutaneous mechanical thrombectomy can be used as an adjunct to thrombolysis in patients with acute limb ischemia and a salvageable limb. Several devices have been developed to perform this procedure. Risks of use include bleeding, distal embolization of the clot with downstream occlusion, and damage to vessels.
Surgical thromboembolectomy is an open procedure performed to remove a clot from an artery. Often, this procedure is performed along with a percutaneous approach to completely remove a clot. This is another alternative treatment for management of an acutely occluded salvageable limb.
Surgical Bypass/Balloon Angioplasty: Traditional surgical approaches provide the patient with better anatomic patency and longer-lasting results at the cost of increased risk of morbidity and mortality and increased resource use. In the BASIL trial (Bypass versus Angioplasty in Severe Ischemia of the Leg), researchers demonstrated that both bypass surgery and balloon angioplasty had equivalent amputation-free survival when used as a first-line treatment option. In the short term, surgery had a greater cost than angioplasty. In the event of an angioplasty failure, surgery still could be a viable option.16
Amputation is the only treatment for non-salvageable limbs. Clinically, these limbs are insensate, immobile/rigid, and may have signs of gangrene. Individuals with terminal illness or who have chronic immobility or flexion contractures may benefit from primary amputation.17
Claudication and Critical Limb Ischemia
Claudication and critical limb ischemia also are included in the differential diagnosis of limb pain. Patients with these diagnoses may present to the ED with limb pain and signs of abnormal extremity perfusion. Although patients may have pain with claudication and critical limb ischemia, the duration of symptoms generally is longer and the pain is less intense than with acute limb ischemia.
Claudication is pain that is increased with exercise and relieved with rest and is caused by arterial flow obstruction. The first-line treatment for claudication is medical management, focusing on smoking cessation, antiplatelet therapy, statins, and treatment of hypertension. Angiotensin-converting enzyme (ACE) inhibitors are the preferred medication to treat hypertension, as this drug class reduces cardiovascular event risk in patients with lower extremity peripheral artery disease (PAD).18 Multidisciplinary team management of patients with diabetes who have PAD is critical for preventing microvascular complications in this population. Meticulous foot care is necessary to prevent ulcer development and tissue loss that can lead to infection.
ED management of claudication should focus on ruling out acute ischemia, connecting patients to outpatient services, and educating them on the need for ongoing medical treatment. Tobacco cessation counseling can be done in a few minutes in the ED setting. About 80% of patients who have PAD are current or former smokers.19 Between 15-20% of patients with claudication will progress to critical limb ischemia. Early detection and aggressive management of claudication is necessary, since the mortality rate is 40% within two years for those who progress to high-grade ischemia and have an amputation.9
Critical limb ischemia is progressive loss of blood flow that increases the risk of tissue loss in the extremity. Rest pain, ulcers, or gangrene in the context of proven arterial insufficiency characterize critical limb ischemia. While acute limb ischemia occurs quickly, over hours to days, critical limb ischemia is more insidious. Although there usually is a slower onset of disease with critical limb ischemia, the condition can be complicated by limb-threatening disease that can lead to sepsis, death, or loss of limb. The spectrum of disease can vary from small ulcers to severe myonecrosis.9
Critical limb ischemia is characterized by pain at rest that primarily affects the foot. Patients may complain of a need to awaken at night to dangle the leg over the side of the bed or to walk a few steps. They may refuse to elevate the leg because of pain. Some patients, especially those with diabetes, may have numbness instead of pain. The exam is significant for absent ankle pulses, redness that is worse with the limb in a dependent position, hair loss, and increased capillary refill time. Erythema that persists when the limb is elevated suggests the presence of infection.
Once confirmed, critical limb ischemia is treated with revascularization. Intervention usually is needed to relieve pain and prevent loss of function of the extremity. A revascularization procedure is indicated for pain that is persistent despite risk factor modification and medical treatment, or for the presence of a nonhealing ulcer. MRA or CTA of the extremity is indicated in stable patients with suspected critical limb ischemia. Amputation may be the only treatment option available for patients with intolerable pain, spreading infection, or severe systemic illness.
Case Presentation
A 64-year-old man with diabetic neuropathy, tobacco use, hypertension, and renal insufficiency presents to the ED with severe, unrelenting pain in the left foot ongoing for several days. He had noticed some breakdown of skin on the plantar surface of the forefoot a few weeks ago, and was referred for further care, but never followed up. Over the past few hours, the color of his foot has changed to a dusky purplish-brown. Small, dark blisters have developed over the entire foot and began to track up his leg during the ED evaluation. He feels somewhat “hot” and nauseated.
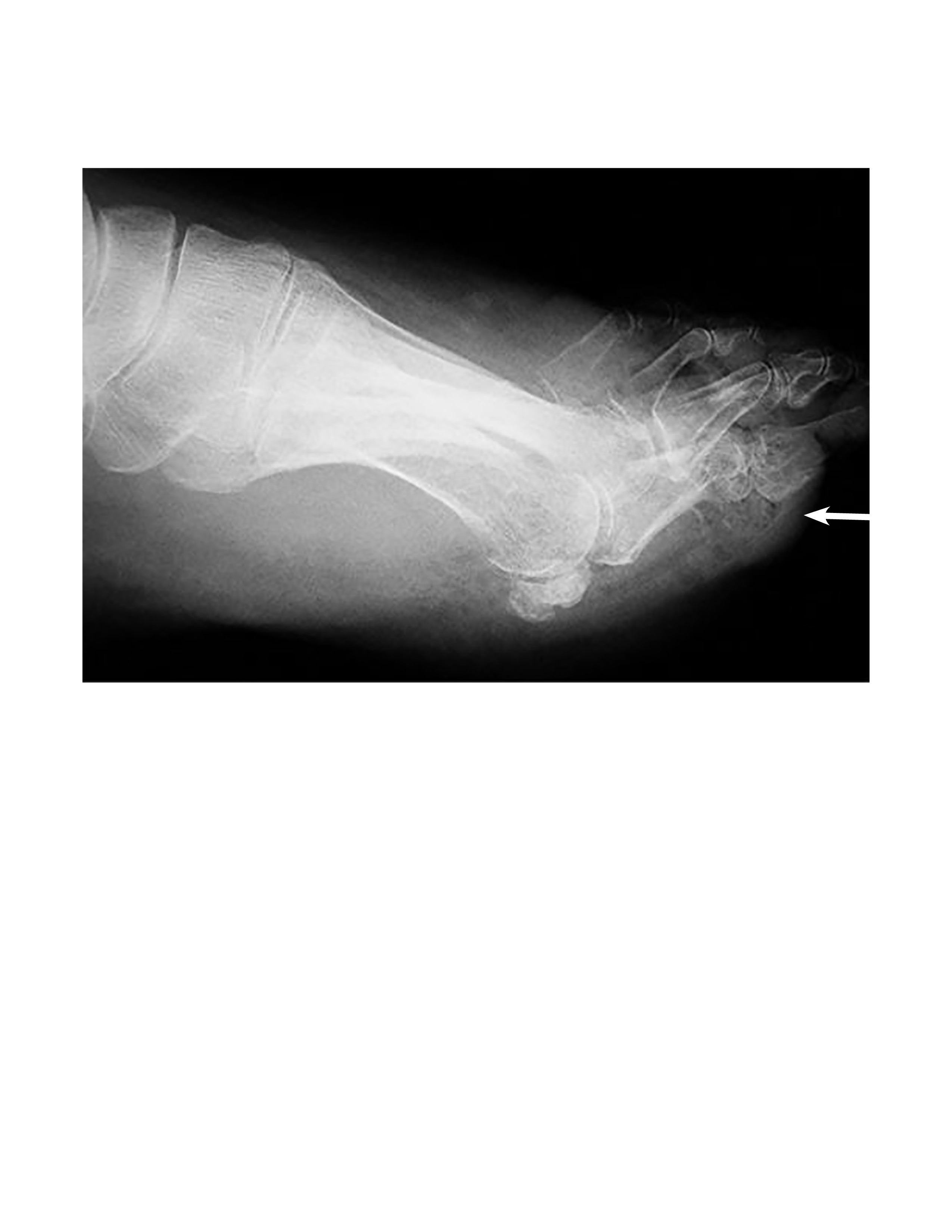
Vitals: Blood pressure 97/54 mmHg, heart rate 115, respiratory rate 28, pulse ox 93% on RA, temperature 101.8° F. (See Figure 5 for the radiograph of the patient’s foot.)
Figure 5. Radiograph of Foot |
 |
|
Source: Image used with permission from: Mark Hardy, DPM, FACFAS |
What Is the Optimal ED Management of a Gangrenous Limb?
Infection in a chronically ischemic limb can take on many forms, from cellulitis to gas gangrene. Although cellulitis is characterized by warmth and erythema with mild pain, gangrene and other necrotizing infections are associated with a high risk of limb loss, sepsis, and death.
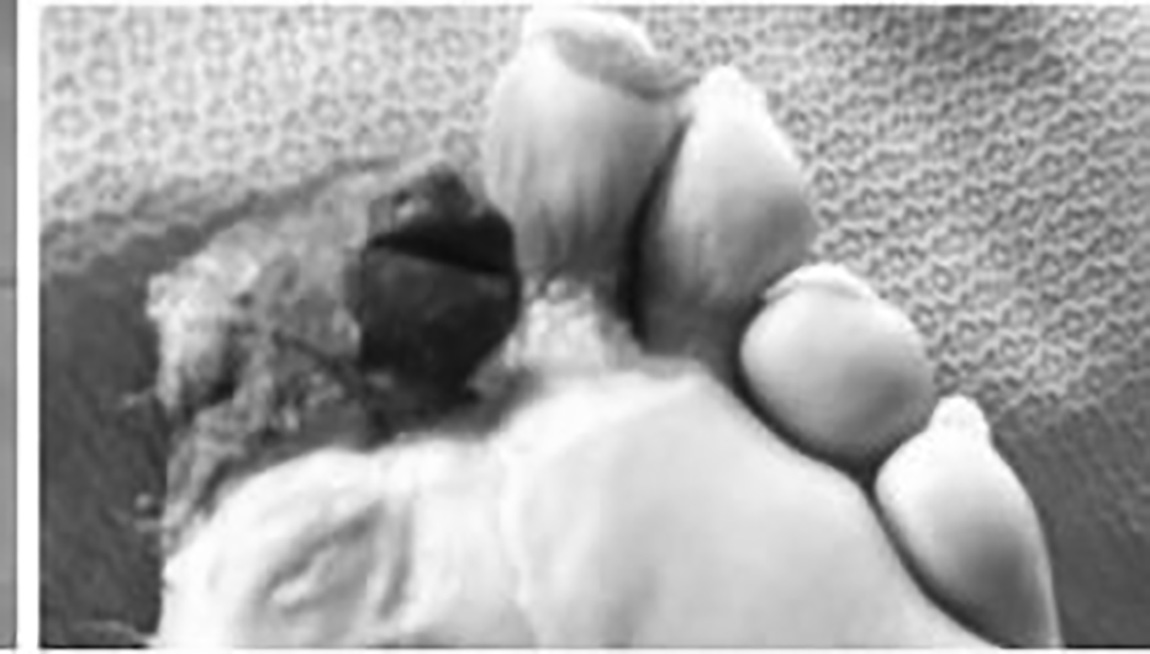
Gangrene is either ischemic or infectious. Dry gangrene is a form of coagulation necrosis with dry, wrinkled skin. In later stages, the skin color darkens. Little tissue liquefaction is present, and there is no infection. (See Figure 6.)
Figure 6. Dry Gangrene of the Great Toe |
 |
|
Source: Image used with permission from: Mark Hardy, DPM, FACFAS |
In contrast, wet gangrene demonstrates copious tissue liquefaction, with warm, red, swollen tissue and severe pain. Wet gangrene commonly complicates diabetic foot infections. The history and physical exam usually are enough to establish a diagnosis. Laboratory investigation to assess for the presence of sepsis and uncontrolled diabetes is warranted. Imaging with plain films should be considered to rule out gas in the tissues. Most of these presentations are polymicrobial (gram-positive, gram-negative, and anaerobic) and require broad-spectrum antibiotic coverage. Treatment options recommended by the Infectious Diseases Society of America (IDSA) include:20
- Piperacillin-tazobactam (3.375 g every 6-8 hours IV) and vancomycin (15 mg/kg IV twice daily);
- Imipenem (1 g every 6-8 hours IV);
- Meropenem (1 g every 8 hours IV);
- Ertapenem (1 g IV daily);
- Cefotaxime (2 g every 6 hours IV) plus metronidazole (500 mg IV every 6 hours) OR clindamycin (600-900 mg IV every 8 hours IV).21
Wet gangrene is a surgical emergency that requires urgent consultation. Obtaining laboratory or imaging results should not delay urgent consultation when wet gangrene is suspected on examination.
Gas gangrene is a bacterial infection frequently related to the presence of exotoxin-producing bacteria, associated with sepsis and systemic illness. It is much less common in the United States than in developing countries, but it should be considered in the differential diagnosis of limb ischemia and infection.
The infectious causes of gas gangrene are subdivided into clostridial (gram-positive) and non-clostridial (gram-negative or mixed) subgroups. These types of bacteria are present in the environment as well as in the human gastrointestinal tract, vagina, and skin. Bacteria associated with gangrene can enter a wound, grow in low-oxygen environments within necrotic tissue, and secrete toxins that further damage soft tissue.22
Alpha toxin is the primary agent associated with morbidity and mortality in gas gangrene. It causes hemodynamic instability, hemolysis, and anemia. Hypotension from alpha toxin may be challenging to reverse because of the toxin’s negative inotropic effects. Bacteremia, sepsis, and multisystem organ failure are common with gas gangrene, and rapid decline is imminent, even with appropriate antibiotic administration. Aggressive resuscitation and early surgical consultation are the cornerstones of therapy.
Gas gangrene progresses over a brief time frame, from hours to days. In the earliest stages, the skin may be bronze, but intact and without bullae. The first symptom usually is pain out of proportion to exam. Pain is relatively sudden in onset, localized, and severe, and the skin rapidly develops tense edema along with brown or purplish blisters. Foul-smelling fluid and gas may emanate from injured tissue. The presence of crepitance on palpation is a highly specific and sensitive finding.
Gas gangrene should be suspected when the following are present:
- History of previous trauma or surgery;
- Muscle swelling, severe pain, or edema;
- Discoloration of wound;
- Presence of thin, gray, “dishwater” discharge;
- Blood-filled, > 5 mm in size, hemorrhagic bullae;
- Unpleasant smell;
- Crepitance.
Immediate surgical consultation should be obtained when gas gangrene is suspected. Early surgical intervention is performed by thoroughly removing all necrotic tissue and releasing swollen compartments via fasciotomy. Multiple trips to the operating room may be necessary to clear all of the compromised tissue. Following radical excision, delayed closure and repair may be pursued. Even with rapid recognition and treatment, the mortality rate is 25%, but the mortality rate is 100% in untreated patients.23
The primary method of establishing the diagnosis of gas gangrene is by physical examination. Laboratory studies may demonstrate evidence of infection, but none are diagnostic. CT or MRI will show gas present in the soft tissues, which also should prompt emergent surgical consultation. Diagnostic testing does not supplant adequate surgical inspection. Survival is increased with earlier surgical intervention.
Early, aggressive resuscitation is key. Circulating bacterial toxins cause damage to small vessels, with leak of fluid into tissues. Fluid requirements may be very high (10-12 liters/day of crystalloid). Hemolysis and disseminated intravascular coagulation may complicate resuscitation efforts.
Bacteria other than Clostridium species can produce tissue gas, so for an undifferentiated case, broad-spectrum coverage is indicated. Initial coverage with vancomycin (15 mg/kg twice daily IV) plus piperacillin/tazobactam (3.375 g every 6 hours IV) or ampicillin/sulbactam (3 g every 6 hours IV) or monotherapy with a carbapenem, according to the IDSA guidelines.22 Once tissue cultures confirm a diagnosis of a clostridial infection, penicillin (2-4 million units every 4-6 hours IV) plus clindamycin (600-900 mg every 8 hours IV) can be started. Hyperbaric oxygen is not recommended because of lack of benefit and potential delay in surgical treatment.22
Pitfalls
Potential pitfalls include:
- failure to identify acute limb ischemia and to communicate the urgency of illness clearly to the receiving vascular surgery team in a timely manner;
- misidentification of acute limb ischemia as a diagnosis that is not time-sensitive, like DVT or cellulitis;
- failure to document the presence of absence of pulses in patients at risk for acute limb ischemia;
- failure to administer aspirin/heparin in the ED to patients with acute limb ischemia;
- failure to refer patients with critical limb ischemia for urgent revascularization;
- failure to treat gangrene as a serious, surgical emergency;
- inappropriate or delayed antibiotic treatment in patients with suspected gangrene.
Pearls
Clinical pearls to remember include:
- Upon identification or strong suspicion of the diagnosis of acute limb ischemia, start heparin as soon as contraindications are ruled out.
- Communicate clearly with surgical team that the patient has limb ischemia, ideally by using the Rutherford classification.
- Do not delay referral for acute limb ischemia or gangrene to vascular surgery pending imaging or lab tests.
- Be sure to consider and document comorbidities to help the surgeon plan for future procedures.
- Carefully counsel all patients with claudication about the risk for progression to critical limb ischemia, and document in your note.
- Consider vasculitis in patients with upper extremity occlusive disease.
- Time is limb.
Conclusion
Limb ischemia should be considered in the differential diagnosis of severe limb pain as a result of the high-risk nature of the disease. A thorough physical examination that includes motor, sensory, and vascular exams is critical to making a rapid diagnosis. Imaging can be helpful when the diagnosis is unclear, but when Doppler pulses are not audible, an urgent contact with the vascular surgeon is needed. Clear communication of the exam and Rutherford classification is helpful to communicate the urgency of the patient’s condition. Prompt revascularization can help salvage limbs and save lives. Unusual presentations of limb pain (especially upper limb ischemia in a young, Asian woman) should lead to concern for vasculitis. Acute limb ischemia has a mortality rate that is the same as acute myocardial infarction and should be treated with a similar urgency. Gangrene can occur as a result of acute or critical limb ischemia and should be treated aggressively with antibiotics, resuscitation, and emergent referral. Appropriate care can lead to a better outcome even if amputation is required.
The authors thank Paul Schoenhagen, MD, and Mark Hardy, DPM, for their assistance with images, and Irene Szentkiralyi for assistance with research.
REFERENCES
- Eliason JL, Wainess RM, Proctor MC, et al. A national and single institutional experience in the contemporary treatment of acute lower extremity ischemia. Ann Surg 2003;238:382-389.
- Norgren L, Hiatt WR, Dormandy JA, et al. Inter-society consensus for the management of peripheral arterial disease (TASC II). J Vasc Surg 2007;45(Suppl S):S5-S67.
- Kuukasjarvi P, Salenius JP. Perioperative outcome of acute lower limb ischaemia on the basis of the national vascualr registry. The Finnvasc Study Group. Eur J Vasc Surg 1994;8:578-583.
- Gabel J, Jabo B, Patel S, et al. Analysis of patients undergoing major lower extremity amputation in the vascular quality initiative. Ann Vasc Surg 2018;46:75-82.
- Applegate RJ, Sacrinty MT, Kutcher MA, et al. Trends in vascular complications after diagnostic cardiac catheterization and percutaneous coronary intervention via the femoral artery, 1998 to 2007. JACC Cardiovasc Interv 2008;1:317-326.
- Callum K, Bradbury A. ABC of arterial and venous disease: Acute limb ischaemia. BMJ 2000;320:764-767.
- Rutherford RB, Baker JD, Ernst C, et al. Recommended standards for reports dealing with lower extremity ischemia: Revised version. J Vasc Surg 1997;26:517-538.
- Morrison HL. Catheter-directed thrombolysis for acute limb ischemia. Semin Intervent Radiol 2006;23:258-269.
- Gerhard-Herman MD, Gornik HL, Barrett C, et al. 2016 AHA/ACC guideline on the management of patients with lower extremity peripheral artery disease: A report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. J Am Coll Cardiol 2017;69:e71-e126.
- Aboyans V, Criqui MH. The epidemiology of peripheral arterial disease. In: Dieter RS, Dieter RA III, Dieter RA Jr, eds. Peripheral Arterial Disease, 1st ed. New York: McGraw-Hill; 2009.
- Santistevan JR. Acute limb ischemia: An emergency medicine approach. Emerg Med Clin North Am 2017;35:889-909.
- Setacci C, de Donato G, Galzerano G, et al. Diagnostic approach to acute limb ischemia. In: Dieter RS, Dieter RA Jr., Dieter RA III, Nanjundappa A, eds. Critical Limb Ischemia: Acute and Chronic. Switzerland: Springer International Publishing; 2017:159-170.
- Met R, Bipat S, Legemate DA, et al. Diagnostic performance of computed tomography angiography in peripheral arterial disease: A systematic review and meta-analysis. JAMA 2009;301:415-429.
- [No authors listed]. Results of a prospective randomized trial evaluating surgery versus thrombolysis for ischemia of the lower extremity: The STILE trial. Ann Surg 1994;220:251-266.
- Ouriel K, Veith FJ, Sasahara AA. A comparison of recombinant urokinase with vascular surgery as initial treatment for acute arterial occlusion of the legs. Thrombolysis or Pheriperhal Arterial Surgery (TOPAS) Investigators. N Engl J Med 1998;338:1105-1111.
- Adam DJ, Beard JD, Cleveland T, et al; BASIL trial participants. Bypass versus angioplasty in severe ischemia of the leg (BASIL): Multicentre, randomised controlled trial. Lancet 2005;366:1925-1934.
- Farber A, Eberhardt RT. The current state of critical limb ischemia: A systematic review. JAMA Surgery 2016;151:1070-1077.
- Ratchford EV. Medical management of claudication. J Vasc Surg 2017;66:275-280.
- Norgren L, Hiatt WR, Dormandy JA, et al. Inter-society consensus for the management of peripheral arterial disease (TASC II). J Vasc Surg 2007;45(suppl):S5-S67.
- Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis 2014;59:e10-e52.
- Yang Z, Hu J, Qu Y, et al. Interventions for treating gas gangrene. Cochrane Database Syst Rev 2015;12:CD010577.
- Stevens DL, Bisno AL, Chambers HF, et al; Infectious Diseases Society of America. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis 2014;59:e10-e52.
- Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft-tissue infections. Clin Infect Dis 2005;41:1373-1406.
Although many cases of extremity pain are the result of mild, self-limited issues, ischemia and gangrene are catastrophic causes of pain that initially can present with nondescript findings. To limit tissue loss and optimize patient outcomes, emergency physicians must be able to distinguish benign limb pain from the earliest stages of high-risk, life- and limb-threatening disease.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
