Pediatric Cardiothoracic Point-of-Care Ultrasound: Part I
AUTHORS
Robert Stenberg, MD, Emergency Medicine Resident, Department of Emergency Medicine, University of North Carolina, Chapel Hill
Daniel Migliaccio, MD, Clinical Assistant Professor, Department of Emergency Medicine, and Head, Ultrasound Education, University of North Carolina, Chapel Hill
Daniel B. Park, MD, Associate Medical Director, Pediatric Emergency Medicine; Director, Pediatric Emergency Ultrasound; Assistant Professor, Departments of Pediatrics and Emergency Medicine, University of North Carolina School of Medicine, Chapel Hill
PEER REVIEWER
Aaron Leetch, MD, Assistant Professor, Departments of Emergency Medicine and Pediatrics; Program Director, Combined Emergency Medicine and Pediatrics Residency, University of Arizona, Tucson
Ultrasound has emerged as a critical tool for use at the bedside to guide not only diagnosis but treatment strategies as well. The first part of this article focuses on the uses and limitations of cardiac ultrasound in the acute setting. Part II will include discussion of cardiac arrest, congenital abnormalities, pneumothorax, pleural effusion, and pneumonia.
— Ann M. Dietrich, MD, FAAP, FACEP, Editor
Introduction
A 3-year-old male with no remarkable past medical history presents with lethargy. He is noted to have increased work of breathing, and is tachycardic, hypotensive, and somnolent. He appears pale, with a prolonged capillary refill. A point-of-care ultrasound (POCUS) demonstrates a hyperdynamic heart with a collapsed inferior vena cava (IVC), consistent with hypovolemic shock. He is given fluid boluses with improvement in his mental status and vital signs and is admitted for severe dehydration.
Being pulled into a room to find a child in obvious shock is quite nerve-racking. Knowing the specific type of shock helps guide management and can alter the care of the patient drastically. A quick, focused echocardiogram can help differentiate the type of shock, as well as help guide the use of fluids, vasopressors, and inotropes. More and more providers are finding POCUS to be a frontline tool. It is easily accessible and enhances critical decision-making.1,2
Cardiac Ultrasound
In the world of POCUS, the cardiac ultrasound can be among the most intimidating. It is a dynamic ultrasound, with what seem to be endless views, Dopplers, and calculations. Pediatric echocardiography seems more daunting, with even more complicated anatomy.
The most important aspects of a bedside ultrasound of the heart can be evaluated easily. An emergency department 2016 critical care consensus focused on appropriate use of bedside ultrasound on critically ill adult patients.3 The authors believed basic users could adequately assess reversible causes of cardiac arrest, cardiogenic shock, and preload responsiveness in pediatric patients. Further, they recognized that some skills, such as identifying congenital heart disease, patent ductus arteriosus, and valvular dysfunction, can be expected only from advanced users. Literature establishing the role of POCUS in the setting of congenital heart disease remains minimal.4
In 2010, the American College of Emergency Physicians and the American Society for Echocardiography released a consensus statement on the use of focused cardiac ultrasound (FOCUS), which emphasized the role of point-of-care echocardiography for evaluating pericardial effusion, systolic function, IVC assessment, and ventricular enlargement. POCUS also can be used for procedural guidance in pericardiocentesis and transvenous pacing.5
Other research has demonstrated that emergency physicians can perform limited echocardiograms adequately.6 In one study, non-cardiology physicians received a one-hour course and two hours of clinical training. After this instruction, they evaluated pediatric intensive care unit patients for pericardial effusion, left ventricular size, and left ventricular systolic function with accuracy rates of 91%, 96%, and 96%, respectively.7
Anatomy/Probe Selection
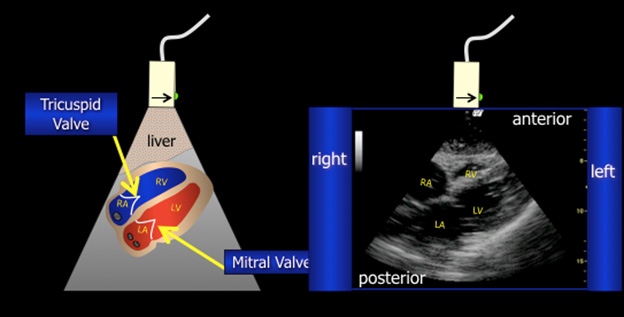
Unlike much of the rest of the body, the main views used to evaluate the heart with ultrasound are not along the traditional axes: transverse, sagittal, and coronal. Instead, the views are more along the axis of the heart (e.g., longitudinal, short, apical). (See Figure 1.) While each side has an atrium, ventricle, atrioventricular valve, and semilunar valve, the normal heart is an asymmetric structure. The left side is normally larger with papillary muscles seen toward the apex. The right side is smaller with greater trabeculations toward the apex, including a moderator band.
Figure 1. Views of Heart |
 |
Traditionally, this anatomy is obtained via the phased array probe, also known as the cardiac probe. It has a small footprint, allowing it to be positioned between ribs, and provides Doppler, high frame rates, and depth often sufficient for obtaining views of the heart.8,9
Positioning and Image Acquisition
Often, when performing POCUS in emergency situations, the provider may not have the time, space, or resources that sonographers have during a comprehensive examination. Below are some tips that can help ensure success in crunch-time situations.
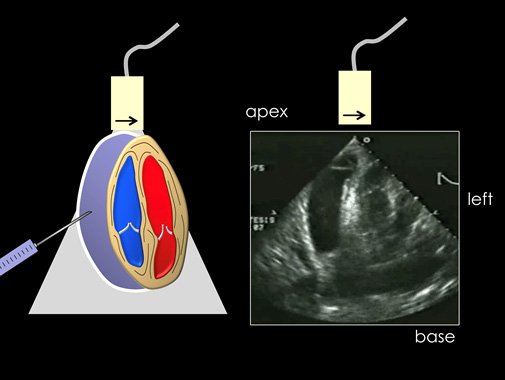
First, obtaining pediatric cardiac views follows the same principles as for adults. Overall, the ideal position is a supine patient, in a left lateral decubitus position. The left nipple is a general landmark. The parasternal long and short views often are found 1-2 cm medial to the nipple and the apical four view inferior. (See Figure 2.)
Figure 2. Obtaining an Apical Four-Chamber in the Left Lateral Decubitus Position |
 |
Consider the angle of the probe relative to the patient. For parasternal views, the image often is optimized when the probe is perpendicular to the chest, and apical/subcostal views are at an acute angle. If the provider can view a good parasternal long, often a good parasternal short is possible. If the probe is anchored in good position, rotate it 90 degrees and vice versa. Stabilizing the hand with the probe during rotation can help maintain the window. In a similar fashion, once the subcostal is in view, it is easy to switch to the IVC view.
The left lung and lingula are large barriers preventing views other than the subcostal views. Further, when an asthma patient’s lungs are hyperinflated and are trapping air, parasternal views can become increasingly challenging. Having the left arm abducted above the head can open the child’s narrow intercostal spaces. Left lateral decubitus position helps swing the heart anteriorly to maximize interface between the heart and intercostals. Potentially, the provider may have a view only at certain points during the respiratory cycle. One tactic is to have the patient take a deep breath and then blow out all the air, keeping it out as long as possible.
With some patients, the provider may not be able to obtain all of the cardiac windows, so the study may be limited. Finally, deliberate practice with experienced providers and repetition will help the clinician proceed with confidence, image acquisition, and speed when seconds matter.
Standard Views for Cardiac POCUS
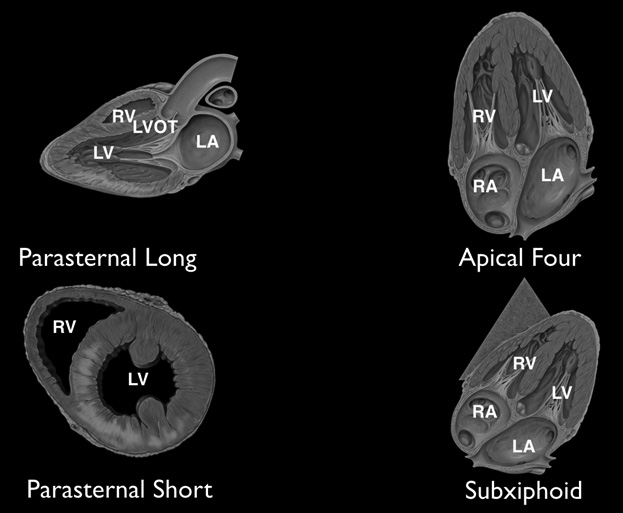
Parasternal Long. The parasternal long view is great for evaluation of the thoracic aorta and is one of the key views for evaluating function. (See Figure 3.)
Figure 3. Parasternal Long View
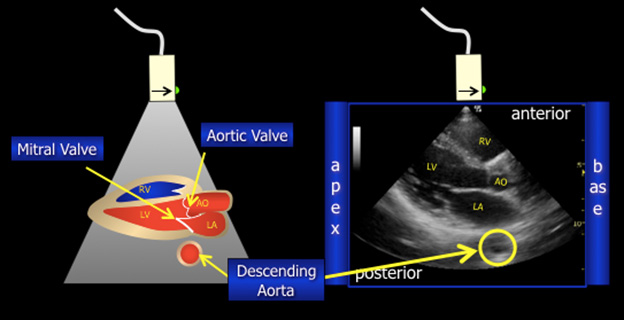
Source: Image courtesy of Thomas Cook, MD.
- Anatomy: The long axis of the left side of heart is seen. The left ventricular inflow, as well as the outflow, is seen. The motion anterior leaflet of the mitral valve is well seen. The right side, left ventricular outflow tract, and left atrium are seen in a row; in a normal heart, these are all roughly the same size.
- Probe marker: Toward right shoulder
- Probe site and angle of probe relative to chest: Lateral to the sternum on left side of chest, usually medial to the nipple (same site as the parasternal short axis). Perpendicular to the chest.
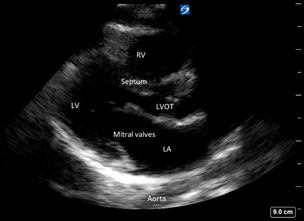
Parasternal long in healthy 5-year-old female
Parasternal Short. The parasternal short view is used for evaluating contractility and valvular function and assessing the septal wall. It can be used for checking the aortic and mitral valve, as well as evaluating the wall motion of the left ventricle (LV). (See Figure 4.)
Figure 4. Parasternal Short View |
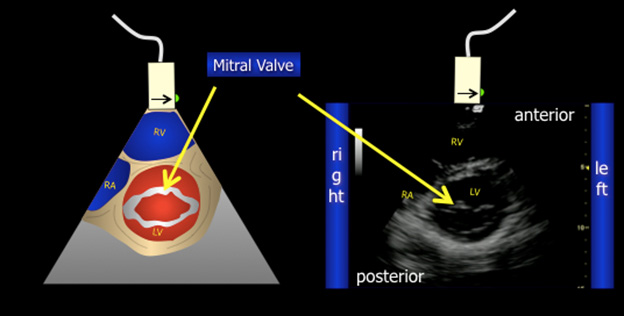 |
|
Source: Image courtesy of Thomas Cook, MD. |
|
|
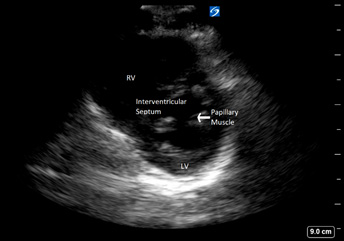
Parasternal short in healthy 5-year-old female |
 |
Apical Four. The apical four view is best for comparing the size of the ventricles. It can be used to evaluate effusion and left ventricular function. (See Figure 5.)
Figure 5. Apical View |
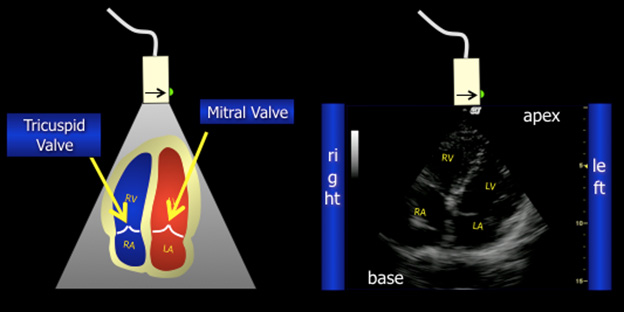 |
|
Source: Image courtesy of Thomas Cook, MD. |
|
|
Apical in healthy 5-year-old female |
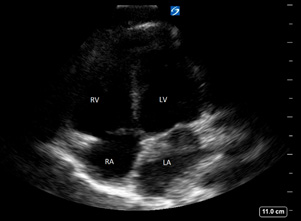 |
Subcostal/Subxiphoid. The subcostal/subxiphoid view is used to evaluate for pericardial effusion as well as for general evaluation of cardiac function. It may be obtained while chest compressions/procedures are being performed. (See Figure 6.)
Figure 6. Subcostal/Subxiphoid View |
|
|
Source: Image courtesy of Thomas Cook, MD. |
IVC View, Longitudinal. The IVC longitudinal view is used to correlate to central venous pressure, with debatable utility in fluid responsiveness, but it can help support clinical scenarios as explained later. (See Figure 7.)
Figure 7. IVC View, Longitudinal View |
|
|
IVC, longitudinal in healthy 5-year-old female |
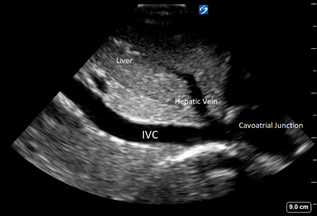 |
Cardiac Function
A 7-year-old previously healthy male presents with a cough and shortness of breath. He is febrile, tachycardic, and ill appearing. A fluid bolus is given, and he seems to be doing worse. A POCUS of his heart reveals poor functioning consistent with myocarditis, and he is admitted to the cardiac ICU.
What is the cardiac function? What does the IVC look like? These focused questions for the emergency provider can change ED management in terms of diagnosis and resuscitation.
For an emergency physician, the estimation of gross contractility and function of the heart is crucial in POCUS. Calculating ejection fraction and grading diastolic dysfunction is out of the scope of practice of POCUS and should be reserved for echocardiograms performed by cardiology. However, the provider should be able to discern if the heart squeeze appears excessive (hyperdynamic), normal, or decreased. Often, decreased systolic function is broken down to mildly, moderately, and severely decreased in some studies, correlating to fractions of 41-55%, 31-40%, and < 30%.10
Providers can watch a few videos on this spectrum and quickly classify as such. Hyperdynamic function, which should not be confused with tachycardia, shows contraction beyond what would be expected; the endocardial borders often are “kissing.” The combination of a hyperdynamic heart with significant respiratory collapsibility of the IVC often supports a relatively volume-depleted state.
In myocarditis, as in the case above, POCUS can alter management and focus care to the underlying etiology. Identifying cardiac dysfunction can help the ED provider treat for cardiogenic shock and avoid other potentially deleterious actions, such as giving a fluid bolus in a volume-overloaded state. In myocarditis, one can expect to see a tachycardic state, and may see a decreased cardiac contractility, including regional wall motion abnormalities.11,12 There also are case reports of associated pericardial effusion.13
Ventricular Walls
A 12-year-old female with a history of cystic fibrosis presents with progressively worsening shortness of breath. She is tachypneic, has increased work of breathing, and appears labored. A POCUS of her heart reveals right ventricle (RV) enlargement, consistent with pulmonary hypertension, and she is admitted to the PICU.
Comparing the size of the ventricles can be quite helpful; the RV should be two-thirds the size of the LV. When the right side is greater than this size, there is a concern for right heart failure. This can be seen in a pulmonary embolism or, more commonly in pediatrics, pulmonary hypertension from underlying lung disease. Signs concerning for right heart strain include:
- RV approaches 1:1 ratio of LV.
- LV on parasternal short axis becomes D-shaped (as opposed to circular) secondary to the right pressures compressing the left.
- McConnell’s sign, which shows the apex of the RV, has significantly more contractility relative to the rest of the RV.
- Dilated IVC.
Data obtained from the point-of-care evaluation of the right heart must be considered in the clinical context of the patient to be used properly and be most helpful. For an acutely ill child with marginal vitals and a clinical picture with a potential for right heart failure (as in the above patient), the POCUS may be quite helpful.14,15 As a caveat for pulmonary embolism, it cannot be ruled out by bedside echocardiogram. In a 2016 study that evaluated 511 patients diagnosed with pulmonary embolism, only 16 of whom were hemodynamically unstable, 71% of the patients had no signs on transthoracic echocardiograms, along with 10% incidental findings. All 16 of the unstable patients had a large hypokinetic right ventricle and either a McConnell’s sign, thrombus seen on echocardiogram, or a 60/60 sign (complex multistep measurement including pulsed-wave Doppler).16 (See Figure 8.)
Figure 8. Apical Four-Chamber View |
|
Apical four-chamber in a patient with a pulmonary embolism showing the right side being much larger than the left |
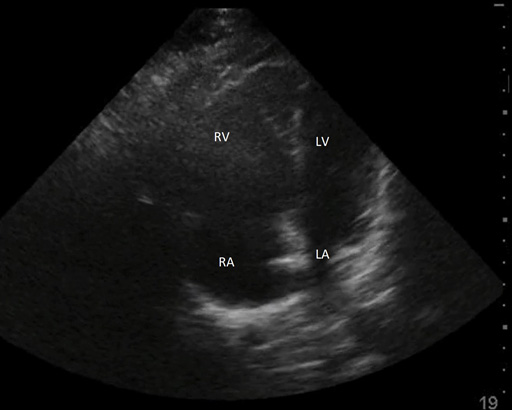 |
A 14-year-old male with no known history who was adopted as a child presents with syncope while running during a track event. He is well appearing, with normal vital signs and a systolic murmur on exam. A POCUS demonstrates hypertrophic cardiomyopathy (HCM), and he is admitted for an echocardiogram performed by cardiology and initiation of treatment.
HCM is a condition emergency providers are taught not to miss. Research has demonstrated that focused cardiac ultrasound can have an effective sensitivity.17 It also has been shown to be easily attainable, with clinicians able to screen adequately after basic ultrasound orientation, a lecture, and a two-hour hands-on session.18 In adults and children near adult size, the general rule is if any LV wall segment (classically the septum) is 15 mm or greater and is not explained by a “loading” condition (e.g., aortic stenosis, hypertension, etc.), there is concern. The 2014 European Society of Cardiology (ESC) suggests for children that the measurement be two standard deviations above the mean, citing Kampmann in Heart 2000 as source for baseline values.19 In that paper, two standard deviations for an LV posterior wall on a 1.1 m2 body surface area, or roughly a 10-year-old child, was 12.6 mm. The ESC suggests at 13 mm, even in adults, HCM can be seen in both genetic and non-genetic forms.
Achieving the most accurate wall thickness possible is important since 1-2 mm can make a large difference in managing the patient. Oblique planes are one of the biggest sources of errors because they can provide falsely elevated numbers. Relying on a parasternal short over a parasternal long may reduce this. Further, the values should be collected at the end of diastole. This ensures that one is not just measuring a contracted heart. Consider using the freeze function and trying to scroll through the recent clip to find what is deemed to be diastole.
One other finding of HCM is systolic anterior motion (SAM) of the mitral valve. The mitral valve or the chordae, which should be closed during systole, can be swinging over the left ventricular outflow tract, playing a role in the obstructive pathology of HCM. Sometimes, this can be seen on POCUS. (See Figure 9.)
Figure 9. Parasternal Long Showing a Thickened Septum |
|
Parasternal long showing a thickened septum as well as systolic anterior motion. The mitral valve is closed and the left ventricle squeezes down. Part of the valve comes over the left ventricular outflow tract. |
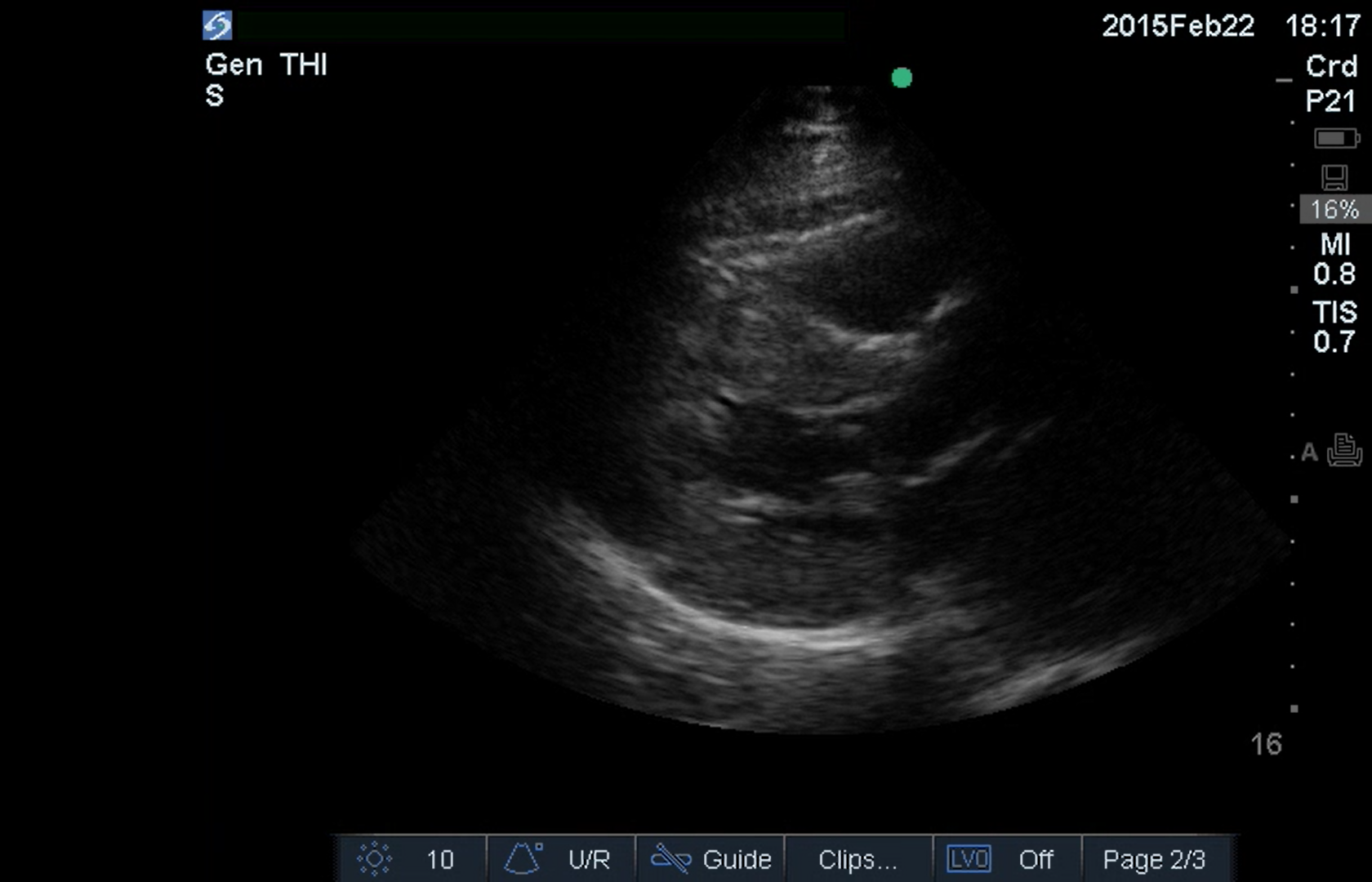 |
SAM is not sensitive and is variably specific. In an older study of 721 echocardiograms from variable cardiac diseases, eight had HCM, 11 had transposition of the great vessels, and in three the SAM was not found to be related; after excluding transposition, specificity was 97%.20 In a 2017 Japanese study, 9,180 echocardiograms found SAM in 60 patients with HCM and 28 patients without HCM.22 The prevalence of SAM after mitral valve repair is another factor that reduces the specificity.22 Overall, in combination with a thickened septum and clinical scenario fitting for HCM, SAM may be supportive.
Pericardial Effusion, Tamponade, Pericardiocentesis
A 9-year-old female with a history of leukemia in remission presents with progressively worsening shortness of breath, which is worse with exertion. She has an elevated heart rate and respiratory rate with a blood pressure of 76/48 mmHg. She appears ill. A POCUS of her heart reveals a large pericardial effusion with diastolic right ventricular collapse consistent with pericardial tamponade. She has an emergent pericardiocentesis and is admitted to the cardiac ICU for a pericardial window.
In both children and adults, pericardial effusions vary from small (0.5-1 cm) to moderate (1-2 cm) to large (> 2 cm). Size does not always correlate with severity. If it suddenly develops as a traumatic hemopericardium, a small amount of fluid can cause tamponade physiology; this correlates to 100-200 mL or a small-to-moderate pericardial effusion.23,24
In a study from Children’s Hospital of Boston, researchers looked at a large echocardiography database noting etiologies of clinically significant pericardial effusions (moderate to large).25 The most common etiology of significant effusions noted were 39% cancer and 37% idiopathic. (See Table 1.) In this scenario, it is important to look for a black stripe of fluid around the heart, anterior to the descending aorta; however, both exudative and hemorrhagic effusions can be echogenic.
Table 1. Etiologies of Patients With Pericardial Effusion |
|
Etiology |
Frequency |
|
Neoplastic |
39% |
|
Idiopathic |
37% |
|
Collagen |
9% |
|
Renal |
8% |
|
Bacterial |
3% |
|
Other (hypothyroidism, viral sepsis, and anorexia; 1 each) |
3% |
|
HIV |
2% |
|
Source: Kühn B, Peters J, Marx GR, Breitbart RE. Etiology, management, and outcome of pediatric pericardial effusions. Pediatr Cardiol 2008;29:90-94. |
|
One of the best mimickers of pericardial effusions is the epicardial fat pad.26 An extension of visceral fat, it tends to be contained anteriorly and has echogenicity (usually isoechoic). If misread, it can lead to unnecessary pericardiocentesis, which can cause myocardial puncture, tamponade, and even death. This is no longer seen only in adults because of increasing childhood obesity rates.27 The best way to differentiate is by looking at multiple views, particularly posteriorly. This is because most effusions should be dependent, and in most scenarios during an echo, that will be posterior. An epicardial fat pad also tends to be best evaluated in systole, as it usually is collapsed during diastole.26,28 It also is important to confirm the effusion is in the pericardial space vs. the pleural space. On the parasternal view, a pleural effusion will traverse posterior to the descending thoracic aorta, whereas a pericardial effusion remains anterior to the aorta.29,30
The most reliable clinical finding for tamponade is pulsus paradoxus, with a pooled sensitivity of 82%.31 Sonographic evidence has shown studies with much higher sensitivities.23 The most reliable are early-diastolic RV collapse when the ventricle should be filling, and end-diastolic right atrial collapse (also called early systolic collapse) when the atria should be filling. (See Figure 10.) At times, it is difficult to discern what moment is diastole vs. systole. Watching the valves open is one easy way to identify diastole. Some providers like to use M-mode in parasternal long view to watch the RV relative to the mitral valve along the septum. In the setting of tachycardia, slowing the clip can help isolate the time of diastole. As with pericardial effusion mentioned earlier, further support of obstructive shock can be found by noting a dilated IVC with minimal respiratory variation. One also may see a swinging heart in a clockwise fashion; however, this has yet to be studied in terms of sensitivities and specificities.32 (See Figures 10, 11, and 12.)
Figure 10. Subxiphoid View |
|
Subxiphoid view demonstrating right atrial collapse in a patient with pericardial tamponade |
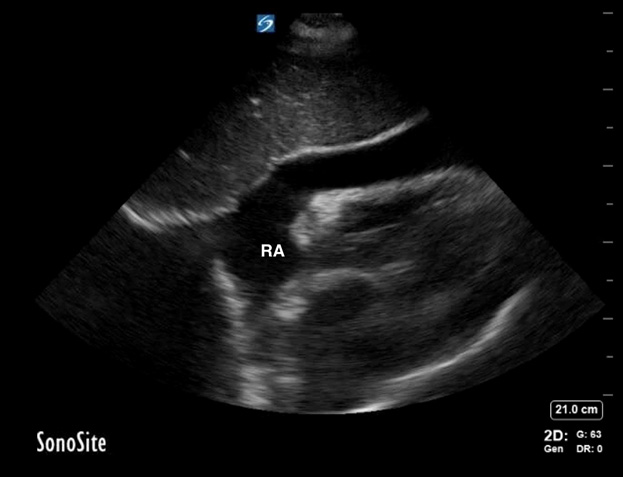 |
Figure 11. Parasternal Long Showing RV Collapse |
|
Parasternal long showing RV collapse with mitral valve open in a patient with tamponade. Also note the echogenic clot in the pericardium. |
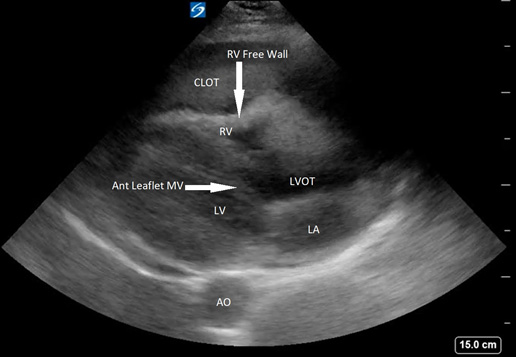 |
Figure 12. M-Mode in the Parasternal Long Axis |
|
M-mode in the parasternal long axis visualizing the activity of the right ventricle, and comparing it to when the mitral valves are opening during diastole |
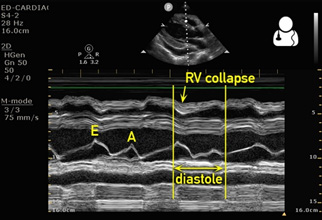 |
|
Source: Creative Commons. Available at: https://www.ultrasoundoftheweek.com/uotw-78-answer/ |
When there is evidence of tamponade, the provider must be ready to intervene. Using ultrasound guidance for pericardiocentesis has been supported by both adult and pediatric literature with decreased complication rates. Literature regarding pediatric pericardiocentesis performed by an emergency provider is sparse. There generally are three approaches. The traditional landmark approach involves a blind subcostal approach aiming toward the left shoulder. In adult patients, ultrasound-guided pericardiocentesis can be performed from a subcostal apical or parasternal long approach. In pediatric patients, providers tend to use parasternal long or subcostal approaches. One overall key goal should be finding the largest pocket of fluid and draining it from an easily accessible location. Given the anatomical complexity and the number of structures near the pericardium, it is important not to damage adjacent structures. Some sources also recommend applying agitated saline to confirm needle tip placement, which should produce bubbles in the pericardial space.11,32-38
The authors of Pediatric Emergency and Critical Care Ultrasound recommend using the parasternal long axis, because it is a more anatomically direct approach to the pericardium.33 If this is done, the provider should insert the needle on the lateral aspect of the ultrasound, focusing on avoiding the internal mammary artery just lateral to the sternum. Needle insertion also should be just above the rib. As with thoracostomies, entrance into the intercostal space is best made superior to the ribs to avoid the neurovascular bundle on the inferior side of the ribs.39 (See Figure 13.)
Figure 13. Pericardiocentesis |
|
Pericardiocentesis from the apical approach with needle tip seen in the pericardial space |
|
Source: Image courtesy of Thomas Cook, MD. |
CONCLUSION
POCUS is an extremely useful tool for physicians to help extend the physical exam. (See Table 2.) It can assist in the management of the child with undifferentiated shock and can add information when a child’s presentation and diagnosis remain obscure. As stated by multiple organizations, adequate documentation and quality assurance are critical. Documentation should include the scope of practice for the POCUS study performed and any limitations. It is important for providers to document what they see, and if there are any possible anomalies on the ultrasound that need further evaluation, perhaps with further imaging. In this manner, POCUS can become an extension of the physical exam and assist in medical decision-making in an appropriate and safe manner.
Table 2. Findings on Cardiac Point-of-Care Ultrasound |
||
Pathology |
Images Required |
Images Demonstrate |
|
Myocarditis/congestive heart failure |
|
|
|
Pulmonary hypertension/pulmonary embolism/Eisenmenger |
|
|
|
Hypertrophic cardiomyopathy |
|
|
|
Pericardial tamponade |
|
|
|
Hypovolemia |
|
|
|
IVC: inferior vena cava; RV: right ventricle; LV: left ventricle; RA: right atria. |
||
Acknowledgments
The authors would like to thank Thomas Cook, MD, for his assistance in creating some of the ultrasound images.
REFERENCES
- Fentress M, Heyne TF, Barron KR, Jayasekera N. Point-of-care ultrasound in resource-limited settings: Common applications. South Med J 2018;111:424-433.
- Istasy V, Thompson D, Belisle SS, et al. The role of point of care ultrasound in the injured paediatric patient. Curr Pediatr Rev 2018;14:41-47.
- Whitson MR, Mayo PH. Ultrasonography in the emergency department. Crit Care 2016;20:227.
- Doniger SJ. Bedside emergency cardiac ultrasound in children. J Emerg Trauma Shock 2010;3:282-291.
- Labovitz AJ, Noble VE, Bierig M, et al. Focused cardiac ultrasound in the emergent setting: A consensus statement of the American Society of Echocardiography and American College of Emergency Physicians. J Am Soc Echocardiogr 2010;23:1225-1230.
- Pershad J, Myers S, Plouman C, et al. Bedside limited echocardiography by the emergency physician is accurate during evaluation of the critically ill patient. Pediatrics 2004;114:e667-e671.
- Spurney CF, Sable CA, Berger JT, Martin GR. Use of a hand-carried ultrasound device by critical care physicians for the diagnosis of pericardial effusions, decreased cardiac function, and left ventricular enlargement in pediatric patients. J Am Soc Echocardiogr 2005;18:313-319.
- Scruggs W, Fox JC. Chapter 2. Equipment. In: Ma O, Mateer JR, Reardon RF, Joing SA, eds. Ma and Mateer’s Emergency Ultrasound, 3e. New York: McGraw-Hill; 2014.
- Szabo TL, Lewin PA. Ultrasound transducer selection in clinical imaging practice. J Ultrasound Med 2013;32:573-582.
- Margossian R, Schwartz ML, Prakash A, et al; Pediatric Heart Network Investigators. Comparison of echocardiographic and cardiac magnetic resonance imaging measurements of functional single ventricular volumes, mass, and ejection fraction (from the Pediatric Heart Network Fontan Cross-Sectional Study). Am J Cardiol 2009;104:419-428.
- Caforio AL, Pankuweit S, Arbustini E, et al; European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: A position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J 2013;34:2636-2648.
- Felker GM, Boehmer JP, Hruban RH, et al. Echocardiographic findings in fulminant and acute myocarditis. J Am Coll Cardiol 2000;36:227-232.
- Bramante RM, Cirilli A, Raio CC. Point-of-care sonography in the emergency department diagnosis of acute H1N1 influenza myocarditis. J Ultrasound Med 2010;29:1361-1364.
- Descotes-Genon V, Chopard R, Morel M, et al. Comparison of right ventricular systolic function in patients with low risk and intermediate-to-high risk pulmonary embolism: A two-dimensional strain imaging study. Echocardiography 2013;30:301-308.
- Lodato JA, Ward RP, Lange RM. Echocardiographic predictors of pulmonary embolism in patients referred for helical CT. Echocardiography 2008;25:584-590.
- Kurnicka K, Lichodziejwska B, Goliszek S, et al. Echocardiographic pattern of acute pulmonary embolism: Analysis of 511 consecutive patients. J Am Soc Echocardiogr 2016;29:907-913.
- Zhang S, Zhu D, Wan Z, Cao Y. Utility of point-of-care echocardiogram in the rapid diagnosis of hypertrophic cardiomyopathy. Am J Emerg Med 2013;31:1280-1282.
- Kerkhof DL, Corrado GD. Response to “Hypertrophic cardiomyopathy in young athletes: Successful screening with point-of-care ultrasound by medical students.” J Ultrasound Med 2018;37:1575.
- Kampmann C, Wiethoff CM, Wenzel A, et al. Normal values of M mode echocardiographic measurements of more than 2000 healthy infants and children in central Europe. Heart 2000;83:667-672.
- Maron BJ, Gottdiener JS, Perry LW. Specificity of systolic anterior motion of anterior mitral leaflet for hypertrophic cardiomyopathy. Prevalence in large population of patients with other cardiac diseases. Heart 1981;45:206-212.
- Uematsu S, Takaghi A, Imamura Y, et al. Clinical features of the systolic anterior motion of the mitral valve among patients without hypertrophic cardiomyopathy. J Cardiol 2017; 69:495-500.
- Loulmet DF, Yaffee DW, Ursomanno PA, et al. Systolic anterior motion of the mitral valve: A 30-year perspective. J Thorac Cardiovasc Surg 2014;148:2787-2793.
- Imazio M, Adler Y. Management of pericardial effusion. Eur Heart J 2013;34:1186-1197.
- Spodick DH. Acute cardiac tamponade. N Engl J Med 2003;349:684-690.
- Kühn B, Peters J, Marx GR, Breitbart RE. Etiology, management, and outcome of pediatric pericardial effusions. Pediatr Cardiol 2008;29:90-94.
- Blanco P, Volpicelli G. Common pitfalls in point-of-care ultrasound: A practical guide for emergency and critical care physicians. Crit Ultrasound J 2016;8:15.
- Elshorbagy HH, Fouda ER, Kamal NM, et al. Evaluation of epicardial fat and carotid intima-media thickness in obese children. Iran J Pediatr 2016;26:e2968.
- Bertaso AG, Bertol D, Duncan BB, Foppa M. Epicardial fat: Definition, measurements and systematic review of main outcomes. Arq Bras Cardiol 2013;101:e18-e28.
- Strony R, Linkenheil E. How to delineate pericardial effusion from pleural effusion on bedside ultrasound. J Emerg Med 2016;50:495-496.
- Goodman A, Perera P, Mailhot T, Mandavia D. The role of bedside ultrasound in the diagnosis of pericardial effusion and cardiac tamponade. J Emerg Trauma Shock 2012;5:72-75.
- Roy CL, Minor MA, Brookhart MA, Choudhry NK. Does this patient with a pericardial effusion have cardiac tamponade? JAMA 2007;297:1810-1818.
- Fowler NO. Cardiac tamponade. A clinical or an echocardiographic diagnosis? Circulation 1993;87:1738-1741.
- Doniger S. Pediatric Emergency and Critical Care Ultrasound. Cambridge, UK: Cambridge University Press; 2013.
- Adler Y, Charron P, Imazio M, et al; ESC Scientific Document Group. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC) Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 2015;36:2921-2964.
- Sagrista Sauleda J, Almenar Bonet L, Angel Ferrer J, et al. [The clinical practice guidelines of the Sociedad Espanola de Cardiology a on pericardial pathology]. In Spanish. Rev Esp Cardiol 2000;53:394-412.
- Maisch B, Seferovic PM, Ristic AD; Task Force on the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology. Guidelines on the diagnosis and management of pericardial diseases executive summary; The Task Force on the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology. Eur J Heart 2004;25:587-610.
- Tsang T, Enriquez-Sarano M, Freeman W, et al. Consecutive 1127 therapeutic echocardiographically guided pericardiocenteses: Clinical profile, practice patterns, and outcomes spanning 21 years. Mayo Clin Proc 2002;77:429-436.
- Tsang TS, El-Najdawi EK, Seward JB, et al. Percutaneous echocardiographically guided pericardiocentesis in pediatric patients: Evaluation of safety and efficacy. J Am Soc Echocardiogr 1998;11:1072-1077.
- Holmes DR Jr, Nishimura R, Fountain R, Turi ZG. Iatrogenic pericardial effusion and tamponade in the percutaneous intracardiac intervention era. JACC Cardiovasc Interv 2009;2:705-717.
Ultrasound has emerged as a critical tool for use at the bedside to guide not only diagnosis but treatment strategies as well. The first part of this article focuses on the uses and limitations of cardiac ultrasound in the acute setting. Part II will include discussion of cardiac arrest, congenital abnormalities, pneumothorax, pleural effusion, and pneumonia.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.