Evaluation and Treatment of Adrenal Insufficiency in the Emergency Department
March 1, 2019
Reprints
AUTHORS
Tiffany Murano, MD, Associate Professor, Department of Emergency Medicine, Rutgers New Jersey Medical School, Newark, NJ
Lana Shaker, MD, Department of Emergency Medicine, Rutgers New Jersey Medical School, Newark, NJ
PEER REVIEWER
Frank LoVecchio, DO, MPH, FACEP, Professor, University of Arizona College of Medicine, Associate Medical Director, Samaritan Regional Poison Control Center, Phoenix, AZ
EXECUTIVE SUMMARY
- Most often adrenal crisis is precipitated by gastrointestinal disease, but it also is precipitated by infection, trauma, surgery, strenuous physical activity, myocardial infarction, hypoglycemia, emotional distress, allergic reactions, and
pregnancy. - Tertiary adrenal insufficiency can occur when exogenous corticosteroids, in doses equivalent to 5 mg or more of prednisolone for four or more weeks, are stopped abruptly.
- Treatment of adrenal crisis should begin as soon as the diagnosis is suspected. Treatment is with fluids and hydrocortisone 100 mg intravenously or intramuscularly followed by an infusion.
- It is important to identify and treat the precipitating cause of adrenal crisis.
Introduction
First described by Thomas Addison in 1855, adrenal insufficiency is the clinical manifestation of glucocorticoid deficiency, with or without mineralocorticoid and adrenal androgen deficiency. Adrenal insufficiency was universally fatal until cortisone was synthesized in 1949, and it continues to be associated with significant morbidity and increased risk of mortality.1
Adrenal insufficiency is classified according to its underlying mechanism, and can be categorized as primary, secondary, or tertiary adrenal failure. Adrenal crisis is a life-threatening emergency presenting with acute hemodynamic decompensation. Treatment of adrenal insufficiency is geared toward determination of the underlying cause, chronic steroid replacement, and prevention or management of acute adrenal crisis with higher-dose parenteral glucocorticoids as well as fluid resuscitation.
Adrenal insufficiency is a challenging diagnosis and often is elusive to physicians.2 Two out of three patients will present three or more times with symptoms of adrenal failure before receiving a correct diagnosis. Many patients may not be diagnosed until after an acute hospitalization with adrenal crisis.2 Early recognition and treatment of adrenal crisis is critical because it remains an important cause of death for patients with adrenal insufficiency. Moreover, patients may deteriorate quickly, resulting in death soon after arrival in the emergency department (ED).3 Patients and other caretakers should be educated on how to prevent an acute crisis. Equally important, the physician should administer an adequate dose of steroids during stress or crisis.4,5 It is essential for emergency physicians to have a high index of suspicion for identifying patients who present with adrenal insufficiency and crisis. Initiating appropriate management in the ED also is necessary. This article addresses the pathophysiology, discusses various clinical presentations, and reviews current evidence-based practices for managing adrenal insufficiency and crisis in the ED.
Epidemiology and Etiology
Primary Adrenal Insufficiency
The most common cause of primary adrenal insufficiency during the first part of the 20th century was tuberculosis. Now, autoimmune adrenal insufficiency is the leading etiology.6 Primary adrenal insufficiency occurs more frequently in women. Although it can present at any age, it most commonly presents between the ages of 30-50 years.7 Approximately one-half of patients with primary adrenal insufficiency also have other coexisting autoimmune diseases.6 (See Table 1.) In developed countries, up to 90% of primary adrenal insufficiency cases are caused by autoimmune adrenalitis, also known as Addison’s disease. In developing countries, tuberculosis, disseminated fungal infections, and HIV/AIDS are the most significant causes of primary adrenal insufficiency.8
Table 1. Summary of Prevalence and Etiologies of Adrenal Insufficiency |
||
|
Type |
Most Common Cause |
Prevalence |
|
Primary Adrenal Insufficiency |
Autoimmune adrenalitis (Addison’s disease)6 |
40-144 per million18 |
|
Secondary Adrenal Insufficiency |
Pituitary adenoma11 |
150-280 per million19 |
|
Tertiary Adrenal Insufficiency |
Chronic exogenous glucocorticoid use12 |
Ranging from 14% to 63% of patients on glucocorticoid therapy; dose dependent20 |
|
Adrenal Crisis |
Gastrointestinal disturbance in patients with primary adrenal insufficiency4 |
6-10 episodes per 100 patients per year15 |
Secondary and Tertiary Adrenal Insufficiency
Secondary adrenal insufficiency is more common than primary adrenal insufficiency and affects women more frequently than men. The peak age of diagnosis is in the sixth decade of life.9,10 Pituitary adenoma is the most common cause of secondary adrenal insufficiency.11 The most common cause of tertiary adrenal insufficiency is long-term exogenous glucocorticoid use, leading to suppression of the hypothalamic secretion of corticotropin-releasing hormone (CRH).12 Collectively, secondary and tertiary adrenal insufficiency also are known as central adrenal insufficiency.
Adrenal Crisis
The incidence of adrenal crisis is estimated to be six to 10 episodes per 100 patients with adrenal insufficiency per year, and it increases with age.4,13,14 Of these six to 10 episodes, one death can be expected.15 In a survey of 840 patients with Addison’s disease in four countries, 8% of patients required hospital treatment for an adrenal crisis annually.16 Adrenal crisis can be the initial presentation of undiagnosed and/or untreated adrenal insufficiency, but it also occurs frequently in patients already on standard therapy for known adrenal insufficiency.
Adrenal crisis can occur with primary, secondary, or tertiary adrenal insufficiency, but it is more common in primary adrenal insufficiency. Adrenal crisis may be recurrent in as many as 65% of patients with primary adrenal insufficiency and 47% of those with secondary adrenal insufficiency.4 Gastrointestinal disturbance is the most common precipitant and is the cause of two-thirds of hospital admissions for adrenal crisis. Other common precipitants include infections, surgical procedures, accidental injuries, strenuous physical activity, myocardial infarction, severe allergic reaction, hypoglycemic events in diabetic patients, pregnancy, and emotional stress.4,13,17
Pathophysiology
The adrenal glands are located above each kidney and consist of the medulla and the cortex. The adrenal medulla secretes catecholamines such as epinephrine and norepinephrine. The adrenal medulla is under sympathetic system control and is not included in the disease process of adrenal insufficiency.
The adrenal cortex has three distinct zones that secrete various hormones under the control of feedback mechanisms. The outermost zone is the zona glomerulosa, which synthesizes aldosterone and is regulated predominantly by the renin-angiotensin system. The zona glomerulosa is not impaired in secondary or tertiary adrenal insufficiency. The next zone, zona fasciculata, secretes cortisol, and is regulated by adrenocorticotropin (ACTH), which is released from the anterior pituitary gland in response to the hypothalamic release of CRH. Cortisol secretion typically is pulsatile and fluctuates in a circadian fashion — highest early in the morning and lowest around midnight. The innermost layer is the zona reticularis and is the location of adrenal adrogen synthesis.
Basal neural input, which follows a circadian rhythm, and stress lead to an increase in CRH release by the hypothalamus. (See Figure 1.) Vasopressin also is released with CRH. CRH and vasopressin stimulate cells of the anterior pituitary gland to increase the release of stored ACTH. CRH also stimulates synthesis of new ACTH. Subsequently, ACTH stimulates release of cortisol by binding to receptors on the cells of the zona fasciculata and reticularis, leading to cortisol release. Increased glucocorticoids exert a feedback inhibition on ACTH secretion directly and also inhibit ACTH secretion indirectly by decreasing CRH and vasopressin secretion.21,22 (See Figure 1.)
Figure 1. Hypothalamus-Pituitary-Adrenal Axis |
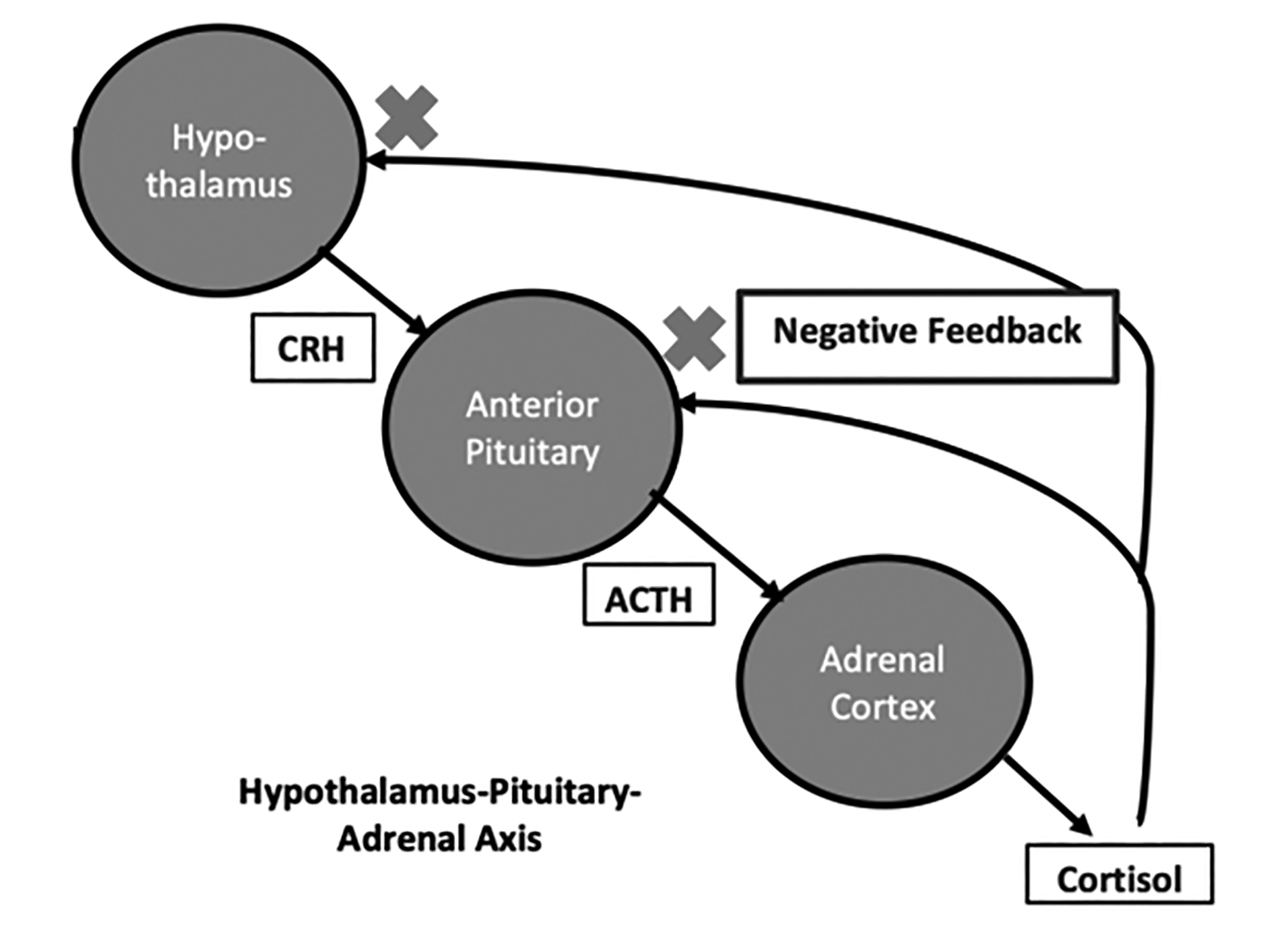 |
|
The hypothalamus produces corticotropin-releasing hormone (CRH), which stimulates the pituitary gland to produce adrenocorticotropic hormone (ACTH). ACTH stimulates the adrenal gland to produce cortisol, which has negative feedback on the production of CRH and ACTH by the hypothalamus and pituitary glands, respectively. |
Primary Adrenal Insufficiency
Primary adrenal insufficiency results from intrinsic adrenal failure, leading to decreased glucocorticoids, mineralocorticoids, and androgens. The decreased levels of these hormones lead to an increase in upstream mechanisms in an effort to increase adrenal hormone production, resulting in increased ACTH by the pituitary and CRH and vasopressin by the hypothalamus. (See Figure 2.) Patients with primary adrenal insufficiency are at an increased risk for adrenal crisis because of the decrease in mineralocorticoids and glucocorticoids.23
Figure 2. Primary, Secondary, and Tertiary Adrenal Insufficiency |
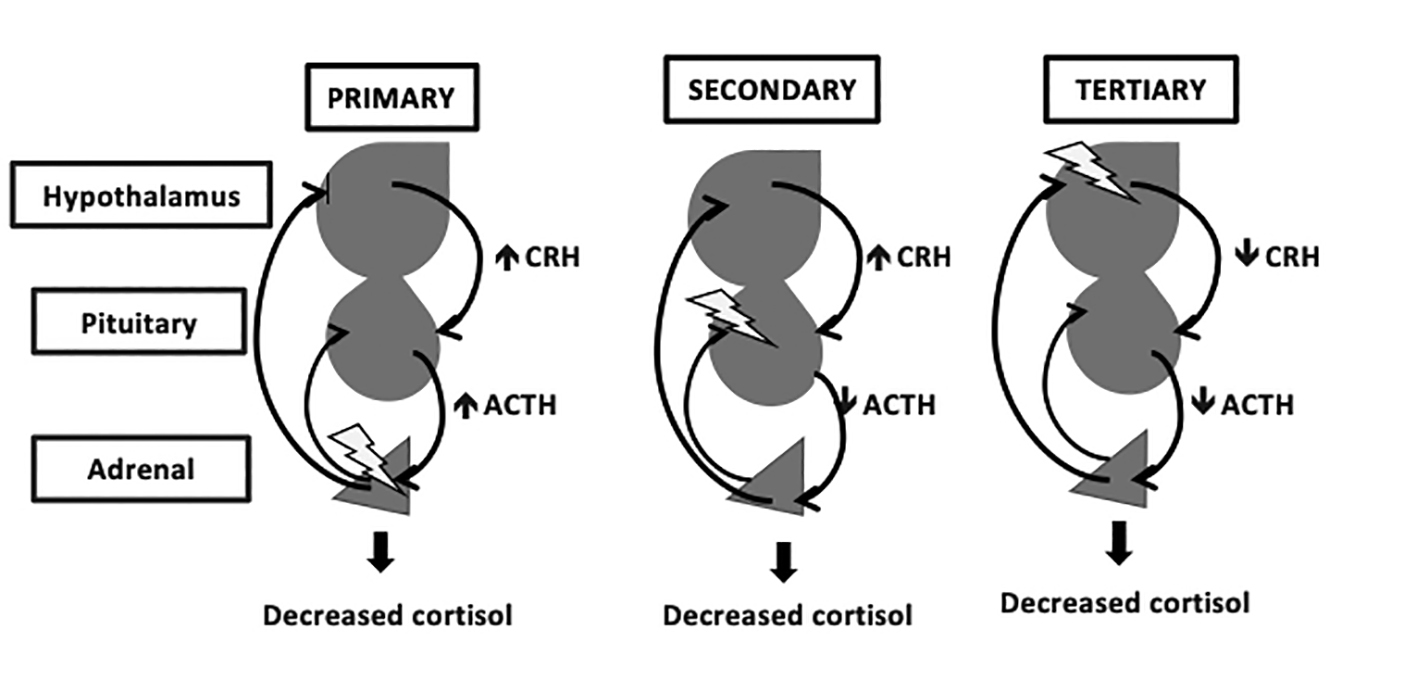 |
|
The organ of deficiency is indicated by the lightning bolt. The positive hormonal regulation is indicated by the arrows on the right side. The negative feedback is indicated by the arrows on the left side. CRH, corticotropin-releasing hormone; ACTH, adrenocorticotropic hormone |
Addison’s disease can be an isolated condition or a part of an autoimmune polyendocrinopathy syndrome.9,24 Sixty percent of patients with Addison’s disease have an additional autoimmune disease.6 Autoimmune-induced primary adrenal insufficiency is characterized by immune-mediated adrenal cortex destruction.25 In this disease entity, the zona glomerulosa is the first zone affected. This leads to the initial feature of adrenal failure, which is characterized by high plasma renin and low aldosterone concentrations, which then is followed by a phase of progressive glucocorticoid deficiency. This glucocorticoid deficiency is characterized as an inadequate response to stress, which then progresses to overt failure.16
Scientists have identified genes related to susceptibility to autoimmune adrenal disease.26 Patients with an autoimmune polyendocrinopathy syndrome can experience various conditions in addition to adrenal insufficiency. These include: chronic mucocutaneous candidiasis, hypoparathyroidism, dental enamel hypoplasia, nail dystrophy, type 1 diabetes mellitus, pernicious anemia, autoimmune thyroid disease, hypogonadism, atrophic gastritis, celiac disease, myasthenia gravis, vitiligo, and/or alopecia. Infectious adenalitis can be caused by tuberculous adrenalitis, HIV/AIDS, fungal infections, cytomegalovirus, syphilis, or African trypanosomiasis.25
Primary adrenal insufficiency can present acutely from bilateral adrenal hemorrhage in patients with antiphospholipid syndrome or other coagulation disorders.27 Some drugs may induce adrenal insufficiency. (See Table 2.) Other causes of primary adrenal insufficiency include adrenal metastases (mainly from lung, stomach, breast, or colon cancer); adrenal infiltration in lymphoma; amyloidosis, sarcoidosis, or hemochromatosis; and bilateral adrenalectomy.
Table 2. Examples of Drugs That Interfere With Adrenal Function |
|
|
Drugs |
Mechanism |
|
Phenytoin, rifampicin, phenobarbital, troglitazone, mitotane, topiramate, levothyroxine |
Increase metabolism of glucocorticoids |
|
Mifepristone, chlorpromazine, imipramine |
Impaired glucocorticoid action |
|
All exogenous glucocorticoids, including topical, inhaled, oral, intra-articular, parenteral; megestrol acetate, medroxyprogesterone acetate, cyproterone acetate, opiates |
Suppress hypothalamic-pituitary-adrenal axis |
|
Ketoconazole, fluconazole, itraconazole, etomidate, metyrapone, aminoglutethimide, trilostane |
Inhibit enzymes involved in cortisol production |
|
Heparin, warfarin, and other anticoagulants |
Adrenal hemorrhage |
|
Ipilimumab, nivolumab |
Autoimmune hypophysitis |
Central Adrenal Insufficiency
Central adrenal insufficiency is the collective classification of secondary and tertiary adrenal insufficiency, and is due to impaired production or action of ACTH. (See Figure 2.) Secondary adrenal insufficiency specifically results from pituitary disease. Potential etiologies are pituitary tumors (including pituitary resection or irradiation), traumatic brain injury, pituitary apoplexy, Sheehan syndrome (peripartum pituitary apoplexy and necrosis), infections, and infiltrative processes. Infections and infiltrative processes can include tuberculosis, meningitis, sarcoidosis, actinomycosis, histiocytosis X, Wegener’s granulomatosis, hemochromatosis, and lymphocytic hypophysitis. ACTH deficiency can be isolated or it can occur with deficiencies in other pituitary hormones. An isolated ACTH deficiency is more likely to be due to an autoimmune process and can be associated with other autoimmune endocrine disorders, such as thyroiditis and type 1 diabetes mellitus. Lastly, genetic causes of ACTH deficiency include loss of function gene mutations.25
Tertiary adrenal insufficiency is caused by disorders of the hypothalamus, resulting in impaired synthesis of CRH, which leads to inhibition of ACTH secretion from the pituitary gland. (See Figure 2.) The most common cause is suppression of the hypothalamic-pituitary-adrenal (HPA) axis by long-term exogenous glucocorticoid use with subsequent withdrawal of the exogenous glucocorticoids. All routes of glucocorticoid administration (oral, inhaled, intranasal, topical, intra-articular, intramuscular, and intravenous) have been associated with suppression of the HPA axis. Suppression can occur with glucocorticoid doses of 5 mg or higher of prednisolone equivalent for longer than four weeks.28 Treatment of Cushing’s syndrome also can lead to tertiary adrenal insufficiency by a similar mechanism. In Cushing’s syndrome, patients have persistent high serum cortisol concentrations, which can suppress the HPA axis. High-dose opiates also have been recognized as a cause of HPA suppression.29 In patients with secondary and tertiary adrenal insufficiency, the adrenal glands typically still are functional, but the lack of hormonal stimulation by ACTH from the pituitary gland or CRH from the hypothalamus leads to decreased cortisol secretion by the adrenal glands. Aldosterone secretion is preserved in this case because it is under feedback control by the renin-angiotensin system.
Adrenal Crisis
Adrenal crisis can present in patients with existing adrenal insufficiency during a major stress event, such as infection or trauma, but it also may be the first presentation of acute cortisol deficiency. Adrenal crisis is associated more often with primary adrenal insufficiency because the mineralocorticoid insufficiency can cause more severe electrolyte and fluid balance disturbances.8 Patients with central adrenal insufficiency are at lower risk of having an adrenal crisis because of preserved mineralocorticoids.23
Many factors may precipitate an adrenal crisis. Infections are considered to be the most common causes. Gastroenteritis is especially hazardous because vomiting and diarrhea impair adequate absorption of glucocorticoids for patients taking maintenance oral therapy.3,30 Systemic bacterial infection initiates an inflammatory cytokine response that leads to cortisol release and reduction in inflammation in patients with an intact HPA axis. However, in patients with hypoadrenalism, the inability to raise the cortisol level can lead to an unrestrained inflammatory response. This response leads to significant tissue damage and systemic effects and is the suspected mechanism resulting in adrenal crisis.14
Certain medications may trigger an adrenal crisis. (See Table 2.) Clinicians always should consider adrenal crisis in cancer patients taking immunotherapy such as ipilimumab and nivolumab, which can cause severe hypophysitis or adrenalitis.15 Thyrotoxicosis or the initiation of thyroid hormone replacement in a patient with undiagnosed adrenal insufficiency also can lead to an adrenal crisis by increasing cortisol metabolism.31,32
Certain disease entities and physiologic states are associated with an increased susceptibility to adrenal crisis. For example, patients with diabetes mellitus are at a higher risk of having an adrenal crisis for unknown reasons.3,33 Pregnant patients have increased glucocorticoid requirements, making them susceptible to adrenal crisis.3 Other factors that increase a patient’s predisposition to an adrenal crisis include psychological/cognitive disorders and/or social isolation, which may impair the patient’s ability to manage his or her disease process. For example, a cognitively impaired patient may fail to take glucocorticoids at stress dosing according to instructions, increasing adrenal crisis risk.
Critical Illness-Related Corticosteroid Insufficiency
Patients who are critically ill activate the HPA axis, leading to increased glucocorticoid production. Cortisol plays a vital role in the systemic response to infection. It contributes to energy generation by shifting to carbohydrate, fat, and protein metabolism. In addition, cortisol increases intravascular fluid retention, enhances inotropic and vasopressor responses, and has anti-inflammatory effects. All of these responses are crucial in critical illnesses such as sepsis.
Cortisol is important for immunomodulation, and insufficient cortisol levels can lead to excess circulating pro-inflammatory cytokines. Elevation of inflammatory cytokines in critically ill patients has been shown to correlate with disease severity, hospital mortality, and adverse long-term outcomes.32,34-36 In some critically ill patients, the HPA axis can become dysregulated, leading to inadequate cellular corticosteroid activity. The underlying mechanisms of how this occurs are multifactorial, complex, and poorly understood. It has been theorized that HPA axis dysregulation may be due to impaired ACTH release and synthesis, and tissue resistance to glucocorticoids.21,23,37-39
Cortisol levels can vary in critically ill patients, making diagnosis of adrenal insufficiency challenging. The term “critical illness-related corticosteroid insufficiency” (CIRCI) was introduced by the American College of Critical Care Medicine to describe this HPA axis dysfunction that occurs during critical illness.21,40 CIRCI is thought to occur in sepsis, septic shock, severe community-acquired pneumonia, acute respiratory distress syndrome, cardiac arrest, head injury, trauma, and burns, and after major surgery.41
Some critically ill patients experience direct structural damage to the adrenal glands from hemorrhage or infarction, also known as Waterhouse-Friderichsen syndrome (WFS). Predisposing factors for WFS include renal failure, disseminated intravascular coagulation, and anticoagulant or tyrosine kinase inhibitor use.42 Impaired blood flow to the pituitary gland leading to ischemia or necrosis also can contribute to a decrease in hypothalamic-pituitary hormone secretion for critically ill patients.43,44 Sepsis is associated with necrosis or hemorrhage within the HPA axis.42 Adrenal insufficiency is a negative prognostic factor for critically ill patients and is associated with an increased mortality rate in septic patients.21
Major trauma can cause tissue necrosis, hemorrhage, and ischemia-reperfusion injuries, leading to initiation of the inflammatory cascade. Patients with severe trauma may develop CIRCI, which can be associated with uncontrolled inflammation, vasopressor dependency, and poor clinical outcomes.40,45 Non-critically ill cirrhosis patients may be affected by adrenal insufficiency, with sources citing a prevalence as high as 47% in patients with cirrhosis,46 leading to the new term “hepato-adrenal” syndrome.
Clinical Features
The signs and symptoms of adrenal insufficiency are highly variable, vague, and nonspecific. For example, patients may present with fatigue, generalized weakness, lethargy, anorexia, weight loss, nausea, vomiting, abdominal pain, mental status changes, and hypotension. Patients with diabetes mellitus can develop an unexplained reduction in insulin requirement.47 Cardiomyopathy also has been described as a rare cardiac manifestation of adrenal insufficiency.48 Clinical features of mineralocorticoid deficiency specifically include salt cravings, myalgias, muscle cramps, and postural dizziness or orthostatic hypotension. Androgen deficiencies can result in decreased axillary and pubic hair (in women), reduced strength, lack of energy or stamina, dry and itchy skin, and loss or impairment of libido (in women). Because of the wide array of presenting symptoms, patients may present to the ED with chief complaints such as weakness, dizziness, or abdominal pain, which could elicit a wide differential diagnosis.
Primary Adrenal Insufficiency
The clinical features of primary adrenal insufficiency result from the deficiency of all adrenocortical hormones, including aldosterone, cortisol, and androgens. Skin hyperpigmentation occurs in 90% of patients with chronic primary adrenal insufficiency and is caused by stimulation of melanocortin receptors by elevated ACTH levels. Changes typically appear on skin creases, scars, the extensor surface of the elbows, knuckles, lips, and gingival mucosa.8,9
Diagnosis of adrenal insufficency should be considered in all patients who present with unexplained collapse and hypotension, especially those with vomiting or diarrhea. Hyperpigmentation, hyponatremia, hyperkalemia, metabolic acidosis, and hypoglycemia should increase the clinical suspicion.49
Central Adrenal Insufficiency
The clinical features of secondary and tertiary adrenal insufficiency are due to isolated glucocorticoid deficiency, as secretion of aldosterone and adrenal androgens is not affected. Additionally, hyperpigmentation will not be present. Patients with pituitary gland abnormalities may have signs and symptoms of deficiency of other anterior pituitary hormones. Vision may be compromised if there is compression of the optic chiasm by a pituitary lesion.
Adrenal Crisis
Adrenal crisis can be the first presentation of adrenal insufficiency. Frequent concomitant features include acute abdominal symptoms, delirium, obtundation, hyponatremia, hyperkalemia, hypoglycemia, and fever.31 (See Table 3.) Currently, there is no universally accepted clinical definition of what constitutes an adrenal crisis. Creating a definition is difficult because there are no diagnostic features or set of common clinical features that have sufficient specificity for a diagnosis of adrenal crisis. Roshworth et al proposed a new definition based on general acceptance of the central physiologic abnormality.23 They defined an adrenal crisis as: “An acute deterioration in health that is associated with absolute (systolic BP < 100 mmHg) or relative (systolic BP < 20 mmHg lower than the patient’s usual BP) hypotension, the features of which resolve following parenteral glucocorticoid administration (demonstrated by a marked resolution of hypotension within 1 hour and improvement of clinical symptoms over 2 hours).”23
Table 3. Clinical Features of Adrenal Crisis |
|
|
History Findings |
Severe weakness, abdominal pain, nausea, vomiting, altered mental status |
|
Physical Exam Findings |
Hypotension, fever, lethargy, abdominal tenderness and guarding, reduced level of consciousness |
|
Diagnostic/Laboratory Findings |
Hyponatremia, hyperkalemia, hypoglycemia, hypercalcemia |
Critical Illness-Related Corticosteroid Insufficiency
CIRCI is associated with increased circulation of biological markers of inflammation and coagulation and inadequate glucocorticoid-mediated anti-inflammatory activity for the severity of the patient’s critical illness.40 Symptoms can be widespread and include systemic, neurological, cardiovascular, digestive, and respiratory effects. (See Table 4.)
Table 4. Clinical Features of Critical Illness-Related Corticosteroid Insufficiency |
|
|
History Findings |
Altered mental status, nausea/vomiting, poor oral intake, weakness |
|
Physical Exam Findings |
Fever, lethargy, reduced level of consciousness, hypotension refractory to fluid resuscitation, decreased sensitivity to catecholamines and vasopressors, high cardiac index, persistent hypoxia |
|
Diagnostic/Laboratory Findings |
Hypoglycemia, hyponatremia, hyperkalemia, metabolic acidosis, hypereosinophilia, hemorrhage or necrosis seen in the pituitary gland, hypothalamus, or adrenal glands |
Differential Diagnosis
A patient with acute adrenal insufficiency or adrenal crisis may present with undifferentiated shock or with vague complaints that encompass a wide range of potential diagnoses. Many of the symptoms of adrenal crisis are similar to those of septic shock. The clinical picture can be obscured as sepsis itself can contribute to HPA axis dysfunction.38 Many patients with adrenal insufficiency may present with abdominal pain, nausea, and/or vomiting with alterations in level of consciousness mimicking diabetic ketoacidosis. Profound dehydration may be present with similar signs and symptoms, including weakness, hypotension, and decreased level of consciousness.
Other conditions that may present similarly include thyroid disorders, early pregnancy (including nausea and vomiting of pregnancy or hyperemesis gravidarum), cyclical vomiting syndrome, mild dehydration, mild electrolyte abnormalities, and psychiatric disorders such as generalized anxiety disorder or major depression. Patients with central causes of adrenal insufficiency may have symptoms related to an intracranial pathology and may elicit a completely different differential diagnosis, such as tension, migraine, or cluster headaches or intracranial mass or hemorrhage.
Diagnostic Studies
In the ED setting, there are no specific diagnostic criteria because other serious illnesses can mimic the features of adrenal insufficiency. Moreover, classic signs, such as hyperpigmentation seen in primary adrenal insufficiency, may not be present.15 Therefore, it is important to have a high index of suspicion for acute adrenal insufficiency in patients with undifferentiated shock.
In patients who are suspected to have adrenal insufficiency, there are three specific aims:
- To determine if the cortisol level is inappropriately low;
- To determine whether the adrenal insufficiency is intrinsic to the adrenal glands (primary) or secondary to pituitary/hypothalamic dysfunction (central); and
- To delineate underlying pathological processes.
This diagnostic workup typically is conducted in an outpatient or inpatient setting by the patient’s primary care physician or specialist.25 The emergency physician may facilitate the diagnosis of adrenal insufficiency in suspected patients by sending paired measurement of serum cortisol and ACTH levels. (See Table 5.)
Table 5. Testing for Adrenal Insufficiency31 |
||
|
Test Name |
Primary Adrenal Insufficiency Findings |
Central Adrenal Insufficiency Findings |
|
Cortisol Testing: Measurement of serum cortisol |
Low (random level |
Low (random level |
|
Cosyntropin Testing: ACTH stimulation test — parenteral administration of 250 mcg of ACTH with subsequent serum cortisol testing at 30 and 60 minutes after administration; can be done at any time of day |
< 18-20 mcg/dL after injection (97% sensitive)50 |
< 18-20 mcg/dL after injection (57-61% sensitive)50 |
|
ACTH Testing: Measurement of serum ACTH |
High ACTH |
Low ACTH or inappropriately normal |
|
ACTH = adrenocorticotropin |
||
It is important to check a complete blood count with a differential, serum electrolytes, and renal function in patients who are suspected to have adrenal insufficiency. Ninety percent of patients with primary adrenal insufficiency will present with hyponatremia and 50% will have hyperkalemia.8 Other common laboratory abnormalities include elevated serum creatinine, hypercalcemia, hypoglycemia, normocytic anemia, eosinophilia, and lymphocytosis. Thyroid-stimulating hormone (TSH) level may be elevated with a normal free T4 level in patients without thyroid disease because of the feedback mechanism of glucocorticoids on TSH release.8,17
Patients with central adrenal insufficiency will present with electrolyte disturbances less commonly, and hyperkalemia will not be present because of the preservation of mineralocorticoid secretion. However, hyponatremia still may occur as a result of a mild syndrome of inappropriate antidiuretic hormone (SIADH) due to increased vasopressin secretion from the hypothalamus, leading to impaired water excretion.51
In patients with acute adrenal crisis, laboratory findings may include hyponatremia, hyperkalemia, hypoglycemia (usually more common in children than adults), and hypercalcemia.
Currently, there are no agreed upon diagnostic tests or laboratory values for the diagnosis of CIRCI. Corticotropin testing is controversial in CIRCI and is not recommended in current clinical practice guidelines.52
Management
In patients with suspected acute adrenal crisis, initiate treatment as soon as possible without waiting for the results of diagnostic testing or procedures.17 For patients with a known history of adrenal insufficiency, steroid medication ideally should be administered within 15 minutes of recognition of possible adrenal crisis.15
Therapeutic principles for patients with adrenal crisis are directed at fluid resuscitation and steroid replacement. (See Table 6.) Fluid administration is based on the patient’s volume status and serum sodium concentration. Recommendations for initial rehydration include 1 liter of intravenous 0.9% saline administered over the first 60 minutes, with frequent hemodynamic monitoring guiding further fluid administration.3,8,17 Steroid replacement is initiated once an adrenal crisis is recognized by administering an initial dose of hydrocortisone 100 mg intravenously (IV) or intramuscularly (IM), followed by either a continuous IV infusion of 200 mg daily or 50 mg injections four times daily until the patient is hemodynamically stable. Patients who are clinically stable may be tapered to a maintenance dose over the following 24 to 72 hours. Hydrocortisone has sufficient mineralocorticoid activity to obviate the need for additional mineralocorticoids, such as fludrocortisone, during initial resuscitation. Once the daily hydrocortisone dose is less than
50 mg, mineralocorticoid administration typically is started.28
Table 6. Summary of Adrenal Crisis Management |
|
The identification and treatment of the precipitating cause or stressor is essential in managing patients with adrenal crisis.
Ensure that there is close monitoring of the patient’s vital signs, regular assessment of serum electrolytes, and management of other medical comorbidities. The patient may require continuous IV infusions of 5% dextrose to correct hypoglycemia.3 In patients with hyponatremia, exercise caution in correction of sodium to < 10 mEq over 24 hours to prevent osmotic demyelination syndrome.
Prevention of adrenal crisis requires early recognition of potential stressors. For patients with known adrenal insufficiency taking glucocorticoid replacement who develop an acute illness, glucocorticoid doses must be adjusted appropriately according to illness severity. For patients with fever or an infection requiring antibiotics or those who require a surgical procedure under local anesthesia, the daily glucocorticoid dose should be increased.31 A common rule for stress dosing of glucocorticoids is the “3 × 3 rule,” which incorporates tripling the glucocorticoid dose for three days.14 Patients with severe diarrhea or vomiting who cannot tolerate oral intake or whose absorption of oral medication may be affected may present to the ED for parenteral therapy.14
Patients with a known diagnosis of primary adrenal insufficiency may run out of or stop taking their prescribed medications. The recommended hydrocortisone regimen for patients with primary adrenal insufficiency is 15 to 25 mg daily in two or three split doses, with the first dose immediately after waking and the last dose not less than six hours before bedtime, and fludrocortisone 50 to 200 mcg as a single daily dose.17
Disposition
Patients in adrenal crisis or suspected of having CIRCI will require hospital admission, and likely intensive care, depending on their hemodynamic stability. Patients with vomiting and diarrhea who are otherwise stable may continue to have impaired absorption of their oral glucocorticoids. These patients are appropriate for hospital admission or observation to ensure appropriate glucocorticoid maintenance therapy, rehydration, and monitoring for signs of impending adrenal crisis. Patients who are stable, have the ability to continue their oral glucocorticoid regimen, and are able to follow up with an outpatient physician promptly may be discharged home. Ensure that these patients have a sufficient supply of hydrocortisone and fludrocortisone, including the additional doses required for illness. Some patients may use a hydrocortisone emergency injection kit (vials of 100 mg hydrocortisone, syringes, and needles) to inject glucocorticoid doses intramuscularly in the event of severe illness, trauma, or persistent vomiting if there will be a delay obtaining medical care.28 All patients should carry a steroid dependency card or wear medical alert bracelets or similar identification.
Controversies
CIRCI
Patients who have an established diagnosis of primary or central adrenal failure, patients taking chronic glucocorticoids before critical illness, and patients experiencing acute adrenal crisis should be treated with systemic glucocorticoids. Hydrocortisone is the preferred corticosteroid and has been used in the treatment of hemodynamic instability in patients with CIRCI, particularly in catecholamine-dependent septic shock. However, identification of patients who may benefit from steroid replacement therapy is controversial.38,53-56 Some research has shown a mortality benefit, while other research has shown no benefit compared to placebo in septic patients.53,55,56 In addition, hydrocortisone therapy may increase a patient’s risk for new infection, hypernatremia, weakness, and hyperglycemia.38
Varying proposed dosing strategies have been cited in the literature for septic “relative adrenal failure,” with some regimens including mineralocorticoid treatment (fludrocortisone) in conjunction with hydrocortisone.27,38,55,57 Traditional treatment is hydrocortisone 200 mg IV daily for three days. The 2016 Surviving Sepsis Campaign guidelines recommend avoiding hydrocortisone in septic shock if adequate fluid resuscitation and vasopressor therapy are able to restore hemodynamic instability. Corticosteroids typically are not recommended in major trauma patients, including those with traumatic brain injury and patients with refractory hypotension.58,59 For patients with acute spinal cord injury, treatment with methylprednisolone has been shown to improve movement, but must be started soon after the injury has occurred, within no more than eight hours.60
Etomidate and Adrenal Insufficiency
Etomidate is a common medication used for induction in rapid sequence intubation (RSI) because of its pharmacokinetic properties and hemodynamic stability.61,62 It has been shown to cause adrenal insufficiency by affecting cortisol synthesis for up to 72 hours, but the clinical consequence of a single dose is controversial.61 When used as an infusion, it has been found to affect mortality and subsequently is no longer used for prolonged sedation.63 Some research has shown single-dose etomidate to increase rates of adrenal insufficiency and mortality in sepsis patients.64 However, evidence has not been consistent, as other literature has shown no significant differences.65-67 Some research has shown that mortality was increased specifically for patients taking multiple vasopressors or with abdominal source of infection.68 Because outcomes such as vasopressor use, hypotension, and mortality in patients with sepsis and severe sepsis still remain to be fully characterized, there are no recommendations or guidelines on etomidate use in these patients. Consider the patient’s hemodynamic status and consider alternative medications for induction in RSI when possible.69,70
Summary
The cortex of the adrenal glands is responsible for secreting glucocorticoids, mineralocorticoids, and androgens. These hormones are under regulation by the HPA axis, with feedback inhibition by CRH, ACTH, and vasopressin. Primary adrenal insufficiency occurs because of primary adrenal failure, whereas secondary and tertiary insufficiency are due to pituitary and hypothalamic causes. Autoimmune adrenal insufficiency is the most common etiology for primary adrenal insufficiency. Pituitary adenoma is the most frequent disease process leading to secondary adrenal insufficiency. The most common cause of tertiary adrenal insufficiency is long-term exogenous glucocorticoid use.
Adrenal insufficiency continues to be associated with significant morbidity and increased risk of mortality. Making the diagnosis of adrenal insufficiency is challenging because patients may present with a wide variety of signs and symptoms and have vague clinical features. Adrenal crisis is a life-threatening emergency and complication in patients with adrenal insufficiency consisting of acute hemodynamic decompensation. CIRCI is due to HPA axis dysfunction and may require treatment with corticosteroids if shock is refractory to vasopressors and fluid resuscitation. There is no current recommendation for adrenal function testing in the ED.
Treatment is geared toward having a high clinical suspicion and administering IV fluids and glucocorticoids as necessary, while also monitoring hemodynamics and electrolytes. Patients with adrenal insufficiency require measures such as stress dosing of glucocorticoids to prevent adrenal crisis.
REFERENCES
- Erichsen MM, Lovas K, Fougner KJ, et al. Normal overall mortality rate in Addison’s disease, but young patients are at risk of premature death. Eur J Endocrinol 2009;160:233-237.
- Bleicken B, Hahner S, Ventz M, Quinkler M. Delayed diagnosis of adrenal insufficiency is common: A cross-sectional study in 216 patients. Am J Med Sci 2010;339:525-531.
- Puar TH, Stikkelbroeck NM, Smans LC, et al. Adrenal crisis: Still a deadly event in the 21st century. Am J Med 2016;129:339 e1-9.
- Hahner S, Loeffler M, Bleicken B, et al. Epidemiology of adrenal crisis in chronic adrenal insufficiency: The need for new prevention strategies. Eur J Endocrinol 2010;162:597-602.
- Wass JA, Arlt W. How to avoid precipitating an acute adrenal crisis. BMJ 2012;345:e6333.
- Erichsen MM, Lovas K, Skinningsrud B, et al. Clinical, immunological, and genetic features of autoimmune primary adrenal insufficiency: Observations from a Norwegian registry. J Clin Endocrinol Metab 2009;94:4882-4890.
- Kong MF, Jeffcoate W. Eighty-six cases of Addison’s disease. Clin Endocrinol (Oxf) 1994;41:757-761.
- Pazderska A, Pearce SH. Adrenal insufficiency — recognition and management. Clin Med (Lond) 2017;17:258-262.
- Arlt W, Allolio B. Adrenal insufficiency. Lancet 2003;361:1881-1893.
- Bates AS, Van’t Hoff W, Jones PJ, Clayton RN. The effect of hypopituitarism on life expectancy. J Clin Endocrinol Metab 1996;81:1169-1172.
- Regal M, Paramo C, Sierra SM, Garcia-Mayor RV. Prevalence and incidence of hypopituitarism in an adult Caucasian population in northwestern Spain. Clin Endocrinol (Oxf) 2001;55:735-740.
- Gomez MT, Magiakou MA, Mastorakos G, Chrousos GP. The pituitary corticotroph is not the rate limiting step in the postoperative recovery of the hypothalamic-pituitary-adrenal axis in patients with Cushing syndrome. J Clin Endocrinol Metab 1993;77:173-177.
- Hahner S, Spinnler C, Fassnacht M, et al. High incidence of adrenal crisis in educated patients with chronic adrenal insufficiency: A prospective study. J Clin Endocrinol Metab 2015;100:407-416.
- Rushworth RL, Torpy DJ. A descriptive study of adrenal crises in adults with adrenal insufficiency: Increased risk with age and in those with bacterial infections. BMC Endocr Disord 2014;14:79.
- Amrein K, Martucci G, Hahner S. Understanding adrenal crisis. Intensive Care Med 2018;44:652-655.
- Betterle C, Scalici C, Presotto F, et al. The natural history of adrenal function in autoimmune patients with adrenal autoantibodies. J Endocrinol 1988;117:467-475.
- Husebye ES, Allolio B, Arlt W, et al. Consensus statement on the diagnosis, treatment and follow-up of patients with primary adrenal insufficiency. J Intern Med 2014;275:104-115.
- National Institute of Diabetes and Digestive and Kidney Diseases. Adrenal Insufficiency and Addison’s Disease. Bethesda, MD: NIH; 2014.
- Ekman B, Fitts D, Marelli C, et al. European Adrenal Insufficiency Registry (EU-AIR): A comparative observational study of glucocorticoid replacement therapy. BMC Endocr Disord 2014;14:1-7.
- Joseph RM, Hunter AL, Ray DW, Dixon WG. Systemic glucocorticoid therapy and adrenal insufficiency in adults: A systematic review. Semin Arthritis Rheum 2016;46:133-141.
- Kanczkowski W, Sue M, Zacharowski K, et al. The role of adrenal gland microenvironment in the HPA axis function and dysfunction during sepsis. Mol Cell Endocrinol 2015;408:241-248.
- Raff H, Sharma ST, Nieman LK. Physiological basis for the etiology, diagnosis, and treatment of adrenal disorders: Cushing’s syndrome, adrenal insufficiency, and congenital adrenal hyperplasia. Compr Physiol 2014;4:739-769.
- Rushworth RL, Torpy DJ, Falhammar H. Adrenal crises: Perspectives and research directions. Endocrine 2017;55:336-345.
- Betterle C, Morlin L. Autoimmune Addison’s disease. Endocr Dev 2011;20:161-172.
- Charmandari E, Nicolaides NC, Chrousos GP. Adrenal insufficiency. Lancet 2014;383:2152-2167.
- Mitchell AL, Pearce SH. Autoimmune Addison disease: Pathophysiology and genetic complexity. Nat Rev Endocrinol 2012;8:306-316.
- Presotto F, Fornasini F, Betterle C, et al. Acute adrenal failure as the heralding symptom of primary antiphospholipid syndrome: Report of a case and review of the literature. Eur J Endocrinol 2005;153:507-514.
- Bancos I, Hahner S, Tomlinson J, Arlt W. Diagnosis and management of adrenal insufficiency. Lancet Diabetes Endocrinol 2015;3:216-226.
- Gibb FW, Stewart A, Walker BR, Strachan MW. Adrenal insufficiency in patients on long-term opioid analgesia. Clin Endocrinol (Oxf) 2016;85:831-835.
- Allolio B. Extensive expertise in endocrinology. Adrenal crisis. Eur J Endocrinol 2015;172:R115-R124.
- Bornstein SR, Allolio B, Arlt W, et al. Diagnosis and treatment of primary adrenal insufficiency: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2016;101:364-389.
- Gomez HG, Gonzalez SM, Londono JM, et al. Immunological characterization of compensatory anti-inflammatory response syndrome in patients with severe sepsis: A longitudinal study. Crit Care Med 2014;42:771-780.
- Meyer G, Badenhoop K, Linder R. Addison’s disease with polyglandular autoimmunity carries a more than 2.5-fold risk for adrenal crises: German Health insurance data 2010-2013. Clin Endocrinol (Oxf) 2016;85:347-353.
- Aisiku IP, Yamal JM, Doshi P, et al. Plasma cytokines IL-6, IL-8, and IL-10 are associated with the development of acute respiratory distress syndrome in patients with severe traumatic brain injury. Crit Care 2016;20:288.
- Fernandez-Botran R, Uriarte SM, Arnold FW, et al. Contrasting inflammatory responses in severe and non-severe community-acquired pneumonia. Inflammation 2014;37:1158-1166.
- Yende S, D’Angelo G, Kellum JA, et al. Inflammatory markers at hospital discharge predict subsequent mortality after pneumonia and sepsis. Am J Respir Crit Care Med 2008;177:1242-1247.
- Elenkov IJ, Iezzoni DG, Daly A, et al. Cytokine dysregulation, inflammation and well-being. Neuroimmunomodulation 2005;12:255-269.
- Schurr JW, Szumita PM, DeGrado JR. Neuroendocrine derangements in early septic shock: Pharmacotherapy for relative adrenal and vasopressin insufficiency. Shock 2017;48:284-293.
- Silverman MN, Sternberg EM. Glucocorticoid regulation of inflammation and its functional correlates: From HPA axis to glucocorticoid receptor dysfunction. Ann N Y Acad Sci 2012;1261:55-63.
- Annane D, Pastores SM, Rochwerg B, et al. Guidelines for the diagnosis and management of critical illness-related corticosteroid insufficiency (CIRCI) in critically ill patients (Part I): Society of Critical Care Medicine (SCCM) and European Society of Intensive Care Medicine (ESICM) 2017. Intensive Care Med 2017;43:1751-1763.
- Annane D, Pastores SM, Arlt W, et al. Critical illness-related corticosteroid insufficiency (CIRCI): A narrative review from a Multispecialty Task Force of the Society of Critical Care Medicine (SCCM) and the European Society of Intensive Care Medicine (ESICM). Intensive Care Med 2017;43:1781-1792.
- Annane D. The role of ACTH and corticosteroids for sepsis and septic shock: An update. Front Endocrinol (Lausanne) 2016;7:70.
- Cohen J, Venkatesh B. Relative adrenal insufficiency in the intensive care population: Background and critical appraisal of the evidence. Anaesth Intensive Care 2010;38:425-436.
- Moraes RB, Czepielewski MA, Friedman G, Borba EL. Diagnosis of adrenal failure in critically ill patients. Arq Bras Endocrinol Metabol 2011;55:295-302.
- Hoen S, Asehnoune K, Brailly-Tabard S, et al. Cortisol response to corticotropin stimulation in trauma patients: Influence of hemorrhagic shock. Anesthesiology 2002;97:807-813.
- Singh RR, Walia R, Sachdeva N, et al. Relative adrenal insufficiency in cirrhotic patients with ascites (hepatorenal syndrome). Dig Liver Dis 2018;50:1232-1237.
- Andrioli M, Pecori Giraldi F, Cavagnini F. Isolated corticotrophin deficiency. Pituitary 2006;9:289-295.
- Alkhateeb M, Alsakkal M, Alfauri MN, Alasmar D. Reversible dilated cardiomyopathy as a complication of adrenal cortex insufficiency: A case report. J Med Case Rep 2018;12:345.
- Laureti S, Vecchi L, Santeusanio F, Falorni A. Is the prevalence of Addison’s disease underestimated? J Clin Endocrinol Metab 1999;84:1762.
- Hamilton DD, Cotton BA. Cosyntropin as a diagnostic agent in the screening of patients for adrenocortical insufficiency. Clin Pharmacol 2010;2:77-82.
- Verbalis JG, Goldsmith SR, Greenberg A, et al. Diagnosis, evaluation, and treatment of hyponatremia: Expert panel recommendations. Am J Med 2013;126:S1-S42.
- Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med 2017;43:304-377.
- Annane D, Sebille V, Charpentier C, et al. Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. JAMA 2002;288:862-871.
- Long B, Koyfman A. Controversies in corticosteroid use for sepsis. J Emerg Med 2017;53:653-661.
- Sprung CL, Annane D, Keh D, et al. Hydrocortisone therapy for patients with septic shock. N Engl J Med 2008;358:111-124.
- Venkatesh B, Finfer S, Cohen J, et al. Adjunctive glucocorticoid therapy in patients with septic shock. N Engl J Med 2018;378:797-808.
- Boonen E, Van den Berghe G. Endocrine responses to critical illness: Novel insights and therapeutic implications. J Clin Endocrinol Metab 2014;99:1569-1582.
- Hoshide R, Cheung V, Marshall L, et al. Do corticosteroids play a role in the management of traumatic brain injury? Surg Neurol Int 2016;7:84.
- Kim G, Young J. Clinical characteristics of trauma patients requiring hydrocortisone treatment for refractory hypotension. Am Surg 2017;83:821-824.
- Bracken MB. Steroids for acute spinal cord injury. Cochrane Database Syst Rev 2012;1:CD001046.
- Groth CM, Acquisto NM, Khadem T. Current practices and safety of medication use during rapid sequence intubation.
J Crit Care 2018;45:65-70. - Dettmer MR, Dellinger RP. The use of etomidate for rapid sequence induction in septic patients. J Thorac Dis 2015;7:1684-1686.
- Ledingham IM, Watt I. Influence of sedation on mortality in critically ill multiple trauma patients. Lancet 1983;1:1270.
- Chan CM, Mitchell AL, Shorr AF. Etomidate is associated with mortality and adrenal insufficiency in sepsis: A meta-analysis. Crit Care Med 2012;40:2945-2953.
- McPhee LC, Badawi O, Fraser GL, et al. Single-dose etomidate is not associated with increased mortality in ICU patients with sepsis: Analysis of a large electronic ICU database. Crit Care Med 2013;41:774-783.
- Alday NJ, Jones GM, Kimmons LA, et al. Effects of etomidate on vasopressor use in patients with sepsis or severe sepsis: A propensity-matched analysis. J Crit Care 2014;29:517-522.
- Gu WJ, Wang F, Tang L, Liu JC. Single-dose etomidate does not increase mortality in patients with sepsis: A systematic review and meta-analysis of randomized controlled trials and observational studies. Chest 2015;147:335-346.
- Rech MA, Bennett S, Chaney W, Sterk E. Risk factors for mortality in septic patients who received etomidate. Am J Emerg Med 2015;33:1340-1343.
- Pantanwala AE, McKinney CB, Erstad BL, Sakles JC. Retrospective analysis of etomidate versus ketamine for first-pass intubation success in an academic emergency department. Acad Emerg Med 2014;21:87-91.
- Jabre P, Combes X, Lapostolle F, et al; KETASED Collaborative Study Group. Etomidate versus ketamine for rapid sequence intubation in acutely ill patients: A multicenter randomized controlled trial. Lancet 2009;374:293-300.
This article addresses the pathophysiology, discusses various clinical presentations, and reviews current evidence-based practices for managing adrenal insufficiency and crisis in the emergency department.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
