Stroke: The Subtle, Atypical, and Enigmatic
March 15, 2019
Reprints
AUTHORS
Tiffany Murano, MD, Associate Professor, Department of Emergency Medicine, Rutgers New Jersey Medical School, Newark
Katie Myers, MD, Department of Emergency Medicine, Rutgers New Jersey Medical School, Newark
PEER REVIEWER
Steven M. Winograd, MD, FACEP, Assistant Clinical Professor of Emergency Medicine, Mt. Sinai Medical School, NY
EXECUTIVE SUMMARY
- Consider acute ischemic stroke whenever neurologic symptoms develop over a few minutes.
- Although the classic sign of stroke is motor weakness, visual impairment, headache, language deficits, dizziness, amnesia, and altered mental status also can be due to stroke.
- Acute arterial dissection almost always presents with neck pain, face pain, or headache. Patients who present with such pain as well as neurologic symptoms should receive imaging with CT angiography.
- Unilateral occipital headache may occur in patients with an acute infarction involving the posterior inferior cerebellar artery.
- In patients with acute vertigo, the presence of a normal head impulse test, direction-changing nystagmus, or skew deviation of gaze fixation suggests a central origin.
- Impairment in alertness and behavioral abnormalities are common in rostral brainstem strokes, especially those due to blockage in the distal tip of the basilar artery.
- Cerebral venous thrombosis is a rare condition that manifests as a headache that is typically worse when supine, and some patients may experience vomiting, visual problems, and motor weakness.
Introduction
Stroke has been recognized in medicine since the time of Hippocrates, 2,400 years ago. Initially called “apoplexy,” meaning “to be struck down by violence” in Greek, stroke was diagnosed by the way it suddenly devastated a person’s ability to function. The aftermath of stroke and the disability it causes have been affecting humankind ever since. Sometimes, the signs and symptoms of stroke are readily apparent to patients and their family members, thanks in large part to the Act FAST public health campaign, which is based on the research by Wall et al.1 This simple acronym reminds the public that symptoms of stroke include facial droop (F), arm weakness (A), and slurred speech (S). The last letter stands for time (T) and is meant to remind patients to act quickly and go to a hospital immediately.1 Emergency physicians (EPs), in consultation with stroke specialist colleagues, diagnose and treat these patients according to the guidelines published by the American Heart Association and the American Stroke Association.2
This article will explore the subtle and enigmatic presentations of stroke — the patients who likely will not present to the emergency department (ED) as a “code stroke” already diagnosed by lay people or EMS. These patients often will present with nonspecific symptoms, such as vision problems, headache, a subtle language deficit, dizziness, or amnesia. Additionally, patients who present with global altered mental status must trigger suspicion for stroke. This article will explore each of these chief complaints with respect to stroke and what constellation of symptoms and physical exam findings should prompt further workup. Then it will examine cerebral venous thrombosis (CVT), a rare type of stroke caused by a venous occlusion, and discuss how its various presentations make diagnosis elusive, particularly in the ED setting.
Epidemiology of Stroke
Every year, about 795,000 people in the United States are diagnosed with a stroke; approximately 610,000 of these are first strokes and 185,000 are recurrent strokes.3 Stroke is the fifth leading cause of death in the United States4,5 and a major cause of disability and loss of functional independence, particularly among the elderly. Stroke reduces mobility in more than half of stroke survivors older than 65 years of age.6 The lifetime risk of stroke for adults worldwide is 25%.6 Men have a higher incidence of stroke than women at younger ages, but the incidence is higher for women by 75 years of age and older.3
Background and Anatomy
The effects of a stroke depend primarily on the location of the blood flow obstruction and the extent of brain tissue affected. Strokes result in neurological dysfunction in the distribution of a vascular territory of the brain, so understanding the vascular territories and the function of the brain tissue they supply is critical to making the correct diagnosis. (See Table 1.) Acute ischemic strokes, which make up 87% of all strokes in the United States, are the main focus of this article. They can be caused by an acute thrombosis of a blood vessel, an embolism from a distant site, or a vascular dissection. Because of blood flow patterns and arterial properties such as size and origin, thrombosis, emboli, and dissection are more likely to affect certain blood vessels, including the internal cerebral artery (ICA) or middle cerebral artery (MCA). Dissections also are more likely to affect certain vessels, the most common of which is the extracranial portion of the internal carotid artery. They can cause ischemic symptoms because of hemodynamic compromise secondary to luminal narrowing or occlusion, thromboembolism, or both.8,9
Table 1. Brain Vascular Territories8-10 |
||
|
Vascular Territory |
Area of the Brain Affected |
Symptoms |
|
MCA |
The cortical branches of the MCA supply the lateral surface of the cortical hemisphere, except for the medial part of the frontal and the parietal lobe (ACA territory), and the inferior part of the temporal lobe (PCA territory). |
|
|
Lateral LSA |
Deep penetrating arteries of the MCA. Their territory includes most of the basal ganglia and the internal capsule. |
|
|
ACA |
The ACA supplies the medial part of the frontal and the parietal lobe and the anterior portion of the corpus callosum, basal ganglia, and internal capsule. |
|
|
Medial LSA |
Arise from the ACA. Heubner’s artery is the largest of the medial LSAs and supplies the anteromedial part of the head of the caudate and the anteroinferior internal capsule. |
|
|
AChA |
Arises from the ICA. Blood supply to the hippocampus, the posterior limb of the internal capsule and the thalamus |
|
|
ICA |
The ICA branches into the MCA, ACA, and AChA; therefore, the vascular territory is the combination of the above. |
|
|
PCA |
Posterior thalamoperforating arteries branch off the P1 segment and supply blood to the thalamus. Cortical branches of the PCA supply the inferomedial part of the temporal lobe, occipital pole, visual cortex, and splenium of the corpus callosum. |
|
|
VA |
The medulla |
|
|
BA |
The pons and midbrain |
|
|
MCA: middle cerebral artery; LSA: lenticulostriate arteries; ACA: anterior cerebral artery; AChA: anterior choroidal arteries; ICA: internal cerebral artery; PCA: posterior cerebral artery, VA: vertebral artery; BA: basilar artery (continued) |
||
Table 1. Brain Vascular Territories (continued)8-10 |
||
|
Vascular Territory |
Area of the Brain Affected |
Symptoms |
|
PICA |
The lateral medulla and the inferior occipital surface of the cerebellum |
|
|
AICA |
The lateral surface of the cerebellum |
|
|
SCA |
The superior and tentorial surface of the cerebellum |
|
|
Watershed |
Watershed infarcts occur at the border zones between major cerebral artery territories as a result of hypoperfusion. |
|
|
Venous thrombosis |
Venous territories |
|
|
PICA: posterior inferior cerebellar artery; AICA: anterior inferior cerebellar artery; SCA: superior cerebellar artery |
||
Stroke Syndromes
Although clinical manifestation of an acute stroke may vary, there are common features that may be aggregated and correlated with specific patterns or stroke syndromes. The following sections will discuss the signs, symptoms, workup, diagnosis, and management of possible strokes by various chief complaints. These include visual complaints, headache, language deficits, dizziness, amnesia, and altered mental status.
Visual Complaints
Visual impairment, in which the visual disturbance is a result of brain damage rather than a problem with the eye, encompasses a broad spectrum of diseases. This is due, in part, to the complex tracts that transmit efferent and afferent signals to the eyes, whether they are processing vision, adjusting light input, coordinating eye movements, or connecting vision with memories, speech, or sound. Conditions include cerebral visual impairment, visual neglect, visual agnosia, various visual perceptual disorders, visual field cuts, lack of facial recognition, and cortical blindness. This section will explore the location of strokes that may affect vision.
Visual complaints are the hallmark of posterior cerebral artery (PCA) territory infarcts because of the location of the visual cortex in the occipital lobe, which is supplied medially by the PCA. (See Figure 1.) The most frequent finding is homonymous hemianopsia. Macular sparing may be present but is variable. If only the inferior portion of the PCA territory is involved, patients will have a superior homonymous quadrantanopia. If only the superior portion of the PCA territory is involved, patients will have an inferior homonymous quadrantanopia. Patients may describe the visual defect as a void or blackness; however, when the entire PCA territory is involved (especially the nondominant hemisphere, which is usually the right), patients may have visual neglect and, therefore, are unaware of their deficit. These patients may present with walking into walls on their neglected side. Oculomotor and pupillary function is preserved in PCA infarcts. The PCA territory also involves the splenium of the corpus callosum, which connects the occipital lobe to the temporal and frontal lobes. Patients with dominant-hemisphere PCA territory infarcts (usually left) frequently also have alexia (loss of the ability to read) without agraphia (loss of the ability to write).11 This is because the visual cortex on the dominant side no longer is able to transmit information to the language centers (Broca’s area and Wernicke’s area). Infarction of the nondominant PCA territory often is accompanied by prosopagnosia (inability to recognize familiar faces) and an inability to visualize the location of places on a map. Additionally, the ability to perform mental visualization or have dreams with visual imagery may be absent.
Figure 1. CT Scan of the Head in a Patient Who Had Vision Loss in the Left Visual Field |
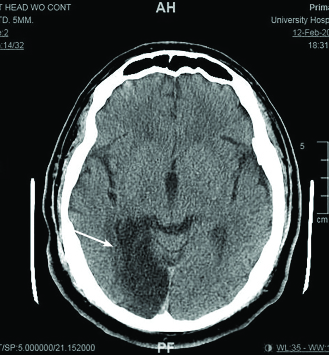 |
|
Imaging shows hypodensity in the right posterior cerebral artery vascular territory. |
Posterior thalamoperforating arteries branch off the P1 segment and supply blood to the thalamus, so patients with proximal P1 infarcts also may have symptoms related to thalamic ischemia. The hallmark of thalamic involvement is hemisensory loss in the face, limbs, and trunk. On examination, touch, pinprick, and position senses are reduced. The combination of hemisensory loss and homonymous hemianopsia is pathognomonic of a proximal PCA stroke.12,13
Vision also may be affected with ischemia in the distribution of the anterior choroidal artery (AChA), because its vascular territory includes the optic tracts and part of the thalamus that contains the lateral geniculate nucleus (LGN). The AChA is the most distal branch of the internal carotid artery (ICA) and originates just after the origin of the posterior communicating artery (PCOM). The afferent neurons that compose the optic tracts synapse in the LGN with the neurons of the optic radiations that transmit information to the visual cortex. With AChA strokes, the regions of infarct most often occur in the posterior limb of the internal capsule (containing the fibers of the corticospinal tract), the internal globus pallidus (iGP), and the LGN.10 As a result, the most common clinical syndrome includes a contralateral hemiparesis and contralateral visual field defect. The visual field defect can consist of a complete homonymous hemianopsia or may manifest as a distinct feature with sparing of the meridian, called sectoranopia. This is due to the arrangement of the afferent visual fibers as they synapse in the LGN. The likely cause of AChA infarction is small-artery focal atherosclerosis secondary to long-standing hypertension and diabetes.10
Transient visual loss (TVL), also known as amaurosis fugax, can occur in one or both eyes, and frequently prompts patients to seek medical attention in the ED. The term amaurosis comes from Greek meaning “dark” and fugax comes from Latin meaning “fleeting.”14 TVL reflects a heterogeneous group of disorders, some relatively benign and others with potential for significant neurological sequelae. Patients with TVL almost always present after the episode has resolved and, therefore, the neurological and ophthalmologic examinations usually are normal. Thus, the history is the most important aspect of the evaluation to elucidate the cause. Important historical features include whether the visual loss affected one or both eyes, the duration of the episode, and a specific description of the symptoms. The most common cause of ischemia to the retina or optic nerve is transient monocular visual loss (TMVL). The most common description of TMVL due to ischemia is blank or fuzzy vision with abrupt onset over a few minutes.15 Carotid artery disease with either plaque embolization or hypoperfusion is a common precipitant. The workup should include ophthalmologic evaluation and carotid imaging. Other considerations include cardiac evaluation, brain magnetic resonance imaging (MRI), electroencephalogram (EEG), hypercoagulable testing, and a workup of vasculitides.
Oculomotor abnormalities frequently are seen in strokes involving the midbrain and pons because of the location of the nuclei of cranial nerves III, IV, and VI, and the medial longitudinal fasciculus. Patients may complain of double vision and dizziness. On exam, they may exhibit oculomotor palsies and pupillary abnormalities. Sometimes these strokes are associated with global altered mental status, which is explained in more detail in the section on altered mental status.
Headache
Nontraumatic headache is a common chief complaint in the ED, accounting for 2-4% of all ED visits annually.16,17 The EP’s goal is to differentiate the small number of patients with life-threatening headaches from the overwhelming majority with benign primary headaches (i.e., migraine, tension, or cluster). Several studies have evaluated the frequency with which secondary (pathological) causes of headache are diagnosed. These studies show some variability, with rates as low as 4% for all headache patients to greater than 14% for those with sudden-onset, severe headaches.17
Patients with an acute arterial dissection are at risk for ischemic stroke. In 80-90% of these patients, acute headache, neck pain, or face pain occurs at symptom onset.18 Although arterial dissections are responsible for only 2% of all ischemic strokes, they are an important cause of stroke in young and middle-age patients. The average age of patients with cervicocerebral arterial dissections is 30 to 50 years, with a mean age of approximately 40 years.7 In patients younger than 45 years of age, arterial dissection is the second leading cause of stroke, accounting for 10-25% of ischemic strokes in this age group.8 Dissection occurs when the structural integrity of the arterial wall is compromised, allowing blood to collect between layers as an intramural hematoma. Strokes can occur either from diminished blood flow from luminal narrowing, or from the development of a thrombus from turbulent blood flow with embolization of a blood clot to a distant artery. Although dissection can result from major head and neck trauma, most dissections occur spontaneously or after minor or trivial injury. Other reported precipitating activities include childbirth, sexual intercourse, coughing or sneezing, and chiropractic neck manipulation.18
Several connective tissue or vascular disorders are associated with dissection, the most common of which is fibromuscular dysplasia. Approximately 15-20% of all cases of cervical dissection are associated with this condition.19 Other associated conditions include Ehlers-Danlos syndrome, Marfan syndrome, osteogenesis imperfecta, cystic medial necrosis, reticular fiber deficiency, homocystinuria, autosomal dominant polycystic kidney disease, alpha-1 antitrypsin deficiency, segmental mediolytic arteriopathy, reversible cerebral vasoconstriction syndrome, and cervical artery tortuosity. Although clinical features may raise the suspicion for dissection, the diagnosis is confirmed with neuroimaging. Therefore, in patients with both stroke-like symptoms and headache or neck pain, CT angiogram or MR angiogram imaging of the neck is warranted to rule out dissection.
Posterior inferior cerebellar artery (PICA) infarction may present with acute unilateral occipital headache, but also usually involves acute persistent vertigo, ataxia, dysmetria, and, occasionally, lateral medullary syndrome. (See Figure 2.) (Also see the section on dizziness.)
Figure 2. MRI Brain in a Patient Who Presented With Nausea, Vomiting, and Headache |
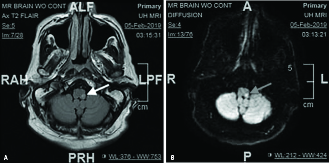 |
|
(A) MRI axial T2-flair. Arrow points to the area of infarct in the posterior inferior cerebellar artery; (B) Corresponding image on the MRI diffusion study. Arrow points to the area of infarct. |
Language Deficits
Although aphasia is a common symptom of stroke, it can be a subtle finding when in isolation. Language processing has three main centers in the brain. Broca’s area is located in the posterior inferior frontal gyrus of the dominant hemisphere and is critical for language production. Wernicke’s area is located in the superior temporal gyrus of the dominant hemisphere and is essential for language comprehension. The angular gyrus is located in the inferior parietal lobe near the visual tracts and is necessary for understanding written language. Other regions of the brain make important contributions to normal language ability. These include the insula, which is integral to normal articulation; several frontal and temporal lobe regions that support sentence-level processing; and large regions of the temporal, occipital, and parietal cortex that support knowledge of words and their meanings.20
Any pathologic process that results in dysfunction of the language network may cause aphasia. (See Table 2.) The most common etiology of acute-onset aphasia is ischemic stroke. Speech can be evaluated for fluency, content, repetition, naming, comprehension, reading, and writing. Based on a thorough assessment of language, a particular aphasia syndrome may be identified, thus allowing for localization to a particular region of the brain.
Table 2. Major Aphasia Syndromes20 |
|||
|
Aphasia Syndrome |
Characteristics |
Brain Territory |
Associated Symptoms |
|
Broca’s aphasia |
|
|
|
|
Wernicke’s aphasia |
|
|
|
|
Conduction aphasia |
|
|
|
|
Global aphasia |
|
|
|
|
Transcortical motor aphasia |
|
|
|
Dizziness
Dizziness and vertigo are common presenting symptoms in the ED, accounting for approximately 4% of all visits.21 The presence of nystagmus on physical exam suggests that dizziness is vertiginous in nature. The etiology of vertigo is classified into two categories, peripheral and central, the more common of which is peripheral. Vertigo, whether central or peripheral in origin, generally is accompanied by postural instability. Peripheral vertigo constitutes disorders such as benign paroxysmal peripheral vertigo, Meniere’s disease, and labyrinthitis. Although central causes are much less common, accounting for about 10% of all causes of dizziness, they can be much more devastating. In the elderly, the incidence of a central cause of vertigo approaches 20%.22 The most common cause of acute central vertigo is ischemic stroke. Patients may present with a similar constellation of symptoms as peripheral vestibular disorders, making the diagnosis more difficult.
However, the presence of subtle findings can differentiate patients with peripheral vertigo from those with a central cause such as stroke. The time course, provoking factors, and aggravating factors of vertigo are the most useful features in establishing the etiology. (See Table 3.) Stroke should be suspected in patients with acute, sustained vestibular syndrome, defined as the rapid onset (over seconds to hours) of vertigo, nausea, vomiting, and gait unsteadiness in association with head-motion intolerance and nystagmus lasting days to weeks.21 The HINTS examination — head impulse test, nystagmus, and test-of-skew — is used to distinguish between peripheral and central causes of vertigo. The presence of any one of three clinical signs (a normal head impulse test, direction-changing nystagmus, or a skew deviation) suggests central rather than peripheral vertigo in patients with an acute, sustained vestibular syndrome. Studies have repeatedly shown superiority of the HINTS exam in diagnosing stroke, even over MRI within the first 48 hours after symptom onset.23,24
In patients with lateral medullary infarcts, vestibulocerebellar symptoms and signs typically are present.25 These signs and symptoms are related to involvement of the vestibular nuclei and their connections and to involvement of the inferior cerebellar peduncle. Common symptoms and signs include feelings of dizziness or of being off-balance, difficulty sitting upright without support, blurred vision or diplopia, nystagmus, ocular torsion, limb ataxia, pain on the ipsilateral side of the face, loss of pain and temperature sensation in contralateral limbs, bulbar muscle weakness manifesting as hoarseness, dysphagia, and asymmetric palate elevation.11
Table 3. Clinical Features of Central vs. Peripheral Vertigo22 |
|
|
Peripheral |
Central |
|
Time Course
|
Time Course
|
|
Nystagmus
|
Nystagmus
|
|
Other Neurological Signs
|
Other Neurological Signs
|
|
Postural instability
|
Postural instability
|
|
Deafness or Tinnitus
|
Deafness or Tinnitus
|
Cerebellar infarction in the distribution of PICA can cause either a vertiginous syndrome (medial PICA affecting the cerebellar vermis) or gait incoordination with abnormal rapid alternating movements and absent or only mild dizziness (lateral PICA affecting the lateral cerebellar hemisphere). (See Figure 3.) When the entire PICA territory is affected, patients frequently have associated headache localized to the ipsilateral occiput or neck. They also may maintain head position with a slight tilt toward the affected side. Approximately one-fifth of the cerebellar infarcts of the PICA territory also involve infarction in the dorsal or dorsolateral medulla. The combination of lateral medullary syndrome and PICA cerebellar infarction results when occlusion of the intracranial vertebral artery occurs and blocks the origin of the PICA and the lateral medullary penetrators. Associated symptoms may include vomiting, gait ataxia, truncal instability, and limb hypotonia.11
Figure 3. Cerebellar Infarct in a 54-Year-Old Male Patient Who Presented With Dizziness |
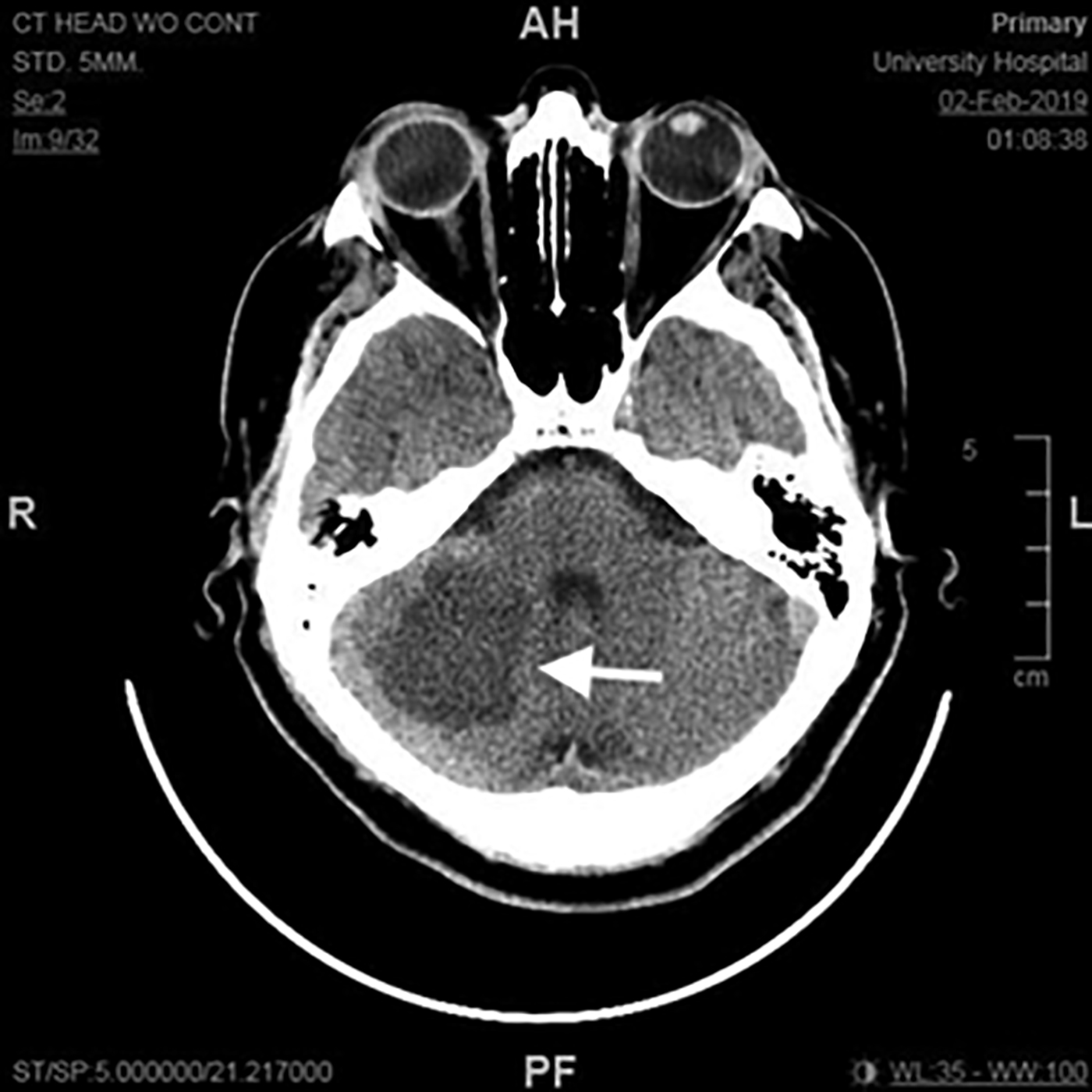 |
|
CT head image shows a large hypodensity in the right cerebellar region. This patient had severe stenosis of the right superior cerebellar artery. |
Amnesia
Transient global amnesia is a clinical syndrome of reversible anterograde amnesia accompanied by repetitive questioning that occurs in middle-age and older individuals. The etiology is unclear, but there appears to be an association with migraine headaches. The prognosis is good, and patients usually have complete recovery and infrequent recurrence.26
Vascular disease is not usually considered in the evaluation of an acutely amnestic patient; however, there have been several case reports describing amnesia as a major initial finding.
The critical structure for the formation and retrieval of new episodic memory is the dominant hemisphere mediobasal temporal lobe and hippocampus, which are both part of the PCA territory.27 Multiple case reports have been presented in the literature in which amnesia is the sole or primary manifestation of stroke.28-30 Patients who present with amnesia warrant a thorough visual field examination, as visual field cuts frequently occur with PCA territory ischemia (see the section on visual complaints). Associated neurological findings, including visual field deficits, alexia, and sensory loss, should warrant further workup of PCA territory stroke.
Altered Mental Status
Pontine stroke from basilar artery occlusion commonly presents as global altered mental status. The basilar artery begins at the medullopontine junction and ends at the junction of the pons and midbrain. Occlusive lesions may occur anywhere along the basilar artery. Motor symptoms include unilateral or bilateral limb weakness with associated hyperreflexia and abnormal spontaneous movements such as shivering, twitching, shaking, or jerking. Incoordination of limb movement is another common motor finding and usually is more pronounced in the legs. In patients who can participate in heel-to-shin testing, the results tend to show clumsiness.
Oculomotor symptoms are a hallmark of pontine stroke because of involvement of the paramedian tegmentum where the medial longitudinal fasciculus is located. Abnormalities may include complete bilateral horizontal gaze palsy, unilateral horizontal conjugate gaze palsy, and unilateral or bilateral internuclear ophthalmoplegia. Patients will have skew deviation, nystagmus, and, frequently, ptosis. Pupils are frequently small (“pinpoint”) but have preserved pupillary response to light, albeit with small amplitude. Lastly, because of the presence of the nuclei of cranial nerves V, VI, and VII in the pons, patients with basilar artery strokes also will show signs of bulbar involvement. The symptoms include facial weakness, dysphonia, dysarthria, dysphagia, and limited jaw movement. The face, pharynx, larynx, and tongue are involved most often. Some patients may completely lose the ability to speak, open their mouth, protrude their tongue, swallow, or move their face. Secretions can pool in the pharynx, making aspiration a potentially serious complication. The condition known as “locked-in syndrome” occurs when patients retain consciousness but lose all other voluntary movements except those of the eyes.11,31
Alertness and behavior abnormalities are common in patients who have rostral brainstem infarcts. An infarct at the distal tip of the basilar artery can cause ischemia and stroke to the rostral brainstem, bilateral thalami, and temporal and occipital lobes supplied by the PCA branches of the basilar artery. This is known as “top of the basilar” syndrome.32 Hypersomnolence and abulia are common, and patients may answer questions with responses that have no relationship to reality. If not completely obtunded, patients may not understand their location in place and time. They may report that they are in a distant geographic location or that they are doing activities that they did in childhood. Patients may develop visual hallucinations, particularly after sundown, that involve vivid colors, objects, and scenes due to involvement of the midbrain peduncles. The midbrain also contains the nuclei of cranial nerves III and IV, so, therefore, it is no surprise that a stroke involving the midbrain affects oculomotor and pupillary function. Patients will have abnormalities of eye position, vertical gaze, and convergence. Upward and downward gaze usually is paralyzed. Regarding convergence, usually one or both eyes are hyperconverged, with an inward and downward location at rest. Because of involvement of the Edinger-Westphal nucleus of cranial nerve III, which innervates the iris and ciliary muscles, the pupils often are fixed and dilated. The superior cerebellar arteries branch from the distal basilar artery, so the superior portion of the cerebellum also may be infarcted. This may manifest as slight dizziness (but much less significant than with PICA infarcts), vomiting, limb dysmetria, gait ataxia, and dysarthria. Notably, sensory and motor function usually are preserved in “top of the basilar” syndrome.11
Cerebral Venous Thrombosis
CVT is a type of stroke that is rare and variable in presentation, and it is often missed, particularly in the ED.
Epidemiology and Risk Factors
CVT has an annual incidence of only 0.22 to 1.57 per 100,000.33,34 It has a variable presentation depending on the location, size, and adverse effects on adjacent brain tissue. Risk factors include the prothrombotic state associated with oral contraceptive use, pregnancy and the puerperium, and malignancy; localized or systemic infection; inflammatory diseases such as systemic lupus erythematosus, Behçet disease, Wegener’s granulomatosis, and sarcoidosis; head injury; and genetic thrombophilic diseases such as antithrombin deficiency, protein C or S deficiency, factor V Leiden mutation, G20210 A prothrombin gene mutation, and hyperhomocysteinemia.34 No underlying etiology or risk factor for CVT is found in less than 10% of children, approximately 13% of adults younger than 65 years of age, and 37% of adults older than 65 years of age.35-37 This seems to imply that increased age also is a risk factor for CVT.
Signs and Symptoms
The symptoms of CVT can be grouped into three major syndromes: isolated intracranial hypertension syndrome, focal syndrome, and encephalopathy. With each of these syndromes, patients may present with one or more signs, symptoms, or neurologic deficits. Isolated intracranial hypertension syndrome is the most common presentation of CVT and is characterized as a headache with or without vomiting, papilledema, or visual problems. Symptoms tend to worsen when the patient lies supine. The focal syndrome can consist of a focal or bilateral deficit, such as leg or hand weakness, focal or generalized seizures, or a combination of these, and often depends on the location of the CVT. (See Table 4.) Approximately 37% of patients present with motor weakness.38 The encephalopathy syndrome seen with CVT manifests as multifocal signs, mental status changes, stupor, or coma. Brain tissue infarction occurs when venous drainage in a certain vascular bed is slowed because of a thrombus, which in turn increases pressure. When this venous pressure surpasses the systolic pressure on the arterial side, there is no forward flow of oxygenated blood, and the tissue becomes ischemic.
Table 4. Location of CVT and Associated Deficits34 |
|
|
Location of CVT |
Focal Deficits |
|
Cavernous sinus |
Ocular signs such as orbital pain, chemosis, proptosis, and oculomotor palsies |
|
Cortical vein thrombosis |
Motor/sensory deficits and seizures |
|
Sagittal sinus |
Motor deficits, bilateral deficits, and seizures |
|
Lateral sinus |
Isolated headache or isolated intracranial hypertension syndrome |
|
Jugular vein |
Isolated pulsatile tinnitus, ± cranial nerve palsies |
|
Posterior fossa veins |
Posterior headache, ± cranial nerve palsies |
Workup and Management
In the setting of a patient with at least one risk factor and signs or symptoms suggestive of CVT, further workup is warranted. Additionally, CVT should be suspected in patients who have atypical neuroimaging features, such as cerebral infarction that crosses typical arterial boundaries, hemorrhagic infarction, or lobar intracerebral hemorrhage of otherwise unclear origin, on routine CT or MRI at presentation.39 In up to 30% of cases, the head CT is normal; however, CT often is the first study performed in the ED and is useful for ruling out other acute or subacute disorders. The second-line imaging study of choice is CT venogram, but MR venography or intra-arterial angiography also can be used if available. Patients with confirmed CVT also warrant screening for thrombophilia, such as testing for protein C and S deficiencies and the factor V Leiden mutation.34
Conclusion
Although rare, subtle strokes are encountered periodically by EPs, frequently without the “code stroke” tipoff by the prehospital providers. The keys to recognizing subtle strokes include taking a good history, performing a thorough neurological examination, and understanding the vascular territories of the brain and their functions.
REFERENCES
- Wall HK, Beagan BM, O’Neill J, et al. Addressing stroke signs and symptoms through public education: The Stroke Heroes Act FAST campaign. Prev Chronic Dis 2008;5:A49.
- Powers WJ, Rabinstein AA, Ackerson T, et al. 2018 guidelines for the early management of patients with acute ischemic stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2018;49:e46-e110.
- Benjamin EJ, Blaha MJ, Chiuve SE, et al. Heart disease and stroke statistics—2017 update: A report from the American Heart Association. Circulation 2017;135:e229-e445.
- Kochanek KD, Murphy SL, Xu J, Arias E. Mortality in the United States, 2016. NCHS Data Brief 2017;293:1-8.
- Murphy SL, Xu J, Kochanek KD, et al. Deaths: Final data for 2015. Natl Vital Stat Rep 2017;66:1-75.
- Feigin VL, Nguyen G, Cercy K, et al. Global, regional, and country-specific lifetime risks of stroke, 1990 and 2016. N Engl J Med 2018;379:2429.
- Schievink WI, Mokri B, O’Fallon WM. Recurrent spontaneous cervical-artery dissection. N Engl J Med 1994;330:393-397.
- Wolf PA. Epidemiology and Prevention. In: Mohr J, Wolf P, Moskowitz M, et al, eds. Stroke: Pathophysiology, Diagnosis, and Management. 5th ed. Philadelphia: Elsevier Saunders; 2011:189-279.
- Smithuis R. Brain ischemia – vascular territories. Radiology Assistant. Available at: http://www.radiologyassistant.nl/en/p484b8328cb6b2/brain-ischemia-vascular-territories.html. Accessed Feb. 1, 2019.
- Liberato B, Krakauer JW. Ischemic stroke: Mechanisms, evaluation, and treatment. In: Schapira AHV, Byrne E, DiMauro S, et al, eds. Neurology and Clinical Neuroscience, 1st ed. Philadelphia: Mosby Elsevier; 2007;551-586.
- Savitz SI, Caplan LR. Vertebrobasilar disease. N Engl J Med 2005;352;2618-2626.
- Georgiadis AL, Yamamoto Y, Kwan ES, et al. Anatomy of sensory findings in patients with posterior cerebral artery territory infarction. Arch Neurol 1999;56:835-838.
- Yamamoto Y, Georgiadis AL, Chang HM, Caplan LR. Posterior cerebral artery territory infarcts in the New England Medical Center Posterior Circulation Registry. Arch Neurol 1999;56:824-832.
- Lavallee PC, Cabrejo L, Labreuche J, et al. Spectrum of transient visual symptoms in a transient ischemic attack cohort. Stroke 2013;44:3312-3317.
- Biousse V, Trobe JD. Transient monocular visual loss. Am J Ophthalmol 2005;140:717-721.
- Ramirez-Lassepas ML, Espinosa CE, Cicero JJ, et al. Predictors of intracranial pathologic findings in patients who seek emergency care because of headache. Arch Neurol 1997;54:1506-1509.
- Goldstein JN, Camargo CA Jr, Pelletier AJ, Edlow JA. Headache in United States emergency departments: Demographics, work-up and frequency of pathological diagnoses. Cephalalgia 2006;26:684-690.
- Schievink WI. Spontaneous dissection of the carotid and vertebral arteries. N Engl J Med 2001;344:898-906.
- Debette S, Markus HS. The genetics of cervical artery dissection: A systematic review. Stroke 2009;40;e459-e466.
- Fridriksson J, den Ouden DB, Hillis AE, et al. Anatomy of aphasia revisited. Brain 2018;141:848-862.
- Kattah JC, Talkad AV, Wang DZ, et al. HINTS to diagnose stroke in the acute vestibular syndrome: Three-step bedside oculomotor examination more sensitive than early MRI diffusion-weighted imaging. Stroke 2009;40:3504-3510.
- Navi BB, Kamel H, Shah MP, et al. Rates and predictors of serious neurological causes of dizziness in the emergency department. Mayo Clin Proc 2012;87:1080-1088.
- Chen L, Lee W, Chambers BR, Dewey HM. Diagnostic accuracy of acute vestibular syndrome at the bedside in a stroke unit. J Neurol 2011;258:855-861.
- Newman-Toker DE, Kerber KA, Hsieh YH, et al. HINTS outperforms ABCD2 to screen for stroke in acute continuous vertigo and dizziness. Acad Emerg Med 2013;20:986-996.
- Searls DE, Pazdera L, Korbel E, et al. Symptoms and signs of posterior circulation ischemia in the New England Medical Center Posterior Circulation Registry. Arch Neurol 2012;69:346-351.
- Quinette P, Guillery-Girard B, Dayan J, et al. What does transient global amnesia really mean? Review of the literature and thorough study of 142 cases. Brain 2006;129:1640-1658.
- Westmacott R, Silver FL, McAndrews MP. Understanding medial temporal activation in memory tasks: Evidence from fMRI of encoding and recognition in a case of transient global amnesia. Hippocampus 2008;18:317-325.
- Ott BR, Saver JL. Unilateral amnesic stroke. Six new cases and a review of the literature. Stroke 1993;24:1033-1042.
- Benson DF, Marsden CD, Meadows JC. The amnesic syndrome of posterior cerebral artery occlusion. Acta Neurol Scand 1974;50:133-145.
- Naldi F, Baiardi S, Guarino M, et al. Posterior hippocampal stroke presenting with transient global amnesia. Neurocase 2017;23:22-25.
- Ferbert A, Brückmann H, Drummen R. Clinical features of proven basilar artery occlusion. Stroke 1990;21:1135-1142.
- Caplan LR. “Top of the basilar” syndrome. Neurology 1980;30:72-79.
- Devasagayam S, Wyatt B, Leyden J, Kleinig T. Cerebral venous sinus thrombosis incidence is higher than previously thought: A retrospective population-based study. Stroke 2016;47:2180-2182.
- Coutinho JM, Zuurbier SM, Aramideh M, Stam J. The incidence of cerebral venous thrombosis: A cross-sectional study. Stroke 2012;43:3375-3377.
- Ferro JM, Canhao P, Bousser MG, et al. Cerebral vein and dural sinus thrombosis in elderly patients. Stroke 2005;36:1927-1932.
- deVeber G, Andrew M, Adams C, et al. Cerebral sinovenous thrombosis in children. N Eng J Med 2001;345:417-423.
- Ichord RN, Benedict SL, Chan AK, et al. Paediatric cerebral sinovenous thrombosis: Findings of the International Paediatric Stroke Study. Arch Dis Child 2015;100:174-179.
- Ferro JM, Canhao P, Stam J, et al. Prognosis of cerebral vein and dural sinus thrombosis: Results of the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT). Stroke 2004;35:664-670.
- Saposnik G, Barinagarrementeria F, Brown RD Jr, et al. Diagnosis and management of cerebral venous thrombosis: A statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2011;42:1158-1192.
This article will explore the subtle and enigmatic presentations of stroke. These patients often will present with nonspecific symptoms, such as vision problems, headache, a subtle language deficit, dizziness, or amnesia.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
