Acute Treatment of Pediatric Migraine
April 1, 2019
Reprints
AUTHORS
Cecilia C. Guthrie, MD, FAAP, FACEP, Associate Professor, Department of Pediatrics, Section of Pediatric Emergency Medicine, The Children’s Hospital at OU Medical Center, Oklahoma City
Yashas Nathani, MD, Fellow, Pediatric Emergency Medicine, The Children’s Hospital at OU Medical Center, Oklahoma City
PEER REVIEWER
Richard Massone, MD, Assistant Director, Our Lady of Lourdes Medical Center ER, Camden, NJ
EXECUTIVE SUMMARY
- Children who experience migraine attacks often have poorer academic performance than their peers, difficulty paying attention while in class, and increased risk of other chronic conditions, such as depression and anxiety.
- Documenting headache characteristics, such as frequency, duration, location, severity, quality, and pattern of headache, as well as triggers, associated symptoms, and family and social history, is crucial in the diagnosis and management. Routine brain computed tomography imaging or magnetic resonance imaging is not indicated for a child who presents with recurrent headaches and has a normal physical and neurological examination.
- Red flags that require neuroimaging include headaches in children younger than 3 years of age, underlying medical conditions (sickle cell disease, immunodeficiency, malignancy, pregnancy, coagulopathy, congenital heart disease, recent head trauma, and seizure disorder), and neurocutaneous syndromes (ash-leaf spots, café-au-lait spots, petechial rash, port wine stain).
- Red flag headache characteristics include early-morning pattern or early-morning waking with headache, nausea, vomiting, worsening headaches with straining, progressive frequency and severity of the headache, changes in mood or mental status, or deteriorating school performance.
- The five phases of migraine are prodrome, aura, headache, resolution, and postdrome. Not all patients experience all phases during each attack.
- The criteria for pediatric migraines include an episodic headache with five or more attacks lasting two to 72 hours that cannot be explained better by medication overuse or another diagnosis. The patient has a non-focal normal neurologic exam. Typical migraine characteristics (such as pulsating quality, pain intensity, and impact on physical activity) and associated symptoms (such as vomiting, nausea, photophobia, and phonophobia) contribute to the diagnosis.
- Researchers have found that intravenous fluids do not improve pain scores and are not associated with sustained headache relief up to 48 hours post-discharge from the ED.
- The reported efficacy of prochlorperazine ranges from 75% to 86%. In one study, metoclopramide was shown to be less effective than prochlorperazine.
- Nonsteroidal anti-inflammatory drugs have been shown to be successful for outpatient therapy of pediatric migraines. Ibuprofen is the most thoroughly investigated agent. Triptans have demonstrated efficacy in treating migraines in children and have been approved for use in children and adolescents. Opioids are not recommended.
A migraine is a type of primary headache that affects children and adults worldwide and commonly these children present to the emergency department (ED). Migraines occur in young children, but may be challenging to diagnose. Migraines can have a great impact on pediatric patients’ quality of life with school absence and limitations on extracurricular activities. Providers can diagnose migraine following criteria in the International Classification of Headache Disorders and must be skilled at recognizing the subtle signs to make an accurate diagnosis.
Treatment of pediatric migraines or possible migraines is difficult secondary to the lack of evidence for effective therapies in pediatric patients. Treatment includes traditional therapy with dopamine receptor antagonists (DRAs), nonsteroidal anti-inflammatory drugs (NSAIDs), triptans, and combination therapies. Newer therapies include magnesium, valproic acid (VPA), and peripheral nerve blocks. The authors review the clinical presentation of migraines in children, guidelines for diagnosis, and therapeutic options.
— Ann M. Dietrich, MD, FAAP, FACEP, Editor
Primary headaches, especially migraines, are a common problem for adults and children who present to the ED. Migraine headaches have been challenging to diagnose, especially in the pediatric population. In addition, there is no consensus on treatment of pediatric migraines in the ED, with the approach to treatment largely extrapolated from adult literature.
Epidemiology
The prevalence of major headaches ranges from 37% to 51% in school-age children and increases to 57% to 82% in adolescence.1 The prevalence increases with age, with nearly 80% of adolescents reporting a significant headache by age 15 years. A common primary headache type is migraine. About 16% of the world’s population suffers from migraine headaches.2 The prevalence of migraine headaches in the pediatric population ranges from 7% to 23%.3 Migraines occur in young children, and the prevalence of migraine increases with age. In a 1997 study by Lee and Olness, children as young as 5 years of age met criteria for migraines per the International Headache Society (IHS).4 In a large-scale meta-analysis of more than 25,000 cases, Lewis et al described the incidence of migraines to be 2% at 3 to 7 years of age, 7% at 7 to 11 years of age, and 20% at 11 to 15 years of age.5 The authors of a Finnish study conducted between 1974 and 2002 demonstrated an increase in prevalence of migraine from 14.5/1,000 to 91.9/1,000 for migraines with and without aura in children 7 years of age.6 Similarly, over a two-year period, the authors of a Taiwanese study showed an increase from 5.2% to 7.4% in migraines in children 13 to 15 years of age.7
A chief complaint of headache is reported in approximately 0.6% to 1.3% of all visits to the pediatric ED.8 Sheridan et al reported that approximately 40% of patients presenting to an ED with a complaint of headache were diagnosed with a primary headache disorder, and approximately three-fourths of these patients were diagnosed with a migraine.9 It also is common for children to present to the ED, typically after the migraine has been ongoing for a couple of days, with one study reporting an average duration of 2.2 days of headache at the time of the ED visit.10
A gender predilection exists in which boys are affected by migraines more frequently before puberty, and females are predominant after puberty, with a ratio of adolescent females to males of 3:1.11 However, the higher prevalence numbers for adolescents may be influenced by the higher likelihood of adolescents presenting to the ED for migraines compared to younger children. Lewis et al noted that 1% of the teenage population and approximately 3% of female teenagers may experience chronic migraines.12
Impact on Life (Morbidity)
Migraine is a leading cause of disability across all age groups. A 2016 article by the Migraine Research Foundation estimated that migraines had an annual economic impact of approximately $36 billion in the United States, resulting from direct medical costs and lost productivity.13 Total annual costs (direct and indirect) were approximated $9,000 higher for migraine patients than costs for demographically similar individuals without migraines.14 The World Health Organization lists migraines as one of the top 20 causes of disability worldwide because of the high costs to individuals and the society.15 For adults, in addition to missed work days and the burden it causes to employers, the impact of migraines can extend to challenges at home when caring for their family members.16
In the pediatric population, disability includes missed school days and inability or decreased ability to participate in extracurricular and social activities. The 1989 National Health Interview Survey reported that out of 975,000 children, there were 164,454 school days missed.17 Compared to their peers, children who experience migraine attacks often have poorer academic performance, difficulty paying attention while in class, and increased risk of other chronic conditions, such as depression and anxiety.17
The Pediatric Quality of Life Inventory v 4.0 (PedsQL 4.0) is a validated measure to assess quality of life in relation to chronic disease in children between 2 and 18 years of age.18 It is a subjective self-evaluation of the child’s functioning and emotional well-being. This questionnaire incorporates responses from children and parents and assesses four domains: emotional, social, school health, and physical health. Using the PedsQL 4.0, Powers et al compared pediatric patients with migraines to healthy children and found impaired quality of life in all areas of functioning in patients with migraine, with adolescents reporting more impairment in school functioning and young children reporting more impairment in social functioning.19 They also found that children with migraines reported disability patterns similar to children with rheumatoid arthritis and cancer.19 Comorbid conditions, such as anxiety and depressive disorders, are more common in children with migraines, and this trend continues in adults.15 In one study of adults hospitalized with a diagnosis of migraine, researchers found that patients with migraines were two times more likely to exhibit suicidal behavior compared to individuals without migraines.20
Assessing the effect of migraine on patients is important, as the ultimate goal of therapy is to improve the patient’s life. The Pediatric Migraine Disability Assessment (PedMIDAS), which is a modification of the Migraine Disability Assessment, was developed as a subjective assessment of the effect of migraine headaches on children and adolescents over a three-month period. Hershey et al found that PedMIDAS was useful for assessing treatment strategies, with improved scores with the use of prophylactic medication.21 In the ED, assessing the effect of migraines on a patient’s life is important to determine the possible need for prophylaxis, which can be a suggestion for further discussion with the primary care physician or specialist, and lifestyle recommendations that can decrease headache triggers.
Pathophysiology
The pathophysiology of migraines is multifactorial, but the exact mechanism of migraine headaches still is largely undefined. Several theories have been proposed over the years and continue to be developed; however, the genetic nature of migraines is clear. Many familial and twin studies have shown that migraine headaches have a high degree of inheritance, with 80% to 90% of first- and second-degree relatives reporting a history of migraines.22 Although no gene or genetic defect has been identified clearly and consistently in migraines with or without aura, three genes have been implicated in familial hemiplegic migraines: P/Q calcium channel (CACNA1A), ATPase (ATP1A2), and sodium channel (SCN1A).22
Genetically, the brain of a migraineur may be more excitable and more vigilant compared to the brain of a person without migraines.23 The inherited alteration of brain excitability may cause the brain to process sensory information differently, even between migraine attacks.2
The trigeminal neurovascular system has been found to contribute to the activation and progression of migraines. The trigeminal sensory afferents that innervate meninges and larger blood vessels in the central nervous system become activated, which in turn activates other pain centers and centers associated with meningeal inflammation.9 Several imaging studies using functional magnetic resonance imaging (fMRI) have shown activation of the trigeminal ganglion in patients with migraines.2 Pain pathways appear to be sensitized with repeated episodes of trigeminal activation and, thus, may contribute to chronic migraines.24
There also is increasing evidence that cortical spreading depression — a transient wave of depolarization of the cortex and the underlying mechanism of aura — is involved in the activation of trigeminal nociception and trigger headache mechanisms.25 It remains unclear how this theory applies to patients who report migraines without aura, which includes the majority of patients with migraines.
Migraine also is thought to be associated with cranial vasodilation. The primary dysfunction may originate from brainstem centers that regulate pain sensation and vascular tone. Neuropeptides, such as 5-hydroxytryptamine (5-HT), and calcitonin gene-related peptide (CGRP) may be responsible for the vascular phenomena seen in migraine.9 5-HT, also known as serotonin, is an inhibitory neuropeptide that may prevent the vasodilatory cascade, while CGRP is a potent vasodilator within the central nervous system and may be a key component in triggering migraine attacks. This may explain the efficacy of triptans and CGRP receptor antagonists in aborting migraine headaches.
Another factor that plays a role in migraine headaches is hormones. Fluctuations in estrogen levels have been implicated in the onset of migraine attacks, with estrogen withdrawal prior to menses being a known trigger.26 Some women taking oral contraceptive medications may experience an increase in headache severity and frequency during placebo weeks with decreased estrogen levels. Similarly, women in the postpartum period may have more headaches due to low estrogen levels.
Diagnosis
Most commonly, the diagnosis of headaches is based on the IHS International Classification of Headache Disorders.27,28 This tool classifies headaches into primary, secondary, and those caused by other cranial neuropathies and headache syndromes. Over time, this tool has been refined, thus helping improve the sensitivity and specificity for diagnosing childhood headaches, especially migraines. (See Table 1.)
Table 1. Pediatric Migraine Criteria28 |
Pediatric MigraineAt least five headaches meeting the following criteria:
Pediatric Migraine With AuraAt least two headaches meeting the following criteria:
|
When evaluating a patient with headache, the provider should differentiate between a primary and secondary headache and determine if the headache is a sign of a life-threatening etiology. Secondary headaches are the most common type of headache (49%) and the most common etiology is infectious.8 Although painful and significantly affecting the patient’s life, primary headache disorders are not life-threatening. Headaches without abnormal neurological or physical examination findings rarely are due to underlying disease processes, such as increased intracranial pressure, central nervous system infection, or malignancy.3
To facilitate an accurate diagnosis, a thorough history and physical examination is required. Documenting headache characteristics, such as frequency, duration, location, severity, quality, and pattern of headache, as well as triggers, associated symptoms, and family and social history, is crucial in the diagnosis and management of the headache. Conducting a comprehensive review of systems, medication use (prescribed, over-the-counter, and illegal/controlled substances), recent events, and a thorough physical examination including a detailed neurological examination further will aid in distinguishing primary from secondary headaches.
It is important to note that routine brain computed tomography (CT) imaging or MRI is not indicated for a child who presents with recurrent headaches and has a normal physical and neurological examination. Neurologic findings are present in 85% of patients with brain tumors within eight weeks and nearly 100% within 24 weeks of illness onset.29 However, neuroimaging should be considered in patients with the red flags described in Table 2.1
Table 2. Red Flags for Considering Neuroimaging for Headaches |
Patient Characteristics
Headache Characteristics
Signs/Symptoms
|
Traditionally, occipital pain has been considered a red flag for pediatric headaches. However, research indicates that occipital headaches are not associated with intracranial pathology more than headaches in other locations.30 Ancillary testing, such as lumbar puncture, laboratory studies, and/or electroencephalogram (EEG), is not indicated routinely for patients with headache in the setting of normal physical and neurological examination.
Primary and Secondary Headaches
Primary headaches include migraine and its variants, tension-type headaches, and other trigeminal autonomic cephalalgias. Tension-type headaches present with pressing or tightening quality of a bilateral headache, with increased pericranial tenderness being the most common abnormal finding. These headaches may or may not be associated with nausea, phonophobia, or photophobia. Trigeminal autonomic cephalalgias usually are unilateral, are accompanied by cranial parasympathetic autonomic features (such as increased lacrimation, nasal congestion, and conjunctival injection), and generally are ipsilateral to the headache. Other forms of primary headaches include cough, exercise, thunderclap, cold stimulus, external pressure, stabbing, nummular, hypnic, sexual activity-associated, and new daily persistent headaches.28 Secondary headaches range from new headaches to significant worsening of pre-existing headaches that are a result of underlying pathology of a systemic disease or trauma. Some etiologies include vascular disorders, malignancy, infection, psychiatric disorders, increased intracranial pressure, trauma to head and/or neck, or substance abuse or withdrawal, among others.
Criteria for Migraine and Its Variants
Migraines can occur with or without an aura. In children, migraine without aura is the most common type and is reported with approximately two-thirds of migraines.1,31 The five phases of migraine are prodrome, aura, headache, resolution, and postdrome.23 Not all patients experience all phases during each attack. The prodromal phase occurs in up to 80% of migraineurs and begins 24 hours prior to the headache.23 Symptoms include yawning, fatigue, changes in appetite, increased energy, frequent urination, difficulty with memory and concentration, irritability, and hyper-/hypo-sexuality. Typical postdrome symptoms include cognitive difficulties, dizziness, fatigue, and concern for recurrence of headache, which may last 24 to 48 hours. Patients describe this phase as feeling like a “hangover” or having been “run over by a truck.”23
The criteria for pediatric migraines include an episodic headache with five or more attacks lasting two to 72 hours that cannot be explained better by medication overuse or another diagnosis. The patient has a non-focal normal neurologic exam. Typical migraine headache characteristics (such as pulsating quality, pain intensity, and impact on physical activity) and associated symptoms (such as vomiting, nausea, photophobia, and phonophobia) contribute to the diagnosis.
Differences between adult and pediatric migraine characteristics have been identified. Migraines in children may have a bilateral or non-throbbing nature and may be shorter in duration compared to those in adults. In addition, children may not be able to communicate symptoms, such as quality or photophobia, so inference from their behavior may be used, such as going to a dark room to lie down. Headache duration in adults ranges from four to 72 hours compared to two to 72 hours in children.27 During the transition from late adolescence to early adulthood, the location of the headache becomes unilateral.28
Migraine with aura has additional neurologic symptoms, which are described as typical, brainstem, hemiplegic, or retinal.28 A typical aura includes visual (zigzag lines with relative scotomas), sensory (unilateral paresthesia often ipsilateral to the headache and involving the hand, face, leg, and/or trunk), and speech/language (aphasia) symptoms. Speech/language aura is the least common among the typical auras. Brainstem aura includes symptoms such as dysarthria, vertigo, tinnitus, hypacusis, diplopia, ataxia (not attributable to sensory deficit), and decreased level of consciousness (Glasgow Coma Scale < 13). Hemiplegic aura involves fully reversible motor weakness lasting less than 72 hours. Retinal migraines include repeated attacks of monocular visual disturbance, including scintillations, scotomata, or blindness, associated with migraine headache. These symptoms are reversible and may occur before, during, or after the headache, and may last up to 60 minutes following the headache. Migraine with aura may be difficult to distinguish from a more serious neurologic event secondary to the accompanying transitory neurologic findings. Migraine with aura may be underdiagnosed in the younger population because of the difficulty obtaining effective and reliable communication about specific aura symptoms. (See Table 1.)
In addition to the classic forms of migraine with or without aura, patients may present to the ED with a migraine variant. An important aspect of a migraine is the recurrent episodic nature of the headache and the ability to distinguish it from a few symptomatic migraine-like attacks. The IHS designates a minimum number of attacks at five.28 In the ED, patients may present with headaches that meet migraine criteria except for the number of previous episodes. These patients may be identified as having probable migraine without aura.28 See Table 3 for migraine variant characteristics and classification.
Table 3. Migraine Variants3 |
||
Migraine Variant |
Description |
Symptoms/Characteristics |
|
Migraine with brainstem aura (formerly known as basilar migraine) |
|
|
|
Chronic migraine |
|
|
|
Familial hemiplegic migraine |
|
|
|
Status migrainosus |
|
|
|
Cyclic vomiting syndrome |
|
|
|
Abdominal migraine |
|
|
Acute Migraine Treatment
For pediatric headaches and migraines, outpatient, nonprescription abortive therapies of ibuprofen or acetaminophen have been shown to be effective. Ibuprofen has been found to be the superior agent.32,33,34 (See Table 4.) Patients may present to the ED for migraine treatment when the headache reaches a level of intolerability and/or the symptoms persist despite home therapy.10,35 In two multicenter studies, children who presented to the ED with a probable migraine headache or migraine by IHS criteria had symptoms for an average of two days.10,35 About 60% of patients with migraines receive oral analgesics prior to ED presentation.10,36
Table 4. Abortive Medications in the ED |
|||
Drug |
Dosing |
Side Effects |
Contraindications |
|
Prochlorperazine Metoclopramide |
0.15 mg/kg (IV), max 10 mg 0.1 mg/kg (IV), |
Extrapyramidal side effects, sedation, prolong QT |
Bone marrow depression |
|
Sumatriptan |
5 to 20 mg (IN) |
History of stroke, cardiovascular disease, uncontrolled hypertension, hemiplegia, migraine |
|
|
Ibuprofen Ketorolac |
10 mg/kg, 0.5 mg/kg (IV), |
Gastrointestinal upset |
|
|
Diphenhydramine |
1 mg/kg, |
Sedation |
Platelet disorders |
|
Magnesium |
25 to 50 mg/kg, |
Nausea, vomiting, hypotension, flushing |
|
|
Valproic acid |
15 mg/kg, |
Nausea, dizziness, tachycardia, paresthesia |
Pregnancy, hepatic disease, mitochondrial disorders, metabolic disorders |
|
IV: intravenously, IN: intranasally |
|||
Systematic reviews of medication trials for pediatric migraine treatments demonstrate the lack of controlled trials of acute treatments for pediatric migraine overall but especially in the ED.34,37 A recent systematic review of acute treatment for pediatric migraines reported seven specific treatment studies performed in the ED.38 Although multiple treatment modalities exist for migraine abortive therapy, the majority of the literature is based on the adult population.36,39,40
Retrospective chart reviews, cohort studies, and database reviews reveal that various treatments are used in treating pediatric migraines in the ED. A multicenter study of children’s hospitals in Canada noted significant variability of treatment between ED sites.10 Eapen et al noted differences in treatment and admission rates between a pediatric and community ED.35 Sheridan et al described significant variability in the evaluation and treatment of pediatric headache in the ED.9 Guidelines in the literature have suggested different first- and second-line treatments.41-43 A retrospective chart review by the Pediatric Emergency Research Canada reported the use of different abortive treatments for pediatric migraines, including dopamine antagonists (specifically, metoclopramide, prochlorperazine, and chlorpromazine) with and without diphenhydramine, NSAIDs, dihydroergotamines, steroids, benzodiazepines, and opioids.10 Bachur et al also noted variability of treatment in a retrospective database study in 2015.44
Intravenous Fluid Hydration
Children with migraine may have symptoms of nausea and vomiting and, therefore, may have decreased oral intake with increased fluid losses. Treatment with intravenous (IV) fluids is variable and has been reported from 10% to 50%.10 Published guidelines for treating pediatric migraines include IV fluid therapy.42,43 Authors of a Canadian multicenter, retrospective review found that the use of IV fluid hydration in the treatment of pediatric migraine is common.45 The authors of a randomized, controlled trial (RCT) found no decrease in pain with IV fluids when comparing the expectation of medicine and no medicine with the fluid after 30 minutes of therapy.46 The use of IV fluids did not improve pain scores and was not associated with sustained headache relief up to 48 hours post-discharge from the ED.47 Since ED patients may receive IV medication therapy, providing fluid therapy may help rehydrate with little risk. In addition, fluid hydration may help provide renal protection with the use of IV nonsteroidal anti-inflammatory treatment, such as ketorolac.48
Dopamine Receptor Antagonist
The role of DRAs for migraine has evolved over time. Initially, DRAs were used for treating the nausea and vomiting associated with migraines. With the development of the trigeminal cascade pathophysiology hypothesis for migraine, DRAs have a role in treatment overall. The DRAs used most commonly are prochlorperazine and metoclopramide.44 Prochlorperazine is the most studied DRA treatment for migraine in children. Metoclopramide has been well studied in the adult population for treatment of migraine. A recent retrospective study comparing prochlorperazine, metoclopramide, and promethazine effectiveness in the ED was the first that specifically defined effectiveness for metoclopramide in pediatric migraine treatment.49 Other less commonly used DRAs are chlorperazine and haldol.
The reported efficacy of prochlorperazine ranges from 75% to 86%.50-54 Sheridan et al found that metoclopramide was less effective than prochlorperazine. Treatment failure occurred with metoclopramide, and the medication resulted in less than 50% pain reduction, odds ratios of 3.5 (95% confidence interval [CI], 0.6-20.22) and 3.2 (95% CI, 0.88-11.4), respectively, when compared to prochlorperazine.49 Adult studies also have shown metoclopramide to be inferior to or no better in effectiveness than prochlorperazine.55,56 Promethazine and chlorperazine are less effective in the treatment of pediatric migraines compared to prochlorperazine and metoclopramide (57% and 60%, respectively).49,51 In addition, a large database study showed treatment with metoclopramide increased the odds for an ED revisit within three days compared to prochlorperazine.44
Side effects from DRAs include dystonia and akathisia. Diphenhydramine has been used as an adjunct medication to prevent extrapyramidal side effects. Vinson and Drotts reported a decrease in akathisia in adults when diphenhydramine was added to prochlorperazine for migraine treatment.57 In 2016, Friedman et al reported no difference in efficacy or side effects in an RCT of adults using metoclopramide with or without diphenhydramine.58
Authors of retrospective studies involving acute migraine treatment with prochlorperazine in pediatric patients reported that the akathisia rate was 0% to 12%.51,54 Brousseau et al reported akathisia in 3% of patients in an RCT involving prochlorperazine.50 In other pediatric retrospective chart reviews with diphenhydramine given in addition to prochlorperazine, the akathisia rate was 0% to 6.5%.43,52 Trottier et al specifically evaluated the rate of akathisia in a pediatric prospective cohort study that involved acute treatment with prochlorperazine and diphenhydramine in the ED. The Vinson scale for akathisia was used for evaluation of akathisia in the ED with telephone follow-up at 48 hours and seven days. Trottier et al observed a definite occurrence in 5%, with possible occurrence in an additional 34%.53 In a study using the Pediatric Health Information System database, Bachur et al reported that 33% of patients treated for pediatric migraine received diphenhydramine. Treatment with diphenhydramine and DRA increased the odds of a return visit to the ED by 27% when compared to DRA treatment alone in pediatric patients.44
NSAIDs
NSAIDs have been shown to be successful for outpatient therapy of pediatric migraines. Ibuprofen is the most thoroughly investigated agent.33,59 Ibuprofen was more effective for pain control than acetaminophen (odds ratio [OR], 2.2;
95% CI, 1.1-4.0) and placebo (OR, 2.9; 95% CI, 1-8.1) in pediatric migraineurs. Ketorolac is an IV NSAID commonly used as a treatment for migraine alone or in combination with other drugs in the ED. Studies have supported the efficacy of ketorolac.60 Brousseau et al demonstrated an efficacy of 55% in pediatric migraines with ketorolac treatment alone.50 In a 2014 RCT, 48% of patients treated with ketorolac did not need a second rescue medication in the ED.61 Naproxen is another NSAID that is effective and commonly is used.62,63
Combination Therapy
When used as part of a hospital migraine treatment guideline, the combination therapy of a DRA and an NSAID with IV fluid administration was found to be more effective than monotherapy. In a cohort study before and after guideline implementation with combination therapy, Leung et al found a significant decrease in the admission rate to 3% for migraine and a decrease in headache pain scores when combination therapy was given once, with admission if headache persisted.42 Kaar et al also reviewed the hospital admission rate and pain score changes before and after implementation of a combination therapy guideline, which also incorporated a repeat dose of DRA as a second-line therapy and an antiepileptic drug as a third-line therapy prior to admission for treatment failure. They found that 12% of treated patients required a second dose of DRA and 39% of patients treated with the second dose were discharged home.43 In an RCT comparing prochlorperazine and ketorolac and then, if needed, crossing over the medications, the combination of both medications was most successful, with 93% effectiveness in discharging the patient home.50 Another combination that has been investigated in the adult population is naproxen and sumatriptan. This combination has been demonstrated to be more efficacious than either agent alone.64 Naproxen also has been studied in adolescents and was shown to have a favorable safety profile.65
Triptans
Triptans act on serotonin receptors (5-HT1B/1D), which have peripheral and central effects on neurovascular vasodilation.23,66 Triptans have demonstrated efficacy in treating migraines in children and have been approved by the U.S. Food and Drug Administration for use in children and adolescents. Routes of delivery include oral agents, nasal sprays, and subcutaneous injection.40 Intranasal sumatriptan is approved for adolescents in Europe.36 The American Academy of Neurology recommends intranasal sumatriptan for acute migraine treatment because there are three studies with class I rating and it has a level A recommendation.67 Triptans have not been studied in the ED for pediatric migraine treatment.
Studies show that the most effective time to give triptans for treatment of migraines is early in the course, while the headache is mild.68 Upon presentation to the ED, the duration of the pediatric migraine is prolonged, with an average reported duration of approximately 48 hours, which may account for the uncommon use of triptans in ED treatment.10,35,44
Magnesium
Magnesium influences neurogenic inflammation that has been associated with migraine pathophysiology.69 A meta-analysis of randomized, double-blind studies showed no beneficial effect at 30 minutes after magnesium treatment in adult migraneurs.69 This result may have been influenced by the limitations of the studies, including inadequate power, the methodology of randomization, possible type II errors, and time constraint of 30 minutes. In 2015, Shahrami et al found increasing efficacy in pain control with magnesium treatment of migraine at 20 minutes (35%), one hour (71%), and two hours (92%) post-treatment.70 A pediatric case series of IV magnesium for acute treatment of headaches found minimal side effects and a response rate that was highest in patients with status migrainosus. Magnesium was given after multiple other medications had failed.71
Opioids
The American Academy of Neurology does not recommend opioids for primary headache treatment.67 Opioid treatment may convert a migraine from an episodic to a chronic headache and potentiate pain with blunting of treatment response.72 A retrospective database study and multi-site chart review of pediatric EDs found that opioid use in the acute treatment of migraines ranges from 3% to 5%.10,44 A retrospective cohort study using commercial claims data from the HealthCore Integrated Research Environment reported that 46% of patients with a diagnosis of headache, excluding trauma, received an opioid prescription at the index date.73 In a retrospective cohort study of state Medicaid claims and the Prescription Drug Monitoring Program, opioid prescribing rates after discharge from an ED or ambulatory care facility were identified. The ED prescribing rate was approximately 4%, which was higher than the ambulatory setting rate of 1%. The authors found an increased probability of an opioid prescription given by non-pediatricians compared to pediatricians after discharge.74
Dihydroergotamine
Dihydroergotamine (DHE) is an ergot alkaloid that acts as a 5-HT1 receptor antagonist and has been found to be effective in treating refractory migraines. Inpatient therapy typically requires multiple doses for resolution of the headache. Contraindications to DHE therapy include stroke, uncontrolled hypertension, pregnancy, and cardiovascular disease. In a pediatric case series in which DHE was given for migraine without aura and status migrainosus, inpatient therapy had 74% to 80% effectiveness.75,76 Patients initially were given dopamine receptor antagonists and then switched to ondansetron prior to the DHE doses to help relieve these symptoms. Initially, the patient may feel worse with side effects from the DHE, such as nausea, vomiting, transient increase in headache, and chest tightness.75,76 In a retrospective chart review, Nelson et al reported 63% of hospitalized patients responded to IV DHE. In addition, the headache resolved in 21% of all hospitalized patients and in 50% of patients diagnosed with status migrainosus.77 In a small case series in which DHE therapy was given for refractory abdominal migraines, five of six patients had symptom resolution or significant improvement.78 In a small outpatient study (n = 12) using a crossover design to compare oral DHE and placebo, researchers did not find a statistically significant improvement in pain.79,80
Peripheral Nerve Block
Inflammation of scalp nerves may contribute to headaches by conducting pain signals. Local anesthetic helps reduce pain by interrupting the nerve signal through inhibition of sodium channels.81 There is no consensus on the specific local anesthetic, injection technique, or dose.82 The local anesthetic may be used alone or in conjunction with corticosteroids.80 Injection sites can be identified at points of maximal tenderness. Peripheral nerve blocks have been used in adults to treat a variety of headaches, including migraines.82 In 2018, the authors of a systematic review and meta-analysis of seven RCTs found that greater occipital nerve (GON) blocks significantly decreased pain intensity and there was no increase in adverse events in adult migraineurs.83
GON blocks have been shown to have the greatest efficacy in children with chronic migraine and new daily persistent headache.84,85 GON blocks also have improved pain in children with post-traumatic headache.84,86 In a retrospective case series, Dubrovksy et al found good therapeutic effect in 93% of patients given a GON block and, occasionally, the lesser occipital nerve and supraorbital nerve, depending on the areas of scalp point tenderness.84
In a prospective RCT, adult migraine patients received GON block, placebo GON block, or IV dexketoprofen and metoclopramide. The authors reported that GON block was as effective as the IV medication and superior to the placebo GON block when the changes in median score were compared.87 The median value change from baseline at 30 and 45 minutes after treatment showed a statistically significant difference when comparing the three arms. Limitations to the study include no blinding of the IV medication, the inability to generalize secondary to a single study site, a possible placebo effect with injections, and a small number of participants in the study (n = 60). In a small (n = 28) randomized, sham-controlled trial of GON blocks in adults, researchers found some effectiveness (30% of participants achieved complete resolution of the headache) at 30 minutes post-treatment. Study participants were eligible if metoclopramide failure had occurred.The participants and outcome assessors were blinded. The study was stopped prior to achieving the projected sample size of 78 because of slow enrollment over 31 months.88
Valproic Acid
VPA is an antiepileptic medication used mainly as prophylaxis for migraine patients. It enhances gamma-aminobutyric acid neurotransmission and blocks the voltage-gated sodium channels and T-type calcium channels.89 Although it has been used as prophylaxis for pediatric migraines, valproic acid was given as an acute migraine treatment in two small retrospective studies.90,91 In a chart review, Reiter et al gave one dose of IV VPA at 1 g, and patients had a subsequent decrease in pain and were discharged home in 78% of visits. A second dose of 500 mg was given in 22% of visits.90 In a case series of 13 patients, IV VPA was given as a second-line therapy, and 83% of the patients had pain improvement and were discharged home.91 Although limitations included missing documentation and small numbers, the authors concluded that VPA may be helpful in the acute treatment of pediatric migraines. In a recent retrospective study on the use of VPA in a continuous infusion after an initial bolus for inpatient therapy for pediatric status migrainosus, researchers reported VPA was well tolerated, and 64% of patients achieved 100% headache resolution by 48 hours from the start of infusion.92
Corticosteroids
The benefits of corticosteroid use in the treatment of acute migraine are uncertain. In a meta-analysis of RCTs in which patients received dexamethasone as adjunct treatment to standard abortive therapy, no benefit in pain reduction was seen; however, a reduction in headache recurrence was noted.93 Conflicting results in other studies show no improvement in recurrence rates.93,94 In a pediatric retrospective chart review, Cobb-Pitstick et al observed no significant decrease in migraine recurrence after acute treatment in the ED or inpatient therapy.95
Propofol
Propofol is an established medication used in procedural sedation in the ED. Studies using low-dose propofol in the acute treatment of migraine have been reported in adult and pediatric patients. In an adult case report96 and pediatric case series,97 low-dose propofol showed efficacy in pain control. In an RCT comparing the efficacy of propofol and subcutaneous sumatriptan, researchers found no difference in pain control at one and two hours after treatment.98 Better antiemetic control was reported with the low-dose propofol. Sheridan et al reported no significant difference in pain reduction in pediatric patients in an RCT comparing low-dose propofol (59%) and standard therapy (NSAID, DRA, and diphenhydramine; 51%).99 Rebound headache was significantly more common in the standard therapy group (67%) compared to the low-dose propofol group (25%). Study limitations included no standardized low-dose propofol and the unblinded and underpowered nature of the study. Questions still remain regarding the effectiveness of propofol in the treatment of pediatric migraine, and further studies are needed.
Treatment Approach
See Figure 1 for an approach to treating children with possible migraine.
Figure 1. Approach to Treating Pediatric Migraine Patients |
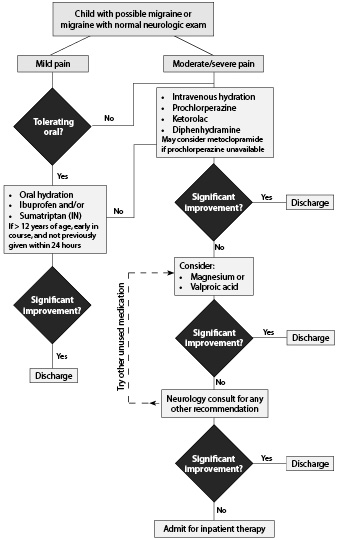 |
- Determine the severity of the patient’s migraine and the patient’s oral tolerance to liquids.
If the patient has a mild headache and is tolerating oral liquids, hydration with oral fluids and ibuprofen may resolve the headache. If nausea and vomiting are present, it may be more beneficial to treat with a DRA and IV hydration. Some published guidelines use ondansetron as the antiemetic.1 A side effect of ondansetron is a headache, so it may be counterproductive. If the headache is mild and early in the course, sumatriptan may be given intranasally.
If the patient has a moderate or severe headache, begin IV hydration and start therapy with a combination of IV ketorolac and DRA, with or without adjunct diphenhydramine. Use prochlorperazine intravenously, as it is most efficacious. If prochlorperazine is unavailable, use metoclopramide.
- Reevaluate the status of the migraine 60 minutes after medications are given.
If the patient is not significantly better, consider magnesium sulfate or VPA as the next treatment. Check for pregnancy before giving VPA to female patients because it is contraindicated in pregnancy. Continue hydrating the patient, especially if giving magnesium sulfate.
- Reevaluate the status of the migraine 60 minutes after the medication is given.
If the patient is not significantly better, administer the alternate option from the previous choice of magnesium sulfate or VPA. In addition, consider contacting a neurology specialist to aid in further medication decision-making.
- Reevaluate the status of the migraine 60 minutes after the medication is given.
A neurology specialist may recommend other medications or admission for inpatient therapy.
- If the pain has resolved or decreased to a mild, tolerable level for the patient on reevaluation, discharge the patient home.
Discharge home with a prescription for naproxen, as medication overuse headache is less likely to occur than with ibuprofen or over-the-counter naproxen. Discuss limiting the number of days the NSAID is used to less than three times per week. Another option is to give a sumatriptan/naproxen combination (10/60 mg or 85/500 mg), which may be taken daily as needed for migraine. Have the patient keep a headache diary to help identify triggers of the migraine and to discuss it with the primary care provider or neurologist. Common triggers for migraine attacks include changes in sleep-wake cycle, stress, dehydration, menstruation (due to fluctuating hormone levels), certain foods, and alcohol.3 Migraine prophylaxis is recommended when headaches occur more than one day per week and significantly affect the patient’s quality of life.40
Return ED Visits
After improvement of a migraine headache in the ED, some patients will experience a recurrence of migraine symptoms and will return to the ED. Headache recurrence and the return rate in different studies have different times of follow-up, from two to three days50 to seven days53 to one month.100 The return rate in pediatric patients has been reported to range from 3% to 5%51 to 11% to 12%.50,53,100 Prescriptions for naproxen and corticosteroids have been given prior to discharge to decrease headache recurrence, with studies showing conflicting success. Analgesic overuse after discharge may contribute to recurrence. In a retrospective review using the hospital’s ED database, Legault et al reported that acute treatment and discharge medicine did not influence the return rate.100 Further study of this problem is needed.
Conclusion
Pediatric migraine is a common presentation to the ED and has a significant effect on quality of life. Acute treatment in the ED is variable, with a large amount of treatment based on the adult literature. Although the headache may resolve with treatment, the headache may recur within days to weeks, which contributes to patients returning to the ED. Further investigations into acute ED treatment and headache recurrence is warranted.
REFERENCES
- Alfonzo M, Chen L. Acute migraine management in children. Pediatr Emerg Care 2015;31:722-727.
- Noseda R, Burstein R. Migraine pathophysiology: Anatomy of the trigeminovascular pathway and associated neurological symptoms, CSD, sensitization and modulation of pain. Pain 2013;154(Suppl 1):1-21.
- Koch T, Oakley CB. Pediatric migraine: Diagnostic criteria and treatment. Contemp Pediatr 2018;35:22-30.
- Lee LH, Olness KN. Clinical and demographic characteristics of migraine in urban children. Headache 1997;37:269-276.
- Lewis DW, Ashwal S, Dahl G, et al. Practice parameter: Evaluation of children and adolescents with recurrent headaches. Report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology 2002;59:490-498.
- Anttila P, Metsähonkala L, Sillanpää M. Long-term trends in the incidence of headache in Finnish schoolchildren. Pediatrics 2006;117:e1197-e1201.
- Wang SJ, Fuh JL, Lu SR, Juang KD. Chronic daily headache in adolescents. Neurology 2006;66:193-197.
- Conicella E, Raucci U, Vanacore N, et al. The child with headache in a pediatric emergency department. Headache 2008;48:1005-1011.
- Sheridan DC, Spiro DM, Meckler GD. Pediatric migraine: Abortive management in the emergency department. Headache 2014;54:235-245.
- Richer LP, Laycock K, Millar K, et al. Treatment of children with migraine in emergency departments: National Practice Variation Study. Pediatrics 2010;126:e150-e155.
- Pakalnis A. Headaches and hormones. Semin Pediatr Neurol 2010;17:100-104.
- Lewis D, Ashwal S, Hershey A, et al. Practice parameter: Pharmacological treatment of migraine headache in children and adolescents: Report of the American Academy of Neurology Quality Standards Subcommittee and the Practice Committee of the Child Neurology Society. Neurology 2004;63:2215-2224.
- Migraine Research Foundation. Migraine fact sheet. Available at: https://migraineresearchfoundation.org/about-migraine/migraine-facts/. Accessed Dec. 11, 2018.
- Bonafede M, Sapra S, Shah N, et al. Direct and indirect healthcare resource utilization and costs among migraine patients in the United States. Headache 2018;58:700-714.
- Kroon Van Diest AM, Ernst MM, Slater S, Powers SW. Similarities and differences between migraine in children and adults: Presentation, disability and response to treatment. Curr Pain Headache Rep 2018;21:48.
- Stang PE, Osterhaus JT. Impact of migraine in the United States: Data from the National Health Interview Survey. Headache 1993;33:29-35.
- Gorodzinsky AY, Hainsworth KR, Weisman SJ. School functioning and chronic pain: A review of methods and measures. J Pediatr Psychol 2011;36:991-1002.
- Varni JW, Seid M, Kurtin PS. PedsQL 4.0: Reliability and validity of the pediatric quality of life inventory version 4.0 generic core scales in healthy and patient populations. Med Care 2001;39:800-812.
- Powers SW, Patton SR, Hommel KA, Hershey AD. Quality of life in childhood migraines: Clinical impact and comparison to other chronic illnesses. Pediatrics 2003;112:e1-5.
- Friedman LE, Zhong QY, Gelaye B, et al. Association between migraine and suicidal behaviors: A nationwide study in the USA. Headache 2018;58:371-380.
- Hershey AD. Pediatric headache. Continuum (Minneap Minn) 2015;21:1132-1145.
- Kabbouche MA, Gilman DK. Management of migraine in adolescents. Neuropsychiatr Dis Treat 2008;4:535-548.
- Jay GW, Barkin RL. Primary headache disorders part I- Migraine and the trigeminal autonomic cephalalgias. Dis Mon 2017;63:308-338.
- Mathew NT. Pathophysiology of chronic migraine and mode of action of preventive medications. Headache 2011;51(Suppl 2):84-92.
- Pietrobon D, Moskowitz MA. Pathophysiology of migraine. Annu Rev Physiol 2013;75:365-391.
- Pakalnis A. Migraine and hormones. Semin Pediatr Neurol 2016;23:92-94.
- Headache Classification Committee of the International Headache Society (IHS). Torelli P, Jensen RH, Tavanaiepour D, et al. The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 2013;33:629-808.
- Headache Classification Committee of the International Headache Society (IHS). Vincent M, Wang S. The International Classification of Headache Disorders, 3rd edition. Cephalalgia 2018;38:1-211.
- Honig PJ , Charney EB. Children with brain tumor headaches: Distinguishing features. Am J Dis Child 1982;136:121-124.
- Irwin SL, Gelfand AA. Occipital headaches and neuroimaging in children. Curr Pain Headache Rep 2018;22:59.
- Ferracini GN, Dach F, Speciali JG. Quality of life and health-related disability in children with migraine. Headache J Head Face Pain 2013;54:325-334.
- Lewis DW. Pediatric migraine headaches. Pediatr Rev 2007;28:43-52.
- Hämäläinen ML, Hoppu K, Valkeila E, Santavuori P. Ibuprofen or acetaminophen for the acute treatment of migraine in children: A double-blind, randomized, placebo-controlled, crossover study. Neurology 1997;48:103-107.
- Damen L, Bruijin JK, Verhagen AP, et al. Symptomatic treatment of migraine in children: A systematic review of medication trials. Pediatrics 2005;116:e295-e302.
- Eapen A, Sivaswamy L, Agarwal R, Thomas R. Management of pediatric migraine in a tertiary care versus community based emergency department: An observational pilot study. Pediatr Neurol 2014;50:164-170.
- Gelfand AA, Goadsby PJ. Treatment of pediatric migraine in the emergency room. Pediatr Neurol 2012;47:233-241.
- Bailey B, McManus BC. Treatment of children with migraine in the emergency department: A qualitative systematic review. Pediatr Emerg Care 2008;24:321-330.
- Patniyot IR, Gelfand AA. Acute treatment therapies for pediatric migraine: A qualitative systematic review. Headache 2016;56:49-70.
- Kacperski J, Kabbouche MA, O’Brien HL, Weberding JL. The optimal management of headaches in children and adolescents. Ther Adv Neurol Disord 2016;9:53-68.
- Patterson-Gentile C, Szperka CL. The changing landscape of pediatric migraine therapy: A review. JAMA Neurol 2018;75:881-887.
- Trottier ED, Bailey B, Dauphin-Pierre S, et al. Practice variation after implementation of a protocol for migraines in children. Eur J Emerg Med 2010;17:290-292.
- Leung S, Bulloch B, Young C, et al. Effectiveness of standardized combination therapy for migraine treatment in the pediatric emergency department. Headache 2013;53:491-197.
- Kaar CRJ, Gerard JM, Nakanishi AK. The use of a pediatric migraine practice guideline in an emergency department setting. Pediatr Emerg Care 2016;32:435-439.
- Bachur RG, Monuteaux MC, Neuman MI. A comparison of acute treatment regimens for migraine in the emergency department. Pediatrics 2015;135:232-238.
- Richer L, Graham L, Klassen T, Rowe B. Emergency department management of acute migraine in children in Canada: A practice variation study. Headache 2007;47:703-710.
- Richer L, Craig W, Rowe B. Randomized controlled trial of treatment expectation and intravenous fluid in pediatric migraine. Headache 2014;54:1496-1505.
- Balbin JEB, Nerenberg R, Baratloo A, Friedman BW. Intravenous fluids for migraine: A post hoc analysis of clinical trial data. Am J Emerg Med 2016;34:713-716.
- Arora S, Wagner JG, Herbert M. Myth: Parenteral ketorolac provides more effective analgesia than oral ibuprofen. Can J Emerg Med 2007;9:30-32.
- Sheridan DC, Laurie A, Pacheco S, et al. Relative effectiveness of dopamine antagonists for pediatric migraine in the emergency department. Pediatr Emerg Care 2018;34:165-168.
- Brousseau DC, Duffy SJ, Anderson AC, Linakis JG. Treatment of pediatric migraine headaches: A randomized, double-blind trial of prochlorperazine versus ketorolac. Ann Emerg Med 2004;43:256-262.
- Kanis JM, Timm NL. Chlorpromazine for the treatment of migraine in a pediatric emergency department. Headache 2014;54:335-342.
- Trottier ED, Bailey B, Dauphin-Pierre S, Gravel J. Clinical outcomes of children treated with intravenous prochlorperazine for migraine in a pediatric emergency department. J Emerg Med 2010;39:166-173.
- Trottier ED, Bailey B, Lucas N, Lortie A. Prochlorperazine in children with migraine: A look at its effectiveness and rate of akathisia. Am J Emerg Med 2012;30:456-463.
- Kabbouche M A, Vockell A-LB, Lecates SL, et al. Tolerability and effectiveness of prochloperazine for intractable migraine in children. Pediatrics 2001;107:e62-64.
- Friedman BW, Esses D, Solorzano C, et al. A randomized controlled trial of prochlorperazine versus metoclopramide for treatment of acute migraine. Ann Emerg Med 2008;52:399-406.
- Coppola M, Yealy DM, Leibold RA. Randomized, placebo-controlled evaluation of prochlorperazine versus metoclopramide for emergency department treatment of migraine headache. Ann Emerg Med 1995;26:541-546.
- Vinson DR, Drotts DL. Diphenhydramine for the prevention of akathisia induced by prochlorperazine: A randomized, controlled trial. Ann Emerg Med 2001;37:125-131.
- Friedman BW, Cabral L, Adewunmi V, et al. Diphenhydramine as adjuvant therapy for acute migraine: An emergency department-based randomized clinical trial. Ann Emerg Med 2016;67:32-39.
- Lewis DW, Kellstein D, Dahl G, et al. Children’s ibuprofen suspension for the acute treatment of pediatric migraine. Headache 2002;42:780-786.
- Taggart E, Doran S, Kokotillo A, et al. Ketorolac in the treatment of acute migraine: A systematic review. Headache 2013;53:277-287.
- Friedman BW, Garber L, Yoon A, et al. Randomized trial of IV valproate vs metoclopramide vs ketorolac for acute migraine. Neurology 2014;82:976-983.
- Andersson PG, Hinge HH, Johansen O, et al. Double-blind study of naproxen vs placebo in the treatment of acute migraine attacks. Cephalalgia 1989;9:29-32.
- Nestvold K, Kloster R, Partinen M, Sulkava R. Treatment of acute migraine attack: Ibuprofen and placebo compared. Cephalalgia 1985;5:115-119.
- Brandes JL, Kudrow D, Stark SR, et al. Sumatriptan-naproxen for acute treatment of migraine. JAMA 2007;297:1443-1454.
- McDonald SA, Hershey AD, Pearlman E, et al. Long-term evaluation of sumatriptan and naproxen sodium for the acute treatment of migraine in adolescents. Headache 2011;51:1374-1387.
- Link AS, Kuris A, Edvinsson L. Treatment of migraine attacks based on the interaction with the trigemino-cerebrovascular system. J Headache Pain 2008;9:5-12.
- Lewis D, Ashwal S, Hershey A, et al. Practice parameter: Pharmacological treatment of migraine headache in children and adolescents: Report of the American Academy of Neurology Quality Standards Subcommittee and the Practice Committee of the Child Neurology Society. Neurology 2004;63:2215-2224.
- Goadsby PJ, Zanchin G, Geraud G, et al. Early vs. non-early intervention in acute migraine — “Act when Mild (AwM).” A double-blind, placebo-controlled trial of almotriptan. Cephalalgia 2008;28:383-391.
- Choi H, Parmar N. The use of intravenous magnesium sulphate for acute migraine: Meta-analysis of randomized controlled trials. Eur J Emerg Med 2014;21:2-9.
- Shahrami A, Assarzadegan F, Hatamabadi HR, et al. Comparison of therapeutic effects of magnesium sulfate vs. dexamethasone/metoclopramide on alleviating acute migraine headache. J Emerg Med 2015;48:69-76.
- Gertsch E, Loharuka S, Wolter-Warmerdam K, et al. Intravenous magnesium as acute treatment for headaches: A pediatric case series. J Emerg Med 2014;46:308-312.
- Tepper SJ. Opioids should not be used in migraine. Headache 2012;52(Suppl 1):30-34.
- Devries A, Koch T, Wall E, et al. Opioid use among adolescent patients treated for headache. J Adolesc Heal 2014;55:128-133.
- Meckler GD, Sheridan DC, Charlesworth CJ, et al. Opioid prescribing practices for pediatric headache. J Pediatr 2019;204:240-244.e2.
- Linder SL. Treatment of childhood headache with dihydroergotamine mesylate. Headache 1994;34:578-580.
- Kabbouche MA, Powers SW, Segers A, et al. Inpatient treatment of status migraine with dihydroergotamine in children and adolescents. Headache 2009;49:106-109.
- Nelson GR, Bale JF, Kerr LM. Outcome and cost of inpatient hospitalization for intravenous dihydroergotamine treatment of refractory pediatric headache. Pediatr Neurol 2017;66:76-81.
- Raina M, Chelimsky G, Chelimsky T. Intravenous dihydroergotamine therapy for pediatric abdominal migraines. Clin Pediatr 2013;52:918-921.
- Hämäläinen ML, Hoppu K, Santavuori PR. Oral dihydroergotamine for therapy-resistant migraine attacks in children. Pediatr Neurol 1997;16:114-117.
- Tobin JA, Flitman SS. Occipital nerve blocks: Effect of symptomatic medication: Overuse and headache type on failure rate. Headache 2009;49:1479-1485.
- Lambrinakos-Raymond K, Dubrovsky AS. Nerve blocks in paediatric and adolescent headache disorders. Curr Opin Pediatr 2018;30:780-785.
- Ashkenazi A, Blumenfeld A, Napchan U, et al. Peripheral nerve blocks and trigger point injections in headache management - A systematic review and suggestions for future research. Headache 2010;50:943-952.
- Zhang H, Yang X, Lin Y, Chen L, Ye H. The efficacy of greater occipital nerve block for the treatment of migraine: A systematic review and meta-analysis. Clin Neurol Neurosurg 2018;165:129-133.
- Dubrovsky AS, Friedman D, Kocilowicz H. Pediatric post-traumatic headaches and peripheral nerve blocks of the scalp: A case series and patient satisfaction survey. Headache 2014;54:878-887.
- Gelfand AA, Reider AC, Goadsby PJ. Outcomes of greater occipital nerve injections in pediatric patients with chronic primary headache disorders. Pediatr Neurol 2014;50:135-139.
- Seeger TA, Orr S, Bodell L, et al. Occipital nerve blocks for pediatric posttraumatic headache: A case series. J Child Neurol 2015;30:1142-1146.
- Korucu O, Dagar S, Çorbacioglu K, et al. The effectiveness of greater occipital nerve blockade in treating acute migraine-related headaches in emergency departments. Acta Neurol Scand 2018;138:212-218.
- Friedman BW, Mohamed S, Robbins MS, et al. A randomized, sham-controlled trial of bilateral greater occipital nerve blocks with bupivacaine for acute migraine patients refractory to standard emergency department treatment with metoclopramide. Headache 2018;58:1427-1434.
- Linde M, Mulleners WM, Chronicle EP, McCrory DC. Valproate (valproic acid or sodium valproate or a combination of the two) for the prophylaxis of episodic migraine in adults. Cochrane Database Syst Rev 2013;(6):CD010611.
- Reiter PD, Nickisch J, Merritt G. Efficacy and tolerability of intravenous valproic acid in acute adolescent migraine. Headache 2005;45:899-903.
- Sheridan DC, Sun B, O’Brien P, Hansen M. Intravenous sodium valproate for acute pediatric headache. J Emerg Med 2015;49:541-545.
- Zafar MS, Stewart AM, Toupin DN, et al. Continuous intravenous valproate as abortive therapy for pediatric status migrainosus. Neurologist 2018;23:43-46.
- Rowe BH, Colman I, Edmonds ML, et al. Randomized controlled trial of intravenous dexamethasone to prevent relapse in acute migraine headache. Headache 2008;48:333-340.
- Fiesseler FW, Shih R, Szucs P, et al. Steroids for migraine headaches: A randomized double-blind, two-armed, placebo-controlled trial. J Emerg Med 2011;40:463-468.
- Cobb-Pitstick KM, Hershey AD, O’Brien HL, et al. Factors influencing migraine recurrence after infusion and inpatient migraine treatment in children and adolescents. Headache 2015;55:1397-1403.
- Sato K, Hida A, Arai N, Takeuchi S. Low-dose intravenous propofol as a possible therapeutic option for acute confusional migraine. Am J Emerg Med 2017;35:195.e5-195.e6.
- Sheridan DC, Spiro DM, Nguyen T, et al. Low-dose propofol for the abortive treatment of pediatric migraine in the emergency department. Pediatr Emerg Care 2012;28:1293-1296.
- Moshtaghion H, Heiranizadeh N, Rahimdel A, et al. The efficacy of propofol vs. subcutaneous sumatriptan for treatment of acute migraine headaches in the emergency department: A double-blinded clinical trial. Pain Pract 2015;15:701-705.
- Sheridan DC, Hansen ML, Lin AL, et al. Low-dose propofol for pediatric migraine: A prospective, randomized controlled trial. J Emerg Med 2018;54:600-606.
- Legault G, Eisman H, Shevell MI. Treatment of pediatric status migrainosus: Can we prevent the “bounce back.” J Child Neurol 2011;26:949-955.
Migraines occur in young children, but may be challenging to diagnose. Migraines can have a great impact on pediatric patients’ quality of life with school absence and limitations on extracurricular activities. Treatment of pediatric migraines or possible migraines is difficult secondary to the lack of evidence for effective therapies in pediatric patients. Treatment includes traditional therapy with dopamine receptor antagonists (DRAs), nonsteroidal anti-inflammatory drugs (NSAIDs), triptans, and combination therapies. Newer therapies include magnesium, valproic acid (VPA), and peripheral nerve blocks. The authors review the clinical presentation of migraines in children, guidelines for diagnosis, and therapeutic options.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
