Volume Overload: Acute Decompensated Heart Failure in the Emergency Department
May 1, 2019
Reprints
AUTHORS
Michael Ullo, MD, PGY-3 Resident Physician, Department of Emergency Medicine, Rutgers New Jersey Medical School, Newark
Gregory Sugalski, MD, Associate Professor, Department of Emergency Medicine, Rutgers New Jersey Medical School, Newark
PEER REVIEWER
Catherine A. Marco, MD, FACEP, Professor of Emergency Medicine and Surgery, Wright State University, Dayton, OH
EXECUTIVE SUMMARY
- Heart failure is a complex clinical syndrome of impaired cardiac pump function.
- About half of patients with heart failure have preserved left ventricular ejection fraction (> 50%).
- Patients with acute heart failure exacerbations and normal to elevated blood pressure have a lower 30-day mortality and respond well to emergency department treatment with nitrates and loop diuretics.
- Early initiation of loop diuretics in the emergency department is associated with lower in-hospital mortality in patients with acute heart failure exacerbations.
- The initial treatment of patients with acute heart failure presenting with hypotension depends on whether they have reduced or preserved ejection fraction; review of previous echocardiograms is helpful in this determination.
- For patients with reduced ejection fraction and the presence of shock, the initial therapy should include the initiation of an inotropic agent such as dobutamine or milrinone.
- Patients with preserved ejection fraction and hypotension should not receive inotropic support. Rather, these patients may benefit from the addition of a vasopressor (norepinephrine, high-dose dopamine, or vasopressin) as well as diuretics.
- Most patients with acute heart failure exacerbations require hospital admission for stabilization. Selected patients can be managed in an emergency department observation unit.
Background
Heart failure is a complex clinical syndrome that is caused by structural or functional impairment of ventricular filling or ejection of blood. This impairment leads to insufficient perfusion and can result in unmet metabolic demands.1
Acute decompensated heart failure (ADHF) is a disease process frequently encountered by emergency physicians and is characterized by the buildup of fluid in the lung interstitium and alveolar spaces. This buildup of fluid in the lungs often is referred to as pulmonary edema. The term “congestive heart failure” has become outdated in the medical literature but it is used to describe patients with evidence of volume overload on clinical exam.
Although the most common cause of pulmonary edema is cardiogenic, it is worthwhile to recognize that there are other noncardiogenic causes. Distinguishing between cardiogenic and noncardiogenic pulmonary edema is a critical step for the emergency physician because the clinical approach may differ. A focused clinical exam, along with laboratory, radiographic, and echocardiographic testing, can help distinguish between the two disease states.
This article will focus primarily on the important aspects of ADHF in the emergency setting. The authors will include a brief synopsis of noncardiogenic pulmonary edema to highlight key principles in the diagnosis and management.
Epidemiology
Heart failure remains one of the most common cardiovascular disorders in the United States with an estimated prevalence of 2%.2,3 The incidence increases with age: 3.7% in patients 75 to 79 years of age, 5.2% in those 80 to 84 years of age, and 7.2% in those 85 years of age and older. The elderly represent more than 80% of decompensated heart failure admissions annually.4,5 Experts hypothesize that the prevalence will rise steadily with an aging population, with an estimated 772,000 new cases in the year 2040.6
Terminology
Because heart failure is a complex entity, understanding of the terminology associated with the disease state is of utmost importance. Although no universal terminology is accepted, it is important for emergency physicians to be familiar with some of the taxonomy.
The New York Heart Association (NYHA) classifies heart failure based on functionality as it relates severity of symptoms. The classes are as follows:7
- Class I: Heart disease with no symptoms or limitations on physical activity;
- Class II (mild): Comfortable at rest, but symptoms of heart failure with ordinary activity;
- Class III (moderate): Comfortable at rest, but symptoms of heart failure with minimal exertion;
- Class IV (severe): Symptoms at rest that greatly impede physical activity.
Another approach to the classification of heart failure is based on ejection fraction (EF). The EF is defined as a measurement, expressed as percentage, of how much blood the left ventricle releases with each ventricular contraction as seen on echocardiography. Table 1 includes descriptions of the different characteristics of this particular classification system.
Table 1. Heart Failure Classification Based on Ejection Fraction7-9 |
|
|
Classification |
Characteristics |
|
Heart failure with preserved ejection fraction |
|
|
Heart failure with mid-range ejection fraction |
|
|
Heart failure with reduced ejection fraction |
|
Describing heart failure can be particularly challenging because a number of different methods are used in the classification process. Early in the disease course, the classification system is useful because it helps guide management. As heart failure continues to progress, the distinction becomes less clear because there is significant overlap in symptomatology and etiology in advanced disease states.
Acute vs. Chronic
Classification often is based on the timing of disease onset. Acute heart failure (AHF) is the abrupt onset of heart failure that typically results after a cardiac insult, such as myocardial infarction or valvular dysfunction. Chronic heart failure is seen in patients who have primary cardiac disease that results in decompensation of cardiac function over time. More than 80% of cases presenting to the emergency department involve the acute decompensation (exacerbation) of chronic heart failure.10
Systolic vs. Diastolic
Systole refers to the portion of the cardiac cycle in which blood is pumped out of the ventricles and into the systemic circulation. Systolic dysfunction is used to describe impairment in contractility (the “pumping” of blood). On the other hand, diastole refers to the filling phase in the cardiac cycle. The diastolic phase is marked by ventricular relaxation and filling. Diastolic dysfunction occurs because of impaired ventricular relaxation. This terminology can be problematic, as most cases of heart failure present with components of both diastolic and systolic dysfunction.
Right-Sided vs. Left-Sided
The physiology of heart failure can be viewed as problems with the left (systemic) side and the right (pulmonary) side of the heart. Although artifactual, this classification system assumes that the two sides of the heart work independently of each other. The reality is that the cardiovascular system is interconnected, and problems in one area can have significant downstream physiologic consequences. With left-sided failure, the pulmonary system can become overloaded, resulting in a host of pulmonary symptoms, such as dyspnea and orthopnea. With right-sided failure, backup into the venous return system results in signs and symptoms related to peripheral edema.
High-Output vs. Low-Output
The term high-output heart failure is used to describe hyperdynamic cardiac dysfunction. Excess adrenergic tone and increased preload can damage cardiac myocytes over time. As this hyperdynamic state persists, the ejection fraction eventually “normalizes,” and the cardiac output becomes similar to the low-output state. Low-output cardiac failure is substantially more common and is associated with impaired ejection fractions. The low-output state typically is caused by ischemic heart disease and other pathologies that damage cardiac myocytes over time.
Etiology
The symptomatology of heart failure is largely due to changes in the ventricular structure of the heart. Remodeling of the ventricles results in cardiac dilation, fibrosis, and reactive hypertrophy.11 These structural changes are the consequence of deficits in cardiac output over time. To improve forward perfusion, neurohormonal modulators attempt to augment cardiac output. These neurohormonal systems serve as the therapeutic target of many pharmaceutical agents used in the chronic management of heart failure.
Primary disease processes of the cardiovascular system contribute to the development of heart failure. Although directly determining the causative factor is difficult, understanding the various etiologies that contribute to this multifactorial disorder is important. The three most common etiologies are ischemic heart disease, hypertension, and diabetes.3
In developed countries, the leading cause of heart failure is coronary artery disease, which is present in more than 70% of affected patients. Thrombosis and subsequent ventricular infarction lead to fibrosis and areas of dyskinesis. Chronic coronary insufficiency results in diffuse myocardial fibrosis, setting the foundation for the development of ischemic cardiomyopathy.12 Patients with a history of myocardial infarction have an eight- to 10-fold increased risk of developing heart failure.13 Hypertension remains the single most important modifiable risk factor for heart failure.7 Diabetes is an independent risk factor for heart failure, with a three-fold increase in prevalence compared to those without diabetes.
Other important but less common etiologies of heart failure include cardiomyopathy and valvular disorders. Diseases of the myocardium contribute greatly to the development of heart failure. Three commonly recognized forms of cardiomyopathy include dilated, hypertrophic, and restrictive. Valvular disease, including severe regurgitation and stenosis, can greatly impair both systolic and diastolic functioning of the heart. Acute dysfunction can lead to fulminant pulmonary edema, while more chronic dysfunction results in gradual structural changes to the heart.14
Risk Factors
There are many well-studied risk factors, both inherited and acquired, associated with the development of heart failure. Age remains a prominent risk factor, with increasing prevalence in those older than 65 years of age.13 Men have a higher risk of developing heart failure than women. African Americans have a higher incidence of and five-year mortality rate from heart failure compared to white Americans.15
Acquired risk factors have been implicated in the development of heart failure. Metabolic syndrome, along with diabetes mellitus and atherosclerotic disease, are associated strongly with myocardial dysfunction. Tobacco use and alcohol abuse also have been implicated. A negative correlation exists between lifetime alcohol consumption and cardiac ejection fraction.16,17
Although less common, exposure to cardiotoxic drugs and toxins can lead to the development of heart failure. Drug-induced cardiomyopathy is a known side effect of chemotherapeutic medications such as 5-fluorouracil, paclitaxel, and the anthracycline class.18 Recreational drugs of abuse, such as cocaine, methamphetamines, and anabolic steroids, have been linked to heart failure.19
Pathophysiology
Normal cardiac physiology centers around cardiac output, which is a measure of the amount of blood pumped by the heart in a given period of time. Cardiac output can be thought of as the product of the heart rate (HR) and stroke volume (SV; Cardiac output = HR × SV). The normal range for cardiac output is 4 to 8 liters per minute and varies based on concurrent metabolic demands.20
Left ventricular (LV) dysfunction is the result of damage to myocardial cells located in the ventricle. Damage to the myocardium results in decreased cardiac output and subsequent hypoperfusion as the heart is unable to distribute blood efficiently into the systemic circulation. At the same time, backflow from unejected blood in the LV results in elevated pulmonary pressures. Increased capillary pressures in the lung parenchyma lead to pulmonary congestion and the hallmark sensation of dyspnea.
Right ventricular (RV) dysfunction typically is regarded as a consequence of LV failure. In a similar fashion, decreased output from the right ventricle leads to increased pressures in the vena cava system. These elevated pressures impair venous drainage from the body, leading to the peripheral edema seen on physical examination.
Clinical Presentation
The signs and symptoms associated with underlying heart failure vary based on the degree to which ventricular function is impaired. It is important for emergency physicians to recognize that heart failure is a spectrum of disease and that presentations can vary greatly based on underlying cardiac dysfunction. Despite this, there are several well recognized manifestations of heart failure that should suggest the possibility heart failure.
Dyspnea is a common symptom and frequently is described by patients as shortness of breath. Early in the disease process, dyspnea occurs with exertion. There may not be much interference with daily functioning, and the disease process may go undiagnosed. As more advanced heart failure develops, dyspnea can present at rest with minimal to no physical activity. Impairments in activities of daily living because of dyspnea represent a significant concern, as poor functional status is an adverse prognostic indicator in patients with heart failure.21 With progression of the disease, orthopnea and paroxysmal nocturnal dyspnea may become salient features in the patient’s history.
Hypoperfusion due to impaired cardiac functioning can result in fatigue and generalized weakness. Advanced stages of heart failure are marked by profound hypotension, representing the inability of the heart to maintain adequate perfusion pressures. Eventually, this can manifest as feelings of lightheadedness and ultimately may lead to syncopal episodes. Hypotension in patients with heart failure has been associated with increased mortality.22
Volume overload is a significant contributor to many of the signs and symptoms within the spectrum of heart failure. Peripheral edema in the lower extremities and dependent areas suggests an excess of intravascular volume. Excess fluid within the alveoli of the lungs may present with a cough associated with frothy sputum. Fluid retention in the body can present with ascites, increased abdominal distention, and an increase in total body weight.
Physical Examination
The physical examination of patients with heart failure will vary based on where on the spectrum of disease the patient falls. Patients with no functional limitations from heart failure may appear well on examination with little to no clinical findings of volume overload. On the other end of the spectrum, patients with severely decompensated heart failure may present in extremis hallmarked by severe respiratory distress and cardiogenic shock.
Findings with well-demonstrated specificity for heart failure include prominent jugular venous distention (JVD), the presence of hepatojugular reflux, a laterally displaced apical impulse, and the presence of a gallop on cardiac auscultation. Findings that are less specific (because they can be found with a host of other medical conditions) include tachypnea, pulmonary rales, peripheral edema, and cachexia in late disease.13,23
Patients presenting to the emergency department with an acute heart failure exacerbation typically are hypertensive and potentially toxic-appearing. Sympathetic activation within the nervous system results in diaphoresis and tachycardia. Pulmonary congestion and impaired ventricular functioning can lead to frank respiratory distress with prominent rales on auscultation.
Differential Diagnosis
Since heart failure is a syndrome comprised of many nonspecific signs and symptoms, physicians must maintain a broad differential when evaluating a patient with a suspected heart failure exacerbation. Although dyspnea is a fairly common finding in patients with heart failure, other etiologies, such as myocardial ischemia and lung pathology such as pneumonia or reactive airway disease, should be considered.24
Edema and evidence of fluid overload on examination involve an equally broad differential. Sodium retention, venous thrombosis and/or insufficiency, medication side effects (e.g., calcium channel blockers), and cirrhosis of the liver all can present with peripheral edema or generalized ascites.
Patients with advanced heart failure typically are hypotensive at baseline because of impaired ventricular ejection and systemic perfusion. Etiologies of hypotension should be considered based on history and physical examination. Sepsis, hemorrhage, and cardiac tamponade are examples of diagnostic considerations that may present with hypotension and, therefore, should be considered in the differential diagnosis.
Diagnosis
The clinical spectrum of heart failure makes each diagnostic encounter unique to the patient’s cardiopulmonary status at any given moment in time. Although outpatient management is essential in the chronic management of heart failure, acute presentations to the emergency department require a specialized diagnostic and therapeutic management plan.
Evaluation of patients with suspected acute decompensated heart failure should begin with a focused history and physical examination. Evidence gained from this portion of the clinical encounter will help guide further testing, and specific features should increase the emergency physician’s suspicion for heart failure as the cause for the patient’s symptoms. When heart failure is suspected, electrocardiography (ECG), chest radiography, ultrasound, and laboratory evidence can assist with making a diagnosis.
An ECG should be obtained and interpreted fairly quickly after the diagnostic encounter is initiated. Careful evaluation for acute myocardial infarction or other evidence of emergent cardiac ischemia should be considered. ECG findings that are consistent with an acute heart failure exacerbation include nonspecific ST/T wave abnormalities, large inverted T waves, and QT interval prolongation.25
Upright chest radiography can reveal findings consistent with heart failure and concomitant pulmonary congestion. The clinician should evaluate chest radiographs for evidence of pulmonary congestion as shown by increased perihilar markings. Unilateral findings are exceedingly rare in acute heart failure presentations.26 Blunting of the costophrenic angles should raise suspicion for pleural effusions, which typically are found in exacerbations of chronic heart failure. (See Figure 1.) Although findings on chest radiography can support the diagnosis of heart failure, it is important to recognize that upwards of 20% of radiographs may be normal in patients with ADHF.27
Laboratory data can provide clinicians with valuable information during the evaluation of heart failure. Routine chemistry testing can be used to evaluate for electrolyte derangements that may be present because of the underlying volume overload. Careful evaluation and trending of serum creatinine is important because patients with advanced heart failure are at risk for cardiorenal syndrome, which is the product of chronic hypoperfusion and elevated venous pressures. Cardiac enzyme testing, including troponin measurement, should be evaluated in conjunction with ECG analysis to exclude acute myocardial infarction or cardiac ischemia.
Figure 1. Cardiomegaly With Pulmonary Vascular Congestion Suggestive of Pulmonary Edema |
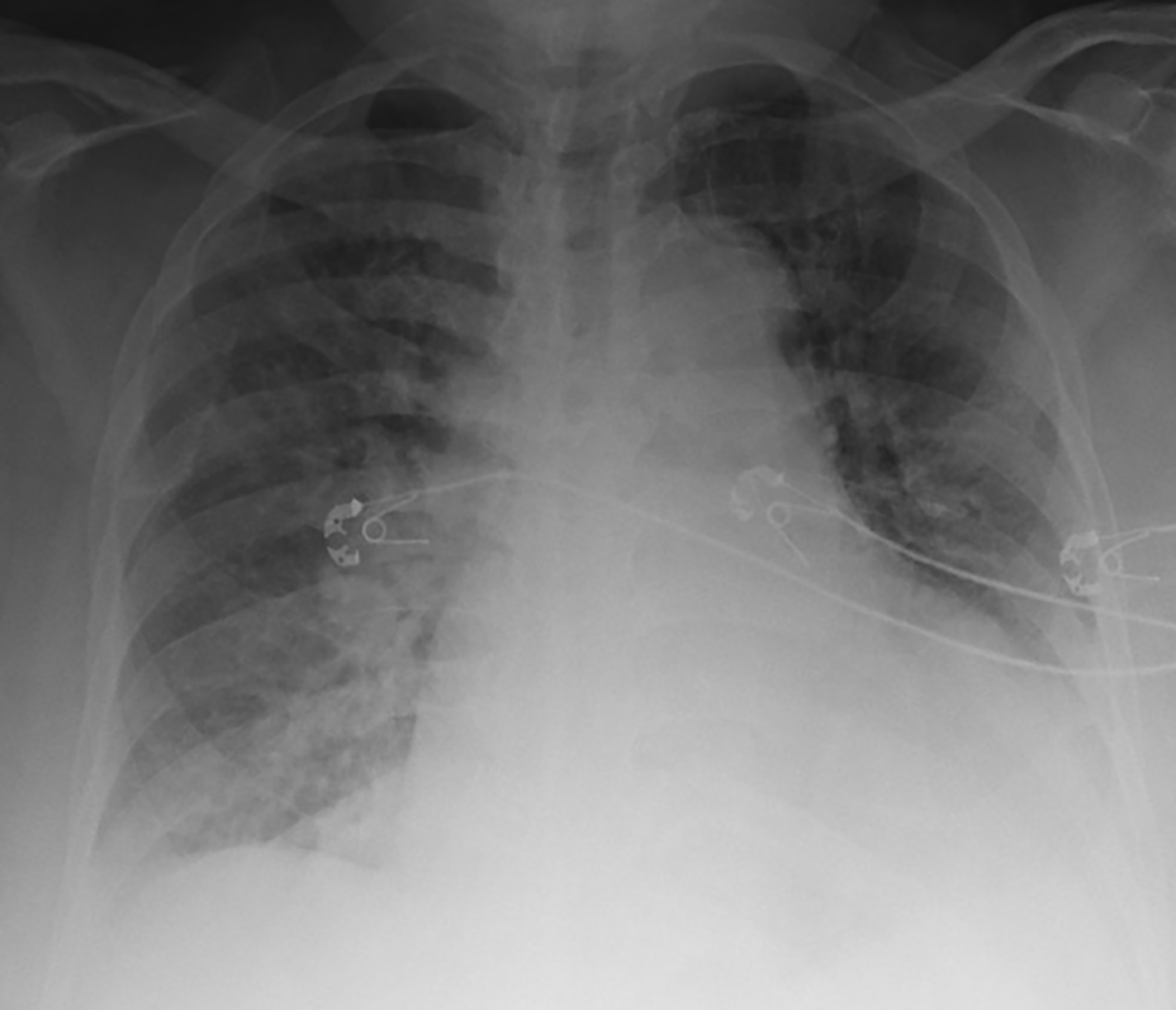 |
|
Portable upright chest X-ray. Mild blunting of left costophrenic angle suggestive of pleural effusion. |
The most recognized laboratory test in the evaluation for heart failure is the serum brain natriuretic peptide (BNP) level. Evaluation of the BNP level is most valuable when the pretest probability for heart failure is intermediate.28 BNP values higher than 400 pg/mL typically are consistent with a diagnosis of heart failure. Intermediate levels, ranging from 200 to 400 pg/mL, should be interpreted with caution because they can neither confirm nor exclude the diagnosis. Finally, low levels of BNP (< 100 pg/mL) are particularly useful because the diagnosis of acute heart failure is extremely unlikely.29 Recent evidence has demonstrated that BNP levels may be falsely low in obese patients. An inverse relationship has been found between body mass index and serum BNP levels.30
Echocardiography plays a pivotal role in the diagnosis of heart failure and the evaluation of cardiac functioning. In fact, leading experts have described it as “the single most useful diagnostic tool in the evaluation of patients with heart failure.”31 Although formal echocardiography by a trained cardiologist can provide valuable information about the structural and functional state of the heart, it rarely is available in the emergent setting. In the emergency department, bedside ultrasound can be a useful tool for quick and reliable information about cardiac activity. Emergency physicians trained in the use of bedside ultrasound can evaluate patients for wall motion abnormalities, estimate ejection fraction, exclude pericardial effusion, and examine for valvular disease.
Adjunctive diagnostic modalities exist to aid in the evaluation of patients with suspected heart failure. Bedside lung ultrasound can be performed quickly during the ultrasonographic evaluation of cardiac functioning. The presence of ultrasonographic B-lines is 94% sensitive and 92% specific when compared to clinical diagnosis.32 The routine use of invasive hemodynamic monitoring is not recommended in the initial evaluation of acute decompensated heart failure.13
Management of Acute Decompensated Heart Failure
Heart failure presentations can vary depending on the stage in which the patient presents to the emergency setting. In the emergency department, roughly 20% of patients present with their first episode of heart failure, while 80% already have presented to the hospital setting for heart failure in the past.10 As with all emergency department encounters, the initial assessment of patients presenting with suspected ADHF should focus on the ABCs of emergency medicine: airway, breathing, and circulation. Patients should be placed on a cardiac monitor and a full set of vital signs should be obtained as intravenous access is attempted.
Acute heart failure typically presents with pulmonary edema. Although pulmonary edema can have cardiogenic and noncardiogenic etiologies, patients with heart failure exacerbations have pulmonary congestion due to cardiac dysfunction and elevated capillary wedge pressures. Noncardiogenic pulmonary edema is a similar but distinct clinical entity that will be discussed briefly at the end of this article. Table 2 provides an overview of the medications often used in the management of acute heart failure in the emergency setting.
Table 2. Medications for Acute Heart Failure |
|||
|
Vasodilating Agents |
|||
|
Medication |
Administration |
Dosing |
Notable Adverse Reactions |
|
Nitroglycerin |
Sublingual |
0.4 mg every 1 to 5 minutes |
Hypotension |
|
Intravenous |
Drip: 5 to 10 mcg/min with increases in increments of 5 to10 mcg/min every 3 to 5 minutes, max 200 mcg/min |
Hypotension, tachyphylaxis |
|
|
Nitroprusside |
Intravenous |
Drip: 5 to 10 mcg/min titrated every five minutes, max 400 mcg/min |
Significant hypotension, cyanide toxicity, rebound vasoconstriction upon discontinuation |
|
Diuretic Agents |
|||
|
Medication |
Administration |
Dosing |
Notable Adverse Reactions |
|
Furosemide |
Intravenous |
No prior use: 20 to 40 mg IV push Prior use: 1 to 2.5 x patient’s home total daily dose divided in half and given as IV push |
Electrolyte disturbances (hypokalemia, hypomagnesemia), hyperuricemia, hypotension, hypovolemia |
|
Bumetanide |
Intravenous |
1 to 3 mg |
|
|
Torsemide |
Intravenous |
10 to 20 mg |
|
|
Adapted from: Tintinalli JE, Stapczynski J, Ma O, et al, eds. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 8e New York: McGraw-Hill; 2016. |
|||
Acute Decompensated Heart Failure With Adequate Perfusion
Management decisions for heart failure exacerbations should be guided based on assessment of perfusion and hemodynamic stability. Patients with stable to elevated blood pressures are demonstrating compensatory mechanisms to maintain systemic perfusion. Patients with such presentations have been shown to have lower mortality than those without substantial myocardial reserves.33
Therapeutic modalities in acute heart failure exacerbations involve reduction of preload and afterload, with careful monitoring to maintain adequate hemodynamic and respiratory function. Hypoxemia should be corrected with supplemental oxygen either via nasal cannula or high-flow face mask. Physicians and nursing staff should be careful not to administer supplemental oxygen routinely because excessive oxygen has been linked to worsened outcomes in decompensated heart failure.34
Patients who do not respond to supplemental oxygen should be considered candidates for noninvasive positive pressure ventilation (e.g., bilevel positive airway pressure [BiPAP] or continuous positive airway pressure [CPAP]) based on mental status and respiratory effort. Use of these measures can restore vital signs rapidly and improve symptomatology by decreasing preload through increased intrathoracic pressures. The use of noninvasive ventilation in conjunction with medical therapy appears to reduce the need for intubation compared to medical therapy alone.35
High-flow nasal cannula (HFNC) has been increasingly popular for treatment of respiratory failure. HFNC provides warmed and humidified oxygen through a nasal cannula at flow rates higher than the traditional nasal cannula. Limited data exist for the role HFNC plays in the management of acute heart failure. In small studies, the use of HFNC was associated with improvement in subjective dyspnea, respiratory rates, and oxygenation.36,37 While more evidence is being gathered, this modality may be useful in the management of acute heart failure.
If patients cannot tolerate or are not suitable for noninvasive ventilation, the emergency physician should consider endotracheal intubation and mechanical ventilation. The need for intubation in acute heart failure is relatively uncommon. Considerations for intubation include altered mental status, inability to tolerate noninvasive ventilation, and continued deterioration despite appropriate medical management. Hypercapnia due to impending respiratory failure may present with alterations in consciousness. Rapid sequence intubation remains the standard approach with administration of a dissociative and paralytic agent. After intubation, ventilatory settings should be titrated carefully to maintain adequate oxygenation with careful monitoring of the patient’s hemodynamic status.
After addressing oxygenation and ventilation, evaluation of cardiovascular hemodynamics can guide the use of vasodilating agents. Nitrates function through the activation of enzymes that relax smooth muscle and lead to vasodilation. At lower doses, nitrates provide vasodilation, which can help reduce preload. Escalation of dosing eventually affects afterload reduction through arteriolar dilation.
In the acute setting, nitroglycerin can be administered either sublingually or transdermally, alleviating the immediate need for intravenous access. Transdermal application can have inconsistent absorption rates, making it a less than ideal agent. Once intravenous access is established, nitroglycerin can be administered as a continuous drip. Nitroglycerin has rapid onset and offset, making it a good agent for rapid fluctuations in blood pressure. The intravenous drip then can be titrated based on vital signs, clinical presentation, and overall response to therapeutic interventions. Use of sodium nitroprusside has fallen out of favor because of the risk of cyanide toxicity and increased rates of unintended hypotension.
Furosemide, a loop diuretic, is another medication frequently used in patients with ADHF. Currently, there is limited statistical evidence on the safety and efficacy of routine use; however, extensive observational experience has demonstrated symptomatic relief.38 The administration of loop diuretics should be considered promptly, as early administration has been associated with lower in-hospital mortality.39 Careful consideration should be given prior to administration of this medication. Patients who are hypotensive or who potentially may need fluid challenges or other medications to augment perfusion may not be appropriate candidates for diuretic use.
The use of opioid analgesics remains a controversial topic in the management of heart failure. Morphine sulfate has properties associated with the release of vasoactive histamine, which can result in downstream vasodilation. Additionally, morphine may play a role in blunting the hyperactive sympathetic nervous system activity seen in patients with ADHF. Administration of opioids carries a significant adverse risk profile, including respiratory depression and worsening of hypotension. A systematic review of the use of opioids in ADHF found no evidence of benefit with some evidence of harm.40
Acute Decompensated Heart Failure With Inadequate Perfusion
In managing the acute heart failure patient with hypotension, it is important to distinguish between those patients with reduced ejection fraction and those with preserved ejection fraction. The management of hypotension in these two groups can differ significantly. For patients with reduced ejection fraction and the presence of shock, the initial therapy should include starting an inotropic agent.23
Second-line treatment in patients with reduced ejection fraction should involve the addition of a vasopressor, although the practitioner should be aware that this may increase left ventricular afterload. In patients with severely reduced ejection fraction (< 25%) and shock, nonpharmacologic treatment strategies, such as intra-aortic balloon pump, extracorporeal circulatory membrane oxygenation (ECMO), or ventricular assist devices, should be considered as a temporizing measure during acute episodes.1
In contrast, patients with preserved ejection fraction and hypotension should not receive inotropic support. Rather, these patients may benefit from the addition of a vasopressor as well as diuretics. In the unique cases of patients presenting with left ventricular outflow obstruction, such as is seen with hypertrophic cardiomyopathy, the therapeutic approach should include beta-blockers, vasopressors, and cautious hydration depending on the presence of pulmonary edema.
It is important to note that regardless of the intervention used to reverse the shock state in a patient with acute heart failure, all measures should be considered temporizing, as there is little to no evidence to suggest that these agents provide any long-term benefit.40
Both dobutamine and milrinone are recommended as temporizing inotropes in the setting of hypotension in patients with reduced ejection fraction.1 As noted earlier, inotropic therapy should not be initiated in patients with preserved ejection fraction or patients with left ventricular outflow obstruction. For patients in whom inotropic support has been initiated, continuous cardiac monitoring is required. The practitioner should consider reducing or stopping the infusion if tachydysrhythmias or worsening hypotension occurs.13
Milrinone is a phosphodiesterase inhibitor that works by increasing cyclic adenosine monophosphate in cardiac cells, thus producing a positive inotropic effect. Milrinone also reduces systemic and pulmonary vascular resistance in addition to improving left ventricular compliance. Milrinone has no beta-adrenergic activity associated with its mechanism of action. Therefore, its effect is not diminished in patients taking beta-blocker therapy.41 Dobutamine works by binding to beta-1 adrenergic receptors, which results in increased stroke volume and cardiac output. It also has a minor effect in decreasing systemic vascular resistance.42
Again, vasopressor therapy should be considered in patients with reduced ejection fraction who are refractory to inotropic support and in patients with preserved ejection fraction or in those with outflow obstruction. Although there is no evidence to support their clinical efficacy or safety, these agents still should be considered as a temporizing measure in patients displaying signs of shock. Commonly used vasopressors include norepinephrine, high-dose dopamine, and vasopressin.43
Management of Chronic Heart Failure
Chronic heart failure is a complex syndrome that progresses over a substantial period of time. Most patients with heart failure will require medical management outside of the emergency department. Despite this, it is still important for emergency providers to be familiar with the foundations of chronic heart failure therapy. The principles behind long-term therapy involve the neurohormonal response of the body to the systemic impact of global heart failure.
Angiotensin converting-enzyme (ACE) inhibitors provide therapy for left ventricular dysfunction by blocking angiotensin II and aldosterone secretion. This class of medication has been demonstrated to increase survival in all classes of chronic heart failure.44
Beta-blocking agents play an important role in increasing ejection fraction and decreasing mortality in chronic heart failure.45 The adrenergic blockade provided by this class helps to combat the long-term activation of the sympathetic nervous system in chronic heart failure. Long-standing elevation of sympathetic activity has deleterious effects on the cardiovascular system and can worsen heart failure. Current guidelines recommend beta-blockers for all patients with symptomatic left ventricular dysfunction.46
Low-dose diuretics are used to prevent the recurrence of heart failure, despite the fact that there are limited data to guide use.47 These medications tend to carry significant side effects, including hypovolemia and electrolyte abnormalities. The potassium-sparing diuretics spironolactone and eplerenone are associated with a reduced risk of death in patients with heart failure.48 They have been shown to improve left ventricular functioning in patients with severe heart failure who already are taking ACE inhibitor and diuretic therapy.49
Digoxin, a cardiac glycoside, works by inhibiting ATP-dependent sodium/potassium pumps in cardiac myocytes. Digoxin has been shown to reduce the rate of hospitalization in chronic heart failure and lower mortality when used in conjunction with ACE inhibitor and diuretic therapy.50
Electrical Therapy
Implantable cardioverter defibrillators (ICD) play an important role in the long-term management of patients with heart failure with severely reduced ejection fraction. Their use in this patient population has been shown to reduce mortality.53 Patients with heart failure are at increased risk of ventricular dysrhythmias, which may lead to sudden cardiac death. In several clinical trials, implantation of an ICD has been shown to decrease mortality when compared to medical therapy alone.54
Patients with heart failure who survived a sudden cardiac arrest or had an episode of sustained ventricular tachycardia are candidates for ICD therapy as secondary prevention. Patients receiving ICDs have a significant reduction in sudden cardiac death.55
Noncardiogenic Pulmonary Edema
Noncardiogenic pulmonary edema is characterized by proteinaceous fluid accumulation in the alveoli of the lungs from a source other than the heart. In heart failure (cardiogenic pulmonary edema), pulmonary congestion is due to elevated pulmonary capillary wedge pressures. Noncardiogenic pulmonary edema presents similarly, except the etiology of fluid accumulation is from a different source.56
The distinction between the sources of pulmonary edema is not always obvious, and in many cases, there may be significant overlap between the two. The history and physical exam in affected patients may demonstrate significant hypoxemia and shortness of breath. Classically, noncardiogenic pulmonary edema presents with radiographic evidence of pulmonary congestion without hemodynamic evidence of cardiac dysfunction. In the emergency department, the ability to obtain invasive hemodynamic monitoring (e.g., Swan-Ganz catheter) is limited. Therefore, the exact etiology of pulmonary edema may be surmised based on the preceding historical features.
The major causes of noncardiogenic pulmonary edema are acute respiratory distress syndrome (ARDS), high altitude pulmonary edema (HAPE), opioid overdose, pulmonary embolism, eclampsia, and transfusion-related acute lung injury (TRALI).57
When a patient presents with evidence of pulmonary edema, the emergency physician must attempt to discern the source of fluid overload. Although basic management principles remain the same, careful attention should be paid to the inciting agent if it can be deduced in the emergency department. Patients with ARDS may require significant ventilatory support with low tidal volumes. If pulmonary embolism is suspected, anticoagulation may be required with thrombolytic therapy. The specific management plan for noncardiogenic pulmonary edema should be tailored to the clinical presentation when evident.
Disposition
The majority of patients presenting to the emergency department with an acute heart failure exacerbation will require admission to the hospital setting. Informed decision-making with patients can help guide disposition based on factors including severity of illness and access to care outside of the hospital setting.
One of the unique challenges in the disposition of patients with acute heart failure in the emergency department is that no risk stratification tools are readily available. Prospective testing of four acute heart failure prediction rules showed no utility in the emergency department setting.56 Therefore, emergency department providers must review each clinical encounter for the presence of high-risk physiologic markers associated with morbidity and mortality in acute heart failure. These markers include renal dysfunction, hypotension, hyponatremia, elevated BNP, and elevated troponin.55
Observation is a reasonable alternative for patients with minimal symptomology who do not require supplemental oxygen and who display low-risk features on diagnostic evaluation. If available, the observation setting can allow for continued diuresis as well as cardiac monitoring with serial cardiac enzyme testing if acute coronary syndrome is being evaluated. Most patients will have complete symptom resolution in 12 to 24 hours. Additionally, the observation unit can help expedite the acquisition of a formal echocardiogram and cardiology consultation. Linkage to outpatient therapy is crucial because follow-up within five days has been shown to decrease readmissions.57 Newly diagnosed cases of mild heart failure from the emergency department may benefit greatly from a period of observation to help with patient education and linkage to outpatient care.
Clinical judgment and response to therapeutic interventions in the emergency department will help guide placement into a monitored regular floor bed or intensive care setting. Physicians should consider resources available in their hospital setting when triaging patients to the inpatient unit. Currently, there is no well-validated guideline to determine appropriate disposition. Patients who require invasive ventilatory support or vasopressor therapy for refractory hypotension typically require intensive care unit (ICU) admission. Roughly 10-12% of patients in the United States are admitted to the ICU for ADHF.58 According to the 2016 European Society of Cardiology heart failure guidelines, a patient should be placed in an intensive care setting if one or more of the following criteria are met:40
- The patient is intubated or likely to require invasive respiratory support/intubation (tachypnea ≥ 25 breaths per minute, accessory muscle usage, and/or refractory hypoxemia).
- Signs and symptoms of hypoperfusion and/or systolic blood pressure < 90 mmHg are present.
- The heart rate is < 40 or > 130 beats/minute.
Regardless of the ultimate disposition, the emergency physician should ensure that patients with heart failure have appropriate access to outpatient care. When available, measures should be taken to engage a patient’s cardiologist in the plan of care. If a patient with newly diagnosed heart failure does not have appropriate follow-up, the emergency physician should attempt to assist with linkage of care. Medication initiation, lifestyle modification, and other interventions can take place in the outpatient setting to help reduce heart failure-related visits to the emergency department.
Summary
Heart failure is an important clinical entity that represents a wide spectrum of illness. Acute presentations in the emergency department can present as a diagnostic challenge. Most patients who present to the emergency department will respond rapidly to nitrates, diuretics, and positive pressure ventilation. Patients in shock with evidence of hypoperfusion may require inotropic or vasopressor support depending on underlying cardiac physiology.
The differential for acute heart failure is broad and will vary based on the patient’s symptomatology. Although no single diagnostic test exists, the emergency physician can use radiography, ultrasonography, electrocardiography, and laboratory testing to help diagnose and manage patients appropriately.
The overwhelming majority of patients presenting to the emergency department with ADHF will require admission.5 As patients’ life expectancy increases with advancements in medical therapy for chronic heart failure, the prevalence of heart failure is expected to increase greatly over the next decade.7
REFERENCES
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2013;62:e147.
- Bragg DA, Crowl A, Manlove E. Hepatitis C: A new era. Prim Care 2017;44:631-642.
- Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics —2013 update: A report from the American Heart Association. Circulation 2013;127:e6.
- Collins SP, Levy PD, Pang PS, Gheorghiade M. The role of the emergency department in acute heart failure clinical trials — Enriching patient identification and enrollment. Am Heart J 2013;165:902-909.
- Heidenreich PA, Albert NM, Allen LA, et al. Forecasting the impact of heart failure in the United States: A policy statement from the American Heart Association. Circ Heart Fail 2013;6:606-619.
- Owan TE, Redfield MM. Epidemiology of diastolic heart failure. Prog Cardiovasc Dis 2005;47:320-332.
- Yancy CW, Jessup M, Bozkurt B, et al. 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J Am Coll Cardiol 2017;70:776-803.
- Ezekowitz JA, O’Meara E, McDonald MA, et al. 2017 Comprehensive Update of the Canadian Cardiovascular Society Guidelines for the Management of Heart Failure. Can J Cardiol 2017;33:1342-1433.
- Udelson JE. Heart failure with preserved ejection fraction. Circulation 2011;124:540.
- Mebazaa A, Gheorghiade M, Piña IL, et al. Practical recommendations for prehospital and early in-hospital management of patients presenting with acute heart failure syndromes. Crit Care Med 2008;36(1 Suppl):129.
- Koitabashi N, Kass DA. Reverse remodeling in heart failure — mechanisms and therapeutic opportunities. Nat Rev Cardiol 2011;9:147-157.
- Lala A, Desai AS. The role of coronary artery disease in heart failure. Heart Fail Clin 2014;10:353-365.
- Heart Failure Society of America; Lindenfeld J, Albert NM, Boehmer JP, et al. HFSA 2010 Comprehensive Heart Failure Practice Guideline. J Card Fail 2010;16:1.
- O’Brien JF, Hunter CL. Heart Failure. In: Walls R, Hockberger R, Gausche-Hill M. Rosen’s Emergency Medicine: Concepts and Clinical Practice. 9th ed. Philadelphia: Elsevier; 2018:986.e6.
- Curtis LH, Whellan DJ, Hammill BG, et al. Incidence and prevalence of heart failure in elderly persons, 1994-2003. Arch Intern Med 2008;168:418-424.
- Leone A. Toxics of tobacco smoke and cardiovascular system: From functional to cellular damage. Curr Pharm Des 2015;21:4370-4379.
- Urbano-Marquez A, Estruch R, Navarro-Lopez F, et al. The effects of alcoholism on skeletal and cardiac muscle. N Engl J Med 1989;320:409-415.
- Bovelli D, Plataniotis G, Roila F; ESMO Guidelines Working Group. Cardiotoxicity of chemotherapeutic agents and radiotherapy-related heart disease: ESMO Clinical Practice Guidelines. Ann Oncol 2010;21 (Suppl 5):277.
- Feenstra J, Grobbee DE, Remme WJ, Stricker BH. Drug-induced heart failure. J Am Coll Cardiol 1999;33:1152-1162.
- Kemp CD, Conte JV. The pathophysiology of heart failure. Cardiovasc Pathol 2012;21:365-371.
- Lainchbury JG, Richards AM. Exercise testing in the assessment of chronic congestive heart failure. Heart 2002;88:538-543.
- Lee DS, Austin PC, Rouleau JL, et al. Predicting mortality among patients hospitalized for heart failure: Derivation and validation of a clinical model. JAMA 2003;290:2581-2587.
- McMurray JJ, Adamopoulos S, Anker SD, et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail 2012;14:803-869.
- Heart Failure Society of America. Evaluation of patients for ventricular dysfunction and heart failure. J Card Fail 2006;12:16.
- Littmann L. Large T wave inversion and QT prolongation associated with pulmonary edema: A report of nine cases. J Am Coll Cardiol 1999;34:1106-1110.
- Attias D, Mansencal N, Auvert B, et al. Prevalence, characteristics, and outcomes of patients presenting with cardiogenic unilateral pulmonary edema. Circulation 2010;122:1109-1115.
- Wang CS, FitzGerald JM, Schulzer M, et al. Does this dyspneic patient in the emergency department have congestive heart failure? JAMA 2005;294:1944-1956.
- Doust JA, Glasziou PP, Pietrzak E, Dobson AJ. A systematic review of the diagnostic accuracy of natriuretic peptides for heart failure. Arch Intern Med 2004;164:1978-1984.
- Maisel AS, Krishnaswamy P, Nowak RM, et al. Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. N Engl J Med 2002;347:161-167.
- Madamanchi C, Alhosaini H, Sumida A, Runge MS. Obesity and natriuretic peptides, BNP and NT-proBNP: Mechanisms and diagnostic implications for heart failure. Int J Cardiol 2014;176:611-617.
- Kirkpatrick JN, Vannan MA, Narula J, Lang RM. Echocardiography in heart failure: Applications, utility, and new horizons. J Am Coll Cardiol 2007;50:381-396.
- Al Deeb M, Barbic S, Featherstone R, et al. Point-of-care ultrasonography for the diagnosis of acute cardiogenic pulmonary edema in patients presenting with acute dyspnea: A systematic review and meta-analysis. Acad Emerg Med 2014;21:843-852.
- Raphael CE, Whinnett ZI, Davies JE, et al. Quantifying the paradoxical effect of higher systolic blood pressure on mortality in chronic heart failure. Heart 2009;95:56-62.
- Park JH, Balmain S, Berry C, et al. Potentially detrimental cardiovascular effects of oxygen in patients with chronic left ventricular systolic dysfunction. Heart 2010;96:533-538.
- Collins SP, Mielniczuk LM, Whittingham HA, et al. The use of noninvasive ventilation in emergency department patients with acute cardiogenic pulmonary edema: A systematic review. Ann Emerg Med 2006;48:269.e4.
- Carratala Perales JM, Llorens P, Brouzet B, et al. High-flow therapy via nasal cannula in acute heart failure. Rev Esp Cardiol 2011;64:723-725.
- Roca O, Pérez-Terán P, Masclans JR, et al. Patients with New York Heart Association class III heart failure may benefit with high flow nasal cannula supportive therapy: High flow nasal cannula in heart failure. J Crit Care 2013;28:741-746.
- Felker GM, O’Connor CM, Braunwald E; Heart Failure Clinical Research Network Investigators. Loop diuretics in acute decompensated heart failure: Necessary? Evil? A necessary evil? Circ Heart Fail 2009;2:56-62.
- Matsue Y, Damman K, Voors AA, et al. Time-to-furosemide treatment and mortality in patients hospitalized with acute heart failure. J Am Coll Cardiol 2017;69:3042-3051.
- National Clinical Guideline Centre (UK). Acute Heart Failure: Diagnosing and Managing Acute Heart Failure in Adults. London: National Institute for Health and Care Excellence (UK); 2014.
- Simonton CA, Chatterjee K, Cody RJ, et al. Milrinone in congestive heart failure: Acute and chronic hemodynamic and clinical evaluation. J Am Coll Cardiol 1985;6:453-459.
- Liang CS, Sherman LG, Doherty JU, et al. Sustained improvement of cardiac function in patients with congestive heart failure after short-term infusion of dobutamine. Circulation 1984;69:113-119.
- Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail 2016;18:891-975.
- Deedwania PC, Carbajal E. Evidence-based therapy for heart failure. Med Clin North Am 2012;96:915-931.
- Barrese V, Taglialatela M. New advances in beta-blocker therapy in heart failure. Front Physiol 2013;4:323.
- Yilmaz MB, Laribi S, Mebazaa A. Managing beta-blockers in acute heart failure: When to start and when to stop? Curr Heart Fail Rep 2010;7:110-115.
- von Lueder TG, Atar D, Krum H. Diuretic use in heart failure and outcomes. Clin Pharmacol Ther 2013;94:490-498.
- Chatterjee S, Moeller C, Shah N, et al. Eplerenone is not superior to older and less expensive aldosterone antagonists. Am J Med 2012;125:817-825.
- Phelan D, Thavendiranathan P, Collier P, Marwick TH. Aldosterone antagonists improve ejection fraction and functional capacity independently of functional class: A meta-analysis of randomised controlled trials. Heart 2012;98:1693-1700.
- Digitalis Investigation Group; Ahmed A, Waagstein F, Pitt B, et al. Effectiveness of digoxin in reducing one-year mortality in chronic heart failure in the Digitalis Investigation Group trial. Am J Cardiol 2009;103:82-87.
- Bardy GH, Lee KL, Mark DB, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med 2005;352:225-237.
- Woods B, Hawkins N, Mealing S, et al. Individual patient data network meta-analysis of mortality effects of implantable cardiac devices. Heart 2015;101:1800-1806.
- Betts TR, Sadarmin PP, Tomlinson DR, et al. Absolute risk reduction in total mortality with implantable cardioverter defibrillators: Analysis of primary and secondary prevention trial data to aid risk/benefit analysis. Europace 2013;15:813-819.
- Ware LB, Matthay MA. Clinical practice. Acute pulmonary edema. N Engl J Med 2005;353:2788-2796.
- Weintraub NL, Collins SP, Pang PS et al. Acute heart failure syndromes: Emergency department presentation, treatment, and disposition: Current approaches and future aims: A scientific statement from the American Heart Association. Circulation 2010;122:1975.
- Auble TE, Hsieh M, McCausland JB, Yealy DM. Comparison of four clinical prediction rules for estimating risk in heart failure. Ann Emerg Med 2007;50:135.e2.
- Hernandez AF, Greiner MA, Fonarow GC, et al. Relationship between early physician follow-up and 30-day readmission among Medicare beneficiaries hospitalized for heart failure. JAMA 2010;303:1716-1722.
- Safavi KC, Dharmarajan K, Kim N, et al. Variation exists in rates of admission to intensive care units for heart failure patients across hospitals in the United States. Circulation 2013;127:923-929.
This article will focus primarily on the important aspects of acute decompensated heart failure in the emergency setting. The authors will include a brief synopsis of noncardiogenic pulmonary edema to highlight key principles in the diagnosis and management.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
