Approach to Pediatric Abdominal Pain in the ED: Part I
August 1, 2019
Reprints
AUTHORS
Jeffrey T. Sakamoto, MD, Resident, Stanford Emergency Medicine Residency Program, Stanford, CA
Monika Thomas-Uribe, MD, MPH, Department of Pediatrics, UCSF-Fresno
Pablo Tarsicio Uribe-Leitz, MD, MPH, Center for Surgery and Public Health: Harvard Medical School, Harvard T.H. Chan School of Public Health, and the Department of Surgery, Brigham & Women’s Hospital, Boston, MA
N. Ewen Wang, MD, Professor of Emergency Medicine, Stanford School of Medicine, Stanford, CA
PEER REVIEWER
Larry B. Mellick, MD, MS, FAAP, FACEP, Vice Chairman for Academic Affairs, Interim Section Chief of Pediatric Emergency Medicine, Assistant Residency Director, Professor of Emergency Medicine, University of South Alabama, Mobile
Executive Summary
- In the approach to pediatric abdominal pain, certain entities are more common in different age groups; an age-based differential diagnosis of abdominal pain is shown in Table 1.
- In the neonate with abdominal pain, always consider necrotizing enterocolitis, malrotation with midgut volvulus, Hirschsprung’s disease, pyloric stenosis, and neonatal testicular torsion.
- The chest exam should focus on identifying abnormal breathing patterns (e.g., tachypnea in sepsis) and identifying causes of referred abdominal pain, such as lower lobe pneumonia or pericarditis. The genitourinary exam should focus on identifying hernias, and the scrotal examination should focus on identifying torsion (tender testicle, horizontal testicle, erythematous scrotum). The anal exam should focus on identifying anal fissures, especially if there is report of hematochezia. The skin exam should focus on identifying rashes (e.g., purpura) and prior surgical incisions.
- The most common presentation of necrotizing enterocolitis includes unexpected and acute onset of feeding intolerance and bloody stools. In general, neonates will be toxic-appearing with tachypnea, pallor, poor skin turgor, and lethargy. The abdominal exam can reveal distention, abdominal erythema/discoloration, and signs of peritonitis due to perforation. Pneumatosis intestinalis may be seen on a radiograph.
- The most common feature of malrotation is bilious vomiting. All neonates with bilious vomiting should be evaluated promptly. If volvulus has occurred, as the intestine becomes ischemic, neonates may develop abdominal distension, peritonitis, hematochezia, and toxic appearance with hemodynamic instability.
Abdominal pain is a common pediatric chief complaint with a diversity of etiologies. Many are benign, but some have the potential for devastating consequences if a timely diagnosis is not made. Understanding and practicing a comprehensive approach facilitates consideration of more serious pathology while allowing for a focused diagnostic plan. This two-part series guides the clinician to a practical clinical approach to pediatric abdominal pain.
— Ann M. Dietrich, MD, Editor
Acute abdominal pain accounts for approximately 5-10% of all pediatric emergency department (ED) visits,1 with more than 900,000 visits annually by children younger than 15 years of age.2 According to the Pediatric Emergency Care Applied Research Network (PECARN) data set, nonspecific abdominal pain was the ED diagnosis for 3.5% of patients with an average ED length of stay of more than three hours.3 Abdominal pain can be a nonspecific diagnosis, but it also can be a symptom of a serious pathology. Children have distinct etiologies of abdominal pain that are epidemiologically different from adults. Therefore, emergency providers (EPs) must be aware of the differential diagnosis and treatment of this chief complaint among pediatric patients. Although causes may differ, the principle of considering and ruling out life-threatening sources remains similar. Pediatric abdominal pain can be caused by a diverse set of pathologies that span the entire spectrum of disease severity.
This two-part series will discuss a brief pathophysiology of abdominal pain and an approach to the initial history and physical exam in a pediatric ED patient population. Part I will focus on the neonate, while part II will focus on the infant, toddler, and child. The authors will discuss an age-based differential diagnosis, appropriate diagnostic studies, and specific treatment for emergent but less common diseases. In addition, they will discuss common benign causes of abdominal pain and their diagnosis and treatment. Abdominal pain due to trauma and obstetric/gynecology causes are beyond the scope of this article.
Pathophysiology
Abdominal pain is caused by the stimulation of nociceptive receptors and afferent sympathetic stretch receptors. It can be classified as visceral or parietal (somatic). Visceral pain is caused by the excessive contraction/tension of small bowel, colon, organ capsules, or mesentery. Etiologies such as infection, toxins, inflammation, or ischemia can lead to visceral abdominal pain. This type of pain typically is characterized as diffuse and dull.
Parietal pain arises from inflammation of the contiguous parietal peritoneum (peritonitis) from local inflammation due to infection, ischemia, blood, perforation, and other conditions (e.g., appendicitis). This type of pain is characterized as sharp, intense, and localized. While an understanding of the pathophysiology of abdominal pain is important, patients (especially children) may not be able to communicate the quality of their pain.
Initial Assessment and Resuscitation
Initial assessment should determine if the child is toxic- or nontoxic-appearing. In all children, assessment of airway, breathing, circulation, (ABCs) vital signs, and responsiveness can be used to determine the toxic vs. nontoxic child. However, compensatory vasoconstrictive mechanisms may mask signs of shock. Older children who can communicate their symptoms, history, and quality of pain may be diagnosed more easily than sick neonates and infants. In neonates and infants specifically, assessment should focus on appearance (tone, interactive, consolability, gaze, strength of cry), work of breathing (tachypnea, retractions), circulation (capillary refill, skin color/turgor, fontanelle), responsiveness, and vital signs.4
Abdominal catastrophes, such as midgut volvulus, incarcerated hernia, intussusception, necrotizing enterocolitis, toxic megacolon, perforation, and others, can cause profound shock and death, especially in neonates and infants. Although this article will not address basic resuscitation measures, in the toxic-appearing child, ABCs should be addressed initially in all cases.
History
The history of a patient with abdominal pain should focus primarily on determining whether the child requires a workup for pathology that may require urgent or emergent surgical intervention. Depending on a child’s age and developmental stage, obtaining a meaningful history in the pediatric patient can be difficult. Parents can help translate a child’s behavior into symptoms. In the neonate/infant, symptoms and a high index of suspicion are essential in initiating a workup, as the abdominal exam may be difficult and unreliable.
The onset, severity, timing, location, progression, exacerbating and alleviating factors, and quality of the abdominal pain should be obtained. Further, it is important to ask about associated symptoms such as fever, nausea, vomiting (bilious vs. nonbilious, bloody vs. nonbloody), dysuria, hematuria, cough, sore throat, relationship with meals, urination, defecation and movement of the pain, similar episodes, and the effect of the pain on level of activity. Past medical history, birth history, past surgical history, travel history, ingestions and food exposure, and sick contacts also are essential. It is important to ask the patient (or parent) about the child’s baseline appetite, food intake, and bowel movements, as well as how the current illness compares to baseline.
Physical Exam
The initial assessment and physical exam should focus on identifying the toxic vs. nontoxic child. Physical exam findings consistent with a surgical abdomen include toxic appearance, abdominal distension, focal tenderness, and signs of peritonitis. In all cases, a systematic examination of the head, chest, abdomen, genitourinary tract, and skin should be completed expediently. The abdominal exam should focus on identifying the location of tenderness, distension, organomegaly, abnormal bowel sounds, and signs of peritonitis such as rebound, guarding, rigidity, or pain with movement.
Examination of the head and neck should focus on identifying pharyngitis or lymphadenopathy, as these can be associated with viral syndromes that cause abdominal pain. The chest exam should focus on identifying abnormal breathing patterns (e.g., tachypnea in sepsis) and identifying causes of referred abdominal pain, such as lower lobe pneumonia or pericarditis. The genitourinary exam should focus on identifying hernias, and the scrotal examination should focus on identifying torsion (tender testicle, horizontal testicle, erythematous scrotum). The anal exam should focus on identifying anal fissures, especially if there is report of hematochezia. The skin exam should focus on identifying rashes (e.g., purpura) and prior surgical incisions.5,6
In neonates and crying infants, the abdominal exam may be challenging. Therefore, distracting and soothing the infant prior to examining the abdomen is essential. Strategies for calming the crying infant to facilitate a physical exam include offering the child a pacifier, examining the child in the caretaker’s arms, facing the child toward the caretaker (away from the physician), or having the parent palpate the abdomen for you. In the older child, the ability to walk, jump, or climb onto the bed to get a sticker can be a good assessment for peritonitis.
Differential Diagnosis
We suggest using an age-based differential diagnosis as an approach to pediatric abdominal pain. (See Tables 1 and 2.)
Table 1. Age-Based Differential Diagnosis for Emergency and Common Causes of Pediatric Abdominal Pain |
||||
Neonate (0-28 days) |
Infant (1-24 months) |
Toddler/Child (2-10 years) |
Adolescent (11-18 years) |
|
|
Emergent/ Urgent |
Necrotizing enterocolitis Malrotation with midgut volvulus Hirschsprung’s disease with toxic megacolon Pyloric stenosis Testicular torsion Obstruction from atresia |
Intussusception Incarcerated hernia Hirschsprung’s disease with toxic megacolon Meckel’s diverticulitis Hemolytic uremic syndrome |
Appendicitis Meckel’s diverticulitis Testicular/ ovarian torsion Intussusception Hemolytic uremic syndrome |
Appendicitis Cholecystitis/ cholangitis Testicular/ ovarian torsion Ectopic pregnancy |
|
Common/ Generally Benign |
Colic Reflux Milk protein allergy |
Gastroenteritis Viral syndrome |
Gastroenteritis Viral syndrome Constipation |
Gastroenteritis Viral syndrome Constipation |
Table 2. Age-Based Differential Diagnosis for Pediatric Non-Gastrointestinal Causes of Apparent Abdominal Pain |
||||
Neonate (0-28 days) |
Infant (1-24 months) |
Toddler/Child (2-10 years) |
Adolescent (11-18 years) |
|
|
Other Causes of Nonspecific Abdominal Pain |
Sepsis Inborn errors of metabolism |
Sepsis Urinary tract infection Foreign body Pneumonia |
Sepsis Urinary tract infection Foreign body Pneumonia Henoch-Schonlein purpura Diabetic ketoacidosis Malignancy Toxic ingestion |
Sepsis Urinary tract infection Foreign body Pneumonia Diabetic ketoacidosis Malignancy Toxic ingestion Pelvic inflammatory disease Tuboovarian abscess Ovarian cyst Hepatitis Nephrolithiasis |
Case #1
A 2-week-old boy presents to the ED with fussiness and bilious emesis. The birth history was uncomplicated. Over the past day, the baby has been eating less and is inconsolable. He began vomiting green-colored emesis eight hours prior to arrival. On exam, the baby appears pale, with a distended abdomen. Vital signs include heart rate of 160 beats per minute, systolic blood pressure of 80 mmHg, and respiratory rate of 40. IV access, fluid bolus, and surgical consultation are initiated.
Differential Diagnosis
In neonates, the differential diagnosis of acute abdominal pain requiring urgent/emergent intervention includes necrotizing enterocolitis (NEC), malrotation with midgut volvulus, pyloric stenosis, Hirschsprung’s disease/toxic megacolon, and neonatal testicular torsion. (See Table 3.)
Table 3. Treatment for Neonatal Gastrointestinal Emergencies7 |
|||
Necrotizing Enterocolitis |
Malrotation |
Pyloric Stenosis |
Hirschsprung’s Disease |
|
Address airway, breathing, and circulation Symptomatic treatment: bowel rest, gastric decompression Antibiotics: ampicillin, cefotaxime or gentamicin, clindamycin or metronidazole Consult surgery |
Address airway, breathing, and circulation Place nasogastric tube Order upper gastrointestinal study Consult surgery |
Assess for dehydration and electrolyte imbalance Stabilize patient with IV fluids and electrolyte replacement Consult surgery |
Address airway, breathing, and circulation Symptomatic treatment Consult surgery If Hirschsprung's-associated enterocolitis is suspected, treat accordingly |
Necrotizing Enterocolitis
NEC refers to ischemic necrosis of the intestinal mucosa associated with severe inflammation, invasion of enteric bacteria, and pneumatosis intestinalis. The pathophysiology is poorly understood, but is likely multifactorial, including genetic predisposition, altered microbiota, immature intestine, and exaggerated immune response.8 NEC affects approximately 7% of low birth weight infants (< 1,500 g) and causes significant morbidity, with mortality rates as high as 30%.8 It primarily affects neonates who are premature (< 32 weeks) and low birth weight (< 1,500 g).
Other risk factors include formula-fed children and microbial dysbiosis (prior antibiotic use, histamine blockers, cow’s milk, enteral feeding).9 However, even term infants can present with NEC, accounting for approximately 10% of cases.10 Infants born < 26 weeks present later at a median of 23 days, while infants born > 31 weeks present earlier at a median of 11 days.11
Clinical Features. Many NEC cases are diagnosed in the neonatal ICU in premature infants;8 thus, term infants who have been discharged home are at increased risk of late presentation. The most common presentation includes unexpected and acute onset of feeding intolerance. Further historical findings may include increased fussiness, bilious vomiting, and bloody stools. In general, neonates will be toxic-appearing with tachypnea, pallor, poor skin turgor, and lethargy. Symptoms may be nonspecific. Therefore, it is important to maintain a high index of suspicion. The abdominal exam can reveal distention. Late findings can reveal abdominal erythema/discoloration and signs of peritonitis due to perforation.8 NEC must be considered in any neonate with new feeding intolerance, abdominal distention, bilious emesis, or toxic appearance.
Diagnostic Imaging and Laboratory Workup. Diagnostic imaging initially should include abdominal X-ray (anterior-posterior and left lateral decubitus) to evaluate for obstruction and free air. Dilated loops of bowel also can be present early in NEC due to ileus.8 Pneumatosis intestinalis (see Figure 1), pneumoperitoneum, or portal venous gas are late signs. Ultrasound has been proposed for diagnosis to evaluate bowel wall thickness, inflammation, and pneumatosis.12 However, routine ultrasound use is not typical in most centers as a part of the initial workup. Laboratory tests should include basic chemistry, complete blood count, coagulation profile, lactate, blood gas, urinalysis, and blood cultures, and may reveal thrombocytopenia, elevated lactate, and hyponatremia.
Figure 1. Necrotizing Enterocolitis |
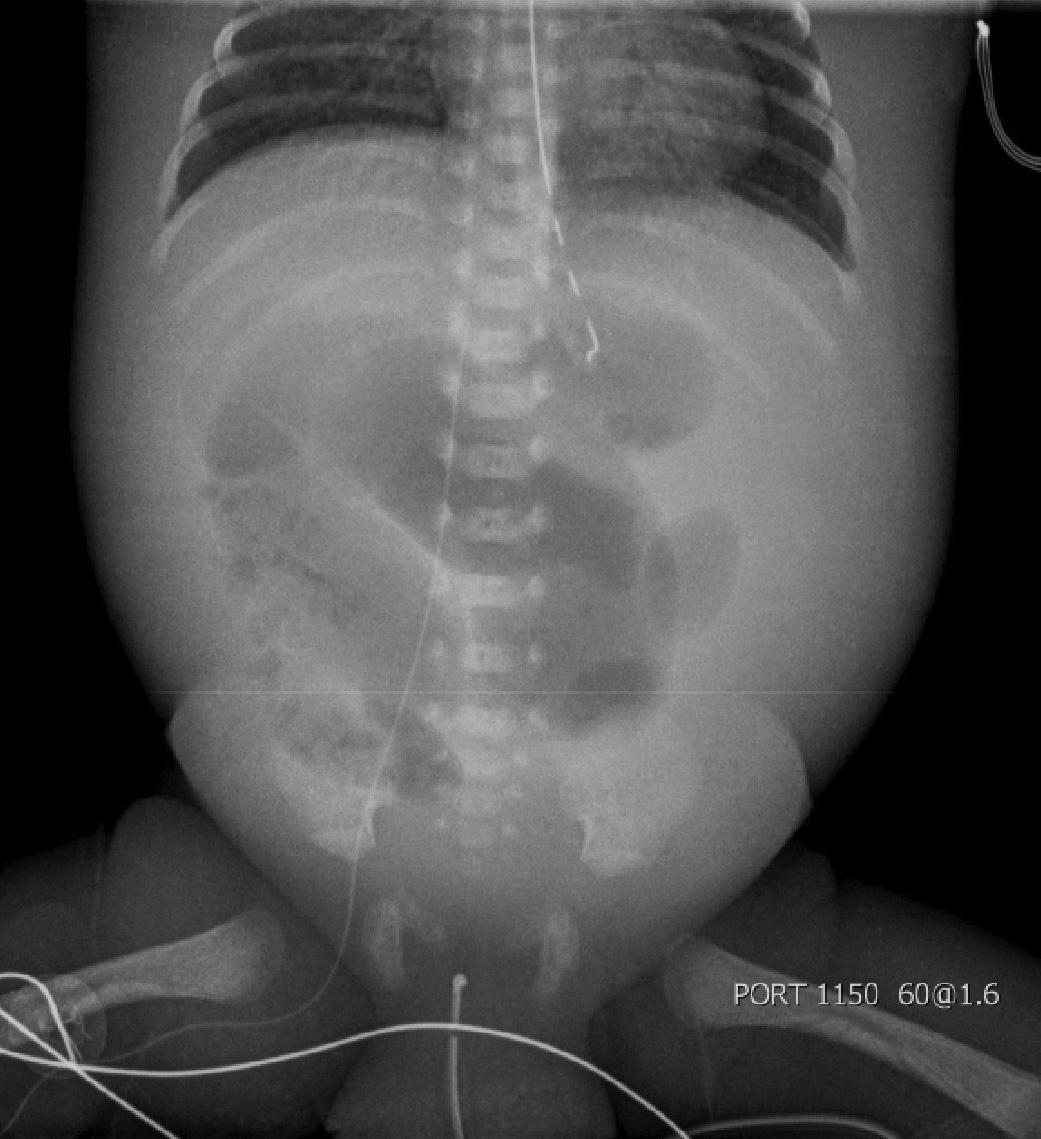 |
|
Plain film of the abdomen demonstrating intramural air (pneumatosis intestinalis) in the right lower abdomen, consistent with necrotizing enterocolitis. |
|
Image courtesy of Casey Grover, MD. |
ED Management/Disposition. Once there is a suspicion for NEC, beyond initial resuscitation addressing airway, breathing, and circulation, antibiotics should be initiated with broad-spectrum coverage using a regimen such as ampicillin, gentamicin, and metronidazole/clindamycin.
With the guidance of pediatric surgical colleagues, the patient should be admitted to the neonatal or pediatric intensive care unit (PICU) for further management. Surgical intervention typically is reserved for cases in which there is evidence of perforation or severe disease.8,13
Malrotation With Midgut Volvulus
Intestinal malrotation occurs during embryogenesis when the duodenum, jejunum, and cecum do not fully rotate. This results in the duodenum located in the right upper quadrant and the cecum located in the upper abdomen. Adhesive (Ladd) bands fix the cecum to the other structures in the right upper quadrant including the abdominal wall, gallbladder, and duodenum. This results in a mesenteric base that rotates easily and predisposes children to volvulus, obstruction, and bowel ischemia.14 (See Figure 2.)
Figure 2. Malrotation With Midgut Volvulus |
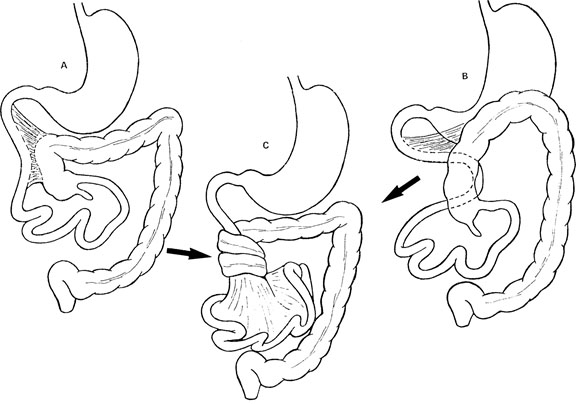 |
|
Image courtesy of Mark Mannenbach, MD, Mayo Clinic. |
Malrotation can occur at an incidence of up to 1 in 6,000 live births.15 Approximately 60% of cases present before age 1 year.15 Although malrotation generally has been considered a neonatal disease, an observational study by Aboagye et al showed that cases of malrotation can present after 1 month of age and even into adulthood.15,16 In addition, up to 30-60% of patients with malrotation have other congenital anomalies, including intestinal webs, congenital diaphragmatic hernia, congenital heart disease (heterotaxy syndrome), omphalocele, gastroschisis, Meckel’s diverticulum, Hirschsprung’s disease, anorectal malformations, and others.14 Malrotation without volvulus can manifest in the older child or adult with an episodic history of distention, nausea, and vomiting that resolves. Malrotation with volvulus or with intermittent volvulus is a surgical emergency and must be recognized and treated immediately.14
Clinical Features. The most common feature of malrotation is bilious vomiting. All neonates with bilious vomiting should be evaluated promptly. If volvulus has occurred, as the intestine becomes ischemic, neonates may develop abdominal distension, peritonitis, hematochezia, and toxic appearance with hemodynamic instability.14 Patients with malrotation without volvulus may present with nonspecific symptoms such as failure to thrive, solid food intolerance, protein-losing enteropathy, malnutrition, and gastrointestinal (GI) bleeding. However, it is possible that patients may have intermittent volvulus. Clinical suspicion for malrotation should be especially high in neonates with a history of premature birth and other congenital anomalies listed above.
Diagnostic Imaging and Laboratory Workup. If the neonate is toxic-appearing with signs of perforation, further imaging is not required, and a pediatric surgeon should be consulted emergently for possible operative management. Because of the risk for ischemia/perforation with volvulus and similar features to NEC, initial diagnostic imaging should include a kidney, ureter, and bladder (KUB) X-ray to evaluate for pneumoperitoneum. At times, a dilated loop of proximal small bowel consistent with obstruction from volvulus may be seen on imaging. (See Figure 3.) Laboratory tests should include basic chemistry, complete blood count, coagulation profile, lactate, blood gas, urinalysis, and blood cultures. Lab results may reveal leukocytosis and elevated lactate.
Figure 3. X-Ray of Neonate With Midgut Volvulus |
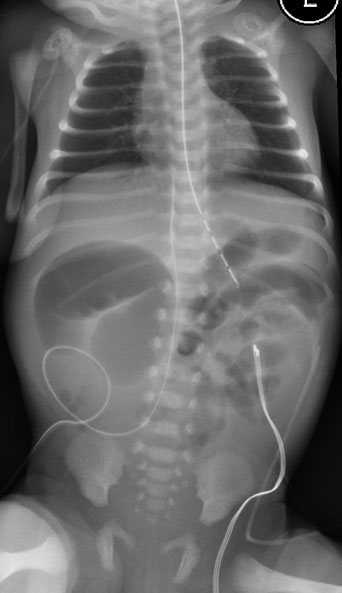 |
|
Asymmetry of the bowel gas with a moderately distended loop of bowel in the right and left side of the abdomen. The newborn has midgut volvulus. |
|
Image courtesy of Dr. Mark Mannenbach, Mayo Clinic. |
If there is a strong suspicion of malrotation with volvulus, a pediatric surgeon should be consulted prior to the imaging study. However, if the patient is stable, after the X-ray, an upper GI contrast series is usually obtained. This is the gold standard diagnostic imaging, with a sensitivity of 96%.17,18 Findings include right-sided ligament of Treitz, corkscrew duodenum, or duodenal obstruction.17,19 However, even with a negative upper GI series, surgeons may pursue operative management if suspicion is high with clinical signs of midgut volvulus as discussed previously.20,21 Ultrasound may be used as an adjunct to diagnose volvulus using the whirlpool sign depicting twisting mesenteric vessels. 22 However, ultrasound should not be used as the sole imaging modality to rule out malrotation.23 (See Figures 4 and 5.)
Figure 4. Upper Gastrointestinal Study Depicting Midgut Volvulus in a Newborn |
|
The duodenal jejunal junction crossed to the left of midline, but did not extend superiorly as expected, and there was no peristalsis identified within the stomach or duodenum. |
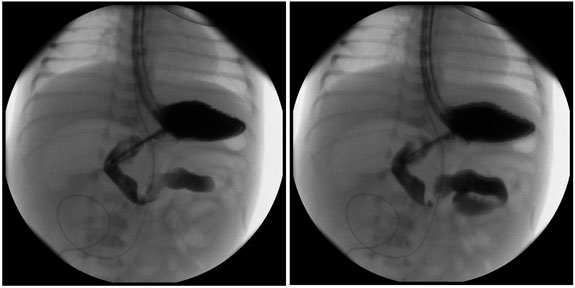 |
Figure 5. Ultrasound Showing SMV/SMA Reversal Sign in Midgut Volvulus |
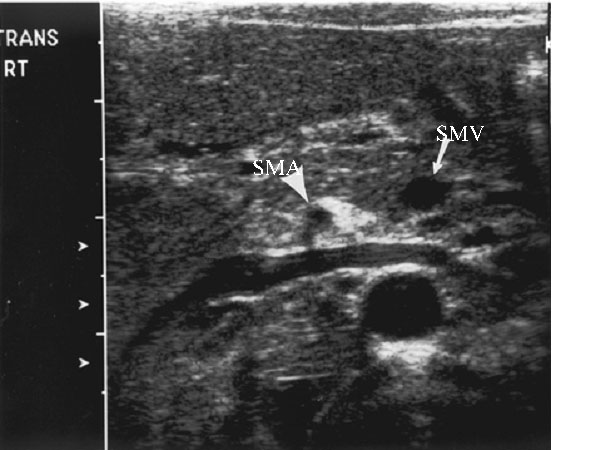 |
|
Image courtesy of Dr. Mark Mannenbach, Mayo Clinic. |
ED Management/Disposition. Initial resuscitation should address airway, breathing, and circulation. Disposition will be made in consultation with the pediatric surgeon for possible immediate operative management. Empiric broad-spectrum antibiotics, such as ampicillin, gentamicin, and metronidazole/clindamycin, should be started.
Pyloric Stenosis
Pyloric stenosis is caused by muscle hypertrophy of the pylorus between the stomach and small intestine. It occurs in up to 3.5 of 1,000 live births.24 It is more common in males, preterm neonates, bottle-fed infants, and babies treated with azithromycin/erythromycin.25,26 Children typically present between 3 and 5 weeks of age.15
Clinical Features. The classic presentation of pyloric stenosis is forceful, nonbilious, postprandial emesis with immediate signs of hunger. If the child has been vomiting for a period of time, there may be signs of dehydration, such as decreased wet diapers, decreased skin turgor, and dry mucous membranes. Physical exam may reveal a dehydrated child with an olive-like mass on palpation of the abdomen. Neonates with pyloric stenosis should not have peritonitis, abdominal distension, hematochezia, or bilious emesis.
Diagnostic Imaging and Laboratory Workup. Point-of-care glucose and electrolytes should be obtained, as patients can present with hypoglycemia, hypochloremia, hyponatremia, hypokalemia, and metabolic alkalosis due to vomiting of gastric contents.
A plain radiograph may reveal a distended stomach. (See Figure 6.) Typically, if an experienced ultrasonographer is present, abdominal ultrasound is the preferred imaging modality, with a sensitivity and specificity approaching 100%.27,28 The thickness, length, and diameter of the pylorus can be measured for diagnosis. (See Figure 7.) If ultrasound is not available or if there is suspicion for other etiologies of emesis, such as malrotation or other causes of obstruction, a pediatric radiologist can be consulted to perform an upper GI contrast study.
Figure 6. Distended Stomach in Pyloric Stenosis |
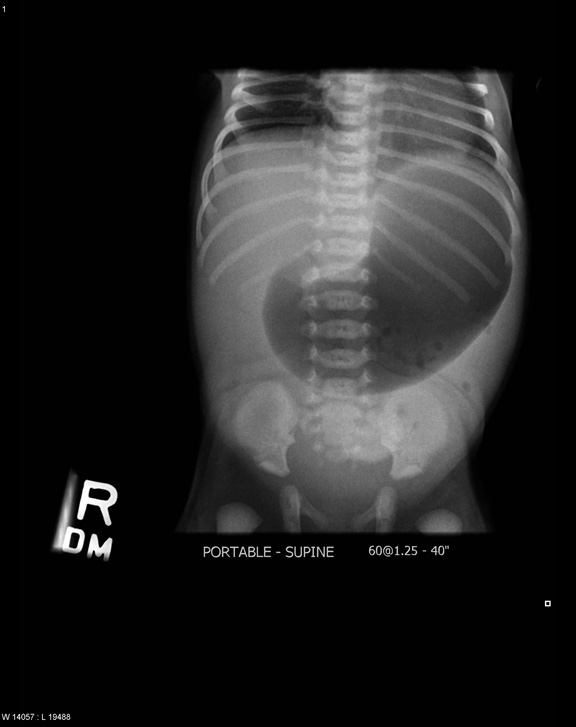 |
Figure 7. Pyloric Stenosis |
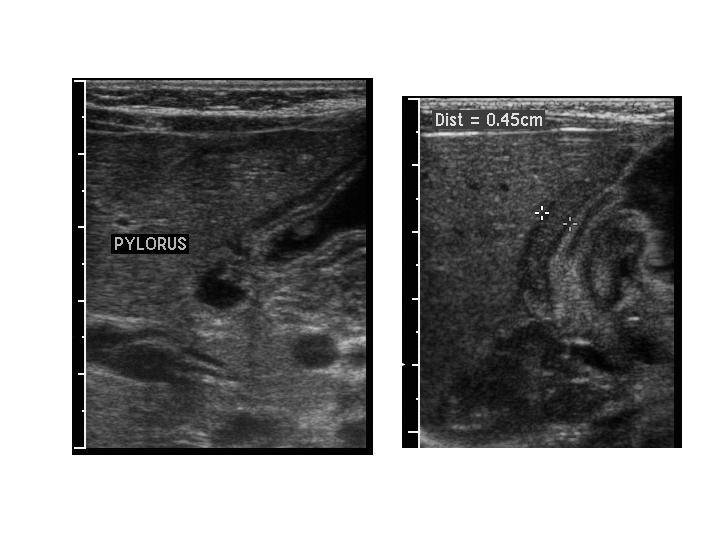 |
ED Management/Disposition. Once the diagnosis is made, patients should have nothing by mouth (NPO). Fluid resuscitation should be initiated and electrolytes should be replenished using isotonic IV fluids, such as normal saline containing glucose and potassium (i.e., 5% dextrose normal saline with 40 meq of potassium). A pediatric surgeon should be consulted. Definitive surgical treatment at this time is laparoscopic pyloromyotomy, which has decreased complications and has a similar success rate compared to open pyloromyotomy.29,30
Aganglionic Megacolon (Hirschsprung’s Disease) and Obstruction
Hirschsprung’s disease (HD) occurs when neural crest cells fail to migrate into the intestinal wall, resulting in a lack of effective peristalsis, leading to obstruction. In the majority of patients, it only affects a short segment of the rectosigmoid colon. HD occurs in approximately 1 in 5,000 live births and typically presents in the neonatal period.31 However, 10% of cases may be diagnosed after 3 years of age.32 Risk factors include male gender, family history, Down syndrome, and other chromosomal abnormalities.31
Patients with HD are at risk of developing obstruction or Hirschsprung’s-associated enterocolitis (HAEC). The incidence of HAEC in patients with HD is approximately 30%, with increasing risk associated with later diagnosis.33
Clinical Features. Classic presentation includes abdominal distension and failure to pass meconium in the first 48 hours of life.34 If meconium is passed, it is scant, and bowel movements typically remain sparse and irregular. A rectal exam may reveal a tight anal sphincter or forceful release of gas/stool known as “squirt sign.” Patients who are toxic-appearing with fever, diarrhea, and abdominal distension may have mechanical obstruction/toxic megacolon or HAEC.35
Diagnostic Imaging and Laboratory Workup. A KUB should be obtained, especially in the toxic-appearing child, to evaluate for signs of obstruction (dilated loops of bowel, airless rectum, cone-shaped transition zone), pneumoperitoneum, and pneumatosis.35
In the nontoxic-appearing child, an unprepped barium enema is generally the preferred initial diagnostic imaging, with sensitivity of 76% and specificity of 97%.36,37 In cases for which this is not diagnostic and high suspicion remains, a suction rectal biopsy is the gold standard for diagnosis, with a sensitivity of 93% and specificity of 100%.37
If needed, laboratory tests should include at least basic chemistry, complete blood count, and liver function tests. If HAEC is suspected, coagulation profile, lactate, blood gas, urinalysis, and blood cultures should be obtained in addition.
ED Management/Disposition. Following initial resuscitation, patients with perforation or HAEC should be started on broad-spectrum IV antibiotics with immediate consult to the pediatric surgeon. Patients with acute obstruction who are toxic-appearing also should receive IV fluids and surgical consult.
Generally, all patients with obstructive features suspected of HD, and especially suspected of HAEC, will be admitted to the hospital or PICU for further management. Inpatient management usually will consist of continued fluid resuscitation, rectal irrigation, ± antibiotics, and diagnostic studies (barium enema, rectal biopsy). In patients with perforation or severe enterocolitis, operative management may be necessary.35
If the child is nontoxic-appearing, not clinically obstructed in the ED, able to tolerate oral hydration, and has reliable follow-up, disposition home with pediatric gastroenterology referral for further workup may be reasonable after consultation with the on-call pediatric surgeon/GI specialist. Once diagnosed, treatment for HD consists of surgical management and removal of the affected colon.38,39
Neonatal Testicular Torsion
Neonatal testicular torsion is an uncommon cause of abdominal pain, with an incidence of approximately six per 100,000 live births, but it does account for 10% of pediatric testicular torsions.40,41 In contrast to testicular torsion in children, neonatal testicular torsion typically is described as extravaginal where the vas, epididymis, fascia, tunica vaginalis, and spermatic cord twist. Intravaginal torsion occurs when the spermatic cord twists within the tunica vaginalis. Further, neonatal testicular torsion can be bilateral and commonly twists from medial to lateral.42
Clinical Features. It is crucial to perform a genitourinary exam in all pediatric patients with apparent abdominal pain. Testicular torsion will present with pain, tenderness, fussiness, and overlying skin changes (erythema, bluish discoloration). Further, the testicle may not transmit light. Lack of cremaster reflex has not been studied in terms of association with neonatal testicular torsion. It has been postulated that an overactive cremaster reflex may be an etiology of neonatal torsion.41
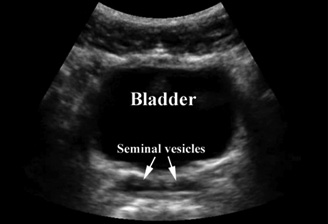
Diagnostic Imaging and Laboratory Workup. Ultrasonography generally is the first-line imaging modality, with sensitivity and specificity approaching 100%.43 (See Figure 8.) Both testicles should be imaged since bilateral torsion can occur.
Figure 8. Testicular Torsion |
|
|
The first Doppler image illustrates compromised vascular flow observed in testicular torsion. The second image of the male pelvis demonstrates the bladder and seminal vesicles in the transverse view. |
|
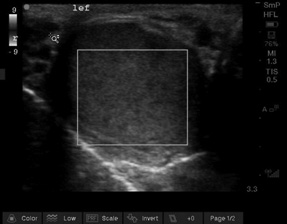 |
 |
|
Image courtesy of Kimberly M. Fender, MD; Daniel B. Park, MD; and Daniel Migliaccio, MD, University of North Carolina, Chapel Hill. |
|
ED Management/Disposition. If there is a high clinical suspicion, pediatric urology should be consulted immediately, since patients can be taken to the operating room without diagnostic imaging. However, the timing and necessity of surgery are controversial without defined guidelines due to poor salvage rates. 44
Common Benign Causes of Abdominal Pain in Neonates
Common benign causes of abdominal pain in neonates include colic, milk protein allergy, and reflux, with prevalences of 8-40%,45 2-3%,46 and 50-60%,47 respectively.
Clinical Features. Patients with these causes of abdominal pain generally will have normal vital signs, oral intake, and wet diapers per day (urine output). The child should be eating normally and growing appropriately. The abdominal exam should be soft without any signs of peritonitis. These are diagnoses of exclusion.
Colic can be distressing to parents. The child typically will seem to cry interminably and inconsolably and can draw back his or her legs in apparent pain. A thorough physical exam should be performed to search for other causes of fussiness. Other sources of pain can include hair tourniquet, corneal abrasion, infection, abdominal catastrophe, nonaccidental trauma, or supraventricular tachycardia. Colic typically starts around the third week of life. Sometimes, it is defined by the “rule of threes,” which include crying for more than three hours per day, more than three days a week, and longer than three weeks. The etiology is not well understood.
Milk protein intolerance may present as a combination of symptoms including rash, wheezing, abdominal discomfort, constipation, and blood-streaked stool.46 The history is key for obtaining the type of formula and diet of the child. Again, it is important to rule out other more serious etiologies, especially if there is blood in the stool. Notably, other benign etiologies of blood in stool include anal fissures and blood from the mother’s irritated nipples.
Reflux typically presents with small volume “spit-up” or nonbloody, nonbilious emesis after feeding.47 If there are signs of failure to thrive, sweating while feeding, or gross blood in stool, other etiologies, such as pyloric stenosis, malrotation, congestive heart failure, supraventricular tachycardia, and obstruction, should be considered.
Diagnostic Imaging and Laboratory Workup. Typically, the diagnosis can be based on the history and physical exam alone without diagnostic imaging or laboratory testing in the ED.
ED Management/Disposition. Patients should be able to tolerate eating and drinking prior to leaving the ED. For colic specifically, observation in the ED to ensure times of calm and oral intake without signs of abdominal pain is reasonable. Treatment of colic consists of nonpharmacologic methods of consoling the child.
Ideally, the specific management of probable milk protein allergy and reflux with medications should be initiated by or with the child’s pediatrician, since he or she will manage subsequent issues. Treatment of milk protein allergy consists of avoiding milk-containing products. Referral to an outpatient primary care pediatrician or nutritionist can aid parents in finding an appropriate formula for their child. Further referral for allergy testing and endoscopy can be considered if symptoms persist.
Treatment strategies of reflux are typically nonpharmacologic and include avoiding milk-containing products, administering smaller feeds, positioning the infant upright after feeding, and burping frequently.47 Pharmacologic therapy, such as histamine type 2 receptor antagonists and proton pump inhibitors, typically are not indicated, with multiple trials failing to show efficacy.48 If symptoms are severe (weight loss, severe irritability), referral to an outpatient pediatric gastroenterologist can be considered for esophagogastroduodenoscopy and initiation of acid suppression.48
Conclusion
Pediatric patients with acute abdominal pain have differential diagnoses that differ from the adult population. Using an age-based differential diagnosis, emergency providers can perform a targeted history, physical, and diagnostic workup to identify the etiology promptly, identify life-threatening illnesses, and diagnose and treat common conditions.
REFERENCES
- Caperell K, Pitetti R, Cross KP. Race and acute abdominal pain in a pediatric emergency department. Pediatrics 2013;131:1098-1106.
- National Hospital Ambulatory Medical Care Survey: 2009 Emergency Department Summary Table. Natl Cent Heal Stat 2012. Available at: https://www.cdc.gov/nchs/data/ahcd/nhamcs_emergency/2009_ed_web_tables.pdf. Accessed April 22, 2019.
- Alpern ER, Stanley RM, Gorelick MH, et al. Epidemiology of a pediatric emergency medicine research network: The PECARN Core Data Project. Pediatr Emerg Care 2006;22:689-699.
- Dieckmann RA, Brownstein D, Gausche-Hill M. The pediatric assessment triangle: A novel approach for the rapid evaluation of children. Pediatr Emerg Care 2010;26: 312-315.
- Miranda A. Abdominal Pain. In: Kliegman RM, Lye PS, Bordini BJ, et al, eds. Nelson Pediatric Symptom-Based Diagnosis. Philadelphia: Elsevier; 2018:161-181.e162.
- Hijaz NM, Friesen CA. Managing acute abdominal pain in pediatric patients: Current perspectives. Pediatric Health Med Ther 2017;8:83-91.
- Amieva-Wang NE, Shandro J, Sohoni A, Fassl B. A Practical Guide to Pediatric Emergency Medicine: Caring for Children in the Emergency Department. Cambridge: Cambridge University Press; 2011.
- Neu J, Walker WA. Necrotizing enterocolitis. N Engl J Med 2011;364:255-264.
- Samuels N, van de Graaf RA, de Jonge RCJ, et al. Risk factors for necrotizing enterocolitis in neonates: A systematic review of prognostic studies. BMC Pediatr 2017;17:105.
- Maayan-Metzger A, Itzchak A, Mazkereth R, Kuint J. Necrotizing enterocolitis in full-term infants: Case-control study and review of the literature. J Perinatol 2004;24:494-499.
- Yee WH, Soraisham AS, Shah VS, et al. Incidence and timing of presentation of necrotizing enterocolitis in preterm infants. Pediatrics 2012;129:e298-304.
- Garbi-Goutel A, Brevaut-Malaty V, Panuel M, et al. Prognostic value of abdominal sonography in necrotizing enterocolitis of premature infants born before 33 weeks gestational age. J Pediatr Surg 2014;49:508-513.
- Robinson JR, Rellinger EJ, Hatch LD, et al. Surgical necrotizing enterocolitis. Semin Perinatol 2017;41:70-79.
- Langer JC. Intestinal rotation abnormalities and midgut volvulus. Surg Clin North Am 2017;97:147-159.
- Aboagye J, Goldstein SD, Salazar JH, et al. Age at presentation of common pediatric surgical conditions: Reexamining dogma. J Pediatr Surg 2014;49:995-999.
- Nehra D, Goldstein AM. Intestinal malrotation: Varied clinical presentation from infancy through adulthood. Surgery 2011;149:386-393.
- Applegate KE. Evidence-based diagnosis of malrotation and volvulus. Pediatr Radiol 2009;39(Suppl 2):S161-163.
- Sizemore AW, Rabbani KZ, Ladd A, Applegate KE. Diagnostic performance of the upper gastrointestinal series in the evaluation of children with clinically suspected malrotation. Pediatr Radiol 2008;38:518-528.
- Long FR, Kramer SS, Markowitz RI, Taylor GE. Radiographic patterns of intestinal malrotation in children. Radiographics 1996;16:547-556; discussion 556-560.
- Ooms N, Matthyssens LE, Draaisma JM, et al. Laparoscopic treatment of intestinal malrotation in children. Eur J Pediatr Surg 2016;26:376-381.
- Hsiao M, Langer JC. Value of laparoscopy in children with a suspected rotation abnormality on imaging. J Pediatr Surg 2011;46:1347-1352.
- Pracros JP, Sann L, Genin G, et al. Ultrasound diagnosis of midgut volvulus: The “whirlpool” sign. Pediatr Radiol 1992;22:18-20.
- Orzech N, Navarro OM, Langer JC. Is ultrasonography a good screening test for intestinal malrotation? J Pediatr Surg 2006;41:1005-1009.
- To T, Wajja A, Wales PW, Langer JC. Population demographic indicators associated with incidence of pyloric stenosis. Arch Pediatr Adolesc Med 2005;159:520-525.
- Eberly MD, Eide MB, Thompson JL, Nylund CM. Azithromycin in early infancy and pyloric stenosis. Pediatrics 2015;135:483-488.
- Zhu J, Zhu T, Lin Z, et al. Perinatal risk factors for infantile hypertrophic pyloric stenosis: A meta-analysis. J Pediatr Surg 2017;52:1389-1397.
- Sivitz AB, Tejani C, Cohen SG. Evaluation of hypertrophic pyloric stenosis by pediatric emergency physician sonography. Acad Emerg Med 2013;20:646-651.
- Niedzielski J, Kobielski A, Sokal J, Krakos M. Accuracy of sonographic criteria in the decision for surgical treatment in infantile hypertrophic pyloric stenosis. Arch Med Sci 2011;7:508-511.
- Kethman WC, Harris AHS, Hawn MT, Wall JK. Trends and surgical outcomes of laparoscopic versus open pyloromyotomy. Surg Endosc 2018;32:3380-3385.
- St Peter SD, Holcomb GW 3rd, Calkins CM, et al. Open versus laparoscopic pyloromyotomy for pyloric stenosis: A prospective, randomized trial. Ann Surg 2006;244:363-370.
- Best KE, Addor MC, Arriola L, et al. Hirschsprung’s disease prevalence in Europe: A register based study. Birth Defects Res A Clin Mol Teratol 2014;100:695-702.
- Constipation Guideline Committee of the North American Society for Pediatric Gastroenterology H, Nutrition. Evaluation and treatment of constipation in infants and children: Recommendations of the North American Society for Pediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr 2006;43:e1-13.
- Menezes M, Puri P. Long-term outcome of patients with enterocolitis complicating Hirschsprung’s disease. Pediatr Surg Int 2006;22:316-318.
- Khan AR, Vujanic GM, Huddart S. The constipated child: How likely is Hirschsprung’s disease? Pediatr Surg Int 2003;19:439-442.
- Gosain A, Frykman PK, Cowles RA, et al. Guidelines for the diagnosis and management of Hirschsprung-associated enterocolitis. Pediatr Surg Int 2017;33:517-521.
- Putnam LR, John SD, Greenfield SA, et al. The utility of the contrast enema in neonates with suspected Hirschsprung disease. J Pediatr Surg 2015;50:963-966.
- De Lorijn F, Reitsma JB, Voskuijl WP, et al. Diagnosis of Hirschsprung’s disease: A prospective, comparative accuracy study of common tests. J Pediatr 2005;146:787-792.
- Coran AG, Teitelbaum DH. Recent advances in the management of Hirschsprung’s disease. Am J Surg 2000;180:382-387.
- Thomson D, Allin B, Long AM, et al. Laparoscopic assistance for primary transanal pull-through in Hirschsprung’s disease: A systematic review and meta-analysis. BMJ Open 2015;5:e006063.
- Kaye JD, Levitt SB, Friedman SC, et al. Neonatal torsion: A 14-year experience and proposed algorithm for management. J Urol 2008;179:2377-2383.
- John CM, Kooner G, Mathew DE, et al. Neonatal testicular torsion—a lost cause? Acta Paediatr 2008;97:502-504.
- Callewaert PR, Van Kerrebroeck P. New insights into perinatal testicular torsion. Eur J Pediatr 2010;169:705-712.
- Kalfa N, Veyrac C, Lopez M, et al. Multicenter assessment of ultrasound of the spermatic cord in children with acute scrotum. J Urol 2007;177:297-301; discussion 301.
- Monteilh C, Calixte R, Burjonrappa S. Controversies in the management of neonatal testicular torsion: A meta-analysis. J Pediatr Surg 2019;54:815-819.
- Stahlberg MR. Infantile colic: Occurrence and risk factors. Eur J Pediatr 1984;143:108-111.
- Rona RJ, Keil T, Summers C, et al. The prevalence of food allergy: A meta-analysis. J Allergy Clin Immunol 2007;120:638-646.
- Nelson SP, Chen EH, Syniar GM, Christoffel KK. One-year follow-up of symptoms of gastroesophageal reflux during infancy. Pediatric Practice Research Group. Pediatrics 1998;102:E67.
- Rosen R, Vandenplas Y, Singendonk M, et al. Pediatric Gastroesophageal Reflux Clinical Practice Guidelines: Joint Recommendations of the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition and the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition. J Pediatr Gastroenterol Nutr 2018;66:516-554.
Abdominal pain is a common pediatric chief complaint with a diversity of etiologies. Many are benign, but some have the potential for devastating consequences if a timely diagnosis is not made. Understanding and practicing a comprehensive approach facilitates consideration of more serious pathology while allowing for a focused diagnostic plan. This two-part series guides the clinician to a practical clinical approach to pediatric abdominal pain.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
