Appropriate Cardiac Testing in an Inappropriate World, Part 2: Cardiac Tests
October 1, 2019
Reprints
AUTHORS
Harvey S. Hahn, MD, FACC, Director, Cardiovascular Fellowship Training Program and Co-Director KPN CV Quality, Kettering Medical Center, Associate Professor of Clinical Medicine, Wright State University/Boonshoft School of Medicine, Kettering, OH, and Loma Linda University, Loma Linda, CA
Erik Poldemann, MD, Cardiology Fellow, Kettering
Medical Center, Kettering, OH
PEER REVIEWER
Jeffrey W. Morgan, DO, MA, FACOI, CS, Dean, School of Osteopathic Medicine in Arizona, Mesa, AZ
EXECUTIVE SUMMARY
This article addresses specifics of the cardiac testing options, considering their appropriate indications, choices of approach, risks, and costs.
• Electrocardiography provides fast, crucial information on several life-threatening conditions, including arrythmias, electrolyte derangements and myocardial ischemia or infarction. It is among the most commonly performed diagnostic procedures in the United States, with sensitivity of detecting an ST-elevation MI low (30-70%) and the specificity in detecting STEMI a respectable 70-100%.
• Coronary artery calcium scoring is a reliable, reproducible, and cost-effective means of measuring a patient’s atherosclerotic plaque burden and aids in predicting risk of major cardiovascular events. Recent data have demonstrated scoring is limited in that age, statin use, and increased exercise levels are all associated with increasing scores but not event rates. A very low score, although portending an excellent prognosis, does not guarantee freedom from coronary artery disease.
• Echocardiography is safe, easily obtainable, and can evaluate the effects of ischemic heart disease, ejection fraction, left ventricular size and filling pressures, and arguably may be the most prognostic test in cardiology.
• A variety of exercise testing, including standard treadmill, exercise myocardial perfusing imaging, stress echo, dobutamine stress echocardiography, pharmacologic PET, and coronary computed tomographic angiography, are discussed with their respective pros and cons.
Part 1 of this two-part series focused on the theory of ordering tests and strategies to minimize unecssary testing. Part 2 looks at when and how to select each individual test given the patient's clinical scenario.
Introduction
In the process of working up a patient with a cardiac complaint, a clinician has a seemingly endless number of testing options. The most appropriate choice depends first and foremost on the patient’s complaint, the history of the present illness, past medical history, body habitus, functional capacity, and costs of the test. (See Figure 1.) It is also important to factor into the decision the associated risks of a test.
Figure 1. Testing Flow Sheet Explained: The Money Diagram64,65 |
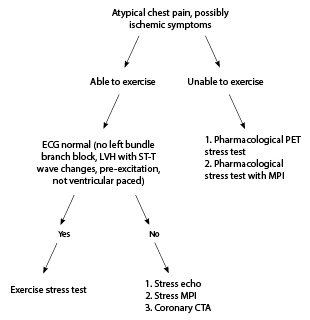 |
|
CTA: computed tomography angiography; ECG: electrocardiogram; LVH: left ventricular hypertrophy; MPI: myocardial perfusion imaging; PET: positron emission tomography |
As a clinician, the first thing to ask once the decision has been made to order a test is, “What is the question I am trying to answer?” Establishing the clinical question to be answered by the test will guide the physician to the ideal test. (See Table 1.) Most of the testing options outlined in this article focus on the evaluation of chest pain and ischemic heart disease.
Table 1. Which Test to Choose |
|
Indication |
Best Test |
|
Asymptomatic |
No testing |
|
Noncardiac chest pain |
No testing |
|
Atypical, possibly ischemic symptoms |
Exercise stress test |
|
Rule out CAD, evaluation of ischemia not needed |
CTA |
|
Low-risk patient, want higher specificity to avoid unnecessary further testing, including invasive angiography |
Stress echocardiography |
|
Higher-risk patient, low threshold to perform cardiac catheterization |
Any nuclear perfusion study |
|
Want to avoid radiation exposure |
Stress echocardiography |
|
CAD: coronary artery disease; CTA: computed tomography angiography |
|
Cardiac Testing Options
There are more costs to a test than just financial strain on the medical system; there is also a financial cost to the patient, as well as costs of time and radiation exposure. (See Table 2.)
Table 2. The Costs of Cardiac Testing |
|||
Test |
Financial Cost |
TimeCost |
Radiation Cost |
|
History and physical exam |
Priceless |
Priceless |
0 |
|
ECG |
$50 |
Minutes |
0 |
|
CT calcium scoring |
$99 |
Minutes |
1 mSv |
|
Echocardiography |
$500-1,000 |
30 minutes |
0 |
|
Exercise stress test |
$175-200 |
30 minutes |
0 |
|
Stress echocardiogram |
$1,500 |
30 minutes |
0 |
|
Stress MPI |
$1,000-3,000 |
~4 hours |
9 mSv |
|
Pharmacologic MPI |
$1,000-3,000 |
~4 hours |
9 mSv |
|
Dobutamine echocardiogram |
$1,500 |
1 hour |
0 |
|
PET |
$3,000 |
1 hour |
1-5 mSv |
|
CCTA, current generation scanners |
$400-900 |
30 minutes |
1 mSv |
|
Cardiac MRI |
$2,500 |
1 hour |
0 |
|
Diagnostic cardiac catheterization |
$2,500 |
1 hour + |
3 mSv + |
|
ECG: electrocardiogram; CCTA: cardiac computed tomography angiography; CT: computed tomography; MPI: myocardial perfusion imaging; MRI: magnetic resonance imaging; mSv: millisievert; PET: positron emission tomography |
|||
The History and Physical Exam
History is still the first cardiac test. A complete history and physical exam are the foundation of an accurate assessment of a cardiac patient. The information gathered from a meticulously executed history and physical exam helps guide a skillful clinician toward the judicious use of diagnostic tests.
A patient’s perception of the quality of the care he or she receives also can be associated with the clinician’s performance of a history and physical exam.1 Eliciting a careful cardiovascular history requires direct communication with the patient; an abundance of information and insight into a condition can be gleaned from direct interaction.1 Time constraints, without doubt, have imposed limits on the clinician’s ability to obtain a careful history and physical exam. The authors believe a proper and focused history and physical examination of the cardiovascular patient can help determine disease severity and prognosis accurately.
The classic article by Diamond and Forrester estimated the probability of coronary artery disease (CAD) based on gender, age, and type of symptoms.2 Using a 50-year-old man as an example, nonanginal chest pain had a disease prevalence of 21.5%; atypical chest pain, 58.9%; and typical angina, 92.0%. Males were more likely to have CAD in all age groups, and prevalence increased with age in both genders.
The exam also is prognostic. Positive findings of murmurs, bruits, and diminished pulses all are important findings to diagnose cardiovascular disease. Blood pressure, heart rate, and fluid status are therapeutic targets to normalize with medical therapy. Abnormal findings on exam, especially those related to heart failure, are extremely prognostic. Killip and Kimball initially described their findings in a post-infarct population in 1967. The researchers showed that with more findings of heart failure (Killip class I, no heart failure findings; Killip class II, S3 gallop or bibasilar rales; Killip class III, pulmonary edema; or Killip class IV, shock), mortality increased in a stepwise fashion.3
Patients with no signs of heart failure had a 6% mortality, although patients with pulmonary edema had a mortality of 38% and those with shock had a mortality of 81%. A follow-up study looked at all-cause mortality in four randomized clinical trials based on the original Killip classification and found that this pattern persisted in modern medicine.4
Electrocardiogram
The practice of clinical electrocardiography - and clinical correlation of the findings from this methodology - has evolved over more than a century.5 Although there have been numerous advances in cardiac imaging in recent years, the electrocardiogram (ECG) still provides crucial information in just minutes regarding the electrical properties of the heart in several life-threatening conditions, including bradyarrhythmias and tachyarrhythmias, electrolyte derangements, and, most of all, myocardial ischemia or infarction.5
Although electrocardiography is an age-old technology, it is still among the most commonly performed diagnostic tests in the United States.6 The sensitivity of a 12-lead ECG in detecting an ST-elevation myocardial infarction (STEMI) is low, and ranges from 30-70%. However, the specificity of ECG in detecting STEMI is more respectable and approaches 70-100%.7 According to the American Board of Internal Medicine and American Academy of Family Physicians, the average cost of an ECG is around $50.8 At the individual patient level, the cost of an ECG may seem a small price to pay to detect cardiac disease; however, this does not take into account the hundreds, or even thousands, of dollars downstream from resulting testing and referrals.
Limited attention has been paid to the appropriate clinical indications for an ECG, likely because of its ease of use, simplicity, and low cost.5 There is value in having a “baseline” ECG, mostly for comparison and to look for acute ischemic changes. It is almost impossible to argue against a routine, baseline ECG.
Risk Scoring: Combined Clinical, ECG, and Biomarker Assessment
After the history and physical exam, there are other ways to assess a patient's risk before ordering more advanced cardiac testing. The most common practice is to combine the clinical assessment with the ECG and biomarkers to stratify patients into different risk categories. This has been done most commonly in the emergency department with point-of-care troponin assays.
The ADAPT trialists looked to an accelerated two-hour diagnostic protocol to assess patients with acute chest pain. Using the Thrombolysis in Myocardial Infarction (TIMI) score, ECG, and two troponin values, the researchers identified 20% of their study population that was low risk and did not need admission or further testing. Of that group, only a single patient had a major adverse cardiac event (MACE), giving the protocol a negative predictive value (NPV) of 99.7%.9
A larger study again used clinical assessment with ECG and troponin evaluation. This study of three community teaching hospitals involved more than 45,000 patients. Of those, 11,230 had two normal troponin values. There were only 20 events for a MACE rate of 0.18%.10 If patients with abnormal vital signs or an abnormal ECG were excluded, there were four events, for a MACE rate of 0.06%. Of those four events, two were non-cardiac and possibly iatrogenic - again making the case for less is more.
One of the most common risk scores is the history, ECG, age, risk factors, and troponin (HEART) score. A recent meta-analysis of the HEART score demonstrated an NPV of 96%.11 Stopyra et al looked at not only MACE rates, but further healthcare utilization after using the HEART Pathway - an electronic health record clinical decision support tool - scoring compared to usual care.13 They found that MACE was not different at one year, but that downstream, cardiac testing was actually higher in the group that used the HEART Pathway.12 Initially, there was less testing in the HEART Pathway group, but by one year there was more testing overall in that group.
The study by Stopyra et al was a small randomized controlled trial of 282 patients.13 A much larger, cohort-designed study of more than 8,400 patients looked at MACE, admissions, and downstream testing for one year before implementing the HEART Pathway, and then for one year after its implementation. In this study, the researchers identified 30.7% of patients as low risk. MACE and death rates were unchanged in the two time periods. There was a 6% decrease in admission for chest pain; early discharges increased from 37% to 43%; and stress testing and angiography decreased a small but significant relative 11%.14
Before selecting a specific cardiac test, the final question to be asked is, “Does this patient even need a test?” A mentor of one of the authors used to ask, “Do you need to know, or is it neat to know?” The question had to be answered before the mentor would agree to a testing strategy. Physicians need to ask the patient if he or she needs to know. As discussed earlier, many patients do not need more reassurance than their doctor’s opinion, which they trust. The National Academy of Medicine’s 2017 report stressed engaging the patient and family in shared decision-making.15 Also, one of the three pillars of evidence-based medicine is “patient values and expectations.”16 Physicians cannot know these unless they talk and listen to their patients. Not only are many tests inappropriate, but so is the old paternalistic model of medicine.
CT Calcium Scoring or CAC Scanning
Coronary artery calcium (CAC) scoring is a reliable, reproducible, cost-effective means of measuring a patient’s atherosclerotic plaque burden and aids in predicting an individual’s risk of a variety of major cardiovascular events.17 CAC scoring, or the quantification of coronary artery calcium, is a non-contrast-enhanced image acquisition technique obtained during a single breath-hold. The scan is obtained in about 10 minutes, is gated by the ECG, and does not require the administration of a beta-blocker to lower the heart rate.18 Computed tomographic (CT) scanners for CAC scoring are readily available and expose a patient to about 1 mSv of radiation without the need for contrast.
Recent studies using coronary angiography, intracoronary ultrasound, and histological studies have demonstrated that CAC quantification by scores correlates well with atherosclerotic plaque burden, but is not associated with the luminal area of a vessel.19 Initial studies showed that progression of CAC was associated with advancement of CAD.20 However, more recent data have demonstrated that CAC scoring is limited in that age,21 statin use,22 and increased exercise level23 all are associated with increasing CAC scores, but not event rates. This limits the use of CAC to monitor disease progression or status serially.
Several methods have been suggested for implementing a CAC in guiding primary prevention strategies. Use of CAC scoring, in a modified approach combined with the atherosclerotic cardiovascular disease (ASCVD) risk estimate, can reclassify a patient’s risk and potentially could influence a significant clinical decision, such as starting statin therapy.24
Although a very low CAC score, or even a score of zero, portends an excellent prognosis, it does not guarantee freedom from CAD. In an interesting study, researchers examined only patients with a CAC of zero and found that 16% had reversible defects consistent with ischemia on PET scanning.25 This is most likely a result of the fact that many coronary lesions, especially the more vulnerable ones, are soft plaques and not calcified.
Pros
- Low cost
- Low radiation dose
- Quick and easy
Cons
- For the same radiation dose, can obtain a CTA of the coronaries, which provides much more information
- Should not be used for monitoring (i.e., no serial studies)
Echocardiography
Echocardiography is the most commonly used cardiac imaging modality to assess the structure and function of the heart.26 The benefit of transthoracic echocardiography (TTE) is that it can be performed relatively quickly, can be transported and done at the bedside, and can be done with little to no risk to the patient. Although TTE is painless, it can lead to further testing downstream and should be ordered rarely in a patient with no symptoms or signs of cardiovascular disease.27 It is rarely appropriate to obtain a transthoracic echocardiogram as a part of “routine surveillance” of left ventricular function with no change in the clinical status of the patient and if the patient has had a TTE within the last year.27
Although TTE is relatively safe and is easily obtainable, in most cases its use should be limited to patients with a change to their clinical status or cardiac exam. A TTE may lead to further unnecessary consultations and to more invasive procedures, which can increase the financial cost to the patient further and can expose him or her to a number of procedural-related complications.28
Echocardiogram is arguably the most prognostic test in cardiology since it provides measurement of several variables that have been independently and reproducibly shown to predict mortality. These include ejection fraction (EF), left ventricular size, right ventricular function, filling pressures (by the E/e’ estimate), Doppler patterns, and pulmonary artery pressures.
Although it has many advantages, TTE can look only at the effects of ischemic heart disease, such as an old myocardial infarction. It cannot detect ischemia unless added to a stress test, which will be discussed later.
Pros
- No radiation
- Quick, easy
- Information on cardiac structure and function/physiology
- Hemodynamic assessment with Doppler has basically supplanted invasive hemodynamic studies in the catheterization laboratory
Cons
- Quality, and thus diagnostic accuracy, limited by body habitus, which is becoming a greater issue with increase in obesity
Stress Testing
It is helpful to think of cardiac stress testing as a combination of two components: a stressing agent (i.e., treadmill, bicycle, or pharmacological) and an imaging agent (ECG, echocardiography, or radionucleotide tracer).29 Determining the ideal combination of stressing agent and imaging agent establishes the appropriateness of the test and is a large part of the art of medicine.
Exercise stress testing or exercise electrocardiography is an important diagnostic tool in the evaluation and management of patients with known or suspected CAD. Myocardial ischemia occurs when the supply of oxygenated blood to myocardial cells is inadequate to meet the delicate balance of supply and demand.
Exercise electrocardiography capitalizes on this principle by stressing this relationship in a controlled environment, where the physiological response of the patient is closely observed.30 Exercise stress testing is relatively safe and effective for detecting significant CAD. According to the American College of Cardiology, exercise electrocardiography has a sensitivity of 68% and specificity of 77% for detecting CAD.31 Exercise stress testing is appropriate in the initial evaluation of patients with suspected or known CAD who present with chest pain and who have an intermediate pre-test probability for CAD.
Prior to ordering an exercise electrocardiography test, a few questions must be asked. First, is the patient physically able to exercise and will the person's exercise capacity allow him or her to achieve the target of 85% of the maximum predicted heart rate? Second, does the patient’s baseline ECG have abnormalities that may interfere with the interpretation of an ECG stress test? These abnormalities include ventricular pre-excitation, ventricular paced rhythms, left bundle branch block, digoxin use with ST-T wave changes, and significant left ventricular hypertrophy with secondary ST-T wave changes. These baseline ECG findings typically preclude a patient from undergoing plain exercise electrocardiographic testing.
Exercise should be the preferred stress modality when it is obvious that the patient has the ability to exercise - or even when it is uncertain. If the target heart rate or metabolic equivalent of task (MET) level is not achieved, then vasodilator stress MPI can be performed.32 Even if the target heart rate is not reached, important physiologic and symptomatic information still can be gleaned: Did the patient develop chest pain? Does the person have a hypertensive response to exercise? Does the patient have chronotropic (heart rate) reserve?
Clinicians should note that most exercise stress tests in patients without known coronary artery disease are performed best by holding cardioactive medications on the day of the stress test (i.e., beta-blockers and calcium channel blockers) to allow for a better detection of the ischemia.30
Exercise stress testing evaluates a patient’s symptoms, exercise capacity, and ECG changes under physiological stress. The ability to produce symptoms is the most important diagnostic portion of the test. Exercise capacity is quantified by METs or exercise time. One MET is defined as the energy expended by sitting quietly, or body oxygen consumption of 3.5 mL/kg of body weight per minute for the average adult.33
Exercise capacity alone is a powerful predictor of cardiovascular events.34 In one study of 6,213 men, some healthy and some with cardiovascular disease, the peak exercise capacity achieved was found to be a stronger predictor of an increased risk of death than clinical variables or established risk factors like hypertension, smoking, and diabetes mellitus.33
Patients in this group whose exercise capacity was < 5 METs had nearly twice the risk of death from any cause as patients whose exercise capacity was > 8 METs.33
The researchers also suggested that for every 1 MET increase in treadmill performance, there was a 12% improvement in overall survival.33 Because poor exercise capacity is a modifiable risk factor, studies also have suggested that exercise capacity improvements over time translate to improved overall prognosis. In another study of nearly 10,000 men, researchers observed a nearly 8% reduction in mortality for every one-minute increase in the time the subject spent on the treadmill.35 Several studies have suggested that patients at intermediate risk for CAD or who have known CAD who achieve ≥ 10 METs on exercise stress ECG testing have a very low prevalence of cardiac mortality, nonfatal myocardial infarction, and late revascularization, regardless of the heart rate they achieve during the test.36 METs are also the most important component of the commonly used Duke treadmill score.37
Pros
- Relatively cheap, easy
- No radiation
- Obtain functional assessment, assess effectiveness of medical therapy, evaluate chronotropic competence
Cons
- Only a 50% predictive value in women or patients with an abnormal baseline ECG
- Many patients now can barely exercise, rendering many tests non-diagnostic
Exercise Myocardial Perfusion Imaging
Before imaging technology allowed for the assessment of myocardial perfusion and function, symptom-limited exercise ECG stress testing was the lone noninvasive method of detecting significant CAD.32 To increase the sensitivity and specificity of a treadmill stress test and to quantify or localize areas of ischemic myocardium, clinicians now can add myocardial perfusion imaging (MPI) to an exercise stress test. The added benefit of exercise MPI is that it provides an assessment of wall motion and ejection fraction as well. Numerous studies have demonstrated that the combination of MPI with symptom-limited exercise enhances the diagnostic and prognostic value of exercise stress testing beyond the measurements obtained from exercise stress ECG testing.32 In one study, the sensitivity of MPI for detecting a coronary artery stenosis ≥ 70% was 86% in male diabetic patients compared with 88% in male nondiabetic patients. The sensitivity of MPI for detecting a coronary artery stenosis ≥ 70% was 93% in female diabetic patients and 86% in nondiabetic patients,38 although the specificity of MPI for detecting significant coronary artery stenosis ranged from 55-64%.38
The major disadvantages of stress MPI using single photon emission computed tomography (SPECT) is the time required to obtain the resting and stress images (up to four hours to obtain both sets of pictures) and radiation exposure to the patient. The average radiation exposure to the patient ranges from 10 to 20 mSv39; for comparison, the average dose of radiation from diagnostic angiography is about 3-4 mSv. MPI also is prone to attenuation artifacts from either breast tissue or the diaphragm, which can lead to equivocal or false-positive findings.
Pros
- Increases the sensitivity and specificity of exercise testing
- Adds EF and wall motion scoring
Cons
- Radiation exposure
- Total test time is about four hours
- About 40% of all scans will have soft tissue attenuation, which will need to be considered
- Women have breast attenuation, and men have diaphragmatic attenuation
Pharmacologic MPI
For patients who are unable to achieve ≥ 85% of their maximum predicted heart rate for age or who have physical limitations precluding them from performing exercise stress testing, pharmacological stress testing is a reasonable option. Adenosine, dipyridamole, and regadenoson are the most commonly used pharmacologic stress agents. These are excellent vasodilator stressing agents because they produce coronary vasodilation and increase myocardial blood flow three to five times that of resting myocardial blood flow.40 When a significant coronary artery stenosis is present and there is flow-limited obstruction, there is a relative difference in radiotracer uptake in the area supplied by the stenotic coronary artery, which appears as a perfusion defect when compared with the resting myocardium.41 The choice of a pharmacological stress agent depends on multiple factors; regadenoson is the primary vasodilator agent used for pharmacological stress MPI because of its ease of use and better tolerability by patients.42 Overall, the sensitivity and specificity of pharmacological stress MPI for detecting significant obstructive CAD ranges from 80-90%.43
Dobutamine, which increases the heart rate and the contractility of the heart, also can be used as a stressing agent for MPI. This increase in heart rate and contractility, in turn, increases the myocardial oxygen demand to a level that is comparable to exercise.41 Dobutamine is generally well-tolerated and safe as a stressing agent for MPI. The target for dobutamine MPI is the 85% age-predicted maximum heart rate. Once this target heart rate is achieved, the radionucleotide is injected over a 10-second period and allowed to circulate for 60 seconds or more.44
Even though pharmacologic agents represent an excellent alternative to exercise stress testing, the inability to exercise is a poor prognostic sign. In a large series of patients, regardless of EF or perfusion results, the one-year mortality was four times higher in the pharmacologic group than in the exercise group.45 This most likely reflects comorbid conditions that affect not only cardiovascular health, but overall health, such as COPD, amputations due to peripheral artery disease, or severe disability.
Pros
- Increases the sensitivity and specificity of exercise testing
- Adds EF and wall motion scoring
Cons
- Radiation exposure
- Total test time is about four hours
- About 40% of all scans will have soft tissue attenuation, which will need to be considered; women have breast attenuation, and men have diaphragmatic attenuation
- With pharmacologic testing, there is minimal physiologic data gathered from the study
Exercise Stress Echocardiogram
Stress echocardiography is another well-validated method of evaluating for ischemic heart disease. Like other stress tests, it is used best in patients with an intermediate pretest probability for CAD. The advantages of echocardiography are that it is readily available, generally inexpensive, and images are acquired without any radiation exposure to the patient. Additionally, stress echocardiography provides an assessment of wall motion, EF, and valvular and diastolic function. Images are acquired at rest and then immediately after peak stress. The hallmark of myocardial ischemia during stress echocardiography is the presence of reduced systolic wall thickening when myocardial oxygen demand exceeds myocardial blood supply.46 The advantage is that these changes to myocardial contraction are detectable on echocardiography prior to the patient experiencing chest pain or the appearance of ST-T wave changes on the ECG.46 Another advantage of stress echocardiography is the relatively short duration of the test; on average, most stress echocardiographic studies can be obtained and interpreted within one hour, potentially increasing the efficiency with which a clinician can decide patient disposition.
Stress echocardiography also is an appropriate test in patients who have abnormalities in their ECG at baseline that preclude interpretation of exercise ECGs.26 Although the sensitivity of stress echocardiography is slightly less than that of stress MPI studies for detection of single-vessel disease, its overall specificity is higher on average.47 The sensitivity of stress echocardiography to detect significant CAD (> 50% coronary artery stenosis by angiography) averages 88%, although the sensitivity is around 83%.26 Image quality during stress echocardiography is greatly affected by a patient’s body habitus; therefore, stress echocardiography may be difficult in obese individuals with poor acoustic windows and may need to be performed with contrast. The quality of the test also greatly depends on local expertise and the experience of the operator obtaining the images.48
Stress echocardiography also has excellent prognostic value, and a normal stress echocardiogram translates to a good prognosis.48 In one study of 1,325 patients who were followed after a normal stress echocardiography study, researchers found that one- and three-year cardiac event-free survival rates were 99% and 97%.49 This suggests that a normal exercise echocardiography study confers a favorable prognosis.
Pros
- Cheap, easy, no radiation
- Obtain functional assessment, assess effectiveness of medical therapy, evaluate chronotropic competence
- Can obtain rest and stress EF, look for contractile reserve, measure changes in pulmonary artery pressure, valve function, or filling pressures pre- and post-exercise, which can greatly aid in discovering if a cardiac cause is leading to symptoms
Cons
- Limited by body habitus, which is becoming a greater issue with increase in obesity; many patients now can barely exercise, rendering many tests non-diagnostic
Dobutamine Stress Echocardiography
When performing stress echocardiography in patients who are unable to exercise, pharmacological agents, including dobutamine and dipyridamole, can be employed. Dobutamine is administered in a graded infusion to achieve the target heart rate. The advantage of pharmacological stress echocardiography is that echocardiographic imaging can be obtained at the exact time of peak stress.78 The procedure itself is very safe, and the risks associated with dobutamine stress echocardiography are relatively low. For dobutamine stress echocardiography, the overall rate of life-threatening events is one per 1,000 tests, or 0.18%.50 This testing modality also is used frequently to assess low-flow aortic stenosis and to evaluate for myocardial viability.
Pros
- No radiation
- Obtain functional assessment, assess effectiveness of medical therapy, evaluate chronotropic competence
- Can obtain rest and stress EF, look for contractile reserve, measure changes in pulmonary artery pressure, valve function, or filling pressures pre- and post-exercise, which can aid in discovering if a cardiac cause is leading to symptoms
Cons
- More complicated because of the need to set up a dobutamine infusion
- Cannot do test on patients who have had beta-blockers
- Limited by body habitus, which is becoming a greater issue with increase in obesity
Pharmacologic PET
Positron emission tomography (PET) is a powerful imaging modality that is used to evaluate myocardial perfusion noninvasively.51 PET stress tests have numerous advantages over SPECT stress imaging. PET stress tests provide higher-resolution images; expose the patient to less radiation than a SPECT scan; and require only 30 minutes to perform. The major disadvantage of PET MPI is the inability to perform the test with exercise as the stressing agent, because of the short half-life of the radioisotopes used. The sensitivity of PET perfusion imaging for detecting angiographic stenosis of ≥ 50% is 91% and specificity is 89%.52 The diagnostic accuracy of PET is also significantly higher than SPECT MPI in all populations, including obese patients, estimated to be 89% vs. 79%.53 Overall, the diagnostic accuracy of MPI with PET imaging is better than SPECT MPI. This can be attributed to PET’s superior temporal resolution and ability to correct for attenuation artifact. Also, several studies in recent years have verified that a normal PET stress test has excellent prognostic value.54,55 Despite its increased cost, PET is emerging as an excellent option for MPI because of its high diagnostic accuracy, ability to correct for soft tissue attenuation artifact, and ability to provide prognostic information.
Pros
- Better resolution than SPECT
- Obtain EF and wall motion
- Better resolution, less radiation, and shorter testing time than SPECT
Cons
- More expensive than SPECT
- Limited availability of PET scanners and isotope generators
- Majority of protocols cannot support exercise studies
Coronary Computed Tomographic Angiography
Coronary computed tomographic angiography (CCTA) is also known as multidetector CT (MDCT), multislice CT (MSCT), 3D CT, and dual source CT. CCTA provides a noninvasive anatomical assessment of the coronaries, which allows clinicians to quantify CAD accurately.56 CCTA images are acquired quickly (in a single breath hold) and can provide information such as EF, wall motion, and images of the aorta to evaluate for dissection. For detecting significant coronary artery plaques, CCTA has a reported sensitivity of 90%
and a specificity of 95%.57
The major limitations of CCTA are radiation exposure, the need for contrast, and the necessity to achieve a slow, regular heart rate.57 Current generation CT scanners expose the patient to much lower radiation doses than previous scanners; 320 detector and dual source scanners typically deliver 1 mSv of radiation to the patient,58 which is the same dose as a CAC score. Slower heart rates improve image quality, and therefore beta-blockers may need to be used in patients with heart rates above 65 beats per minute.59
Many recent studies have verified the prognostic utility of CCTA. A study of 1,559 patients who underwent CCTA found that a negative CCTA had a NPV of 99.3% in excluding major adverse cardiac events for 30 days after the initial presentation.60 Similarly, disease burden obtained by CCTA has similar prognostic value as that of invasive catheterization.61 Furthermore, CCTA not only shows the lumen, but also shows the whole vessel and lesion composition. Low attenuation plaque (soft plaque) is mostly lipid and more likely to rupture and lead to acute coronary syndromes.62 This is in direct contrast to what CAC measures - hard, calcified plaque, which is much more stable. CCTA can predict by their appearance high-risk lesions more likely to rupture. Lesions that exhibit positive remodeling (remember the Glagov phenomena mentioned in Part 1), disrupted plaque, ulcerations or intraplaque dye penetration,62 or the “napkin ring” sign63 all are at high-risk of rupturing and causing ischemic symptoms.
Recently, the Prospective Multicenter Imaging Study for Evaluation of Chest Pain (PROMISE) trial examined the initial strategy of CCTA vs. functional stress testing in stable diabetic patients with symptoms suggestive of CAD. The PROMISE trial found that in diabetic patients with stable chest pain, a CCTA strategy resulted in fewer adverse cardiovascular outcomes than an initial functional stress testing strategy.64
Pros
- Fast, easy, cheap, low radiation exposure
- Get a “free” aortogram in this study to evaluate for aortic dissection also
Cons
- Dye load, so renal dysfunction limits its application
- Unstable and high heart rates make gating more difficult and reduce the quality and diagnostic ability of the study
Summary
To choose the best and most appropriate test for the patient, keep these points in mind:
- Less is more.
- Do not order a test that you will not do anything with.
- Do not order preoperative tests that will only delay needed surgery.
- Do not order “routine” anything.
- Do not order high-tech tests on low-risk patients.
- Do not try to reassure the patient or yourself.
- Be honest about personal conflict of interest (COI).
REFERENCES
- Zipes D, Libby P, Bonow R, et al. The medical history and physical examination. In: Braunwald’s Heart Disease. 11th Edition. Philadelphia: Elsevier; 2019:404-405
- Diamond GA, Forrester JS. Analysis of probability as an aid in the clinical diagnosis of coronary-artery disease. N Engl J Med 1979;300:1350-1358.
- Killip T, Kimball JT. Treatment of myocardial infarction in a coronary care unit. A two year experience with 250 patients. Am J Cardiol 1967;20:457-464.
- Khot UN, Khot MB, Bajzer CT, et al. Prevalence of conventional risk factors in patients with coronary heart disease. JAMA 2003;290:898-904.
- Zipes D, Libby P, Bonow R, et al. Electrocardiogram. In: Braunwald’s Heart Disease. 11th Edition. Philadelphia: Elsevier; 2019:649-651.
- U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. National Ambulatory Medical Care Survey: 2010 Summary Tables. Available at: https://www.cdc.gov/nchs/data/ahcd/namcs_summary/2010_namcs_web_tables.pdf. Published 2010. Accessed June 1, 2019.
- Garvey JL, Zegre-Hemsey J, Gregg R, et al. Electrocardiographic diagnosis of ST segment elevation myocardial infarction: An evaluation of three automated interpretation algorithms. J Electrocardiol 2016;49:728-732.
- American Academy of Family Physicians. EKGs and exercise stress tests. Philadelphia, PA: Choosing Wisely;2016. Available at: http://www.choosingwisely.org/wp-content/uploads/2018/02/EKGs-And-Exercise-Stress-Tests-AAFP.pdf. Accessed June 1, 2019.
- Than M, Cullen L, Aldous S, et al. 2-Hour accelerated diagnostic protocol to assess patients with chest pain symptoms using contemporary troponins as the only biomarker. The ADAPT trial. J Am Coll Cardiol 2012;59:2091-2098.
- Weinstock MB, Weingart S, Orth F, et al. Risk for clinically relevant adverse cardiac events in patients with chest pain at hospital admission. JAMA Intern Med 2015;175:1207-1212.
- Fernando SM, Tran A, Cheng W, et al. Prognostic accuracy of the HEART score for prediction of major adverse cardiac events in patients presenting with chest pain: A systematic review and meta-analysis. Acad Emerg Med 2019;26:140-151.
- Mark DG, Huang J, Chettipally U, et al. Performance of coronary risk scores among patients with chest pain in the emergency department. J Am Coll Cardiol 2018;31:606-616.
- Stopyra JP, Riley RF, Hiestand BC, et al. The HEART Pathway randomized controlled trial one-year outcomes. Acad Emerg Med 2019;26:41-50.
- Mahler SA, Lenoir KM, Wells BJ, et al. Safely identifying emergency department patients with acute chest pain for early discharge. HEART Pathway accelerated diagnostic protocol. Circulation 2018;138:2456-2468.
- Frampton SB, Guastello S, Hoy L, et al. Harnessing evidence and experience to change culture: A guiding framework for patient and family engaged care. NAM Perspectives 2017;1:1-38.
- Sackett DL, Rosenberg WM, Gray JA, et al. Evidence based medicine: What it is and what it isn’t. BMJ 1996;312:71.
- Blaha MJ, Mortensen M, Kianoush S, et al. Coronary artery calcium scoring: Is it time for a change in methodology? JACC Cardiovasc Imaging 2017;10:923-937.
- Zipes D, Libby P, Bonow R, et al. Coronary artery calcium scoring. In: Braunwald’s Heart Disease. 11th Edition. Philadelphia: Elsevier; 2019:324-328.
- Baumgart D, Schmermund A, Goerge G, et al. Comparison of electron beam computed tomography with intracoronary ultrasound and coronary angiography for detection of coronary atherosclerosis. J Am Coll Cardiol 1997;30:57-64.
- Schmermund A, Baumgart D, Mohlenkamp S, et al. Natural history and topographic pattern of progression of coronary calcification in symptomatic patients. Arterioscler Thromb Vasc Biol 2001;21:421-426.
- McClelland RL, Chung H, Detrano R, et al. Distribution of coronary artery calcium by race, gender, and age. Results from the Multi-Ethnic Study of Atherosclerosis (MESA). Circulation 2006;113:30-37.
- Puri R, Nicholls SJ, Shao M, et al. Impact of statins on serial coronary calcification during atheroma progression and regression. J Am Coll Cardiol 2015;65:1273-1282.
- DeFina LF, Radford NB, Barlow CE, et al. Association of all-cause and cardiovascular mortality with high levels of physical activity and concurrent coronary artery calcification. JAMA Cardiol 2019;4:174-181.
- Greenland P, Blaha MJ, Budoff MJ, et al. Coronary calcium score and cardiovascular risk. J Am Coll Cardiol 2018;71:434-447.
- Schenker MP, Dorbala S, Hong EC, et al. Interrelation of coronary calcification, myocardial ischemia, and outcomes in patients with intermediate likelihood of coronary artery disease: A combined positron emission tomography/computed tomography study. Circulation 2008;117:1693-1700.
- Zipes D, Libby P, Bonow R, et al. Echocardiography. In: Braunwald’s Heart Disease. 11th Edition. Philadelphia: Elsevier; 2019:174-244.
- Bhatia RS, Dudzinski DM, Malhotra R, et al. Educational intervention to reduce outpatient inappropriate echocardiograms: A randomized control trial. JACC Cardiovasc Imaging 2014;7:857-866.
- American College of Cardiology. Echocardiograms for heart valve disease. Washington, DC: Choosing Wisely; 2012. Available at: http://www.choosingwisely.org/patient-resources/echocardiograms-for-heart-valve-disease/. Accessed June 1, 2019.
- Balady GJ, Arena R, Sietsema K, et al. Clinician’s guide to cardiopulmonary exercise testing in adults: A scientific statement from the American Heart Association. Circulation 2010;122:191-225.
- Zipes D, Libby P, Bonow R, et al. Exercise testing in coronary artery disease. In: Braunwald’s Heart Disease. 11th Edition. Philadelphia: Elsevier; 2019:159-163.
- Bourque JM, Beller GA. Exercise stress ECG without imaging. JACC Cardiovasc Imaging 2015;8:1309-1321.
- Gonzalez J, Beller G. Choosing exercise or pharmacologic stress imaging, or exercise ECG testing alone: How to decide. J Nucl Cardiol 2017;24:555-557.
- Myers J, Prakash M, Froelicher V. Exercise capacity and mortality among men referred for exercise testing. N Engl J Med 2002;346:793-801.
- Bourque JM, Holland BH, Watson DD, et al. Achieving an exercise workload of ≥ 10 METS predicts a very low risk of inducible ischemia: Does myocardial perfusion imaging have a role? J Am Coll Cardiol 2009;54:538-545.
- Blair SN, Kohl HW, Barlow CE, et al. Changes in physical fitness and all-cause mortality. A prospective study of healthy and unhealthy men. JAMA 1995;273:1093-1098.
- Bourque JM, Charlton GT, Holland BH, et al. Prognosis in patients achieving ≥ 10 METS on exercise stress testing: Was SPECT imaging useful? J Nucl Cardiol 2011;18:230-237.
- Shaw LJ, Peterson ED, Shaw LK, et al. Use of a prognostic treadmill score in identifying diagnostic coronary disease subgroups. Circulation 1998;98:1622-1630.
- Kumar D, Sethi RS, Bansal S, et al. Diagnostic accuracy of stress myocardial perfusion imaging in Indian diabetic patients: A single centre experience. Indian J Nucl Med 2017;32:177-183.
- Gupta A, Bajaj N. Reducing radiation exposure from nuclear myocardial perfusion imaging: Time to act is now. J Nucl Cardiol 2017;24:1856-1859.
- Chan SY, Brunken RC, Czernin J, et al. Comparison of maximal myocardial blood flow during adenosine infusion with that of intravenous dipyridamole in normal men. J Am Coll Cardiol 1992;20:979-985.
- Chareonthaitawee P, Aske JW. Overview of stress radionuclide myocardial perfusion imaging. UpToDate. Available at: https://www.uptodate.com/contents/overview-of-stress-radionuclide-myocardial-perfusion-imaging?search=overview-of-stress-radionuclide-myocardial-perfusion-imaging)&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1. Accessed June 15, 2019.
- Rossen JD, Quillen JE, Lopez AG, et al. Comparison of coronary vasodilation with intravenous dipyridamole and adenosine. J Am Coll Cardiol 1991;18:485-491.
- Iskandrian AE, Bateman TM, Belardinelli L, et al. Adenosine versus regadenoson comparative evaluation in myocardial perfusion imaging: Results of the ADVANCE phase 3 multicenter international trial. J Nucl Cardiol 2007;14:654-658.
- Henzlova MJ, Cerqueira MD, Mahmarian JJ, et al. Stress protocols and tracers. J Nucl Cardiol 2007;13:e80-90.
- Sharir T, Germano G, Kavanagh PB, et al. Incremental prognostic value of post-stress left ventricular ejection fraction and volume by gated myocardial perfusion single photon emission computed tomography. Circulation 1999;100:1035-1042.
- Senior R, Monaghan M, Becher H, et al. Stress echocardiography for the diagnosis and risk stratification of patients with suspected or known coronary artery disease: A critical appraisal. Supported by the British Society of Echocardiography. Heart 2005;91:427-436.
- Gottdiener JS. Overview of stress echocardiography: Uses, advantages, and limitations. Prog Cardiovasc Dis 2001;43:315-334.
- Al-Shehri H, Small G, Chow B, et al. Cardiac CT, MR, SPECT, ECHO, and PET: What test, when? Appl Radiol 2011;40:13-22.
- McCully RB, Roger VL, Mahoney DW, et al. Outcome after normal exercise echocardiography and predictors of subsequent cardiac events: Follow-up of 1,325 patients. J Am Coll Cardiol 1998;31:144-149.
- Varga A, Garcia MA, Picano E. Safety of stress echocardiography (from the International Stress Echo Complication Registry). Am J Cardiol 2006;98:541-543.
- Bengel FM, Higuchi T, Javadi MS, et al. Cardiac positron emission tomography. J Am Coll Cardiol 2009;54:1-15.
- Di Carli MF, Dorbala S, Meserve J, et al. Clinical myocardial perfusion PET/CT. J Nucl Med 2007;48:783-793.
- Bateman TM, Heller GV, McGhie AI, et al. Diagnostic accuracy of rest/stress ECG-gated Rb-82 myocardial perfusion PET: Comparison with ECG-gated Tc-99m sestamibi SPECT. J Nucl Cardiol 2006;13:24-33.
- Chow BJ, Wong JW, Yoshinaga K, et al. Prognostic significance of dipyridamole-induced ST depression in patients with normal 82Rb PET myocardial perfusion imaging. J Nucl Med 2005;46:1095-1101.
- Yoshinaga K, Chow BJ, Williams K, et al. What is the prognostic value of myocardial perfusion imaging using rubidium-82 positron emission tomography? J Am Coll Cardiol 2006;48:1029-1039.
- Levsky JM, Haramati LB, Spevack DM, et al. Computed tomography angiography versus stress echocardiography in acute chest pain: A randomized controlled trial. JACC Cardiovasc Imaging 2018;11:1288-1297.
- De Filippo M, Capasso R. Coronary computed tomography angiography (CCTA) and cardiac magnetic resonance (CMR) imaging in the assessment of patients presenting with chest pain suspected for acute coronary syndrome. Ann Transl Med 2016;4:255.
- Chen MY, Shanbhag SM, Arai AE. Submillisievert median radiation dose for coronary angiography with a second-generation 320-detector row CT scanner in 107 consecutive patients. Radiology 2013;267:76-85.
- Roobottom C, Mitchell G, Iyengar S. The role of non-invasive imaging in patients with suspected acute coronary syndrome. Br J Radiol 2011;84:269-279.
- Takakuwa KM, Keith SW, Estepa AT, et al. A meta-analysis of 64-section coronary CT angiography findings for predicting 30-day major adverse cardiac events in patients presenting with symptoms suggestive of acute coronary syndrome. Acad Radiol 2011;18:1522-1528.
- Andreini D, Pontone G, Mushtaq S, et al. A long-term prognostic value of coronary CT angiography in suspected coronary artery disease. JACC Cardiovasc Imaging 2012;5:690-701
- Madder RD, Chinnaiyan KM, Marandici AM, et al. Features of disrupted plaques by coronary computed tomographic angiography: Correlates with invasively proven complex lesions. Circ Cardiovasc Imaging 2011;4:105-113.
- Otsuka K, Fukuda S, Tanaka A, et al. Napkin-ring sign on coronary CT angiography for the prediction of acute coronary syndrome. JACC Cardiovasc Imaging 2013;6:448-457.
- Dorbala S. Standard myocardial perfusion and cardiac FDG PET protocols and associated patient radiation doses. Image Wisely campaign. American College of Radiology 2012. Available at: https://www.imagewisely.org/-/media/Image-Wisely/Files/Nuclear-Medicine/Standard-Myocardial-Perfusion.pdf?la=en. Accessed June 16, 2019.
- Einstein AJ. Effects of radiation exposure from cardiac imaging. How good are the data? J Am Coll Cardiol 2012;59:553-565.
This article addresses specifics of the cardiac testing options, considering their appropriate indications, choices of approach, risks, and costs.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
