Suspected Acute Coronary Syndrome
October 1, 2019
Reprints
AUTHORS
Andrew J. Matuskowitz, MD, Department of Emergency Medicine, Medical University of South Carolina, Charleston
Aalap Shah, MD, Department of Emergency Medicine, Medical University of South Carolina, Charleston
Jordan McCarthy, MD, Department of Emergency Medicine, Medical University of South Carolina, Charleston
Patrick McNeal, PA, Department of Emergency Medicine, Medical University of South Carolina, Charleston
Richard R. Bayer II, MD, Division of Cardiology, Department of Medicine, Medical University of South Carolina, Charleston
PEER REVIEWER
William J. Brady, MD, Professor, Emergency Medicine, University of Virginia School of Medicine, Charlottesville
EXECUTIVE SUMMARY
• The HEART and Emergency Department Assessment of Chest Pain Accelerated Diagnostic pathways are better able to risk-stratify for 30-day major adverse cardiac events than clinical gestalt.
• High-sensitivity cardiac troponin testing can rapidly exclude acute myocardial infarction upon presentation.
• High-sensitivity troponin assays are not interchangeable; the limit of detection, upper reference limit, and value diagnostic of acute myocardial infarction are assay-specific.
• Coronary CT angiography (CCTA) can be used in patients without prior myocardial infarction or coronary revascularization.
• Stress myocardial perfusion testing can be used when CCTA is contraindicated.
The workup of suspected acute coronary syndrome (ACS) in the emergency department (ED) is an ever-evolving process, and staying up-to-date can be difficult. Guideline updates, new risk stratification decision rules, and emerging noninvasive imaging — not to mention high-sensitivity troponins — can overwhelm the the physician, leading to a dizzying array of practice variation between institutions and providers.
Once clear signs of acute myocardial infarction (AMI) are excluded by a 12-lead electrocardiogram (ECG) and cardiac troponins, risk assessment is necessary to estimate the likelihood of ACS and future major adverse cardiac events (MACE). Standardizing chest pain management for such patients more accurately identifies those at increased risk, reduces iatrogenic complications associated with unnecessary cardiac testing, unburdens overcrowded hospitals, and reduces healthcare-associated costs.
This article addresses controversies associated with ED evaluation of suspected ACS. Whether providers practice in a resource-limited environment or not, this review aims to empower ED providers to maximize diagnostic precision in a patient-centered and resource-conscious way.
Background and Significance
Chest pain is the second most common chief complaint in EDs and accounts for approximately 7 million to 8 million ED visits annually in the United States.1,2 Although only about 10% of patients ultimately are diagnosed with ACS, hospital admission rates for chest pain are commonly greater than 50% and have been documented as high as 70%.3-6 This over-triage practice strains hospital resources and exposes patients to unnecessary testing and iatrogenic risks. The cause of this phenomenon likely is multifactorial. Fear of litigation, risk intolerance, and an often-quoted statistic that ED providers miss ACS at a rate of 2% may have contributed.7 The quoted 2% miss rate was published in the bygone era of creatine kinase MB (CK-MB) cardiac biomarkers that possessed lower accuracy than contemporary troponins.8
Identifying ACS in those with the disease while minimizing unnecessary cardiac testing is critical to providing higher value, patient-centered care. No ACS assessment process is 100% accurate, so some cases will not be detected despite appropriate evaluation. The acceptable miss rate for ACS diagnosis is a matter of debate. ED providers vary in risk tolerance, but most believe that a 30-day ACS miss rate of < 1% is acceptable.9,10 Statistically speaking, this translates to a sensitivity (the proportion of patients with ACS who are identified correctly as having ACS) that approaches 99%, and a negative predictive value (NPV; the proportion of patients with a negative workup who do not have ACS) greater than 99%.11 This review will consider 99% sensitivity and at least 99% NPV as the measures needed to rule out ACS and short-term MACE adequately. When the 30-day MACE risk is estimated to be greater than 1%, patients are identified as at increased risk and require further cardiac workup. Of note, this review focuses exclusively on the evaluation of suspected ACS, but it is essential for emergency providers to consider other cardiac and noncardiac causes of chest pain.
Classifying Acute Coronary Syndromes
Understanding the spectrum of ACS is important. Most broadly, ACS represents a heterogenous group of life-threatening disease processes, including ST-elevation myocardial infarction (STEMI) and non-ST-elevation ACS (NSTE-ACS), the latter of which is subdivided further into non-ST-elevation myocardial infarction (NSTEMI) and unstable angina. A STEMI is characterized by the presence of ST-elevations in two consecutive leads on an ECG in an appropriate clinical context and indicates a high likelihood of complete coronary occlusion. STEMIs comprise roughly 30% of patients with ACS and require emergent invasive coronary angiography.12 The remaining 70-75% of patients with ACS include NSTEMI and unstable angina.13 NSTEMI is characterized by an elevated troponin level and an absence of significant ST-elevation. Unstable angina is characterized by myocardial ischemia at rest or with minimal exertion in the absence of cardiomyocyte necrosis. Consequently, patients with unstable angina do not have rises in cardiac troponin indicative of myocardial injury.14 (See the section on Contemporary Troponins.) Finally, MACE generally refers to adverse clinical outcomes, including STEMI, AMI, death, and, depending on the study, coronary revascularization (i.e., percutaneous coronary intervention [PCI] or coronary artery bypass grafting [CABG]).
Typical vs. Atypical Symptoms
The foundation for estimating pretest probability of ACS remains the history and physical exam. Typical chest pain is characterized as substernal chest pressure or discomfort that worsens with exertion or emotional stress and improves with rest or nitroglycerin.15 Other typical symptoms include dyspnea, diaphoresis, nausea, and presyncope. Atypical symptoms include weakness, epigastric discomfort or indigestion, or sharp chest pain.16 Although atypical symptoms present less commonly, women, older adults, people with diabetes, and patients with dementia are more likely to present atypically.17
Must-Know ECG Basics
The mortality rate of untreated STEMI is approximately 30%.18 Because survival rates increase significantly with faster PCI door-to-balloon times, the American Heart Association and American College of Cardiology (AHA/ACC) guidelines recommend ECG screening for all patients with suspected ACS within 10 minutes of first medical contact.19 A liberal use of ECG is encouraged to detect atypical presentations of ACS.20 Ischemic ECG findings include ST-elevation or depression, T-wave inversions, and Q-waves. In the appropriate clinical context, acute ST-depression ≥ 0.05 mV in two or more leads is suggestive of ACS, and acute ST-depression > 0.1 mV is associated with an 11% rate of myocardial infarction (MI) and death at one year.14 Although they are less specific, T-wave inversions are associated with a positive likelihood ratio for ACS of 1.8.21 The prognostic value of isolated T-wave inversions is unclear, but deep, symmetric T-wave inversions in the anterior leads often are related to significant stenosis of the proximal left anterior descending coronary artery.14 Patients with symptoms concerning for ACS who have ST-depressions or T-wave inversions should be categorized as high risk and likely require hospital admission. Finally, a normal ECG may be found in approximately 6% of patients with NSTE-ACS, underscoring the importance of assessing biomarkers.22
Single vs. Serial ECGs
To the authors’ knowledge, no studies have been conducted to establish whether single or serial ECGs improve outcomes in acute chest pain. However, case reports have shown that ECGs can become ischemic or diagnostic of STEMI following an initial unremarkable ECG.23 Because cardiac ischemia is a dynamic process, the AHA recommends serial ECG monitoring every 15-30 minutes for patients with concerning persistent or recurrent symptoms.19
Contemporary Troponins
Contemporary cardiac troponin I (cTnI) and troponin T (cTnT) assays are used commonly to assess for AMI.19 CK-MB biomarkers, which are not unique to heart muscle, lack sufficient sensitivity and specificity and should be discarded.24,25 An elevated contemporary cardiac troponin (cTn) level is defined as greater than the 99th percentile upper reference limit (URL) in a healthy population. An AMI is defined as a combination of rising or decreasing cardiac biomarkers with at least one cTn level above the 99th percentile URL and symptoms, acute ECG changes, or imaging findings consistent with myocardial ischemia.26 Contemporary troponin levels elevate within two to four hours of injury, and peak values generally occur 18-24 hours after insult.27 A single negative cTn level can exclude AMI when obtained more than six to nine hours after the onset of ongoing chest pain. However, when symptoms are intermittent or less than six hours, repeat measurements for up to six hours after symptom onset is recommended.19
Mild Troponin Elevations
An elevated contemporary troponin by itself portends a worse short-term prognosis, but some patients have persistent, usually low-level troponin elevation and do not automatically require hospitalization.28 Chronic unchanging elevations usually reflect structural heart disease, such as heart failure, or impaired renal clearance.29,30 Reviewing previous baseline troponin levels can be particularly useful in these circumstances. On the other hand, increasing troponin values suggest acute disease. Non-ACS causes of acutely elevated or changing troponin levels include, but are not limited to, Takotsubo cardiomyopathy, tachyarrhythmias, pulmonary embolism, stroke, sepsis, and gastrointestinal bleeding.30 When mildly elevated initial troponin values do not change after two to three hours, treat the underlying illness accordingly.
Delta Troponin Levels
Characterizing the degree of changing troponin values to diagnose AMI remains controversial. The European Society of Cardiology (ESC) and AHA/ACC guidelines suggest using a troponin change > 20% with an elevated baseline troponin level to identify AMI.14,19 Other studies suggest that there may be a slight advantage to using an absolute numerical change, or delta, which depends on the specific assay.31,32 Unfortunately, there is no universally accepted delta value. If using an absolute value change, refer to your institution’s specific troponin assay to determine the change diagnostic of AMI. If none is available, consider using the 20% change cutoff instead.
ED Discharge vs. Further Assessment
Differentiating between low-risk patients who can be discharged safely without further testing in the ED and those at increased risk who may benefit from further assessment can be difficult. In patients with chest pain for whom ACS is suspected but the ECG and troponins are unremarkable, the AHA/ACC guidelines provide well-meaning but vague recommendations. “Reasonable” options include patient observation with serial ECGs and troponins, noninvasive cardiac imaging, or discharge with stress testing within 72 hours.19 However, the risks associated with cardiac testing, additional costs, and increased length of stay (LOS) attributed to further cardiac workup should be measured against each patient’s intrinsic risk of ACS. For instance, widespread use of advanced cardiac imaging without appropriate risk-benefit assessment results in increased downstream cardiac testing, contrast-associated risks, and higher radiation doses. Numerous studies in low-risk patients have found that stress testing or coronary CT angiography (CCTA) does not improve patient outcomes and has high false-positive rates, leading to further unnecessary testing.33-36 Contrast-associated nephropathy and adverse reactions to contrast dye are uncommon but non-negligible risks associated with CCTA.37,38 Similarly, CCTA and stress myocardial perfusion testing impart radiation risks.39 Although the lifetime cancer risks from these tests are low, they must be considered as part of the patient’s broader risk assessment.40,41
Iatrogenic risks and unnecessary healthcare utilization associated with cardiac testing underscore the importance of appropriate pre-imaging risk stratification. Although clinical impression, or gestalt, often is invoked, reliability is at best modest; as noted in a study of experienced ED physicians, “typicality” of chest pain was poorly sensitive (39.3%-53.1%) and specific (65.8%) for AMI and serious coronary artery disease (CAD).42 On the other hand, risk-stratification decision rules, such as the HEART Pathway and Emergency Department Assessment of Chest Pain Accelerated Diagnostic Pathway (EDACS-ADP), offer several advantages. Decision rules estimate risk objectively, provide judgment when clinical risk is unclear, reduce physician practice variation, facilitate standardized communication with consultants, and offer evidence-based justification for decisions if adverse outcomes occur.43 Ultimately, decision rules help determine who can be discharged safely with follow-up and who requires further workup.
Risk Stratification Decision Rules
Importantly, not all decision rules are created equal. For instance, the Thrombolysis in Myocardial Infarction (TIMI) score was developed not to identify which patients are likely to have ACS in the acute chest pain population, but to identify which patients known to have ACS are likely to die within the next 14 days.44 It has been tested in the undifferentiated chest pain population but failed to stratify patients into separate risk groups accurately and missed 1.7% cases of MACE in discharged patients.45 Similarly, the Accelerated Diagnostic Protocol for Chest Pain (ADAPT) and Vancouver decision rules, while adequately sensitive for MACE, identify only about 20-30% as low risk.6,46,47 Currently, three of the most commonly used risk stratifying decision rules are the HEART score, HEART Pathway, and EDACS-ADP.
HEART Score
The HEART score classifies patients into low-, moderate-, and high-risk groups based on a component score of five elements: History, ECG, Age, Risk factors, and a single contemporary Troponin.48 (See Table 1.) According to the original and subsequent validation studies, patients classified as low risk had 1.0-1.7% incidence of MACE at six weeks.48-50 A recent multicenter, randomized trial compared outcomes using the HEART score to usual care. Investigators found that the six-week MACE incidence was 1.3% lower than with usual care.51 Most recently, in a meta-analysis assessing 29 HEART score studies, researchers found that the HEART score had a pooled sensitivity of 95.9%.52 Although the HEART score does not meet the generally accepted 99% sensitivity threshold, it marks a transition to an increased focus on the patient history, which has greatly influenced future risk-stratifying decision rules.
Table 1. HEART Score |
|||
|
History |
Highly suspicious |
2 points |
Mostly high-risk features |
|
Moderately suspicious |
1 point |
Mixture of high- and low-risk features |
|
|
Slightly suspicious |
0 points |
Mostly low-risk features |
|
|
High-Risk Features
|
Low-Risk Features
|
||
|
Electrocardiogram |
New ischemic changes |
2 points |
|
|
Nonspecific changes |
1 point |
|
|
|
Normal |
0 points |
|
|
|
Age |
≥ 65 years |
2 points |
|
|
45-64 years |
1 point |
||
|
< 45 years |
0 points |
||
|
Risk Factors |
≥ 3 risk factors or history of atherosclerotic disease |
2 points |
|
|
1-2 risk factors |
1 point |
||
|
No risk factors |
0 points |
||
|
Troponin |
> 3 × normal limit |
2 points |
|
|
1-3 × normal limit |
1 point |
||
|
≤ normal limit |
0 points |
||
|
Sources: Adapted from: Huis in’t Veld MA, Cullen L, Haler SA, et al. The fast and the furious: Low-risk chest pain and the rapid rule-out protcol. West J EM 2017;18. https://creativecommons.org/licenses/by/4.0/. Cakal ED. Is troponin enough? International Emergency Medicine Project. July 17, 2018. |
|||
HEART Pathway
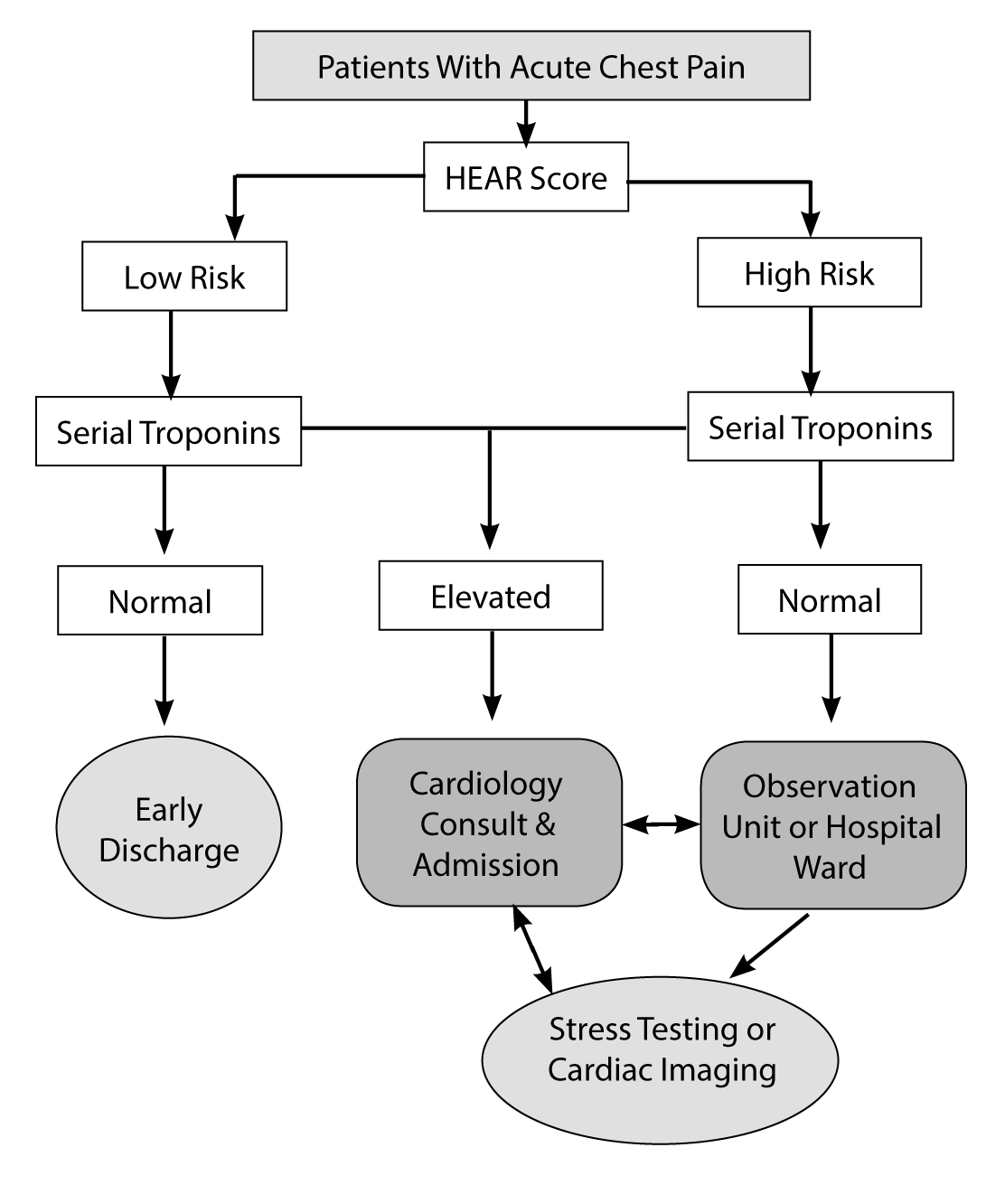
To improve upon the HEART score’s predictive value, the HEART Pathway was developed. The HEART Pathway combines a HEAR (History, ECG, Age, Risk factors) score with serial troponins collected at zero and three hours. (See Figure 1.) Patients with an ischemic ECG or an elevated initial or serial troponin are automatically identified as high risk. Patients with HEAR scores ≤ 3 and negative serial troponins are considered low risk; patients with HEAR scores ≥ 4 or who have a troponin above the 99th percentile URL are considered high risk. In the HEART Pathway Randomized Controlled Trial, researchers randomized patients to the HEART Pathway or usual care. The HEART Pathway identified 46.8% of patients as low risk and safe for discharge, none of whom had a MACE at 30 days. Compared to usual care, the HEART Pathway increased ED discharges by 21%, decreased length of stay (LOS) by 12 hours, and decreased cardiac testing by 12%.5 Impressively, 0% of the low-risk patients had MACE at one year.53 Validation studies have confirmed the value of the HEART Pathway. In one prospective pre-post study involving three institutions and 8,474 total patients, Mahler et al found that implementation of the HEART Pathway using an interactive decision support tool reduced hospitalization by 6% and detected slightly more AMIs. Importantly, MI or death occurred in only 0.4% of low-risk patients in the postimplementation group.54 Numerous external validations of the HEART Pathway found that patients identified as low risk were discharged with < 1% risk of MACE within four to six weeks.55-58
Figure 1. HEART Pathway |
 |
|
Source: Reprinted with permission from Mahler SA, Stopyra JP, Apple FS, et al. Use of the HEART Pathway with high sensitivity cardiac troponins: A secondary analysis. Clin Biochem 2017;50:7. |
HEART Score and HEART Pathway Limitations
The HEART score and HEART Pathway have several limitations. In the HEART score, risk is estimated based on a single troponin. Unless chest discomfort has been ongoing for at least six hours, contemporary serial troponins should be collected and separated by at least three hours.19 The absence of a required second troponin in the HEART score likely contributes to its suboptimal sensitivity. Additionally, a low-risk HEART score ≤ 3 can be achieved with only an elevated troponin and ischemic ECG, both of which are independent risk factors for death.28,59,60 Unless an elevated troponin is mild and unchanged from baseline — as may be the case in chronic heart failure or renal disease — such patients automatically should be considered high risk.29
Neither the HEART score nor HEART Pathway should be used to risk-stratify patients with a prior MI or revascularization. Such patients inherently have an increased risk for MACE. Also, the history component of both rules is fairly subjective and may differ depending on the provider.61 Provider variation in the history component can be reduced by assigning two points if three or more high-risk features are present, one point if two high-risk features are present, and zero points if only one high-risk feature is present.62 (See Table 2.) Alternatively, two points can be assigned if only high-risk features are present, one point if both high- and low-risk features are present, and zero points if only low-risk features are present.63 If your institution uses the HEART Pathway, consider minimizing provider variation by standardizing the history component using one of these strategies.
EDACS-ADP
Compared to the HEART Pathway, the EDACS-ADP has demonstrated similar sensitivity and NPV, and it has shown promise in identifying a higher proportion of low-risk patients who can forego further cardiac testing. EDACS contains clinical characteristics that are assigned positive or negative point values. (See Table 2.) The EDACS-ADP identifies patients as safe for discharge if EDACS < 16 with no new ischemic ECG findings and negative serial troponins at zero and two hours after presentation. In the initial derivation and validation study, EDACS identified 51.3% of its cohorts as low risk and was 99-100% sensitive.64 Subsequent validation studies found similarly outstanding sensitivities and NPVs. When compared to other ED-based risk stratification decision rules, the EDACS-ADP identified more patients as safe for discharge than ADAPT (41.6-51.3% vs. 20-30.5%), the Vancouver Rule (41.6% vs. 25.7%), and a variation of the HEART score (60.8% vs. 51.8%).47,63,65 It is worth noting that one study using prospectively collected data from the HEART Pathway randomized controlled trial found that the EDACS-ADP was only 88.2% sensitive for MACE. This was a small, single-center trial, and the authors noted that differences in healthcare practices may have contributed to the lower sensitivity.11 Specifically, in the original New Zealand EDACS studies, fewer patients with MACE had revascularizations.11,47,64 In contrast, the U.S. study had higher revascularization rates, which are of unclear benefit, and may be more reflective of practice patterns than true ACS. Despite this anomalous study, the pooled 95% confidence interval of the two prospective and two retrospective EDACS-ADP studies demonstrated an NPV of 99.23% to 99.94%.63 These pooled findings are promising, but a large, prospective U.S. trial to assess generalizability would further support the safety of the EDACS-ADP.
Table 2. Emergency Department Assessment of Chest Pain Accelerated Diagnostic Pathway (EDACS-ADP) |
|
|
Clinical Characteristics |
Score |
|
A) Age 18-45 |
+2 |
|
46-50 |
+4 |
|
51-55 |
+6 |
|
56-60 |
+8 |
|
61-65 |
+10 |
|
66-70 |
+12 |
|
71-75 |
+14 |
|
76-80 |
+16 |
|
81-85 |
+18 |
|
≥ 86 |
+20 |
|
B) Male sex |
+6 |
|
C) 18-50 years and either I) known coronary artery disease or II) ≥ 3 risk factors |
+4 |
|
D) Symptoms and signs Diaphoresis Radiates to arm or shoulder Pain occurred or worsened with inspiration |
+3 +5 -4 |
|
Pain is reproduced by palpation |
-6 |
|
EDACS Total EDACS-ADP Management Recommendations Low Risk Recommendation |
____ (I) EDACS < 16 (II) No ischemia on ECG (III) 0 and 2 h troponins negative Patients safe for discharge to early outpatient follow-up investigation |
|
Not Low Risk Recommendation |
(I) EDACS ≥ 16 (II) New ischemia on ECG (III) Either 0 or 2 h troponin positive Proceed with usual care with further observation and delayed troponin |
|
Sources: Adapted from: Huis in’t Veld MA, Cullen L, Haler SA, et al. The fast and the furious: Low-risk chest pain and the rapid rule-out protcol. West J EM 2017;18. https://creativecommons.org/licenses/by/4.0/. Than M, Flaws D, Sanders S, et al. Development and validation of the Emergency Department Assessment of Chest pain Score and 2 h accelerated diagnostic protocol. Emerg Med Australas 2014;26:34-44. |
|
High-Sensitivity Troponins
The U.S. Food and Drug Administration (FDA) has finally approved several high-sensitivity cardiac troponin (hs-cTn) assays for use in the United States.14,66 Compared to contemporary troponin assays, hs-cTn assays have higher NPV for AMI, have reduced time to detection of AMI, and result in an approximately 4% absolute increase in the detection of AMI caused by atherothrombi.14
Excluding AMI
High-sensitivity troponins are defined as those that can detect cardiac troponin levels in ≥ 50% of healthy individuals. Unlike contemporary troponins, which require three- to six-hour serial testing, hs-cTns have shown remarkable effectiveness at excluding AMI using zero- and one-hour testing. For instance, early multicenter hs-cTn studies, which used a 0 h/1 h algorithm, found that approximately 60-63% of patients were excluded for an AMI and had 99-100% NPV when the hs-cTnT level was less than the 99th percentile URL and had an absolute change of < 2 ng/L within one hour.67-69 Pooled NPV for AMI in patients identified as a “rule-out” cohort exceeded 98% in several large validation studies.14 The 2015 ESC guidelines subsequently approved the 0 h/1 h algorithm, provided that it includes a clinical assessment of the likelihood of ACS and 12-lead ECG.
Evidence also suggests that a single initial hs-cTn below the limit of detection essentially may exclude the diagnosis of AMI. One large, 15-site, observational study using an hs-cTnT assay demonstrated that a single undetectable hs-cTnT level on arrival had a 99.4% NPV for AMI.70 These findings reinforce that hs-cTns are a powerful tool to exclude AMI in the ED setting. But do hs-cTn assays also exclude 30-day MACE reliably?
Excluding MACE
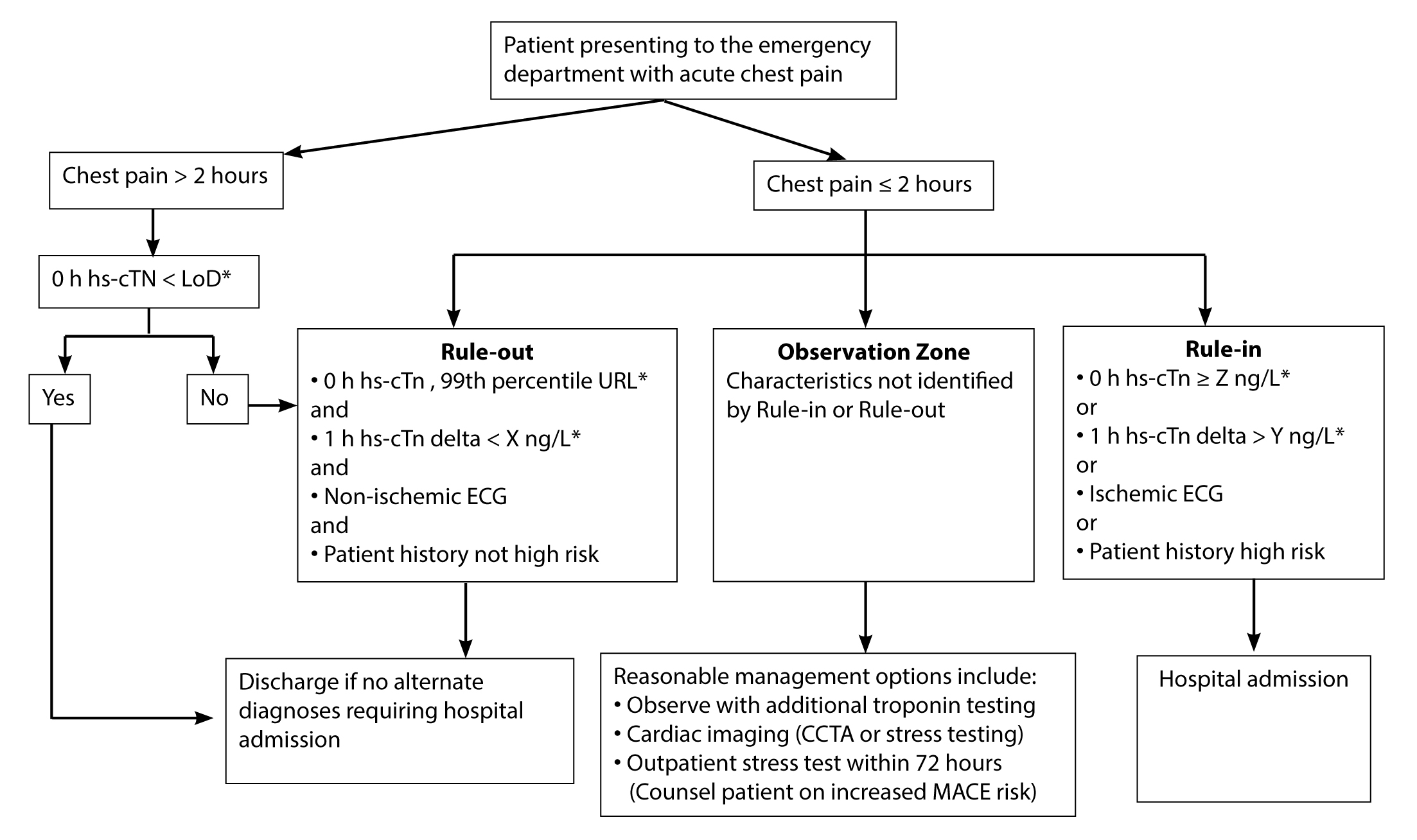
In the 15-site study noted earlier, investigators also assessed 30-day MACE in patients with hs-cTn levels < 19 ng/L (the 99th percentile URL) at zero and three hours. They found that serial hs-cTnT levels < 19 ng/L had 99.3% NPV for MACE at 30 days.70 Similarly, just as a single initial hs-cTn assay below the limit of detection may exclude AMI sufficiently, it also may exclude 30-day MACE sufficiently. A large meta-analysis involving an hs-cTnI assay found that in patients presenting more than two hours after symptom onset without ischemic ECG findings and hs-cTnI below the limit of detection at presentation had a 99.6% NPV for AMI and cardiac death at 30 days.71 Additional large studies using different hs-cTn assays are needed to assess 30-day MACE, but these findings provide compelling evidence that hs-cTns reliably identify a sufficient proportion of very low-risk patients who can be discharged safely without further cardiac testing. (See Figure 2.)
Figure 2. Suggested High-Sensitivity Troponin Workflow |
 |
|
* Denotes assay-specific values. LoD = Limit of detection; X = Absolute delta consistent with no clear ischemic disease; Y = Absolute delta consistent with clear ischemic disease; Z = Value consistent with probable acute myocardial infarction. |
The Lower Specificity Controversy
The increased detection of elevated troponin levels that occurs with hs-cTn assays comes at the cost of lower specificity. Lower specificity makes many physicians concerned that a higher proportion of patients with mildly increased hs-cTn levels will increase cardiac testing and hospitalizations unnecessarily. This concern is reasonable, but it should be measured against the total proportion of patients expected to be included or excluded effectively for ACS using hs-cTn assays. Using 0- and 1-hour hs-cTn time intervals, four large, multicenter studies found that the majority of patients were classified accurately into “rule-in” and “rule-out” groups where the disposition was clear. The “rule-out” groups comprised 50-63% of patients with 99-100% NPV for AMI, and the “rule-in” groups comprised 10%-17% of patients, of whom 74-84% had an AMI. The remaining 23-31% of patients in these studies had mildly elevated or changing hs-cTn levels, classifying them in an “observation zone,” 8-23% of whom had an AMI.67-69,72 The “observation zone” is a heterogenous group, and further workup depends on the clinical presentation. In this cohort, reasonable options include further serial hs-cTn testing (e.g., at three hours), rapid CCTA or stress testing, or admission to an observation unit.14 (See Figure 2.)
Combining High-Sensitivity Troponins With Decision Rules
The added value of combining risk stratification decision rules with hs-cTn assays is unclear. The initial and validation EDACS-ADP studies used hs-cTn assays and classified a respectable 42-51% as low risk, with 100% NPV for 30-day MACE.47,64 Similarly, researchers using an hs-cTn assay identified 37.2% of patients with HEART scores ≤ 3, of whom none had MACE at 30 and 180 days.73 A larger multinational study assessed a modified HEART Pathway that combined the HEAR score with 0- and 1-hour hs-cTn testing. Low risk was defined as a HEAR score ≤ 3 with an hs-cTnT < 14 ng/L at presentation and a change of < 3 ng/mL at one hour. Forty percent of patients were identified as very low risk and < 1% experienced AMI or death at 30 days. Conversely, in patients with negative 0- and 1-hour hs-cTn levels, a HEAR score ≥ 4 resulted in 2% risk of AMI or death at 30 days.62
Further studies are needed that directly assess the added value of hs-cTn levels in combination with decision rules compared to hs-cTn levels with gestalt and ECG alone. Until such time, both strategies seem reasonable and justifiable.
Reduced Prevalence of Unstable Angina
Although hs-cTn assays are expected to increase the detection of AMI and correspondingly decrease the diagnosis of unstable angina, unstable angina is unlikely to disappear altogether. The aforementioned hs-cTn studies have demonstrated a remarkable capacity to exclude AMI, but normal or unchanging hs-cTn levels do not completely exclude ACS.74 Unstable angina can be identified retrospectively in patients who were discharged after AMI was excluded but subsequently were diagnosed with AMI after the initial visit. Ischemic ECG patterns, abnormal noninvasive imaging, and flow-limiting coronary stenosis benefiting from PCI occasionally occur despite normal hs-cTn levels.75 For instance, in the McCord et al study described earlier, patients with unremarkable hs-cTn levels and a HEAR score ≥ 4 still had 30-day MACE of 2%. This not-insignificant rate suggests that unstable angina will play a smaller but ongoing role in the future.62
The Takeaway
High-sensitivity troponins are highly effective for excluding disease, and the expected increase in minimally elevated troponins seems to be outmatched by the higher proportion of patients classified in discreet rule-in and rule-out groups. When initial hs-cTn levels are below the 99th percentile URL or relatively unchanged at one hour, both AMI and 30-day MACE seem to be excluded with a high NPV. Similarly, when symptom onset is greater than two hours, a single hs-cTn below the limit of detection effectively excluded AMI and 30-day MACE in early studies. Integrating decision rules (e.g., HEART Pathway and EDACS-ADP) with serial hs-cTns has been shown to rule out ACS within 30 days reliably, but at this time, it is not clearly superior to the combined use of hs-cTns, gestalt, and ECG. One final point: hs-cTnI and hs-cTnT assays are not interchangeable. The limit of detection, 99th percentile URL, and value diagnostic of AMI is assay specific.67,76 Additionally, hs-cTnI assays contain sex-specific cutoffs for the 99th percentile URL, whereas hs-cTnT assays do not. Be sure to follow the assay specific instructions at your institution.
Next Steps for Patients at Increased ACS Risk
Further workup is necessary in patients classified as at increased risk by objective risk-stratification decision rules, mildly elevated or changing hs-cTn levels, or a combination of the two. Choosing between inpatient observation, noninvasive cardiac imaging, or discharge with stress testing within 72 hours requires nuanced decision-making. Observation or short-say units provide a suitable option for patients thought to have an increased risk of ACS. When no observation unit is available, CCTA and myocardial perfusion stress testing may be ideal alternatives. A fourth, often overlooked option includes shared decision-making with patients. The AHA/ACC guidelines support shared decision-making.19 One randomized trial comparing shared decision-making to usual care found that in patients with an estimated 2% risk of short-term MACE, shared decision-making increased patient knowledge and engagement in decision-making and actually decreased the rate of observation unit admission for stress testing.77 However, patients who choose to depart from validated, evidence-based recommendations must be made aware of their increased MACE risk.
Noninvasive Imaging in ACS
Noninvasive cardiac imaging is indicated in patients identified as at increased risk for ACS without ischemic ECG changes and negative troponins. Stress ECG, stress myocardial perfusion, stress echocardiography, cardiac magnetic resonance imaging (CMR), and CCTA are approved by the AHA/ACC guidelines.19 Choosing between rest and stress testing options depends on time and whether the patient has had a prior MI or revascularization procedure. Resting studies typically are performed more quickly than stress testing.78 In general, patients without a prior MI or revascularization may undergo resting myocardial perfusion testing or CCTA. These modalities provide an anatomic assessment of perfusion defects and coronary vessel stenoses, respectively. Both have been tested in randomized trials, as opposed to the resting echocardiograms and CMR that are not as well studied.35,79,80 In patients with a prior MI or revascularization, resting myocardial perfusion and CCTA do not adequately identify current ischemia. Instead, stress testing, myocardial perfusion, echocardiography, or CMR is required.78 Given the abundance of data supporting their use, this review focuses on the strengths and weakness of CCTA and myocardial perfusion stress testing.
Coronary CT Angiography
Coronary CT angiography using a 64-slice CT scanner provides an anatomical assessment of the degree of stenosis in the coronary lumen and degree of calcium burden in the surrounding vessels. While calcium scoring has limited value in the ED setting, the degree of coronary stenosis is a highly useful surrogate for ACS and offers several advantages to stress testing.81 CCTA has demonstrated sensitivities of approximately 95% (confidence interval [CI] 93% to 97%) and NPV > 99%.35,79,82 In the multicenter Acrin-PA Trial specifically, no patients with less than 50% stenosis had MACE at 30 days.35 Two large, randomized controlled trials comparing CCTA with standard care demonstrated a decreased LOS by six to 12 hours compared with stress testing or usual care. CCTA also tripled the number of patients discharged from the ED.35,79 More recently, the SCOT-HEART investigators found that CCTA plus standard care significantly decreased the rate of death and MI at five years compared to standard care alone. The authors asserted that CCTA identified mild and moderate CAD in patients earlier, which resulted in more preventive medical therapies.83
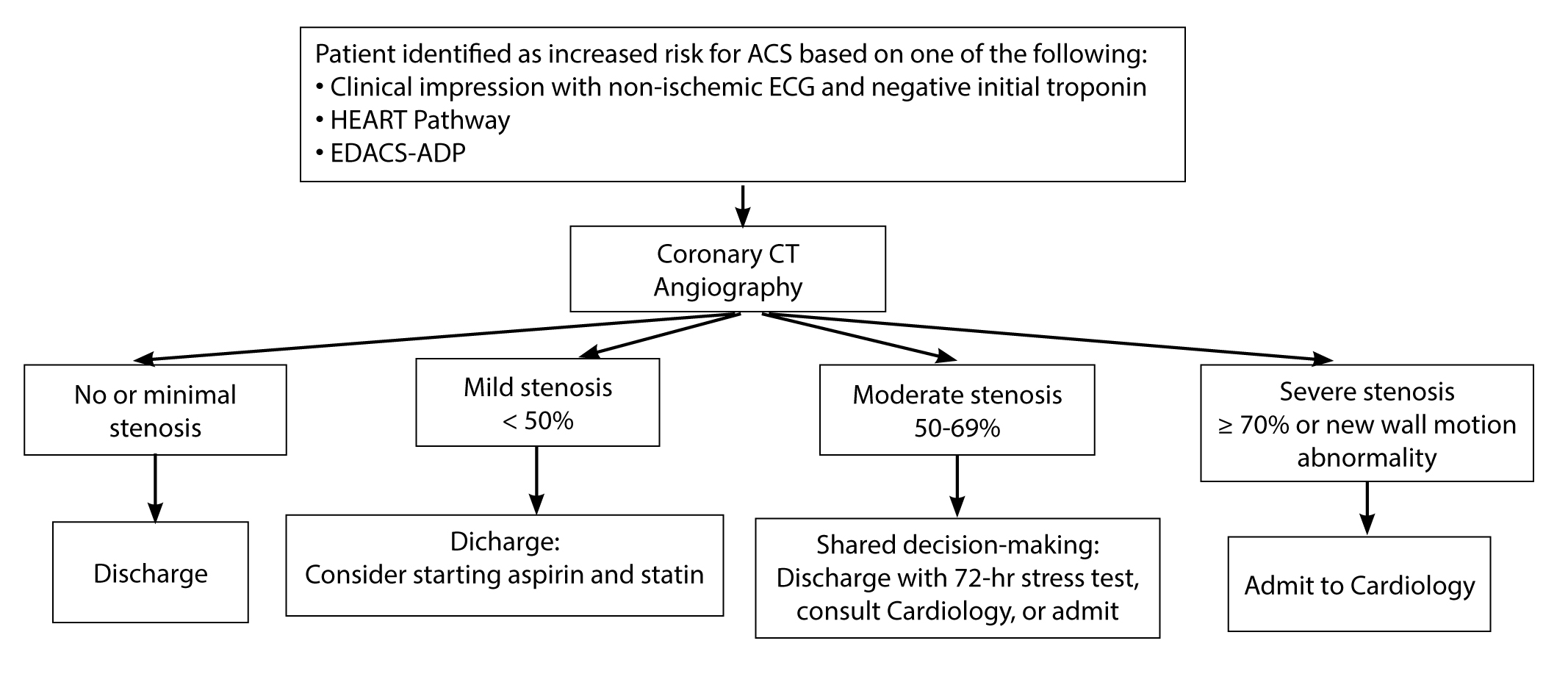
The main limitations of CCTA are specificity and positive predictive value (PPV). One review reported that PPV using invasive coronary angiography as the refence standard ranged from 50-90%, depending on the study.78 However, the majority of patients undergoing CCTA in several studies were found to have no, minimal, or mild stenosis (< 50%), which correlated with < 1% short-term MACE risk.35,84 Therefore, the strength of CCTA is its ability to quickly reclassify a large proportion of patients previously thought to be at increased risk for ACS as safe for discharge. For institutions with CCTA, a reasonable management approach is depicted in Figure 3.35,84,85
Figure 3. Coronary CT Angiography Suggested Workflow |
 |
Stress Myocardial Perfusion Testing
Stress myocardial perfusion testing detects the presence and extent of inducible perfusion abnormalities suggestive of ischemia as well as the presence of prior infarction.78 Compared to CCTA, stress myocardial perfusion testing has lower sensitivities (70-85%) and specificities (80-90%).86 One meta-analysis directly compared CCTA and myocardial perfusion stress tests and found that CCTA had a pooled sensitivity of 99%, while myocardial perfusion stress testing had a much lower pooled sensitivity of 73%.87 As a rule-in test, myocardial perfusion imaging testing also demonstrated higher false-positive rates than CCTA.36
CCTA can be performed following a patient’s first negative troponin result while still in the ED, whereas stress testing typically occurs following admission to inpatient or observation units or within 72 hours of patient discharge.
Despite several advantages of CCTA, stress testing continues to play an important diagnostic role in acute chest pain. Stress testing induces ischemic changes, which provides a functional assessment not possible with CCTA.78 Furthermore, stress testing can be used when CCTA is absolutely or relatively contraindicated, including in patients with known contrast-associated reactions, previous cardiac stent placement or CABG, tachycardia, atrial fibrillation, or morbid obesity.88 Finally, many institutions do not have 64-slice CCTA scanning capabilities or trained cardiothoracic imaging radiologists. In such cases, stress testing is reasonable and appropriate.
Summary
High-sensitivity troponins, observation units, and CCTA are powerful modalities that assist in identifying ACS, but they are not always available. In resource-limited environments, ED providers can still use evidence-based strategies that reliably identify ACS while minimizing patient risks, overcrowding, and healthcare costs. In patients without ischemic ECG changes and negative contemporary troponins, consider using the HEART Pathway or EDACS-ADP prior to reflexive hospital admission or observation. These decision rules often identify more patients as low risk than gestalt. Moreover, patients identified as low risk have short-term MACE rates
< 1% and can be discharged with routine follow-up. On the other hand, higher-risk patients often can be referred for outpatient stress testing within 72 hours. Regardless of institutional resources, adhering to evidence-based practices enhances care delivery to patients.
REFERENCES
- Rui P, et al. National Hospital Ambulatory Medical Care Survey: 2016 Emergency Department Summary Tables. 2016, U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics: Hyattsville, MD.
- National Hospital Ambulatory Medical Care Survey: 2015 Emergency Department Summary Tables. U.S Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics. 2015.
- Venkatesh AK, et al. Variation in US hospital emergency department admission rates by clinical condition. Med Care 2015;53:237-244.
- Mahler SA, et al. Can the HEART score safely reduce stress testing and cardiac imaging in patients at low risk for major adverse cardiac events? Crit Pathw Cardiol 2011;10:128-133.
- Mahler SA, et al. The HEART Pathway randomized trial: Identifying emergency department patients with acute chest pain for early discharge. Circ Cardiovasc Qual Outcomes 2015;8:195-203.
- Than M, et al. 2-Hour accelerated diagnostic protocol to assess patients with chest pain symptoms using contemporary troponins as the only biomarker: The ADAPT trial. J Am Coll Cardiol 2012;59:2091-2098.
- Pope JH, et al. Missed diagnoses of acute cardiac ischemia in the emergency department. N Engl J Med 2000;342:1163-1170.
- Reinhardt SW, et al. Noninvasive cardiac testing vs clinical evaluation alone in acute chest pain: A secondary analysis of the ROMICAT-II randomized clinical trial. JAMA Intern Med 2018;178:212-219.
- Than M, et al. What is an acceptable risk of major adverse cardiac event in chest pain patients soon after discharge from the emergency department?: A clinical survey. Int J Cardiol 2013;166:752-754.
- Kline JA, et al. Pretest probability assessment derived from attribute matching. BMC Med Inform Decis Mak 2005;5:26.
- Stopyra JP, et al. Performance of the EDACS-accelerated diagnostic pathway in a cohort of US patients with acute chest pain. Crit Pathw Cardiol 2015;14:134-138.
- Steg PG, et al. Baseline characteristics, management practices, and in-hospital outcomes of patients hospitalized with acute coronary syndromes in the Global Registry of Acute Coronary Events (GRACE). Am J Cardiol 2002;90:358-363.
- Go AS, et al. Heart disease and stroke statistics — 2013 update: A report from the American Heart Association. Circulation 2013;127:e6-e245.
- Roffi M, et al. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: Task Force for the Management of Acute Coronary Syndromes in Patients Presenting without Persistent ST-Segment Elevation of the European Society of Cardiology (ESC). Eur Heart J 2016;37:267-315.
- Diamond GA, Forrester JS. Analysis of probability as an aid in the clinical diagnosis of coronary-artery disease. N Engl J Med 1979;300:1350-1358.
- Swap CJ, Nagurney JT. Value and limitations of chest pain history in the evaluation of patients with suspected acute coronary syndromes. JAMA 2005;294:2623-2629.
- Culic V, et al. Symptom presentation of acute myocardial infarction: Influence of sex, age, and risk factors. Am Heart J 2002;144:1012-1017.
- Terkelsen CJ, et al. System delay and mortality among patients with STEMI treated with primary percutaneous coronary intervention. JAMA 2010;304:763-771.
- Amsterdam EA, et al. 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: Executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014;130:2354-2394.
- O’Gara PT, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: Executive summary: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 2013;127:529-555.
- Fanaroff AC, et al. Does this patient with chest pain have acute coronary syndrome? The Rational Clinical Examination Systematic Review. JAMA 2015;314:
1955-1965. - Thygesen K, et al. Third universal definition of myocardial infarction. J Am Coll Cardiol 2012;60:1581-1598.
- Velez J, et al. Serial electrocardiography. Am J Emerg Med 2002;20:43-49.
- Keller T, et al. Serial changes in highly sensitive troponin I assay and early diagnosis of myocardial infarction. JAMA 2011;306:2684-2693.
- Reichlin T, et al. Early diagnosis of myocardial infarction with sensitive cardiac troponin assays. N Engl J Med 2009;361:858-867.
- Thygesen K, et al. Fourth Universal Definition of Myocardial Infarction (2018). J Am Coll Cardiol 2018;72:2231-2264.
- Babuin L, Jaffe AS. Troponin: The biomarker of choice for the detection of cardiac injury. CMAJ 2005;173:1191-1202.
- Zethelius B, et al. Troponin I as a predictor of coronary heart disease and mortality in 70-year-old men: A community-based cohort study. Circulation 2006;113:1071-1078.
- Hollander JE. Managing troponin testing. Ann Emerg Med 2016;68:690-694.
- Vasile VC, Jaffe AS. High-sensitivity cardiac troponin for the diagnosis of patients with acute coronary syndromes. Curr Cardiol Rep 2017;19:92.
- Mueller M, et al. Absolute and relative kinetic changes of high-sensitivity cardiac troponin T in acute coronary syndrome and in patients with increased troponin in the absence of acute coronary syndrome. Clin Chem 2012;58:209-218.
- Reichlin T, et al. Utility of absolute and relative changes in cardiac troponin concentrations in the early diagnosis of acute myocardial infarction. Circulation 2011;124:136-145.
- Dawson M, et al. Low-risk young adult patients with chest pain may not benefit from routine cardiac stress testing: A Bayesian analysis. Crit Pathw Cardiol 2010;9:170-173.
- Hamilton B, et al. Stress testing in young low-risk patients with potential acute coronary syndromes. Am J Emerg Med 2012;30:639-642.
- Litt HI, et al. CT angiography for safe discharge of patients with possible acute coronary syndromes. N Engl J Med 2012;366:1393-1403.
- Hartsell S, et al. False-positive rates of provocative cardiac testing in chest pain patients admitted to an emergency department observation unit. Crit Pathw Cardiol 2014;13:104-108.
- Mehran R, et al. Contrast-associated acute kidney injury. N Engl J Med 2019;380:2146-2155.
- SCOT-HEART investigators. CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): An open-label, parallel-group, multicentre trial. Lancet 2015;385:2383-2391.
- Douglas PS, et al. Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med 2015;372:1291-1300.
- Committee to Assess Health Risks from Exposure to Low Levels of Ionizing Radiation. Health risks from exposure to low levels of ionizing radiation. BEIR VII Phase 2. 2006, Washington, DC: National Academies Press.
- Mettler FA Jr., et al. Effective doses in radiology and diagnostic nuclear medicine: A catalog. Radiology 2008;248:254-263.
- Carlton EW, et al. ‘Chest pain typicality’ in suspected acute coronary syndromes and the impact of clinical experience. Am J Med 2015;128:1109-1116 e2.
- Innes GD. Can a HEART pathway improve safety and diagnostic efficiency for patients with chest pain? Ann Emerg Med 2019;74:181-183.
- Antman EM, et al. The TIMI risk score for unstable angina/non-ST elevation MI: A method for prognostication and therapeutic decision making. JAMA 2000;284:835-842.
- Chase M, et al. Prospective validation of the Thrombolysis in Myocardial Infarction Risk Score in the emergency department chest pain population. Ann Emerg Med 2006;48: 252-259.
- Scheuermeyer FX, et al. Development and validation of a prediction rule for early discharge of low-risk emergency department patients with potential ischemic chest pain. CJEM 2014;16:106-119.
- Than MP, et al. Effectiveness of EDACS versus ADAPT accelerated diagnostic pathways for chest pain: A pragmatic randomized controlled trial embedded within practice. Ann Emerg Med 2016;68:93-102 e1.
- Six AJ, et al. Chest pain in the emergency room: Value of the HEART score. Neth Heart J 2008;16:191-196.
- Backus BE, et al. A prospective validation of the HEART score for chest pain patients at the emergency department. Int J Cardiol 2013;168:2153-2158.
- Backus BE, et al. Chest pain in the emergency room: A multicenter validation of the HEART Score. Crit Pathw Cardiol 2010;9:164-169.
- Poldervaart JM, et al. Effect of using the HEART Score in patients with chest pain in the emergency department: A stepped-wedge, cluster randomized trial. Ann Intern Med 2017;166:689-697.
- Fernando SM, et al. Prognostic accuracy of the HEART Score for prediction of major adverse cardiac events in patients presenting with chest pain: A systematic review and meta-analysis. Acad Emerg Med 2019;26:140-151.
- Stopyra JP, et al. The HEART Pathway randomized controlled trial one-year outcomes. Acad Emerg Med 2019;26:41-50.
- Mahler SA, et al. Safely identifying emergency department patients with acute chest pain for early discharge. Circulation 2018;138:2456-2468.
- Hyams JM, et al. Impact of the HEART pathway on admission rates for emergency department patients with chest pain: An external clinical validation study. J Emerg Med 2018;54:549-557.
- Oliver JJ, et al. An external validation of the HEART pathway among emergency department patients with chest pain. Intern Emerg Med 2018;13:1249-1255.
- Sharp AL, et al. Improving emergency department care for low-risk chest pain. NEJM Catal 2018;2018. [Epub 2018 Apr 18].
- Smulowitz PB, et al. Impact of implementation of the HEART pathway using an electronic clinical decision support tool in a community hospital setting. Am J Emerg Med 2018;36:408-413.
- de Lemos JA, et al. Association of troponin T detected with a highly sensitive assay and cardiac structure and mortality risk in the general population. JAMA 2010;304:2503-2512.
- Hamm CW, et al. ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: The Task Force for the management of acute coronary syndromes (ACS) in patients presenting without persistent ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J 2011;32:2999-3054.
- Wu WK, et al. Documentation of HEART score discordance between emergency physician and cardiologist evaluations of ED patients with chest pain. Am J Emerg Med 2017;35:132-135.
- McCord J, et al. Prognostic utility of a modified HEART score in chest pain patients in the emergency department. Circ Cardiovasc Qual Outcomes 2017;10. doi: 10.1161/CIRCOUTCOMES.116.003101.
- Mark DG, et al. Performance of coronary risk scores among patients with chest pain in the emergency department. J Am Coll Cardiol 2018;71:606-616.
- Than M, et al. Development and validation of the Emergency Department Assessment of Chest pain Score and 2 h accelerated diagnostic protocol. Emerg Med Australas 2014;26:34-44.
- Flaws D, et al. External validation of the emergency department assessment of chest pain score accelerated diagnostic pathway (EDACS-ADP). Emerg Med J 2016;33:618-625.
- Kontos MC. Poll Results: Ultra-High-Sensitivity Troponin Assays. American College of Cardiology. Oct. 17, 2018. Available at: https://www.acc.org/latest-in-cardiology/articles/2018/10/17/11/21/poll-results-ultra-high-sensitivity-troponin-assays. Accessed Sept. 8, 2019.
- Mueller C, et al. Multicenter evaluation of a 0-hour/1-hour algorithm in the diagnosis of myocardial infarction with high-sensitivity cardiac troponin T. Ann Emerg Med 2016;68:76-87.e4.
- Reichlin T, et al. One-hour rule-out and rule-in of acute myocardial infarction using high-sensitivity cardiac troponin T. Arch Intern Med 2012;172:1211-1218.
- Reichlin T, et al. Prospective validation of a 1-hour algorithm to rule-out and rule-in acute myocardial infarction using a high-sensitivity cardiac troponin T assay. CMAJ 2015;187:E243-E252.
- Peacock WF, et al. Efficacy of high-sensitivity troponin T in identifying very-low-risk patients with possible acute coronary syndrome. JAMA Cardiol 2018;3:104-111.
- Chapman AR, et al. Association of high-sensitivity cardiac troponin I concentration with cardiac outcomes in patients with suspected acute coronary syndrome. JAMA 2017;318:1913-1924.
- Rubini Gimenez M, et al. One-hour rule-in and rule-out of acute myocardial infarction using high-sensitivity cardiac troponin I. Am J Med 2015;128:861-870.e4.
- Santi L, et al. The HEART score with high-sensitive troponin T at presentation: Ruling out patients with chest pain in the emergency room. Intern Emerg Med 2017;12:357-364.
- Smulders MW, et al. Acute chest pain in the high-sensitivity cardiac troponin era: A changing role for noninvasive imaging? Am Heart J 2016;177:102-111.
- Mokhtari A, et al. A 1-h combination algorithm allows fast rule-out and rule-in of major adverse cardiac events. J Am Coll Cardiol 2016;67:1531-1540.
- Mahler SA, et al. Use of the HEART Pathway with high sensitivity cardiac troponins: A secondary analysis. Clin Biochem 2017;50:401-407.
- Hess EP, et al. The chest pain choice decision aid: A randomized trial. Circ Cardiovasc Qual Outcomes 2012;5:251-259.
- Rybicki FJ, et al. 2015 ACR/ACC/AHA/AATS/ACEP/ASNC/NASCI/SAEM/SCCT/SCMR/SCPC/SNMMI/STR/STS appropriate utilization of cardiovascular imaging in emergency department patients with chest pain: A joint document of the American College of Radiology Appropriateness Criteria Committee and the American College of Cardiology Appropriate Use Criteria Task Force. J Am Coll Cardiol 2016;67:853-879.
- Hoffmann U, et al. Coronary CT angiography versus standard evaluation in acute chest pain. N Engl J Med 2012;367:299-308.
- Udelson JE, et al. Myocardial perfusion imaging for evaluation and triage of patients with suspected acute cardiac ischemia: A randomized controlled trial. JAMA 2002;288:2693-2700.
- Schenker MP, et al. Interrelation of coronary calcification, myocardial ischemia, and outcomes in patients with intermediate likelihood of coronary artery disease: A combined positron emission tomography/computed tomography study. Circulation 2008;117:1693-1700.
- Janne d’Othee B, et al. A systematic review on diagnostic accuracy of CT-based detection of significant coronary artery disease. Eur J Radiol 2008;65:449-461.
- SCOT-HEART Investigators, et al. Coronary CT angiography and 5-year risk of myocardial infarction. N Engl J Med 2018;379:924-933.
- Goldstein JA, et al; CT-STAT Investigators. The CT-STAT (Coronary Computed Tomographic Angiography for Systematic Triage of Acute Chest Pain Patients to Treatment) trial. J Am Coll Cardiol 2011;58:1414-1422.
- Schlett CL, et al. Prognostic value of CT angiography for major adverse cardiac events in patients with acute chest pain from the emergency department: 2-year outcomes of the ROMICAT trial. JACC Cardiovasc Imaging 2011;4:481-491.
- Amsterdam EA, et al. Testing of low-risk patients presenting to the emergency department with chest pain: A scientific statement from the American Heart Association. Circulation 2010;122:1756-1776.
- Nielsen LH, et al. The diagnostic accuracy and outcomes after coronary computed tomography angiography vs. conventional functional testing in patients with stable angina pectoris: A systematic review and meta-analysis. Eur Heart J Cardiovasc Imaging 2014;15:961-971.
- Taylor AJ, et al. ACCF/SCCT/ACR/AHA/ASE/ASNC/NASCI/SCAI/SCMR 2010 Appropriate Use Criteria for Cardiac Computed Tomography. J Cardiovasc Comput Tomogr 2010;4:407.e1-33.
The workup of suspected acute coronary syndrome in the emergency department is an ever-evolving process, and staying up-to-date can be difficult. This review aims to empower providers to maximize diagnostic precision in a patient-centered and resource-conscious way.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
