Postpartum Emergencies
October 15, 2019
Reprints
AUTHORS
Catherine A. Marco, MD, Professor, Emergency Medicine and Surgery, Wright State University, Boonshoft School of Medicine, Dayton, OH
Kelli Thomas, MD, Resident, Department of Emergency Medicine, Wright State University, Boonshoft School of Medicine, Dayton, OH
Walter Rzecznik, BS, Northeast Ohio Medical University (NEOMED), Rootstown, OH
PEER REVIEWER
Steven Winograd, MD, FACEP, Attending Physician, Mt. Sinai Queens Hospital Center, Assistant Clinical Professor of Emergency Medicine, Mt. Sinai Medical School, Jamaica Queens, NY
EXECUTIVE SUMMARY
• Preeclampsia and eclampsia may occur after the birth of the child. The risk for these disorders lasts for several months
postpartum.
• Treatment of severe preeclampsia and eclampsia in the postpartum period is the same as during pregnancy, with magnesium as the first-line drug.
• Postpartum hemorrhage occurs most commonly immediately after delivery, but can occur up to 12 weeks after delivery.
• Severe hemorrhage may require massive transfusion with blood and clotting factors. Desmopressin and tranexamic acid have been used in severe cases. Postpartum hemorrhage is caused most often by uterine atony.
• Breastfeeding may be complicated by mastitis or breast abscess. While the two may look similar on physical exam, ultrasound can differentiate them rapidly.
Introduction
Postpartum emergencies may include a variety of clinical presentations, ranging from minor concerns to life-threatening emergencies. Problems of pregnancy comprise 1.3% of emergency department (ED) visits annually.1 About 25% of postpartum patients with pregnancy complications seek ED care within the six months following delivery.2 Among postpartum patients, about 1% will require readmission.3 The most common ED complaints include obstetric wound complication, fever, abdominal pain, breast complications, and hypertension. Common postpartum emergencies include pain, fever, hemorrhage, hypertension, preeclampsia, eclampsia, infection, and depression. ED management should include a thorough history, including date and route of delivery, procedural complications, pregnancy history, and current symptoms. The physical exam should include vital signs, lung, cardiac, and abdominal examinations. Diagnostic studies should be ordered to address the specific emergency, and may include focused assessment with sonography in trauma (FAST) examination or pelvic ultrasound. Management should include initial stabilization, followed by disease-specific treatment. Among ED visits for postpartum complications, approximately 22% will require readmission.
Postpartum Pain
Pain and fatigue are the most commonly reported complaints among women in the early postpartum period.4 It has been reported that 90% of women experience postpartum pain that interferes with childcare and other activities of daily living.5 Effective pain management is crucial for both mother and child. Untreated pain has been associated with a greater amount of opioid use, depression, and development of chronic pain. Common types of postpartum pain include the perineum, breasts, pelvic/uterine areas, incision, and headache.
Following a vaginal delivery, 73% of primiparas and 37% of multiparas reported significant perineal pain. Incisional pain after cesarean delivery was the most common complaint of women up to two months postpartum.6 Development of chronic pain following vaginal or cesarean delivery occurs at a rate of about 10%.7 Women with nausea or nausea with vomiting during pregnancy are at higher risk for experiencing pelvic girdle pain four to six months postpartum compared to symptom-free mothers. Women who start with a higher prepregnancy body mass index (BMI), complete less than 12 years of education, and have a longer time between menarche and pregnancy experience more pelvic girdle pain on average.8 Perineal trauma in assisted vaginal delivery, episiotomy, and obstetric lacerations are associated with more pain and higher need for narcotic pain medication. These are risk factors for the development of significant postpartum pain. Nearly 85% of vaginal deliveries are reported to have documented perineal or vaginal trauma, with 60-70% of these requiring surgical repair.9
The causes of postpartum pain are extensive and may be related to uterine involution, uterine contractions, perineal/vaginal trauma, breast engorgement, hemorrhoids, hematoma, wound infection, uterine infection, and venous thrombosis. Chronic pain following birth may develop from dysregulation of inflammatory pathways or neuropathic pain induced by vaginal birth trauma or surgical manipulation.
Uterine involution occurs two to four days postpartum and consists of spontaneous uterine contractions as it returns to its nonpregnant state. When the uterus is more distended, such as with a multiple gestation pregnancy or polyhydramnios, this may be considerably uncomfortable. Breastfeeding increases endogenous oxytocin during lactation.
Postoperative pain after cesarean delivery occurs due to direct trauma to tissue, including nerve endings, and the development of an acute inflammatory response. Hematoma, wound infection, uterine infection, and venous thrombosis also should be considered as etiologies for postpartum pain in the appropriate clinical setting. About 11% of patients have wound infections after cesarean delivery.6 Headache in the postpartum period may be related to preeclampsia, hypercoagulability, or post-dural puncture, but most are commonly due to primary headache conditions.10 Postpartum lumbar back pain may be musculoskeletal in nature or related to the epidural insertion site.11
In patients presenting with postpartum abdominal pain, consider the following: endometritis, urinary tract infection, ovarian rupture, bladder rupture, and mesenteric vein thrombosis. Ovarian vein thrombosis can occur on either ovary and may present with lower right or left quadrant abdominal/pelvic pain. Patients with preeclampsia may have right upper quadrant (RUQ) pain due to liver inflammation. Patients presenting with perineal pain should be evaluated for vaginal/perineal laceration, hematoma, and local tissue infection. Rectal pain after birth may be caused by hemorrhoids due to increased intra-abdominal pressure during active labor. Patients may have incisional pain after cesarean delivery; however, infection, hematoma, and evisceration should be considered.
Patients with chest pain, shortness of breath, or leg swelling should be evaluated for venous thrombosis, including deep venous thrombosis and pulmonary embolism. In patients with headache, the differential diagnosis should include primary headache disorders in addition to subarachnoid hemorrhage, idiopathic intracranial hypertensions, dural venous sinus thrombosis, preeclampsia, meningitis, pituitary apoplexy, and post dural-puncture headache.
After exclusion of serious illness, the focus may turn to pain management. The World Health Organization’s (WHO) analgesic ladder uses a three-tiered approach to medication-induced analgesia for patients with postpartum pain. This includes initiation with non-opioid analgesia, including nonsteroidal anti-inflammatory drugs (NSAIDs) or acetaminophen before initiation of lower morphine-equivalents, such as codeine, hydrocodone, oxycodone, or oral morphine. Stronger opioids, such as parenteral morphine, hydromorphone, and fentanyl, should be reserved for patients who do not receive relief from the lower-tier medications.
Postpartum patients who are breastfeeding may express concern regarding the safety of analgesic medications. NSAIDs, including ketorolac, are found in minimal concentrations in breast milk, are considered safe, and are the preferred first-line agent for postpartum pain. However, opioids are found in much larger concentrations in breast milk, and medications containing codeine or tramadol are not recommended for patients who are breastfeeding. Shared decision-making with the patient should include discussion of the risk-benefit ratio, including signs of opioid toxicity in the infant. Opioid prescriptions should be limited to the least quantity possible.
Breast pain may be related to nipple trauma, inappropriate latch of the baby, mastitis, or clogged ducts. Breast pain from milk engorgement of the non-breastfeeding mother can be managed with cool compresses and compressive bras. Nipple trauma can be treated with breast shields or topical lanolin.
Uterine cramping and perineal pain may be reduced by applying ice packs or cold gel packs to the affected area in addition to using pharmacologic analgesics. Hemorrhoids may be treated with topical steroids or anesthetic creams. Exercise and physical therapy have been shown to improve postpartum pelvic girdle and low back pain.12 Effective postpartum pain management encompasses both pharmacologic and nonpharmacologic components.
Postpartum Hemorrhage
Postpartum hemorrhage is defined as blood loss of 1,000 mL or more or signs or symptoms of hypovolemia within 24 hours of delivery.13 Secondary postpartum hemorrhage refers to excessive bleeding between one day and 12 weeks following delivery. Hemorrhage is the leading cause of obstetric postpartum maternal death worldwide.14 Often in the ED or prior to patient arrival, it is difficult to estimate blood loss. Underestimation of peripartum blood loss may result in delayed diagnosis and treatment. Overestimation of blood loss may lead to unnecessary and potentially harmful therapies.15 Hemorrhage after delivery may be identified by estimated or measured qualitative blood loss in conjunction with classification of hypovolemic shock.
Obstetric hemorrhage is the leading cause of maternal morbidity and mortality in the world, killing more pregnant women than any other medical condition.16 Severe hemorrhage is correlated to significant morbidity including end-organ dysfunction and pituitary necrosis seen in Sheehan syndrome. The incidence of postpartum hemorrhage with significant morbidity and mortality is increasing in the United States, partially because of the trend of advanced maternal age and obesity. Although the prevalence of postpartum hemorrhage is on the rise, it is considered to be largely preventable.
The causes of postpartum hemorrhage can be recalled as the four Ts, which include tone, tissue, trauma, and thrombin. Tone refers to the muscular contraction of the uterus following birth, or the absence of it, as in uterine atony. Uterine atony is the most common cause of postpartum hemorrhage and accounts for 70-80% of cases.17,18 Tissue encompasses placental pathology, such as retained placenta and abnormal implantation in placenta percreta, accreta, and increta. Trauma to the uterus or cervix, uterine inversion, or birth canal lacerations can contribute to postpartum bleeding. Thrombin refers to coagulation disorders and thrombin dysfunction.
Recognition of abnormal postpartum bleeding is critical in addition to recognition of symptoms of acute anemia, including tachycardia, hypotension, altered mental status, syncope, lightheadedness, chest pain, or shortness of breath. Estimated blood loss of
1,000 mL or more, or signs of hemodynamic instability or symptomatic acute blood loss anemia within 24 hours of vaginal delivery is diagnostic of postpartum hemorrhage.19 The physical exam should be focused on identifying vital sign abnormalities in addition to comprehensive abdominal, perineal, and internal examination.
Initial therapy should include resuscitation with intravenous (IV) crystalloids and blood transfusion, if indicated. If uterine atony is suspected, uterine massage should be performed. To perform bimanual uterine massage, place a fist in the anterior fornix and compress the uterine fundus against a hand in the suprapubic region. Additional medical therapy should be undertaken in consultation with obstetrics, and may include an agent such as oxytocin. (See Table 1.)
Table 1. Dosing and Use of Uterotonics for Postpartum Hemorrhage |
||
|
Medication |
Dose |
Cautions |
|
Oxytocin |
10 units IV/IM bolus or 20-40 units in 1 L crystalloid |
Concentrated IV boluses can cause hypotension, high doses may lead to hyponatremia |
|
Methylergonovine (Methergine) |
0.2 mg IM every two to four hours |
Avoid in hypertension |
|
Carboprost (Hemabate) |
250 mcg IM every 15 minutes up to eight doses |
May cause bronchospasm, nausea, vomiting, diarrhea, hypertension |
|
Misoprostol |
1 mg per rectum or orally |
Side effects may include nausea, vomiting, diarrhea |
|
IV, intravenous; IM, intramuscular |
||
Blood product administration is fundamental in resuscitation of active bleeding and hemorrhagic shock. When bleeding is severe and several units of packed red blood cells (pRBCs) will not be adequate, initiate the institution’s massive transfusion protocol (MTP) to alert the blood bank. Massive transfusion is defined as the administration of 10 units or more of packed red blood cells in 24 hours. Administration of pRBCs in this quantity causes dilution of coagulation factors, contributes to the inability to form adequate clots, and significantly diminishes the likelihood of hemostasis. MTP often calls for administration of 1:1:1 ratio of pRBCs to platelets to cryoprecipitate.
Coagulopathy may be suspected or encountered after dilution of natural clotting factors. Adjuncts to blood product administration target activation of the clotting cascade to promote clot formation. Fibrinogen is the first clotting factor to deplete in acute blood loss, and low levels may correlate to the severity of bleeding. Fibrinogen usually is replaced via cryoprecipitate with 150-350 mg in each unit of cryoprecipitate. Guidelines recommend maintenance of the fibrinogen level of greater than 200 mg/dL in patients with active bleeding and initiation of massive transfusion protocol. Each unit of cryoprecipitate will increase serum fibrinogen by about 10 mg/dL.20 Desmopressin may be used to increase secretion of von Willebrand factor from endothelial cells, improving platelet function. The usual IV dose is 0.3 mcg/kg (max 25-30 mcg/kg), given over 25 to 30 minutes.20 Important side effects of desmopressin to consider are hyponatremia and tachyphylaxis.
Tranexamic acid (TXA) has been studied in various settings of acute blood loss and hemorrhagic shock, in trauma surgery literature. It binds to plasmin and plasminogen, inhibiting fibrin degradation. Administration of 1 g IV bolus followed by 1 g over the next eight hours is associated with decreased bleeding rates and reduced need for surgical intervention for postpartum bleeding. This benefit has been noted to diminish when medication administration is delayed for more than three hours following delivery. Early use of TXA is associated with reduced maternal mortality.21 Use of TXA does not increase the risk of thrombotic complications. Data are limited, but there have been no adverse neonatal events described with the use of TXA during childbirth, although it is known to cross the placenta. TXA is cleared renally and is contraindicated in patients with severe kidney disease. Concentrations of higher doses may be related to thrombotic complications and seizures.22
Other blood product adjuncts, such as prothrombin complex concentrates and recombinant-activated factor VII, are recommended only in specific circumstances because of their undesirable safety profiles. Thromboelastography and thromboelastometry are viscoelastic tests that may be used to help guide blood production administration therapy. Their benefit has been proven in reduction of traumatic mortality, but research is not yet available for their specific use in postpartum hemorrhage. Use of these tests may depend on institution availability and provider comfort with interpretation.
Examination of the external genitalia, the vaginal vault and walls, and the cervix is required to detect delivery-associated lacerations that may contribute to postpartum hemorrhage. Lacerations should be repaired in cephalocaudal fashion with synthetic, absorbable suture.23 Gel foam or other absorbent hemostatic packing may be used to help control bleeding. Antibiotic prophylaxis may be considered for higher-grade lacerations. To exclude cervical lacerations, two pairs of ring forceps may be used to expose the tissue circumferentially. If lacerations are suspected and cannot be repaired, vaginal packing may be used until definite repair can be completed. Vaginal packing also may be considered following large laceration repairs to help prevent hematoma formation.
Retained placenta occurs in 2-3% of vaginal deliveries and can cause postpartum hemorrhage and/or contribute to uterine atony. Bedside ultrasound or manual exploration may be used to identify retained products. If bimanual massage is unsuccessful, curettage and suction may be required.
Uterine inversion occurs when the fundus of the uterus prolapses through the cervix and contributes to postpartum hemorrhage when present. If the uterine fundus is nonpalpable on exam, this diagnosis must be considered. Once identified, the uterine fundus should be returned to its proper anatomic position as soon as possible using a closed fist. If the uterus remains inverted, edema causes incarceration and reduces the chances of successful repositioning. Surgical exploration is required beyond repositioning maneuvers with emergent obstetrical consultation.
In any patient with postpartum hemorrhage, uterine rupture must be considered. The diagnosis can be made by physical examination during bimanual exploration of the uterus. Bedside ultrasound also may be used to identify free abdominal fluid or complex pelvic collections related to uterine rupture. Definitive obstetrical surgical management is indicated.
Other uterine bleeding following vaginal delivery may be managed with balloon tamponade until definitive surgical management is available. Mechanical uterine tamponade may be a viable option for the hemodynamically stable patient with uterine atony unresponsive to medical management. Intrauterine balloons can be placed under direct visualization with speculum exam or manually. Ultrasound may be used to confirm intrauterine placement. Balloon tamponade is reported to be effective for control of atonic uterine bleeding in up to 80% of cases. Other hemodynamically stable patients may be candidates for uterine artery embolization when available; however, unstable patients should undergo emergent hysterectomy.
Postpartum patients should routinely be admitted to obstetrical service for monitoring and management. In the setting of significant postpartum hemorrhage, patients may require intensive care admission and continued resuscitation.
Postpartum Hypertension, Preeclampsia, and Eclampsia
Hypertension during pregnancy can be divided into four categories: preeclampsia, chronic hypertension, chronic hypertension with superimposed preeclampsia, and gestational hypertension. Hypertension complicating pregnancy is defined as systolic blood pressure of 140 mmHg or greater and/or diastolic blood pressure of 90 mmHg or greater on two or more occasions at least four hours apart. Preeclampsia is defined as hypertension combined with proteinuria, or end organ damage (thrombocytopenia, impaired liver function, renal insufficiency, pulmonary edema, or visual or neurological dysfunction).24 Gestational hypertension is defined as blood pressure elevation after 20 weeks of gestation without the above associated findings. Eclampsia is the presence of grand mal seizure or coma with preeclampsia.25 HELLP syndrome (hemolysis, elevated liver enzymes, and low platelet count) is a variant of preeclampsia. Laboratory abnormalities are summarized in Table 2.
Table 2. Laboratory Findings in Preeclampsia |
|
|
Organ Dysfunction |
Laboratory Result |
|
Proteinuria |
|
|
Thrombocytopenia |
|
|
Renal insufficiency |
|
|
Liver dysfunction |
|
Hypertension may exist in the postnatal period as preeclampsia or eclampsia causing significant morbidity and mortality, including postpartum depression, seizures, HELLP syndrome, pulmonary edema, renal failure, and future cardiovascular disease such as peripartum cardiomyopathy.
Hypertension in pregnancy is one of the components of the deadly triad of maternal mortality, grouped with hemorrhage and sepsis.26 The spectrum of hypertension affecting pregnancy may present up to six weeks after delivery.27 Hypertension in pregnancy is reported to affect up to 10% of pregnancies, and 3-5% of these pregnancies present with preeclampsia. Postpartum hypertension may complicate about 2% of pregnancies.28 Forty-four percent to 50% of seizures associated with eclampsia occur in the postpartum period; therefore, it has been reported that eclampsia may be most common after birth. Nearly half of the seizures are noted to occur beyond 48 hours after delivery, so the majority of new mothers will already be discharged home. Half of women with preeclampsia who sustain intracranial hemorrhage develop this in the postpartum period.29
Research regarding the etiology and pathophysiology of preeclampsia is ongoing, but the leading theory involves abnormal placentation. The normal placenta is generated with low-resistance, high-flow vascularity. In preeclampsia, abnormal cytotrophoblast invasion and deficient spiral artery remodeling cause narrower arterioles and considerably higher resistance. Hypertension ensues to compensate for this increased resistance. Placental ischemia and endothelial injury cause oxidative stress and angiogenic factor imbalances that are believed to cause severe features of preeclampsia.30 Genetic factors may contribute to the development of preeclampsia, since a twofold to fourfold increase in risk has been noted in women with a primary relative diagnosed with preeclampsia.
Recent literature has proposed primary involvement of the cardiovascular system in the development of preeclampsia. Forty percent of echocardiograms of patients with preeclampsia have shown global diastolic dysfunction, compared to 14% of controls. This myocardial dysfunction is due to the compensatory remodeling in response to increased afterload of hypertension. In combination with endothelial activation causing extravascular pooling of fluid, cardiac insufficiency leads to a predisposition to develop acute pulmonary edema. The duration of the disease has been correlated directly to the severity of cardiovascular dysfunction.31
Advanced maternal age, obesity, ethnicity, diabetes mellitus, chronic hypertension, hyperlipidemia, primiparity, multiple gestation pregnancy, in vitro fertilization, systemic lupus erythematosus, and other thrombophilia disorders have been identified as risk factors for preeclampsia development.
The clinical presentation of preeclampsia is variable, and the majority of patients are asymptomatic. Symptoms suggest that the disease is severe. Pulmonary edema may present with shortness of breath, dyspnea, cough, or chest pain. Oxygen saturation at presentation and 48 hours after may be used to predict adverse maternal outcomes. Because of extravascular pooling of fluid due to low oncotic pressure, women may develop peripheral edema. As cardiac demand increases, there is the potential for development of myocardial infarction. Patients may present with moderate to severe right upper quadrant or epigastric pain and tenderness due to liver involvement. Patients also may present with a chief complaint of nausea and vomiting.
Neurologic involvement is believed to be due to cerebrovascular vasospasm or the inability of cerebral blood flow to be regulated appropriately in response to a sudden spike in blood pressure. Patients may report headache with or without associated visual changes, such as scotomas, blurred vision, or diplopia. Complete blindness is rare but has been reported in severe cases of preeclampsia. Patients with cerebral edema may present with lethargy, confusion, or coma. Convulsions or seizures are diagnostic for eclampsia.
Postpartum patients are at increased risk of intracranial hemorrhage. Presenting symptoms may include severe headache, altered mental status, focal neurologic signs, or severe headache in the setting of hypertension.32 Computed tomography (CT) scan is the initial modality of choice for patients when there is concern for intracranial hemorrhage. If subarachnoid hemorrhage is found, CT angiography or MRI angiography is indicated.
Physical examination of the hypertensive postpartum patient should include a complete cardiorespiratory examination to evaluate for cardiomyopathy and/or pulmonary edema. A fundoscopic examination may reveal papilledema, retinal hemorrhage, or retinal edema. Further neurologic assessment may reveal hyperreflexia with or without clonus due to neurologic excitability. Abdominal examination may reveal right upper quadrant or epigastric tenderness to palpation.
Patients presenting to the ED with signs or symptoms concerning for preeclampsia may have been undergoing preventive therapy. Although no intervention to date has been proven effective for the prevention of preeclampsia, it is beneficial to be aware of the possible prevention strategies. Use of antiplatelet agents, such a low-dose aspirin (81 mg), as anti-inflammatory agents has mixed results in effective prevention, but they have not been associated with significant risk, and may be used in women at high risk of developing preeclampsia. Low-dose aspirin in high-risk women is recommended starting in the first trimester. Other preventive strategies, such as vitamin C and E supplementation, dietary salt intake restriction, and other nutritional interventions, are not recommended. Bed rest or restriction of physical activity is not recommended for the primary prevention of preeclampsia.
Classically, hypertension with proteinuria is diagnostic for preeclampsia. Proteinuria is defined as 300 mg or more on 24-hour urine collection, or a protein-creatinine ratio of 0.3 or greater. In the ED, urinalysis by dipstick with 1+ protein may be suggestive of proteinuria. Spot urine protein to creatinine ratio also may be used as an alternative to the classic 24-hour urine collection. Urine protein (Up)/urine creatinine (Ucr) ratio greater than 0.3 g/gcr is diagnostic for proteinuria. Amniotomy and local tissue edema may affect the accuracy of clean catch urine samples, falsely elevating the protein content in the urine. It may be prudent to obtain specimens via straight catheterization to avoid erroneous results.33 Preeclampsia also may be diagnosed as hypertension with other laboratory findings summarized in Table 3.
Table 3. Definitions of Preeclampsia |
|
Mild Preeclampsia |
|
|
Severe Preeclampsia |
|
HELLP syndrome is a further subset of preeclampsia in which hypertension is combined with hemolysis, elevated liver enzymes, and low platelet count. (See Table 4.) To clinically differentiate HELLP from thrombocytopenic purpura, lactate dehydrogenase may be obtained. Eclampsia may be diagnosed when new-onset grand mal seizures are present in a woman with preeclampsia.
Table 4. Laboratory Findings in HELLP Syndrome |
|
|
Laboratory Test |
Results |
|
Platelet count |
< 100,000/μL |
|
Liver function tests |
Moderate elevation (typically < 500 U/L) |
|
Bilirubin |
> 1.2 mg/dL |
|
Renal function tests |
Normal or elevated BUN and creatinine |
|
Peripheral smear |
Schistocytes |
|
Coagulation profile |
Abnormal |
|
Lactate dehydrogenase |
> 600 U/L suspicious for hemolytic anemia |
|
HELLP, hemolysis, elevated liver enzymes, low platelet count; BUN, blood urea nitrogen |
|
Asymptomatic hypertension in the pregnant or postpartum patient may be related to preeclampsia, but other diagnoses should be considered. Blood pressure measurement should be taken with the patient in the appropriate position: seated with the legs uncrossed and the back and arm supported to bring the arm to the level of the right atrium. Ideally, the patient should remain in this position for five minutes prior to initial blood pressure assessment. It is important to use an adequately sized blood pressure cuff. When an accurate blood pressure measurement reading is elevated, further evaluation is required.
Medications commonly used in the postpartum period that are associated with elevated blood pressure include NSAIDs, decongestants, and ergot alkaloids, such as methylergonovine. Secondary causes of hypertension should be considered, as renal parenchymal disease, thyroid dysfunction, fibromuscular dysplasia, pheochromocytoma, Cushing’s syndrome, or hyperaldosteronism may be an underlying condition.34
Symptoms associated with preeclampsia are vague and generally nonspecific, leaving a large differential diagnosis to consider. For presentations of hematologic symptoms, such as bleeding, coagulation abnormality, disseminated intravascular coagulation (DIC), or shock, the differentials considered should include idiopathic thrombocytopenic purpura, thrombotic thrombocytopenic purpura, or septic shock. The differential diagnosis of visual disturbances includes retinal detachment, retinal artery or vein thrombosis, trauma, retinal ischemia, or central serous retinopathy. Right upper quadrant or epigastric pain, nausea and vomiting, and abnormal liver function tests may be due to viral hepatitis, drug-induced hepatotoxicity, acute pancreatitis, cholestasis, or gastritis. Patients who present with chest pain, shortness of breath, or hypoxia may have pulmonary edema, pulmonary embolism, pneumonia, coronary artery disease, or peripartum cardiomyopathy. Seizure or severe headache may be due to epilepsy, hypoglycemia, hypertensive encephalopathy, central venous sinus thrombosis, amniotic fluid embolism, idiopathic intracranial hypertension, or intracranial hemorrhage.
Therapy for hypertension, preeclampsia, and eclampsia should be started emergently. The use of hydralazine, alpha methyldopa, beta-blockers, and nifedipine is reported extensively in the literature, but no single medication has been proven superior to another. See Table 5 for dosage recommendations for commonly used antihypertensive agents. In the pregnant patient, angiotensin converting-enzyme (ACE) inhibitors, angiotensin receptor blockers, and sodium nitroprusside should be avoided because of safety concerns.35 Management of hypertension in the pregnant patient also may include delivery, depending on the gestational age and severity of maternal disease.
Magnesium sulfate is recommended for preeclampsia with severe features. Some symptoms that may precede seizures include headache, altered mental status, blurred vision, scotomata, clonus, and right upper quadrant pain. Parenteral magnesium is continued for 24 hours after initial diagnosis. A loading dose of magnesium of 4-6 g IV over 15-20 minutes is followed by an infusion of 1-2 g per hour. Vital signs and urine output should be monitored every hour, and deep tendon reflexes should be checked after the loading dose and every two hours during the infusion.
Patients with asymptomatic hypertension without preeclampsia in the postnatal period may be treated as outpatients, with or without administration of antihypertensive medications. (See Table 5.) Close follow-up with an obstetrics specialist should be arranged, and the patient should return to the ED for worsening hypertension or the development of symptoms of preeclampsia, such as leg swelling.
Severe hypertension, preeclampsia, and eclampsia are indications for admission and consultation with obstetrics for blood pressure management and continued parenteral administration of magnesium.
Table 5. Antihypertensive Medications in Pregnancy |
||
|
Antihypertensive |
Oral Dosing |
IV Dosing |
|
Nifedipine |
10-30 mg, max 120 mg/day |
|
|
Hydralazine |
10-50 mg, max 300 mg/day |
5-10 mg bolus, max 45 mg |
|
Labetalol |
300-2,400 mg divided daily |
20 mg bolus followed by 40 mg then 80 mg q10 min max 300 mg |
|
Clonidine |
0.1-0.2 mg, max 2.4 mg/day |
|
|
Captopril (postpartum only) |
6.25-12.5 mg, max 200 mg/day |
|
|
Methyldopa |
500-2,000 mg divided daily, max 3 g daily |
|
Postpartum Infections
Fever is an important sign of infection in the postpartum period. Common etiologies include endometritis, mastitis, pyelonephritis, thrombophlebitis, or upper respiratory infection.36
Endometritis (inflammation of the endometrial layer of the uterus) should be considered in all postpartum patients with fever and abdominal or pelvic pain. The incidence of endometritis is 1-3% following vaginal delivery and 13-90% following cesarean delivery.37 Risk factors for endometritis include cesarean delivery, multiple gestation, young maternal age, long duration of labor, prolonged rupture of membranes, internal fetal monitoring, meconium-stained amniotic fluid, extremes of age, low socioeconomic level, digital examination after 37 weeks of gestation, and maternal human immunodeficiency virus infection. Symptoms may include abdominal pain, pelvic pain, fever, malodorous vaginal discharge, or incisional pain or discharge for post-cesarean delivery patients. Bacterial pathogens typically arise from the genital tract and may include Group B Streptococcus, Enterococcus, Ureaplasma urealyticum, Peptostreptococcus, Bacteroides bivius, Chlamydia trachomatis, Neisseria gonorrhoeae, Gardnerella vaginalis, Mycoplasma hominis, or polymicrobial infection. Patients with mild infection may be treated as outpatients with an oral agent, such as amoxicillin-clavulanic acid 850 mg PO twice a day, clindamycin 300 mg three times a day for 10 days, or doxycycline 100 mg twice a day for 10 days (avoid doxycycline in breastfeeding patients) plus metronidazole 500 mg PO twice daily. Patients requiring inpatient therapy should be treated with IV antibiotics, such as clindamycin (900 mg IV q 8 hours) plus gentamicin (1.5 mg/kg IV q 8 hours).38 Other regimens may include cefoxitin 2 g q 6 hours plus vancomycin 1 g q 12 hours, or cefotetan plus vancomycin, or cefotaxime plus vancomycin.
Mastitis commonly presents with breast pain, malaise, and fever.39 Mastitis is due to breast engorgement, clogged ducts, or incomplete emptying. Common pathogens causing mastitis include Staphylococcus aureus, Escherichia coli, and Streptococcus pyogenes. Ultrasound may be helpful in distinguishing abscess from mastitis. Mastitis will appear as hypoechoic fluid surrounding fat lobules without a discrete fluid collection, while an abscess will appear as a discrete fluid collection. Treatment should include frequent breast emptying (breastfeeding may be continued or a breast pump may be used), analgesia, and antibiotic therapy with an agent such as dicloxacillin 500 mg four times a day for 14 days, cephalexin 500 mg four times a day for 14 days, or clindamycin 300 mg four times a day for 14 days. Trimethoprim-sulfamethoxazole should be avoided in lactating mothers of infants younger than 2 months of age.
Perinatal Depression
Postpartum psychiatric conditions include anxiety, psychosis, depression, and suicidal thoughts.40,41 According to the DSM-5, perinatal depression occurs during pregnancy or in the four weeks following delivery.42 Many experts define postpartum depression as depression that occurs in the first year after delivery.43 An estimated 25% of postpartum women experience depressive symptoms, and approximately 6-13% of postpartum women experience major depression.44-46 Risk factors include single motherhood, poor social support, history of untreated depression or anxiety during pregnancy, low socioeconomic status, unwanted pregnancy, smoking, birth complications, gestational diabetes, hypertension, and stress in the partner relationship.47-50 Perinatal depression is associated with poor maternal-infant bonding, reduced duration of breastfeeding, and maternal suicide risk. Symptoms may include a combination of fatigue, sadness, depressed mood, poor maternal-infant bonding, guilt, anhedonia, sleep or eating disturbance, difficulty concentrating, feelings of worthlessness, or suicidal thoughts.
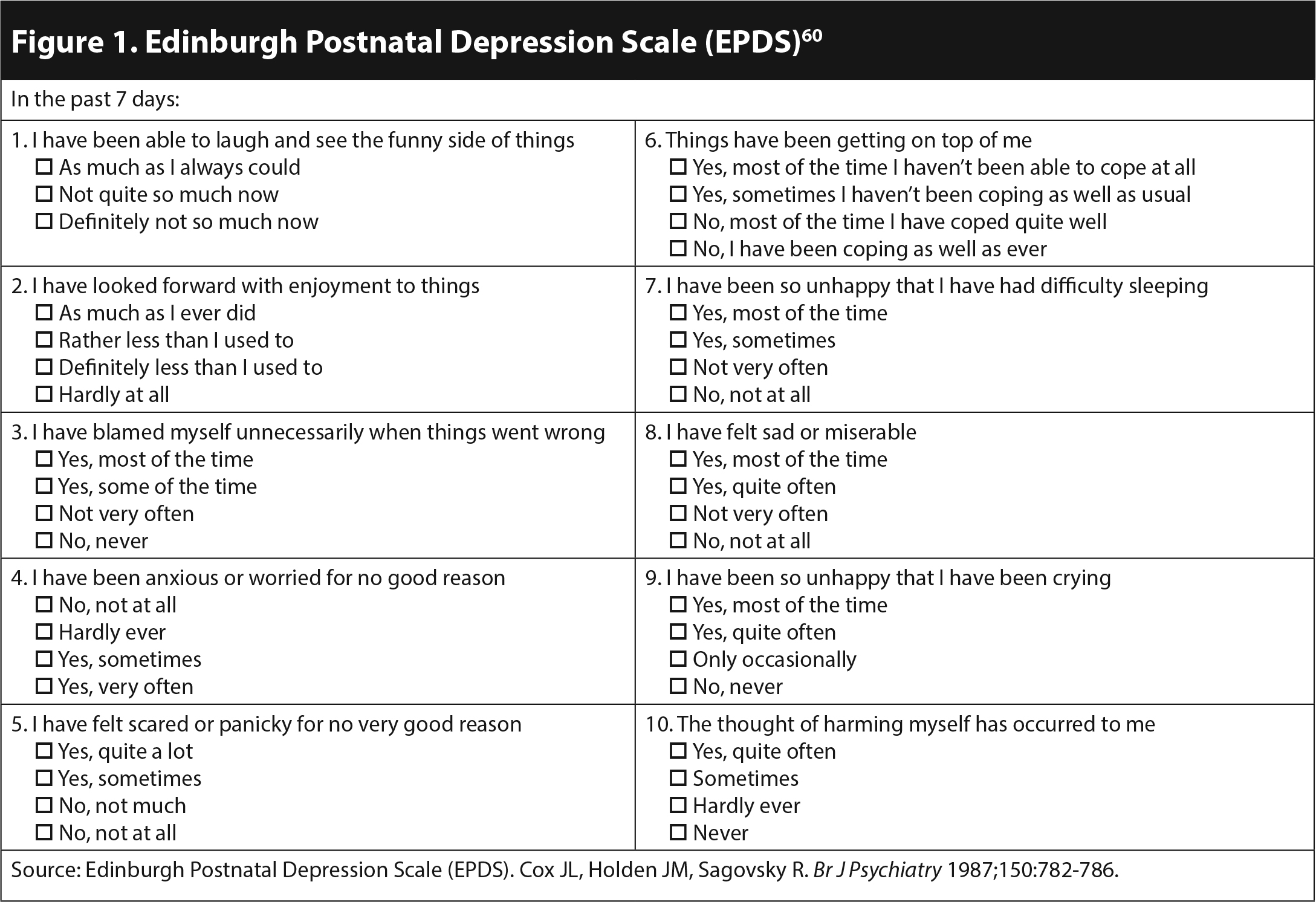
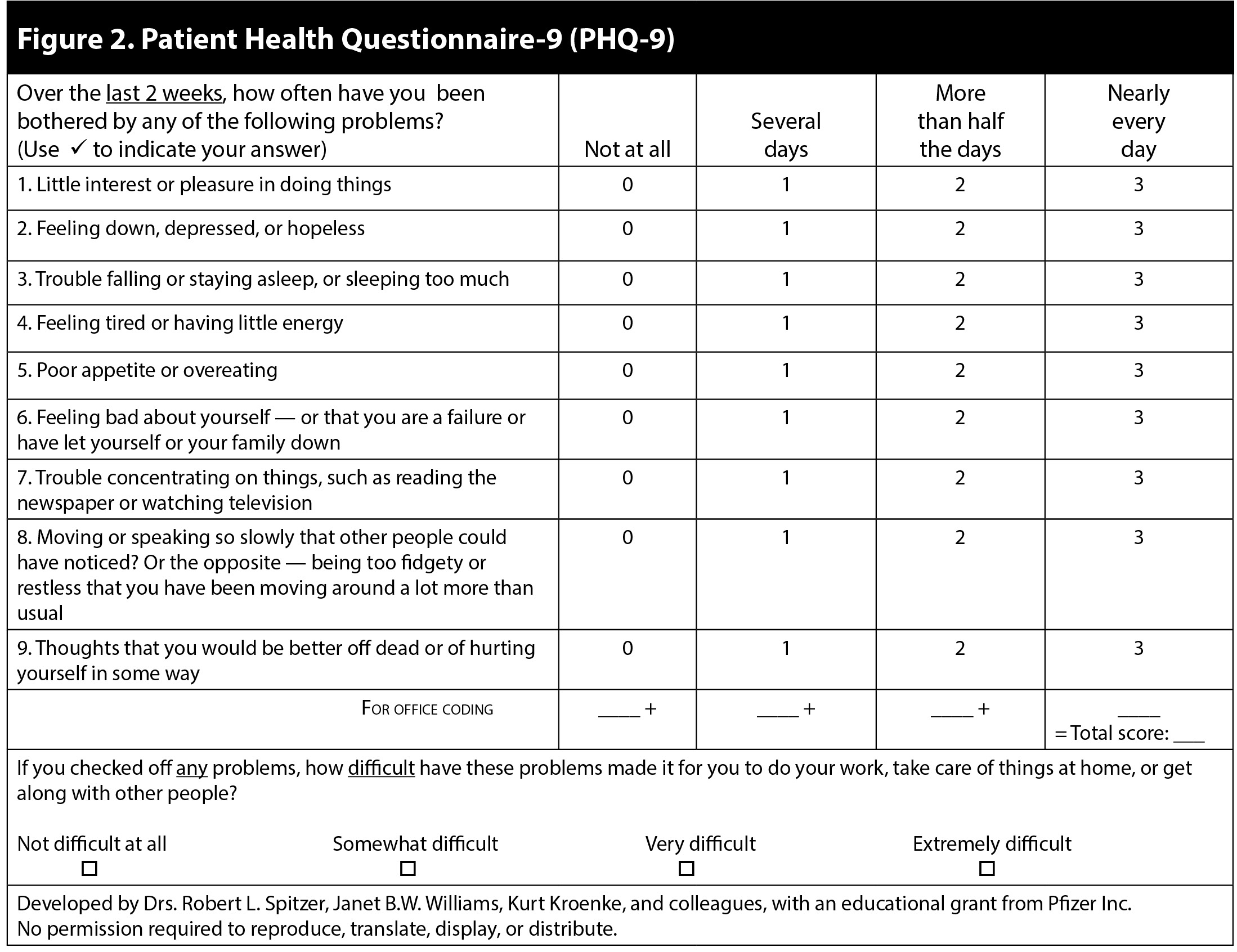
Postpartum women should be screened for depressive symptoms. A screening instrument may be used, such as the Edinburgh Postnatal Depression Scale (EPDS) (see Figure 1), the Patient Health Questionnaire (PHQ-9) (see Figure 2), the Postpartum Depression Screening Scale, or the Beck Depression Inventory-II (BDI-II).53-59
|
|
Identifying suicidal ideation and initiating psychiatric treatment are important. Risk factors for suicidal thoughts include pre-existing suicidality, discontinuation of psychotropic medications, sleep disturbances, stillbirth, and intimate partner violence.62,63 Assessment of suicide risk is essential. All postpartum women should be screened for depression using a standardized, validated tool.64 Several validated tools are available, including PHQ-2, the Patient Safety Screener, the Tool for Assessment of Suicide Risk (TASR) adolescent screener, the Ask Suicide-Screening Questions (ASQ), the Columbia-Suicide Severity Rating Scale (C-SSRS), and the Suicide Assessment Five-step Evaluation and Triage (SAFE-T).65-68
Treatment of perinatal depression may include nonpharmacologic therapy and pharmacologic therapy. Nonpharmacologic therapy may involve cognitive behavior therapy, interpersonal psychotherapy, exercise, behavioral activation, maternal-child interaction guidance, and peer support.69-73 Pharmacologic treatment should be initiated for severe depression or failure to respond to nonpharmacologic interventions. Treatment may be initiated in consultation with a mental health provider. Most commonly, treatment is initiated with a selective serotonin reuptake inhibitor (SSRI), such as sertraline or paroxetine as indicated.74-78
Patients with major depression or suicidal ideation may benefit from inpatient psychiatric treatment. Patients with minor depressive symptoms may be referred for outpatient mental health services.
Conclusion
Patients who present with postpartum complications should undergo a full evaluation, including history of present symptoms, birth history, and physical examination. Laboratory testing should be performed when appropriate. Therapy for the specific condition should be undertaken in consultation with obstetrics. Inpatient therapy is indicated for patients who are unstable, suicidal, or unable to complete outpatient therapy. Patients may be managed appropriately as outpatients if they are able to comply with therapy, care for themselves and their infants, and follow up with obstetrics in a timely fashion.
REFERENCES
- Rui P, Kang K, Ashman JJ. National Hospital Ambulatory Medical Care Survey: 2016 Emergency Department Summary Tables. 2016. Available at: https://www.cdc.gov/nchs/data/nhamcs/web_tables/2016_ed_web_tables.pdf. Accessed Sept. 27, 2019.
- Harris A, Chang HY, Wang L, et al. Emergency room utilization after medically complicated pregnancies: A Medicaid claims analysis. J Womens Health (Larchmt) 2015;24:745-754.
- Brosseau EC, Danilack V, Cai F, Matteson KA. Emergency department visits for postpartum complication. J Women’s Health (Larchmt) 2018;27:253-257.
- [No authors listed]. ACOG Committee Opinion No. 742: Postpartum Pain Management. Obstet Gynecol 2018;132:e35-e43.
- Fahey JO. Best practices in management of postpartum pain. J Perinat Neonatal Nurs 2017;31:126-136.
- Carter EB, Temming LA, Fowler S, et al. Evidence-based bundles and cesarean delivery surgical site infections: A systematic review and meta-analysis. Obstet Gynecol 2017;130:735-746.
- Munro A, George RB, Chorney J, et al. Prevalence and predictors of chronic pain in pregnancy and postpartum. J Obstet Gynaecol Can 2017;39:734-741.
- Chortatos A, Iversen PO, Haugen M, et al. Nausea and vomiting in pregnancy — association with pelvic girdle pain during pregnancy and 4-6 months post-partum. BMC Pregnancy Childbirth 2018;18:137-144.
- Zhang Y, Huang L, Ding Y, et al. Management of perineal pain among postpartum women in an obstetric and gynecological hospital in China: A best practice implementation project. JBI Database System Rev Implement Rep 2017;15:165-177.
- O’Neal MA. Headaches complicating pregnancy and the postpatrum period. Pract Neurol 2017;17:191-202.
- Wilkes D, Martinello C, Medeiros FA, et al. Ultrasound-determined landmarks decrease pressure pain at epidural insertion site in immediate post-partum period. Minerva Anestesiol 2017;83:1034-1041.
- Sakamoto A, Nakagawa H, Nakagawa H, Gamada K. Effect of exercise with a pelvic realignment device on low-back and pelvic girdle pain after childbirth: A randomized control study. J Rehabil Med 2018;50:914-919.
- Menard MK, Main EK, Currigan SM. Executive summary of the reVITALize initiative: Standardizing obstetric data definitions. Obstet Gynecol 2014;124:150-153.
- Khan KS, Wojdyla D, Say L, et al. WHO analysis of causes of maternal death: A systematic review. Lancet 2006;367:1066-1074.
- Andrikopoulou M, D’Alton ME. Postpartum hemorrhage: Early identification challenges. Semin Perinatol 2019;43:11-17.
- Nathan LM. An overview of obstetric hemorrhage. Semin Perinatol 2019;43:2-4.
- Mousa HA, Blum J, Abou El Senoun G, et al. Treatment for primary postpartum haemorrhage. Cochrane Database Syst Rev 2014;(2):CD003249.
- Affronti G, Agostini V, Brizzi A, et al. The daily-practiced post-partum hemorrhage management: An Italian multidisciplinary attended protocol. Clin Ter 2017;168:e307-e316.
- Committee on Practice Bulletins – Obstetrics. Practice Bulletin No. 183: Postpartum Hemorrhage. Obstet Gynecol 2017;130:e168-e186.
- Pacheco LD, Saade GR, Hankins GDV. Medical management of postpartum hemorrhage: An update. Semin Perinatol 2019;43:22-26.
- WOMAN Trial Collaborators. Effect of early tranexamic acid administration on mortality, hysterectomy, and other morbidities in women with post-partum haemorrhage (WOMAN): An international, randomised, double-blind, placebo-controlled trial. Lancet 2017;389:2105-2116.
- Pacheco LD, Hankins GDV, Saad AF, et al. Tranexamic acid for the management of obstetric hemorrhage. Obstet Gynecol 2017;130:765-769.
- Gilmandyar D, Thornburg L. Surgical management of postpartum hemorrhage. Semin Perinatol 2019;43:27-34.
- Mol BWJ, Roberts CT, Thangaratinam S, et al. Pre-eclampsia. Lancet 2016;387:999-1011.
- American College of Obstetricians and Gynecologists: Task Force on Hypertension in Pregnancy. Hypertension in pregnancy. Obstet Gynecol 2013;122:1122-1131.
- Malik R, Kumar V. Hypertension in pregnancy. In: Islam MS. Hypertension: From Basic Research to Clinical Practice. Switzerland: Springer; 2017:375-393.
- Clark SL, Belfort MA, Dildy GA, et al. Emergency department use during the postpartum period: Implications for current management of the puerperium. Am J Obstet Gynecol 2010;203:38.e1-6.
- Sharma KJ, Kilpatrick SJ. Postpartum hypertension: Etiology, diagnosis, and management. Obstet Gynecol Surv 2017;72:248-252.
- Cairns AE, Pealing L, Duffy JMN, et al. Postpartum management of hypertensive disorders of pregnancy: A systematic review. BMJ Open 2017;7:e018696.
- Hariharan N, Shoemaker A, Wagner S. Pathophysiology of hypertension in preeclampsia. Microvasc Res 2017;109:34-37.
- Perry H, Khalil A, Thilaganathan B. Preeclampsia and the cardiovascular system: An update. Trends Cardiovasc Med 2018;28:505-513.
- Fairhall JM, Stoodley MA. Intracranial haemorrhage in pregnancy. Obstet Med 2009;2:142-148.
- Herrera Gonzalez A, Quinn S, Lennon J, et al. Amniotomy affects the urine protein creatinine ratio accuracy in the diagnosis of preeclampsia. Obstet Gynecol 2017;129:32S.
- Charles L, Triscott J, Dobbs B. Secondary hypertension: Discovering the underlying cause. Am Fam Physician 2017;96:453-461.
- World Health Organization. WHO recommendations: Drug treatment for severe hypertension in pregnancy. Geneva: World Health Organization; 2018.
- Belfort MA, Clark SL, Saade GR, et al. Hospital readmission after delivery: Evidence for increased incidence of non-urogenital infection in the immediate postpartum period. Am J Obstet Gynecol 2010;202:35.e1-7.
- Rivlin ME. Endometritis. Medscape. Available at: https://emedicine.medscape.com/article/254169. Accessed Sept. 16, 2019.
- Mackeen AD, Packard RE, Ota E, Speer L. Antibiotic regimens for postpartum endometritis. Cochrane Database Syst Rev 2015;(2):CD001067.
- Berens PD. Breast pain: Engorgement, nipple pain, and mastitis. Clin Obstet Gynecol 2015;58:902-914.
- Rodriguez-Cabezas L, Clark C. Psychiatric emergencies in pregnancy and postpartum. Clin Obstet Gynecol 2018;61:615-627.
- Osborne LM. Recognizing and managing postpartum psychosis: A clinical guide for obstetric providers. Obstet Gynecol Clin North Am 2018;45:455-468.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Publishing; 2013: 5-25.
- Stuart-Parrigon K, Stuart S. Perinatal depression: An update and overview. Curr Psychiatry Rep 2014;16:468.
- Wisner KL, Sit DK, McShea MC, et al. Onset timing, thoughts of self-harm, and diagnoses in postpartum women with screen-positive depression findings. JAMA Psychiatry 2013;70:490-498.
- Gavin NI, Gaynes BN, Lohr KN, et al. Perinatal depression: A systematic review of prevalence and incidence. Obstet Gynecol 2005;106:1071-1083.
- Stewart DE, Vigod S. Postpartum depression. N Engl J Med 2016;375:2177-2186.
- Lancaster CA, Gold KJ, Flynn HA, et al. Risk factors for depressive symptoms during pregnancy: A systematic review. Am J Obstet Gynecol 2010;202:5-14.
- Centers for Disease Control and Prevention. Prevalence of self-reported postpartum depressive symptoms – 17 states, 2004-2005. MMWR Morb Mortal Wkly Rep 2008;57:361.
- Ko JY, Rockhill KM, Tong VT, et al. Trends in postpartum depressive symptoms - 27 states, 2004, 2008, and 2012. MMWR Morb Mortal Wkly Rep 2017;66:153-158.
- Howard MM, Mehta ND, Powrie R. Peripartum depression: Early recognition improves outcomes. Cleve Clin J Med 2017;84:388-396.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Publishing; 2013.
- Wheaton N, Al-Abdullah A, Haertlein T. Late pregnancy and postpartum emergencies. Emerg Med Clin North Am 2019;37:277-286.
- O’Hara MW, Stuart S, Watson D, et al. Brief scales to detect postpartum depression and anxiety symptoms. J Womens Health (Larchmt) 2012;21:1237-1243.
- Petrozzi A, Gagliardi L. Anxious and depressive components of Edinburgh Postnatal Depression Scale in maternal postpartum psychological problems. J Perinat Med 2013;41:343-348.
- Santos IS, Tavares BF, Munhoz TN, et al. Patient Health Questionnaire-9 versus Edinburgh Postnatal Depression Scale in screening for major depressive episodes: A cross-sectional population-based study. BMC Res Notes 2017;10:57.
- Moraes GP, Lorenzo L, Pontes GA, et al. Screening and diagnosing postpartum depression: When and how? Trends Psychiatry Psychother 2017;39:54-61.
- Smith EK, Gopalan P, Glance JB, Azzam PN. Postpartum depression screening: A review for psychiatrists. Harv Rev Psychiatry 2016;24:173-187.
- Learman LE. Screening for depression in pregnancy and the postpartum period. Clinic Obstet Gynecol 2018;61:525-532.
- Bobo WV, Yawn BP. Concise review for physicians and other clinicians: Postpartum depression. Mayo Clin Proc 2014;89:835-844.
- Cox JL, Holden JM, Sagovsky R. Detection of postnatal depression. Development of the 10-item Edinburgh Postnatal Depression Scale. Br J Psychiatry 1987;150:782-786.
- Patient Health Questionnaire-9 (PHQ-9). Available at: http://www.phqscreeners.com/sites/g/files/g10016261/f/201412/PHQ-9_English.pdf. Accessed Sept. 22, 2019.
- Sit D, Luther J, Buysse D, et al. Suicidal ideation in depressed postpartum women: Associations with childhood trauma, sleep disturbance and anxiety. J Psychiatr Res 2015;66-67:95-104.
- Palladino CL, Singh V, Campbell J, et al. Homicide and suicide during the perinatal period: Findings from the National Violent Death Reporting System. Obstet Gynecol 2011;118:1056-1063.
- Committee on Obstetric Practice. The American College of Obstetricians and Gynecologists Committee Opinion no. 630. Screening for perinatal depression. Obstet Gynecol 2015;125:1268-1271.
- Suicide Assessment Five-Step Evaluation and Triage (SAFE-T) Substance Abuse and Mental Health Services Administration. 2009. Available at: https://www.integration.samhsa.gov/images/res/SAFE_T.pdf. Accessed Aug. 31, 2019.
- National Institute of Mental Health. Ask Suicide-Screening Questions (ASQ) Toolkit. Available at: https://www.nimh.nih.gov/research/research-conducted-at-nimh/asq-toolkit-materials/index.shtml. Accessed Aug. 31, 2019.
- Columbia University Medical Center. The Columbia Lighthouse Project. The Columbia Protocol C-SSRS versions including e-screening version. Available at: http://www.cssrs.columbia.edu. Accessed Aug. 31, 2019.
- Milgrom J, Gemmill AW. Screening for perinatal depression. Best Pract Res Clin Obstet Gynaecol 2014;28:13-23.
- O’Connor E, Rossom RC, Henninger M, et al. Primary care screening for and treatment of depression in pregnant and postpartum women. JAMA 2016;315:388-406.
- Letourneau NL, Dennis CL, Cosic N, Linder J. The effect of perinatal depression treatment for mothers on parenting and child development: A systematic review. Depress Anxiety 2017;34:928-966.
- Pritchett RV, Daley AJ, Jolly K. Does aerobic exercise reduce postpartum depressive symptoms? A systematic review and meta-analysis. Br J Gen Pract 2017;67:e684-e691.
- O’Hara MW and Engeldinger J. Treatment of postpartum depression: Recommendations for the clinician. Clin Obstet Gynecol 2018;61:604-614.
- Poyatos-Leon R, Garcia-Hermoso A, Sanabria-Martinez G, et al. Effects of exercise-based interventions on postpartum depression: A meta-analysis of randomized controlled trials. Birth 2017;44:200-208.
- Latendresse G, Elmore C, Deneris A. Selective serotonin reuptake inhibitors as first-line antidepressant therapy for perinatal depression. J Midwifery Womens Health 2017;62:317-328.
- Kimmel MC, Cox E, Schiller C, et al. Pharmacologic treatment of perinatal depression. Obstet Gynecol Clin North Am 2018;45:419-440.
- Alwan S, Friedman JM, Chambers C. Safety of selective serotonin reuptake inhibitors in pregnancy: A review of current evidence. CNS Drugs 2016;30:499-515.
- Susser LC, Sansone SA, Hermann AD. Selective serotonin reuptake inhibitors for depression in pregnancy. Am J Obstet Gynecol 2016;215:722-730.
- Kim DR, Epperson CN, Weiss AR, Wisner KL. Pharmacotherapy of postpartum depression: An update. Expert Opin Pharmacother 2014;15:1223-1234.
Postpartum emergencies may include a variety of clinical presentations, ranging from minor concerns to life-threatening emergencies. Common postpartum emergencies include pain, fever, hemorrhage, hypertension, preeclampsia, eclampsia, infection, and depression.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.