Taming of the Flu: What’s New for 2020
Introduction
As the winter months descend upon the Northern Hemisphere, it is once again time for healthcare providers to focus on the prevention and treatment of the influenza virus. According to the Centers for Disease Control and Prevention (CDC), the 2018-2019 flu season was the longest season in the past 10 years. Influenza-related illnesses began in November 2018, reached a peak in February 2019, and returned to baseline levels in April 2019.1
Overall, the 2018-2019 flu season was moderate in nature, with the influenza A virus causing the majority of illness. Last year’s flu season saw a lower rate of hospitalization for adults and a similar rate of hospitalization for children when compared to the previous season. There were an estimated 37.4 to 42.9 million flu cases in the United States during last year’s flu season, with upward of 20.1 million medical visits, 647,000 hospitalizations, and 61,200 deaths.2
The current flu season has seen a sharp rise in cases when compared with previous flu seasons, but it is unknown whether this is a harbinger of a particularly severe flu season or simply an early spike in flu activity.3 As of week 48 of 2019, 12 states were already reporting higher than normal levels of influenza-like illness activity.4 Of note, the United States currently is seeing a predominance of influenza B cases, which typically are not seen until the latter half of the flu season. This strain is the most commonly identified subtype in age groups 0-4 years and 5-24 years, with influenza A being more common in adults 65 years of age or older.3,4
The 2019-2020 flu season is already among us, and it is imperative that those healthcare practitioners on the frontline have current knowledge of prevention and treatment strategies. Currently available data suggest that nationally, flu activity is high as of Jan. 4, 2020, but severity (hospitalizations and deaths) is not high at this point in the season. Both influenza A and B strains have been identified, and their predominance varies by region and age group.
Background
Influenza is an acute respiratory illness responsible for significant seasonal epidemics each year. The disease is transmitted by the influenza virus, an enveloped RNA virus that is part of the Orthomyxoviridae family. Despite commonly being a self-limited illness, the virus causes significant morbidity and mortality each year.
Influenza A and influenza B are responsible for the majority of clinically significant influenza infections. Influenza A is primarily implicated in significant pandemics that greatly affect public health. This virus is typed according to the antigenic characteristics of envelope glycoproteins, specifically hemagglutinin and neuraminidase. Influenza A has three major subtypes of hemagglutinin (H1, H2, and H3), along with two subtypes of neuraminidase (N1 and N2). Influenza B is not classified by subtype and has not been shown to cause pandemics.
As of Sept. 29, 2019, 10,444 specimens had been tested by public health laboratories, with 2,460 positive specimens. Of the positive specimens, 44.1% of cases have been identified as influenza A and 55.9% as influenza B. Currently, influenza B is more common in children and young adults, while influenza A is the predominant strain in patients older than 65 years of age. For patients 25-64 years of age, both strains have been identified equally.4
Additional subtypes of influenza, C and D, are less commonly associated with human disease. Influenza C produces a self-limited respiratory illness that is more common in children. Influenza D is believed to have originated in cattle and is largely responsible for bovine respiratory disease.5
The burden of influenza on the population each year is significant, with as many as 35.6 million cases in the United States since 2010, resulting in up to 56,000 deaths. Of the 35.6 million cases, as many as 710,000 patients were hospitalized secondary to influenza. The widespread use of the influenza vaccine continues to be the most effective method of prevention.
The CDC estimates that the vaccine has prevented 5.1 million influenza illnesses, 2.5 million influenza-related medical visits, and 71,000 hospitalizations.6
Global Impact
Throughout history, several major global outbreaks of influenza have been documented. These outbreaks are due to the ability of viral cells to change their antigenic structure rapidly. While influenza traditionally is regarded as a human disease entity, it is important to recognize that animals, such as birds and pigs, serve as important reservoirs for viral strains. These unique properties of the virus are responsible for the significant outbreaks that have shaped the history of human health.
While the influenza pandemic of 1918 is perhaps the most well-known of all outbreaks, influenza has affected human history for centuries. The first documented influenza outbreak may have occurred as early as 1510. While it would have been impossible for early historians to know the exact nature of the disease, this illness bore similar traits to the modern influenza virus that is well described today. Historical accounts of a rapid onset fever with respiratory symptoms that occurred via trade routes or in major population centers suggest that influenza epidemics and pandemics have been present throughout the course of human history.7
H1N1 Pandemic, 1918
The H1N1 avian flu virus had a major worldwide impact from 1918 to 1919. Historical estimates suggest that nearly 500 million people across the globe were infected, representing roughly one-third of the entire population. In the United States, the emergence of the virus occurred during the spring of 1918, when military personnel were reported to have flu-like symptoms. Transmission of the virus resulted in the death of 50 million worldwide, with roughly 675,000 deaths in the United States.8,9 In 2005, researchers successfully re-created the virus in an effort to learn more about the emergence of pandemic viruses.10
H2N2 Pandemic, 1957-1958
In February 1957, the H2N2 strain of the influenza A virus resulted in the pandemic known as the “Asian flu.” Located predominantly in East Asia, the virus spread to coastal U.S. cities during the summer of 1957. Approximately 1.1 million deaths were documented worldwide, with close to 116,000 fatalities in the United States.11,12
H3N2 Pandemic, 1968
The H3N2 strain of the influenza A virus was first documented to reach the United States in September 1968. The estimated number of deaths was 1 million worldwide, with roughly 100,000 fatalities in the United States.13
H1N1 Pandemic, 2009
In April 2009, a novel strain of the influenza A virus, H1N1, resulted in a significant outbreak of respiratory illness. The strain emerged in Mexico and quickly spread across the United States, with more than 55 million hospitalizations worldwide. Attempts to curb infection resulted in the production of a specially formulated vaccine. The clinical impact of this virus resembled previous strains of influenza; however, increased morbidity occurred among younger adults with comorbidities. In the United States, more than 10,000 patients died due to complications from the infection. The World Health Organization declared that the pandemic was over by April 2010.14,15
H3N2 Variant Influenza, 2011
First identified in 2011, the H3N2 strain of the influenza A virus has been responsible for more than 400 cases of influenza, with the majority identified after July 2012.16 This particular strain was found to originate in swine, with the addition of the M gene from the H1N1 influenza A virus. Most affected individuals reported contact with swine prior to illness. Cases of this strain typically are milder and self-limited.
Avian H7N9 Influenza, 2013
The novel avian H7N9 strain was first identified in China in 2013 and has been responsible for annual epidemics during the flu season. As with the H3N2 influenza strain, there is limited evidence of direct person-to-person transmission. Most cases have been associated with the handling of poultry prior to illness. Patients with confirmed avian H7N9 flu had severe disease, with mortality rates anywhere from 27-36%, with pneumonia as a common complication.17,18
Epidemiology
The influenza virus carries a clinically significant health burden that tends to occur seasonally, with peak activity in the winter months. This phenomenon is associated with the antigenic shifts and antigenic drifts seen in the type A strain of the virus. Major changes in the virus’s glycoproteins, neuraminidase and hemagglutinin, are termed antigenic shifts. These shifts are responsible for significant epidemics and pandemics that affect large populations. Minor changes in the structure of these glycoproteins result in antigenic drift, which tends to result in more localized outbreaks.
The incidence of influenza is difficult to characterize because it is not always diagnosed and varies based on the disease burden during each season or unique epidemic. Furthermore, the incidence varies based on different age groups. The World Health Organization currently estimates that annual influenza epidemics result in 1 billion infections worldwide, with 3 to 5 million cases of severe disease.19 Meta-analysis of hospitalized patients in the United States suggests an annual incidence around 8%.20 Seasonal disease causes 250,000 to 500,000 deaths worldwide and between 5,000 and 50,000 deaths in the United States.21
Flu Season
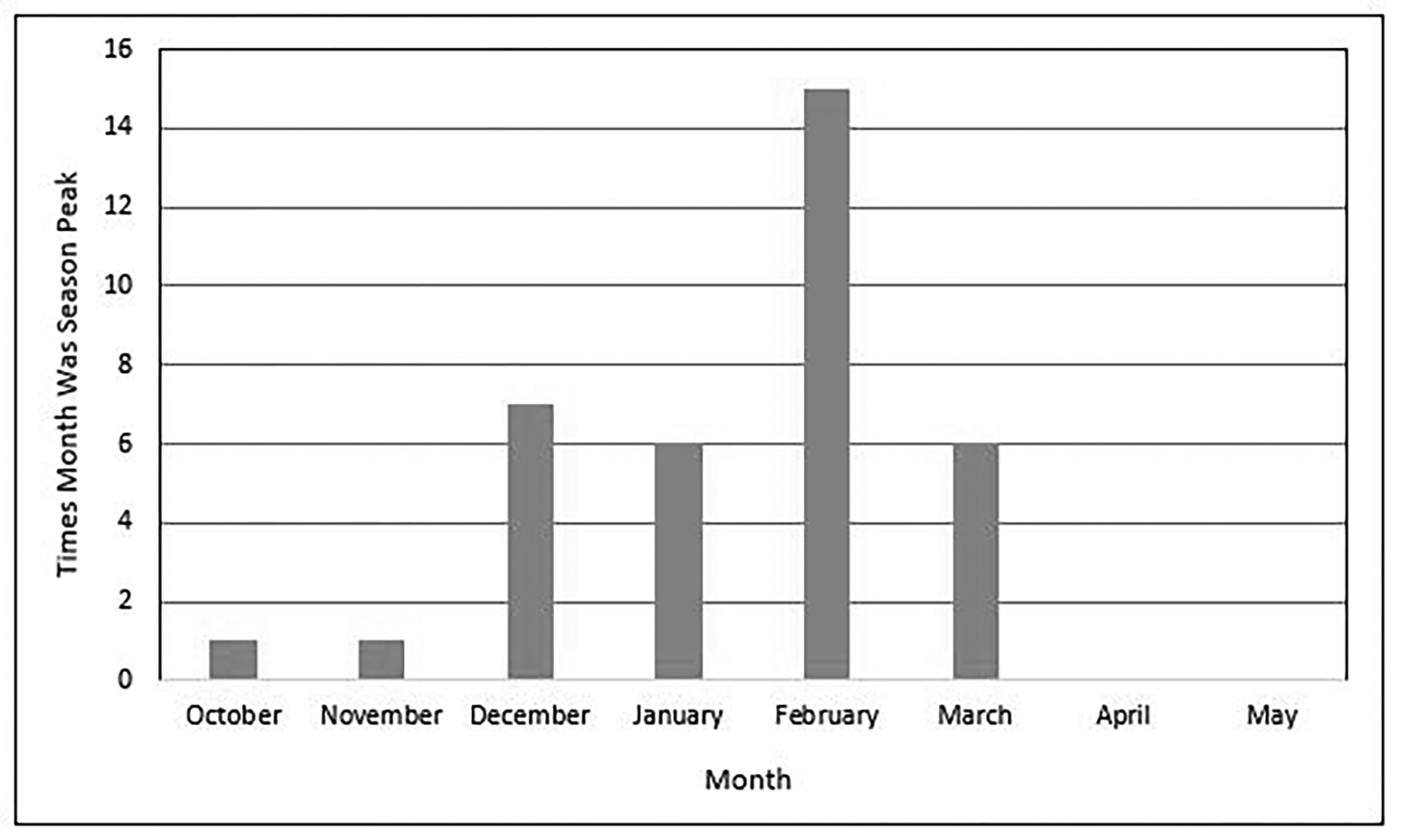
While the influenza virus circulates year-round, flu season typically is defined as the period when infection with influenza is the highest. In the United States, the CDC maintains annual data with weekly influenza reports available to the public. The incidence of respiratory secretions testing positive for influenza begins to rise in October and peaks throughout the winter months in the Northern Hemisphere.22 (See Figure 1.) Despite this, it is important for clinicians to remember that influenza circulates continuously throughout the year and may be present at any time.
Figure 1. Peak Month of Flu Activity 1982-1983 Through 2017-2018 |
 |
|
Source: Centers for Disease Control and Prevention. Influenza: The flu season. Available at: www.cdc.gov/flu/about/season/flu-season.htm. |
Although the flu season typically occurs during the winter months in both the Northern and Southern hemispheres, flu season may occur at any period during the year in tropical regions and may affect travelers. Similarly, sporadic outbreaks may occur on cruise ships and with airline travel.23,24
Etiology
The influenza virus (genus influenzavirus) is a negative-sense, single-strand RNA virus. (See Figure 2.) Influenza A, B, C, and D have been identified, with types A and B responsible for the bulk of human pathology. The A and B viral genome contains segments of genetic material that encode viral proteins that facilitate viral replication and entry into host cells. The hemagglutinin protein allows for viral entry, while the neuraminidase protein allows for viral release.
Figure 2. Influenza Virus Particles |
 |
|
Source: National Institute of Allergy and Infectious Diseases (NIAID) |
Risk Factors for Infection
Risk factors for infection with influenza are similar to those for other viral pathologies that cause disease in humans. Children without significant previous exposure to the virus are at a higher risk for infection with more severe features. Because of their decreased immune system function and significant medical comorbidities, older adults are at higher risk for severe illness that often requires hospitalization.25
Patients with neuromuscular disease and lung pathology also are more susceptible to infection. This risk is thought to be due to impaired handling of respiratory secretions.26 Females who are pregnant have an increased risk throughout pregnancy, with a peak during the third trimester that persists through the postpartum period.27
Individuals who are obese are at higher risk of infection because of several proposed mechanisms. Reduced efficacy of vaccination as well as increased viral replication have been described in patients with a significantly elevated body mass index.28
Pathophysiology
Influenza virus is spread from person to person mainly via large droplet transmission that occurs during sneezing and coughing. Transmission from host to recipient occurs when both parties are within close contact to each other, typically less than 6 feet, since the large droplets do not remain aerosolized for significant periods of time.29 The virus enters the respiratory tract and targets the epithelial cells for subsequent replication. The virus binds to the cell wall through an interaction between the virus’s hemagglutinin glycoprotein and sialic acid glycoproteins on the host cell wall. Once the virus enters the cell and replicates, the neuraminidase glycoprotein helps release replicated virions. This process leads to eventual apoptosis of the affected cells and subsequent spread of the virus.
Infectious Course
The incubation period for influenza is typically one to four days. Viral shedding is thought to occur within 24 to 48 hours before symptom onset, although there is less of a viral burden than when the patient is symptomatic.29 Peak viral shedding has been shown to occur two days after the onset of symptoms.30 Longer periods of shedding may occur with certain patient populations, such as children, older adults, immunocompromised patients, or those with chronic medical conditions.31-33
Uncomplicated influenza infection typically manifests with an acute onset of fever, headache, myalgias, and malaise. Following these initial symptoms, patients commonly experience symptoms associated with respiratory tract disease, such as cough, sore throat, and rhinorrhea.34 Patients with uncomplicated influenza usually recover from their illness within two to five days, although complete symptom resolution may take upward of one week or more.
Clinical Presentation
The clinical presentation of uncomplicated influenza mimics other respiratory viral syndromes. Patients often present with the abrupt onset of symptoms within one to four days of exposure to the virus. Symptomatology can range from mild to life-threatening and can last for up to two weeks.35,36
The initial symptoms after infection include high fevers, intense myalgias, headaches, and anorexia. As the infection progresses, patients tend to exhibit respiratory tract symptoms, such as nasal congestion, rhinorrhea, sore throat, and development of a nonproductive cough. The presence of gastrointestinal symptoms, such as vomiting and diarrhea, rarely are associated with influenza in adult patients. In contrast, nausea and vomiting may be key elements of the history elicited for pediatric patients.
The physical examination often is remarkable for a fever with the presence of posterior cervical adenopathy and erythematous mucous membranes of the nasopharyngeal passages. The oropharynx may appear hyperemic, and the lung examination can vary from benign to rales secondary to superimposed pneumonia. Tachycardia may present as a response to the febrile state or dehydration secondary to decreased oral intake. Children may demonstrate erythematous tympanic membranes suggestive of acute otitis media.
Patients at the extremes of age may present atypically. Exacerbations of underlying chronic medical comorbidities may be the initial presentation for older patients. Careful consideration should be applied when evaluating this subset of patients, since diagnosis is not always straightforward.15,35,36
Differential Diagnosis
Influenza presents similarly to other respiratory viruses.
A thorough history and physical examination by the physician can help guide the evaluation of patients for potential etiologies. Upper respiratory infections caused by other viral agents, such as adenovirus, rhinovirus, and coronavirus, present similarly to influenza. Overlapping symptoms, such as rhinorrhea, myalgias, and cough, can predominate the clinical picture. Other infectious pathologies, such as meningitis, pneumonia, and pyelonephritis, should be considered in the evaluation of patients with flu-like symptoms. A thorough travel history and exposure history should be pursued to screen for other etiologies, such as dengue fever or Ebola. This broad differential can make diagnosis challenging.
In the summer of 2019, e-cigarette or vaping product use-associated lung injury (EVALI) became the center of attention after outbreaks were reported to the CDC. Although the pathogenesis is not entirely understood, pathologic findings are heralded by fibrinous pneumonitis, diffuse alveolar damage, or organizing pneumonia. Symptoms of EVALI are centered primarily around cardiopulmonary complaints, such as shortness of breath, cough, and chest pain. Constitutional symptoms, such as malaise, fevers, and chills, also have been reported.37
While more information is being gathered on EVALI, the progression into influenza season sparked new recommendations from the CDC. Because of significant symptom overlap between EVALI and influenza, healthcare providers are encouraged to ask patients about the use of e-cigarette or vaping products. Consideration should be given to the active ingredients and how the products were obtained.38 Patients suspected of EVALI often require hospital admission and consultation with specialists from pulmonology and infectious disease.
As the influenza season continues to progress, healthcare providers are encouraged to emphasize the importance of influenza vaccination in patients who use e-cigarette or vaping products. Currently, there are not enough data to demonstrate that patients with EVALI are at higher risk for complications from the influenza virus or other respiratory illnesses. Therefore, it is recommended that clinicians consider the possibility of EVALI when determining the disposition for patients presenting with influenza-like symptoms.
Diagnosis and Testing
Influenza can be diagnosed clinically based on a thorough history and physical examination without the need for routine diagnostic testing. The abrupt onset of a febrile respiratory illness with systemic symptoms, such as myalgias and headaches, during peak months can guide clinicians toward a presumptive diagnosis.39,40
Laboratory testing is available and should be reserved for instances in which confirmatory testing will affect patient care. In infants and young children, nasal aspirates and swabs are the preferred specimens for testing. Older children and adults should provide specimens from the nasopharynx. In mechanically ventilated patients, endotracheal aspirates or bronchiolar lavage fluid should be obtained for evaluation of the lower respiratory tract.41,42
Suggested uses for rapid diagnostic testing include hospitalized patients, patients with significant medical comorbidities, and documentation for institutional purposes. Individual providers should contact their laboratory for detailed information regarding the performance of the specific diagnostic test used at their institution.
Rapid Antigen Testing
The rapid antigen test can identify viral nucleoprotein antigens in respiratory specimens.41 Data suggest that this testing method carries a pooled sensitivity of 62% and specificity of 98%.42 Although these tests can produce results in as quick as 15 minutes, they tend to have lower sensitivity than reverse transcriptase polymerase chain reaction (PCR).
Polymerase Chain Reaction
The PCR method of diagnosis is typically more burdensome since it can take upward of eight hours and is not always readily available. Despite these drawbacks, PCR analysis may be useful in select patients since sensitivity and specificity approach 100%.43
Viral Culture
Viral culture, typically viewed as the gold standard for diagnosis, has a turnaround time of 48-72 hours. Therefore, it is not used for initial clinical management in the emergency setting and instead may carry a benefit for public health screening.41
Complications
Patients with influenza also may develop more severe complications as a direct result of their infection. Well-established complications include primary influenza pneumonia, secondary bacterial pneumonia, myositis, rhabdomyolysis, acute myocardial infarction, myocarditis, pericarditis, central nervous system involvement, and toxic shock syndrome.
Although it is one of the rarest complications of influenza infection, primary influenza pneumonia is considered to be one of the most severe complications. Clinicians should consider primary pneumonia in patients with persistent symptoms and recurrent fevers. Subsequent secondary bacterial pneumonia is recognized frequently as a complication in patients older than 65 years of age.44 Radiologic imaging of the chest can assist with diagnosis. Streptococcus pneumoniae is the most common bacteria implicated. Clinicians also should recognize the increasing prevalence of community-acquired methicillin-resistant Staphylococcus aureus as a causative agent.45,46
Management
The mainstay of management for most adults and children infected with the influenza virus remains supportive care since the disease state is usually self-limited. Otherwise healthy patients without significant comorbidities may achieve symptomatic relief with over-the-counter medications and adequate oral hydration.
When initiated promptly, antiviral therapy has been shown to play a role in shortening the duration of influenza symptoms. The greatest benefit of these medications has been demonstrated when given to febrile patients within the first 24 to 30 hours of presentation.47-49
The two major classes of antiviral medications for influenza are neuraminidase inhibitors (oral oseltamivir, intranasal zanamivir, and intravenous peramivir) and adamantanes (oral amantadine and rimantadine). Because of concerns that adamantanes are effective only against influenza A and that most viral strains are highly resistant to this drug class, their use is not routinely recommended. Consequently, neuraminidase inhibitors predominate as the antiviral therapy of choice when treating suspected or confirmed cases of influenza in the overwhelming majority of patients.50
Current clinical guidelines from the CDC and the Infectious Diseases Society of America (IDSA) recommend initiation of neuraminidase inhibitors as soon as possible with confirmed or suspected cases of influenza in patients with severe illness, hospitalized patients, and patients at high risk of complications. The decision to administer therapy should not be delayed or depend on the results of diagnostic testing.42,50
Otherwise healthy adults and children with presumed or confirmed influenza may be treated electively as outpatients with neuraminidase inhibitors. Studies in these populations demonstrated a small decrease in duration of illness in adults by approximately one day.51 Shared decision-making with the patient on a case-by-case basis can help guide the decision to initiate medical therapy.
Oseltamivir (U.S. brand name: Tamiflu) is currently one of the most frequently prescribed medications used in the management of influenza. The typical dosage regimen for oseltamivir is described in Table 1. Adult patients with a creatinine clearance less than 60 should have their dose adjusted to 30 mg twice daily.52 While the drug is considered pregnancy class C, the American College of Obstetricians and Gynecologists (ACOG) recommends presumptive treatment with antiviral medication during pregnancy.50 Treatment should be initiated within 48 hours of the onset of illness and continued for five days. Common side effects include headache, nausea, and vomiting.
Table 1. Oseltamivir Dosing for Treatment52 |
|
Adults |
|
|
Children (≥ 1 year of age) |
|
|
Infants (< 1 year of age) |
|
Patients who require hospitalization or who are deemed to be at high risk for influenza complications should be treated with a neuraminidase inhibitor regardless of duration of symptoms. These risk factors include age older than 65 years, pregnancy, and chronic comorbid medical conditions. Initiation of therapy should not be delayed while waiting for diagnostic or confirmatory testing.50,53
Chemoprophylaxis for patients with exposure to suspected or confirmed cases of influenza remains a controversial topic. In general, routine use of chemoprophylaxis with antivirals is not recommended. Patients exposed to influenza who are at high risk for complications and cannot be vaccinated because of contraindications should be considered as candidates.50
Emerging Therapy
In October 2018, the United States Food and Drug Administration (FDA) announced the approval of single-dose baloxavir marboxil (U.S. brand name: Xofluza) for treatment of acute, uncomplicated influenza in people 12 years of age and older.54 This oral medication works by blocking mRNA synthesis of endonucleases responsible for viral proliferation.55 Results from a randomized controlled trial comparing baloxavir marboxil to oseltamivir and placebo demonstrated superiority in the reduction of viral load one day after initiation of pharmacotherapy for patients receiving baloxavir marboxil.56 It remains unclear whether baloxavir marboxil is superior to oseltamivir for hospitalized or immunocompromised patients, or in those with neuraminidase inhibitor-resistant influenza infections.57
In October 2019, baloxavir was approved by the FDA for expanded indications, including those with potential high-risk complications. One randomized trial showed that baloxavir had a shorter median time to resolution when compared to oseltamivir or placebo.56
Concern about emerging resistance to baloxavir developed in Japan during the 2018-2019 influenza season. Children without known exposure to this medication were found to have a strain of influenza virus with acquired resistance to baloxavir.1,58 In this year’s flu season (through Nov. 16, 2019), 169 influenza samples had been tested for susceptibility to the antiviral medications currently available. Zanamivir and baloxivir were 100% effective against the current strains, while oseltamivir and peramivir each had one case of reduced efficacy (0.6%).4
Prevention
Vaccination remains the cornerstone for primary prevention of the flu. While effective in preventing infection with common strains of the virus, there remains significant potential for illness despite vaccination.59
Given the high rates of new strains that develop from significant antigenic variation, vaccines are reformulated annually to match expected circulating strains. These formulations are generally developed six months in advance of flu season based on surveillance data from the previous year. According to the CDC, for the 2018-2019 flu season, the vaccine was found to be approximately 47% effective.
The seasonal influenza vaccine is recommended for yearly administration in patients 6 months of age and older who do not have any known contraindications for vaccination. This includes pregnant women (regardless of gestational age) and women who are breastfeeding.60
Currently, three types of influenza vaccines are available in the United States:
- The inactivated influenza vaccine is administered intramuscularly and is available in tetravalent and quadrivalent forms for patients 6 months of age and older.
- The recombinant hemagglutinin vaccine is available for patients 18 years of age and older.
- The intranasal, live attenuated influenza vaccine is available for otherwise healthy and nonpregnant patients, age 2 through 49 years.
These preparations are typically available in the fall in preparation for peak incidence during the winter months.61,62
For the 2019-2020 flu season, both trivalent and quadrivalent vaccines will be available. All vaccines will contain hemagglutinin from the H1N1, H3N2, and influenza B virus strains. The quadrivalent vaccine will contain an additional hemagglutinin from another influenza B virus strain.63
Several formulations of the vaccine are available for the current season, with no specific recommendations available for the use of one over the other. Quadrivalent vaccines are believed to convey greater protection with the
addition of another influenza B hemagglutinin. A summary of the various available influenza vaccines is included in Table 2.
Table 2. Influenza Vaccines68 |
|||
|
Name |
Type |
Age |
Precautions |
|
Afluria Quadrivalent |
Inactivated influenza virus |
Various |
|
|
Fluarix Quadrivalent |
Inactivated influenza virus |
≥ 6 months |
|
|
FluLaval Quadrivalent |
Inactivated influenza virus |
≥ 6 months |
|
|
Fluzone Quadrivalent |
Inactivated influenza virus |
Various |
|
|
Flucelvax Quadrivalent |
Inactivated influenza virus |
≥ 4 years |
|
|
Fluzone High-Dose |
Inactivated influenza virus |
≥ 65 years |
|
|
Fluad |
Inactivated influenza virus |
≥ 65 years |
|
|
Flublok Quadrivalent |
Recombinant hemagglutinin |
≥ 18 years |
|
|
FluMist Quadrivalent |
Live, attenuated virus |
2 through 49 years |
Same as above as well as:
|
Contraindications to vaccination include severe allergy to previous formulations of the influenza vaccine in the past. Additionally, several of the available forms of the vaccine contain small amounts of egg protein. It was previously thought that egg allergy was a contraindication to vaccination; however, recent data have demonstrated that vaccination may be given in patients with known egg allergy with appropriate precautions. These patients should be referred to an allergy specialist for additional evaluation prior to administration.
The intranasal, live attenuated vaccine should be withheld from patients who are immunocompromised. This vaccine also is contraindicated in patients who are pregnant, 50 years of age or older, and those who have recently taken influenza antiviral medication within the past 48 hours.62
Recently, researchers have been working on the establishment of a universal vaccination. This vaccine would function to elicit the creation of protective antibodies in vaccinated patients against well-conserved viral proteins.64,65
Disposition
Outpatient Management From the ED
The vast majority of patients diagnosed with influenza from the ED may be discharged safely home with strong return precautions. Patients discharged from the ED should follow up with a primary care provider for re-evaluation in two days for reexamination and monitoring of the patient’s clinical course. Prior to discharge, patients should demonstrate an ability to ambulate steadily and tolerate oral intake without any difficulty. Additionally, vital signs should be re-evaluated prior to discharge planning for persistent hypoxemia, tachycardia, or tachypnea. The febrile state is expected with the disease process, and clinicians should consider the effects of administration of antipyretics when evaluating the significance of fever in the ED.
Discharge instructions should educate patients on signs and symptoms that should prompt a patient to seek immediate medical attention. Persistent fevers, inability to tolerate oral intake, confusion, or changes in mental status should be reinforced clearly as reasons to return to the ED. On discharge, patients diagnosed with influenza should be educated on hand washing, respiratory hygiene, and cough etiquette to decrease the risk of viral spread.
Supportive care with antipyretics, rest, and hydration are the mainstays of outpatient therapy. Treatment with an antiviral agent may be initiated without diagnostic testing in the appropriate clinical setting. Patients should be counseled regarding the risks and benefits of medical therapy before initiation with a neuraminidase inhibitor.
Observation
Certain patients may benefit from observation in the hospital or ED, depending on institutional practices. Patients should be placed on droplet precautions to limit the spread of viral illness within the healthcare facility.66 The observation setting may be a useful disposition for patients who may benefit from serial cardiopulmonary examinations and/or intravenous hydration secondary to clinically significant dehydration. Patients at the extremes of age without significant medical comorbidities may benefit from this extended period of medical observation. Prior to discharge, patients should be re-evaluated for symptomatic control and ability to tolerate oral intake. If a patient’s clinical response to symptomatic therapy fails to improve, strong consideration should be given for admission to the hospital for further management in the inpatient setting.
Hospitalization
Over the past eight years, more than 700,000 patients have required hospitalization because of influenza.6 The decision to admit a patient to the hospital for flu can be particularly challenging, as no well-validated scoring system exists to guide disposition. This decision should be based on clinical judgment in conjunction with assessment of the patient’s risk factors and disease burden.
A subset of patients presenting to the ED should be considered strongly for admission to the hospital because of concern for decompensation from the viral illness. Adults older than 65 years of age, pregnant women, children younger than 5 years of age, as well as individuals with comorbid conditions should be considered for admission.53 (See Table 3.) Additionally, patients who were previously evaluated by a medical provider and who failed to improve with outpatient management should be considered.
Table 3. Comorbid Conditions That Increase the Risk of Influenza Complications53 |
|
Patients with influenza may deteriorate to acute respiratory distress syndrome (ARDS), requiring possible intubation and intensive care for hypoxemic respiratory failure. In these patients, extracorporeal membrane oxygenation (ECMO) may serve as bridge therapy during the acute illness.67 The EP should proceed cautiously with patients who present with hypoxemia because these patients may progress to respiratory failure. Patients who are deemed to be at high risk for worsening of clinical conditions due to comorbidities or changes in mental status should be screened for intensive care admission when available. Consultation with a pulmonologist or infectious disease specialist can be obtained in the inpatient setting depending on a patient’s clinical course or past medical history when applicable.
Summary
Influenza outbreaks occur each year with their nature and extent largely determined by the virus’s glycoprotein structure and antigenic properties. These outbreaks typically occur during the winter months and can confer high morbidity to the general patient population. Increased mortality rates are seen in young children, older adults, and those with chronic comorbid medical conditions.
During the winter months, physicians should maintain a high suspicion for influenza in all patients presenting with an acute febrile respiratory illness. While the differential diagnosis is broad, the history and physical examination can guide the physician toward a presumptive diagnosis without the need for additional testing. Laboratory testing is available and should be reserved for those patients in whom the diagnosis may change the patient’s clinical management.
Treatment is indicated for patients with severe disease or those at risk for complications. Initiation of antiviral treatment should be within 48 hours of symptom onset, while the vast majority of otherwise healthy patients can be managed with supportive care only. Complicated cases of influenza may require hospitalization and treatment with antiviral therapy regardless of symptom duration.
Prevention against the influenza virus is primarily achieved through the annual influenza vaccine. This vaccine attempts to protect patients from the most commonly circulating strains of the virus. This is accomplished by obtaining routine surveillance data about viral characteristics during each flu season.
Emerging therapy, including single-dose antiviral medications and universal vaccination, may shape the rapidly evolving nature of the influenza virus and its burden on human health.
REFERENCES
- Xu X, Blanton L, Elal AIA, et al. Update: Influenza activity in the United States during the 2018-19 season and composition of the 2019-20 influenza vaccine. MMWR Morb Mortal Wkly Rep 2019;68:544-551.
- Centers for Disease Control and Prevention. 2019-2020 U.S. flu season: Preliminary burden estimates. Available at: https://www.cdc.gov/flu/about/burden/preliminary-in-season-estimates.htm. Accessed Dec. 13, 2019.
- Centers for Disease Control and Prevention. Weekly U.S. influenza surveillance report. ILI activity map. Available at: https://www.cdc.gov/flu/weekly/index.htm#ILIActivityMap. Accessed Dec. 13, 2019.
- Centers for Disease Control and Prevention. Weekly U.S. influenza surveillance report. Public Health Laboratories. Available at: https://www.cdc.gov/flu/weekly/#PublicHealthLaboratories. Accessed Dec. 13, 2019.
- Flynn O, Gallagher C, Mooney J, et al. Influenza D virus in cattle, Ireland. Emerg Infect Dis 2018;24:389-391.
- Reed C, Chaves SS, Daily Kirley P, et al. Estimating influenza disease burden from population-based surveillance data in the United States. PloS One 2015;10:e0118369.
- Morens DM, Taubenberger JK, Folkers GK, Fauci AS. Pandemic influenza’s 500th anniversary. Clin Infect Dis 2010;51:1442-1444.
- Jester B, Uyeki T, Jernigan D. Readiness for responding to a severe pandemic 100 years after 1918. Am J Epidemiol 2018;187:2596-2602.
- Belser JA, Tumpey TM. The 1918 flu, 100 years later. Science 2018;359:255.
- Centers for Disease Control and Prevention. Reconstruction of the 1918 influenza pandemic virus. Available at: https://www.cdc.gov/flu/about/qa/1918flupandemic.htm. Accessed Dec. 14, 2019.
- Viboud C, Simonsen L, Fuentes R, et al. Global mortality impact of the 1957-1959 influenza pandemic. J Infect Dis 2016;213:738-745.
- Langmuir AD. Epidemiology of Asian influenza. With special emphasis on the United States. Am Rev Respir Dis 1961;83:2-14.
- Glezen WP. Emerging infections: Pandemic influenza. Epidemiol Rev 1996;18:64-76.
- Reed C, Angulo FJ, Swerdlow DL, et al. Estimates of the prevalence of pandemic (H1N1) 2009, United States, April–July 2009. Emerg Infect Dis 2009;15:2004-2007.
- Walls RM, Hockberger RS, Gausche-Hill M. Rosen’s Emergency Medicine: Concepts and Clinical Practice. 9th ed. Philadelphia: Elsevier; 2018.
- Schicker RS, Rossow J, Eckel S, et al. Outbreak of influenza A(H3N2) variant virus infections among persons attending agricultural fairs housing infected swine — Michigan and Ohio, July-August 2016. MMWR Morb Mortal Wkly Rep 2016;65:1157-1100.
- Gao R, Cao B, Hu Y, et al. Human infection with a novel avian-origin influenza A (H7N9) virus. N Engl J Med 2013;368:1888-1897.
- Yu H, Cowling BJ, Feng L, et al. Human infection with avian influenza A H7N9 virus: An assessment of clinical severity. Lancet 2013;382:138-145.
- World Health Organization. Influenza (Seasonal) Fact Sheet. Nov. 6, 2018. Available at: https://www.who.int/en/news-room/fact-sheets/detail/influenza-(seasonal). Accessed Dec. 14, 2019.
- Tokars JI, Olsen SJ, Reed C. The seasonal incidence of symptomatic influenza in the United States. Clin Infect Dis 2018;66:1511-1518.
- Takhar SS, Moran GJ. Serious viral infections. In: Tintinalli JE, Stapczynski J, Ma O, et al, eds. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide. 8th ed. New York: McGraw-Hill; 2016.
- Centers for Disease Control and Prevention. Influenza: The flu season. Available at: www.cdc.gov/flu/about/season/flu-season.htm. Accessed Dec. 14, 2019.
- Moura FE. Influenza in the tropics. Curr Opin Infect Dis 2010;23:415-420.
- Brownstein JS, Wolfe CJ, Mandl KD. Empirical evidence for the effect of airline travel on inter-regional influenza spread in the United States. PLoS Med 2006;3:e401.
- Thompson WW, Shay DK, Weintraub E, et al. Influenza-associated hospitalizations in the United States. JAMA 2004;292:1333.
- Keren R, Zaoutis TE, Bridges CB, et al. Neurological and neuromuscular disease as a risk factor for respiratory failure in children hospitalized with influenza infection. JAMA 2005;294:2188.
- Neuzil KM, Reed GW, Mitchel EF, et al. Impact of influenza on acute cardiopulmonary hospitalizations in pregnant women. Am J Epidemiol 1998;148:1094-1102.
- Van Kerkhove MD, Vandemaele KA, Shinde V, et al. Risk factors for severe outcomes following 2009 influenza A (H1N1) infection: A global pooled analysis. PLoS Med 2011;8:e1001053.
- Brankston G, Gitterman L, Hirji Z, et al. Transmission of influenza A in human beings. Lancet Infect Dis 2007;7:257-265.
- World Health Organization Writing Group, Bell D, Nicoll A, et al. Non-pharmaceutical interventions for pandemic influenza, international measures. Emerg Infect Dis 2006;12:81-87.
- Carrat F, Vergu E, Ferguson NM, et al. Time lines of infection and disease in human influenza: A review of volunteer challenge studies. Am J Epidemiol 2008;167:775-785.
- Leekha S, Zitterkopf NL, Espy MJ, et al. Duration of influenza A virus shedding in hospitalized patients and implications for infection control. Infect Control Hosp Epidemiol 2007;28:1071-1076.
- Klimov AI, Rocha E, Hayden FG, et al. Prolonged shedding of amantadine-resistant influenzae A viruses by immunodeficient patients: Detection by polymerase chain reaction-restriction analysis. J Infect Dis 1995;172:1352-1355.
- Loeb M, Singh PK, Fox J, et al. Longitudinal study of influenza molecular viral shedding in Hutterite communities. J Infect Dis 2012;206:1078-1084.
- Cohen YZ, Dolin R. Influenza. In: Kasper DL, Fauci AS, Hauser SL, et al, eds. Harrison’s Principles of Internal Medicine. 19th ed. New York: McGraw Hill Book Company; 2015:1209.
- Centers for Disease Control and Prevention. Clincial signs and symptoms of influenza. Available at: https://www.cdc.gov/flu/professionals/acip/clinical.htm. Accessed Dec. 14, 2019.
- Layden JE, Ghinai I, Pray I, et al. Pulmonary illness related to e-cigarette use in Illinois and Wisconsin — Preliminary report. N Engl J Med 2019. doi: 10.1056/NEJMoa1911614. [Epub ahead of print].
- Jatlaoui TC, Wiltz JL, Kabbani S; Lung Injury Response Working Group. Update: Interim guidance for health care providers for managing patients with suspected e-cigarette, or vaping, product use-associated lung injury — United States, November 2019. MMWR Morb Mortal Wkly Rep 2019;68:1081-1086. Available at: https://www.cdc.gov/mmwr/volumes/68/wr/mm6846e2.htm. Accessed Dec. 14, 2019.
- Centers for Disease Control and Prevention. Information for clinicians on rapid diagnostic testing for influenza. Available at: http://www.cdc.gov/flu/professionals/diagnosis/rapidclin.htm. Accessed Dec. 14, 2019.
- Centers for Disease Control and Prevention. Influenza signs and symptoms and the role of laboratory diagnostics. Available at: http://www.cdc.gov/flu/professionals/diagnosis/labrolesprocedures.htm. Accessed Dec. 14, 2019.
- Chartrand C, Leeflang MM, Minion J, et al. Accuracy of rapid influenza diagnostic tests: A meta-analysis. Ann Intern Med 2012;156:500.
- Harper SA, Bradley JS, Englund JA, et al. Seasonal influenza in adults and children — Diagnosis, treatment, chemoprophylaxis, and institutional outbreak management: Clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis 2009;48:1003-1032.
- Abraham MK, Perkins J, Vilke GM, Coyne CJ. Influenza in the emergency department: Vaccination, diagnosis, and treatment: Clinical practice paper approved by American Academy of Emergency Medicine Clinical Guidelines Committee. J Emerg Med 2016;50:536-542.
- Chertow DS, Memoli MJ. Bacterial coinfection in influenza: A grand rounds review. JAMA 2013;309:275.
- Wolter N, Tempia S, Cohen C, et al. High nasopharyngeal pneumococcal density, increased by viral coinfection, is associated with invasive pneumococcal pneumonia. J Infect Dis 2014;210:1649-1657.
- Schwarzmann SW, Adler JL, Sullivan RJ Jr, Marine WM. Bacterial pneumonia during the Hong Kong influenza epidemic of 1968-1969. Arch Intern Med 1971;127:1037.
- Cooper NJ, Sutton AJ, Abrams KR, et al. Effectiveness of neuraminidase inhibitors in treatment and prevention of influenza A and B: Systematic review and meta-analyses of randomised controlled trials. BMJ 2003;326:1235.
- Heneghan CJ, Onakpoya I, Thompson M, et al. Zanamivir for influenza in adults and children: Systematic review of clinical study reports and summary of regulatory comments. BMJ 2014;348:g2547.
- Dobson J, Whitley RJ, Pocock S, Monto AS. Oseltamivir treatment for influenza in adults: A meta-analysis of randomised controlled trials. Lancet 2015;385:1729.
- Centers for Disease Control and Prevention. Influenza antiviral medications: Summary for clinicians. Available at: http://www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Accessed Dec. 14, 2019.
- Jefferson T, Demicheli V, Deeks J, Rivetti D. Neuraminidase inhibitors for preventing and treating influenza in healthy adults. Cochrane Database Syst Rev 2000;CD001265.
- Oseltamivir. Lexi-Drugs. Lexicomp. Wolters Kluwer Health, Inc. Riverwoods, IL. Available at: http://online.lexi.com. Accessed Jan. 10, 2020.
- Fiore AE, Fry A, Shay D, et al. Antiviral agents for the treatment and chemoprophylaxis of influenza — recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2011;60:1-24.
- U.S. Food and Drug Administration. FDA approves new drug to treat influenza [news release]. Oct. 24, 2018. Available at: https://www.fda.gov/news-events/press-announcements/fda-approves-new-drug-treat-influenza. Accessed Dec. 14, 2019.
- Heo Y-A. Baloxavir: First global approval. Drugs 2018;78:693-697.
- Hayden FG, Sugaya N, Hirotsu N, et al. Baloxavir marboxil for uncomplicated influenza in adults and adolescents. N Engl J Med 2018;379:913-923.
- Uyeki TM. A step forward in the treatment of influenza. N Engl J Med 2018;379:975-977.
- Takashita E, Kawakami C, Ogawa R, et al. Influenza A(H3N2) virus exhibiting reduced susceptibility to baloxavir due to a polymerase acidic subunit I38T substitution detected from a hospitalised child without prior baloxavir treatment, Japan, January 2019. Euro Surveill 2019;24. doi: 10.2807/1560-7917.ES.2019.24.12.1900170.
- Kilbourne ED. Influenza immunity: New insights from old studies. J Infect Dis 2006;193:7-8.
- [No authors listed]. ACOG Committee Opinion No. 732: Influenza vaccination during pregnancy. Obstet Gynecol 2018;131:e109-e114.
- Grohskopf LA, Sokolow LZ, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices — United States, 2018-19 influenza season. MMWR Rec Rep 2018;67:1-20.
- Committee on Infectious Diseases. Recommendations for prevention and control of influenza in children, 2017-2018. Pediatrics 2017;140. doi: 10.1542/peds.2017-2550.
- U.S. Food & Drug Administration. 2019 Meeting Materials, Vaccines and Related Biological Products Advisory Committee. Available at: https://www.fda.gov/advisory-committees/vaccines-and-related-biological-products-advisory-committee/2019-meeting-materials-vaccines-and-related-biological-products-advisory-committee. Accessed Dec. 14, 2019.
- Lambert LC, Fauci AS. Influenza vaccines for the future. N Engl J Med 2010;363:2036-2044.
- Erbelding EJ, Post DJ, Stemmy EJ, et al. A universal influenza vaccine: The strategic plan for the National Institute of Allergy and Infectious Diseases. J Infect Dis 2018;218:347-354.
- Centers for Disease Control and Prevention. Prevention strategies for seasonal influenza in healthcare settings. Available at: https://www.cdc.gov/flu/professionals/infectioncontrol/healthcaresettings.htm. Accessed Dec. 14, 2019.
- Australia and New Zealand Extracorporeal Membrane Oxygenation (ANZ ECMO) Influenza Investigators, Davies A, Jones D, et al. Extracorporeal membrane oxygenation for 2009 influenza A(H1N1) acute respiratory distress syndrome. JAMA 2009;302:1888.
- Grohskopf LA, Alyanak E, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices — United States, 2019–20 Influenza Season. MMWR Recomm Rep 2019;68:1-21.
The 2019-2020 flu season is already among us, and it is imperative that those healthcare practitioners on the frontline have current knowledge of prevention and treatment strategies. Both influenza A and B strains have been identified, and their predominance varies by region and age group.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
