Long-Lasting, Woman-Controlled Contraception Is Here
March 1, 2020
Reprints
By Jeffrey T. Jensen, MD, MPH
Leon Speroff Professor and Vice Chair for Research, Department of OB/GYN, Oregon Health & Science University, Portland
SOURCES: Vieira CS, Fraser IS, Plagianos MG, et al. Bleeding profile associated with 1-year use of the segesterone acetate/ethinyl estradiol contraceptive vaginal system: Pooled analysis from Phase 3 trials. Contraception 2019;100:438-444.
Tuazon JP, Sitruk-Ware R, Borlongan CV. Beyond contraception and hormone replacement therapy: Advancing Nestorone to a neuroprotective drug in the clinic. Brain Res 2019;1704:161-163.
Gemzell-Danielsson K, Sitruk-Ware R, Creinin MD, et al. Segesterone acetate/ethinyl estradiol 12-month contraceptive vaginal system safety evaluation. Contraception 2019;99:323-328.
Archer DF, Merkatz RB, Bahamondes L, et al. Efficacy of the 1-year (13-cycle) segesterone acetate and ethinylestradiol contraceptive vaginal system: Results of two multicentre, open-label, single-arm, phase 3 trials. Lancet Glob Health 2019;7:e1054-e1064.
I decided to introduce the new one-year contraceptive vaginal ring system to you in a special feature to help highlight the most im-portant information about the product in one report. Before I review the data, I want to acknowledge and disclose my relation to this product: I served as a senior advisor to the Population Council on the International Committee for Contraception for much of the ring development program; I served as an investigator in the Eunice Kennedy Shriver National Institute of Child Health and Human Development-funded Contraception Clinical Trial Network (CCTN) that performed the Phase III clinical trial; and most recently, I serve as a consultant for TherapeuticsMD, the small company that will market the ring in the United States. While these opportunities have provided me with a unique inside understanding of this product, they also represent a potential source of influence. Even though these potential conflicts of interest are listed in this publication, I want to be fully transparent about these relationships. My institution, Oregon Health & Science University, also manages potential conflicts of interest for faculty. With that out of the way, let’s talk about Annovera.
Why is a new method needed?
High rates of unintended pregnancy provide evidence that the available contraceptive method mix does not meet the needs of all women. We also know that short-acting methods like oral contraceptives have higher failure rates than long-acting reversible contraceptive (LARC) methods like intrauterine devices (IUDs) or implants. Hubacher and colleagues conducted a partially randomized patient preference trial1 where they recruited women seeking a short-acting method (pills or injectable) and randomized consenting participants to receive either the short-acting method or a long-acting reversible method of their choice, free of charge. Women who declined randomization were invited to participate in an observational cohort using their preferred method at their own cost. The 12-month method continuation probabilities were similar among the women who received a short-acting method (63% preference short-acting group, 53% randomized short-acting group), but significantly higher (78%) among those randomized to LARC. More impressively, the 12-month cumulative unintended pregnancy probabilities were 6.4% and 7.7% among the preference and randomized short-acting groups, but only 0.7% among those randomized to LARC. This study adds to other convincing population-based research that supports the contraceptive superiority of LARC methods over short-acting methods. LARC uptake has increased substantially over the last two decades,2 and this correlates with a reduction in unintended pregnancy.3
So, it appears we have cracked the nut; increase access to LARC methods, and all women will use them. Only one problem; they won’t. Not all women want to use an IUD or implant. Some have had problems with a device, and others balk at the idea of a method that they do not control. In an effort to make use of short-duration methods easier, products like the patch and monthly combination vaginal ring have been introduced. However, these products have not shown greater effectiveness than the Pill.4 One explanation could be that women have difficulty continuing with a prescription that must be refilled regularly. Pittman and colleagues found that only 30% of participants using short-acting hormonal methods enrolled in the Contraceptive CHOICE Project obtained refills on time, with ring use a predictor of non-adherence.5 While extended use of the ring may have explained this result, the very low adherence rates support that starting late increases failure rates. So fewer trips to the pharmacy might help. This concept drove the development of the one-year ring.
What is Annovera?
Annovera received U.S. Food and Drug Administration (FDA) approval in August 2018, and a soft market introduction occurred during the fourth quarter of 2019, with full introduction in January 2020. Similar to other combined hormonal methods, the system is designed for 21 days of continuous use, followed by a seven-day ring-free interval, during which the ring is removed, washed, and stored. Unlike other products, the same ring is used for subsequent cycles for an entire year (13 cycles total). Thus, the product represents a new class of “long-lasting” contraceptives. One trip to the pharmacy provides a year of contraception that is woman-controlled.
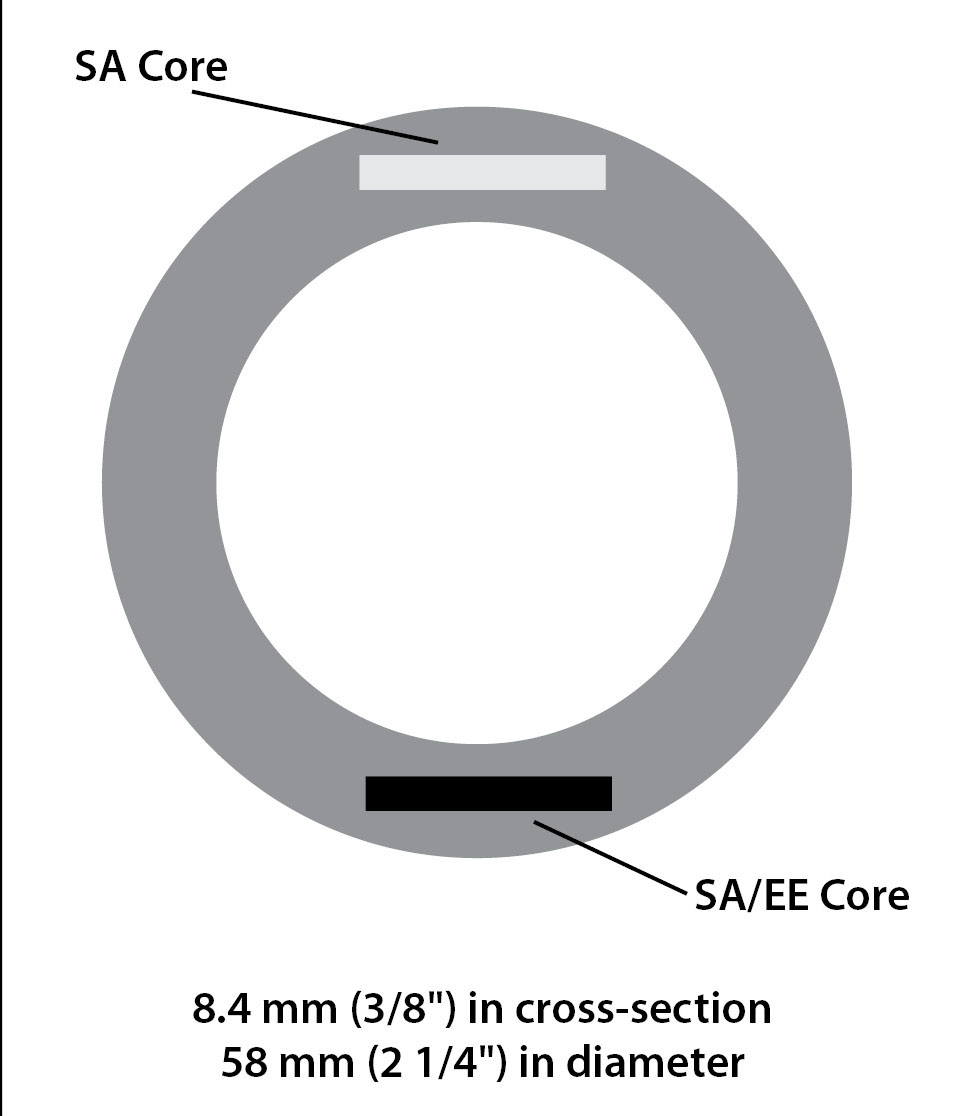
This system is a soft and flexible silicone elastomer contraceptive vaginal ring (CVR) that is noticeably different from the ethyl vinyl acetate (EVA) monthly ring. Compared to the EVA ring, the silicone material is very soft to the touch and pliable, and not noticed by a woman (or her partner) when correctly placed in the vagina. The CVR has an outer diameter of 56 mm and the cross-sectional diameter is 8.4 mm. The patient places the system in the vagina, directing the ring toward the upper fornix and cervix. Two silicone elastomer cores, 3 mm wide, sealed within hollow channels in the body of the ring, release the contraceptive steroids; one measures 11 mm in length and releases segesterone acetate (SA) and the second core measures 18 mm and releases both ethinyl estradiol (EE) and SA. (See Figure 1.) A new CVR contains 103 mg SA and 17.4 mg EE; this provides an average release rate of 150 mcg/day of SA and
13 mcg/day of EE. A major advantage of this CVR over the etonogestrel/EE ring is that it is stable at room temperature and does not require refrigeration. With the 21/7 schedule of use, the same ring is used over 13 consecutive cycles. Release levels of hormones decline gradually over the year of use, but they remain sufficient to provide adequate contraception for several more weeks in the event that a ring is not changed at the correct time.
Figure 1. Structure of the Annovera Ring |
 |
|
SA: segesterone acetate; EE: ethinyl estradiol Adapted with permission from TherapeuticsMD. |
This product represents the first commercial use of SA, a 19-norprogesterone derivative with reduced binding to the androgen receptor, estrogen receptor, and mineralocorticoid receptor compared to 19-nortestosterone derived progestins.6 SA is not bioavailable following oral administration, so the progestin has been evaluated for delivery as a gel, implant, and ring. SA does not bind sex hormone binding globulin (SHBG) in circulation.6
Once released from the ring, SA and EE are absorbed through the vaginal epithelium and enter the circulation through the venous network supplying the upper vagina. Although the vaginal route of delivery avoids first-pass hepatic metabolism, this does not reduce the impact of EE on hepatic globulin production. Therefore, this product carries the same contraindications and thrombosis risk as other combined hormonal contraceptive methods.
How effective is Annovera?
Two identically designed, open-label, single-arm Phase III studies conducted between 2006 and 2009 provide the primary evidence for contraceptive efficacy, bleeding patterns, and safety of the SA/EE CVR. The first study was conducted by the CCTN at 15 U.S. sites, and the Population Council conducted the second international study at 12 sites in the United States, Latin America, Europe, and Australia. The pooled analyses published in The Lancet provide the main results for safety and efficacy presented to the FDA,7 with additional safety data published in Contraception.8
The primary efficacy endpoint (Pearl Index for women younger than 35 years of age) included 2,265 participants. Forty pregnancies occurred, leading to a calculated Pearl Index of 2.98 (95% confidence interval [CI], 2.13–4.06) per 100 woman-years. Of interest, the Pearl Index was 2.10 among women who did not report any unscheduled ring removals greater than two hours. The Pearl Index was significantly lower among the European participants compared to participants at the other study sites, and also varied by participant age.
What is the bleeding pattern with use of this CVR?
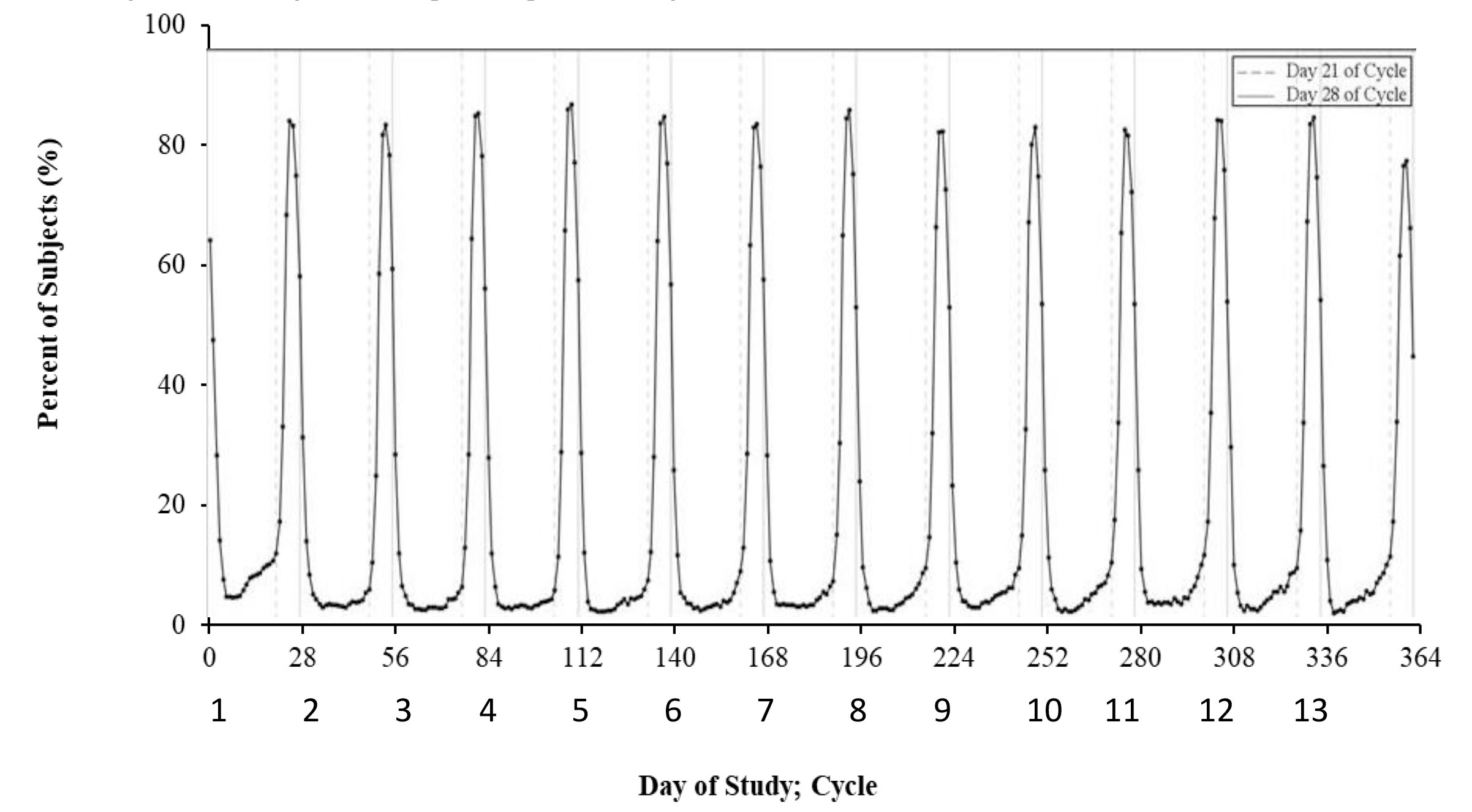
Pooled analysis of bleeding diaries from the Phase III trials included data from 16,408 cycles from 2,070 participants according to World Health Organization (WHO) terminology, with spotting defined as bloody discharge not requiring sanitary protection, normal bleeding defined as flow requiring sanitary protection, and heavy bleeding defined as flow heavier than normal menses.9 The vast majority of participants reported scheduled bleeding or spotting during CVR use (92-95% per cycle) with a mean of 4.9 ± 1.1 days per cycle and great consistency over the year-long study. (See Figure 2.) Only 1.8% of participants discontinued the CVR due to unacceptable bleeding.
Figure 2. Percent of Subjects Experiencing Bleeding or Spotting in the Phase III Studies |
 |
|
Reprinted with permission from TherapeuticsMD. |
What about safety?
In the Phase III clinical trials, 15 serious adverse events occurred, which were deemed possibly or probably related to the CVR. Four women (0.2%) experienced nonfatal venous thromboembolism (VTE); three had risk factors (two had body mass index [BMI] greater than 29 kg/m2 and one was found to have Factor V Leiden mutation). Among women with a BMI less than 29 kg/m2, the VTE rate was 10.8/10,000 woman-years (95% CI, 8.9-13.1). Other serious adverse events included two allergic reactions and 10 spontaneous abortions. Five subjects reported gallbladder disease; one required a cholecystectomy. The general side effect profile appears no different than other combined products.
The occurrence of VTE in obese women during the Phase III trials led to a decision by the Data Safety Monitoring Board to stop enrollment and discontinue participation of obese women. This decision artificially increased the total discontinuation rate for the product during the clinical trial. It also led to the inclusion of special language in the label.
Can Annovera be used in obese women?
The decision to stop enrollment and discontinue participation of obese women during the Phase III trial resulted in the inclusion of a statement in the package insert in the indications and usage section: Annovera is “not adequately evaluated in females with a body mass index of > 29 kg/m2.” To be fair, most of the combined methods currently on the market have not been adequately evaluated in this high-risk population. The U.S. Medical Eligibility Criteria list use of combined hormonal contraception as category 2 for obese women, and do not distinguish products.10
While the available data suggest that the risk of VTE with use of the SA/EE ring will be similar to other combined methods, in our litigious society it makes sense to discuss this controversy with patients considering use of the method. History tells us that new methods attract unhealthy users. I worry that some clinicians may think the vaginal administration of hormones will reduce risks. While this is true for estradiol, vaginal administration of EE still results in an increase in prothrombotic globulins, and the experience with the monthly ring shows a similar risk for VTE. The completion of an FDA-mandated Phase IV post-marketing study for Annovera will provide prospective comparative data similar to studies completed evaluating EE/drospirinone, estradiol valerate/dienogest pills, and the EE/etonogestrel ring. This should clarify this issue, but it is years away.
Which women are good candidates for Annovera?
Looking carefully at all of the data, this CVR provides a great option for patients who desire a long-lasting product that is easy to use and woman-controlled. This ring has many advantages similar to LARC methods but does not require clinician placement or removal. One main counseling point requires emphasis: This ring provides highly-effective contraception when it stays in the vagina. In the clinical trials, women who reported any ring removal (except for the seven-day ring-free week) experienced significantly higher failure rates. The explanation is that SA has a very short half-life. While the product label tells women that ring removal for less than two hours does not require back-up contraception, I worry that some women conclude that repeated removal for short intervals is acceptable. The clinical trial results strongly suggest that short-term removal commonly exceeds two hours; it is not hard to imagine how removal for sex could lead to a ring being out overnight. With repeated acts of intercourse, a discontinuous pattern of suppression could lead to follicle growth and ovulation.
No product is good for every woman. In my opinion, women who feel the need to remove the ring for any reason (outside of the scheduled removal week) are not ideal candidates for this method. The ring does not increase the risk of vaginitis and does not require removal for hygiene. Reinforcing this message may encourage perfect use.
The next obvious question would be whether continuous use (no cycle interruption) makes sense. Our experience with other combined methods, including the EE/etonogestrel ring, suggests that continuous use of the SA/EE ring would be acceptable. How this will affect bleeding is not known. I expect that clinical trials will provide better data over the coming years.
REFERENCES
- Hubacher D, Spector H, Monteith C, et al. Long-acting reversible contraceptive acceptability and unintended pregnancy among women presenting for short-acting methods: A randomized patient preference trial. Am J Obstet Gynecol 2017;216:101-109.
- Daniels K, Abma JC. Current contraceptive status among women aged 15-49: United States, 2015–2017. NCHS Data Brief No. 327, Dec. 2018.
- MacCallum-Bridges CL, Margerison CE. The Affordable Care Act contraception mandate & unintended pregnancy in women of reproductive age: An analysis of the National Survey of Family Growth, 2008-2010 v. 2013-2015. Contraception 2020;101:34-39.
- Sundaram A, Vaughan B, Kost K, et al. Contraceptive failure in the United States: Estimates from the 2006-2010 National Survey of Family Growth. Perspect Sex Reprod Health 2017;49:7-16.
- Pittman ME, Secura GM, Allsworth JE, et al. Understanding prescription adherence: Pharmacy claims data from the Contraceptive CHOICE Project. Contraception 2011;83:340-345.
- Sitruk-Ware R, Nath A. The use of newer progestins for contraception. Contraception 2010;82:410-417.
- Archer DF, Merkatz RB, Bahamondes L, et al. Efficacy of the 1-year (13-cycle) segesterone acetate and ethinylestradiol contraceptive vaginal system: Results of two multicentre, open-label, single-arm, phase 3 trials. Lancet Glob Health 2019;7:e1054-e1064.
- Gemzell-Danielsson K, Sitruk-Ware R, Creinin MD, et al. Segesterone acetate/ethinyl estradiol 12-month contraceptive vaginal system safety evaluation. Contraception 2019;99:323-328.
- Vieira CS, Fraser IS, Plagianos MG, et al. Bleeding profile associated with 1-year use of the segesterone acetate/ethinyl estradiol contraceptive vaginal system: Pooled analysis from Phase 3 trials. Contraception 2019;100:438-444.
- Curtis KM, Tepper NK, Jatlaoui TC, et al. U.S. medical eligibility criteria for contraceptive use, 2016. MMWR Recomm Rep 2016;65:1-103.
Annovera is designed for 21 days of continuous use, followed by a seven-day ring-free interval, during which the ring is removed, washed, and stored. Unlike other products, the same ring is used for subsequent cycles for an entire year (13 cycles total).
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
