Infectious Disease Alert Updates
By Carol A. Kemper, MD, FACP
Clinical Associate Professor of Medicine, Stanford University, Division of Infectious Diseases, Santa Clara Valley Medical Center
Dr. Kemper reports no financial relationships relevant to this field of study.
Step-Wise Interventions for Hospital-Onset Clostridioides difficile
SOURCE: Rohde JM, Jones K, Padron N, et al. A tiered approach for preventing Clostridioides difficile infection. Ann Intern Med 2019;171(Suppl 7):S45- S52.
Clostridium difficile, also known as Clostridioides difficile, infection (CDI) has emerged as the most important — and insubmissive — healthcare-associated infection (HAI). From 2001 to 2010, cases of CDI doubled in hospital patients in the United States, with an increased incidence of 4.5 cases to 8.2 cases per 1,000 non-maternal adult discharges during the 10 years of observation.1 The incidence of CDI was highest among those 65 years of age and older (11.6 cases per 1,000 adult discharges). In 2011, CDI caused an estimated 29,000 deaths in the United States. By 2015, government data indicated the rate had further increased to 14.2 cases per 1,000 hospital discharges, although better metrics found that 79% of these infections were present on admission and 21% occurred during the hospital stay.2 Nonetheless, healthcare-associated exposure is still believed to be the most common source for community-acquired CDI, with 80% of affected patients reporting contact with the healthcare system. Interestingly, while government data indicate the rate of CDI is slowly increasing throughout the United States, hospital-acquired CDI has modestly declined.
Combating CDI in the hospital environment requires a multi-pronged approach, including maximized hand hygiene and contact precautions, enhanced environmental measures, minimization of host risk factors through antimicrobial stewardship, and improved diagnostics. From personal experience, I can attest to the importance of each of these interventions. None by itself is sufficiently effective. In 2016, the Centers for Disease Control and Prevention (CDC) launched a quality initiative to provide a non-randomized, tiered approach to four cohorts of hospitals and long-term acute care facilities struggling with higher rates of CDI. A series of interventions was introduced — and then certain interventions were amplified later when needed. I will add my own personal comments to this list.
These CDC-based initiatives included:
• Reinforced antimicrobial stewardship: Antimicrobial stewardship remains the single most important intervention in all U.S. guidelines. In my experience, curtailing the use of unnecessary antibiotics and limiting the duration of antibiotics is helpful, but many hospitalized patients require antibacterials, putting them at immediate risk for active CDI, especially if colonized. You just cannot get beyond that fact. I would add to this recommendation that Infectious Disease consultation has been shown to improve antimicrobial use and reduce days of hospitalization.
• Improved C. difficile (CD) diagnostics: Current guidelines advocate for the use of two types of tests in a step-wise approach: testing for the presence of organism (molecular or other), and enzyme immunoassay for the presence of CD toxin production. The ability to test separately for the presence of the organism and toxin production has greatly expanded our ability to manage patients with colonization vs. those with active disease. Unfortunately, modern methods of testing remain confusing to many physicians, and patients with colonization may not be tested for toxin production and may receive unnecessary treatment (and an incorrect diagnosis). Further, repeated testing and multiple tests are no longer needed, and only those with clinically significant symptoms should be tested for toxin production. From my perspective, the most strategic advantage of the newer diagnostics is the ability to detect fecal colonization, allowing for preemptive contact isolation and enhanced environmental measures.
• Precautions and equipment: Contact precautions and appropriate personal protective equipment (PPE) with gowns and gloves is key to cutting down on hospital transmission of the organism, regardless of colonization or active CDI.
• Reinforced hand hygiene: Even with the appropriate use of contact precautions and PPE, hand hygiene remains one of the most important interventions. At least 20% to 40% of CDI in the hospital can be attributed to transmission of organism and/or spores via hands. Newer technologies to monitor hand hygiene better would be welcome.
• Environmental measures: From my perspective, this is the final and perhaps the most important intervention a hospital can invest in. Good cleaning staff are important. Since CD spores can persist in the environment for months, a room with a prior occupant with CD clearly puts the next occupant at risk. We provide enhanced daily room cleaning while a patient with CDI is hospitalized, and “terminal cleaning” is done at exit. The number of room changes is limited when possible.
Several personal comments to add to this list:
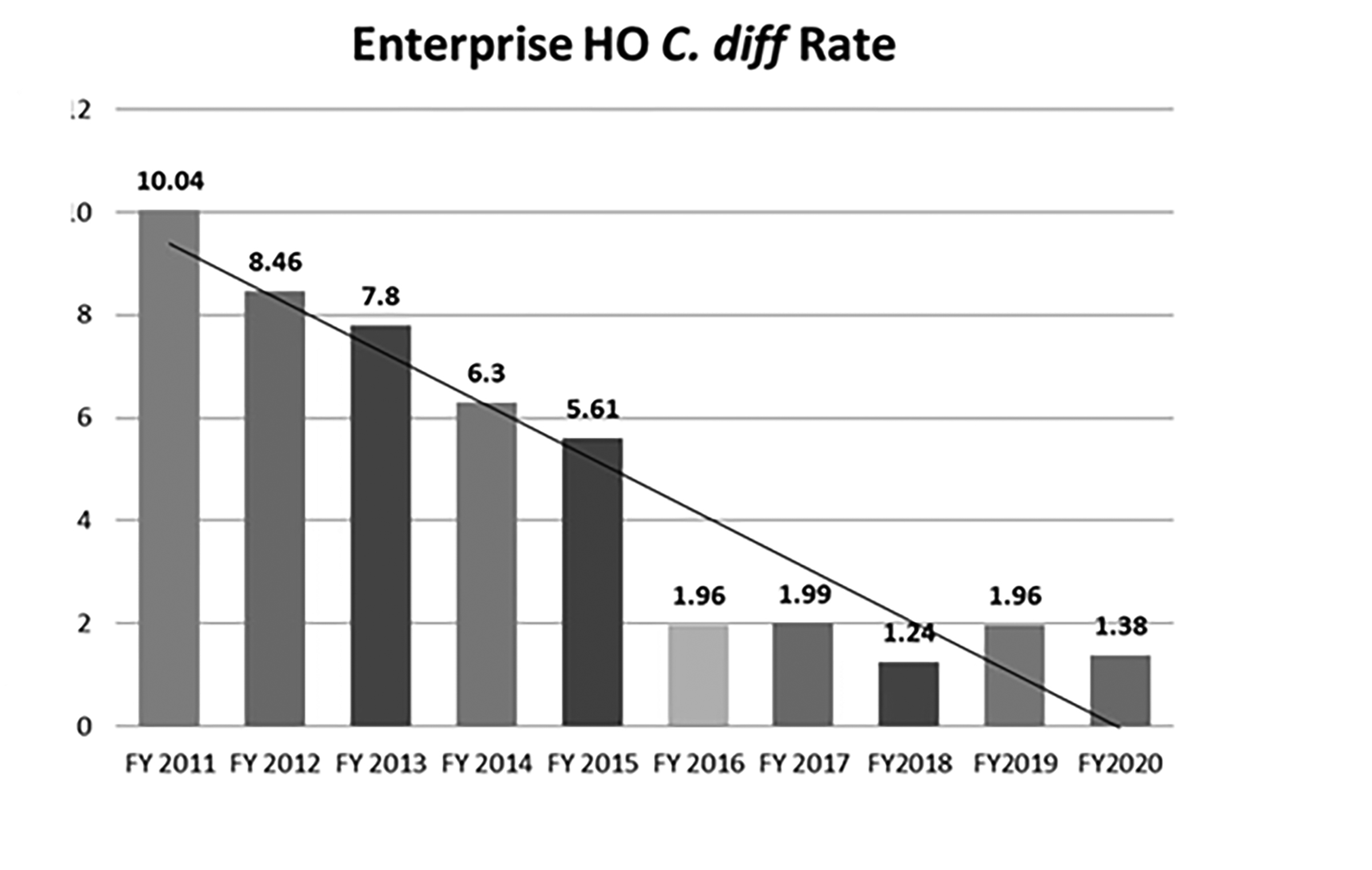
• For the past 10 years, our facility in Mountain View, CA, has worked hard to implement a series of step-wise measures, similar to those outlined above, aimed at reducing hospital-onset CDI (HO-CDI). I am proud to say our facility’s HO-CD rates are now among the lowest in California. (See Figure 1.) This was not done in a controlled fashion, but simply as we gained information and experience, and as resources were available. From the figure, you can see the resulting gradual improvement in HO-CD rates at our facility.
Figure 1: C. difficile Hospital Onset Rate (per 10,000 Patient Days), Fiscal Years 2011-2020 |
 |
• Beginning in 2009, we launched a Hand Hygiene Campaign, beefed up our antimicrobial stewardship program, hired a PharmD to manage the program, and introduced increasingly more aggressive contact precautions.
• Patients with CDI are maintained on contact precautions for a minimum of two months following successful completion of treatment. Data suggest that excretion of spores/organism is greatest in the two weeks following completion of treatment, not during treatment.
• In 2014, two-step testing became available, and we began routinely screening high-risk admissions for CD colonization, including patients with a history of CD (ever), patients admitted from long-term care or from an outside facility, and patients on hemodialysis. We provide surveillance screening for about 600 admissions every quarter, with an overall rate of colonization in asymptomatic high-risk persons of ~16%. Rates of CD colonization remain highest in those with a history of CDI (46%), followed by admissions from skilled care (13%), and dialysis patients (12%). (Previously, 30-day readmissions were screened, but the rates of CD colonization were so low, this step was not economical.) Unfortunately, surveillance is not billable and is done purely at the hospital’s expense. Providing funding for this important public health safety measure would be helpful to hospitals. My guess is that many hospitals cannot afford this important measure.
• Further, patients with a history of CDI or colonization are “flagged” upon readmission, immediately go back into contact isolation, and receive surveillance rectal swab PCR testing. Contact precautions are removed when two separate surveillance swabs obtained one week apart are negative (not while receiving agents active against CD).
• Beginning in 2013, environmental measures were constantly improved. Terminal cleaning is performed in all critical care and CD rooms. We used our own data to fight intended cuts in cleaning and fought for additional staff.
• Routine adenosine triphosphate (ATP) monitoring following room cleaning was introduced, both as a means to provide feedback and education to the cleaning staff, but also to ensure quality cleaning. Environmental cleaning staff receive regular competencies, using ATP monitoring.
• With the gradual implementation of all of the above from 2009-2015, our facility was able to reduce the HO-CDI rates by more than half. But it was not until our facility went the extra step and invested in ultraviolet (UV) disinfection in 2015 that HO-CDI rates significantly dropped. (See Figure 1.) UV disinfection now is done on every CD room and every critical care room at patient exit, following terminal cleaning. The hospital originally invested in three UV robots, but has expanded to five. Despite this, the use of UVC is limited by hours in the day — about 30 additional minutes are required per room for cleaning staff to set up the robot and run the program. Although it does not replace good elbow-grease cleaning methods, this added measure has been highly effective.
• At present, ~40% of our HO-CDI cases are known to be colonized on presentation to the hospital. In other words, despite our best efforts, patients with recognized colonization on presentation who developed active CDI during hospitalization are classified as HO-CDI. Current Centers for Medicare and Medicaid Services (CMS) punitive measures unfairly burden hospitals like ours that support a large skilled nursing population with a high rate of CD colonization, and undermine hospital efforts to control this important disease.
• Now that patient colonization can be confirmed, what next? It has yet to be determined whether preemptive treatment for CD colonization in patients admitted to the hospital, who are receiving antibacterials, is appropriate. Recognizing that ~40% of these patients will develop active CDI has prompted many of us to preemptively treat such patients. Urgent data are needed regarding which patients at risk are candidates for preemptive treatment, as well as the optimal approach.
REFERENCES
- Pechal A, Lin K, Allen S, Reveles K. National age trend in Clostridium difficile infection incidence and health outcomes in United States community hospitals. BMC Infect Dis 2016;16:682.
- Barrett ML,Owens PL. Clostidium difficile hospitalizations, 2011-2015. Aug. 18, 2018. U.S. Agency for Healthcare Research and Quality. Available at: https://www.hcup-us.ahrq.gov/reports/HCUPCDiffHosp2011-2015Rpt081618.pdf. Accessed Feb. 11, 2020.
Step-Wise Interventions for Hospital-Onset Clostridioides difficile
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
