Extracorporeal Membrane Oxygenation in the Emergency Department
March 15, 2020
Reprints
AUTHORS
Chandni Ravi, MD, Department of Emergency Medicine, Rutgers New Jersey Medical School, Newark, NJ
Ilya Ostrovsky, MD, Assistant Professor, Department of Emergency Medicine, Ultrasound Director, Rutgers New Jersey Medical School, Newark, NJ
PEER REVIEWER
Kimberly Boswell, MD, Assistant Professor of Emergency Medicine, University of Maryland School of Medicine, Baltimore
EXECUTIVE SUMMARY
- Extracorporeal membrane oxygenation (ECMO) is a process whereby blood is drained from a vein, pumped through a membrane oxygenator that removes carbon dioxide (CO2) and replenishes oxygen, and reintroduced into the body via an artery or a vein.
- ECMO can be considered in the emergency department for cardiac arrest refractory to resuscitation measures, shock refractory to fluids resuscitation and vasopressors, and acute severe respiratory failure.
- ECMO is associated with a high complication rate, both during cannulation and ongoing management; at least one significant complication has been noted to occur in more than half of the patients receiving ECMO.
- The decision to institute ECMO in the emergency department requires a major institutional commitment, and support from cardiology, vascular surgery, and critical care specialists is crucial.
Cardiopulmonary bypass in the emergency department (ED)? You must be crazy, or blindingly arrogant. Well, maybe. But to contemplate heroic measures in the ED requires a bit of arrogance. As described in this issue of Emergency Medicine Reports, extracorporeal membrane oxygenation (ECMO) can be lifesaving in selected patients, albeit with a high rate of complications and some long-term effects. Only a few emergency physicians in select hospitals in urban areas possibly will be able to implement ECMO in the ED. For the rest of us in community hospitals, awareness of this therapy and an understanding of potential candidates is the purpose of this article.
— Joseph Stephan Stapczynski, MD, Editor
Introduction
Extracorporeal life support (ECLS) comprises several modalities for temporary mechanical cardiopulmonary support, including extracorporeal carbon dioxide removal (ECCO2R), cardiopulmonary bypass (CPB), and extracorporeal membrane oxygenation (ECMO). ECMO provides cardiac and respiratory support in patients who have potentially reversible causes of cardiac or respiratory failure. While CPB uses a heart-lung machine to completely bypass cardiopulmonary circulation, ECMO functions by augmenting oxygenation, ventilation, and cardiac output.1 Deoxygenated blood is drawn from venous circulation; pumped through an external membrane oxygenator, which eliminates carbon dioxide (CO2) and replenishes oxygen; and returned to venous or arterial circulation.
Because of the growth in ECMO research and training of intensivists, the use of ECMO in the United States increased 433% from 2006 to 2011.2 The number of U.S. hospitals offering ECMO has grown from 108 in 2008 to 264 today.3 About 14,000 adult patients received ECMO between 1990 and 2014, with a rate of survival to discharge of 57% for those in respiratory failure and 41% for those in cardiac failure.4
Initiation of ECMO by the emergency department (ED) is a relatively new development, but with significant potential for growth. This is particularly seen at centers already performing ECMO in the intensive care unit (ICU).5 Familiarity with the indications and mechanisms of ECMO is important for emergency providers on the frontlines of managing critically ill patients who require aggressive resuscitation.
History
The first successful use of extracorporeal circulation in humans with a heart-lung machine was by Gibbon in 1953 for the repair of an atrial septal defect.6 In 1971, ECMO was used successfully for 75 hours in a 24-year-old patient with respiratory failure in the setting of traumatic transection of the thoracic aorta, who eventually recovered.7 Robert Bartlett became known as the “Father of ECMO” for his successful use of ECMO in a neonatal patient with meconium aspiration in 1975.8 National registry data collected between 1980 and 1987 demonstrated a survival rate of 81% in newborn patients with severe respiratory failure supported by ECMO.9 This inspired a series of randomized, controlled trials that showed encouraging results of ECMO use in neonates.10-12
The use of ECMO in the adult population was met with mixed results. Initial reports were discouraging, with randomized, controlled trials failing to show any statistical difference in survival between the conventional intensive care and ECMO groups.13,14 The conventional ventilatory support vs. extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR) trial was the first study of ECMO in adult patients to demonstrate an increase in survival. Patients with acute respiratory distress syndrome (ARDS) did better when managed at an ECMO center (but not necessarily placed on ECMO) compared to conventional management.15 ECMO also was used widely to treat adult patients during the H1N1 influenza pandemic of 2009.16
The ECMO to rescue lung injury in severe ARDS (EOLIA) study was a large, multicenter, randomized, controlled trial which demonstrated that ECMO was safe and appropriate to use with no increased mortality compared to conventional therapy. Although EOLIA was meant to study the effects of early ECMO initiation, it was terminated because of a high crossover rate. However, it highlighted the role of ECMO as a rescue modality that may improve mortality in patients who fail conventional and other rescue therapies for ARDS.17
What Is ECMO?
In ECMO, deoxygenated blood is drained from a vein, pumped through a membrane oxygenator that removes CO2 and replenishes oxygen, and then is reintroduced into the body via an artery or a vein.
Principles of ECMO
The main purpose of ECMO is to facilitate successful gas exchange of both carbon dioxide and oxygen. The membrane oxygenator functions similar to the native lung in many ways — removing dissolved carbon dioxide from the blood and adding oxygen to the hemoglobin within red cells. The movement of these gases across the membrane is dependent on different factors. In general, CO2 removal is more efficient than oxygen addition, leading to a potential imbalance in gas exchange. Oxygen exchange across the membrane oxygenator is dependent on the thickness of the blood film, membrane material, fraction of inspired oxygen (FiO2), and hemoglobin concentration. Carbon dioxide exchange is determined by the total surface area, blood flow, and “sweep gas” flow rate. Occasionally, CO2 may need to be added to the sweep gas to prevent respiratory alkalosis due to the excessive removal of CO2.18
Unlike traditional CPB in which the heart is stopped, ECMO does not require total anticoagulation with heparin to prevent thrombus formation because of the higher blood flow rates (4 L/min vs. 2 L/min with CPB). An ECMO circuit also lasts for weeks compared to hours with CPB.19
Configuration in ECMO
Venovenous (VV) ECMO is used in cases of isolated respiratory failure with preserved cardiac function. It takes deoxygenated blood from the venous circulation and returns oxygenated blood to the veins. The ECMO circuit exists in series with the lungs.20
In contrast, venoarterial (VA) ECMO provides partial or complete respiratory and circulatory support. It takes deoxygenated blood from the venous system and returns oxygenated blood to the arterial system. The circuit functions in parallel to the native heart and lungs.20 A dialysis membrane may be added to either circuit to provide continuous renal replacement therapy.21,22
ECMO in the ED is used primarily either as a temporizing measure for refractory shock (ECLS) or in the setting of cardiac arrest (extracorporeal cardiopulmonary resuscitation or ECPR).4,23
The ECMO Circuit
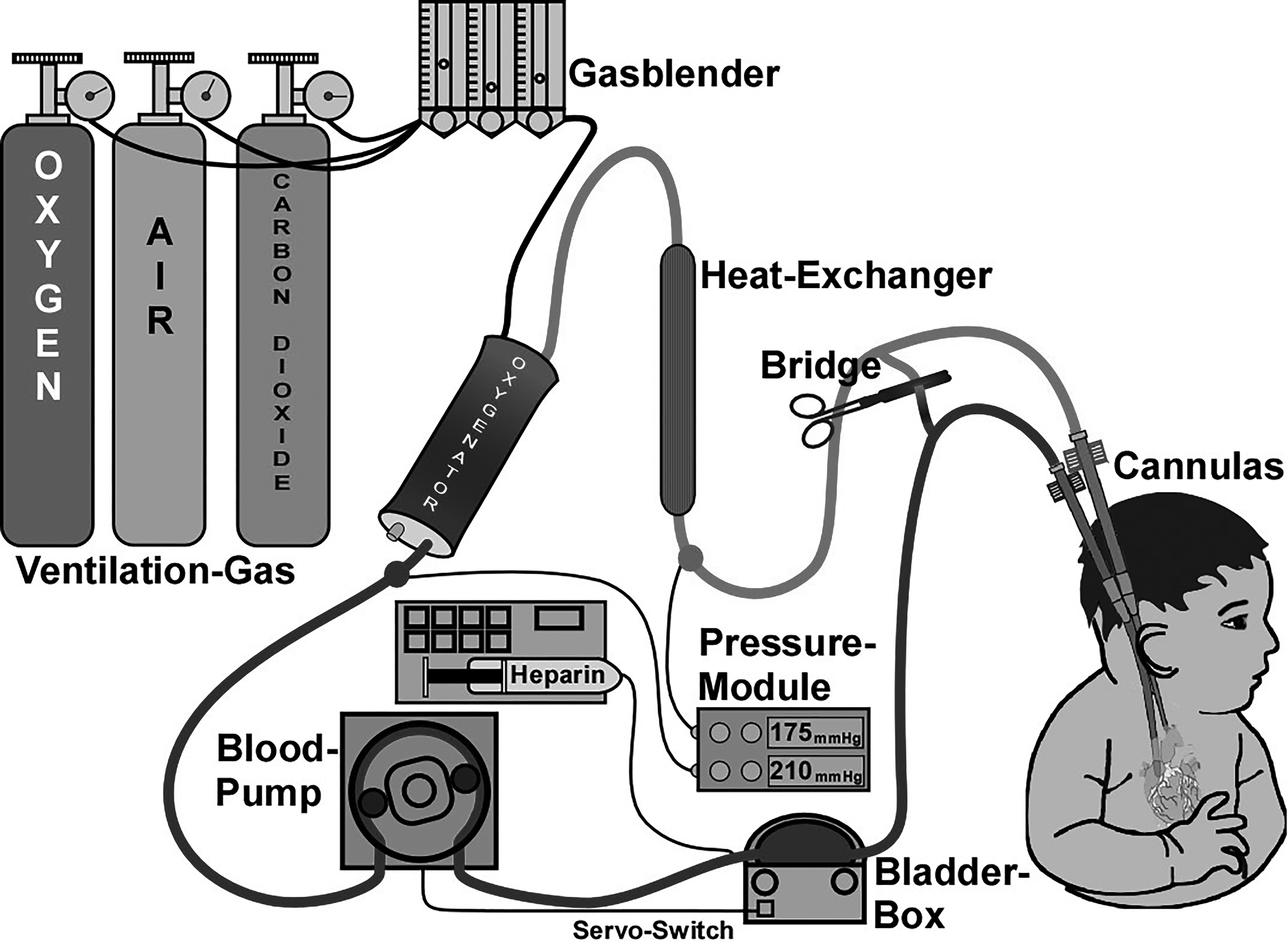
The basic ECMO circuit is a closed system consisting of a venous drainage cannula, pump, membrane oxygenator, heat exchanger, blender, central console, in-line monitors, and return cannula.20,23 (See Figure 1.)
Figure 1. Extracorporeal Membrane Oxygenation Circuit |
 |
|
Source: Jürgen Schaub / Wikimedia Commons / CC-BY-SA-2.0-DE / https://commons.wikimedia.org/wiki/File:Ecmo_schema-1-.jpg. |
Pumps
The pump is an essential component of the ECMO machine. It functions to circulate blood through the ECMO circuit as well as back to the patient while augmenting venous outflow. The pump must provide adequate flow to the patient within a safe range of pressures. Low output pressure can result in hemolysis, while a high output pressure can result in rupture of the circuit.24 Two types of pumps are used in ECMO devices: a roller pump and a centrifugal pump.
Roller pumps function by progressively compressing segments of circuit tubing and pushing blood through a curved raceway. This continuous peristalsis of the tubing system generates negative pressure that pulls venous blood from the patient, as well as positive pressure that pushes oxygenated blood back into the patient. The advantage of roller pumps is that they provide continuous flow in the circuit independent of preload. Their main disadvantage is that they cause hemolysis.18,20,24
Centrifugal pumps are smaller and consist of a pump head that contains a magnetically driven impeller spinning at a high rate. This generates a pressure differential that results in flow of blood across the pump. Centrifugal pumps are associated with a smaller degree of hemolysis.25 However, they also are associated with certain disadvantages, such as stagnation and heating in the pump head, thrombus formation in the outlet line at low flow rates, and inability to maintain a set flow rate.
Newer generations of centrifugal pumps have been designed to overcome most of these disadvantages and largely have replaced roller pumps. For instance, centrifugal pumps with magnetic levitations were found to be associated with improved outcomes.26 These modifications to centrifugal pumps have been key in making extended support via ECMO feasible.
Gas Exchange Membranes
The gas exchange membrane in an ECMO circuit performs the function of oxygen addition and CO2 removal. The efficiency or rated flow of an oxygenator is measured as the amount of desaturated blood that can be nearly fully saturated per minute. Membranes have been developed using various materials, including silicone rubber, polypropylene, polymethylpentene (PMP), polyvinyl chloride, polyurethane, and stainless steel.24
The two most commonly used membranes are the silicone membrane oxygenator and the hollow fiber oxygenator (HFO). The Kolobow silicone rubber membrane lung has been used in ECMO machines for decades. The silicone membrane was found to be very efficient at gas exchange; however, it had a few drawbacks. The size of the membrane requires adjustment based on the size of the patient. Therefore, hospitals needed to obtain different ECMO machines for the pediatric and adult populations. Resistance to flow was found to be high, which made the silicone rubber membrane less suitable for use with centrifugal pumps. Transportation also was challenging because of the bulky design and the longer duration required for priming.27
The newer class of hollow fiber PMP oxygenators have addressed most of these issues. These hollow fibers are capillary tubes that contain gas inside and blood on the surface. This allows gas exchange via a countercurrent mechanism. HFO membranes are more durable, extremely efficient at gas exchange, demonstrate minimal plasma leakage, and have a relatively low resistance to blood flow. Additionally, they have a lower incidence of thrombus formation, are easier to prime, are more compact, and are well suited for use with centrifugal blood pumps.28
Heat Exchanger
Loss of heat occurs during ECMO because of the exposure of the patient’s blood to a large extracorporeal surface area, particularly in children. For this reason, all ECMO circuits require a heat exchanger to maintain temperatures between 37°C and 40°C. Heat exchangers typically use the flow of heated water countercurrent to blood flow. Excessive heat must be prevented because it will lead to hemolysis and bubble formation. Newer generation membrane oxygenators contain an integrated heat exchange device, which reduces the number of components and simplifies the ECMO circuit design.18
In-Line Monitors
ECMO circuits contain in-line monitors that measure blood flow, chemistry, pressure, pH, oxyhemoglobin concentration, and partial pressure of CO2 (pCO2). These monitors are placed on both the venous and arterial ends of the circuit. They assist in the detection of problems, such as bubbles in the system, oxygenator failure, increased pressures, and disconnections. The oxyhemoglobin saturation on the venous access limb of the circuit in VA ECMO is especially useful in assessing the adequacy of oxygen delivery.24
Cannulation
Venovenous
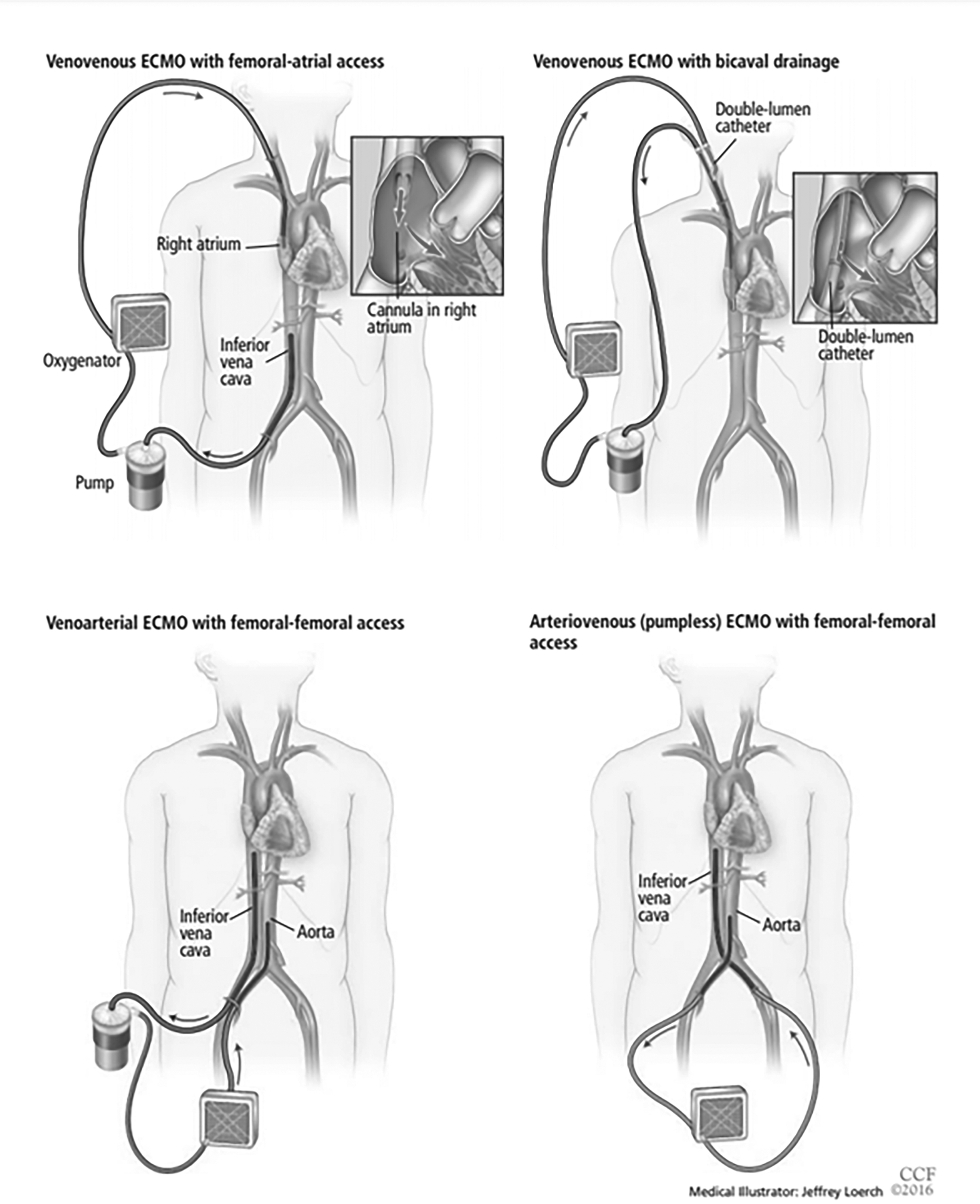
Three options are available for VV ECMO cannula placement (see Figure 2):
Figure 2. Four Configurations of Extracorporeal Membrane Oxygenation |
 |
|
Reprinted with permission from: Kulkarni T, Sharma NS, Diaz-Guzman E. Extracorporeal membrane oxygenation in adults: A practical guide for internists. Cleve Clin J Med 2016; 83(5):373-384. doi:10.3949/ccjm.83a.15021. Copyright © 2016 Cleveland Clinic Foundation. All rights reserved. |
- Femoro-atrial access typically involves drainage of deoxygenated blood from the femoral vein and return of oxygenated blood to the right atrium via the right internal jugular vein.23
- Bicaval access employs a single dual-lumen catheter that drains venous blood via the internal jugular vein and returns oxygenated blood directly into the right atrium.29 Single cannulation has the benefits of reduced recirculation and cannula site infection. However, transesophageal echocardiography or fluoroscopy is required to ensure appropriate placement.
- Bifemoral access involves the removal of deoxygenated blood from the inferior vena cava (IVC) via the femoral vein on one side and return of oxygenated blood to the right atrium via the contralateral femoral vein. Bifemoral access has a few advantages. Both femoral veins are large and easily accessible, thereby allowing rapid initiation of VV ECMO. Proximity to the femoral artery is useful in case VA support is required. Femoral cannulation also does not require transesophageal echocardiography or radiography for its placement.30
Venoarterial
Two basic options are available for VA ECMO cannula placement.
- Central cannulation is performed via a sternotomy in the operating room because it involves direct drainage of venous blood from the right atrium and return into the proximal ascending aorta. The main advantage of central cannulation is good venous drainage with reliable arterial return, thereby ensuring high flow rates perfusing the entire body. It is not without disadvantages because of its invasive nature and the need to immobilize the patient. Potential complications include sternotomy issues, aortic dissection, and thromboembolic events.31
- Peripheral cannulation placement is much easier because it can be performed via percutaneous approach at the bedside. Usually, a femoral vein and the ipsilateral femoral artery are used for vascular access. Other options include the internal jugular vein for venous access and the carotid or subclavian/ axillary artery for arterial access. The tip of the cannula is positioned in the mid-right atrium, and the arterial cannula is positioned in the common iliac artery.
Femoral arterial cannulation is associated with many risks, such as lower limb ischemia, pulmonary edema, and watershed phenomenon (the mixing of de-oxygenated blood from the patient’s left ventricle with oxygenated blood from the ECMO unit) resulting in coronary and cerebral hypoxia. These risks are only partially present in subclavian arterial cannulation.32 Performing cannulation under resuscitation conditions may be challenging, and the use of transesophageal echocardiography (TEE) can be very useful in these cases.
Anticoagulation
Bleeding and thrombotic complications are the major complications seen with ECMO. According to the Extracorporeal Life Support Organization (ELSO) Registry Report, the rate of circuit replacement due to thrombotic complications is about 20%.33 As blood is exposed to large non-endothelial surfaces in the ECMO circuit, inflammatory and coagulation cascades are stimulated. The underlying disease process also can give rise to both a bleeding tendency and a hypercoagulable state, depending on its effect on the hemostatic system.
Therefore, patients placed on ECMO need to be anticoagulated. The most common anticoagulant used is unfractionated heparin, which is administered as a continuous infusion.34 In ECLS, heparin typically is administered as a 100 IU/kg bolus once vascular access has been established. The heparin infusion is titrated to maintain an activated clotting time of 160-220 or an activated partial thromboplastin time of 45-55 seconds.35 Low molecular weight heparin also can be given subcutaneously in adult patients during ECMO, but it is used less commonly. Direct thrombin inhibitors (e.g., bivalirudin) and factor Xa inhibitors (e.g., rivaroxaban, argatroban) have been described but with few large-scale studies.36 Anticoagulation is withheld in patients who are at risk of bleeding.
ECMO in the ED
The advent of newer ECMO equipment and supportive evidence from prior studies has paved the way for its use as salvage therapy in the ED. Two terms associated with its use in the ED are ECLS and extracorporeal cardiopulmonary resuscitation (ECPR). ECLS is described as ECMO used to temporize a critically ill patient, while ECPR refers to the use of VA ECMO in cardiac arrest.
ECPR has been shown to play a role in ongoing refractory cardiac arrest,18 severe accidental hypothermia,37 near-drowning,38 pulmonary embolism,39 septic shock,40 and even severe electrolyte abnormalities.41 ECMO in the ED buys time for diagnostic workup or serves as a bridge to therapeutic interventions, such as percutaneous coronary intervention, rewarming, or damage control surgery in trauma.41
Indications for ECMO in the ED
The decision to initiate ECMO in a patient in the ED is complex and varies based on institutional policies, patient factors, and the underlying disease process. In general, younger and healthier patients with an acute reversible insult leading to rapid cardiopulmonary collapse with a definitive management plan in place tend to have better outcomes. The following are some indications for ED ECMO:
VA ECMO for Cardiac Arrest (ECPR). For the emergency provider, application of VA ECMO in ECPR has been the most exciting development. ECPR may be useful in reducing the time from arrest to restoration of cerebral perfusion.1 Ultrasound-guided femoral arterial and venous catheters are inserted with ongoing cardiopulmonary resuscitation (CPR). Once connected to the extracorporeal circuit, complete cardiopulmonary bypass is confirmed, and CPR is discontinued.
Currently there is increasing evidence that patients in out-of-hospital cardiac arrest started on ECPR have better outcomes when compared to traditional CPR. Successful initiation of ECMO by ED physicians has been demonstrated with improved neurologically intact survival.42,43 Witnessed arrest or ED arrest, ventricular fibrillation or ventricular tachycardia as the initial rhythm, age range 18 to 70 years, presumed cardiac cause, and minimal interruption in chest compressions have been associated with positive outcomes and are instances in which ECPR should be considered. The outcomes of ECPR with in-hospital cardiac arrest have been relatively more promising, likely because of the shorter time to initiation of bypass.
The main limitation of studies on ECPR is that most of the data are retrospective and from single centers; therefore, they are prone to bias and confounders. Neurological outcomes following ECPR also may appear to be better in the ECMO group because these patients also were more likely to receive therapeutic hypothermia and percutaneous coronary intervention, both of which are interventions known to improve outcomes following cardiac arrest.
VA ECMO for Refractory Shock (ECLS). ECLS has shown promising results when used in septic shock or cardiogenic shock related to acute myocardial infarction, massive pulmonary embolism, or myocarditis.1 The inability to wean a patient from CPB following cardiac surgery and primary graft failure following cardiac transplantation are other common indications for ECMO.18 These are conditions with potential for recovery with time or definitive treatment (percutaneous coronary intervention, device implantation, or cardiac transplant). However, ECMO increases left ventricular afterload and oxygen demand due to retrograde aortic flow and potentially can worsen left heart failure. To combat this, a left ventricular drain often is added to the circuit.
VV ECMO for Severe Acute Respiratory Failure. Persistent hypoxemia despite maximum ventilator therapy in acute respiratory failure is an indication for VV ECMO. Since it allows the lungs to rest, VV ECMO prevents the deleterious effects of positive pressure injury, ventilator complications, and oxidative stress.44 Most commonly, it has been used in patients with ARDS or as a bridge in those awaiting lung transplantation. The EOLIA trial demonstrated that emergency ECMO improves outcomes by “buying time” in extremely hypoxemic patients.45 The CESAR trial found that patients who received ECMO in ARDS had a higher survival rate without severe disability at six months.15 Several scoring systems, such as the Murray score, age-adjusted oxygenation index score, and acute physiology of stroke score, have been used to determine appropriate candidacy. According to the ELSO Registry Report, ECLS should be considered when the risk of mortality is 50% or greater and is indicated when the risk of mortality is 80% or greater. Unfortunately, employing these scoring systems in the emergent setting might be challenging.
Contraindications to ED ECMO
Conditions that are associated with a particularly poor outcome despite ECMO therapy typically are contraindications to its use. These include advanced age (> 65 years), chronic conditions such as end-stage heart failure or chronic obstructive pulmonary disease, irreversible multi-organ failure, untreatable malignancy, or conditions precluding the use of anticoagulation such as intracranial hemorrhage.
Patients with aortic dissections or aortic regurgitation are not ECMO candidates because there is a risk of propagating the dissection or over-distending the left ventricle leading to worsening congestive heart failure. For patients with ARDS, prolonged mechanical ventilation with high airway pressures is a relative contraindication.
Contraindications to ECPR include prolonged CPR without adequate tissue perfusion, unrecoverable heart disease, and non-transplant or left ventricular assist device candidates. Patients in these cases are relegated to a “bridge-to-nowhere” in that they cannot survive without ECMO and there is no hope for recovery.
Certain situations that previously were contraindications to ECMO are being reconsidered. Pregnancy and postpartum patients were considered high risk for bleeding and, therefore, not suitable candidates. There has been a change in this opinion recently because it has been demonstrated that they are at no increased risk for catastrophic bleeding when compared to non-
pregnant patients.46
Traditionally, multi-trauma and severe traumatic brain injury were thought to be absolute contraindications. The advent of heparin-bonded circuits has obviated the need for systemic anticoagulation, therefore allowing ECMO use in select patients.47 Morbid obesity also once was considered a contraindication because of complex cannulation, but a lack of association between body weight and mortality has been demonstrated, likely because of the development of newer cannulas.48
Candidate Selection
The key to successful outcomes with the use of ECMO in the ED is the selection of patients who are appropriate candidates. The provider should consider the underlying diagnosis, patient-specific risk factors, anticipated duration of support, viable exit strategy, indications, and contraindications. While scores cannot substitute for clinical gestalt, and candidacy ultimately is determined by the provider, several scores have been described to provide objective assistance in this process. The use of scoring systems also is beneficial when developing a protocol at the departmental or hospital level.
The PRESERVE49 and RESP50 scores have been investigated for identifying suitable patients for use of
VV ECMO in ARDS in the ICU setting. Therefore, they may not be generalizable to the ED population, who predominantly require VA ECMO for ECPR. Furthermore, external validation of PRESERVE and RESP scores demonstrated poor performance in patients with severe ARDS receiving ECLS.
The Survival After Veno-arterial ECMO (SAVE) score is designed to assist with the prediction of survival for adult patients undergoing ECMO for refractory cardiogenic shock. Scoring parameters include diagnosis, age, weight, pre-ECMO diastolic blood pressure and pulse pressure, duration of intubation pre-ECMO, peak inspiratory pressures, and the presence of multi-system dysfunction.51 The modified SAVE score, which is equivalent to the combined lactate value and SAVE score, showed improved outcome predictions for patients who underwent urgent VA ECLS in the ED.52 The ENCOURAGE score may be useful in predicting mortality in patients with an acute myocardial infarction in cardiogenic shock on VA ECLS.53
Another scoring system that may be particularly useful in the ED setting is the pre-ECMO Simplified Acute Physiology Score (SAPS) II score. SAPS II has been studied in patients who require initiation of ECMO in the ED for acute circulatory and/or respiratory failure. Pre-ECMO SAPS II scores of ≤ 80 were associated with higher mortality at 28 and 60 days when compared to those with scores of > 80.54
Providers should bear in mind that while they are interesting and potentially useful in the future, many of these scores are not used in routine practice. The external validation that has taken place is based on studies with small numbers.
Complications of ECMO
ECMO is associated with a high complication rate during both cannulation and ongoing management.55 (See Table 1.) At least one significant complication has been noted to occur in more than half of the patients on ECMO.56 Complications are commonly due to cannulation, bleeding, or infection.
Table 1. Complications Associated With ECMO |
|
Mechanical Complications |
|
|
Hemorrhagic Complications |
|
|
Other Complications |
|
Since ECMO patients need aggressive systemic anticoagulation because of the risk for arterial thrombosis, they are prone to hemorrhage at the cannulation site as well as other sites. Intracerebral hemorrhage (ICH) is the most devastating hemorrhagic complication seen in these patients and can lead to significant disability. Studies with higher rates of computerized tomography (CT) have described up to a 20% incidence of ICH. Because monitoring the patient’s neurological status accurately while the patient is sedated on ECMO is challenging, early head CT on admission has been proposed as a strategy for the timely detection of ICH.57 In patients who are at a higher risk for bleeding or who may have developed bleeding already, using heparin-bonded circuits without systemic anticoagulation or targeting lower levels of anticoagulation are recommended strategies.35
Complications related to cannulation include thrombus formation, limb ischemia, dissection or perforation of a vessel, cannula migration, pseudoaneurysm formation, and accidental decannulation leading to hemorrhage. Since ECMO requires a high rate of blood flow, large diameter cannulas are used. These can mechanically occlude flow to the distal artery. In a study of femoral arterial cannulation in
VA ECMO, peripheral vascular complications were reported in about 20% of patients, the majority (89%) of whom required surgery in the form of angioplasty, bypass, or amputation.58 The use of a distal perfusion catheter may help avoid ischemia. Risks of incorrect placement of cannulas is reduced with ultrasound-guided needle and guidewire insertion and TEE or fluoroscopy to confirm placement.
Infections are common in patients receiving ECMO. These may be catheter-related bloodstream infections, cannulation site infections, or worsening sepsis in patients with ARDS. Acute tubular necrosis leading to renal failure and the need for hemodialysis also is common and is reported in about 13% of patients on ECMO.4
Logistics/Systems of Care
For successful implementation of an ED ECMO program, a multi-disciplinary institutional commitment is necessary. Predetermined inclusion and exclusion criteria, as well as the threshold for discontinuing ECMO when a patient fails to improve, must be defined. Coordination is essential between emergency medical services (EMS), emergency medicine physicians, nursing staff, the blood bank, the cardiac catheterization lab, vascular surgeons, neurocritical care, and the intensive care unit. Prehospital EMS providers must be able to identify and transport potential ECMO candidates quickly. Emergency physicians must be competent in ECMO cannulation. Cardiology must be able and willing to provide definitive care for suitable patients in the form of left ventricular assist device or transplant. Capabilities for several days of bridging ECMO therapy and transplant must be available 24/7 from an infrastructural as well as nursing standpoint. Finally, a formal neuroprognostication scheme with continuous goals of care discussions by palliative care, case management, and social work is paramount.
In general, higher annual hospital ECMO volumes (> 30/year) are associated with improved mortality.59 Institutional capabilities in terms of the number of simultaneous ECMO patients also must be considered. Regional networks and communication with ECMO referral centers may be needed at many hospitals with resource limitations.
Institution-specific practices and guidelines likely exist, and abiding by those is crucial. Few emergency medicine physicians are able to cannulate for ECMO currently, and collaboration with surgical colleagues (cardiac, vascular, trauma, etc.) is necessary in most institutions to place a patient on ECMO.
Ethics
Because of the increasing use of ECMO, novel ethical dilemmas have emerged. For instance, should discussions with patients regarding their desires for resuscitation in the event of cardiac or respiratory arrest include a discussion about ECMO? Who decides when to stop the circuit and under what circumstances?60 Other complexities include the pre-existing code status, degree of input from the surrogate decision maker, ethical obligation of the physician, costs involved, and finally long-term quality of life. Some studies report long-term neurocognitive abnormalities in more than 50% of ECMO survivors.1 These factors must be weighed carefully when considering the use of extracorporeal measures in a patient.
REFERENCES
- Mosier JM, Kelsey M, Raz Y, et al. Extracorporeal membrane oxygenation (ECMO) for critically ill adults in the emergency department: History, current applications, and future directions. Crit Care 2015;19:431.
- Sauer CM, Yuh DD, Bonde P. Extracorporeal membrane oxygenation use has increased by 433% in adults in the United States from 2006 to 2011. ASAIO J 2015;61:31-36.
- Extracorporeal Life Support Organization (ELSO). ECMO Registry. Ann Arbor, MI. January 2020.
- Kulkarni T, Sharma NS, Diaz-Guzman E. Extracorporeal membrane oxygenation in adults: A practical guide for internists. Cleve Clin J Med 2016;83:373-384.
- Tonna JE, Johnson NJ, Greenwood J, et al; Extracorporeal REsuscitation ConsorTium (ERECT) Research Group. Practice characteristics of emergency department extracorporeal cardiopulmonary resuscitation (eCPR) programs in the United States: The current state of the art of emergency department extracorporeal membrane oxygenation (ED ECMO). Resuscitation 2016;107:38-46.
- Gibbon JH Jr. Application of a mechanical heart and lung apparatus to cardiac surgery. Minn Med 1954;37:171-185.
- Hill JD, O’Brien TG, Murray JJ, et al. Prolonged extracorporeal oxygenation for acute post-traumatic respiratory failure (shock-lung syndrome). Use of the Bramson membrane lung. N Engl J Med 1972;286:629-634.
- Bartlett RH, Gazzaniga AB, Jefferies MR, et al. Extracorporeal membrane oxygenation (ECMO) cardiopulmonary support in infancy. Trans Am Soc Artif Intern Organs 1976;22:80-93.
- Toomasian JM, Snedecor SM, Cornell RG, et al. National experience with extracorporeal membrane oxygenation for newborn respiratory failure. Data from 715 cases. ASAIO Trans 1988;34:140-147.
- Bartlett RH, Roloff DW, Cornell RG, et al. Extracorporeal circulation in neonatal respiratory failure: A prospective randomized study. Pediatrics 1985;76:479-487.
- O’Rourke PP, Crone RK, Vacanti JP, et al. Extracorporeal membrane oxygenation and conventional medical therapy in neonates with persistent pulmonary hypertension of the newborn: A prospective randomized study. Pediatrics 1989;84:957-963.
- [No authors listed]. UK collaborative randomised trial of neonatal extracorporeal membrane oxygenation UK Collaborative ECMO Trail Group. Lancet 1996;348:75-82.
- Morris AH, Wallace CJ, Menlove RL, et al. Randomized clinical trial of pressure-controlled inverse ratio ventilation and extracorporeal CO2 removal for adult respiratory distress syndrome. Am J Respir Crit Care Med 1994;149:295-305.
- Zapol WM, Snider MT, Hill JD, et al. Extracorporeal membrane oxygenation in severe acute respiratory failure. A randomized prospective study. JAMA 1979;242:2193-2196.
- Peek GJ, Mugford M, Tiruvoipati R, et al. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): A multicentre randomised controlled trial. Lancet 2009;374:1351-1363.
- Australia and New Zealand Extracorporeal Membrane Oxygenation (ANZ ECMO) Influenza Investigators, Davies A, Jones D, et al. Extracorporeal membrane oxygenation for 2009 influenza A(H1N1) acute respiratory distress syndrome. JAMA 2009;302:1888-1895.
- Combes A. Extracorporeal membrane oxygenation (ECMO) for severe acute respiratory distress syndrome (ARDS). The EOLIA (ECMO to rescue Lung Injury in severe ARDS) trial: A multicenter, international, randomized, controlled open trial [in French]. Reanimation 2011;20:49-61.
- Allen S, Holena D, McCunn M, et al. A review of the fundamental principles and evidence base in the use of extracorporeal membrane oxygenation (ECMO) in critically ill adult patients. J Intensive Care Med 2011;26:13-26.
- Kolla S, Awad SS, Rich PB, et al. Extracorporeal life support for 100 adult patients with severe respiratory failure. Ann Surg 1997;226:544-566.
- Jayaraman AL, Cormican D, Shah P, Ramakrishna H. Cannulation strategies in adult veno-arterial and veno-venous extracorporeal membrane oxygenation: Techniques, limitations, and special considerations. Ann Card Anaesth 2017;20(Suppl):S11–S18.
- Marasco SF, Lukas G, McDonald M, et al. Review of ECMO (extra corporeal membrane oxygenation) support in critically ill adult patients. Heart Lung Circ 2008;17(Suppl 4):S41-S47.
- Van Dyk M. The use of CRRT in ECMO patients. The Egyptian Journal of Critical Care Medicine 2018;6:95-100.
- Squiers JJ, Lima B, DiMaio JM. Contemporary extracorporeal membrane oxygenation therapy in adults: Fundamental principles and systematic review of the evidence. J Thorac Cardiovasc Surg 2016;152:20-32.
- Lequier L, Horton SB, McMullan DM, Bartlett RH. Extracorporeal membrane oxygenation circuitry. Pediatr Crit Care Med 2013;14(Suppl 1):S7-S12.
- Toomasian JM, Bartlett RH. Hemolysis and ECMO pumps in the 21st century. Perfusion 2011;26:5-6.
- Luciani GB, Hoxha S, Torre S, et al. Improved outcome of cardiac extracorporeal membrane oxygenation in infants and children using magnetic levitation centrifugal pumps. Artif Organs 2016;40:27-33.
- Kolobow T, Bowman RL. Construction and evaluation of an alveolar membrane heart-lung. Trans Am Soc Artif Intern Organs 1963;9:238-243.
- Khoshbin E, Roberts N, Harvey C, et al. Poly-methyl pentene oxygenators have improved gas exchange capability and reduced transfusion requirements in adult extracorporeal membrane oxygenation. ASAIO J 2005;51:281-287.
- Bermudez CA, Rocha RV, Sappington PL, et al. Initial experience with single cannulation for venovenous extracorporeal oxygenation in adults. Ann Thorac Surg 2010;90:991-995.
- Burrell AJC, Ihle JF, Pellegrino VA, et al. Cannulation technique: Femoro-femoral. J Thorac Dis 2018;10(Suppl 5):S616-S623.
- Pavlushkov E, Berman M, Valchanov K. Cannulation techniques for extracorporeal life support. Ann Transl Med 2017;5:70.
- Napp LC, Kühn C, Hoeper MM, et al. Cannulation strategies for percutaneous extracorporeal membrane oxygenation in adults. Clin Res Cardiol 2016;105:283-296.
- Extracorporeal Life Support Organization. Registry Report. Ann Arbor: University of Michigan; 2013.
- Mulder MM, Fawzy I, Lancé MD. ECMO and anticoagulation: A comprehensive review. Neth J Crit Care 2018;26:6-13.
- Swol J, Belohlávek J, Brodie D, et al. Extracorporeal life support in the emergency department: A narrative review for the emergency physician. Resuscitation 2018;133:108-117.
- Cho HJ, Kim DW, Kim GS, Jeong IS. Anticoagulation therapy during extracorporeal membrane oxygenator support in pediatric patients. Chonnam Med J 2017;53:110-117.
- Jarosz A, Darocha T, Kosiński S, et al. Extracorporeal membrane oxygenation in severe accidental hypothermia. Intensive Care Med 2015;41:169-170.
- Kim KI, Lee WY, Kim HS, et al. Extracorporeal membrane oxygenation in near-drowning patients with cardiac or pulmonary failure. Scand J Trauma Resusc Emerg Med 2014;22:77.
- Yusuff HO, Zochios V, Vuylsteke A. Extracorporeal membrane oxygenation in acute massive pulmonary embolism: A systematic review. Perfusion 2015;30:611-616.
- Takauji S, Hayakawa M, Ono K, Fujita S. Venoarterial extracorporeal membrane oxygenation for severe sepsis and septic shock in 3,195 adults. Crit Care Med 2016;44(Suppl 1):440-440.
- Palatinus JA, Lieber SB, Joyce KE, Richards JB. Extracorporeal membrane oxygenation support for hypokalemia-induced cardiac arrest: A case report and review of the literature. J Emerg Med 2015;49:159-164.
- Johnson NJ, Acker M, Hsu CH, et al. Extracorporeal life support as rescue strategy for out-of-hospital and emergency department cardiac arrest. Resuscitation 2014;85:1527-1532.
- Bellezzo-JM, Shinar Z, Davis DP, et al. Emergency physician-initiated extracorporeal cardiopulmonary resuscitation. Resuscitation 2012;83:966.
- Schmidt M, Stewart C, Bailey M, et al. Mechanical ventilation management during extracorporeal membrane oxygenation for acute respiratory distress syndrome: A retrospective international multicenter study. Crit Care Med 2015;43:654-664.
- Gattinoni L, Vasques F, Quintel M. Use of ECMO in ARDS: Does the EOLIA trial really help? Crit Care 2018;22:171.
- Sharma NS, Wille KM, Bellot SC, Diaz-Guzman E. Modern use of extracorporeal life support in pregnancy and postpartum. ASAIO J 2015;61:110-114.
- Ontaneda A, Annich GM. Novel surfaces in extracorporeal membrane oxygenation circuits. Front Med (Lausanne) 2018;5:321.
- Al-Soufi S, Buscher H, Nguyen ND, et al. Lack of association between body weight and mortality in patients on veno-venous extracorporeal membrane oxygenation. Intensive Care Med 2013;39:1995-2002.
- Schmidt M, Zogheib E, Rozé H, et al. The PRESERVE mortality risk score and analysis of long-term outcomes after extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. Intensive Care Med 2013;39:1704-1713.
- Schmidt M, Bailey M, Sheldrake J, et al. Predicting survival after extracorporeal membrane oxygenation for severe acute respiratory failure. The Respiratory Extracorporeal Membrane Oxygenation Survival Prediction (RESP) score. Am J Respir Crit Care Med 2014;189:1374-1382.
- Schmidt M, Burrell A, Roberts L, et al. Predicting survival after ECMO for refractory cardiogenic shock: The survival after veno-arterial-ECMO (SAVE)-score. Eur Heart J 2015;36:2246-2256.
- Chen WC, Huang KY, Yao CW, et al.The modified SAVE score: Predicting survival using urgent veno-arterial extracorporeal membrane oxygenation within 24 hours of arrival at the emergency department. Crit Care 2016;20:336.
- Muller G, Flecher E, Lebreton G, et al. The ENCOURAGE mortality risk score and analysis of long-term outcomes after VA-ECMO for acute myocardial infarction with cardiogenic shock. Intensive Care Med 2016;42:370-378.
- Kim KI, Lee HS, Kim HS, et al. The pre-ECMO simplified acute physiology score II as a predictor for mortality in patients with initiation ECMO support at the emergency department for acute circulatory and/or respiratory failure: A retrospective study. Scand J Trauma Resusc Emerg Med 2015;23:59.
- Zangrillo A, Landoni G, Biondi-Zoccai G, et al. A meta-analysis of complications and mortality of extracorporeal membrane oxygenation. Crit Care Resusc 2013;15:172-178.
- Combes A, Leprince P, Luyt CE, et al. Outcomes and long-term quality-of-life of patients supported by extracorporeal membrane oxygenation for refractory cardiogenic shock. Crit Care Med 2008;36:1404-1411.
- Cavayas YA, Del Sorbo L, Fan E. Intracranial hemorrhage in adults on ECMO. Perfusion 2018;33(1_suppl):42-50.
- Aziz F, Brehm CE, El-Banyosy A, et al. Arterial complications in patients undergoing extracorporeal membrane oxygenation via femoral cannulation. Ann Vasc Surg 2014;28:178-183.
- Barbaro RP, Odetola FO, Kidwell KM, et al. Association of hospital-level volume of extracorporeal membrane oxygenation cases and mortality. Analysis of the extracorporeal life support organization registry. Am J Respir Crit Care Med 2015;191:894-901.
- Courtwright AM, Robinson EM, Feins K, et al. Ethics committee consultation and extracorporeal membrane oxygenation. Ann Am Thorac Soc 2016;13:1553-1558.
Extracorporeal membrane oxygenation (ECMO) can be lifesaving in selected patients, albeit with a high rate of complications and some long-term effects. Awareness of this therapy and an understanding of potential candidates is the purpose of this article.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
