COVID-19: Pandemic 2020
July 1, 2020
Reprints
AUTHOR
James Wilde, MD, FAAP, Professor of Emergency Medicine, Augusta University, Augusta, GA
PEER REVIEWER
Dean L. Winslow, MD, Professor of Medicine, Division of General Medical Disciplines, Division of Infectious Diseases and Geographic Medicine, Stanford University School of Medicine
EXECUTIVE SUMMARY
• Coronavirus disease 2019 (COVID-19) is caused by a coronavirus, which is responsible for diseases such as severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS), but also for the common cold.
• Many patients with COVID-19 may be asymptomatic but capable of spreading the disease. Older patients and those with comorbidities are at risk for more severe disease and death. Healthcare workers tend to get more severe disease as well.
• Children do not seem to be as susceptible to the disease, but there are reports of a Kawasaki-like illness associated with COVID.
• To date, there is no effective treatment for the disease other than supportive care.
• There are many different presentations of the disease, not just respiratory. Therefore, precautions should be taken to protect oneself during the pandemic.
Coronavirus disease 2019 (COVID-19) is a rapidly spreading disease, and our knowledge is rapidly growing. This monograph will concentrate on the history of the pandemic. While some management is discussed, these change rapidly with time. It is important to stay abreast of the most recent recommendations from sources such as the National Institutes of Health and Centers for Disease Control and Prevention.
— Sandra M. Schneider, MD, Editor
Background
The world is in the midst of a pandemic caused by a novel coronavirus. The pathogen, now named severe acute respiratory syndrome coronavirus-2, or SARS-CoV-2, came to world attention in January 2020. Initial reports used the term “Wuhan coronavirus” to designate the new virus. The disease it causes has come to be known as COVID-19, short for coronavirus disease 2019.1 When the first human cases were identified in Wuhan, China, in December 2019, the virus likely moved from its animal reservoir in late November and crept silently into Wuhan hospitals over the next few weeks.2,3 The alarm was sounded by several brave Chinese physicians in December, when they started noting unusual cases of pneumonia marked by acute respiratory distress syndrome (ARDS). On Dec. 31, Chinese authorities alerted the World Health Organization (WHO) China Country office that 27 cases of pneumonia of unknown origin had been detected in and around Wuhan. By Jan. 9, Chinese researchers announced that they had mapped the new coronavirus’s genome, which appeared to match the genome of a coronavirus previously isolated from bats. As of Jan. 20, there were 278 cases reported in China, mostly in and around Wuhan, and four cases outside of China, with an apparent case fatality rate of 2%. The first case of this novel coronavirus was identified in Thailand on Jan. 13, in Japan on Jan. 15, and in South Korea on Jan. 20. WHO started the first of their daily updates on the progress of COVID-19 on Jan. 20.
By Jan. 26, a total of 2,014 cases had been confirmed globally, including 29 cases from 10 countries outside of China. In 26 of those 29 cases, there was a travel history from Wuhan City, China. On Jan. 17, the Centers for Disease Control and Prevention (CDC) began public health entry screening at three U.S. international airports that received the largest number of travelers from Wuhan. On Jan. 21, the CDC activated its emergency operations center, and on Jan. 31, President Trump declared a public health emergency and announced restrictions on travel from China.
Origins of COVID-19
The first Situation Update from WHO regarding COVID-19 was published online on Jan. 20. In that update, the events surrounding the origins of the outbreak were summarized.4 The initial report concerned 44 patients who were identified with pneumonia of initially unknown etiology from Dec. 16, 2019, to Jan. 2, 2020, in Wuhan City, China, a city of more than 11 million people in Hubei Province.5 On Jan. 11, WHO received further information from Chinese authorities that the outbreak was associated with an outdoor seafood market in Wuhan City that featured assorted live animals for sale. Specimens obtained from patients admitted to a Wuhan hospital on Dec. 27 were analyzed by a team from the Chinese Center for Disease Control and Prevention and were found to contain a novel virus with 86.9% sequence identity to a previously published SARS-like coronavirus.6 Electron micrographs of infected cells showed a virion with distinctive spikes consistent with the Coronaviridae family. Investigators from the Wuhan Institute of Virology examined samples from seven patients with severe pneumonia and found a coronavirus with 96% identity at the whole-genome level to bat coronavirus.7 Although it appears that this virus was originally derived from bats, there is speculation about an as-yet unconfirmed intermediate host that allowed it to pass into humans. One prime candidate for the intermediate host is the pangolin, an animal that is sold for medicinal purposes and as a culinary delicacy in China.8
There also has been speculation on social media and in common print media that the virus originated from the Wuhan Institute of Virology as a pathogen that had been engineered to become infectious for humans. There is no conclusive proof for this hypothesis. Detailed analysis of the genome by researchers at the Scripps Research Institute concluded that SARS-CoV-2 “is not the product of purposeful manipulation.”9 An April 30 news release from the Office of the Director of National Intelligence stated, “The Intelligence Community also concurs with the wide scientific consensus that the COVID-19 virus was not manmade or genetically modified.”10
The Virus
Coronaviruses are single-stranded zoonotic RNA viruses that have many hosts in the animal kingdom.11,12 Prior to the identification of SARS-CoV-2, there were six coronaviruses known to infect humans. Four of these coronaviruses are common causes of upper respiratory infections in children and tend to circulate in humans in a seasonal pattern between December and May. Coronaviruses can mutate and recombine, and occasionally those mutated strains can spread from animals to humans. This is what happened in the case of both SARS-CoV, which emerged briefly in 2002 and disappeared by 2003, and MERS-CoV, which emerged in 2013 and is still circulating in parts of the Middle East. Infections due to SARS-CoV and MERS-CoV both caused high mortality in victims, with death rates of approximately 10% and 30%, respectively. Both are beta coronaviruses, like SARS-CoV-2.
An interesting paper in 2007 discussed the ability of viruses to mutate and jump species and even speculated that, “The presence of a large reservoir of SARS-CoV like viruses, in horseshoe bats together with the culture of eating exotic animals in southern China, is a time bomb. The possibility of the reemergence of SARS and other novel viruses from animals or laboratories and therefore the need for preparedness, should not be ignored.”13
Early studies showed that the SARS-CoV-2 virus uses S-proteins on the surface of the virion to bind to host receptors, and that these proteins have a high affinity for human angiotensin converting-enzyme 2 (ACE2) receptors.1 These receptors are the primary binding site used by SARS-CoV-2 to gain access to human cells during infection.14 ACE2 receptors are found in many human cell types, including in the lungs, heart, kidneys, and intestines. They are found throughout epithelial cells of the nose, mouth, and lungs. They are particularly abundant on type 2 pneumocytes in the alveoli.
The pathophysiology of SARS-CoV-2 might be explained in part by interference with ACE2 receptors.15 The renin-angiotensin system is a biochemical pathway that converts angiotensin I to angiotensin II (ANG II). It is an important mechanism for the regulation of blood pressure. ANG II increases blood pressure, but it also can lead to inflammation. Left unchecked, it actually may cause damage to human tissues. ACE2 helps to modulate that effect by breaking down ANG II. When SARS-CoV-2 binds to ACE2 receptors, it leads to decreased activity of the enzyme, which leaves ANG II relatively unchecked and, thus, able to cause higher degrees of inflammation. Research is ongoing to determine whether manipulation of the interaction between ACE2 and SARS-CoV-2 might lead to prophylactic or therapeutic interventions.
Transmission, Incubation Period, Contagion, and Viral Shedding
The CDC has summarized the data on transmission of the virus in their Interim Infection Prevention and Control Recommendations.16 The available evidence indicates that SARS-CoV-2 is spread primarily by respiratory droplets after a cough or sneeze. These droplets may gain access to an uninfected person by inhalation or by landing in the mouth, nose, or eyes. The degree of transmission from contaminated surfaces is not clear. Another report from the CDC provides evidence for transmission from singing during a choir practice.17
Early in the pandemic, the incubation period was estimated to be five to 14 days, with a median of 5.1 days, based on cases with clearly identified exposure and symptom onset.18 The mathematical model used to calculate this interval also indicates that 97.5% of people will have symptoms by day 11, and 99% of people will have symptoms by day 14. This and other similar estimations of the incubation period are the basis for the current length of quarantine periods for patients with known exposures to COVID-19.
The period of contagion for respiratory infections, such as influenza, are well known to start before the actual onset of symptoms in the index patient. COVID-19 also appears to be transmissible before the onset of symptoms. One report from China evaluating a family cluster clearly documented transmission during the incubation period.19 Another Chinese study showed that 12.6% of transmission may be pre-symptomatic.20 A study from Singapore very early in the outbreak in that country showed that pre-symptomatic transmission had occurred in at least 6.4% of the first 157 locally acquired infections.21 A study in a U.S. skilled nursing facility demonstrated a mean duration of three days from a positive test for COVID to the onset of symptoms.22 The testing cycle thresholds (Ct) for the real-time polymerase chain reaction (RT-PCR) results in that study were no different between COVID-19-positive patients who were symptomatic, pre-symptomatic, and asymptomatic, suggesting that the viral load is similar for all groups and, thus, all are potentially infectious.
An early report from China demonstrated that the highest viral load was detected in the nasopharynx at the time of symptom onset, with levels dropping sharply by day 10 of illness.23 This is in sharp contrast to SARS-CoV, which was notable for increased levels of infectiousness at seven to 10 days after the onset of illness, making containment of that 2003 outbreak much more practical.24 Another group from China looked at temporal patterns of viral shedding in 94 patients and found a similar pattern of high viral load in throat swabs at the onset of symptoms, declining rapidly over 10 days, but remaining detectable for up to three weeks.25 Based on an analysis of 77 transmission pairs, the researchers inferred that infectiousness actually may peak zero to two days before symptom onset, with as much as 44% of secondary cases becoming infected before symptom onset in the index case. Another group from Hong Kong tested sputum rather than nasopharyngeal swabs and again found a high viral load at presentation. Of interest, that group also detected SARS-CoV-2 in blood in 22% of patients tested and in rectal swabs in 27%. No patients had virus detected in urine. This group also demonstrated serum antibodies to two different COVID-19 proteins in 88% to 100% of patients 10 days or later after symptom onset.26
Data on viral load and communicability helps to inform recommendations for discontinuing isolation of patients with documented COVID-19 infection. The current recommendation from the CDC is to discontinue isolation 10 days after the onset of symptoms if the symptoms are improving and at least three days have passed since the last fever. In that recommendation, CDC experts acknowledge that low-level viral shedding or residual particles of viral RNA may continue for as long as six weeks after infection. It is not known if PCR-positive samples beyond day 10 of illness represent infectious virus, but once patients are clinically recovered, they are “likely no longer infectious.”27
Estimates of Basic Reproduction Number
One of the early priorities in studying the transmission of COVID-19 was to calculate a basic reproduction number, also known as R0. This number provides an estimate of the transmissibility of a virus, or how many additional new infections can be expected in a naïve population after exposure to an infectious person. There are several different methods for calculating the R0, which can change based on actions taken to protect the uninfected, such as isolation of infected individuals and social distancing. The first estimate was based on data obtained from China from Dec. 31 to Jan. 28, the first month of the outbreak and before travel to other provinces was restricted.28 These authors estimated an R0 of 2.68. Since then, many other authors have calculated an R0 for COVID-19 based on varying patient populations. One summary of 12 different estimates found a median of 2.78 and a range of 1.5 to 6.49.29 An R0 greater than 1 indicates that the number infected is likely to increase, while an R0 less than 1 indicates that the number of infected will decrease.
Outbreak in China
When the world first learned about the novel coronavirus in Wuhan in mid-January, the outbreak was still relatively small. By Jan. 20, there were only 278 confirmed cases inside China, and six deaths.4 On Jan. 23, the Chinese government instituted a quarantine and halted all train and air travel from Wuhan City to other parts of China in an effort to contain the spread of the virus. Two days later, all of Hubei province was placed under quarantine. However, by that time, a large and rapidly growing number of infections had already been detected in China outside of Hubei province, including at least 10 in Beijing and Shanghai. On Jan. 31, both Italy and the United States halted all airline flights between their countries and China. By that same date, two cases had already been detected in Italy and six cases had been detected in the United States. The number of infected patients rose rapidly in China during the next five weeks. (See Table 1.)
Table 1. Total Infections and Deaths Due to COVID-19, China, 2020 |
||
|
Date |
Number of Confirmed Cases |
Number of Deaths |
|
Jan. 20 |
278 |
6 |
|
Jan. 31 |
9,720 |
213 |
|
Feb. 15 |
66,576 |
1,524 |
|
March 1 |
79,968 |
2,873 |
|
March 15 |
81,048 |
3,204 |
|
April 1 |
82,631 |
3,321 |
|
April 15 |
83,745 |
3,352 |
|
Source: World Health Organization Situation Update |
||
Some perspective is helpful in looking at these statistics. Even as late as Feb. 15, when China reported more than 66,000 infections and 1,524 deaths, the world outside of China had 526 total cases of COVID-19 and only two deaths. No country outside of China had yet reached a total of 100 cases. Further analysis of the data available on the WHO website shows that as of Feb. 15, more than 80% of the cases reported in China were in Hubei province, the home province of Wuhan. This is despite the fact that Hubei accounts for less than 5% of the total population of China. Why the infection was so much more concentrated in Hubei (and remains so to this day) is difficult to explain, but it might explain why extreme measures regarding social distancing and sheltering in place had not yet been undertaken outside of China.
Several early reports from China described the characteristics of COVID-19 infections.30,31 One report described 99 patients recruited from a single center from Jan. 1 to Jan. 20 in Wuhan.32 In this study, 49% of patients were said to have a history of exposure to the seafood market in Wuhan. The mean age was 55.5 years, and COVID-19 was confirmed by RT-PCR in all. The primary symptoms were fever (83%), cough (82%), and shortness of breath (31%). A small minority had sore throat (5%), rhinorrhea (4%), and diarrhea (2%). Two-thirds of patients had markedly increased erythrocyte sedimentation rate, ferritin level, and C-reactive protein. All had evidence for pneumonia, and at the time of publication, 11% had died and 62% were still in the hospital.
Another study looked at 1,099 patients infected with SARS-CoV-2 in 30 provinces in China.30 All patients had laboratory proven COVID-19. Forty-three percent of the patients were from Wuhan, and 72% of the remainder had contact with residents of Wuhan. Nine hundred twenty-six of 1,099 patients were classified as having “non-severe” disease. The mean incubation period was four days, and the mean age of the patients was 43 years. Fever was present in 43% at the time of admission, but fever was noted in 89% at some time during the hospital stay. Nine hundred seventy-five chest computed tomography (CT) scans were performed, and 86% showed abnormal results, including ground glass opacities in 56%. Eighteen percent of patients with non-severe disease had no abnormalities on chest radiograph or chest CT scan. Lymphopenia was present in 83%, and most of the patients had elevations of C-reactive protein. Overall, severe illness occurred in 16% after admission to the hospital, and only five patients died. The median duration of hospitalization was 12 days.
The largest case series to date described the epidemiology of the outbreak as of Feb. 11.31 At that point, there were 44,672 confirmed cases in China, and another 28,000 suspected cases. Analysis of the confirmed cases showed only 1% were younger than 10 years of age, another 1% were 10-19 years of age, and 87% were older than 30 years of age. Mild cases accounted for 81% of patients, severe for 14%, and critical for 5%. The overall case fatality rate was 2.3%.
A plateau in the number of infected patients appeared to be reached in China by the beginning of March, two months after the outbreak was first identified. After March 1, the total number of infected has risen by only about 5%, and few new infections have been detected in China since May 1. As of June 1, the total number of deaths in China was just under 5,000, for an overall case fatality rate of 5.5%. But just as China appeared to be reaching the end of its outbreak, events took a marked turn for the worse in the rest of the world.
The Diamond Princess Outbreak
The largest cluster of COVID-19 infections outside of mainland China during the month of February occurred from Feb. 3 to Feb. 23 aboard the Diamond Princess cruise ship.33 The ship was quarantined in Yokohama, Japan, on Feb. 3 after returning from a two-week cruise to three countries. The quarantine was ordered when authorities in Japan learned that a passenger had departed the ship in Hong Kong on Jan. 25 and subsequently was found to have COVID-19 infection. The ship had approximately 3,700 passengers and crew. The median age of the passengers was 69 years. On Feb. 5, passengers were quarantined in their cabins, but crew members continued to work. Initial testing focused on passengers with fever or respiratory symptoms as well as their close contacts. All those with positive tests were taken off the ship and hospitalized. After this cohort, testing was expanded to allow phased disembarkation of passengers. Eventually, those without symptoms or close contacts were required to complete a 14-day quarantine on board the ship before they could depart.
A total of 712 passengers and crew (19.2%) had positive tests for SARS-CoV-2. More than 46% of positive patients were asymptomatic at the time of the test. Among 381 symptomatic patients, 9.7% required intensive care, and nine died (1.3%). As of March 13, 11 symptomatic U.S. passengers remained in the hospital in Japan.
This outbreak represented an interesting case study in the early stages of the COVID pandemic. It demonstrated that the disease is highly communicable, but infection is not inevitable, especially if uninfected people are removed from sources of contagion. Eighty percent of the passengers and crew of the Diamond Princess escaped infection. The case fatality rate of 1.3% in a largely elderly cohort of infected people also gave a glimpse into what could be expected as the infection continued to spread around the globe. The large number of asymptomatic patients who were positive for the virus provided a hint into the difficulty of containing any large outbreak.
COVID Appears in Europe
Both Iran and Italy reported marked increases in COVID-19 infections starting in the last week of February, two to three weeks before the rest of Europe and North America began to see acceleration in the number of infections. Why these countries were affected so much earlier than surrounding countries is unclear. The situation was particularly dire in Italy, with the Lombardy region being the epicenter.34 The outbreak in Italy served as an illustration of what was to come to the United States. Soon after Italy was inundated, the rest of Europe began to experience high rates of infection as well. As of the end of May, infection rates were in steep decline all over Europe, but the toll was high. As of May 31, Europe had 180,000 total deaths, half the world total, and far above the 102,000 at that point in the United States.
The Seattle Nursing Home Outbreak
On Feb. 28, a case of COVID-19 was identified in a skilled nursing facility in King County, WA.35 This facility cared for 130 residents, with a staff of 170. At the time the index patient was identified, at least 45 residents and staff had symptoms of respiratory illness. The CDC aided local public health officials to investigate the outbreak. By March 18, 101 residents, 50 healthcare personnel, and 16 visitors, all of whom were epidemiologically linked to the facility, were confirmed with COVID-19 infections. Most of the residents with confirmed infections had respiratory symptoms, but seven residents had no symptoms at all.
The index patient in the outbreak, a 73-year-old woman, was admitted to the hospital on Feb. 27 and died on March 2. The median age of infected facility residents was 83 years. Fifty-four percent were hospitalized, and the case fatality rate was 33.7%. Ninety-four percent of facility residents had chronic underlying health conditions.
Among the 16 infected visitors, 50% were hospitalized, but only one died (6%). The median age for this group was 62.5 years. Only three of 50 infected healthcare personnel were hospitalized, and none of them died. The median age for this group was 43.5 years.
This episode illustrated the potential for rapid spread of COVID-19 in a vulnerable population of elderly adults. Until this outbreak was identified, the primary screening algorithms focused on people with known contacts to COVID-19 patients or travelers from regions with high levels of COVID-19 activity. In response to this outbreak, on March 10, the governor of Washington ordered a policy of mandatory screening of healthcare workers and visitor restrictions for all licensed nursing homes and assisted living facilities in the state.
Another report from the same region and time period illustrated the potential benefit of social distancing.36 During March 5-9, two residents of a senior independent and assisted living community in Seattle were hospitalized with confirmed COVID-19 infections. On March 6, social distancing began, including isolation of residents in their rooms, with no communal meals or activities. In addition, no visitors were allowed in the facility, and staff member screening began. Eighty residents (mean age 86 years) and 62 staff members (mean age 42 years) participated in SARS-CoV-2 testing on March 10, and again seven days later. Results were positive for SARS-CoV-2 in three residents and two staff members on the first round of testing. In the second round of testing, only one new positive result was found, in an asymptomatic resident.
Assays to Diagnose COVID-19
Testing for SARS-CoV-19 using an RT-PCR assay began in CDC labs on Jan. 18. Testing in U.S. public health labs started on Feb. 1, but because of initial problems with the test developed by the CDC for state lab use, testing outside of the CDC did not increase substantially until Feb. 27. By the middle of March, the vast majority of testing was being done at the state and local level, and by March 14, public health laboratories using the CDC assay were no longer required to submit samples to the CDC for confirmation.
On March 12, Roche Molecular Systems, Inc., became the first of numerous commercial entities to obtain an emergency use authorization (EUA) to develop their own diagnostic test for COVID based on the assay developed by the CDC. Since then, EUAs have been issued to dozens of additional manufacturers. This has opened the door for testing to be done at the individual hospital level, primarily focused in academic medical centers. Virtually all of these assays are based on RT-PCR technology. On April 1, the first antibody-based assay was announced by Cellex Inc., which offered an IgG/IgM rapid test. Many more have been developed since then, but because of the urgency of making tests available, these manufacturers were not required to submit to the rigors of the Food and Drug Administration (FDA) approval process.
There have been varying reports in the medical literature regarding the sensitivity of these assays. A small study in the Chinese literature examined the detection of SARS-CoV-2 by RT-PCR in various clinical specimens from patients with COVID-19 disease. In this study, 93% of bronchoalveolar lavage fluid specimens were positive for the virus. The rate of positive results was 63% for nasal swabs, and 32% for pharyngeal swabs.37 In a much larger study of 1,014 patients published in the Feb. 26 online journal Radiology, only 59% of patients who were diagnosed clinically with COVID-19 infection had a positive throat swab by RT-PCR.38 This study, like others in publication, was hampered by the lack of a definitive “gold standard” to determine which patients actually had confirmed COVID-19 infection.
According to the Infectious Diseases Society of America (IDSA), there were at least 25 different commercially available nucleic acid amplification tests for SARS-CoV-2 as of May 6.39 These tests were rushed to market under an EUA, and most have no clinical performance data. While these tests are highly specific, there is little to no information on their clinical sensitivity by anatomic site or by time during the course of the disease. Moreover, the current literature does not even have a standard definition for infection with COVID-19. The IDSA statement does recommend the use of nasopharyngeal swabs over throat swabs or saliva alone to test for COVID-19. Given the poor quality of the available evidence, we are severely hampered in our ability to interpret the assays in terms of predictive value, particularly in settings where the prevalence is low. In practice, this means that a positive test is likely to represent a true positive; the patient likely has COVID. However, a negative test does not rule out infection with COVID. Much work remains to be done to assess for the accuracy of existing molecular diagnostic tests.
Estimated Impact of COVID-19
By the middle of February, it had become apparent that COVID-19 would spread throughout the world. But at this point, there had been fewer than 2,000 deaths in China and two deaths outside China. The debate began over how much of an impact the virus would have in terms of numbers infected and overall case fatality rates (CFRs).
In an editorial in the New England Journal of Medicine co-authored by Anthony Fauci of the National Institute of Allergy and Infectious Diseases, the authors theorized the CFR “may be considerably less than 1%” despite early estimates from China that it was approximately 2%.40 This was based on the likelihood that many asymptomatic or minimally symptomatic cases were escaping detection. They further speculated that the overall impact might be on the order of what would be expected from seasonal influenza, with a CFR of 0.1%, or possibly as severe as the CFR seen with the influenza pandemics of 1957 or 1968.
One of the most influential estimates of the potential impact of COVID-19 was published by a team from the Imperial College of London on March 15.41 Their model was based on several assumptions: an R0 of 2.4; an incubation period of 5.1 days; a CFR of 0.9%; and infectiousness beginning 12 hours before symptoms. An estimate of deaths then was generated, with the added assumption that no efforts would be made to suppress or mitigate the rate of infection. This was designed to provide a worst-case scenario to policy makers. The model predicted 510,000 deaths in Great Britain and 2.2 million deaths in the United States by Aug. 1, 2020. These authors pointed out that a number of non-pharmacologic interventions could be undertaken to reduce these numbers, which would lead to “flattening the curve,” a reference to various reductions in the height of the mortality curve depending on what efforts were undertaken for mitigation or suppression. This term has been used incessantly in the press and on social media ever since, largely because it provides a graphic illustration to the public of why social distancing has become necessary.
Shortly after this estimate was released to the public, U.K. Prime Minister Boris Johnson announced the implementation of stringent measures to increase social distancing. The United States saw equally swift measures to “flatten the curve” at the same time. By the end of March, almost all U.S. primary, secondary, and university-level schools had discharged their students for the term, most to complete their work online. At the same time, most governors in the United States ordered nonessential businesses to close and send their employees home.
The Institute for Health Metrics and Evaluation (IHME) at the University of Washington has featured a prominent model to estimate mortality.42 Their estimates differ markedly from the one from the Imperial College of London, in part because they are based on the levels of social distancing in place at the time of the estimate. On March 30, they estimated that the United States would see between 39,000 and 142,000 deaths due to COVID-19 by Aug. 1, with a median estimate of 82,000. One month later, with social distancing starting to ease in many U.S. states, the numbers were little changed. By May 26, with many businesses opening back up and people no longer maintaining high levels of social distancing, the estimate had increased to 116,000 to 173,000, with a median of 131,000 deaths by Aug. 1. As of June 5, the total number of deaths nationwide had reached 108,000, and the projection for the total by Aug. 1 was 142,000.
The models thus far have been quite fluid, with constant updates. The obvious question is, how accurate are they? One study of modeling efforts during the Ebola outbreak in West Africa showed that models are unreliable for long-term forecasts of infectious disease outbreaks, and probably are not accurate more than a few weeks ahead of time.43
As of June 21, there have been 2.3 million confirmed cases of COVID-19 in the United States, with 120,000 deaths. At first glance, this would seem to indicate that the CFR is 6%, far higher than the estimates based on the Diamond Princess cruise ship experience. However, it is still unknown how many subclinical cases or unrecognized cases have occurred. Thus, the true CFR at this point is unknown. Calculation of a true CFR will only be possible when there are data from large-scale seroprevalence surveys, something the CDC is starting to coordinate at selected sites around the country.44 Once we are able to measure antibodies that indicate past infection in representative samples, we will be able to understand the true extent of infection in the U.S. population. New York started seroprevalence surveys in early May, and preliminary data indicate that 11.4% to 19.9% of the general population may have antibodies to COVID-19.45 If even the lower end of the range is accurate, this could lower the estimate of CFR by tenfold or greater, meaning the CFR may be closer to 0.6%. However, caution is in order, since the serologic assays themselves have just been developed, and the specificities are not clearly defined. High rates of cross-reactivity with conventional coronaviruses could artificially inflate the numbers of “confirmed” infected patients.
COVID-19 in the United States
The first U.S. case of COVID-19 was detected on Jan. 21, 2020. Throughout the month of February, the number of COVID cases in the United States increased very slowly, with a week passing between the identification of the 12th and 13th cases. By March 1, only 62 U.S. cases had been documented. But the months of March and April saw explosive spread of the virus in the United States, with marked increases in infections and deaths starting around the middle of March. (See Table 2.)
Table 2. Total Cases and Deaths from COVID-19, United States, 2020 |
||
|
Date |
Total |
Total Deaths |
|
Jan. 20 |
0 |
0 |
|
Jan. 31 |
6 |
0 |
|
Feb. 15 |
15 |
0 |
|
March 1 |
62 |
0 |
|
March 15 |
1,678 |
41 |
|
April 1 |
163,199 |
2,850 |
|
April 15 |
578,268 |
23,476 |
|
May 1 |
1,035,353 |
55,337 |
|
May 15 |
1,361,522 |
82,119 |
|
June 1 |
1,734,040 |
102,640 |
|
Source: World Health Organization |
||
The initial epicenter was in the Seattle region, starting with the large number of patients in the skilled nursing homes recounted earlier. But the epicenter then shifted quickly to the New York and New Jersey regions. New York City has had both the largest number of cases and the largest number of deaths to date. As of June 11, New York and New Jersey accounted for 38% of all deaths in the United States, and 19.4% of all U.S. deaths have been in New York City alone.46 Adding the deaths from the rest of the state brings the most recent total for New York to more than 30,000, or more than one-quarter of the total deaths. Of note, although California is the most populous state, it so far accounts for only 4% of all deaths.
The CDC COVID Tracker website provides a snapshot of the outbreak in the United States with a variety of data sets available, including total number of cases diagnosed, deaths by state, deaths by county, and deaths by age group. Fifty-nine percent of the deaths to date have been in patients older than 75 years of age, 21% in patients 65-74 years, and 15% in patients 50-64 years. Only 4% of deaths have been in patients younger than 50 years. This is consistent with an earlier report showing patients older than age 65 years accounted for 31% of cases, 45% of hospitalizations, 53% of intensive care unit (ICU) admissions, and 80% of deaths.47
CDC data on U.S. adult patients admitted to a hospital with laboratory confirmed COVID-19 infection during the first month of the outbreak (March 1-8) indicate that 75% were older than age 50 years, and 89% had underlying medical conditions, including hypertension (50%), obesity (48%), chronic lung disease (35%), diabetes mellitus (28%), and cardiovascular disease (28%). Fever was noted at admission in 71%, cough in 80%, shortness of breath in 80%, diarrhea in 27%, and nausea or vomiting in 24%.48 Rhinorrhea was reported in 7%. There also have been reports of unusual neurologic symptoms, particularly olfactory and taste disorders (anosmia and dysgeusia).49
Published clinical reports regarding COVID-19 in the United States are quite sparse as of June 1, with most of the data coming from the New York area. Almost all of the clinical studies focus on the most severely ill patients admitted to ICUs. One study focused on the 22% of patients admitted to the hospital who were critically ill and required care in the ICU.50 The median age was 62 years, and 78% were older than 50 years. Eighty-two percent had at least one comorbidity, the most common of which were hypertension, diabetes, and obesity. Symptoms included fever in 71%, shortness of breath in 74%, cough in 66%, and diarrhea in 12%. Less than 10% had rhinorrhea or sore throat. Patients were followed up to 28 days. Seventy-nine percent of patients required invasive mechanical ventilation for a median of 18 days. Sixty-six percent of patients required vasopressors, and 31% required renal replacement therapy. Ultimately, 39% died.
With such a marked number of deaths in such a short period, it would seem reasonable that total deaths from all causes in the United States should have increased during this time, and indeed they have. The CDC publishes a weekly tally of deaths due to all causes in the United States.51 As expected, this tally varies by the time of year, with more deaths reported during the winter when respiratory viruses are in circulation, and decreases during the summer. The data began to show an increase in the number of deaths during the week ending March 28, when the excess reached 6.3% to 10.1% of the expected value. This excess grew to 34.8% to 39.6% above the national baseline during the week ending April 11. It remained above the baseline as of May 30. This increased rate corresponds to the period during which COVID-19 infections and deaths appeared in the United States, as shown in Table 2. This provides ample evidence that COVID-19 has caused marked increases in death rates in the United States. (See Figures 1 and 2.)
Figure 1. Total Number of COVID-19 Deaths in the U.S. Reported to the CDC, by Age Group |
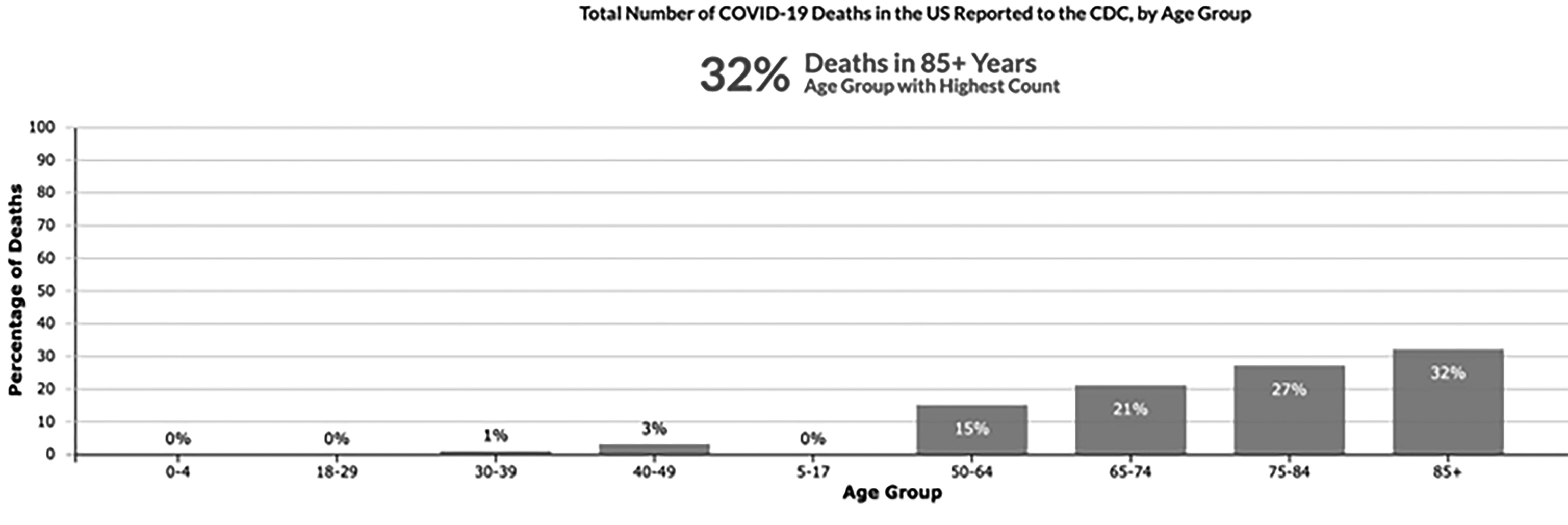 |
|
Source: Centers for Disease Control and Prevention |
Figure 2. Weekly Number of Deaths (from all Causes) |
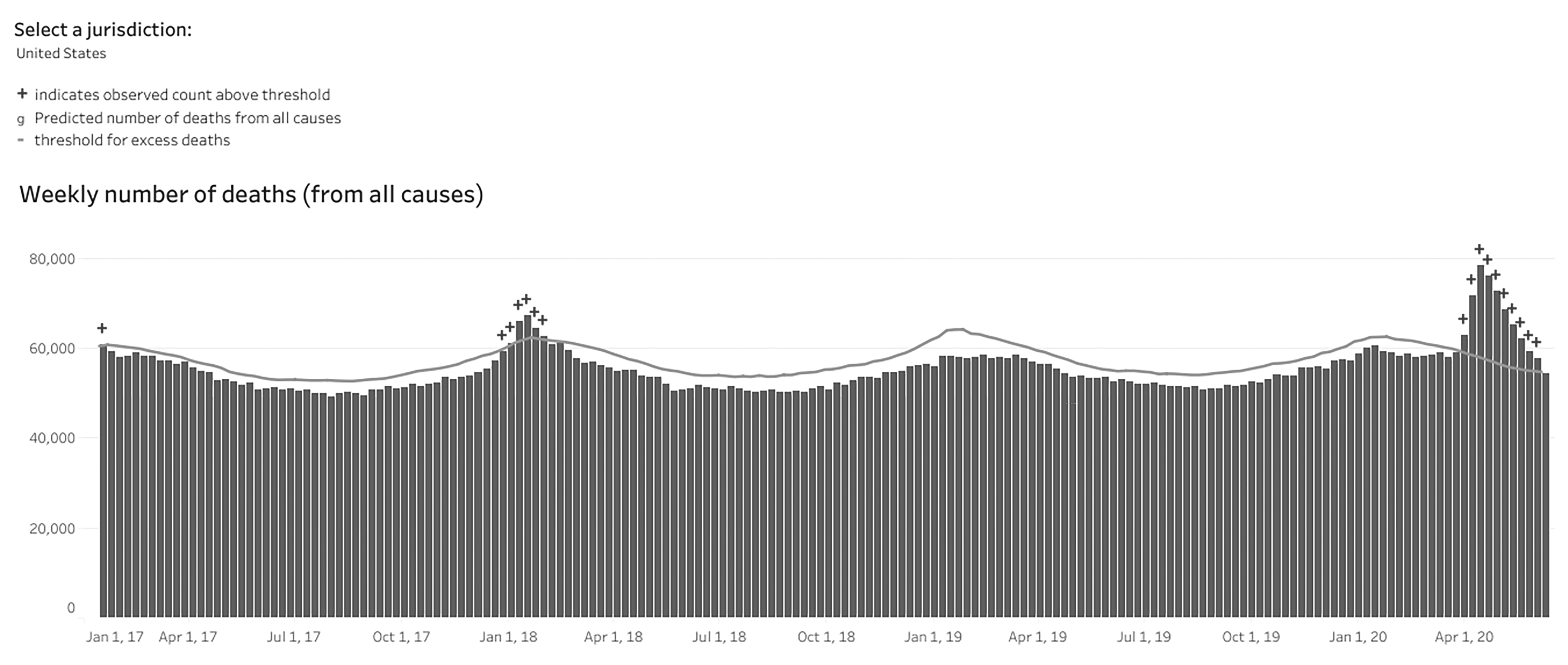 |
|
Source: Centers for Disease Control and Prevention |
COVID-19 can present as mild, moderate, or severe disease. The pattern observed in China and now in the United States is that most patients with COVID-19 have mild disease, particularly in the younger age groups. The CDC states that in the absence of hypoxia or pneumonia, most infected people do not require admission to the hospital and can manage their illness at home.52 Indicators for severe disease include marked tachypnea, hypoxemia, ratio of partial pressure arterial oxygen to fraction of inspired oxygen less than 300, and large lung infiltrates on chest radiograph.53 If patients have these indicators, further management in the hospital may be warranted.
For patients who develop severe disease, the median time to dyspnea is five to eight days, median time to ARDS is eight to 12 days, and median time to ICU admission is 10-12 days after the onset of symptoms.52 Therefore, if patients are managed outside the hospital, careful monitoring is warranted for one to two weeks, particularly if the patients have underlying health problems that predispose them to more severe disease.
Guidelines have been developed to aid clinicians in their management of patients with severe COVID-19 infection. Readers are directed to:
- Alhazzani W, et al. Surviving Sepsis Campaign: Guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19). Critical Care Medicine 2020;48:e440-e469.
- Infectious Diseases Society of America Guidelines on the Treatment and Management of Patients with COVID-19 (www.idsociety.org/COVID19guidelines)
- National Institutes of Health COVID Treatment Guidelines. Available online at https://www.covid19treatmentguidelines.nih.gov/
- ACEP COVID-19 Field Guide. https://www.acep.org/corona/covid-19-field-guide
COVID-19 Illness: Selected Important Features
Patients infected with COVID-19 can present with a variety of nonspecific symptoms, as summarized earlier. Paramount among them is ARDS. Early reports from China described abnormal chest CT scans in most of the critically ill patients with COVID. Chest X-rays show bilateral multifocal opacities. CT of the chest often shows typical bilateral ground glass opacities.54 In some reports from China, clinical findings such as these were used to establish the diagnosis even if the virus was not detected by RT-PCR testing. Mechanical invasive ventilation has been required for large numbers of the most critically ill patients, and mortality in these patients has been quite high, ranging from 40% to 70%, according to CDC. Many experts have found prone ventilation to be helpful in severely ill patients. Guidelines on the management of COVID-19 from the Surviving Sepsis Campaign recommend 12-16 hours of prone ventilation per day for mechanically ventilated adults with moderate to severe ARDS.55
A number of reports have indicated that COVID-19 induces a hypercoagulable state, especially in the patients who are most ill.56,57 The available data indicate patients hospitalized with COVID-19 infection have an incidence of thrombotic complications between 16% and 49%. Autopsies on patients who have died from COVID-19 have shown blood clots throughout the microvasculature, affecting multiple organ systems, including the lungs. Recommendations for monitoring and treating for this hypercoagulable state have been published recently, and they include measurement of D-dimers, prothrombin time (PT), fibrinogen, and platelet counts. The International Society on Thrombosis and Haemostasis states that low molecular-weight heparin therapy should be considered in all patients hospitalized with COVID-19 infection.58
A weekly surveillance summary of COVID-19 activity in the United States is maintained by the CDC in its online COVID View.59 The most recent report is for the week ending June 6. The data show a number of trends. First, the percentage of specimens testing positive for COVID-19 has continued to drop for at least five weeks in public health, commercial, and clinical laboratories, and is now close to 5% after peaking around 20% to 25%. This does not appear to be due to larger numbers of tests being done on asymptomatic patients, because the number of specimens tested actually has dropped since the middle of May. Further, the weekly graph showing the percentage of medical visits for COVID-19-like illness (CLI) also has been on a consistent seven-week decline. Finally, the graph for the percentage of total deaths due to pneumonia, influenza, or COVID-19 (PIC) has been declining consistently after peaking five weeks ago at 26.8%; the most recent level is 9.8%. (See Figure 3.) Taken in aggregate, these data indicate that COVID-19 is on the decline nationally, but this is variable by state.
Figure 3. U.S. Mortality: Death Certificates Listing Pneumonia, Influenza, and COVID-19 |
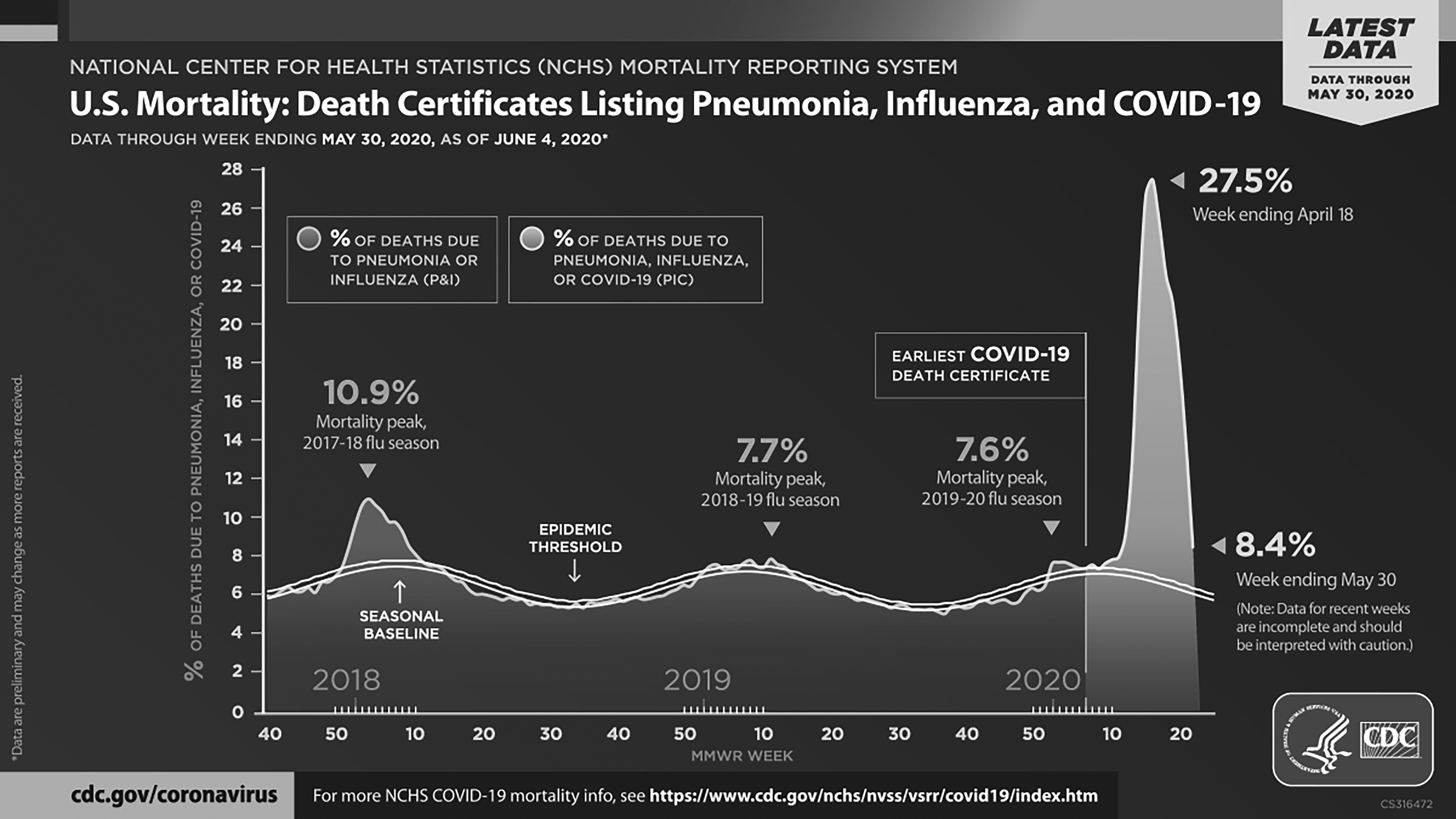 |
|
Source: Centers for Disease Control and Prevention |
COVID-19 in Healthcare Workers
The first report focusing on COVID-19 in healthcare workers (HCW) came from China in February.31 In this large case series of 44,672 confirmed infections, 3.8% were HCW. Overall, 15% of infections in HCW were classified as severe or critical, but the CFR was quite low at five out of 1,716. No other details are provided.
On April 17, the U.S. CDC COVID-19 response team provided early information on COVID-19 in U.S. healthcare personnel (HCP). The reporting period was from Feb. 12 to April 9. A total of 315,531 COVID-19 cases had been reported to the CDC as of that date, including 9,282 who were identified as HCP. There were 27 deaths among HCP. As of May 30, there were 64,479 cases among HCP, with 309 deaths.60 Both of these reports are likely undercounts because of the fact that for both periods, less than 25% of the total number of confirmed COVID-19 cases reported to the CDC contained any information on HCP status. We know that HCP are dying in significant numbers from this pathogen, but the true extent is yet unknown.
A study from Italy examined mental health effects during the height of the COVID-19 outbreak there.61 Data were collected between March 27 and March 31 via an online questionnaire based on validated mental health screening tools. This was a self-selected sample, so response rates could not be measured. A total of 1,379 HCW completed the questionnaire. Overall, 49% endorsed symptoms of posttraumatic stress, 25% reported symptoms of depression, 20% reported symptoms of anxiety, and 8% reported insomnia.
COVID-19 in Children
There is little information on COVID-19 in children, in part because it appears they are infected at a markedly lower rate than adults. One study from China described the epidemiology of COVID-19 in children, but in this study, only one-third of the cases were confirmed.62 Two-thirds of the cases were diagnosed on clinical grounds alone, primarily symptoms combined with exposure to known COVID-19 patients. Included cases were from the first month of the Chinese outbreak (Jan. 16 to Feb. 8). More than 90% of the pediatric patients had asymptomatic, mild, or moderate infections, and only one child died. This study also showed mildly higher risk for severe or critical infections in children younger than 12 months of age compared to children older than 1 year.
Another study focused solely on 171 children with proven COVID-19 in China from Jan. 28 through Feb. 26 at Wuhan Children’s Hospital, the only hospital assigned to treat COVID-19-infected children in Wuhan.63 In this cohort, 16% were asymptomatic, three patients required intensive care support, and there was one death. Only 41.5% of these COVID-19-infected children had fever at any time during their illness.
A report from the U.S. CDC summarized the data on U.S. children as of April 2.64 At that point, there were 239,000 cases of COVID-19 diagnosed in the United States, with 5,443 deaths. Only 2,572 (1.7%) of the confirmed cases were in children younger than 18 years of age. Seventy-three percent had symptoms of fever, cough, or shortness of breath, compared with 93% of adults. Hospitalization rates were substantially lower than those for adults, and there were three deaths. Seventy-one percent of the cases came from New York or New Jersey. Unfortunately, data were missing for 70% of the pediatric cohort, making it difficult to describe fully the clinical characteristics of these patients.
Data on symptoms in children are available from Chinese and U.S. reports and are quite similar.63,64 Fever was noted in 41% to 56%, cough in 49% to 54%, shortness of breath or tachypnea in 13% to 29%, and diarrhea in 9% to 13%. Less than 10% had rhinorrhea. Most children do not require admission to a hospital.
A cross-sectional study of children admitted to U.S. pediatric intensive care units (PICUs) was published in May.65 Forty-six hospitals in the United States and Canada participated, but 30 hospitals reported no admissions to their PICU during the study period of March 14 to April 3. A total of 48 children younger than 21 years of age were included in the study; less than half were younger than 10 years of age. All had diagnoses confirmed by PCR from nasal swabs. Eighteen required invasive ventilation, and two died. Forty percent of the patients were considered “medically complex.”
Reports started appearing in Italy and France in April describing an outbreak of severe multisystem inflammatory syndrome in children (MIS-C) that appear to be associated with COVID-19 infections.66,67 Symptoms overlap considerably with Kawasaki disease, although the CDC case definition requires involvement of at least two organs (heart, kidneys, lungs, blood, gastrointestinal tract, skin, or nervous system ).68 The case definition also requires evidence for current or recent COVID-19 infection. Patients with MIS-C tend to be a little older than the typical patient with Kawasaki disease, around 10 years, but anyone younger than 21 years can be affected. Patients have elevated inflammatory markers, and many have severe cardiac dysfunction. In the report from France, 80% required inotropic support and 20% required extracorporeal membrane oxygenation. Most children have responded well to high-dose immunoglobulin and steroids.
A useful website for tracking severe COVID-19 infections in children can be found at www.myvps.org. This site tracks 181 PICUs in the United States and Canada. As of May 29, a total of 395 children younger than 18 years of age have been admitted to a PICU in either country, an average of two per participating hospital. Sixteen children younger than 18 years of age have died. Two-thirds of the deaths have occurred in New York or New Jersey, which also account for more than 30% of the total deaths in the United States. For context, it is helpful to note that as of this date, more than 100,000 patients had died in the United States alone due to COVID-19. It is interesting to note that this site also documented a total of 110 PICU staff who were infected with SARS-CoV-2 as of May 29, or less than one HCW per participating hospital. This site illustrates as well as any of the publications noted earlier that children largely have avoided both severe disease and death due to SARS-CoV-2 compared to the adult population.
COVID-19 is a disease that largely affects adults. Why is there such a marked difference? A recent study found that nasal epithelium shows age-dependent ACE2 expression, providing an interesting theory about the lower infection rate in children.69 This study showed much lower ACE2 expression in children younger than 10 years of age, with increases seen successively in every age group up to age 60 years.
Medications for COVID-19
There have been several candidate therapies discussed in the literature from early in the pandemic, with much of the focus on hydroxychloroquine, IL-6 receptor antagonists, and remdesivir. Many more medications are likely to enter trials in the future, especially with the ability to identify candidates through the use of computer-based drug discovery.70
Hydroxychloroquine has received much of the attention in the media, but studies to date have been disappointing. One early non-randomized study of hydroxychloroquine from France claimed to show a marked reduction in the viral burden.71 However, there were several problems with this study, including a non-randomized sample of only 26 patients; the lack of any clinical endpoints, such as likelihood of intubation or death; and the exclusion of six patients from the analysis. A large study from New York focused on patients admitted to the hospital with moderate to severe COVID-19 infections, 58.9% of whom were begun on hydroxychloroquine shortly after admission.72 Patients were not randomized, and patients who received hydroxychloroquine were compared to those who did not. There was no difference in the primary outcome measures: likelihood of intubation or death. To date, no large, randomized trials have been completed to show evidence of benefit from hydroxychloroquine.
An early study from China evaluated a combination of lopinavir and ritonavir for COVID-19 infection in a randomized, controlled trial with 199 patients.73 There was no difference in time to improvement or in 28-day mortality.
Remdesivir was evaluated in a randomized, double-blind, placebo-controlled trial of 1,063 patients from multiple sites around the world.74 One thousand sixty-three patients were randomized. The median age was 59 years, and 89% had severe disease at the time of enrollment. The median time between symptom onset and enrollment was nine days. Patients in the remdesivir group recovered in 11 days, compared to 15 days for controls, a statistically significant reduction of four days. Mortality was lower in the remdesivir group, but the difference was not significant. Serious adverse events were similar in the two groups. Remdesivir is available under an EUA for the treatment of COVID-19 in adults and children with severe COVID-19 disease.
A recent study suggested the use of dexamethasone for patients with severe symptoms. There are no data that suggest that early initiation of treatment will be preventive, as the dexamethasone appears to work in the second phase of the disease during the cytokine storm.75
The data to this point are not encouraging regarding medical interventions for COVID-19. So far, no “magic bullet” has been identified that has shown a significant impact on mortality. The focus at this point is on pushing forward with more randomized, controlled trials of various medications, and to avoid empiric use of medications with unproven benefit.
Conclusion
The world has been in the grip of a new pandemic for the past six months, one that has caused more than 400,000 deaths worldwide to date. Although the vast majority of deaths have been in adults older than 45 years of age, younger adults and children also are at risk. The peak of infection in the United States for the first wave appears to have occurred in mid-April, but infections continue to occur even now, in the summer months, at substantial rates. We do not know if we face a marked increase in a second wave, or even when that second wave may occur. At this point, it appears that COVID-19 is going to be a constant and possibly growing threat until a vaccine is produced to protect the population.
REFERENCES
A complete list of references is available online: https://bit.ly/2NpGc8h.
Coronavirus disease 2019 (COVID-19) is a rapidly spreading disease, and our knowledge is rapidly growing.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
