Heated High-Flow Nasal Cannula Oxygen Therapy and Noninvasive Positive Pressure Ventilation
January 15, 2021
Reprints
AUTHORS
Catherine A. Marco, MD, FACEP, Professor of Emergency Medicine, Department of Emergency Medicine, Wright State University Boonshoft School of Medicine, Dayton, OH
Jennifer M. Oakes, DNAP, MSNA, CRNA, Associate Program Director for Nurse Anesthesia, Texas Christian University, Fort Worth
PEER REVIEWERS
Alexander S. Niven, MD, Critical Care Specialist, Mayo Clinic, Rochester, MN
Vibhu Sharma, MD, Pulmonary Disease and Critical Care Medicine, University of Colorado, Aurora
EXECUTIVE SUMMARY
- Oxygen delivery by high-flow nasal cannula (HFNC) provides increased oxygenation by increasing the oxygen in the nasopharyngeal dead space. Patients are able to eat, drink, and talk, and, therefore, it is more comfortable than wearing an oxygen mask.
- HFNC can be used in patients with COVID-19 as they develop acute respiratory failure. However, HFNC is not as efficient at removing carbon dioxide as intubation.
- Initial settings include a flow rate of 30 mL/minute, which can be raised as tolerated by the patient. The maximum flow rate is 60 mL/minute. FiO2 can be set up to 100%.
- Abdominal distention and barotrauma can occur from high flow settings.
Introduction
Adequate oxygenation is a priority in all acutely ill emergency department (ED) patients. Oxygen may be delivered through a variety of devices, including low-flow systems (including nasal cannula or face mask) or high-flow systems (including nonrebreather masks and Venturi masks). The fraction of inspired oxygen (FiO2) delivered by these systems may be variable.
Noninvasive ventilation (NIV) strategies are a good option for patients who do not tolerate low-flow or high-flow systems or who remain hypoxic. NIV includes bilevel positive airway pressure (BiPAP) and continuous positive airway pressure (CPAP). NIV provides positive end expiratory pressure (PEEP) by a tight-fitting mask. NIV provides improved oxygenation and ventilation for patients who tolerate the mask.
Heated high-flow nasal cannula (HFNC) oxygen is a safe and effective method of oxygenation. It does not provide as much PEEP as NIV, but may be useful in patients who are unable to tolerate NIV. An understanding of the physiologic benefits, indications, complications, and step-by-step instructions for settings and parameters is necessary to optimally use this therapy.
History
The nasal cannula apparatus was first patented in 1949. Since then, the concept of delivering needed oxygen therapy through the nasal passage has grown in popularity and use. Simple nasal cannulas are limited in the amount of flow and pressure that can pass through this apparatus. Modifications and improved designs have occurred over the years to optimize this form of enhanced oxygen delivery. When additional oxygen delivery is needed, the next step is to change from a nasal cannula to a mask. Even mask apparatuses have a limited maximum oxygen delivery flow of 15 L/minute. Masks can be equipped with reservoirs and one-way valves to reduce air entrainment, so nonrebreather (NRB) masks can increase FIO2 to 80% or greater. One of the more recent modifications is the design allowing for higher flows, humidification, and greater carbon dioxide washout.
The concept of high-flow nasal oxygen therapy was first introduced in the early 2000s.1 Initially, this system was used to treat apnea in premature infants. Applications were expanded later through a variety of pediatric respiratory issues. The growing literature supporting this treatment modality has since expanded the use into adult critical care environments and is becoming a practical option in emergency medicine and anesthesia.2
HFNC System and Mechanism of Action
HFNC oxygen therapy is delivered by an air/oxygen blender, an active humidifier, and a single heated tube that delivers high-flow oxygen via a soft, silicone nasal cannula that is available in a variety of sizes.3 It is designed to fit into the nares comfortably without occluding the flow of gases. Therefore, this is considered an open-circuit device.1
HFNC systems are designed to deliver oxygen up to 60 L/minute with variable FiO2 and temperature.4 FiO2 can be adjusted between 21% to 100%. FiO2 should be adjusted to keep the SaO2 between 88% to 92%, as clinically indicated. Humidification also is a component of current systems, with a set humidification of 44 mg H2O/L (100% relative humidity). Temperature settings are adjustable, but 37°C is considered optimal in most situations.
Humidification is preset to 100% but can vary based on flow and temperature. This variability is usually transient and minor.1 The flow should be adjusted to assist in optimal lung expansion during inspiration and increase end-expiratory lung volume as needed.5 Initial flow should be set at 30-35 L/minute and can be adjusted upward as tolerated.
Physiologic Advantages
The advantages of HFNC are numerous. The high flow rate results in decreased inspiratory effort, improved oxygenation, reduced respiratory rate, reduced minute ventilation, and more homogeneous ventilation distribution.6-8 The high flow rate also results in higher concentrations of oxygen delivery, due to minimal entrainment of room air. HFNC is delivered by soft and pliable nasal prongs, and usually it is well tolerated. Patients can easily speak, eat, and drink while wearing the device. Warming and humidification of inspired oxygen is more effective and can decrease the work of breathing.
Although the nasopharyngeal dead space is constant, HFNC replaces the mixed gases in these areas with oxygen, turning the dead space into a functional oxygen reservoir. HFNC delivers a mild PEEP effect by increased nasopharyngeal airway pressure.9 This PEEP effect is improved with the mouth closed. A chin strap may be used to assist patients with keeping the mouth closed. It has been estimated that each increase of
10 L/minute results in approximately 0.7 cm H2O of airway pressure. Physiologic benefits are summarized in Table 1.
Table 1. Physiologic Advantages of High-Flow Nasal Cannula Oxygen |
|
Clinical Indications
Numerous oxygen delivery systems are available and should be used as therapeutic modalities appropriate for the clinical setting. (See Table 2.) The typical starting point of oxygen therapy progresses from a spontaneously ventilating patient breathing room air to a patient with supplemental oxygen delivered via a nasal cannula. An oxygen-sparing nasal reservoir cannula (nasal pendant) is another effective oxygenation method.10 When this proves to be insufficient, then a mask (simple or nonrebreather) may be attempted. The next step would be noninvasive ventilation (CPAP or BiPAP). If these measures are not sufficient to treat hypoxia adequately, intubation and mechanical ventilation are appropriate. (See Figure 1.) The additional option of HFNC can fit into this decision tree prior to noninvasive ventilation or if noninvasive ventilation is poorly tolerated because of patient discomfort or claustrophobia, which is very common. There is some evidence of improved oxygenation and reduced hypercapnia in patients with acute hypercapnic respiratory failure.11
Figure 1. Oxygenation Algorithm |
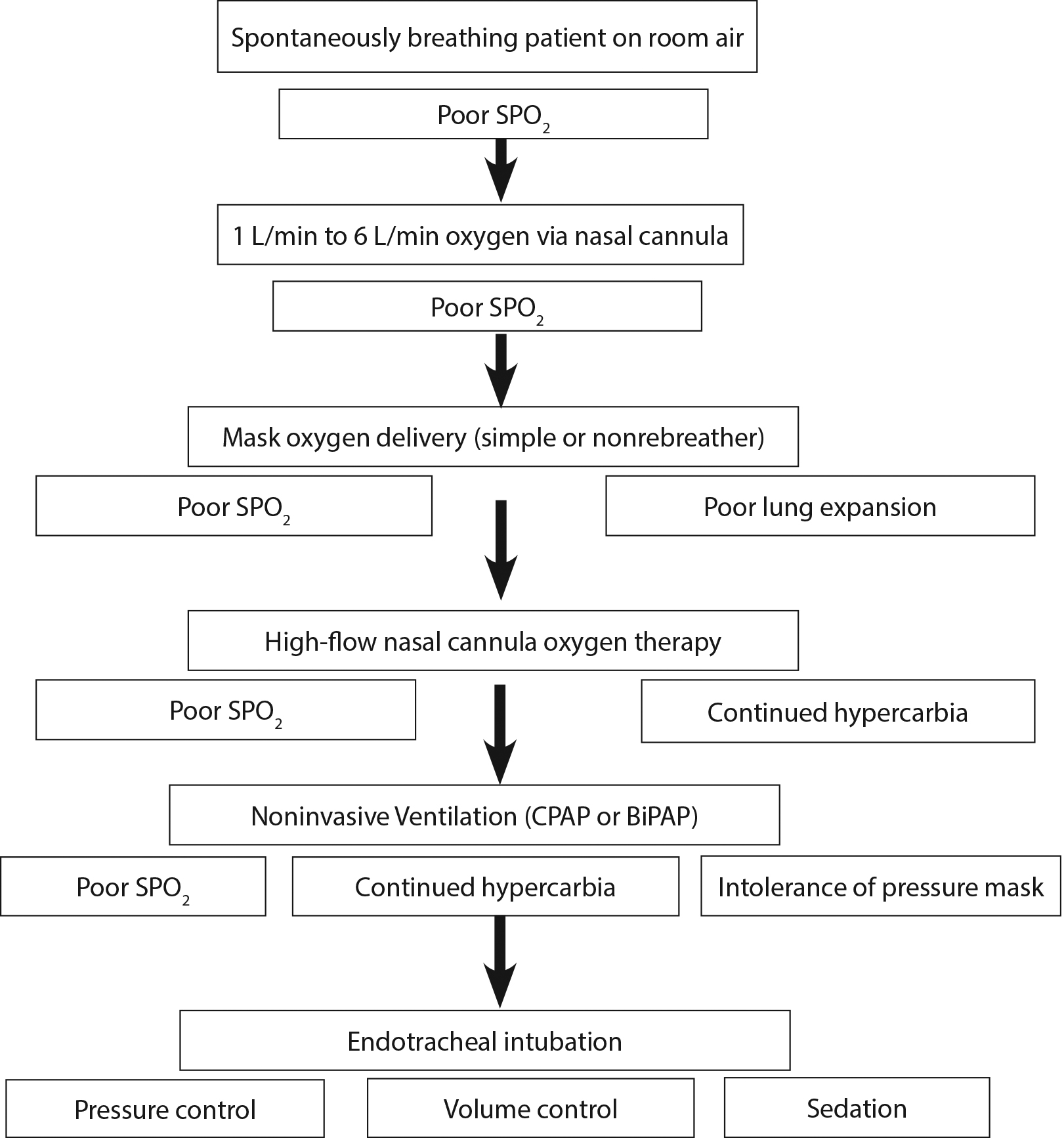 |
|
*This figure represents an overview of oxygenation therapies. Choice of therapy should be based on acuity and condition. CPAP = continuous positive airway pressure; BiPAP = bilevel positive airway pressure |
Table 2. Oxygen Delivery Systems |
|||
|
System |
Advantages |
Disadvantages |
Initial Settings |
|
Blow by oxygen |
|
|
|
|
Nasal cannula |
|
|
|
|
Face mask |
|
|
|
|
Nonrebreather mask |
|
|
|
|
High-flow nasal cannula |
|
|
|
|
Noninvasive ventilation (CPAP, BiPAP) |
|
|
|
|
Self-inflating ventilation bag (AmbuBag, etc.) |
|
|
|
|
Endotracheal intubation |
|
|
|
|
PEEP = positive end expiratory pressure; CPAP = continuous positive airway pressure; BiPAP = bilevel positive airway pressure |
|||
HFNC can be an effective oxygenation technique for a variety of clinical indications, including hypoxemic respiratory failure, acute exacerbation of chronic obstructive pulmonary disease (without hypercapnia), acute congestive heart failure, acute respiratory distress syndrome (ARDS), post-extubation oxygenation, pre-intubation oxygenation, and other hypoxic conditions.1,12-16 (See Table 3.) Although some clinical indications still are controversial (i.e., ARDS), emerging data will elucidate HFNC’s definitive role in airway management in these settings.
Table 3. Clinical Applications |
|
HFNC can be an effective oxygenation therapy for patients with hypoxemic respiratory failure and do-not-resuscitate or do-not-intubate orders.17 HFNC has demonstrated reduced need for intubation and a reduced mortality among patients with acute hypoxemic respiratory failure.18-20 In patients with acute hypoxemia, HFNC is associated with lower mortality compared with standard oxygen therapy.19
A recent meta-analysis showed improved patient comfort and dyspnea, with no difference in mortality or intubation, compared to standard oxygenation.21
HFNC has been studied as an effective treatment for COVID-19. HFNC has been shown to effectively oxygenate patients with COVID-19 and respiratory failure, and may reduce the need for invasive ventilation.22-26 HFNC does not produce increased air or surface contamination when compared to standard oxygen masks.27 A surgical mask may be placed over the cannula to minimize dispersion of particles.28 Although data are limited at this time, evidence suggests its efficacy in the setting of hypoxia without hypercarbia for COVID-19 patients.
Contraindications
HFNC should not be used in patients with facial trauma, recent surgery, or other facial anomalies that preclude use of a high-flow nasal prong unit. (See Table 4.) The use of HFNC in patients with ARDS is controversial. Emerging data will be useful in elucidating its potential role in the treatment of ARDS.
Table 4. Contraindications to HFNC |
|
Complications
HFNC is generally well tolerated. Potential complications include abdominal distension, barotrauma, or delay in endotracheal intubation. (See Table 5.) Abdominal distension could be related to the flow rate and/or incompetent esophageal sphincter control. Barotrauma also can be related to the flow rate and/or air trapping. The delay in invasive ventilation should be avoided by proper monitoring and clinical judgment.
Table 5. Potential Complications of HFNC |
|
Indications of HFNC failure are declining SpO2, increased respiratory rate, and paradoxical chest movement with respirations.1 When these are observed, alternative noninvasive methods or endotracheal intubation should be considered. Care must be taken that HFNC does not delay clinically indicated intubation.29
Instructions for Practical Use
The device should be cleaned properly, and a new circuit, filter, and nasal cannula should be used for each patient. Particularly when used for patients with suspected COVID-19 or other contagious conditions, proper cleaning of the entire apparatus is essential. The nasal canula is designed to fit in the patient’s nares without occluding passages.
Sizing
The size of the nasal cannula should be selected based on the patient’s age and size. (See Figure 2.) Pediatric sizes are available and are based on the patient’s weight.
Figure 2. High-Flow Nasal Cannula Prongs |
 |
|
Photo courtesy of Catherine A. Marco, MD, with acknowledgement of Sarah Dick, volunteer. |
Fitting
A head strap usually is included with the nasal cannula package. The nasal cannula prongs should be inserted into the nares and then the strap is adjusted to fit around the back of the head. Care should be taken to not allow the strap to slide down onto the neck or to pass over the ears.
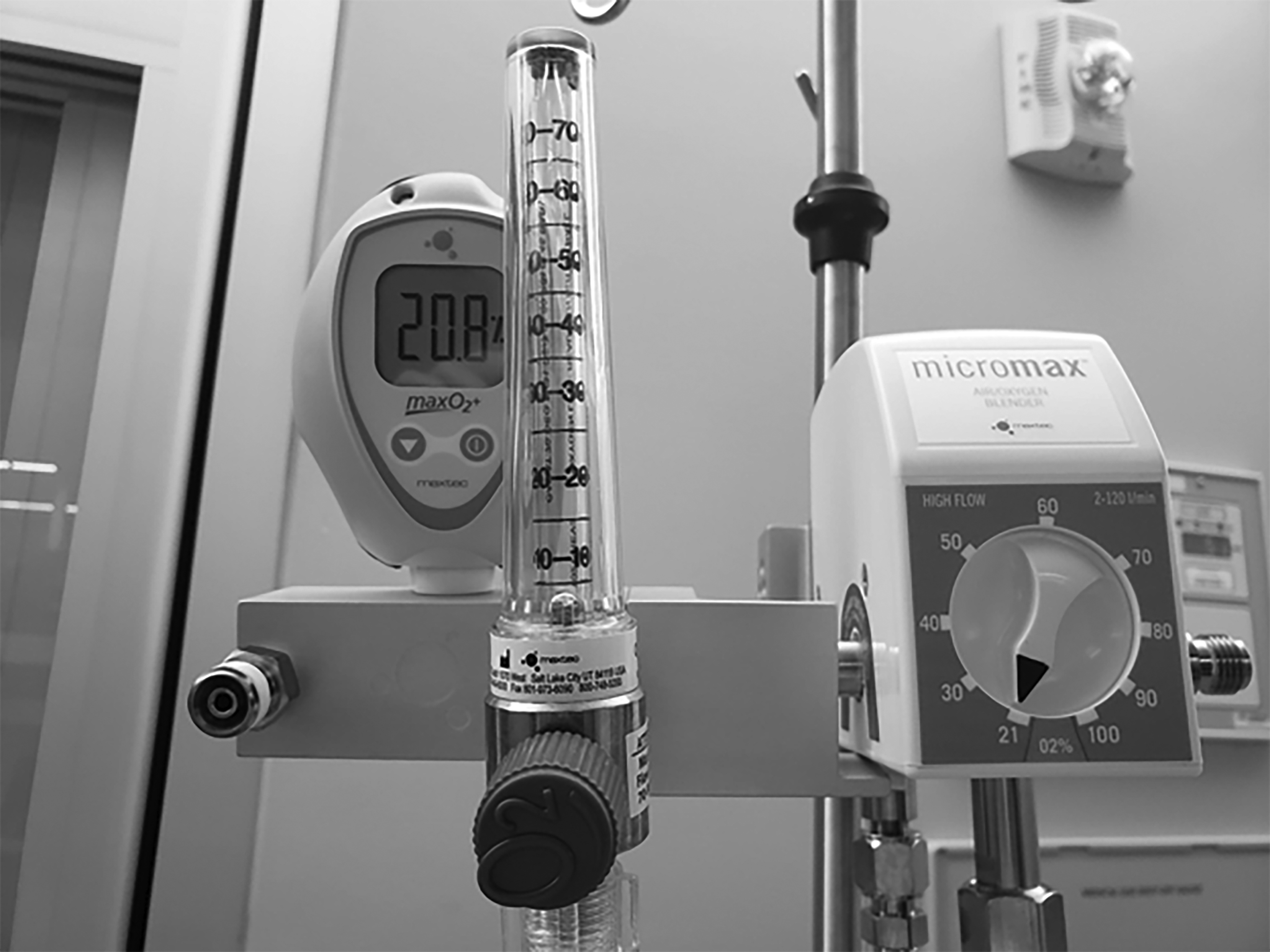
Adjusting Flow Rate
For adults, the flow rate ranges from 5 L/minute to 60 L/minute. (See Figure 3.) A typical starting rate is usually 30 L/minute. This may be increased as tolerated by the patient until the desired physiologic effects are seen. It also is recommended that all settings be confirmed and that flow begin prior to placing the cannula on the patient.
Figure 3. High-Flow Nasal Cannula Controls to Adjust FiO2 and Flow Rates |
 |
Adjusting FiO2
FiO2 ranges from 21% to 100%. The gas blender will adjust the FiO2 based on the oxygen input that is set. FiO2 can be adjusted between 21% (room air) and 100%.
Temperature
The initial temperature setting should be 37°C. Temperature sensors are intrinsic with the device and are set with safety features to prevent patient injury. Some systems require manually turning on the heater and temperature monitoring.
Humidification
Sterile water is needed to provide adequate humidification. It is important to use only sterile water. Normal saline or lactated Ringers solution is contraindicated. Humidification initially should be set at 100%.
Patient Instructions
Patients should be instructed not to remove the cannula unless instructed by a healthcare professional. They should be aware of the basic functions of the machine and what to expect prior to administration. Patients should breathe normally and not attempt to occlude nasal or oral airways during treatment.
Noninvasive Positive-Pressure Ventilation
Noninvasive positive-pressure ventilation (NIPPV or NIV) is a form of positive pressure ventilation typically delivered by face mask or nasal mask. NIPPV provides an effective means to assist patients with ventilation and oxygenation. NIPPV has proven benefits in numerous clinical settings, including acute hypercapnic respiratory failure, acute exacerbation of chronic obstructive pulmonary disease, and acute cardiogenic pulmonary edema.30-37 Other clinical conditions that may benefit from NIPPV include respiratory distress secondary to COVID-19, acute hypoxemic respiratory failure, asthma, pneumonia, ARDS, postoperative, or chest trauma-induced acute respiratory failure.19,38-45 Despite initial concerns about aerosols generated during NIPPV and HFNC, studies have demonstrated no increased aerosol generation compared to 6 L O2 by nasal cannula.46 NIPPV also may have benefit for preoxygenation prior to endotracheal intubation.47-49
The physiologic benefits of NIPPV include increased tidal volume, increased minute ventilation, improved pulmonary compliance, and reduced alveolar atelectasis. NIPPV increases intrathoracic pressure and may shift pulmonary edema into the vasculature, and decrease venous return and afterload, which may improve cardiac function. These physiologic benefits lead to reduced work of breathing, improved mental status, improved mortality, and decreased rate of endotracheal intubation.
Contraindications to NIPPV include the need for emergent intubation, and other emergent life-threatening conditions, such as acute upper gastrointestinal hemorrhage, severe epistaxis, or hemodynamic instability. Relative contraindications include facial trauma or an anatomic abnormality that precludes appropriate mask fit, airway obstruction (upper airway mass, etc.), and the inability to protect the airway (including altered mental status, copious secretions, neuromuscular disorders, or stroke). NIPPV is considered a short-term intervention and is not appropriate for conditions requiring long-term ventilatory support.
Complications are rare and may include local irritation, discomfort, anxiety, nasal dryness, pulmonary barotrauma, pneumothorax, gastric distension, and aspiration.
NIV includes bilevel NIV (bilevel positive airway pressure or BiPAP), continuous positive airway pressure (CPAP), pressure support ventilation (PSV), and other modes (e.g., neurally adjusted ventilatory assist [NAVA]). Bilevel NIV has been studied widely, has proven benefit in improving alveolar ventilation, and is used widely.
Mechanisms of action for improved ventilation and oxygenation include improved alveolar ventilation, preload reduction, and decreased left ventricular afterload. Multiple studies have demonstrated a reduced rate of intubation and improved mortality.
NIV is a reasonable option to treat respiratory distress for patients who decline endotracheal intubation, including hospice patients or patients with do not resuscitate (DNR) orders.
To administer NIPPV, the correct mask size should create a tight seal. Typical initial settings are spontaneous mode with IPAP of 8-10 cm H2O and EPAP 3-5 cm H2O.50 Patients should be monitored closely for mental status, airway protection, work of breathing, oxygen saturation, heart rate, and blood pressure.
REFERENCES
- Spicuzza L, Schisano M. High-flow nasal cannula oxygen therapy as an emerging option for respiratory failure: The present and the future. Ther Adv Chronic Dis 2020;11:2040622320920106.
- Humphreys S, von Ungern-Sternberg BS, Skowno J, et al. High-flow oxygen for children’s airway surgery: Randomised controlled trial protocol (HAMSTER). BMJ Open 2019;9:e031873.
- Nishimura M. High-flow nasal cannula oxygen therapy in adults: Physiological benefits, indication, clinical benefits, and adverse effects. Respir Care 2016;61:529-541.
- Thille A, Muller G, Gacouin A, et al. Effect of postextubation high-flow nasal oxygen with noninvasive ventilation vs high-flow nasal oxygen alone on reintubation among patients at high risk of extubation failure: A randomized clinical trial. JAMA 2019;322:1465-1475.
- Corley A, Caruana LR, Barnett AG, et al. Oxygen delivery through high-flow nasal cannulae increase end-expiratory lung volume and reduce respiratory rate in post-cardiac surgical patients. Br J Anaesth 2011;107:998-1004.
- Mauri T, Turrini C, Eronia N, et al. Physiologic effects of high-flow nasal cannula in acute hypoxemic respiratory failure. Am J Respir Crit Care Med 2017;195:1207.
- Sztrymf B, Messika J, Mayot T, et al. Impact of high-flow nasal cannula oxygen therapy on intensive care unit patients with acute respiratory failure: A prospective observational study. J Crit Care 2012;27:324.e9.
- Roca O, Riera J, Torres F, Masclans JR. High-flow oxygen therapy in acute respiratory failure. Respir Care 2010;55:408.
- Parke R, McGuinness S, Eccleston M. Nasal high-flow therapy delivers low level positive airway pressure. Br J Anaesth 2009;103:886.
- Mulondo J, Maleni S, Aanyu-Tukamuhebwa H, et al. Efficacy and safety of oxygen-sparing nasal reservoir cannula for treatment of pediatric hypoxemic pneumonia in Uganda: A pilot randomized clinical trial. BMC Pulm Med 2020;20:230.
- Yuste ME, Moreno O, Narbona S, et al. Efficacy and safety of high-flow nasal cannula oxygen therapy in moderate acute hypercapnic respiratory failure. Rev Bras Ter Intensiva 2019;31:156-163.
- Nishimura M. High-flow nasal cannula oxygen therapy in adults. J Intensive Care 2015;3:15.
- Messika J, Ben Ahmed K, Gaudry S, et al. Use of high-flow nasal cannula oxygen therapy in subjects with ARDS: A 1-year observational study. Respir Care 2015;60:162.
- Carratalá, Perales JM, Llorens P, Brouzet B, et al. High-flow therapy via nasal cannula in acute heart failure. Rev Esp Cardiol 2011;64:723.
- Doshi PB, Whittle JS, Dungan G 2nd, et al. The ventilatory effect of high velocity nasal insufflation compared to non-invasive positive-pressure ventilation in the treatment of hypercapneic respiratory failure: A subgroup analysis. Heart Lung 2020;49:610-615.
- Haywood ST, Whittle JS, Volakis LI, et al. HVNI vs NIPPV in the treatment of acute decompensated heart failure: Subgroup analysis of a multi-center trial in the ED. Am J Emerg Med 2019;37:2084-2090.
- Peters SG, Holets SR, Gay PC. High-flow nasal cannula therapy in do-not-intubate patients with hypoxemic respiratory distress. Respir Care 2013;58:597-600.
- Rochwerg B, Granton D, Wang DX, et al. High flow nasal cannula compared with conventional oxygen therapy for acute hypoxemic respiratory failure: A systematic review and meta-analysis. Intensive Care Med 2019;45:563.
- Ferreyro BL, Angriman F, Munshi L, et al. Association of noninvasive oxygenation strategies with all-cause mortality in adults with acute hypoxemic respiratory failure: A systematic review and meta-analysis. JAMA 2020;324:57-67.
- Ou X, Hua Y, Liu J, et al. Effect of high-flow nasal cannula oxygen therapy in adults with acute hypoxemic respiratory failure: A meta-analysis of randomized controlled trials. CMAJ 2017;189:E260.
- Monro-Somerville T, Sim M, Ruddy J, et al. The effect of high-flow nasal cannula oxygen therapy on mortality and intubation rate in acute respiratory failure: A systematic review and meta-analysis. Crit Care Med 2017;45:e449.
- Raoof S, Nava S, Carpati C, Hill NS. High-flow, noninvasive ventilation and awake (nonintubation) proning in patients with coronavirus disease 2019 with respiratory failure. Chest 2020;158:1992-2002.
- Agarwal A, Basmaji J, Muttalib F, et al. High-flow nasal cannula for acute hypoxemic respiratory failure in patients with COVID-19: Systematic reviews of effectiveness and its risks of aerosolization, dispersion, and infection transmission. Can J Anaesth 2020;67:1217-1248.
- Demoule A, Vieillard Baron A, Darmon M, et al. High-flow nasal cannula in critically ill patients with severe COVID-19. Am J Respir Crit Care Med 2020;202:1039-1042.
- Ni YN, Luo J, Yu H, et al. Can high-flow nasal cannula reduce the rate of endotracheal intubation in adult patients with acute respiratory failure compared with conventional oxygen therapy and noninvasive positive pressure ventilation?: A systematic review and meta-analysis. Chest 2017;151:764-775.
- Ni YN, Luo J, Yu H, et al. The effect of high-flow nasal cannula in reducing the mortality and the rate of endotracheal intubation when used before mechanical ventilation compared with conventional oxygen therapy and noninvasive positive pressure ventilation. A systematic review and meta-analysis. Am J Emerg Med 2018;36:226-233.
- Leung CCH, Joynt GM, Gomersall CD, et al. Comparison of high-flow nasal cannula versus oxygen face mask for environmental bacterial contamination in critically ill pneumonia patients: A randomized controlled crossover trial. J Hosp Infect 2019;101:84-87.
- Leonard S, Strasser W, Whittle JS, et al. Reducing aerosol dispersion by high flow therapy in COVID-19: High resolution computational fluid dynamics simulations of particle behavior during high velocity nasal insufflation with a simple surgical mask. J Am Coll Emerg Physicians Open 2020;1:578-591.
- Kang BJ, Koh Y, Lim CM, et al. Failure of high-flow nasal cannula therapy may delay intubation and increase mortality. Intensive Care Med 2015;41:623.
- Keenan SP, Sinuff T, Cook DJ, Hill NS. Which patients with acute exacerbation of chronic obstructive pulmonary disease benefit from noninvasive positive-pressure ventilation? A systematic review of the literature. Ann Intern Med 2003;138:861.
- Osadnik CR, Tee VS, Carson-Chahhoud KV, et al. Non-invasive ventilation for the management of acute hypercapnic respiratory failure due to exacerbation of chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2017;7:CD004104.
- Lindenauer PK, Stefan MS, Shieh MS, et al. Outcomes associated with invasive and noninvasive ventilation among patients hospitalized with exacerbations of chronic obstructive pulmonary disease. JAMA Intern Med 2014;174:1982-1993.
- Masip J, Roque M, Sánchez B, et al. Noninvasive ventilation in acute cardiogenic pulmonary edema: Systematic review and meta-analysis. JAMA 2005;294:3124.
- Berbenetz N, Wang Y, Brown J, et al. Non-invasive positive pressure ventilation (CPAP or bilevel NPPV) for cardiogenic pulmonary oedema. Cochrane Database Syst Rev 2019;4:CD005351.
- Gray A, Goodacre S, Newby DE, et al. Noninvasive ventilation in acute cardiogenic pulmonary edema. N Engl J Med 2008;359:142.
- Collins SP, Mielniczuk LM, Whittingham HA, et al. The use of noninvasive ventilation in emergency department patients with acute cardiogenic pulmonary edema: A systematic review. Ann Emerg Med 2006;48:260.
- Yeow ME, Santanilla JI. Noninvasive positive pressure ventilation in the emergency department. Emerg Med Clin North Am 2008;26:835.
- Faraone A, Beltrame C, Crociani A, et al. Effectiveness and safety of noninvasive positive pressure ventilation in the treatment of COVID-19-associated acute hypoxemic respiratory failure: A single center, non-ICU setting experience. Intern Emerg Med 2020; Nov 22:1–8.
- Xu XP, Zhang XC, Hu SL, et al. Noninvasive ventilation in acute hypoxemic nonhypercapnic respiratory failure: A systematic review and meta-analysis. Crit Care Med 2017;45:e727.
- Stefan MS, Nathanson BH, Lagu T, et al. Outcomes of noninvasive and invasive ventilation in patients hospitalized with asthma exacerbation. Ann Am Thorac Soc 2016;13:1096-1104.
- Stefan MS, Priya A, Pekow PS, et al. The comparative effectiveness of noninvasive and invasive ventilation in patients with pneumonia. J Crit Care 2018;43:190.
- Zhang Y, Fang C, Dong BR, et al. Oxygen therapy for pneumonia in adults. Cochrane Database Syst Rev 2012;CD006607.
- Keenan SP, Sinuff T, Cook DJ, Hill NS. Does noninvasive positive pressure ventilation improve outcome in acute hypoxemic respiratory failure? A systematic review. Crit Care Med 2004;32:2516.
- Althoff MD, Holguin F, Yang F, et al. Noninvasive ventilation use in critically ill patients with acute asthma exacerbations. Am J Respir Crit Care Med 2020;202:1520.
- Chiumello D, Coppola S, Froio S, et al. Noninvasive ventilation in chest trauma: Systematic review and meta-analysis. Intensive Care Med 2013;39:1171.
- Miller DC, Beamer P, Billheimer D, et al. Aerosol risk with noninvasive respiratory support in patients with COVID-19. J Am Coll Emerg Physicians Open 2020;1:521-526.
- Harbut P, Gozdzik W, Stjernfält E, et al. Continuous positive airway pressure/pressure support pre-oxygenation of morbidly obese patients. Acta Anaesthesiol Scand 2014;58:675-680.
- Baillard C, Fosse JP, Sebbane M, et al. Noninvasive ventilation improves preoxygenation before intubation of hypoxic patients. Am J Respir Crit Care Med 2006;174:171.
- Nong L, Liang W, Yu Y, et al. Noninvasive ventilation support during fiberoptic bronchoscopy-guided nasotracheal intubation effectively prevents severe hypoxemia. J Crit Care 2020;56:12.
- [No authors listed]. Noninvasive ventilation for severe cardiogenic pulmonary edema. Evidence-Based Medicine Consult. https://www.ebmconsult.com/articles/noninvasive-ventilation-cpap-bipap-cardiogenic-pulmonary-edema
Adequate oxygenation is a priority in all acutely ill emergency department patients. Oxygen may be delivered through a variety of devices, including low-flow systems or high-flow systems.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
