Tibolone Fracture Trial
Tibolone Fracture Trial
Abstract & Commentary
By Leon Speroff, MD, Editor, Professor of Obstetrics and Gynecology, Oregon Health and Science University, Portland, is Editor for OB/GYN Clinical Alert.
Synopsis: A randomized clinical trial reported that tibolone reduced vertebral fractures by 45% and non-vertebral fractures by 26%.
Source: Cummings SR, et al. The effects of tibolone in older postmenopausal women. New Engl J Med 2008;359:697-708.
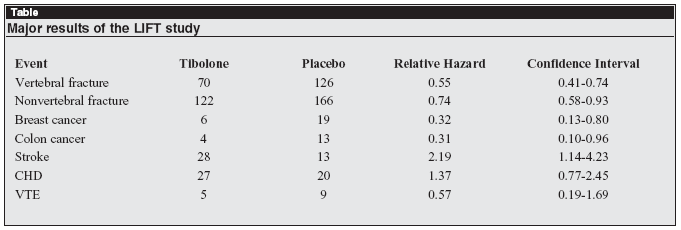
The lift study (Long-Term Intervention on Fractures with Tibolone) was a randomized, placebo-controlled multicenter trial in 22 countries of tibolone, 1.25 mg, given daily over 3 years. The 4538 women who participated in the trial were age 60-85, all at high risk of fractures because of osteoporosis, and all treated with calcium and vitamin D supplementation. The study was stopped in February 2006 after a mean treatment of 34 months because of an increased risk of stroke. The risks of all events were assessed after 5 years of follow up. Major results are listed in the Table, below.
The reduction of fractures was about four times as great in women who already had a vertebral fracture upon entry to the study compared with women who had not had a fracture at baseline. It is noteworthy that the number of falls in the treated group was 25% less. The increase in stroke was greater in the oldest women (older than age 70).
Commentary
Although tibolone is available in 90 countries, it is not an approved drug in the United States. Nevertheless it is obtainable over the internet, and it is important to be aware of the results from recent randomized trials. Based on previous bone density studies, the results of the LIFT trial on fracture reduction were not unexpected. The magnitude of the effect is roughly comparable to that with estrogen, bisphosphonates, and raloxifene (with the important exception being a lack of effect of raloxifene on hip fractures). The reduction of breast cancer was comparable to that reported with tamoxifen and raloxifene, but this was not a primary endpoint of the study. Although the difference was not statistically significant, there were 4 cases of endometrial cancer in the tibolone group and none in the placebo group.
The reported risk of stroke is similar to that observed with estrogen. In the Women's Health Initiative, no increase in stroke was observed in women younger than age 70 who had an absence of stroke risk factors. It seems prudent to avoid the use of tibolone in elderly women and in women who are at risk for stroke (specifically those with hypertension, diabetes, or atrial fibrillation, and those who smoke).
The OPAL study (Osteoporosis Prevention and Arterial effects of tiboLone) was a 3-year, randomized, double-blind trial in 6 U.S. centers and 5 European centers, treating 866 postmenopausal women with either 2.5 mg tibolone daily, 0.625/2.5 mg daily of conjugated estrogens/medroxyprogesterone acetate, or placebo.1 Unfortunately, the OPAL trial did not achieve its goal of providing robust data on cardiovascular effects, due to the older age of the women and the notably different results in American and European women. There continues to be good reason to believe that tibolone will have a neutral effect in terms of coronary heart disease.
Tibolone treatment of postmenopausal women is as effective as estrogen therapy in relieving hot flushes, preventing bone loss, and increasing vaginal lubrication, but it stimulates libido to a greater degree than estrogen. There is less breast tenderness and mastalgia with tibolone. Endometrial safety has been reported to be comparable to that achieved with continuous combined estrogen-progestin regimens, and with a lower rate of breakthrough bleeding. The previously reported increased risks of breast cancer and endometrial cancer in observational studies very likely represent "preferential prescribing" of tibolone in Europe. Women prescribed tibolone in Europe more often had chronic breast disease, a personal history of breast cancer, previous dysfunctional uterine bleeding, hypertension, and previous uterine operations. Most importantly, more women prescribed tibolone had a history of treatment with unopposed estrogen. Thus, clinicians were more likely to prescribe tibolone to women they believed were at higher risks for these two cancers, and this would yield higher rates in treated groups compared with control groups. The standard dose of tibolone for many years was 2.5 mg daily, but the new studies support the use of the lower dose, 1.25 mg, with no apparent loss of efficacy. Tibolone continues to be an appropriate choice for hormonal therapy, suitable for many postmenopausal women.
Reference
- Bots ML, et al. The effect of tibolone and continuous combined conjugated equine oestrogens plus medroxy-progesterone acetate on progression of carotid intima-media thickness. Eur Heart J 2006;27:746-755.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
