Special Feature: Maintenance Therapy for Ovarian Cancer: Are We There Yet?
Special Feature
Maintenance Therapy for Ovarian Cancer: Are We There Yet?
By Robert L. Coleman, MD, Associate Professor, University of Texas; M.D. Anderson Cancer Center, Houston, is Associate Editor for OB/GYN Clinical Alert.
Dr. Coleman reports no financial relationship to this field of study.
July of this year represented the 5-year anniversary of the publication of Gynecologic Oncology Group (GOG) protocol 178, a pivotal trial demonstrating the significant impact of 9 additional months of paclitaxel chemotherapy to women who had achieved a complete clinical response to primary therapy.1 The trial was so remarkably positive for women randomized to receive one additional year of monthly paclitaxel therapy that it was closed prematurely for ethical concerns — meeting a predefined early closure benchmark based on progression-free survival (median PFS: 28 months vs 21 months, P = 0.0023).
Those unfamiliar with this stream of clinical research may be surprised to know that the results have largely been disregarded. The primary impact has been inconsistent or incomplete use of the strategy as studied, or use of other approaches lacking in evidence base. I draw emphasis to evidence-based treatment, as completion therapy or "consolidation" is frequently recommended or prescribed; what's missing is the evidence base.
This Special Feature will explain the impetus to develop effective maintenance treatment, present what has been explored in phase III prospective trials, and discuss why, despite the positive results of the pivotal trial, patients and physicians shun routine use of the "winning arm" of this trial. Ongoing and planned confirmatory concepts will also be introduced.
The Problem
Women with advanced stage ovarian cancer have effective chemotherapeutic options for care administered after surgical cytoreduction (debulking). Response rates vary from 50% to 80% with about half of these being complete; that is, no evidence of disease following exam and imaging and resolution of their elevated biomarker (usually CA125).2,3
In the office, the discussion with this fortunate patient is positive and upbeat and frequently overshadows attendant cumulative toxicities related to primary therapy, such as neuropathy, fatigue, myalgias, and alopecia. Despite the enthusiastic interaction, probability of recurrence within 2 years is substantive (about 50%-60%) and its occurrence is usually fatal.3 Even in the setting of a negative reassessment surgical procedure, the risk is only marginally reduced.
Given these statistics, it is logical to hypothesize the clinical observations might be the result of incomplete response to an abbreviated front-line chemotherapy program (6 cycles of therapy). Indeed, part of the historical rationale to perform a second-look surgery in these patients was to provide some guidance as to when therapy could be safely discontinued. Over time, convention settled with 6-8 cycles as adequate. Maintenance therapy is that treatment administered in the specific setting of complete clinical or pathological remission of disease following primary therapy (see Figure, below).
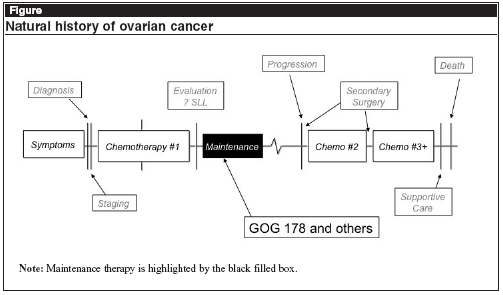
The Strategy
Given the contention that recurrence may have resulted from incomplete primary therapy, several studies were conducted under the premise that additional treatment could favorably impact these women. A simple strategy involved doubling the exposure to primary chemotherapy.
For example, Hakes et al reported a randomized phase III study comparing survival outcomes in women administered 5 cycles of chemotherapy followed by observation compared to those women given 5 additional cycles of the same therapy.4 Patients having residual disease at second-look surgery were crossed over to the 10-cycle arm. Complete response rate was similar, as was survival; additional chemotherapy only added toxicity.
Another strategy, in this same theme, involves the administration of a chemotherapeutic, which has non-cross-resistant cytotoxicity characteristics. Two large randomized clinical trials investigated the topoisomerase I inhibitor, topotecan, which has documented clinical efficacy in women with recurrent ovarian cancer.5,6 The two trials were slightly different in design, but each prescribed the novel agent for 4 additional cycles following best response to primary therapy. Despite the appeal, no difference in the primary endpoint (PFS) was observed.
To round out the "more is better" theme comes a provocative phase III study, which enrolled 110 women with stage III/IV ovarian cancer who, at second-look, were identified with either no visible disease or small-volume persistent disease, to either standard-dose cyclophosphamide and carboplatin or high-dose cyclophosphamide and carboplatin with stem cell support.7 As expected, a low, but measurable, treatment-related mortality rate (3%) and lower completed therapy rate was observed in the high-dose arm, but no effect on PFS or overall survival (OS) was recorded. It appears this avenue is unfruitful.
Another strategy, based on our observations of successful treatment of women with early stage disease, is radiation. Since the at-risk target tissue in these patients involves the peritoneal cavity, two strategies were investigated for maintenance: whole abdominal radiation therapy (WART) and intraperitoneal radio-phosphorus (32P).
Sorbe and colleagues conducted a three-arm randomized trial of WART, chemotherapy, or observation following second-look surgery.8 In the cohort of women identified as being in pathological complete remission (negative second-look surgery), no difference was observed between either therapy arm and observation. A similar experience was observed in GOG protocol 93, in which women with pathological complete response were randomized to either 32P or observation; PFS and OS were similar.9
The ability for ovarian cancer cells to grow in the host suggests faulty, escaped, or bypassed immunoregulatory processing. There is intense interest in leveraging the potential immune host response for prevention and therapy in this disease.9
Relative to maintenance therapy, several attempts have been unsuccessfully made. In one, interferon-a was administered (up to 2 years) subcutanteously to 149 women in complete clinical remission following primary therapy.10 Compared to 151 women observed without therapy, no difference in median survival was observed (27 months vs 33 months).
In another, antibody to CA125 (oregovomab) was administered to 145 women with normal CA125 levels and no tumor following primary therapy.11 Progression-free survival, the primary endpoint, was no different between the arms. Post-hoc analysis suggested a benefit may exist in those women with highly expressing CA125 tumors that demonstrated early response to initial chemotherapy. However, in a follow-up randomized study, even in this enriched population, no benefit was observed for the experimental agent.
A new anti-idiotypic CA-125 antibody (abagovomab) is currently under investigation in this setting.12
Since early events in carcinogenesis involve specific interactions of the tumor cell and its microenvironment, several investigators have opined that targeted therapy could be beneficial in the prevention of disease progression or relapse.13 One randomized clinical trial of a unique biological agent has been reported. In this study, 243 women completing primary chemotherapy were randomized to tanomastat, a matrix metalloproteinase inhibitor, or placebo.14 Therapy was to continue until progression or 5 years. Despite its novelty, no difference was observed between the cohorts. While disappointing, the premise in this strategy is extremely relevant and likely to serve as an avenue to future investigation, as the menu of interesting compounds expands.
The Kicker
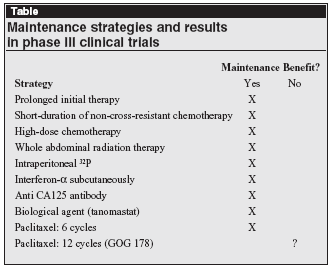
The investigational environment, vis-à-vis this clinical experience, is an important context upon which to consider the results of GOG 178 (see Table, left). There has been much criticism of the study and scrutiny of its results, particularly in light of a similar trial, reported in abstract form, which was closed prematurely due to an unplanned futility analysis demonstrating no impact of 6 cycles of paclitaxel vs no further therapy.15 The contribution of this latter study to our knowledge base, unfortunately, is suspect, at best, due to this unplanned issuance.
Nevertheless, there are several valid shortcomings to GOG 178, including an incomplete evaluation of the impact of toxicity resulting from the additional therapy, and the lack of overall survival information. Further, it has been intimated that no statistical difference in overall survival is expected between the treatment cohorts.16 No doubt, these factors, aligned with patient trepidation, have influenced clinicians' practice patterns. In the absence of a confirmatory trial, it is clear that expansion of maintenance therapy will be justifiably muted.
Fortunately, the GOG and other cooperative groups have embraced this challenge (e.g., novel chemotherapeutics, vaccine, and biologicals), and ongoing are trials to address each of these deficiencies, not the least of which is the consideration that not every patient is destined to recur. Our current inability to a priori identify these individuals requires that the study eligibility be broadly defined and intensifies the discrimination of treatment-induced and treatment-enhanced toxicity relative to any attained efficacy endpoints. Future work in patient genomic profiling may avail this requirement.
The Wrap-up
The clinical concern of patients with advanced ovarian cancer and their health care providers is simply — cure. Statistically, only marginal gains on this endpoint have been observed over the last 3 decades despite the array of therapies available. However, median survival has nearly doubled and continues to improve, largely the result of this therapeutic environment. Strategies to push the envelope, such as maintenance therapy, are worthy ventures, as one just might be identified which could provide the direct inhibition of disease re-growth and provide an avenue for sustained disease-free survival.
References
- Markman M, et al. Phase III randomized trial of 12 versus 3 months of maintenance paclitaxel in patients with advanced ovarian cancer after complete response to platinum and paclitaxel-based chemotherapy: A Southwest Oncology Group and Gynecologic Oncology Group trial. J Clin Oncol 2003;21:2460-2465.
- McGuire WP, et al. Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. N Engl J Med 1996;334:1-6.
- Cannistra SA. Cancer of the ovary. N Engl J Med 2004;351:2519-2529.
- Hakes TB, et al. Randomized prospective trial of 5 versus 10 cycles of cyclophosphamide, doxorubicin, and cisplatin in advanced ovarian carcinoma. Gynecol Oncol 1992;45:284-289.
- De Placido S, et al. Topotecan compared with no therapy after response to surgery and carboplatin/paclitaxel in patients with ovarian cancer: Multicenter Italian Trials in Ovarian Cancer (MITO-1) randomized study. J Clin Oncol 2004;22:2635-2642.
- Pfisterer J, et al. Randomized phase III trial of topotecan following carboplatin and paclitaxel in first-line treatment of advanced ovarian cancer: A gynecologic cancer intergroup trial of the AGO-OVAR and GINECO. J Natl Cancer Inst 2006;98:1036-1045.
- Cure H, et al. Phase III randomized trial of high-dose chemotherapy (HDC) and peripheral blood stem cell (PBSC) support as consolidation in patients (pts) with advanced ovarian cancer (AOC): 5-year follow-up of a GINECO/FNCLCC/SFGM-TC study. 2004 ASCO Annual Meeting Proceedings 2004;22:5006.
- Sorbe B. Consolidation treatment of advanced (FIGO stage III) ovarian carcinoma in complete surgical remission after induction chemotherapy: A randomized, controlled, clinical trial comparing whole abdominal radiotherapy, chemotherapy, and no further treatment. Int J Gynecol Cancer 2003;13:278-286.
- Buckanovich RJ, et al. Endothelin B receptor mediates the endothelial barrier to T cell homing to tumors and disables immune therapy. Nat Med 2008;14:28-36.
- Hall GD, et al. Maintenance treatment with interferon for advanced ovarian cancer: Results of the Northern and Yorkshire gynaecology group randomised phase III study. Br J Cancer 2004;91:621-626.
- Berek JS, et al. Randomized, placebo-controlled study of oregovomab for consolidation of clinical remission in patients with advanced ovarian cancer. J Clin Oncol 2004;22:3507-3516.
- Sabbatini P, et al. Phase I study of abagovomab in patients with epithelial ovarian, fallopian tube, or primary peritoneal cancer. Clin Cancer Res 2006;12:5503-5510.
- Spannuth WA, et al. Angiogenesis as a strategic target for ovarian cancer therapy. Nat Clin Pract Oncol 2008;5:194-204.
- Hirte H, et al. A phase III randomized trial of BAY 12-9566 (tanomastat) as maintenance therapy in patients with advanced ovarian cancer responsive to primary surgery and paclitaxel/platinum containing chemotherapy: A National Cancer Institute of Canada Clinical Trials Group Study. Gynecol Oncol 2006;102:300-308.
- Conte PF, et al. Final results of After-6 protocol 1: A phase III trial of observation versus 6 courses of paclitaxel (Pac) in advanced ovarian cancer patients in complete response (CR) after platinum-paclitaxel chemotherapy (CT). 2007 ASCO Annual Meeting Proceedings Part I 2007;25:5505.
- Markman M, et al. Survival (S) of ovarian cancer (OC) patients (pts) treated on SWOG9701/GOG178: 12 versus (v) 3 cycles (C) of monthly single-agent paclitaxel (PAC) following attainment of a clinically-defined complete response (CR) to platinum (PLAT)/PAC. 2006 ASCO Annual Meeting Proceedings Part I 2006;24:5005.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
