Evaluation and Management of Blunt Trauma Patients in the Emergency Department
Evaluation and Management of Blunt Trauma Patients in the Emergency Department
Authors: Howard Werman, MD, Professor of Emergency Medicine, Department of Emergency Medicine, The Ohio State University, Columbus; and Erica C. Kube, MD, Clinical Instructor, Chief Resident, Department of Emergency Medicine, The Ohio State University, Columbus.
Peer Reviewer: Jonathan Glauser, MD, Department of Emergency Medicine, MetroHealth Medical Center, The Cleveland Clinic, Cleveland, OH.
Introduction
Victims of blunt trauma are frequently encountered in the emergency department (ED). Forty percent of all ED visits each year are attributed to injury, which equates to about 40 million ED visits annually. Additionally, approximately one-third of intensive care unit (ICU) admissions in the United States are trauma-related. In regions that have an organized trauma system, patients with major trauma are stabilized and transported to the closest designated trauma center per the Advanced Trauma Life Support (ATLS) paradigm. Trauma centers are so designated by their ability to provide comprehensive care for the traumatized patient, both acutely and in the rehabilitative phases of their recovery. Data have shown that major trauma patients who are transferred to verified trauma centers have lower in-hospital mortality rates.1 Additionally, studies have shown that trauma patients treated at American College of Surgeons (ACS)-designated level 1 trauma centers have better survival outcomes than patients treated at ACS-designated level 2 trauma centers.2,3 A large proportion of trauma patients initially are assessed and managed in hospitals not verified as trauma centers. Care and stabilization in these facilities are a crucial part of an organized trauma system.
Epidemiology
Trauma is the leading cause of death in most developed countries for persons aged 1-44 and is estimated to become the second or third most common cause of death across all age groups by 2020. Motor vehicle crashes are the most common etiology, accounting for the greatest number of injuries and deaths. Falls are the second most common cause of unintentional injury, with infants and the elderly being at highest risk. The incidence of falls declines from infancy up to 14 years of age but then reemerges as a major cause of blunt trauma in the elderly. It is clear that trauma is a large burden on the health care system; data have shown that for every death related to injury, there are 233 ED visits, 19 admissions, and 450 follow-up physician office visits.
According to the Bureau of Labor Statistics, there are 4 million non-fatal injuries among U.S. workers in the private sector. The overall rate of 4.2 cases per 100 full-time equivalents was the lowest level in the past 5 years. Jobs in agriculture, mining, construction, and manufacturing have among the highest rates of injury, whereas the service professions were far less likely to suffer work-related injury.4 Fatal workplace accidents due to vehicle-related injuries have been substantially reduced in recent years, whereas fatal work-related falls have increased. The highest case fatality rates among all occupations occur in mining (24.8 cases per 100,000 employed) and agriculture (27.3 cases per 100,000 employed). Workers in the fishing industry and loggers have the highest occupation-specific fatality rate. Blumen5 noted a case fatality rate of 196 cases per 100,000 employed in the air medical transport industry.
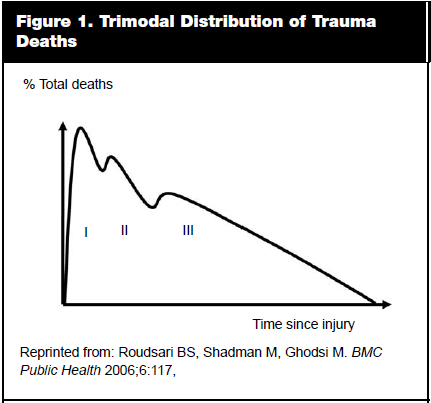
There is a trimodal distribution of death in victims of trauma. (See Figure 1.) The first peak occurs within seconds to minutes after the insult; these patients typically die from brain or spinal cord injury or rupture of aorta, heart, or other large vessels. The single most important factor that can reduce mortality in these early deaths is a comprehensive injury prevention program. Improved vehicle design, passenger restraint systems, and protective head gear are examples of interventions that have been shown to reduce trauma mortality. The second peak occurs minutes to several hours after the insult; these patients die from hemopneumothorax, ruptured spleen, liver lacerations, epidural and subdural hematomas, pelvic fractures, or blood loss from multiple injuries. The third peak occurs days to weeks later and is usually due to multiple organ failure and sepsis. Clearly, the role in the ED is to identify and address those patients at risk of early mortality, quickly recognize those injuries that can lead to death if not properly treated, and arrange for definitive care of such patients in an expeditious manner. Early aggressive resuscitation of major trauma victims also can prevent death in the third peak following injury typically caused by multi-system organ failure.
 |
There is a definite role for emergency physicians to have an impact on future injury in the period immediately after the traumatic injury has occurred, often referred to as the "teachable moment." While some traumatized patients are risk-takers and will be resistant to changes in their behaviors, other patients may have had a lapse of self-control or a moment of carelessness and would be receptive to an emergency physician discussing their endangering behavior and advising changes in the future.6 This is also an opportunity to intervene with patients suspected of drug or alcohol abuse.7 Every physician-patient encounter is an opportunity to encourage a change in the patient's behavior and lead to a reduction of trauma morbidity and mortality in the future.
Mechanism of Blunt Injury
It is important to anticipate what injuries a patient will have based on the history of the accident, as there are patterns to injury based on the particular mechanism. Specific information about the details of the event including speed of travel, position of the patient, use of restraint systems, and the height of fall should be determined by a careful history obtained from the patient, bystanders, and prehospital personnel. While the mechanism of injury is useful for predicting injury patterns, it has been shown to be a very poor predictor of appropriate triage decisions.8 This is especially true if anatomical and physiologic criteria are not considered. Several studies have looked only at mechanism of injury and severity of the actual injuries to the patient. With auto accidents specifically, studies have evaluated which mechanisms or patterns of the accident put the patient at risk for serious injury, potentially requiring a higher level of trauma care. Palanca9 found that a prolonged extrication time, vehicle intrusion, and ejection from the vehicle were the only three criteria statistically associated with major injury. Recently, Boyle10 evaluated patients with mechanism of injury as the sole triage criteria and found that only a fall greater than 5 m or entrapment more than 30 minutes were associated with severe injuries.
Blunt trauma most commonly is the result of motor vehicle collisions (MVCs), motorcycle crashes, automobile versus pedestrian, falls, assaults, and, rarely, blast injuries. Blunt trauma typically produces an inelastic collision in which kinetic energy is lost in deformation of both the vehicle and the patient involved in the collision. Strain refers to stress produced in the tissue by blunt forces. Depending on the direction of the force, this can result in tensile strain, shearing, compression, or over-pressure. This leads to specific injuries including tearing, shearing injuries, contusion, and hollow viscus rupture. (See Table 1.) With motor vehicle collisions, three separate impacts are described: In the first, the motor vehicle strikes an object; in the second, the victim collides with the motor vehicle; in the third, the soft tissues of the body strike the supporting structures in the body. Further injury from recoil is possible. Mortality is related directly to the amount of force that is transferred to the patient. Head-on collisions carry the highest mortality, whereas lower mortality rates are seen with side impact (20-35%), rollover (8-15%), and rear impact (3-5%).11,12 The speed of impact also is a major determinant of outcome, with 5% mortality for collisions at 20 mph versus an 85% mortality for collisions at speeds in excess of 50 mph.
 |
The importance of passenger restraint is emphasized by the fact that ejection from the vehicle increases the risk of major injury 300-500%, and the risk of spinal column injury is 1 in 13.13 Additionally, it is known that the current 3-point restraints required in all vehicles are 45% effective in preventing fatal injury in cars and 60% effective in preventing fatal injury in pickup trucks, SUVs, and vans.14 Data also have shown that 3-point restraints reduce severe injuries in front and front-angle crashes by 58%, compared with a reduction of 17% with lap belts only.15 On the other hand, ejection from the vehicle carries an increased risk of ICU admission, higher injury severity score (ISS), significant head injury, and higher mortality.16
Unlike occupants of automobiles, motorcyclists and bicyclists are not protected by the structure of the vehicle or its restraining devices. The only sources of protection for these patients are any protective clothing or helmet that they are wearing. Injuries typically are sustained by the following mechanisms: frontal or lateral impact, ejection, falling off the bike, or "laying the bike down." There can be injury to any of the extremities, chest, abdomen, pelvis, or head depending on how the motorcyclist or bicyclist and the bike came to a stop. It is important to inquire about any protective devices that the patient was using at the time of the incident, such as protective clothing or a helmet. Helmets have been shown to decrease the amount of injury to the head by dispersing energy.
Pedestrian versus automobile injury is a fairly common problem, most often in urban areas, with children being most frequently involved. Most injuries to pedestrians, in order of incidence, are to the chest, head, and lower extremity. There are three described impacts in pedestrian versus automobile collisions. First, the pedestrian strikes the vehicle's bumper. This can result in pelvis or leg injuries in adults and chest and abdominal injuries in children, although these injury patterns are changing with changes in the design of vehicles. These injuries ultimately depend on where the patient's point of impact was in relation to the vehicle, which will vary among small passenger cars, sport utility vehicles, and trucks. The second impact occurs when the pedestrian strikes the hood and windshield of the automobile; this usually results in injuries to the head, chest, and abdomen. The third impact occurs when the pedestrian strikes the ground; this results in spine and head injuries.
Falls result in injury by abrupt deceleration. The most important determinant of injury is the height of the fall. The extent of injuries also will depend on the contact surface, the speed at which the body was falling, the elasticity of the involved tissues, and the position of the body when it contacted the surface. Harder contact surfaces increase the rate of deceleration and cause more injuries than softer surfaces. A larger surface area for contact will disperse the deceleration energy and result in less severe injuries than a smaller surface area. As with all previous examples, it is very important to gather as much detail about the conditions associated with the injury to allow prediction of what injuries the patient may have.
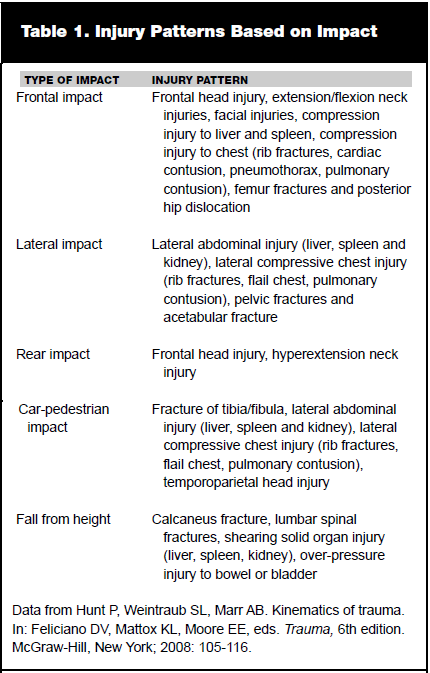
Blast injuries are divided into primary, secondary, and tertiary. Primary blast injuries are a direct result of the pressure wave created by a blast traveling at speeds of 3000 to 8000 meters per second; gas-filled organs tend to be most severely affected by this pressure wave. (See Figure 2.) The pressure wave dissipates within a short distance of the explosion and, thus, primary blast injuries are not typically the most prevalent injuries associated with explosions. Blast injuries are non-linear, complex, and may be enhanced by reflection of the pressure wave off solid objects. Thus, blasts in closed spaces produce more devastating injuries. Enhanced explosive devices generate a secondary explosion and, thus, cause a wider area of damage. The most common primary blast injury is ruptured tympanic membrane. Less common injuries include lung contusion, pneumothorax, alveolar rupture, retinal detachment, intraocular hemorrhage, air embolism, and colonic rupture. Traumatic amputations from blasts carry a high mortality.17
 |
Secondary blast injuries are a result of flying objects striking the patient. The resultant injuries are the major cause of death among victims of blast injury. The injury mechanics are very similar to penetrating missile trauma. Tertiary injuries occur when the patient is thrown against another object or the ground as the result of the wind forces that follow the initial pressure wave, potentially resulting in additional injuries. An additional pattern is quaternary injury produced by burns, toxins, asphyxiants, or radiological agents released by the explosion.
Head Trauma
It is estimated that there are 2 million cases of brain injury annually in the United States, and approximately 10% of these patients die before arrival at the hospital. The most common overall cause of traumatic death is injury to the central nervous system. More than 80% of patients with head injuries ultimately will be classified as mild, 10% as moderate, and 10% as severe. For patients suspected of having a brain injury, the primary focus should be patient stabilization and prevention of further injury to the brain by maintaining adequate perfusion and oxygenation of the brain tissue. Additional focus should be on identification of expanding mass lesions where rapid assessment, diagnosis, and evaluation by a neurosurgeon must be ensured. Mild traumatic brain injury (concussion) will be reviewed in an upcoming issue of Emergency Medicine Reports and therefore this section will focus mainly on more serious injuries.
It has been proposed that serum biomarkers released at the time of injury may herald significant cerebral trauma. When cellular damage occurs, proteins including S100 proteins, neuron-specific enolase, and cleaved Tau protein are released into the extracellular matrix, the cerebrospinal fluid, and, ultimately, into the serum if there is disruption of the blood-brain barrier. Such compounds are elevated in severe trauma.18
Non-contrast CT of the head is the imaging modality of choice in acute head injury; however, it is critical that obtaining the CT scan not delay transfer of the patient to a trauma center with neurosurgical capabilities when necessary. Classic findings of a subdural or epidural hematoma typically are visualized on initial CT scans, whereas initial findings in brain contusion or intracranial hematomas may be subtle or not seen on initial scans. The treating physician should maintain a high index of suspicion for these conditions in the appropriate clinical setting.
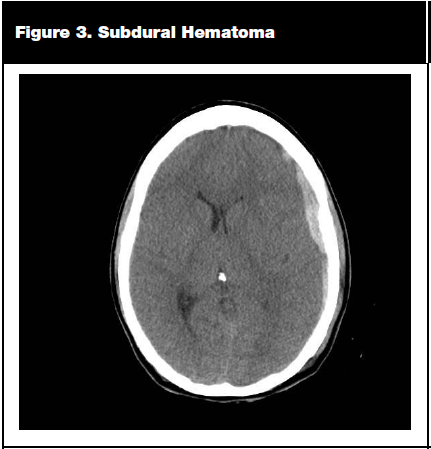
Subdural hematoma represents the most common traumatic mass lesion and is estimated to occur in 20-40% of severely head-injured patients. A subdural hematoma is a collection of blood between the dura and arachnoid meningeal layers and is usually a result of disruption of the bridging veins with associated injury to underlying brain parenchyma. On CT scan, they are classically identified as a crescent-shaped density. (See Figure 3.) Treatment involves early stabilization and consultation with a neurosurgeon; morbidity and mortality depend on the severity of the injury to the underlying brain parenchyma and how rapid the onset is of mass effect from the space-occupying blood.
 |
Epidural hematomas result from blood collecting in the potential space between the dura and the inner table of the skull. They usually result from laceration of the middle meningeal artery caused by injury to the temporal bones or from laceration of the dural sinuses, which leads to venous bleeding into the epidural space. Epidural hematoma appears as a lens-shaped density on head CT. (See Figure 4.) In contrast to the typical association of underlying brain tissue with subdural hematomas, epidural hematomas result from injury to the skull and rarely involve the underlying brain parenchyma. Patient morbidity and mortality result from the direct mass effect of the clot and resultant herniation. Thus, an epidural hematoma must be identified and evacuated in a timely fashion.
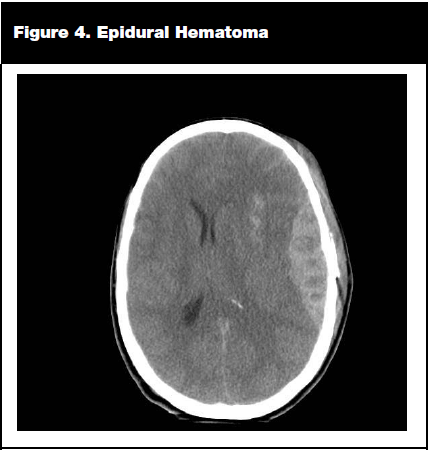 |
Contusion to the brain parenchyma itself also is seen in up to 30% of severe head injuries. These contusions are the result of movement of the brain within the cranial vault and can appear at a site distant from the site of the blunt trauma; an example is the so-called "contracoup injury." Contusions can take 12-24 hours to appear on the CT scan, with a depressed level of consciousness being the only physical sign of the injury. As these injuries progress, they can increase in size due to progressive edema, which can result in mass effect within the brain and declining patient level of consciousness.
Subarachnoid hemorrhages (SAH) and intraventricular hemorrhages also can result from trauma. These hemorrhages may be less dangerous for the patient because the blood is either spread out over the surface of the brain or within the ventricles; such lesions do not generally cause mass effect. However, the presence of SAH should alert the clinician that the patient has a 63-77% increased risk of associated cerebral contusion and a 44% increased risk of subdural hematoma.19
As stated above, it is critically important to identify these injuries and ensure adequate oxygenation and perfusion of the brain tissues. While the treating physician cannot alter the injury that has occurred, he or she can minimize further injury to surrounding brain parenchyma with appropriate management in the ED. When patients with suspected or confirmed intracranial injury are identified, the emergency physician always should start with the ABCD assessment and consider early intubation. If the patient will require transfer to a trauma facility with neurosurgical capabilities, it is especially prudent to assume control of the airway and prevent hypoxia. Additionally, it has been estimated that a single episode of hypotension increases mortality in an adult with head injury by 50%.20 Therefore, adequate fluid resuscitation is paramount in ensuring that the vulnerable brain tissue is not further deprived of adequate perfusion. Hyperventilation was advocated in the past for patients with head injuries. It was previously thought that a decreased PCO2 would cause cerebral vasoconstriction that would result in decreased cerebral blood flow and lower intracranial pressure. Further research on this matter has suggested that aggressive hyperventilation actually may be detrimental to the brain tissue because it decreases cerebral blood flow and thereby causes ischemia.21,22 At present, routine hyperventilation generally is discouraged except in the case of increased intracranial pressure (ICP) with impending herniation. In that situation, hyperventilation may be used for short intervals to decrease the risk of ischemia until definitive treatment can be accomplished. In line with preventing further injury, it is recommended that patients with significant head injury be treated in the ED with anti-epileptic medications, such as carbamazepine and phenytoin, to prevent seizures.23
With injury to the brain tissue, there is potential for swelling, which also increases ICP. Several therapeutic modalities have been studied to decrease the swelling and subsequent rise in ICP. Mannitol is useful for decreasing ICP by drawing extracellular water into the vascular space and subsequently decreasing the volume of intracranial contents. An intact blood-brain barrier is required for mannitol administration to be effective so that extracellular water can move down the osmolar gradient into the more concentrated blood. Additionally, the diuresis associated with mannitol use can lead to systemic hypotension and hypoperfusion; thus, the drug is contraindicated in hypotensive patients. With continuous mannitol infusion, there has been an observed phenomenon called "rebound effect," in which accumulation of mannitol in the extracellular spaces causes a reverse osmotic gradient and subsequent accumulation of water in the brain. The use of corticosteroids in the setting of brain injury also has been reevaluated, with the most recent literature indicating that steroids are of no benefit and may, in fact, result in increased mortality. Thus, steroids should not be used routinely for these patients.24 The increased mortality is believed to be the result of impaired wound healing, induction of diabetes, and immunosuppression.
Once the patient with significant head injury is either suspected or diagnosed, it is important to engage in early discussion with a neurosurgeon or transferring institution to arrange for rapid transport to the appropriate facility. According to the ATLS protocol, any delay in transport to obtain a head CT is inappropriate. Instead, the patient should be transported rapidly to the facility most prepared to provide definitive treatment.
Chest Trauma
Chest injuries are estimated to be responsible for 20-25% of all trauma deaths and are a common cause of morbidity in traumatized patients. Only 10-15% of chest trauma patients ultimately will require operative management; most are treated with analgesia, intubation, pulmonary toilet, and tube thoracostomy. Thus, the stabilizing physician should be comfortable with performing these procedures.
It is important to consider the possible injuries based on the mechanism of injury. With high-energy mechanisms such as MVCs or falls from great heights, one must suspect thoracic trauma and investigate appropriately. It is also very important that tubes, such as endotracheal and thoracostomy tubes, be placed when appropriate prior to initiating patient transfer to the appropriate facility.
Radiologic studies have a critical role in the evaluation of chest trauma; however, imaging never should replace the role of good clinical assessment. Patients with suspected chest trauma, as evidenced by airway compromise, respiratory distress, massive crepitus, paradoxical chest wall movement, or absent or markedly diminished breath sounds, should have their airways secured, vascular access obtained, and have a needle or tube thoracostomy performed before proceeding with the standard portable chest radiograph (CXR). The CXR is helpful in identifying or suggesting osseous injury, pulmonary contusion, pneumothorax, and mediastinal hematoma. Remember to consider patient position in interpreting the initial chest radiograph.
The presence of life-threatening injuries can be missed on CXR. CT is a more sensitive imaging modality to evaluate all soft-tissue and bony structures within the chest. One should consider imaging thoracic vasculature with CT scan in patients with high-energy impacts or evidence of significant trauma to the chest, as evidenced by scapular fractures or first or second rib fractures. There is emerging evidence that chest CT may be more sensitive in identifying significant injuries following blunt trauma.25,26
There is an expanding role for ultrasound to aid in the diagnosis of thoracic injuries, which has been referred to as the extended FAST. Ultrasound has the benefits of being a rapid bedside test without risks of ionizing radiation and can assist the user in identifying thoracic injuries, such as lung contusion27 and pnemothorax.28 Some also have recommended obtaining a subdiaphragmatic view to evaluate for the presence of pleural fluid.27 Sisley, et al., have demonstrated that the detection of traumatic hemothorax with ultrasound has a sensitivity of 97.5% and a specificity of 99.7%.29 For the detection of pneumothorax, the sonographer uses sonographic signs to assess the integrity of contact between the visceral and parietal pleura; the absence of this interface may indicate the presence of air in the pleural space.
Pulmonary contusions are the most common injury to the lung in blunt trauma, occurring in 17-75% of patients. They are a result of injury to the walls of the alveoli, causing the leakage of blood from pulmonary vessels into the interstitial and alveolar spaces and typically occur in the lung tissue adjacent to the bony structures of the rib cage. As stated above, CXR may be helpful in identifying pulmonary contusion. However, contusions may not be visible for up to 6 hours after the injury. CT of the chest is much more sensitive and, in some studies, is being used to help predict the chances of the patient having complications in the future.27
Pneumothorax has been reported in 20-25% of trauma patients and 30-40% of blunt chest trauma patients. It typically is caused by a broken rib that lacerates the underlying lung parenchyma, but it also can result from the rupture of the lung from high intrathoracic pressure during impact. It is identified clinically by a lack of breath sounds on the affected side, and radiologically by a defined line that represents the visceral pleura and a lack of lung markings beyond this line on CXR. (See Figure 5.) CT scan can identify 10-50% of pneumothoraces that are not seen on CXR. (See Figure 6.) If the pneumothorax is greater than 20% or the patient is symptomatic, the patient generally is treated with chest tube placement.
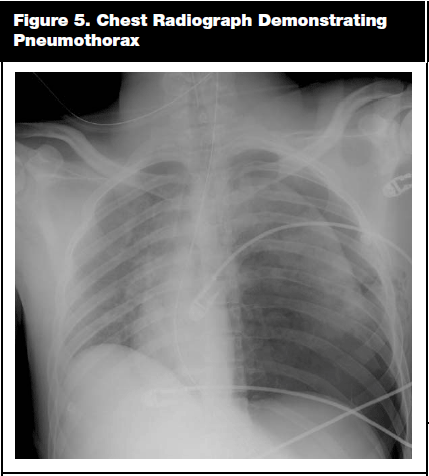 |
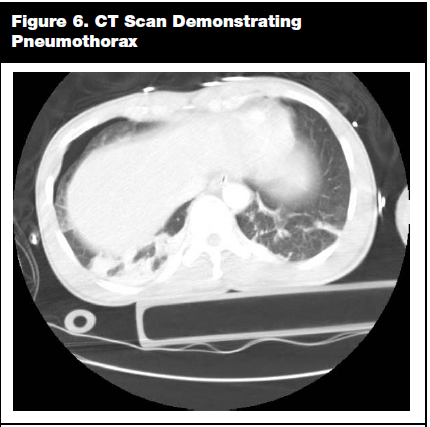 |
Tension pneumothorax results from the progressive accumulation of air in the pleural space, causing high intrathoracic pressures. This can result in compression of the vena cava, impairing venous return and decreasing cardiac output. Clinically, this appears as absent breath sounds on the side of the pneumothorax, tracheal deviation to the opposite side, hypotension, severe respiratory distress, and neck vein distention. Treatment is immediate needle decompression with a 14- or 16-gauge intravenous catheter in the second intercostal space, with resultant conversion to a simple pneumothorax. This needle decompression then is followed by tube thoracostomy to treat the pneumothorax.
Hemothorax, the collection of blood in the pleural space, is seen in up to 50% of blunt chest trauma patients. Bleeding can be from the chest wall, lung, diaphragm, mediastinum, or pleura. If the volume of blood is large enough, it can be visualized on an early CXR. CT is highly sensitive for detecting even a small amount of blood and can be used to elucidate the source of the bleeding. Initial treatment involves chest tube placement and consultation with the trauma and/or thoracic surgeon. Patients with greater than 1500 mL of blood on initial chest tube placement or less than 1500 mL initially but ongoing bleeding at a rate of 200 mL/hr for 2-4 hours should be taken to the operating room for management of their intrathoracic bleeding.
The most common skeletal injury in blunt chest trauma is rib fracture. The most concerning aspect of rib fractures is the potential injury to the underlying lung parenchyma. When there are several broken ribs in succession on the same side, there is a potential for flail chest. This results in a free-floating segment of the chest wall that moves paradoxically with respiration and can cause hemodynamic instability. These injuries are uniformly associated with pulmonary contusion and carry a mortality rate of 10-20%. Trinkle30 showed that the respiratory insufficiency associated with flail chest was due to the underlying pulmonary contusion rather than as a result of the paradoxical movement of the flail segment as was previously thought. Pulmonary contusion can be seen on chest x-ray as a hazy opacity at the site of injury. This injury may not be obvious immediately on initial chest x-ray and may only appear after fluid has diffused from the vascular space into the alveolar space. CT scan of the chest is more sensitive than chest x-ray in identifying early pulmonary contusion and therefore is recommended as the imaging modality of choice in patients with thoracic trauma to more thoroughly identify all associated injuries.31
Multiple rib fractures should always suggest that the patient has a flail chest. Treatment of flail chest involves adequate analgesia, careful fluid resuscitation, non-invasive or invasive mechanical ventilation, oxygen supplementation, aggressive pulmonary toilet, and consideration of surgical chest wall stabilization. The pain associated with flail chest should be treated aggressively as the patient is stabilized and all injuries are evaluated. Consideration for epidural analgesia has been advocated to prevent the risk of respiratory depression associated with parenteral opioids. Appropriate fluid resuscitation is recommended, especially as patients with flail chest usually have other significant traumatic injuries; however, there is some concern that aggressive fluid resuscitation can worsen underlying pulmonary contusion. Some studies have shown that early resuscitation using hypertonic saline may be beneficial by decreasing neutrophil-induced lung injury,32 whereas other studies dispute this finding.33
It was previously thought that obligatory intubation and mechanical ventilation were necessary for improved ventilation; however, work by Trinkle30 showed that patients with flail chest who were managed without tracheostomy or mechanical ventilation recovered well. In fact, his work suggests that mechanical ventilation is not always necessary and can be associated with increased morbidity and mortality. The use of non-invasive ventilation has been advocated in flail chest to reduce the incidence of intubation and subsequent complications.34 On the other hand, unstable patients who require mechanical ventilation should be intubated promptly in the emergency department. This is particularly true for patients undergoing advanced imaging and transfer, and not immediately available to the physician, as patients with flail chest can decompensate quickly and may require emergent airway management. There also is a role for surgical fixation of the flail segment. This procedure typically is employed if thoracotomy is performed for other injuries or if the patient has required prolonged ventilation without improvement. A prospective study by Tanaka35 showed that patients who had surgical stabilization of their flail chest segment required fewer ventilator days, shorter ICU stays, lower incidence of pneumonia, and were more likely to return to full-time work compared to patients who were medically managed only.
Blunt cardiac injury (BCI) is seen in 17-75% of patients with blunt trauma, depending on the mechanism.36 BCI results from direct force applied to the heart, and the presenting symptoms range from no findings to fatality. Most blunt cardiac injuries result from motor vehicle accidents, but they also can be seen with blast injuries or falls. As with all aspects of trauma evaluation, the physician must maintain a high index of suspicion regarding potential blunt cardiac injuries based on the mechanism of injury. Diagnosis of cardiac injury is established by electrocardiogram (ECG), myocardial biomarkers. and echocardiography. An ECG should be performed on any patient suspected of having BCI. The most common abnormality seen in BCI is sinus tachycardia, followed by premature atrial or ventricular contractions. Studies have shown that 85% of patients who developed complications from BCI had an abnormal ECG on hospital admission.37 Cardiac biomarkers also have been advocated in the evaluation of patients with suspected BCI. CK-MB has been used to identify patients with BCI, but elevations in CK-MB have not predicted which patients ended up having complications from the BCI. Troponin T measurement has been shown to have greater diagnostic value than CK values.38 Echocardiography also can be used to evaluate for any suspected abnormalities in cardiac wall motion, pericardial effusion, or valvular disruption in the evaluation of patients with BCI. The routine use of echocardiography in suspected blunt trauma patients is not recommended as there are no data that show echocardiography can be used to predict complications from BCI.39 Most patients with BCI develop complications (dysrhythmias) early; however, patients often are monitored for the first 24 hours after the injury.
Any patient who requires intubation, non-invasive ventilation such as BiPAP or CPAP, needle or tube thoracostomy, or who has the potential for deterioration should be transported to a trauma center for further evaluation. In particular, minor chest trauma in the geriatric patient can result in devastating outcomes40; hospitalization of such patients should be considered.
Abdominal and Pelvic Trauma
Most blunt abdominal trauma is the result of MVCs; the remainder are from assaults and falls. The management of abdominal trauma patients relies heavily on frequent reassessment of the abdomen, as nearly half of patients with no obvious findings on initial physical examination are found to have positive findings during laparotomy. If the patient is hypotensive, rapid assessment and simultaneous resuscitation are required. Focused abdominal sonography in trauma (FAST) or diagnostic peritoneal lavage (DPL) can be used to detect the presence of hemoperitoneum and the need for laparotomy in hypotensive patients. If the patient is hemodynamically stable, the treating physician can afford the time to order a CT scan of the abdomen/pelvis to gather more detailed information about the injuries. Depending on the skills of the assessing physician and capabilities of the sending facility, early transfer should be considered if abdominal trauma is suspected.
FAST is a relatively easy technique to master and has become part of routine emergency medicine practice. FAST has been shown to have a sensitivity of 85% for detection of any intraabdominal injury; when considering only injuries that require surgical intervention, the sensitivity increases to 97%.41 FAST has replaced the DPL in most institutions as an initial tool to evaluate for hemoperitoneum. It should be emphasized that the FAST scan can identify only intraperitoneal fluid and provides no information about retroperitoneal injuries. The FAST examination also is limited in its ability to provide information about diaphragmatic, pancreatic, and vascular injuries. In most trauma centers, hemodynamically unstable patients with a positive FAST scan are taken directly for laparotomy.
CT scan of the abdomen/pelvis can be utilized to evaluate solid organ injury, identify intraabdominal fluid, and provide information about retroperitoneal injury, an anatomic area not well evaluated by FAST or DPL. CT scan has accuracy rates of 92-98%, with pancreatic and hollow viscus injuries being the most often missed injuries.42 CT scanning should be considered only in hemodynamically stable patients evaluated in non-trauma hospitals and should not delay transfer where indicated for other injuries.
The spleen is the most commonly injured intra-abdominal organ. Patients with left lower rib fractures should be considered high risk for splenic injury, as should any blunt abdominal trauma patient with abdominal pain and/or signs of hypovolemia. Splenic injuries can be identified with a positive FAST scan indicating blood in the peritoneal cavity. (See Figure 7.) In a stable patient, CT scanning allows for definitive identification and grading of the splenic injury. Definitive treatment of splenic injury depends on the degree of the laceration or injury, and can range from close observation to embolization of the splenic artery or splenectomy.
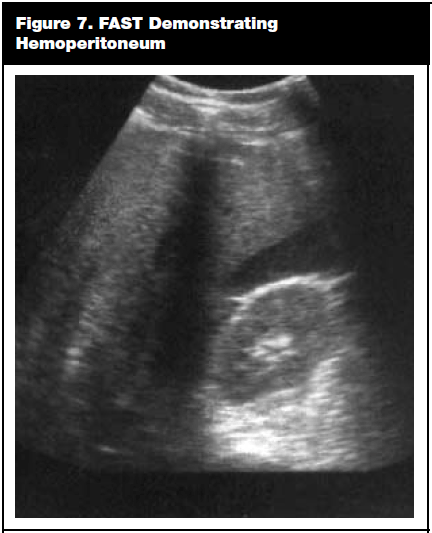 |
Liver injury is evaluated and treated in a manner similar to splenic injuries. Most hemodynamically stable patients with liver injuries can be managed non-operatively with observation. Hemodynamically unstable patients can be managed operatively or angiographically with embolization. The FAST examination can be used to detect free intraperitoneal fluid associated with liver injury. In experienced hands, ultrasound can be used to evaluate the liver parenchyma for defects indicative of subcapsular or contained parenchymal injury.
There also can be injury to the hollow organs within the abdominal cavity, which are inherently more difficult to detect with physical examination and ultrasound. Even with CT scan, injuries to the bowel and pancreas can be missed. Perforation of the bowel may not be evident clinically early in the patient's course until peritoneal signs have developed. Some have suggested that DPL be used in these cases to evaluate for bowel injury, but this strategy may not be indicated in all settings. As always, one must have a high index of suspicion for serious injury based on the mechanism and consider transfer for further evaluation and observation if initial imaging studies do not provide definitive information regarding possible injuries.
Pelvic fractures result in the death of 10-15% of patients, and they can result in life-threatening hemorrhage. Approximately 9% of blunt abdominal trauma patients suffer from pelvic fractures. MVCs are the most common cause of pelvic fractures. All patients with suspected pelvic trauma should undergo the usual ATLS evaluation on presentation to the ED. Hemodynamically unstable patients should be evaluated fully for the source of their bleeding, which includes evaluation for chest and abdominal injuries. When pelvic fractures are identified, it is critical to stabilize the pelvis to prevent further hemorrhage. In the prehospital setting and during transfer, this can be accomplished with military anti-shock trousers (MAST), commercial pelvic binders, or a simple cravat tied around the pelvis. Stabilization of the pelvis should tamponade bleeding and shunt blood from the pelvis and lower extremity into the chest to perfuse the vital organs.
With pelvic fractures, the treating physician must remember that the patient may have associated injuries to the rectum, vagina, bladder, or urethra. This is particularly true with displaced fractures involving the pubic rami and sacroiliac joints. Digital rectal examination should be performed in males to evaluate for a high-riding prostate, which is a sign of injury to the urethra. These patients should be examined for scrotal hematoma. The external genitalia in all patients should be evaluated for any evidence of bleeding and, if present, the patient should undergo evaluation of the urethra prior to insertion of a Foley catheter via retrograde urethrogram or cystogram. Recent literature reports a mortality rate of 70% with open pelvic fractures.43
Plain films of the pelvis have only 80% sensitivity at diagnosing pelvic fractures.44 CT scan of the pelvis is the preferred imaging modality to identify all injuries to the bones and pelvic organs. Definitive management recently has focused on angiographic control of hemorrhage by interventional radiology and external fixation by orthopedics. Hemorrhage associated with pelvic fractures may be either venous or arterial in origin; achieving control of this hemorrhage is vital to properly managing these injuries. There has been some controversy regarding whether embolization or external fixation should be used initially to manage pelvic hemorrhage. Miller45 evaluated the role of embolization and fixation in the management of hemorrhage associated with pelvic trauma. According to the authors, the most important question to answer is whether the bleeding is thought to be arterial in origin. Hypotensive patients who do not respond to initial resuscitation should be presumed to have an arterial source. If the bleeding is arterial, then rapid angiography and embolization is recommended; if not, then external fixation may be employed. Stable patients who undergo CT scan of the abdomen and pelvis who have a contrast blush associated with their pelvic fracture also should be presumed to have arterial bleeding and be managed with angiography.45 External fixation has a significant role in hemorrhage control as reducing the pelvic volume tamponades furthers bleeding. External fixation for open-book (anterior compression) pelvic fracture may not be sufficient to control associated bleeding.46 In such injuries, there is a definite role for both angiography with embolization along with external fixation in the management of these severe injuries. As with all previous examples, early consultation with the orthopedic surgeon in patients with significant pelvic trauma is critical to assuring appropriate definitive treatment.
The use of seat belts has increased due to public awareness and laws mandating their use. While seat belt use reduces serious injuries and lowers mortality, injuries also may be sustained from the seat belt itself. There is a constellation of injuries related to the use to seat belts, called the "seatbelt syndrome." These injuries are the result of compression or hyperflexion that occurs during the time of impact that can lead to the classic triad of abdominal wall ecchymosis (the so-called "seat belt sign"), abdominal injuries, and spinal fractures. Despite the fact that severe injury results from the seat belt itself, the data indicate that the protective benefits of seat belt use far outweigh any potential risks.
Stabilization
The principles of ATLS can be utilized to stabilize patients with major blunt trauma for transfer. The first priority remains the airway, which should be secured in patients with major head and thoracic trauma along with patients with abdominal or pelvic trauma and hemodynamic instability. Full spinal immobilization should be maintained during the transfer. Although there are protocols for spinal clearance in the field and ED, there is evidence that patients who require transfer to a trauma center may be at risk for occult spinal injuries.47
Intravenous access should be obtained with the goal of restoring intravascular volume. Two large-bore intravenous lines should be established prior to transport. Intraosseous access has emerged as an alternative method of intravascular access in critically ill patients.48 Early resuscitation should include crystalloid infusion with blood products being considered once 2-4 L of crystalloid (either normal saline or lactated Ringers) have been administered. If the patient does not respond with improvements in tachycardia or hypotension and the etiology is known or presumed to be related to hemorrhage, the physician should consider administration of blood products. The endpoints of fluid resuscitation remain controversial.49,50 In some centers serum lactate is used to monitor the fluid resuscitation status of the patient.
Appropriate fluid resuscitation in trauma is controversial and is beyond the scope of this review. The prevailing practice of aggressive fluid resuscitation was derived from early experiments using a fixed-volume model of hemorrhagic shock that showed prompt restoration of circulating volume improved mortality.51 Later experiments using a continuous hemorrhage model disputed these findings and demonstrated that animals that were deliberately "under-resuscitated" actually had better survival.52 This led to the landmark study by Bickel53 in which victims of trauma were randomized to receive either aggressive prehospital and ED fluid resuscitation or minimal fluids prior to operative repair. Those patients in whom fluids were withheld until the patient was taken to the operating room had a better survival rate (70% versus 62% [p = 0.04]). It should be noted that this study was conducted in a setting with primarily penetrating trauma and rapid transport in an urban environment. This has led to the concept of "permissive hypotension" in which prehospital and ED fluid resuscitation is minimized until the patient undergoes surgical control of hemorrhage. This concept derives from the fact that patients with significant hemorrhage often are coagulopathic, acidotic, and hypothermic during and after resuscitation, all of which are worsened with excessive fluid administration. It is felt that vessel spasm, hypotension, and the patient's natural coagulation cascade will temporize bleeding; excessive fluid administration causes dilution of the natural coagulation factors and decreases blood viscosity and reversal of vessel spasm. The military surgical literature cites examples in which wounded soldiers arrived at medical facilities without active hemorrhage from amputated limbs. When aggressive fluid resuscitation was initiated, significant hemorrhage emerged that was not controlled with tourniquet application. Surgeons from the first and second World Wars also have described this phenomenon previously.54 Current military practice is to minimize prehospital fluid and blood product administration in combat scenarios, as long as the patient has normal mental status and a palpable radial pulse.
Application of permissive hypotension has not been well studied in blunt trauma, especially in settings with long transport times such as rural environments. However, there is an emerging sentiment that one should appropriately resuscitate unstable, hypotensive patients, but use caution as over-resuscitation actually may diminish the ability to gain control of hemorrhage until the patient is definitively managed by the trauma surgeon.
Tube thoracostomy should be performed prior to transfer, but other procedures and diagnostic studies should not delay transfer to a trauma center when appropriate. Tube thoracostomy should be performed by physicians trained in the technique with evidence of continued skill competence, as a complication rate of up to 30% is reported with this procedure.55,56 Direct consultation with the accepting trauma surgeon or ED physician should include information about the exact mechanism of injury, diagnostic findings, therapeutic interventions, and discussion about the appropriate mode of transfer (air versus ground). All available medical records, radiographs, laboratory studies, and prehospital reports should be transported with the patient.
Summary
Patients with blunt trauma frequently are encountered in the emergency department. The potential for early mortality and morbidity should be considered in all blunt trauma victims with a significant mechanism of injury. An organized assessment for life-threatening head, chest, abdominal, and pelvic injuries should be conducted with a recognition that definitive care may be provided after transfer to a certified trauma center. Proper stabilization measures including airway control, fluid resuscitation, and appropriate diagnostic studies should be performed prior to transfer. Airway control, assuring proper oxygenation and maintaining cerebral perfusion, are the hallmarks of early management of head injury. Attention to proper oxygenation and ventilation of patients with early tube thoracostomy is definitive in most patients with thoracic injury. Finally, pelvic stabilization and intravenous fluids are key management principles in abdominal and pelvic trauma.
References
1. Newgard CD, McConnell KJ, Hedges JR, et al. The benefit of higher level of care transfer of injured patients from nontertiary hospital emergency departments. J Trauma 2007; 63(5):965-971.
2. McConnell KJ, Newgard CD, Mullins RJ, et al. Mortality benefit of transfer to level I versus level II trauma centers for head-injured patients. Health Serv Res 2005;40:435-457.
3. Demetriades D, Martin M, Salim A, et al. The effect of trauma center designation and trauma volume on outcome in specific severe injuries. Ann Surg 2005;242:512-517.
4. US Department of Labor – Bureau of Labor Statistics. Injuries, illnesses, and fatalities. US Department of Labor web site http://www.bls.gov/iif/). USDL 08-1182. August 20, 2008.
5. Blumen IJ. A safety review and risk assessment in air medical transport. In: Air Medical Physician Handbook (2002). Association of Air Medical Physicians, Salt Lake City, 2002.
6. Johnston BD, Rivara FP, Droesch RM, et al. Behavior change counseling in the emergency department to reduce injury risk: A randomized, controlled trial. Pediatrics 2002;110:267-274.
7. Sommers MS, Dyehouse JM, Howe SR, et al. Effectiveness of brief interventions after alcohol-related vehicular injury: A randomized controlled trial. J Trauma 2006;61:523-531.
8. Broyle MJ. Is mechanism of injury alone in the prehospital setting a predictor of major trauma a review of the literature. J Trauma Manag Outcomes 2007;1:4.
9. Palanca S, Taylor DM, Bailey M, et al. Mechanisms of motor vehicle accidents that predict major injury. Emerg Med 2003;15:423-428.
10. Boyle MJ, Smith EC, Archer F. Is mechanism of injury alone a useful predictor of major trauma? Injury 2008;39:986-992.
11. Mackay M. Mechanisms of injury and biomechanics: Vehicle design and crash performance. World J Surg 1992;16:420-427.
12. Gikas PW. Mechanisms of injury in automobile crashes. Clin Neurosurg 1972;19:175-190.
13. Grossman MD. Patterns of blunt injury. In: Peitzman AB, Rhodes M, Schwab CW, et al, eds. The Trauma Manual, 2nd edition. Philadelphia: Lippincott Williams & Wilkins; 2002:4-9.
14. Blincoe L, Seay A, Zaloshnja E, et al. (2002). The economic impact of motor vehicle crashes, 2002. Report no. DOT HS 809 446. Washington, DC: National Highway Traffic Safety Administration.
15. Durbin DR, Arbogast KB, Moll EK. Seat belt syndrome in children: A case report and review of the literature. Pediatr Emerg Care 2001;17:474-477.
16. Gongora E, Acosta JA, Wang DS, et al. Analysis of motor vehicle ejection victims admitted to a Level I trauma center. J Trauma 2001;51:854-859.
17. Covey D. Blast and fragment injuries of the musculoskeletal system. J Bone Joint Surg Am 2002;84:1221-1234.
18. Korfias S, Stranjalis G, Boviatsis E, et al. Serum S-100B protein monitoring in patients with severe traumatic brain injury. Intensive Care Med 2007;33:355-360.
19. Letarte P. The brain. In: Feliciano DV, Mattox KL, Moore EE, eds. Trauma, 6th edition. New York: McGraw-Hill, Inc.;2008:397-418.
20. Peitzman AB, Rhodes M, Schwab CW, et al. The Trauma Manual, 2nd ed. Philadelphia: Lippincott Williams and Wilkins; 2002.:133.
21. Warner KJ, Sharar SR, Copass MK, et al. The impact of prehospital ventilation on outcome after severe traumatic brain injury. J Trauma 2007;62: 1330-1336.
22. Davis DP, Idris AH, Sise MJ, et al. Early ventilation and outcome in patients with moderate to severe traumatic brain injury. Crit Care Med 2006;34: 1202-1208.
23. Brain Trauma Foundation; American Association of Neurological Surgeons; Congress of Neurological Surgeons; Joint Section on Neurotrauma and Critical Care, AANS/CNS, Bratton SL, Chestnut RM, Ghajar J, et al. Guidelines for the management of severe traumatic brain injury. XIII. Antiseizure prophylaxis. J Neurotrauma 2007;24 Suppl 1:S83-S86.
24. Roberts I, Yates D, Sandercock P, et al; CRASH trial collaborators. Effect of intravenous corticosteroids on death within 14 days in 10,008 adults with clinically significant head injury (MRC CRASH trial): Randomized placebo controlled trial. Lancet 2004;364:1321-1328.
25. Brink M, Deunk J, Dekker HM, et al. Added value of routine chest MDCT after blunt trauma: Evaluation of additional findings and impact on patient management. AJR Am J Roentgenol 2008;190:1591-1598.
26. Trupka A, Waydhas C, Hallfeldt KK, et al. Value of thoracic computed tomography in the first assessment of severely injured patients with blunt chest trauma: Results of a prospective study. J Trauma 1997;43:405-411.
27. Soldati G, Testa A, Silva FR, et al. Chest ultrasonography in lung contusion. Chest 2006;130:533-538.
28. Blaivas M, Lyon M, Duggal S. A prospective comparison of supine chest radiography and bedside ultrasound for the diagnosis of traumatic pneumothorax. Acad Emerg Med 2005;12:844-849.
29. Sisley AC, Rozycki GS, Ballard RB, et al. Rapid detection of traumatic effusion using surgeon-performed ultrasonography. J Trauma 1998;44:291-296.
30. Trinkle JK, Richardson JD, Franz JL, et al. Management of flail chest without mechanical ventilation. Ann Thoracic Surg 1975;19:355-365.
31. Trupka A, Waydhas C, Hallfeldt KK, et al. Value of thoracic computed tomography in the first assessment of severely injured patients with blunt chest trauma: Results of a prospective study. J Trauma 1997;43:405-411.
32. Hashiguchi N, Lum I, Romeril E, et al. Hypertonic saline resuscitation efficacy may require early treatment in severely injured patients. J Trauma 2007;62:299-306.
33. Cohn SM, Fisher BT, Rosenfield AT, et al. Resuscitation of pulmonary contusion: Hypertonic saline is not beneficial. Shock 1997;8:292-299.
34. Vidhani K, Kause J, Parr M. Should we follow ATLS guidelines for the management of traumatic pulmonary contusion: The role of non-invasive ventilatory support. Resuscitation 2002;52:265-268.
35. Tanaka H, Yukioka T, Yamaguti Y, et al. Surgical stabilization of internal pneumatic stabilization? A prospective randomized study of management of severe flail chest patients. J Trauma 2002;52:727-732.
36. Ellie MD. Blunt cardiac injury. Mt. Sinai J Med 2006;73:542-552.
37. Foil MB, Mackersie RC, Furst SR, et al. The asymptomatic patient with suspected myocardial contusion. Am J Surg 1990;160:638-642.
38. Ferjani M, Droc G, Dreux S, et al. Circulating cardiac troponin T in myocardial contusion. Chest 1997;111:427-433.
39. Karalis DG, Victor MF, Davis GA, et al. The role of echocardiography in blunt chest trauma: A transthoracic and transesophageal echocardiographic study. J Trauma 1994;36:53-58.
40. Bulger EM, Arneson MA, Mock CN, et al. Rib fractures in the elderly. J Trauma 2000;48:1040-1046.
41. Farahmand N, Sirlin CB, Brown MA, et al. Hypotensive patients with blunt abdominal trauma: Performance of screening US. Radiology 2005;235: 436-443.
42. Miller PR, Croce MA, Bee TK, et al. Associated injuries in blunt solid organ trauma: Implications for missed injury in nonoperative management. J Trauma 2002;53:238-242.
43. Rice PL, Rudolph M. Pelvic fractures. Emerg Med Clin North Am 2007;25: 795-802.
44. Duane TM, Dechert T, Wolfe LG, et al. Clinical examination is superior to plain films to diagnose pelvic fractures compared to CT. Am Surg 2008;74:476-479.
45. Miller PR, Moore PS, Mansell E, et al. External fixation or arteriogram in bleeding pelvic fracture: Initial therapy guided by markers of arterial hemorrhage. J Trauma 2003;54:437-443.
46. Grimm MR, Vrahas MS, Thomas KA. Pressure-volume characteristics of the intact and disrupted pelvic retroperitoneum. J Trauma 1998;44:454-459.
47. Werman HA, White LJ, Herron H, et al. Clinical clearance of spinal immobilization in the air medical environment: A feasibility study. J Trauma 2008;64:1539-1542.
48. Cooper BR, Mahoney PF, Hodgetts TJ, et al. Intra-osseous access (EZ-IO) for resuscitation: UK military combat experience. J R Army Med Corps 2007;153:314-316.
49. Kwan I, Bunn F, Roberts I; WHO Pre-Hospital Trauma Care Steering Committee. Timing and volume of fluid administration for patients with bleeding. Cochrane Database Syst Rev 2003;(3):CD002245.
50. Wade CE, Holcomb JB. Endpoints in clinical trials of fluid resuscitation of patients with traumatic injuries. Transfusion 2005;45(1 Suppl):4S-8S.
51. Shires GT, Coln D, Carrico J, et al. Fluid therapy in hemorrhagic shock. Arch Surg 1964;88:688-693.
52. Mapstone J, Roberts I, Evans P. Fluid resuscitation strategies: A systematic review of animal trials. J Trauma 2003;55:571-589.
53. Bickel WH, Wall MJ, Pepe PE, et al. Immediate versus delayed fluid resuscitation for hypotensive patients with penetrating torso injuries. N Engl J Med 1994;331:1105-1109.
54. Cannon WB, Faser J, Collew EM. The preventive treatment of wound shock. JAMA 1918;47:618.
55. Bailey RC. Complications of tube thoracostomy in trauma. J Accid Emerg Med 2000;17:111-114.
56. Aylwin CJ, Brohi K, Davies GD, et al. Pre-hospital and in-hospital thoracostomy: Indications and complications. Ann R Coll Surg Engl 2008;90:54-57.
Victims of blunt trauma are frequently encountered in the emergency department (ED). Forty percent of all ED visits each year are attributed to injury, which equates to about 40 million ED visits annually. Additionally, approximately one-third of intensive care unit (ICU) admissions in the United States are trauma-related.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
