Drug Criteria & Outcomes: Aprepitant (Emend) Formulary Evaluation
Drug Criteria & Outcomes: Aprepitant (Emend) Formulary Evaluation
Part 2: Clinical Trials Summary, Recommendations, and Criteria for Use
By Omar Dudar, PharmD
Candidate
Harrison School of Pharmacy
Auburn (AL) University
Written while on clinical
rotation at Huntsville (AL) Hospital
Clinical trial summary
Phase IIa studies
Four Phase IIa studies have been conducted with aprepitant to establish
a time period for therapy, dose, and a regimen of combination therapy with aprepitant
for the Phase IIb trial.
Cocquyt, et al conducted the first randomized, double-blind, clinical trial to evaluate the efficacy of the NK1 antagonist (aprepitant) (see Table 4). The purpose of the trial was to evaluate the prevention of emesis during the acute and delayed phases of chemotherapy with cisplatin-naïve patients. In this earlier trial the pro-drug of aprepitant "L-758,298" was given intravenously to 30 patients at doses of 60 mg or 100 mg daily for seven days. The standard group of 23 patients received only ondansetron 32 mg intravenously for the course of seven days and was compared with the L-758,298 group (designated aprepitant IV group). Both treatment groups were evaluated for efficacy of "no emesis" through the seven days of therapy. Nausea was assessed as a secondary endpoint with the use of a four-point scale.
Summary: During the delayed phase, the aprepitant IV group’s prevention of emesis was significantly improved compared to the ondansetron group, P = 0.005.
Other results
- Average nausea score distribution for the delayed phase was lower in the aprepitant IV group compared to the ondansetron group (P = 0.15).
- There were no serious side effects in the aprepitant IV group.
Authors’ conclusions: Single daily doses of IV aprepitant were sufficient to suppress delayed nausea and vomiting associated with cisplatin treatment and also to decrease the rate of vomiting during the acute phase. The data collected seem to concur with the assumption that there is a different mechanism for the acute vs. delayed phases of vomiting.
Campos, et al organized a randomized, multicenter, double-blind, parallel-group, controlled clinical trial to evaluate the prevention of acute and delayed emesis after cisplatin 70 mg/m2 or more for each of the groups listed in Table 5. Note: MK-869 was the former name given to the first oral dosage form of aprepitant in Table 5, below.
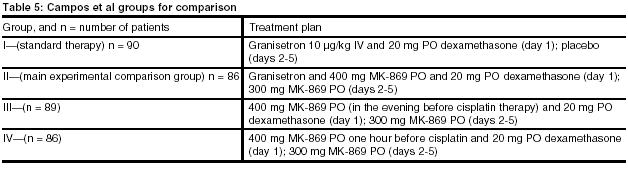
Rescue therapy: Metoclopramide 20-30 mg PO four times a day (qid) or 1-2 mg/kg metoclopramide IV qid for the first day, and dexamethasone 8 mg PO twice a day (bid) (days 2-5) as needed (PRN) vomiting.
Inclusion criteria:
- 16 years or older.
- Negative assay for serum B human chorionic-gonadotropin (for safety of the fetus).
Exclusion criteria:
- Karnofsky scale score of less than 60; allergy to metoclopramide, dexamethasone, or granisetron; emesis before cisplatin (24 hours before day 1); seizure treatment in the past two months, severe illness other than neoplasm; or lab criteria for hemoglobin, WBC, platelets, AST, ALT, alkaline phosphatase, bilirubin, and Scr.
Summary of results
Acute phase (no emesis)
- Group II vs. Group I (80% vs. 57%), P < 0.01, 90% confidence interval (8-38%).
- Group III (46%), no P value given.
- Group IV (43%), no P value given.
Acute phase (no emesis and no rescue therapy)
- Group II vs. Group I (75% vs. 51%), P < 0.01.
- Group III (44%), no stated P value.
- Group IV (41%), no stated P value.
Delayed Phase (no emesis)
- Group I (29%).
- Group II (63%).
- Group III (51%).
- Group IV (57%).
(P < 0.01 for groups II, III, and IV vs. group I)
Summary of results
- Quality of life (QOL)
measure of nausea reviewed from nausea diary as secondary endpoint.
- Diary with visual
analog scale (0-100 mm).
— No nausea = 0; 100 = nausea as bad as it can get.
- Diary with visual
analog scale (0-100 mm).
- Distribution of scores in delayed period was lower in comparison with Group II vs. Group I (P < 0.05 for days 1-5 and days 2-5) (see Table 6).
- During the first 24 hours, the difference between Group II vs. Group I was statistically significant for fewer nausea episodes, P < 0.05.
- Delayed phase: Group
II was associated with a lower frequency of nausea than Group I, P < 0.05.
- The difference between Group III vs. Group I was statistically significant, P < 0.05.
- Delayed (day 2): Frequency of nausea for Groups II, III, and IV was lower than Group I. (The difference of Group IV vs. Group I was statistically significant, P < 0.05; other P values were not stated.)
Global satisfaction (for treatment with cisplatin documented on day 2 and day 6)
- QOL measure of satisfaction
of anti-emetic treatment on a visual analog scale (0-100 mm).
- 0 = not at all satisfied; 100 = completely satisfied.
- Results for groups I, II, III, and IV on day 2 were 94, 98, 96, and 96, and day 6 were 92, 95, 96, and 98, respectively.
- There was no statistically significant difference in scores on the scale from 0-100 mm for satisfaction.
Authors’ conclusions
- Experimental therapy with aprepitant (Group II) displayed decreased incidence of acute and delayed phase emesis as compared to Group I.
- In the acute period, the proportion of patients without emesis was increased by 23% in Group II compared to Group I.
- During the acute phase of emesis, Group II results showed no further benefit vs. Groups III and IV.
- Treatment for Group II showed benefit in the delayed phase to decrease emesis rates.
- Once-daily dosing of aprepitant and standard therapy is sufficient to reduce emesis significantly vs. the standard therapy (Group I).
Strengths and weaknesses: Strengths and weaknesses of the Campos et al trial are summarized in Table 7, below.

The two other Phase IIa trials assessed aprepitant in combination with dexamethasone (Van Belle et al) and dexamethasone together with a 5-HT3-RA (Navari et al). These two trials had similar endpoints and design as the Campos trial and are not addressed in detail.
Summary of important findings of the trials
- During acute and delayed phases, aprepitant as monotherapy or coadministered with corticosteroids shows less efficacy in CINV compared to 5-HT3-RA.
- Once-a-day dosing of aprepitant demonstrated efficacy in CINV.
- Activity of aprepitant in the delayed phase compared to 5-HT3-RA in the acute phase demonstrates distinct mechanisms of emesis during these times.
- Triple therapy of aprepitant, dexamethasone, and 5-HT3-RA on day 1 and aprepitant on subsequent days is more efficacious than the standard therapy without aprepitant (dexamethasone and 5-HT3-RA day 1, and dexamethasone on subsequent days).
Other trials
Poli-Bigelli et al organized a multicenter, randomized, double-blind, placebo-controlled trial to compare aprepitant in combination therapy with ondansetron and dexamethasone to the standard therapy. In this study, 283 patients were randomized to the aprepitant group, leaving 286 for standard therapy treatment (see Table 8, below).

Patients reported the frequency of their CINV in a diary over the five-day period for assessment of outcomes: date and time for emesis, and retching. Nausea was evaluated with a visual analogue scale (VAS), and a Functional Living Index-Emesis (FLIE) questionnaire was completed on day 6 for final evaluation. The study ended between day 19 and 29 when the patients returned.
Result summary for emetic prevention: The results for complete response, which focused on the four-day duration, was characterized by no emesis and no use of rescue therapy for the overall study time. During the acute phase, the difference between Group 1 and Group 2 was statistically significant for preventing vomiting. During the delayed phase, Group 1 results also showed a statistically significant improvement from Group 2 (see Table 9, below).
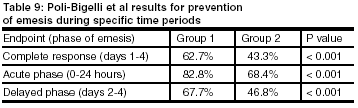
QOL measure: The FLIE questionnaire showed more patients in the aprepitant group (74.7%) compared with 63.5% in the standard therapy group having minimal or no impact of CINV on their daily lives.
Safety: The most common adverse drug reactions of the aprepitant group compared to standard group were neutropenia (1.8% vs. 2.1%), dehydration (1.6% vs. 0.7%), septic shock (1.1% vs. 0.7%), dyspnea (1.1% vs. 0.7%), and respiratory insufficiency (1.8% vs. 0.45).
Authors’ conclusions: Poli-Bigelli et al concluded that aprepitant can help in the delayed phase of nausea and vomiting associated with highly emetogenic chemotherapy, such as cisplatin > 70 mg/m2. A more frequent use of rescue medication was observed in the standard group vs. the aprepitant group. Overall side effects were similar in occurrence between the two groups; however, the aprepitant group exhibited a greater incidence of neutropenia, dehydration, septic shock, dyspnea, and respiratory insufficiency.
Strengths and weaknesses: Strengths and weaknesses of the Poli-Bigelli et al trial are summarized in Table 10, below.

Chawla et al conducted a multicenter, randomized, double-blind, placebo-controlled study of cisplatin-naïve cancer patients to find an appropriate dose of aprepitant to use with standard CINV prevention therapy. The study group included patients 18 years or older with solid tumors and a Karnofsky score of 60 or more, who would be started on a cisplatin regimen > 70 mg/m2. Patients were excluded if they had other treatment with nonapproved drugs within four weeks of study, or abnormal lab values: WBC < 3,000/mm3, ANF < 1,500/mm3, platelet count < 100,000/mm3, LFTs > 2.5 times upper limit, bilirubin > 1.5 times upper limit, or creatinine > 1.5 times upper limit. Patients with CNS malignancy or those who were actively infected were excluded for safety reasons. The primary endpoint was complete response: no emesis and no rescue therapy. Patients were randomized and stratified by gender to either of three groups, with those in Group 1 receiving placebo instead of aprepitant (see Table 11).
QOL assessment: Patients kept a diary and assessed their nausea with a visual analog scale for a secondary endpoint.
Comments: For each endpoint examined, prevention of emesis in Group 3 was statistically significant compared to Group 1 (see Table 12). Group 21 was statistically significant vs. standard therapy (Group 1) for the delayed and complete response, when considering reported P values alone.
Safety: Tolerability was assessed for 214 patients receiving aprepitant 125/80 mg dose, 120 patients taking the aprepitant 40/25 mg doses, and 212 patients on standard therapy. The aprepitant 125/80 mg Group 3 had the highest rate of adverse events with a 1.06 relative risk (P = 0.448), and the relative risk of discontinuation due to adverse events was 1.32 (P = 0.804). Incidence of serious clinical adverse events was higher in the aprepitant groups than in the standard therapy: 21.5 % in the aprepitant 125/80 mg (Group 3), 16.7% in the 40/25 mg group (Group 21), and 12.3% in the standard therapy (Group 1). Adverse events common in 10% or more of the patients were fatigue, constipation, diarrhea, nausea, neutropenia, anorexia, headache, and hiccups. Out of 580 patients on study therapy, 131 (22.6%) had one or more laboratory adverse event (specific laboratory values and tests were not stated in the study). Standard therapy had the highest incidence at 8.5%. There was an increased rate of infections, including febrile neutropenia in the aprepitant group at 13% compared to the standard therapy group at 4.2%.
Authors’ conclusions: Authors concluded aprepitant at either dose level (40/25 mg or 125/80 mg) reduced the rate of emesis at the overall, acute, and delayed phases more frequently than standard therapy. Follow-up on the aprepitant 375/250 mg group showed efficacy similar to the 125/80 mg group, and there were no differences in the occurrence of clinically significant drug interactions between groups. The 125/80 mg aprepitant group displayed improved results for endpoints vs. the 40/25 mg aprepitant group. Tolerability from pooled data indicates the adverse effects reported were similar to population standards for those with cancer. Aprepitant’s effect on increasing dexamethasone levels may be linked to greater efficacy and increased incidence of febrile neutropenia.
Strengths and weaknesses: Strengths and weaknesses of the Chawla et al trial are summarized in Table 13, below.

Two other Phase III multicenter, randomized, parallel, double-blind, controlled trials (Warr D, et al) evaluated 1,105 patients randomized to aprepitant therapy or standard therapy, and involved six cycles of chemotherapy (see Table 14, below).
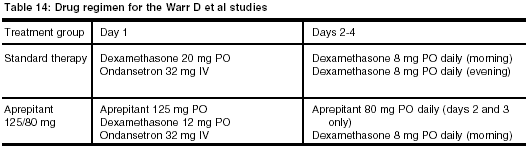
Endpoints of the study were evaluated at the acute phase (0-24 hours), delayed phase (25-120 hours) after cisplatin therapy, and overall (0-120 hours) post-cisplatin therapy for complete response (no emetic episodes and no rescue therapy.) (See Table 15.)
Secondary endpoints: Other endpoints were based on the visual analog scale (0-100 mm) ratings for: complete protection (VAS score < 25 mm), no emetic episodes, no rescue therapy; no emesis (no emetic episodes even with rescue therapy); no nausea, VAS score < 5 mm maximum; and no significant nausea (VAS < 25 mm maximum).
QOL endpoints: Quality of life was assessed using the FLIE questionnaire. The primary endpoint on this scale was from 0 to 126 with > 108 defined as minimal or no impact of nausea and vomiting on daily life. Patients had a diary in which to record nausea, vomiting, and use of rescue medication to be evaluated with the FLIE on day 6 of therapy. In this study, more than 108 on the FLIE was achieved by 74% vs. 64% of the aprepitant and standard therapy groups, respectively, P < 0.05. Therapy with cisplatin was repeated for six cycles and evaluated for the frequency of complete response, and achievement of this primary endpoint was found to be significantly higher (P < 0.006) in the group receiving aprepitant, with rates for aprepitant of 61% (n = 516) in cycle 1 and 59% (n = 89) by cycle 6 vs. rates for standard therapy of 46% (n = 522) in cycle 1 and 40% (n = 78) by cycle 6. Adverse events were mild to moderate in intensity for the standard therapy group and aprepitant groups, and were pooled from both Phase III studies.
Authors’ conclusions: The authors conclude that the primary endpoint for complete response was greater in the aprepitant group than the standard therapy for acute and delayed phases, with a P < 0.001. First emesis occurred at longer time after cisplatin administration in the aprepitant group than for standard therapy. These studies defined the dosing regimen, combination drugs, optimal time phase effects of aprepitant with combination drugs, and length of treatment. Some weaknesses of the study include reporting bias with the FLIE scale, and use of undefined emetogenic chemotherapy between groups. Strengths include large sample population, multi-regional study, double-blinding, stated alpha value and power, and ability to show difference in groups with a large study population.
Cost
A complete three-day dose of aprepitant costs approximately $240 using Tri-fold pack units, compared to ondansetron’s 32 mg IV one-time dose of approximately $200. A combination therapy with single-dose ondansetron 32 mg IV and three-day aprepitant regimen would cost approximately $440. A dexamethasone oral regimen for four days costs approximately $5-$10.
Recommendations
Nausea and vomiting are two of the most feared and devastating consequences of chemotherapy, considering the possibility of broken bones, torn esophagus, surgical sutures tears, and overall intolerance of chemotherapy. Patients undergoing highly emetogenic chemotherapy may experience emesis, despite standard antiemetic therapy in 50% of cases. Acute-phase prevention of emesis with standard therapy fails in 25% of patients, and for 50% during the delayed phase, indicating a clear need for CINV standard therapy improvement.
The benefit aprepitant offers is an increase in emesis prevention rate for the acute and delayed phases compared to standard therapy. Studies indicate a different mechanism of action esxists between the acute and delayed phases that could have a link to substance P-NK1 receptor antagonism. Clinical trials tested cisplatin-naïve patients at a highly emetogenic dose of 70mg/m2. Trial data also demonstrate that aprepitant improves secondary measures for nausea prevention and positive outcomes for the effect of CINV on quality of life. As other highly emetogenic drugs were allowed for clinical trial patients, a broad use of aprepitant for CINV might be explored.
Some of the limitations to aprepitant include the potential for drug interactions and adverse events. As a mild inhibitor of CYP3A4, aprepitant has a higher chance of drug interactions than standard therapy alone, so dose reduction of corticosteroids has been suggested as an example intervention. Adverse events were similar between aprepitant and standard therapy with exception of fatigue, constipation, diarrhea, neutropenia, headache, and hiccups. Despite propensity for these adverse events, patients in Phase III trials were able to tolerate aprepitant therapy, and criteria can restrict the use of aprepitant when benefit outweighs the risk.
Another concern is the added aprepitant cost to standard therapy, as three days of aprepitant therapy is approximately $240 compared to about $200 for ondansetron infusion. Even though aprepitant adjunct therapy is more expensive, criteria for judicious use of aprepitant from trial results will help to contain costs and allow benefit for those who meet the need for inclusion. The addition of aprepitant to the formulary for patients who meet the criteria listed below is therefore recommended.
List of highly emetogenic drugs
Severe
- Cisplatin (Platinol), carboplatin (Paraplatin), carmustine (BICNU) — < 250 mg/m2; dactinomycin (Cosmegen), daunorubicin (Cerubidine), doxorubicin (Adriamycin) — > 60 mg/m2, irinotecan (Camptosar), methotrexate (Folex PFS) — > 1 g/m2; procarbazine (Matulane) — PO doses, cyclophosphamide (Cytoxan), cytarabine (Cytosar), dacarbazine (DTIC-Dome), mechlorethamine (Mustargen), and streptozocin (Zanosar).
Criteria for use
- Start aprepitant even in patients who have not tolerated highly emetogenic therapy in the past.
- Do not use aprepitant for nonchemotherapy-related emesis, such as postoperative, rescue emetic therapy, or radiation-induced emesis.
- Use aprepitant in adults older than 18 years, due to trial exclusion of younger patients.
- Monitor for side effects when aprepitant is co-administered with drugs metabolized by CYP 450 enzymes.
- Do not use aprepitant PRN or as monotherapy.
- Use only in combination with a 5HT3-RA + a corticosteroid, e.g., dexamethasone.
Resources
- Aprepitant (Emend) for prevention of nausea and vomiting due to cancer chemotherapy. Med Lett Drugs Ther 2003;45:62-63.
- Campos D, Pereira JR, Reinhardt RR, et al. Prevention of cisplatin-induced emesis by the oral neurokini-1 antagonist, MK-869, in combination with granisetron and dexamethasone or with dexamethasone alone. J Clin Oncol 2001;19:1759-1767.
- Chawla SP, Grunberg SM, Gralla RJ, et al. Establishing the dose of the oral NK1 antagonist aprepitant for the prevention of chemotherapy-induced nausea and vomiting. Cancer 2003;97:2290-2300.
- Cocquyt V, Van Belle S, Reinhardt RR, et al. Comparison of L-758,298, a pro-drug for the selective neurokinin-1 antagonist, L-754,030, with ondansetron for the prevention of cisplatin-induced emesis. Eur J Cancer 2001;37:823-825.
- GlaxoSmithKline. Zofran Injection Product Information. Research Triangle Park, NC; 2001.
- Granisetron (drug evaluation in electronic form). MICROMEDEX Healthcare Series. Englewood, CO: MICROMEDEX; 2003.
- Hansten P, Horn J. Drug Interactions — Hansten & Horn’s Analysis & Management: A Clinical Perspective and Analysis of Current Development. St. Louis: Wolters Kluwer Health; 2003:347, 878.
- Merck & Co. Emend formulary information request. West Point, PA; 2003.
- Merck & Co. Emend product information. Whitehouse Station, NJ; 2003.
- Ondansetron (drug evaluation in electronic form). MICROMEDEX Healthcare Series. Englewood, CO: MICROMEDEX; 2003.
- Poli-Bigelli S, Rodrigues-Pereira J, Carides AD, et al. Addition of the neurokinin-1 receptor antagonist aprepitant to standard anti-emetic therapy improves control of chemotherapy-induced nausea and vomiting. Results from a randomized, double-blind, placebo-controlled trial in Latin America. Cancer 2003;97:3090-3098.
- Thomas T. Nausea and Vomiting. In: Dipiro JT, et al. Pharmacotherapy: A Pathophysiologic Approach. 5th ed. New York: McGraw-Hill; 2003:247-249.
- Warr D, Gralla RJ, Hesketh, PJ, et al. The oral NK1 antagonist aprepitant for the prevention of chemotherapy induced nausea and vomiting: Two randomized, double-blind, placebo controlled trials. Proc Am Soc Clin Oncol 2003;22:726 (abstr 2919).
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
