Strategies for the Pill: Providers share views
Strategies for the Pill: Providers share views
When it comes to oral contraceptives (OCs), what is your current practice when it comes to prescribing pills in extended- or continuous- regimens? More providers are prescribing pills in this manner, say respondents to the 2008 Contraceptive Technology Update Contraception Survey. About 62% say they increased use of such pill regimens in the last year.
Look for another dedicated extended regimen pill to enter the marketplace. In October 2008, the Food and Drug Administration approved Pomona, NY-based Barr Pharmaceuticals' New Drug Application for LoSeasonique, a new extended-cycle oral contraceptive. Under the pill's extended-cycle regimen, women take combination tablets containing 0.10 mg levonorgestrel and 0.02 mg of ethinyl estradiol daily for 84 consecutive days, followed by 0.01 mg ethinyl estradiol tablets for seven days. The company says the new pill will be shipped to trade customers and be available by prescription in the first quarter of 2009.1
One pill is packaged for continuous use: Lybrel from Wyeth Pharmaceuticals of Collegeville, PA. The drug, which won approval in 2007, is designed to be taken 365 days a year, without a placebo phase or pill-free interval. Other dedicated extended-regimen pills include Barr/Duramed's Seasonale and Seasonique, and Quasense, manufactured by Watson Pharmaceuticals of Corona, CA.
What has led to the uptick in use of such regimens? Ullainee Hartman, WHNP, a women's health nurse practitioner at Family Planning of the Big Horns, a reproductive health care clinic in Sheridan, WY, says it is a combination of her increased awareness of the benefits of such regimens, as well as patients' requests.
The menstrual benefits of suppressing periods include a reduction in dysmenorrhea, menorrhagia, premenstrual syndrome, and perimenopausal symptoms, such as hot flashes, night sweats, and irregular monthly periods. Nonmenstrual benefits include a reduction in menstrual migraines, endometriosis, and acne, as well as an improved sense of well-being.2
Women want the option of fewer periods. Results of a 2008 national survey show that while about three-quarters of women polled would prefer to get their period less often, just 8% report having tried extended or continuous contraceptive pills.3
When counseling a woman on when to start her first pill pack, what is your strategy for pill initiation? Most participants in the 2008 Contraception Survey say they are using the Quick Start method, which is immediate initiation of pills before start of the next menses. A total of 65% of responses indicated such use, an increase from 2007's 62% figure.
Quick Start is a user satisfaction issue, observes Mimi Zieman, MD, clinical associate professor in the Department of Gynecology and Obstetrics at Emory University School of Medicine in Atlanta. In speaking on the subject at the recent Contraceptive Technology Quest for Excellence Conference, Zieman noted that being able to provide multiple pill packs upon Quick Start initiation aids in helping women continue with the method.4
Many women might experience nausea when beginning use of a new OC. Which pill do survey participants prescribe for women who have experienced nausea on previous OCs?
While Alesse, a monophasic 20 mcg pill from Wyeth Pharmaceuticals, Collegeville, PA, continues to lead in this category, its 33% ranking declined from 2007's 56%. Other top choices included Loestrin (19.8%), a 20 mcg pill from Barr/Duramed, and Ortho Tri-Cyclen Lo (9.3%), a multiphasic 25 mcg pill from Ortho-McNeil Pharmaceutical, Raritan, NJ.
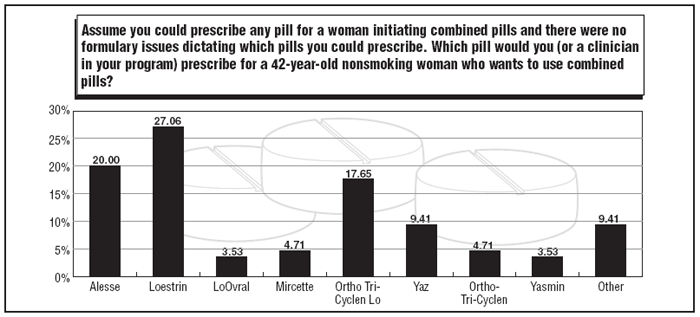
If a woman is in her 40s, healthy, and a nonsmoker, she is a potential candidate for combined oral contraceptives. When it comes to pill options for older women, 2008 survey participants named Loestrin (27%), followed by Alesse (20%). This standing reverses positions held by the two pills in the 2007 survey (19% and 44%, respectively). Ortho Tri-Cyclen Lo remained in the third spot (17.6%). (See the graphic, below, on top pills for older women.)
When it comes to women who smoke and the Pill, about 78% of survey participants say they will not write prescriptions for women ages 35-39 who smoke 10 cigarettes a day. For women ages 40 and older who smoke 10 cigarettes a day, about 92% say they will not prescribe OCs, which falls in line with similar figures in 2007.
When talking with patients about smoking, share these facts from the American Lung Association:
- In 2006, 20.2 million (17.8%) of women smoked in the United States. Annually, cigarette smoking kills an estimated 178,408 women in the United States.
- Women who smoke double their risk for developing coronary heart disease. Women who smoke also have an increased risk for developing cancers of the oral cavity, pharynx, larynx, esophagus, pancreas, kidney, bladder, and uterine cervix.5
While oral contraceptives provide reliable birth control, they also offer noncontraceptive benefits. Use of combined OCs leads to reduced risk of ovarian cancer and endometrial cancer.6 About 30% of 2008 survey participants says they specifically prescribed the Pill in the last year to help women decrease their risk of cancer of the ovary.
When it comes to initiating combined OC use in postpartum women who are not breast- feeding, about 49% of 2008 survey participants say they will begin pill use four to six weeks after delivery. About 20% say they start combined pills one to three weeks postpartum, while about 18% begin OC use upon hospital discharge.
For breast-feeding women who wish to use progestin-only pills, about 48% indicate they will initiate pill use four to six weeks postpartum, while about 25% say they begin progestin-only pill use one to three weeks following delivery. About 15% state they start minipills upon hospital discharge. Two percent initiate progestin-only pill use at first menses; 10% list other options.
References
- Barr Pharmaceuticals Inc. FDA Approves Duramed's LoSeasonique Oral Contraceptive. Press release. Accessed at phx.corporate-ir.net.
- Sulak PJ, Kuehl TJ, Ortiz M, et al. Acceptance of altering the standard 21-day/7-day oral contraceptive regimen to delay menses and reduce hormone withdrawal symptoms. Am J Obstet Gynecol 2002; 186:1,142-1,149.
- Harris Interactive, prepared for The National Women's Health Resource Center. Menstrual Management Survey Report. Aug. 29, 2008. Accessed at www.healthywomen.org/.
- Zieman M. Quick Start, extended regimens and shortened pill-free interval: New products and protocols for contraceptive management. Presented at the Contraceptive Technology Quest for Excellence Conference. Atlanta; October 2008.
- American Lung Association. Smoking and women fact sheet. Accessed at www.lungusa.org.
- Nelson A. "Combined Oral Contraceptives." In: Hatcher RA, Trussell J, Nelson AL, et al. Contraceptive Technology: 19th revised edition. New York City: Ardent Media; 2007.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
