Concussion Management for the Emergency Clinician
Concussion Management for the Emergency Clinician
Author: David W. Wright, MD, Co-Director for Research, Emergency Medicine Research Center, Department of Emergency Medicine, Emory University, Atlanta, GA.
Peer Reviewer: Thomas D. Kirsch, MD, MPH, FACEP, Associate Professor, Department of Emergency Medicine, Johns Hopkins University, Baltimore, MD.
Introduction
Concussions, also known as mild traumatic brain injuries (mTBI), create challenges for the emergency care provider. Only recently is good evidence available from well designed studies to guide diagnosis and therapy. A number of points now can be made with some confidence. First, patients with mTBI are an exceedingly common problem. Second, there are short- and long-term consequences of mTBI that should not be ignored. Third, after much debate and years of controversy, there is emerging agreement on a definition for mTBI. Fourth, return to play/school/work recommendations are more evidence-based, which provide a logical and practical way to instruct patients on what to do after an mTBI and how to return safely to their normal routines. In addition to these relatively significant developments, new studies provide further clarification of mTBI and how best to manage these patients.
Case 1: An 18-year-old male presents with a chief complaint of moderate headache, nausea, vomiting x 3 for the past 12 hours. His mom was told "there has been a virus going around at his school." He was healthy prior to the onset of these symptoms. He was a star athlete on his high school football team and played in a game the evening before. At the game he had to sit out a full quarter of play due to getting his "bell rung." There was no loss of consciousness. He continued to play, although he stated he was not feeling well. His symptoms are unremarkable, other than the headache and nausea/vomiting. No diarrhea. His vital signs are: Blood pressure 118/70, heart rate 101, respiratory rate 14, and he was afebrile. The physical examination was normal. He was awake and alert.
What is the differential diagnosis? Did he receive a concussion the night before? Is he at risk for intracranial injury? Does he need a CT? Can he return to practice?
Case 2: An 81-year-old female presents after a witnessed mechanical ground-level fall during the night at the nursing home. She has a history of atrial fibrillation and severe dementia. Her medication list includes labetolol, lisinopril, warfarin, and memantine. There are no other records available at this time. She is awake but confused, but her normal baseline mental status is not clear at this point. Her vital signs are normal, and her examination is unremarkable other than a small forehead contusion and her confused mental state.
What should this patient's work-up entail? Should she be admitted to the hospital for observation? What are her risk factors?
Background
The CDC estimates that there are more than 1.6-3.8 million traumatic brain injuries (TBI) each year in the United States.1,2 An overwhelming majority (approximately 80%) of these cases are classified as "mild" (mTBI).3,4 More than 20% of the soldiers returning from Iraq and Afganistan have sustained an mTBI.5 The problem is so significant that TBI has been labeled the "signature injury" of these conflicts. The culmination of new data from the sports medicine field and the growing number of injured soldiers have generated renewed interest in TBI.
The diagnosis of mTBI historically has been very challenging, in large part due to the continuing debate over the clinical definition. Most physicians agree on the physical signs and symptoms of a moderate and severe TBI (measured by the Glasgow Coma Scale [GCS] score; severe = 3-8 and moderate = 9-13), which are characterized by profound alterations in consciousness.6 However, patients with a mild TBI (GCS 14-15) by definition have very subtle changes in mental status, and fewer than 10% result in an initial loss of consciousness.7,8 mTBI should be suspected in any patient who has sustained a loss of consciousness, has amnesia around the event, or initially is confused or dazed. Many of these patients do not initially seek medical attention or are under-diagnosed by the medical community, making the determination of true incidence of mTBI impossible. Even when the diagnosis is suspected, patients are not hospitalized and observed as often as in the past. There is a real need for enhanced surveillance and evaluation for mTBI in the emergency room and outpatient primary care setting.2,9
Although an mTBI is often considered by the patient and lay public to be insignificant, there can be significant morbidity and even mortality. A substantial number of mTBI patients experience initial objective neuropsychological difficulties involving memory, attention, and executive functioning.10-19 These symptoms are often called post-concussive syndrome, which will be discussed in detail later in this issue. A significant subset of patients are left with persistent subjective cognitive complaints that disrupt their social relationships and their ability to resume leisure and work-related activities.20 Second impact syndrome may occur when a second concussion occurs prior to recovery from the first. Rapid onset of cerebral edema and death can occur.21-26 This syndrome is most often seen in athletes who prematurely return to play. The importance of the morbidity (i.e., prolonged cognitive deficits and affective and personality changes) and mortality (i.e., second impact syndrome) produced by mTBI is increasingly appreciated.21-26 There is good evidence that repetitive concussions result in long-term cognitive deficits and structural damage to the brain.24,26,27
Depression is a common secondary complication of mTBI.28-32 Early detection of mTBI is critical to patient education and treatment and potentially could prevent the secondary complications of depression and anxiety. Mittenberg and colleagues compared the effectiveness of standard hospital treatment and discharge instructions to education concerning the symptoms and their management in mTBI patients.33 At six months postinjury, 28% of patients who received standard treatment met ICD-10 criteria for post-concussion syndrome, compared to only 11% of the preventative treatment group. Patients in the preventative group also reported significantly shorter overall symptom duration, fewer symptoms, fewer symptomatic days in the previous week, and lower symptom severity levels. One implication of this research is that early detection could lead to interventions to mitigate the morbidity associated with mTBI.
The lack of a firm pathophysiological finding and a poor understanding of the underlying biomechanical forces that lead to mild cognitive deficits further complicate the diagnosis of mTBI. Although mechanical tolerances have been proposed for moderate and severe TBI, determination of thresholds for mTBI is complicated by poor patient recounts and delayed or inaccurate clinical diagnoses.34,35 Early and reliable diagnosis of mTBI may not only assist the patient with rehabilitation and improved outcome, but it will also provide a tool to correlate the acute response to a mild insult with the mechanical circumstances of the injury. Neuropsychological testing is a useful tool for detecting the often subtle changes resulting from mTBI but is rarely available to the emergency clinician.15,16,19,36,37
Pathophysiology
Moderate/Severe TBI. A more thorough understanding of the pathophysiology of severe TBI has emerged over the past 2 decades. Once thought to be a single event, TBI is now known to include the activation of a complex biochemical cascade that is cytotoxic and leads to expansion of the initial injury and an increase in long-term morbidity and mortality of the patient.38-40 These two phases of injury are fundamentally different and have been divided in the literature into the primary and secondary injury. The primary injury can include contusions (bruises to brain parenchyma), hematomas (subdural, epidural, intraparenchymal, intraventricular, and subarchanoid), diffuse axonal injury (DAI), direct cellular damage (neurons, axons, and other supportive cells), tearing and shearing of the tissues, loss of the blood-brain barrier, disruption of the neurochemical homeostasis, and loss of the electrochemical function. Secondary injury refers to the damage caused by the series of deleterious cellular and subcellular events (also know as the secondary cascade) that follow. This is not to be confused with the term "secondary insults," which is used in the clinical literature to describe conditions or circumstances (e.g., hypotension, hypoxemia, elevated ICP) that, if not treated, worsen the outcome of the patient with an acute TBI.
The secondary cascade is extremely complex and includes excessive release of glutamate into the presynaptic space, activation calcium channels (e.g., N-Methyl D-Aspartate, AMPA, and others41-46), an overwhelming influx of calcium into the cell, and a dis-equilibrium of other critical ions such as sodium and potassium.38 The calcium and ion shifts lead to mitochondrial damage, production of free radicals, activation and phosphoralation of enzymes, loss of membrane integrity, damage to ion transport systems, and a host of other dysfunctional events. The death of cells in the injured tissues leads to the production and release of cytokines and other pro-inflammatory enzymes. In addition, both cytotoxic edema (formed within the cell from loss of cell membrane integrity and ion exchange) and extracellular edema (from the breakdown of the blood-brain barrier and alteration of Aquaporins and other water exchange mechanisms) lead to swelling.47 Since the brain is essentially in a confined space, pressure rises and structures are damaged. Herniation of brain tissue and brain death can result.
Minor TBI. The underlying pathophysiology for mild TBI is not as clear. A leading hypothesis is that mTBI results in a predominantly metabolic insult as opposed to a structural one.39,40,48-55 Ion shifts result in mitochondrial dysfunction and depletion of intracellular energy stores. The brain may be exceedingly vulnerable during this time and when taxed (low oxygen state, recurrent insult, or concussion), could result in an exponential increase in dysfunction and cellular injury. Even minor insults could have profound effects. In fact, anything that increases metabolic demand, such as heavy mental or physical exercise, could worsen the injury. This concept of metabolic injury explains the return of symptoms with physical and mental activity and guides the new approach to return to play and work recommendations. It also explains why structural imaging techniques, such as CT, are poor determinants of injury. Studies using functional imaging techniques tend to support this theory. There is evidence that structural injury also occurs. The fact that structural abnormalities cannot be identified on MRI or CT does not preclude their existence. Indeed, new imaging techniques such as diffusion tensor imaging are revealing some of the underlying cellular-level damage that occurs.56-60 The predominant form of structural injury in mTBI is a form of diffuse axonal injury. The end result is an injury that puts the brain in a vulnerable state and suggests the need for "brain rest" and avoidance of secondary injuries.
Diagnosis
The definitions developed by the American Congress of Rehabilitation Medicine, the CDC, and the American Academy of Neurology (AAN) are essentially the same and now have been widely adopted by the medical community.61,62 This definition includes:
Exposure to a blunt force or acceleration deceleration injury and any period of transient confusion, disorientation, impaired consciousness, loss of consciousness for less than 30 minutes, and any period of dysfunction in memory (amnesia) associated with the event, neurological or neuropsychological dysfunction.
A simpler version now advocated is "any trauma-induced alteration in mental status."63
Loss of consciousness (LOC) is not a defining element of a concussion. The lack of LOC does not eliminate the possibility of underlying pathology, the presence of positive CT, or persistent symptoms. LOC remains an important risk factor for injury, but is only one of several risk factors (vomiting, focal neurologic complaint, etc.) after injury.64-68
For the emergency care provider, the diagnosis of mTBI essentially resides on the history surrounding the exposure, i.e., was there any alteration in the mental state at the time of the event or subsequent to the event? This definition includes the layman's account of getting your "bell rung," "seeing stars," or feeling dazed or confused as a result of the injury. The presence of amnesia further solidifies the diagnosis and may be associated with more significant injury. Patients who have loss of consciousness lasting longer than 30 minutes or who have a Glasgow Coma Score of less than 14 are not considered "mTBI" and should be evaluated as moderate or severe, depending on the examination. Note that GCS 13, previously defined as mild, is now classified as moderate, as the risk for complications for these patients is similar to moderate TBI patients. A tool for proper screening and evaluation (Acute Concussion Management – ACE) has been developed and is available in the Center for Disease Control (CDC) Physicians' Tool Kit through the CDC web site (www.cdc.gov/ncipc/tbi/physicians_tool_kit.htm).
Many factors complicate the diagnosis. The presence of alcohol, other injuries, and clarity of the events (dependence on patient recollection) can make it unclear whether the patient sustained a concussion. Currently there are no widely available and reliable tests that can confidently rule a concussion in or out. Serum markers, discussed later, are evolving and may provide a new critical role in diagnosis and management. CT is an important modality to rule out injuries that need neurosurgical interventions, but as stated above, is negative in most patients with mTBI. A CT does not predict post-concussive syndrome.
The clinical signs and symptoms associated with an mTBI are listed in Table 1.69 It should be noted that clinical symptoms may begin immediately after the insult or may be delayed for days to weeks. Therefore, the lack of current (at the time of evaluation) signs and symptoms does not exclude an mTBI if the historical account is consistent. Another complicating factor is that many of these signs and symptoms are nonspecific and overlap with other conditions.
 |
Physical Examination Findings
The physical examination in isolated mTBI often is normal, but it is important to document a full examination and to look for clues of injury mechanism severity and neurological impairment. As with all emergency department patients, the ABCs (airway, breathing, and circulation) should be evaluated, stabilized when indicated, and documented. The cervical spine should always be assessed and documented. Central cervical tenderness or an inability to obtain a reliable examination (e.g., intoxication, confusion, etc.) should prompt CT imaging of the spine. The presence of contusions or lacerations to the scalp may provide some insight when patients have an unclear history (e.g., intoxication, etc.).
A thorough neurological examination is also important. Focal findings are rare in mTBI and should alert the clinician to potentially significant intracranial pathology. A Glasgow Coma Score, although insensitive to subtle dysfunction associated with mTBI, is important to document. Signs of global impairment, such as confusion, perseveration, amnesia, or other findings, should also be assessed and recorded.
Mild TBI may result in impairment in balance, and an examination of gait or a balance challenge can expose ongoing symptoms. However, the most consistent abnormalities in mTBI are subtle impairments in cognitive function. Assessing cognitive function in the emergency setting is challenging. The "gold standard" neuropsychological examination is impractical in the acute setting. The Mini Mental Status Exam is relatively brief and easy to administer but is rarely performed and may be insensitive to subtle abnormalities. However, some assessment of cognition should be performed.
Grading Concussions
The practice of grading concussions into categories is still widely employed but is not evidence-based and can lead to errors in management. More than 20 different classification systems exist.36 These systems served a purpose for standardizing care and aiding in the decision process for returning to play and work when there were no solid data to direct care. However, they have also added more confusion to the management of concussion due to the sheer number of classification schemes, the lack of consistency, and the lack of evidence to support them.
A more scientific and evidence-based approach for management is now emerging. As discussed above (pathophysiology section), metabolic disturbances uncouple the energy supply-demand equilibrium, setting up a vulnerable state for neuronal cells.36,70 Symptoms reflect the underlying metabolic disturbances and therefore can guide management. When symptoms return with cognitive or physical exercise, it is a warning that mismatch may be occurring. Cessation of activity is advised, including a reduction of the level of activity. (See Return to Activity section.)
Imaging
CT is an important tool for determining if a life-threatening lesion is present. CT has many advantages over other imaging modalities. It is fairly ubiquitous in emergency departments in the United States, it is exquisitely sensitive to the presence of blood, and it guides management in the acute setting. But, which head-injured patients should receive a CT scan? Which patients with a negative CT scan can be discharged safely? In 2002, the American College of Emergency Physicians put out evidence-based guidelines on these topics.71 These guidelines were updated in 2008.68
Several well designed studies have addressed the significance of symptoms with respect to their ability to predict a positive finding on CT.64-67,72-76 The primary goals of these studies were to create decision rules to determine who was at risk for a significant intracranial lesion and who would need neurosurgical intervention. Another goal was to decrease the utilization of CT. The evidence does not address who will deteriorate if the CT is positive, or the relationship between positive or negative CT and the duration of post-concussive symptoms.
The data demonstrate fairly consistently that patients with mTBI and GCS of 14-15 will have a lesion on CT about 15% of the time, and fewer than 1% will require neurosurgical intervention.68
The first evidence-based decision rule, the New Orleans Criteria (see Table 2) was developed by Haydel, et al.73 The study had two phases, a rule-development phase (520 subjects) and a validation phase (909 subjects). In the study, 6.5% had an intracranial lesion, and 0.4% needed surgical intervention. Inclusion criteria for the study were an mTBI with GCS of 15 and either LOC or amnesia. The rule was 100% sensitive for detecting intracranial lesions on CT; however, it was only 5.3% specific.
 |
Another well validated decision rule is from Stiell, et al.72 Stiell developed the Canadian CT head rule (CCHR) based on prospectively evaluating 3,121 mTBI patients with a GCS of 14-15 for lesions leading to neurosurgical intervention or who were deemed "clinically important." The CCHR (see Table 2) was 100% sensitive at detecting their definition of clinically significant lesions. However, there is a very important caveat: The CCHR rules use a different definition for significant injury. The CCHR dismisses the significance of many positive CT findings (solitary contusions less than 5 mm, smear subdurals less than 4 mm thick, isolated pneumocephalus, and closed depressed skull fractures that do not penetrate the inner table, stating that these do not deteriorate). Follow-up assessment to document deterioration, however, was based on phone survey data and did not take into account non-neurosurgical sequelae.
Smits, et al. compared the NOC and CCHR in 3181 adults with mTBI and a GCS of 14-15.66 The NOC was 100% sensitive for neurosurgical lesion and 98% sensitive for an intracranial lesion, but was only 5.3% specific. The CCHR was 100% sensitive for neurosurgical lesion but only 83% sensitive for an intracranial lesion with an improved specificity of 37% and 39%, respectively. Steill, et al. also compared the two rules and found similar results except they reported higher specificity for significant intracranial lesions; 13% NOC and 51% for CCHR. CT use would be reduced by 3% using the NOC and 37% with the CCHR.
A major downside of these two decision rules is that all patients in these trials had either LOC or amnesia. A majority of mTBI do not result in LOC and, as shown later, LOC is not the best predictor of intracranial pathology. Importantly, these rules cannot be applied to patients on anticoagulants or children, as they were excluded in the validation studies.
Several European studies also have attempted to create mTBI decision rules. The National Institute for Clinical Excellence (NICE) and the Neurotraumatology Committee of the World Federation of Neurosurgical Societies (NCWFNS) more recently have evaluated clinical signs and symptoms.64,67 These decision rules have been applied to large data sets and shown to be relatively sensitive (NICE – 94% for neurosurgical lesions, 82% for intracranial lesions, and NCWFNS – 100% for neurosurgical lesions and intracranial injuries). Ibanez, et al. evaluated 1101 mTBI patients with GCS 14-15 and found approximately 2% of patients without LOC had intracranial lesions and 0.6% required surgery (similar rates to those with LOC).
One of the most important findings from these studies is the relative significance of certain elements of the history and physical. For example, nausea and vomiting after the concussion has an odds ratio comparable to LOC for a positive CT. (See Table 3.) Other factors are as important as LOC, if not more.
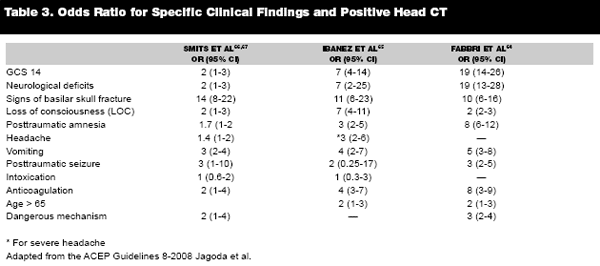 |
These decision rules can guide clinical practice, but each clinician has to decide what is an acceptable risk in his or her own practice and recognize that none of these rules address short- or long-term non-neurosurgical sequelae such as post-concussive syndrome.
Magnetic resonance imaging (MRI) can detect subtle lesions that CT imaging cannot. However, despite the enhanced capabilities of the MRI, it is still not sensitive for mTBI. MRIs are not readily available at most facilities, are more expensive, and have other limitations related to the magnetic field. Moreover, it is not clear how that the additive information provided translates into any clinical importance.
Functional MRI (fMRI) has been a useful research tool. fMRI can correlate functional elements, such as oxygen demand, to neuropsychological testing paradigms.77-79 It will remain an important research tool but is not likely to be available to the emergency care provider in the near future.
Another promising imaging technique is Diffusion Tensor Imaging (DTI).56-60 DTI images the disruption of water molecules that flow along axons. Since the primary insult after injury is now believed to be diffuse axonal injury (DAI), it is providing significant insight into the pathophysiology of mTBI. As with MRI and fMRI, it is not readily available to the primary clinician.
Biomarkers
Biomarkers hold a lot of promise for screening, better classification of injury, and guiding therapy. Biomarkers would be useful to detect mTBI in cases where the diagnosis is difficult (intoxicated patients, poor historical record, etc). A number of different markers have been studied (Tau, NSE, S100b, and others). Until recently, there simply was not enough data to support the use of any of these markers. A lack of specificity for neuronal injury plagues most of these markers. However, S100b may be sufficiently sensitive to detect the presence of injury.68 Biberthaler, et al. reported that S100b has a 99% sensitivity and 30% specificity for detecting a positive CT when measured within 3 hours of a suspected mTBI.80 Other studies had similar findings, depending on the cutoff level used and the detection methodology.81,82 There are limitations to S100b. Serum levels rise and fall quickly, so time from injury would be critical to the relevance of a negative level. The current study provides only a dichotomous answer (concussion or not). Second, the laboratory test for S100b is not widely available. However, this is the first biomarker that could actually provide useful information in the clinical setting of acute TBI.
Cognitive Screening and Psychometrics
Cognitive testing in the ED has been limited to brief memory screens such as the Mini Mental Status Exam (MMSE). Other tools, primarily developed for sports, such as the Standardized Assessment for Concussion (SAC) and the newer Sports Concussion Assessment Tool (SCAT) are rarely performed in the ED and have not been validated in this setting. Although brief, these screens still require 10-15 minutes of dedicated time with the examiner to perform. These factors limit their widespread application in the ED.
Psychometrics, or neuropsychological (NP) testing, has been the cornerstone for assessing the long-term effects of an mTBI. NP testing measures cognitive function and can detect very subtle deficiencies.15,36,83-87 Testing consists of a battery of individual tests that evaluate a number of domains required for normal brain function (memory, attention, concentration, executive function, reaction time, etc). For TBI, the most important battery covers cognitive deficits such as short-term and long-term memory, reaction time, and executive function.
Classical NP testing involves a battery of paper tests that require more than 2 hours to perform. Obviously this type of testing is too time-intensive and not practical for the ED. A number of computerized versions have been developed that are shorter and easier to administer. Several of these tests are being employed by sports programs to guide recovery from concussion. They are best when baseline scores can be obtained (such as preseason testing) to compare within individuals. Baseline testing is not practical for the ED, and normative controls add more variability. However, cognitive testing in the ED may soon be available with more novel approaches being developed.
Treatment
There are no pharmacological treatments for mTBI. The primary objectives of the emergency department clinician are to identify patients who have intracranial lesions requiring neurosurgical intervention, identify patients who might deteriorate, and guide return to normal activities. Historically, emergency clinicians have done a good job of identifying intracranial lesions and recognizing patients who might deteriorate, but have not performed well in providing evidenced-based discharge instructions or return to activity information. In defense, much of the literature on this topic is fairly new and still under development. However, there are enough data to provide recommendations with moderate certainty.
Treatment begins with proper identification of a patient with mTBI. Once recognized, a history and physical examination should be performed with a focus on factors that increase the risk of intracranial pathology, as outlined in Table 3.68
Acute head trauma patients who are at risk for intracranial pathology and in whom a noncontrast head CT should be performed are those with focal neurologic deficits, vomiting, moderate to severe headache, age 65 years or greater, physical signs of a basilar skull fracture, GCS score less than 15, coagulopathy (or anticoagulant therapy), or a dangerous mechanism of injury.
In addition, patients with alcohol and drug intoxication, short-term memory deficits, physical evidence of trauma above the clavicle and/or posttraumatic seizures, who have had loss of consciousness or posttraumatic amnesia also should be considered for a CT.
It is always wise to discharge the patient to the care of a responsible individual and provide instructions to both the patient and the caregiver. Several studies have documented poor understanding of discharge instructions in mTBI patients.88,89 If a CT is not clinically indicated, or the S100b is below the cutoff point, the patient should receive careful instructions on delayed symptoms and when to return to the ED, in addition to the instructions on return to activity and follow up. For whatever reason, in patients for whom a CT is indicated but not available, observation is recommended. There are no data to support any specific time window for observation.
Follow-up
Discharge planning should include explicit instructions regarding follow up. The CDC's Physicians' Tool Kit is an excellent resource for discharge instructions and follow up. Patients who are symptom free and have returned to their normal activities of daily living do not specifically need to follow up. Patients continuing to have symptoms for more than 3 weeks should seek follow up with a qualified healthcare provider who has experience and or training in mTBI. It is recommended that every ED identify local providers with such training. Most sports medicine training programs include mTBI curriculum. These experts can manage the ongoing symptoms, refer for further testing (imaging and or neuropsychological evaluation), and inform the patients and family members about how to navigate their environment.
Return to Activity: Play/School/Work (PSW)
Based on the pathophysiology and the recognition that symptoms reflect an underlying vulnerable state, a graded return to PSW has been developed. This recovery program is simple and has many advantages over previous recommendations and guidelines. In many guidelines, an arbitrary recovery period of 7 days after a "grade II" concussion was recommended.90 This time was not evidence-based and happened to coincide with weekly sporting events as opposed to evidence of recovery. For non-athletic return to work or school, no formal recommendations have been developed. However, adoption of the sports return-to-play scheme (with some modifications) is logical and consistent with the symptom-based approach. (See Table 4.)
 |
Protracted Symptoms. There are no consistent findings that can predict the frequency or intensity of post-concussive symptoms. Most studies have shown almost complete resolution of symptoms by 6 weeks in 80% of patients.92 The most common symptoms involve headache, difficulty concentrating, and short-term memory difficulties. The treatment of symptoms includes rest, including "brain rest," and therapies targeted toward specific symptoms (NSAIDs, hydration, etc.). Aspirin should be avoided to prevent any bleeding complications.
Post-Concussive Syndrome (PCS)
Patients often report a series of physical, emotional, and cognitive symptoms in the days and weeks after their mTBI. While most mTBI patients will go on to make a full recovery, some individuals will have persistent symptoms for months to years that can cause significant disability. The estimated prevalence of PCS varies widely, with about 20-40% of subjects reporting symptoms at three months and about 15% with symptoms at 1 year.12,93 The most commonly reported post-concussion symptoms are headache, dizziness, decreased concentration, memory problems, sleep disturbances, irritability, fatigue, visual disturbances, judgment problems, depression, and anxiety.94,95 When a cluster of symptoms becomes chronic after an mTBI, it is often called persistent post-concussive symptoms (PPCS) or post-concussion syndrome (PCS). There are two overlapping but distinct official definitions for PCS; one is listed in the ICD 10th Revision of the International Classification of Diseases (ICD-10) clinical criteria, and the other is in the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV). Adding to the confusion, the terms post-concussive symptoms (symptoms that occur within 4 weeks after the injury) and post-concussive syndrome (symptoms that last longer than 1 month – ICD 10 definition, or more than 3 months – DSMV-IV definition) are mistakenly interchanged and both have the "PCS" acronym. The underlying pathophysiology of PCS is equally unclear. There is so much symptom overlap with post-traumatic stress disorder (PTSD) and other non-traumatic conditions that making the distinction based on clinical grounds is not currently possible. Moreover, none of the clinical findings at the time of the injury (CT scan, loss of consciousness, etc.) reliably predict PCS. Nevertheless, it is clear that a small percentage of patients will have persistent symptoms that are associated with their original injury and are often disabling. Neuropsychological testing and a symptom checklist are the cornerstones for the diagnosis and management. The primary approach is to treat the symptoms. Appropriate referral to a neuropsychologist or mTBI clinic is necessary to manage these patients effectively.
Special Considerations
Coagulapathy. Patients on warfarin and other blood thinners require a note of caution. Intuitively, there is an increased risk for intracranial hemorrhage in patients on anticoagulants who sustain a TBI. Some experts believe that even trivial injuries, such as ground-level falls can result in catastrophic intracranial hemorrhages, especially in elderly patients. A number of factors likely play a role. First, most patients on anticoagulants are elderly, and it is well documented that elderly patients have poorer outcomes after TBI. Second, anatomical differences (atrophied brain, vulnerable bridging veins, etc.) and decrease parenchymal compliance increase the risk for intracranial hemorrhage in elderly patients. Third, patients on anticoagulants often have other co-morbidities that can affect overall outcome after a TBI. There are no prospective studies that definitively demonstrate the risk. Several retrospective studies of adequate quality demonstrate at least a four-fold increase in poorer outcome if the INR is elevated.79,96-98 Patients on anticoagulants that are sub-therapeutic may not have a significant increased risk, but that is not certain as this time.
The true incidence for intracranial hemorrhage in patients on warfarin who have sustained an mTBI is not known. However, hemorrhages in patients on warfarin and who have an elevated INR have an exceedingly high mortality (more than 80%).99 In general, all patients on warfarin with a suspected TBI should have an emergent head CT and INR checked. Most patients with intracranial hemorrhage will need immediate anticoagulant reversal with FFP, vitamin K, or possibly other appropriate reversal agents and emergent neurosurgical consultation.
Another, more controversial question is what to do with anticoagulated patients after a negative CT. Delayed hemorrhage is reported in the literature.100 There are no data on the risk of delayed hemorrhage after very minor head injuries in patients with a negative CT and who are anticoagulated. The degree of anticoagulation likely plays an important role, but it is uncertain to what degree. Cohen, et al. reported that an alarming number of patients (~32 out of 45) admitted with a normal head CT and elevated INR after TBI deteriorated. Mortality in these patients was more than 80%.99 Cohen suggests, and the author agrees, that patients with an abnormal INR (2 or greater) should be admitted and observed, even with a normal initial CT. It is also not clear if antiplatelet agents (e.g., aspirin, ticlid, clopidogrel) increase the risk of intracranial bleeding in the setting of mTBI.
Sports Concussions. In addition to the general discussions of mTBI, detecting concussions in athletes can be more challenging. Athletes in general do not want to be pulled from the active roster or the game and tend to downplay their symptoms. For athletic trainers and coaches, the problem becomes more profound in the chaotic environment of a game where the athlete is energized and emotionally involved. Pressure on the team to perform is not isolated to the players, but also applies to the coaches and staff. Athletic trainers and sports medical personnel often are positioned between the needs of the team and the health of the athlete. Sports medicine-trained physicians and certified athletic trainers typically are well versed in concussion recognition and management. In fact, in organized sports at the college level or above, most athletes who sustain concussions are managed outside the ED unless imaging is required. However, in teams or sports without any trained health personnel, the athlete often will go unnoticed entirely.
In athletes, decisions on return to play should be managed very carefully and preferably by health providers trained in concussion management. The role of the ED physician is to inform the athlete that he or she should not return to activities or training until cleared by the team trainer/physician. The reason behind this additional caution is the increased risk and exposure of the athlete to repeat concussion and injury. Recurrent concussions are shown to worsen outcome and may lead to second impact syndrome.101,102 A graded activity program should be instituted to clear athletes for play.103,104 (See Return to PSW section.)
Second Impact Syndrome. Second impact syndrome is a rare event that results in rapid cerebral edema and high mortality.24,105,106 The pathophysiology and the predictors are not well understood. It is hypothesized that a second impact before the brain has reset and/or recovered from the first causes a loss of autoregulation, ion imbalance, and rapid cerebral edema. This explanation fits well with the concept of the metabolic disturbance, energy demand mismatch, and the vulnerable state of the brain after a concussion. Almost all cases of second impact syndrome have occurred in young athletes. However, the rarity of the condition is evidence that there are other factors that predispose and/or contribute to this devastating condition.
Recurrent Concussions. There is now strong evidence that recurrent concussions are more significant than the initial one and make the patient more susceptible to further concussions.101,102 Young age appears to be a risk factor for the increased susceptibility.107-109 Lovell, et al. have shown diminished cognitive function, slower recovery times, and prolonged disability in injured high school and college athletes. It is controversial as to whether this translates to older athletes, such as those in professional sports.110 The increased vulnerability is of special concern because of the lack of consistent medical oversight in youth sports.
Dementia Pugilistica. Dementia pugilistica is a special condition of early onset dementia related to boxing and other contact sports that involve multiple direct blows to the head.25,111,112 Concussions are common in boxing. Indeed, it is the only sport whose whole intent is to produce a concussion, the KO (knock out). There is some evidence connecting head injury and Alzheimer's disease in some patients.113
Case Resolutions
Case I: The 18-year-old male with a chief complaint of moderate headache, nausea, vomiting x 3 for the past 12 hours had a subarachnoid hemorrhage and a smear subdural on CT. He had suffered a concussion the night before ("bell rung") during the football game. Even though he had no history of loss of consciousness, the elements in his history that should have warned of potential intracranial injury were moderate to severe headaches and > 2 episodes of vomiting. He was admitted to the hospital for observation. He required no surgery and was discharged 2 days later in good condition.
What is the differential diagnosis? He could have had a viral syndrome or gastroenteritis, but one must be suspicious of a concussion with significant intracranial injury given the recount of his injury during the game and the symptoms described.
Did he receive a concussion the night before? Yes.
Is he at risk for intracranial injury? Yes, moderate to severe headache and vomiting more than 2 times are risk factors for intracranial injury despite no history of loss of consciousness.
Does he need a CT? Yes.
Can he return to practice? No. Given the degree of intracranial injury, it would be recommended he not be involved in contact sports for the season, and possibly the rest of his life. If he had no intracranial injury and reported the same symptoms, the recommendation should be not to engage in significant activity until his symptoms resolve and then begin a graded return-to-play program under the supervision of an experienced medical provider.
Case 2: The 81-year-old female patient was at high risk for intracranial hemorrhage (ICH) due to the warfarin she was taking for atrial fibrillation. Her INR was 3.5. Patients with supra-therapeutic INR levels have a high degree of risk for ICH, which, if it occurs, has a mortality of more than 80%. The patient should be admitted for observation even if the CT scan is negative. There is evidence of delayed ICH in these patients. Some physicians would recommend FFP to protect against the potential for ICH. If the patient's INR is in the therapeutic range, it is not clear if there is a significant increase in ICH. However, a period of observation would be prudent.
What should this patient's work-up entail? A CT scan and INR should be obtained.
Should she be admitted to the hospital for observation? Yes, if the INR is supra-therapeutic or the CT shows evidence of hemorrhage.
What are her risk factors? The patient is on warfarin and elderly. Both increase the risk of intracranial hemorrhage.
Conclusion
Mild TBI is a significant global health problem. Recent advances in the understanding of mTBI have led to a more conservative approach to management. Recognition is the key to appropriate intervention. The emergency clinician should be well versed in the signs and symptoms of mTBI, understand the appropriate work-up, and better inform patients about their injury.
References
1. Langlois JA, Rutland-Brown W, Wald MM. The epidemiology and impact of traumatic brain injury: A brief overview. J Head Trauma Rehabil 2006:21:375-378.
2. Thurman D. The epidemiology and economics of head trauma. In: Miller L, ed. Head Trauma: Basic, Preclinical, and Clinical Directions. New York: Wiley and Sons: 2001.
3. No authors listed. Injury Fact Book. Atlanta: National Center for Injury Prevention and Control: 2002.
4. Centers for Disease Control and Prevention. Traumatic Brain Injury. 2003. Available from: http://www.cdc.gov/ncipc/factsheets/tbi.htm.
5. Warden D. Military TBI during the Iraq and Afghanistan wars. J Head Trauma Rehabil 2006;21:398-402.
6. Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet 1974;2:81-84.
7. Cantu RC. Head injuries in sports. Br J Sports Med 1996;30:239-289.
8. Cantu RC. Reflections on head injuries in sport and the concussion controversy. Clin J Sport Med 1997;7:83-84.
9. Thurman D, Guerrero J. Trends in hospitalization associated with traumatic brain injury. JAMA 1999;282:954-957.
10. Gronwall D. Cumulative and persisting effects of concussion on attention and cognition in mild head injury. Levin HS, Eisenberg HM, ed. New York: Oxford University Press; 1989:153-162.
11. Gronwall D, Wrightson P. Delayed recovery of intellectual function after minor head injury. Lancet 1974;2:605-609.
12. Binder LM, Rohling ML, Larrabee GJ. A review of mild head trauma. Part I: Meta-analytic review of neuropsychological studies. J Clin Exp Neuropsychol 1997;19:421-431.
13. Dikmen S, Machamer J, Temkin N, et al. Neuropsychological recovery in patients with moderate to severe head injury: 2 year follow-up. J Clin Experimental Neuropsychology 1990;12:507-519.
14. Dikmen S, Machamer JE. Neurobehavioral outcomes and their determinants. J Head Trauma Rehabil 1995;10:74-86.
15. Dikmen S, Mclean A, Temkin N. Neuropsychological and psychosocial consequences of minor head injury. J Neurol Neurosurg Psychiatry 1986;49:1227-1232.
16. Hinton-Bayre AD, Geffen GM, Geffen LB, et al. Concussion in contact sports: Reliable change indices of impairment and recovery. J Clin Exp Neuropsychol 1999;21:70-86.
17. Macciocchi SN, Barth JT, Alves W, et al. Neuropsychological functioning and recovery after mild head injury in collegiate athletes. Neurosurgery 1996;39:510-514.
18. Ponsford J, Willmott C, Rothwell A, et al. Factors influencing outcome following mild traumatic brain injury in adults. J Int Neuropsychol Soc 2000;6:568-579.
19. Levin HS, Mattis S, Ruff RM, et al. Neurobehavioral outcome following minor head injury: A three-center study. J Neurosurg 1987;66:234-243.
20. Van Der Naalt J. Prediction of outcome in mild to moderate head injury: A review. J Clin Exp Neuropsychol 2001;23:837-851.
21. Cantu RC. Catastrophic injury of the brain and spinal cord. Virginia Medical Quarterly 1996;123:98-102.
22. Cantu RC. Head and spine injuries in the young athlete. Clin Sports Med 1988;7:459-472.
23. Cantu RC. Head and spine injuries in youth sports. Clin Sports Med 1995;14:517-532.
24. Cantu RC. Second-impact syndrome. Clin Sports Med 1998;17:37-44.
25. Erlanger DM, Kutner KC, Barth JT, et al. Neuropsychology of sports-related head injury: Dementia pugilistica to post concussion syndrome. Clin Neuropsychol 1999;13:193-209.
26. Kelly JP, Nichols JS, Filley CM, et al. Concussion in sports. Guidelines for the prevention of catastrophic outcome. JAMA 1991;266:2867-2869.
27. Cantu RC, Voy R. Second-impact syndrome a risk in any contact sport. Physician and Sports Medicine 1995;23:27.
28. Fenton G, Mcclelland R, Montgomery A, et al. The postconcussional syndrome: Social antecedents and psychological sequelae. Br J Psychiatry 1993;162:493-497.
29. Mathias JL, Coats JL. Emotional and cognitive sequelae to mild traumatic brain injury. J Clin Experimental Neuropsychology 1999;21:200-215.
30. Rutherford WH, Merrett JD, McDonald JR. Symptoms at one year following concussion from minor head injuries. Injury 1979;10:225-230.
31. Levin HS. Prediction of recovery from traumatic brain injury. J Neurotrauma 1995;12:913-922.
32. Levin HS, High WM, Goethe KE, et al. The Neurobehavioural Rating Scale: Assessment of the behavioural sequelae of head injury by the clinician. J Neurol, Neurosurg Psychiatry 1987;50:183-193.
33. Mittenberg W, Tremont G, Zielinski RE, et al. Cognitive-behavioral prevention of postconcussion syndrome. Arch Clin Neuropsychol 1996;11:139-145.
34. Lighthall JW, Dixon CE, Anderson TE. Experimental models of brain injury. J Neurotrauma 1989;6:83-97.
35. Margulies SS, Thibault LE. A proposed tolerance criterion for diffuse axonal injury in man. J Biomech 1992;25:917-923.
36. Lovell M. The neurophysiology and assessment of sports-related head injuries. Neurol Clin 2008;26:45-62; viii.
37. Leininger BE, Gramling SE, Farrell AD, et al. Neuropsychological deficits in symptomatic minor head injury patients after concussion and mild concussion. J Neurol Neurosurg Psychiatry 1990;53:293-296.
38. Mcintosh TK, Smith DH, Meaney DF, et al. Neuropathological sequelae of traumatic brain injury: Relationship to neurochemical and biomechanical mechanisms. Lab Invest 1996;74:315-342.
39. Hovda DA. Oxidative need and oxidative capacity following traumatic brain injury. Crit Care Med 2007;35:663-664.
40. Hovda DA, Lee SM, Smith ML, et al. The neurochemical and metabolic cascade following brain injury: Moving from animal models to man. J Neurotrauma 1995;12:903-906.
41. Nakazawa M, Matsuno K, Mita S. Activation of sigma1 receptor subtype leads to neuroprotection in the rat primary neuronal cultures. Neurochem Int 1998;32:337-343.
42. Peng TI, Jou MJ, Sheu SS, et al. Visualization of NMDA receptor-induced mitochondrial calcium accumulation in striatal neurons. Exp Neurol 1998;149:1-12.
43. Tavalin SJ, Ellis EF, Satin LS. Inhibition of the electrogenic Na pump underlies delayed depolarization of cortical neurons after mechanical injury or glutamate. J Neurophysiol 1997;77:632-638.
44. Uematsu D, Greenberg JH, Araki N, et al. Mechanism underlying protective effect of Mk-801 against NMDA-induced neuronal injury in vivo. J Cereb Blood Flow Metab 1991;11:779-785.
45. Weaver CE, Marek P, Park-Chung M, et al. Neuroprotective activity of a new class of steroidal inhibitors of the N-methyl-D-aspartate receptor. Proc Natl Acad Sci USA 1997;94:10450-10454.
46. Zhang L, Rzigalinski BA, Ellis EF, et al. Reduction of voltage-dependent Mg2+ blockade of NMDA current in mechanically injured neurons. Science 1996;274:1921-1923.
47. Marmarou A. A review of progress in understanding the pathophysiology and treatment of brain edema. Neurosurg Focus 2007;22:E1.
48. Hillered L, Enblad P. Nonischemic energy metabolic crisis in acute brain injury. Crit Care Med 2008;36:2952-2953.
49. Marcoux J, Mcarthur DA, Miller C, et al. Persistent metabolic crisis as measured by elevated cerebral microdialysis lactate-pyruvate ratio predicts chronic frontal lobe brain atrophy after traumatic brain injury. Crit Care Med 2008;36:2871-2877.
50. Belli A, Sen J, Petzold A, et al. Metabolic failure precedes intracranial pressure rises in traumatic brain injury: A microdialysis study. Acta Neurochir (Wien) 2008;150:461-469; discussion 470.
51. Prins ML. Cerebral metabolic adaptation and ketone metabolism after brain injury. J Cereb Blood Flow Metab 2008;28:1-16.
52. Fiskum G. Mitochondrial participation in ischemic and traumatic neural cell death. J Neurotrauma 2000;17:843-855.
53. Lifshitz J, Sullivan PG, Hovda DA, et al. Mitochondrial damage and dysfunction in traumatic brain injury. Mitochondrion 2004;4:705-713.
54. Vespa P, Bergsneider M, Hattori N, et al. Metabolic crisis without brain ischemia is common after traumatic brain injury: A combined microdialysis and positron emission tomography study. J Cereb Blood Flow Metab 2005;25:763-774.
55. Hovda DA, Yoshino A, Kawamata T, et al. The increase in local cerebral glucose utilization following fluid percussion brain injury is prevented with kynurenic acid and is associated with an increase in calcium. Acta Neurochir Suppl (Wien) 1990;51:331-333.
56. Rutgers DR, Fillard P, Paradot G, et al. Diffusion tensor imaging characteristics of the corpus callosum in mild, moderate, and severe traumatic brain injury. AJNR Am J Neuroradiol 2008;29:1730-1735.
57. Wilde EA, Mccauley SR, Hunter JV, et al. Diffusion tensor imaging of acute mild traumatic brain injury in adolescents. Neurology 2008;70:948-955.
58. Rutgers DR, Toulgoat F, Cazejust J, et al. White matter abnormalities in mild traumatic brain injury: A diffusion tensor imaging study. AJNR Am J Neuroradiol 2008;29:514-519.
59. Bazarian JJ, Zhong J, Blyth B, et al. Diffusion tensor imaging detects clinically important axonal damage after mild traumatic brain injury: A pilot study. J Neurotrauma 2007;24:1447-1459.
60. Inglese M, Makani S, Johnson G, et al. Diffuse axonal injury in mild traumatic brain injury: A diffusion tensor imaging study. J Neurosurg 2005;103:298-303.
61. Cantu RC, Aubry M, Dvorak J, et al. Overview of concussion consensus statements since 2000. Neurosurg Focus 2006;21:E3.
62. Mccrory P, Johnston K, Meeuwisse W, et al. Summary and Agreement Statement of the 2nd International Conference on Concussion in Sport, Prague 2004. Clin J Sport Med 2005;15:48-55.
63. Practice Parameter: The Management of Concussion in Sports (Summary Statement). Report of the Quality Standards Subcommittee. Neurology 1997;48:581-585.
64. Fabbri A, Servadei F, Marchesini G, et al. Clinical performance of Nice recommendations versus Ncwfns proposal in patients with mild head injury. J Neurotrauma 2005;22:1419-1427.
65. Ibanez J, Arikan F, Pedraza S, et al. Reliability of clinical guidelines in the detection of patients at risk following mild head injury: Results of a prospective study. J Neurosurg 2004;100:825-834.
66. Smits M, Dippel DW, De Haan GG, et al. External validation of the Canadian CT Head Rule and the New Orleans Criteria for CT Scanning in patients with minor head injury. JAMA 2005;294:519-1525.
67. Smits M, Dippel DW, De Haan GG, et al. Minor head injury: Guidelines for the use of CTa multicenter validation study. Radiology 2007;245:831-838.
68. Jagoda AS, Bazarian JJ, Bruns JJ, et al. Clinical Policy: Neuroimaging and decisionmaking in adult mild brain injury in the acute setting, in ACEP and CDC Clinical Policy. 2008.
69. Centers for Disease Control. Heads Up: Brain injury in your practice: A tool kit for physicians, National Center for Injury Prevention and Control and (NCIPC), eds. Atlanta: Centers for Disease Control and Prevention: 2007.
70. Giza CC, Hovda DA. The neurometabolic cascade of concussion. J Athl Train 2001;36:228-235.
71. Jagoda AS, Cantrill SV, Wears RL, et al. Clinical Policy: Neuroimaging and decisionmaking in adult mild traumatic brain injury in the acute setting. Ann Emerg Med 2002;40:231-249.
72. Stiell IG, Laupacis A, Wells GA. Indications for computed tomography after minor head injury. Canadian CT Head and Cervical-Spine Study Group. N Engl J Med 2000;343:1570-1571.
73. Haydel MJ, Preston CA, Mills TJ, et al. Indications for computed tomography in patients with minor head injury. N Engl J Med 2000;343:100-105.
74. Stiell IG, Clement CM, Rowe BH, et al. Comparison of the Canadian CT Head Rule and the New Orleans Criteria in patients with minor head injury. JAMA 2005;294:1511-1518.
75. Stein SC, Fabbri A, Servadei F, et al. A critical comparison of clinical decision instruments for computed tomographic scanning in mild closed traumatic brain injury in adolescents and adults. Ann Emerg Med 2008, Mar 11.
76. Fabbri A, Vandelli A, Servadei F, et al. Coagulopathy and Nice recommendations for patients with mild head injury. J Neurol Neurosurg Psychiatry 2004;75:1787-1788.
77. Chen JK, Johnston KM, Frey S, et al. Functional abnormalities in symptomatic concussed athletes: An FMRI study. Neuroimage 2004;22:68-82.
78. Ptito A, Chen JK, Johnston KM. Contributions of functional magnetic resonance imaging (FMRI) to sport concussion evaluation. NeuroRehabilitation 2007;22:217-227.
79. Mcallister TW, Sparling MB, Flashman LA, et al. Early plateau of brain activation on FMRI in response to increased working memory task difficulty one month after mild traumatic brain injury (mTBI). J Neuropsychiatry Clin Neurosciences 2000;12:159-160.
80. Biberthaler P, Linsenmeier U, Pfeifer KJ, et al. Serum S-100B concentration provides additional information for the indication of computed tomography in patients after minor head injury: A prospective multicenter study. Shock 2006;25:446-453.
81. De Kruijk JR, Leffers P, Menheere PP, et al. S-100b and neuron-specific enolase in serum of mild traumatic brain injury patients. A comparison with health controls. Acta Neurologica Scandinavica 2001;103:175-179.
82. Herrmann M, Jost S, Kutz S, et al. Temporal profile of release of neurobiochemical markers of brain damage after traumatic brain injury is associated with intracranial pathology as demonstrated in cranial computerized tomography. J Neurotrauma 2000;17:113-122.
83. Blostein PA, Jones SJ, Buechler CM, et al. Cognitive screening in mild traumatic brain injuries: Analysis of the neurobehavioral cognitive status examination when utilized during initial trauma hospitalization. J Neurotrauma 1997;14:171-177.
84. Brooks DN. Measuring neuropsychological and functional recovery. In: Levin HS, ed. Neurobehavioral Recovery from Head Injury. New York: Oxford University Press: 1987:57-72.
85. Brooks J, Fos LA, Greve KW, et al. Assessment of executive function in patients with mild traumatic brain injury. J Trauma 1999;46:159-163.
86. Cicerone KD, Smith LC, Ellmo W, et al. Neuropsychological rehabilitation of mild traumatic brain injury. Brain Injury 1996;10:277-286.
87. Collins MW, Grindel SH, Lovell MR, et al. Relationship between concussion and neuropsychological performance in college football players. JAMA 1999;282:964-970.
88. Levitt MA. Minor head injury. Ann Emerg Med 1994;23:1143-1144.
89. Saunders CE, Cota R, Barton CA. Reliability of home observation for victims of mild closed-head injury. Ann Emerg Med 1986;15:160-163.
90. No authors listed. Practice Parameter: The management of concussion in sports (summary statement). Report of the Quality Standards Subcommittee. Neurology 1997;48:581-585.
91. Aubry M, Cantu R, Dvorak J, et al. Summary and Agreement Statement of the 1st International Symposium on Concussion in Sport, Vienna 2001. Clin J Sport Med 2002;12:6-11.
92. Jagoda A, Riggio S. Mild traumatic brain injury and the postconcussive syndrome. Emerg Med Clin North Am 2000;18:355-363.
93. Ingersoll CD. Long-term effects of closed head injuries in sport. Sports Med 1993;16:342-354.
94. Ryan LM, Warden DL. Post concussion syndrome. Int Rev Psychiatry 2003;15:310-316.
95. Kushner D. Mild traumatic brain injury: Toward understanding manifestations and treatment. Arch Intern Med 1998;158:1617-1624.
96. Karni A, Holtzman R, Bass T, et al. Traumatic head injury in the anticoagulated elderly patient: A lethal combination. Am Surg 2001;67:1098-1100.
97. Mina AA, Knipfer JF, Park DY, et al. Intracranial complications of preinjury anticoagulation in trauma patients with head injury. J Trauma 2002;53:668-672.
98. Li J, Brown J, Levine M. Mild head injury, anticoagulants, and risk of intracranial injury. Lancet 2001;357:771-772.
99. Cohen DB, Rinker C, Wilberger JE. Traumatic brain injury in anticoagulated patients. J Trauma 2006;60:553-557.
100. Reynolds FD, Dietz PA, Higgins D, et al. Time to deterioration of the elderly, anticoagulated, minor head injury patient who presents without evidence of neurologic abnormality. J Trauma 2003;54:492-496.
101. Iverson GL, Gaetz M, Lovell MR, et al. Cumulative effects of concussion in amateur athletes. Brain Inj 2004;18:433-443.
102. Collins MW, Lovell MR, Iverson GL, et al. Cumulative effects of concussion in high school athletes. Neurosurgery 2002;51:1175-1179; discussion 1180-1171.
103. Guskiewicz KM, Bruce SL, Cantu RC, et al. Research based recommendations on management of sport related concussion: Summary of the National Athletic Trainers' Association Position Statement. Br J Sports Med 2006;40:6-10.
104. Lovell M, Collins M, Bradley J. Return to play following sports-related concussion. Clin Sports Med 2004;23:421-441, ix.
105. Mccrory PR, Berkovic SF. Second impact syndrome. Neurology 1998;50:677-683.
106. Saunders RL, Harbaugh RE. The second impact in catastrophic contact-sports head trauma. JAMA 1984;252:538-539.
107. Reddy CC, Collins MW, Gioia GA. Adolescent sports concussion. Phys Med Rehabil Clin N Am 2008;19:247-269, viii.
108. Daniel JC, Olesniewicz MH, Reeves DL, et al. Repeated measures of cognitive processing efficiency in adolescent athletes: Implications for monitoring recovery from concussion. Neuropsychiatry, Neuropsychology, & Behavioral Neurology 1999;12:167-169.
109. Field M, Collins MW, Lovell MR, et al. Does age play a role in recovery from sports-related concussion? A comparison of high school and collegiate athletes. J Pediatr 2003;142:546-553.
110. Lovell MR, Collins MW, Iverson GL, et al. Recovery from mild concussion in high school athletes. J Neurosurg 2003;98:296-301.
111. Adams CW, Bruton CJ. The cerebral vasculature in dementia pugilistica. J Neurology, Neurosurgery & Psychiatry 1989;52:600-604.
112. Roberts GW. Immunocytochemistry of neurofibrillary tangles in dementia pugilistica and Alzheimer's disease: Evidence for common genesis. Lancet 1988;2:1456-1458.
113. Van den Heuvel C, Thornton E, Vink R. Traumatic brain injury and Alzheimers disease: A review. Prog Brain Res 2007;161:303-316.
Concussions, also known as mild traumatic brain injuries (mTBI), create challenges for the emergency care provider.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
