Special Feature: Radiologic Hazards and Acute Radiation Exposure
Radiologic Hazards and Acute Radiation Exposure
By Theodore C. Chan, MD, FACEP
Growing concern over terrorism and the possible use of weapons of mass destruction, including nuclear and radiologic devices, has drawn attention to the need to prepare for potential, large-scale radiation incidents. Such events may cause mass injuries or have very little clinical impact, but are likely to induce widespread fear and panic due to public misinformation and preconceived notions regarding nuclear and radiologic incidents.
Widespread, uncontrolled radiation exposure has occurred in the past as a result of atomic bomb detonations and fall-out, nuclear reactor accidents, and other intentional and non-intentional mishaps with the use of radioactive material and devices. In fact, radioactive material is used widely in society at university and research laboratories, government agencies, military units, and hospitals and medical centers, where radioactive isotopes are used for various diagnostic and therapeutic purposes. Despite its use in this setting, most metropolitan-area hospitals still are not prepared adequately for a radiologic event.1
Radiation Physics
There are several forms of ionizing radiation that vary in their physical properties, body tissue absorption, and biologic effects. Alpha particles are a particulate emission that travel short distances in air and penetrate less than 0.1 mm in tissue. As a result, alpha particles are hazardous primarily when inhaled, ingested, or deposited on open wounds. Beta particles are also a particulate emission, but travel farther distances in air and penetrate more deeply into tissue. As a result, these particles can be hazardous with skin and eye exposure, as well as inhalation or ingestion. Similarly, neutron particulate emission can travel long distances in air and can cause injury both with external and internal contamination. Gamma rays are an electromagnetic wave energy that can travel long distances through air and penetrate most materials with the exception of lead, concrete, or steel. Gamma rays penetrate easily through tissue and can cause internal and external injury. Similarly, x-ray radiation easily penetrates tissues and deposits its energy deep in the body.
The radiation dose absorbed by specific tissues is measured in rads (or the international Gray [Gy] unit equivalent to 100 rads). For example, a fetal x-ray exposure dose from a maternal, two-view chest x-ray is 0.00007 rads.2 Because different forms of radiation have different biologic effects at the same absorbed dose, the effective dose often is measured in rems (or the international unit sievert [sv], which is equivalent to 100 rems). When dealing with beta particles and gamma rays, the rem essentially is equivalent to the rad.1
Important factors minimizing the effects of radiation are distance, time, and shielding. The absorbed dose decreases with the square of the distance from the source, so that doubling the distance diminishes the dose rate to one-fourth. Radioactivity also decays with time depending on the source material and its half-life. As a result, time management of exposures remains a key component of minimizing absorption. Appropriate shielding can minimize exposure significantly. Clothing or paper can provide shielding from alpha particles. Beta particles are stopped by materials such as glass, aluminum, plastic, and other metals. Dense materials such as lead can shield tissues from gamma ray radiation.
Biologic Effects
Radiation can have both acute and long-term effects. High doses of radiation cause cellular death, resulting in acute injury. Generally, rapidly dividing cells, such as intestinal mucosa or bone marrow cells, are most vulnerable to radiation. Lower doses of radiation may not cause cell death, but can induce cellular damage that can lead to malignant transformation years later.3
Acute radiation exposure can result in a number of clinical syndromes, including a gastrointestinal syndrome associated with nausea, vomiting, and diarrhea from death of intestinal mucosal stem cells; hematopoietic syndrome associated with bone marrow suppression, leukopenia, and thrombocytopenia; and a cutaneous syndrome in which extensive skin injuries occur such as radiation burns, ulceration, and necrosis.4 The table lists the clinical effects based on acute whole-body radiation exposure.
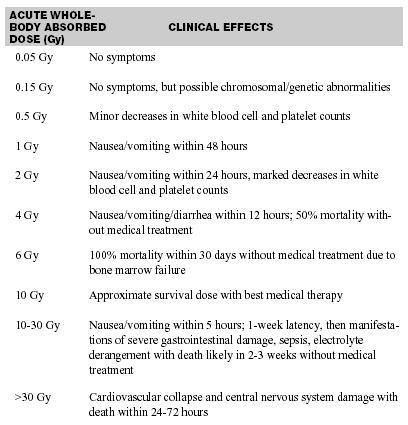
As a result of bone marrow suppression, a reduction in lymphocyte count can be seen in exposed individuals. In fact, the absolute lymphocyte count at 48 hours after exposure can be a marker of exposure, injury, and prognosis.6 Counts at 48 hours above 1500 cells/mm3 indicate no significant acute injuries with an overall excellent prognosis for the patient. Lymphocyte counts from 1000-1500 cells/mm3 indicate an acute absorbed whole-body dose of 0.5-1.9 Gy resulting in clinical symptoms but an overall good prognosis. Counts of 500-999 cells/mm3 indicate a 2.0-3.9 Gy exposure, severe injury and fair prognosis, whereas counts of 100-499 cells/mm3 suggest an overall poor prognosis. Counts of less than 100 cells/mm3 indicate a radiation exposure greater than 8 Gy resulting in a high incidence of death.
Decontamination and Treatment
External contamination occurs with radioactive exposure to skin and clothing. Management should focus on removing and controlling the spread of radioactive materials. Removal of clothing usually results in elimination of nearly 90% of contamination in most cases.1 Skin decontamination can be performed effectively with the use of water and detergents. Open wounds can be rinsed with saline. All contaminated materials should be placed in large, labeled plastic bags that undergo proper disposal.
Management of internal contamination focuses on a number of approaches: reduction of absorption, dilution, blockage, displacement (by non-radioactive substances), elimination from tissues, and chelation. A number of medications and chelating agents are under investigation for these uses, but many require administration either just before or within hours of exposure. Potassium iodide (KI) or iodate has been recommended after high-dose radiation exposure (i.e., detonation of a nuclear device) to prevent radioiodine from accumulating in the thyroid gland.7 Recommended daily doses are 130 mg for adults, 65 mg for ages 3-18 years, 32 mg for ages 1 month to 3 years, and 16 mg for infants younger than one month of age. As with other agents, KI must be administered within hours of exposure.
Management of radiologic incidents requires knowledge of the radiation source, dispersal pattern, numbers of victims and associated injuries (i.e., traumatic or blast injuries in the event of a nuclear or "dirty" bomb device), and local health care facilities and resources. Patients should be decontaminated and assessed for dosage exposure, associated injuries, and clinical signs and symptoms of radiation injury. For example, patients who report nausea, vomiting, or diarrhea must be evaluated for acute whole-body exposure. Supportive measures and treatment should be initiated as dictated by the patient’s presentation and available resources.
Provider safety remains of paramount importance during such events. For responding field personnel, the use of appropriate personal protective equipment, and just as importantly, time management on the scene, should focus on minimizing exposure and dosage absorbed. Recommended exposure limits for responders to radiation disaster events vary from 5-25 rem per event to exposures of no more than 0.1 Gy per hour during the event (in comparison, recommendations for total occupation exposure are 5 rem per year).8
After appropriate decontamination of victims, there is little risk of radiation transfer to health care facilities. In fact, medical personnel at the site of the Chernobyl disaster dealing with decontaminated victims received less than 10 milli-Gy of radiation.1 Medical personnel still should wear protective clothing and gloves in conformity with universal precautions, and respiratory equipment and respirators generally are not required at the hospital.
Resources
Additional resources and information on radiation exposure and injuries can be obtained 24 hours a day from the U.S. Department of Energy Radiation Emergency Assistance Center/Training Site (865-576-1005). Information also is available on its website at www.orau.gov/reacts. In addition, a handbook on medical management of radiologic casualties is available at www.afrri.usuhs.mil.
Dr. Chan, Associate Clinical Professor of Medicine, Emergency Medicine, University of California, San Diego, is on the editorial board of Emergency Medicine Alert.
References
1. Mettler FA Jr, Voelz GL. Major radiation exposure what to expect and how to respond. N Engl J Med 2002;346:1554.
2. Roberts JR. Radiographic imaging during pregnancy: Plain x-rays. Emerg Med News 2002;23:45.
3. Pierce DA, Preston DL. Radiation-related cancer risks at low doses among atomic bomb survivors. Radiat Res 2000;154:178.
4. Fliedner TM, et al. Stem cell responses after radiation exposure: A key to the evaluation and prediction of its effects. Health Phys 1996;70:787.
5. Gusev I, et al. Medical Management of Radiation Accidents. 2nd ed. Florida: CRC Press; 2001.
6. Goans RE, et al. Early dose assessment following severe radiation accidents. Health Phys 1997;72:513.
7. National Council on Radiation Protection and Measurements. Protection of the thyroid gland in the event of releases of radioiodine. Report no. 55. Washington DC: 1977.
8. International Commission on Radiological Protection. 1990 Recommendations of the International Commission on Radiological Protection. Vol. 21, No. 1-3 of Annals of the ICRP. Oxford, England: Pergamon Press; 1991.
Growing concern over terrorism and the possible use of weapons of mass destruction, including nuclear and radiologic devices, has drawn attention to the need to prepare for potential, large-scale radiation incidents.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
